Culture Media and Methods, Identification of Bacteria by Conventional, Automated and Molecular Methods
Culture Media
The basic constituents of culture media are:
Table of Contents
Peptone: Mixture of partially digested proteins
- Agar: It is used for solidifying the culture media. It has no nutritional property.
- It is prepared from seaweeds (red algae of species Gelidium and Gracilaria).
- Agar is preferred over gelatine, as it is bacteriologically inert, and it melts at 98°C and usually solidifies at 42°C
- Concentration of agar:
- For solid agar: 1–2% (Japanese agar 2% or New Zealand agar 1.2%)
- For semisolid agar: 0.5%
- For solid agar to inhibit Proteus swarming: 6%
- Others: Meat extract, Yeast extract, Blood and serum, Water and Electrolytes (NaCl).
Read And Learn More: Micro Biology And Immunology Notes
Simple/Basal Media
They contain minimum ingredients that support the growth of non-fastidious bacteria. Examples include:
- Peptone water: It contains peptone (1%) + NaCl (0.5%) + water
- Nutrient broth: It is made up of peptone water + meat extract (1%)
- Nutrient agar: It is made up of nutrient broth + 2% agar
The basal media are used for:
- Testing the non-fastidiousness of bacteria
- They serve as the base for the preparation of many other media
- Nutrient broth is used for studying the bacterial growth curve
- Nutrient agar is the preferred medium for:
- Performing the biochemical tests such as oxidase, catalase and slide agglutination
- To study the colony character and Pigment demonstration.
Enriched Media
When a basal medium is added with additional nutrients such as blood, serum or egg, it is called enriched medium. They also support the growth of fastidious bacteria, e.g.:
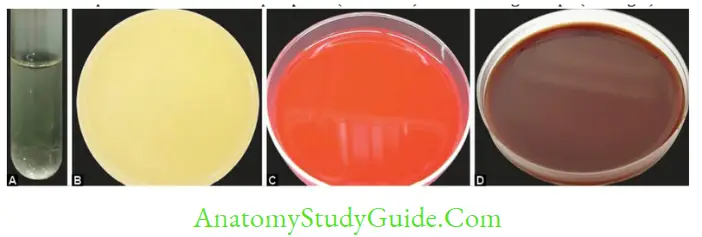
- Blood agar: Prepared by adding 5–10% of sheep blood to the molten nutrient agar at 45°C.
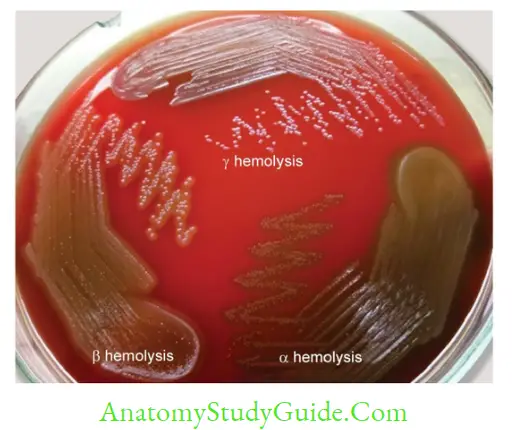
It is used to test the hemolytic property of the bacteria. - Chocolate agar: It is the heated blood agar, blood is added to the molten nutrient agar at 70°C. It is more nutritious than blood agar, and even supports Haemophilus influenzae.
- Loeffler’s serum slope is used for isolation of Corynebacterium diphtheriae.
- Blood culture media: Used for blood culture. They are of two types:
Monophasic medium is made-up of brain-heart infusion (BHI) broth
-
- Biphasic medium has a liquid phase (BHI broth) and a solid agar slope (BHI agar).

Enrichment Broth
Liquid media that allow certain organism (pathogens) to grow and inhibit others (normal flora):
- Selenite F and Tetrathionate broth used for Salmonella and Shigella
- Alkaline peptone water (APW) used for Vibrio cholerae.
Selective Media
Solid media that allow certain organism (pathogens) to grow and inhibit others (normal flora):
- Lowenstein Jensen (LJ) medium is used for isolation of Mycobacterium tuberculosis
- Thiosulphate Citrate Bilesalt Sucrose (TCBS) agar used for isolation of Vibrio species
- For the isolation of enteric pathogens such as Salmonella and Shigella from stool:
- DCA (Deoxycholate Citrate Agar)
- XLD (Xylose Lysine Deoxycholate) agar
- Potassium tellurite agar (PTA) is used for isolation of Corynebacterium diphtheriae.
- Wilson Blair bismuth sulphite medium: It is used for isolation of Salmonella typhi.
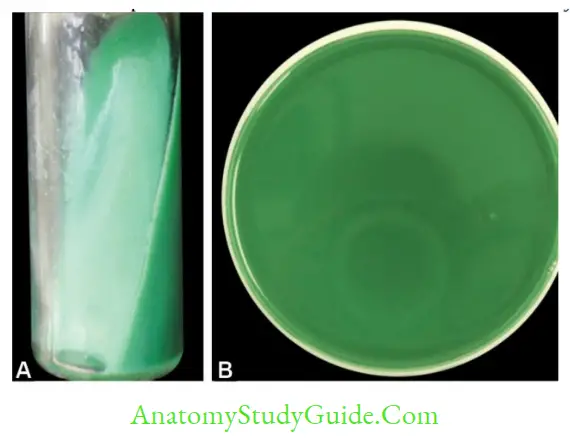
Transport Media
They are used for the transport of specimens containing delicate organism or when the delay is expected. Bacteria do not multiply in the transport media, they only remain viable.
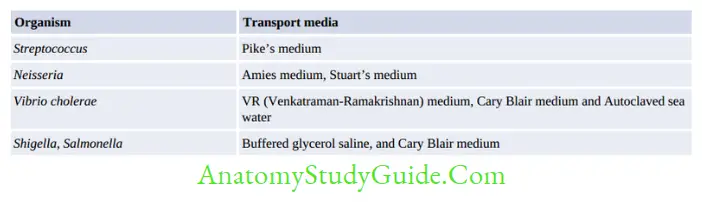
Differential Media
These media differentiate between two groups of bacteria by using an indicator.
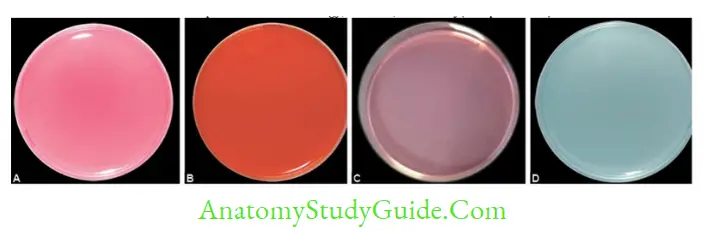
- MacConkey agar: It differentiates organisms into LF or lactose fermenters (produce pink colonies, e.g. E. coli and Klebsiella) and NLF (produce colorless colonies, e.g. Shigella and Salmonella).
- CLED agar (Cysteine lactose electrolyte-deficient agar): This is similar to MacConkey agar, differentiates between LF and NLF. It is used as an alternative to combination of blood agar and MacConkey agar, for the processing of urine specimens:
- Advantages over MacConkey agar: It is less inhibitory than MacConkey agar, supports gram-positive bacteria (except β hemolytic Streptococcus) and Candida.
- Advantage over blood agar: It can prevent the swarming of Proteus.
Anaerobic Culture Media
Anaerobic media contain reducing substances which take up oxygen and create lower redox potential and thus permit the growth of obligate anaerobes such as Clostridium.
Examples are:
- Robertson’s cooked meat (RCM) broth: It contains chopped meat particles (beef heart),which provide glutathione and unsaturated fatty acids.
- Other anaerobic media include:
- BHIS agar: Brain heart infusion agar with supplements (vitamin K and hemin)
- Thioglycollate broth, Anaerobic blood agar
- Neomycin blood agar, Egg yolk agar and Phenylethyl agar.
Culture Methods
Various Aerobic Culture Methods
- Streak culture by using loop with intermittent heating: It is the routinely used method.
- Lawn or carpet culture: Useful for Carrying out antimicrobial susceptibility testing by disk diffusion method, Bacteriophage typing, and producing large amount of bacterial
growth for preparation of bacterial antigens and vaccines. - Stroke culture (zigzag fashion by straight wire): Used for citrate, urease and TSI (triple sugar iron test).
- Stab culture: Stabbing the semisolid agar butt by a straight wire. It is used for: 1. maintaining stock cultures, 2. OF test 3. Mannitol motility medium, 4. Nutrient agar semisolid butts,5. TSI (here both stroke and stab cultures are made).
- Liquid culture:
- Uses: Liquid cultures are useful for: 1. blood culture, (2) for sterility testing.
- Advantages: Used for: (1) When bacteria load is less, (2) specimens (e.g. blood) containing antibiotics can be neutralized by dilution in the medium, (3) when large yields of bacteria are required, (4) for demonstration of bacterial growth curve.
- Disadvantages: Liquid cultures do not provide a pure culture from a mixed inoculum.
- Pour-plate culture: Quantitative culture method, used to estimate viable bacterial count.
Incubatory Conditions
- Most of the pathogenic organisms grow best at 37°C.
- Candle jar: It provides capnophilic atmosphere (3–5% CO2). This is useful for Brucella abortus, Streptococcus, pneumococcus and gonococcus.
- Microaerophilic bacteria such as Campylobacter and Helicobacter require 5% oxygen.
Anaerobic Culture Methods
- Production of vacuum.
- By displacement and combustion of oxygen: This principle is used in:
- McIntosh and Filde’s anaerobic jar (It involves evacuation of air and replacement with hydrogen gas manually)
- Anoxomat System (principle is same, but done by automated instrument).
- Gas pak System (Absorption of oxygen chemically, e.g. using alkaline pyrogallol).
Indicators of anaerobiosis are:
-
- Chemical indicator: Reduced methylene blue
- Biological indicator: Pseudomonas
- Anaerobic Glove Box (or anaerobic chamber)
- By reducing agents: Such as glucose, thioglycollate, meat, cysteine, and ascorbic acid.
- PRAS media (Prereduced, Anaerobically Sterilized).
Preservation of Microorganisms
- Short-term methods: (1) Sub-culturing, (2) Immersing the culture in glycerol, or sterile distilled water, (3) Freezing at –20°C and (4) Drying (for moulds and spores).
- Long-term methods: By Ultra temperature freezing and Lyophilization (freeze-drying).
Identification Of Bacteria
Identification of bacteria can be done by: (1) conventional (culture and identification by biochemical reactions), (2) automated culture techniques and (3) molecular methods.
Conventional Methods
Conventional method consists of following steps:
- Specimens are subjected to direct microscopy (Gram stain or any other special stain).
- Followed by conventional culture on blood agar and MacConkey agar or any other special media.
- Colonies grown on culture media are subjected to culture smear and motility testing.
- Based on the culture smear report, the appropriate biochemical reactions are put for bacterial identification.
Automated Culture Techniques
Conventional culture methods often yield poor results because of low bacterial load. Therefore,various automated blood culture techniques have been in use since last decade.
Advantages: The major advantages of automated blood culture techniques are:
- Continuous automated monitoring (once in every 15–20 min by the instrument).
- Other advantages: More sensitive,↑ yield, rapid, less labor intensive.
Disadvantages: (1) High cost (2) inability to observe the colony morphology as liquid medium is used, (3) no separate detection in mixed cultures, (4) radioactive hazards for BACTEC.
Automated methods include:
- Automated culture methods (include automated blood culture systems) and
- Automated bacterial identification systems.
Automated Blood Culture Methods
- BacT/ALERT 3D
Principle of BacT/ALERT is based on colorimetric detection of growth in contrast to fluorometric detection by BACTEC.- CO2 produced by growing microorganisms diffuses across the membrane into the sensor where it reacts with water generating hydrogen ions.
As the concentration of hydrogen ions increases and the pH decreases, the blue-green sensor becomes yellow, a change that is detected by colorimetric method.
- CO2 produced by growing microorganisms diffuses across the membrane into the sensor where it reacts with water generating hydrogen ions.
- BacT/ALERT VIRTUO: It is an advanced form of BacT/ALERT that offers several advantages such as: (1) automatic loading and unloading of bottles, (2) faster detection of growth, (3) can determine the volume of blood present in bottles.
- BACTEC: Earlier versions of this system were based on the use of radioisotopes to detect growth. They are not in use currently. The current versions are based on fluorescent detection.
- VersaTREK: It works on the principle that the CO2 liberated from bacteria, causes a pressure change which is then detected by manometry.
Automated Systems for Bacterial Identification
- MALDI–TOF (Matrix-assisted laser desorption/ionization time-of-flight), e.g. VITEK MSIt can identify bacteria, fungi, and mycobacteria with a turnaround time of few minutes and with absolute accuracy
- VITEK 2, Phoenix and MicroScanWalkAway systems- for automated identification and antimicrobial susceptibility test.
Molecular Methods
Nucleic acid amplification techniques (NAATs) include:
- Polymerase chain reaction (PCR) and its modification including Real time PCR
- Ligase chain reaction (LCR) and Transcription-mediated amplification (TMA)
- Nucleic acid sequence-based amplification (NASBA)
- Strand displacement amplification (SDA)
- Biofire FilmArray
- Loop-Mediated Isothermal mplification (LAMP).
Polymerase Chain Reaction (PCR)
PCR is a technology in molecular biology used to amplify a single or few copies of a piece of
DNA to generate millions of copies of DNA. It was developed by Kary B Mullis.
Principle: PCR involves three basic steps.
- DNA extraction from the organism
- Amplification of extracted DNA: The extracted DNA is subjected to repeated cycles (30–35 numbers) of amplification in a thermocycler which takes about 3–4 hours.
Each amplification cycle has three steps:
-
- Denaturation at 95°C: This involves separation the dsDNA into two separate ssDNA.
- Primer annealing (55°C): Primer is a short oligonucleotide complementary to a small sequence of the target DNA. It anneals to the complementary site on ssDNA.
- Extension of the primer (72°C): This step is catalyzed by TaPolymerase enzyme.
Gel electrophoresis of amplified product: The amplified DNA is electrophoretically migrated according to their molecular size to form bands; seen under UV rays.
Advantages: PCR has the following advantages compared to the conventional culture:
- More sensitive: It can amplify very few copies of a specific DNA, so it is more sensitive.
- More specifically: By use of primers targeting specific DNA sequence of the organism
- Detects the organism: (1) either from sample, (2) to confirm culture isolate.
- Detect the organisms that are highly fastidious or noncultivable by conventional culture methods.
- Detect genes coding drug resistance (e.g. mecA gene detection in Staphylococcus aureus)
- Detects genetic diseases such as sickle cell anemia, phenylketonuria, etc.
Disadvantages:
- Conventional PCR detects only the DNA, but not the RNA (detected by RT-PCR).
- Qualitative, not quantitative (Quantitation is done by real time PCR).
- Viability: PCR cannot differentiate between viable or nonviable organisms.
- False positive amplification may occur due to contamination with environmental DNA.
- False negative: by PCR inhibitors present in some specimens such as blood, feces, etc.
Modification of PCR
- Reverse transcriptase PCR (RT-PCR): For amplifying RNA, RT-PCR is done.
After RNA extraction, the first step is addition of reverse transcriptase enzyme that converts RNA into DNA.
Then, the amplification of DNA and gel documentation steps are similar to that described for conventional PCR.
Useful for detection of RNA viruses or 16SrRNA genes of the organisms. - Nested PCR: The amplified products of the first round PCR is subjected to another round of amplification using a second primer targeting a different gene of same organism.
- More sensitive: Double round of amplification yields high quantity of DNA.
- More specific: Use of two primers targeting same organisms makes more specific.
- Disadvantage: There is more chance of contamination of the PCR tubes, which may lead to false positive results.
- Multiplex PCR: It uses more than one primer which can detect many DNA sequences of several organisms in one reaction.
- Syndromic approach: To diagnose infectious syndrome caused by more than one organism.
- For example, for the etiological diagnosis of pyogenic meningitis, different primers targeting the common agents of pyogenic meningitis, such as pneumococcus, meningococcus, and H. influenzae can be added simultaneously in the same reaction tube
- Contamination issues: There are chances of the reaction tubes being contaminated with environmental DNA.
Real-time PCR (rt-PCR)
Real-time PCR, though expensive, but has many advantages over a conventional PCR:
- Quantitative, hence can be used for monitoring treatment response, e.g. in HIV or HBV.
- Takes less time: As the amplification can be visualized even when the amplification cycle is going on.
- Contamination rate is extremely less.
- Sensitivity and specificity of rt-PCR assays are extremely higher.
Detection of amplification products of real-time PCR
- Nonspecific methods use SYBR green dye that stains any nucleic acid nonspecifically.
- Specific methods use fluorescently labeled DNA probe such as (1) TaqMan or hydrolysis probe, (2) Molecular beacon and (3) FRET (Fluorescence Resonance Energy Transfer) probe.
Biofire Filmarray
- It is a completely automated multiplex nested PCR system where all the steps from sample preparation to amplification, detection and analysis are performed automatically by the system;
- Four panels are available such as respiratory, gastrointestinal, meningitis-encephalitis and blood culture identification panels;
- It has excellent sensitivity and specificity with turnaround time of 1 hour.
- However, the higher cost of the panels limits its wide use.
Loop-Mediated Isothermal Amplification (LAMP)
- LAMP is an isothermal nucleic acid amplification technique. It provides several advantages over PCR. Amplification is carried out at a constant temperature of 60–65°.
- Advantages: Polymerase with strand displacement ability, Cheaper and easy to perform, Amplicons are detected directly by naked eyes either by turbidity or visual fluorescence detection
- Uses: LAMP assay has been approved for tuberculosis
- Drawback: It shows high false positive results due to cross-contamination between the reaction tubes
Microbial Typing
Microbial typing refers to characterization of an organism beyond its species level. It is used to determine the relatedness between different microbial strains of the same species and thereby helps to (1) investigate outbreaks, and (2) determine the source and routes of infections.
Genotypic methods are more reliable and have better reproducibility and discriminative power than phenotypic methods, however, they are expensive.
Leave a Reply