Pharmaceutical Organic Chemistry Long Question And Answers 2021
Question 1. Define electrophilic substitution reaction. Explain the mechanism of nitration and sulphonation of benzene.
Answer:
Table of Contents
Electrophilic substitution of benzene:
- Benzene is the one where an electrophile substitutes the hydrogen atom of benzene. As the aromaticity of benzene is not disturbed in the reaction, these reactions are highly spontaneous.
- Basic examples of electrophilic substitution reactions of benzene are nitration, sulfonation, halogenation, Friedel Craft’s alkylation, and acylation, etc.
The Mechanism for Electrophilic Substitution of Benzene:
An electrophilic substitution reaction generally involves three steps:
1. Generation of electrophile:
Due to the presence of Lewis acid, generation of electrophile takes place. As the Lewis acid accepts the electron pair from the attacking reagent.
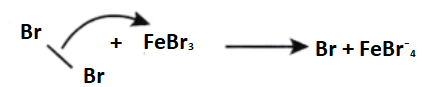
2. Formation of arenium ion:
The electrophile generated attacks on the benzene ring to form a positively charged cyclohexadienyl cation better called an arenium ion containing one sp³ hybridized carbon atom. The positive charge is effectively distributed over three carbon atoms by resonance which makes it partially stable
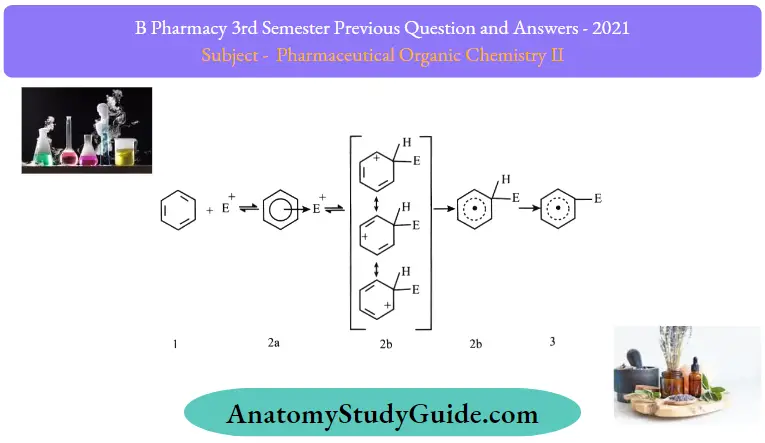
As the delocalization electron stops at an sp3 hybridized carbon atom, the arenium ion is not aromatic in nature.
3. Removal of positive charge from the carbocation intermediate:
The arenium ion finally loses its proton from sp3 hybridized carbon to a Lewis base restoring the aromaticity.
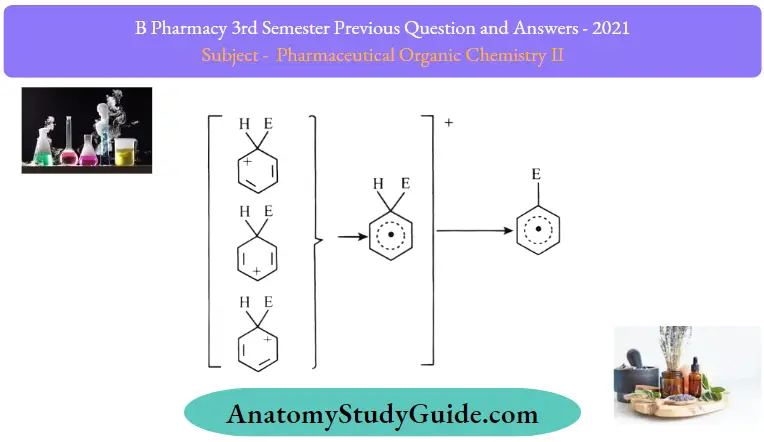
Few examples of electrophilic aromatic substitution:
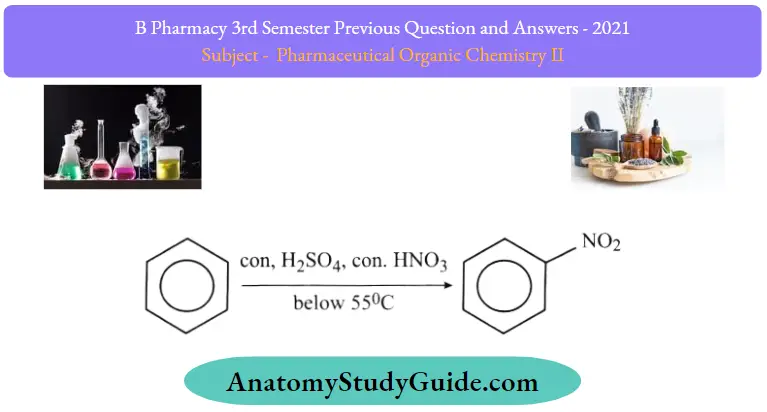
1. Nitration of Benzene:
Benzene reacts with nitric acid at 323-333k in the presence of sulphuric acid to form nitrobenzene. This reaction is % known as the nitration of Benzene.
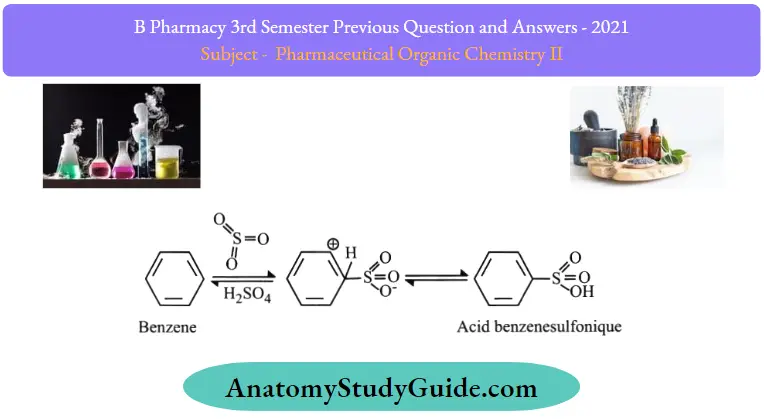
2. Sulfonation of Benzene:
Sulfonation of benzene is a process of heating benzene with fuming sulphuric acid (H2SO4 +SO3) to produce benzenesulfonic acid. The reaction is reversible.
Question 2. Write any three chemical reactions of benzoic acid.
Answer:
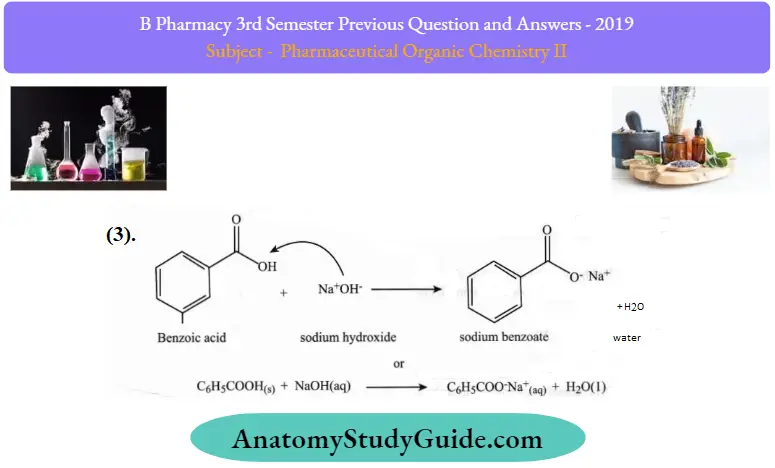
- Formation of Salt: Benzoic acid has a carboxyl group, which is acidic and so it reacts with a base like sodium hydroxide to produce a salt, sodium benzoate C6H5COO” and Na+. It is an iron compound and when sodium benzoate is treated with an acid such as HCl it forms the reactant back.
- Formation of Esters: When benzoic acid reacts with any alcohol in the presence of sulfuric acid. Here sulfuric acid acts as a dehydrating f7k agent. In reaction, it removes water molecules produced by the combination of acid and alcohol to form a pleasant¬ smelling ester, ethyl benzoate. This reaction is an esterification reaction.
- Formation of Acid Halides: When benzoic acid reacts with thionyl chloride SOCI2 or Phosphorus pentachloride PCI5, it forms benzoyl chloride which is an example of acyl or acid halide. Benzoyl chloride is highly reactive and so it reacts with an amine such as methylamine CH3NH2 or with ammonia NH3 to form benzamide C6H5CONH2
Question 3. Explain the acidity and effect of substituents on the acidity of phenols.
Answer:
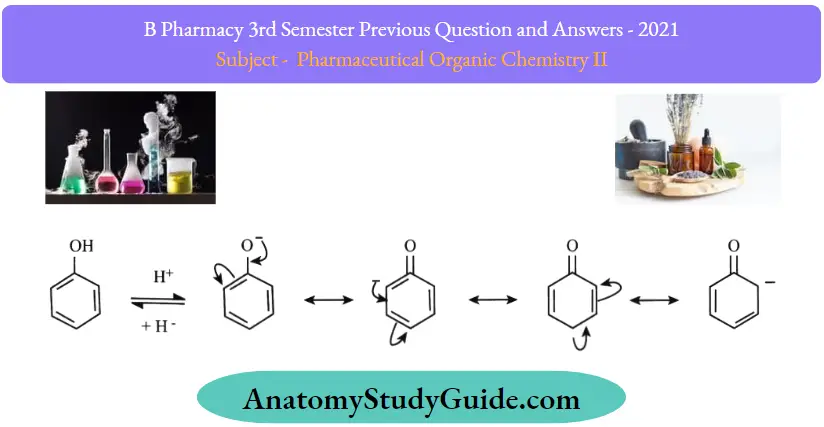
The acidity of phenols is due to their ability to lose hydrogen ions to form phenoxide ions.
- In a phenol molecule, the sp2 hybridized carbon atom ofthe benzene ring attached directly to the hydroxyl group acts as an electron-withdrawing group.
- This sp2 hybridized carbon atom of a benzene ring attached directly to the hydroxyl group has higher electronegativity in comparison to the hydroxyl group.
- Due to the higher electronegativity of this carbon atom in comparison to the hydroxyl group attached, electron density decreases on the oxygen atom.
- The decrease in electron density increases the polarity of the O-H bond and increases the ionization of phenols.
- Thus, the phenoxide ion is formed. The phenoxide ion formed is stabilized by the delocalization of negative charge due to the resonance in the benzene ring.
- Phenoxide ion has greater stability than phenols, as in the case of phenol charge separation takes place during resonance.
Acidity of phenols:
- The resonance structures of phenoxide ions explain the delocalization of negative charge.
- In the case of substituted phenols, the acidity of phenols increases in the presence ofthe electron-withdrawing group.
- This is due to the stability of the phenoxide ion generated.
- The acidity of phenols further increases if these groups are attached at ortho and para positions.
- This is because the negative charge in phenoxide ion is mainly delocalized at the ortho and para positions ofthe attached benzene ring.
- On the other hand, the acidity of phenols decreases in the presence of electron-donating groups as they prohibit the formation of polyoxide ions.
Question 4. Explain drying, semi-drying, and nondrying of oils with examples. Explain the significance and reactions of hydrolysis and hydrogenation of oils and fats.
Answer:
- Drying oil: Drying oil is a kind of oil that hardens when exposed to air and forms a tough, elastic layer on the surface to which it’s applied.
- Example: Linseed oil.
- A semi-drying oil: A semi-drying oil is an oil that partially hardens when it is exposed to air. This is as opposed to a drying oil, which hardens completely, or a non-drying oil, which does not harden at all. Oils with an iodine number of 115-130 are considered semi-drying.
- Example: Semi-drying oils are com oil, cottonseed oil, etc.
- Non-drying oil: Non-drying oil is a highly saturated oil (such as olive oil) that is unable to solidify when exposed in a thin film to air.
- Examples: Avocado oil, olive oil, etc.
Significance of oils and fats:
- To convert a liquid oil into a solid fat.
- When solid fats ofthe right consistency are expensive or not available, hydrogenation, sometimes in combination with other processes such as interesterification or fractionation, may provide a way to produce the desired fat.
Hydrolysis:
The reaction can be catalyzed by acid, base, or lipase, but it also occurs as an uncatalyzed reaction between fats and water dissolved in the fat phase at suitable temperatures and pressures.
Nonenzymatic ester hydrolysis and the soap-making process:
In an aqueous solution, esters are subject to hydrolysis to the corresponding carboxylic acid and alcohol components.
Hydrolysis Mechanism:
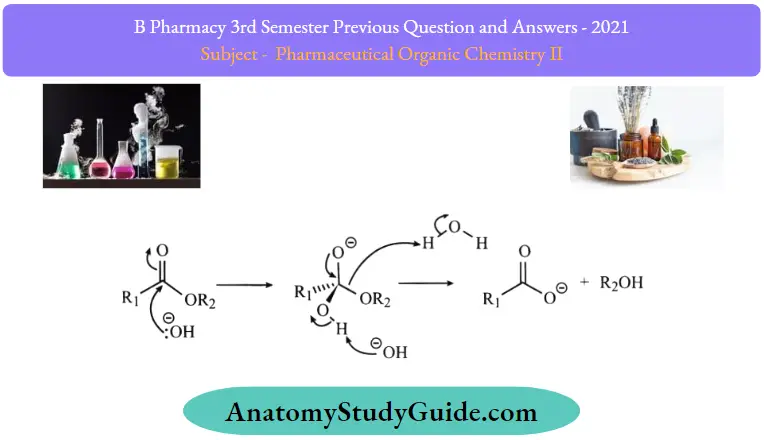
- Step 1: The hydroxide nucleophiles attack at the electrophilic C of the ester C=0, breaking the n bond and creating the tetrahedral intermediate.
- Step 2: The intermediate collapses, reforming the C = O results in the loss ofthe leaving group the alkoxide, RO–, leading to the carboxylic acid.
- Step 3: An acid/base reaction. A very rapid equilibrium where the alkoxide, functions as a base deprotonating the carboxylic acid, RCO2H, (an acidic workup would allow the carboxylic acid to be obtained from the reaction).
Question 5. Explain the principle involved in any one method of determination of iodine value. Give its significance.
Answer:
Iodine value:
It is the measure of the degree of unsaturation of fats, wax, and oils which it determines the amount of iodine that is taken up by 100 gms of fats or oil.
Iodine Principle:
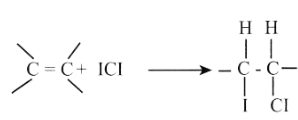
Fatty acids react with a halogen [ iodine] resulting in the addition ofthe halogen at the C=C double bond site. In this reaction, iodine monochloride reacts with the unsaturated bonds to produce a di-halogenated single bond, of which one carbon has bound an atom of iodine.
After the reaction is complete, the amount of iodine that has reacted is determined by adding a solution of potassium iodide to the reaction product.
⇒ ICl + KI → KCl + I2
This causes the remaining unreacted ICI to form molecular iodine. The liberated I2 is then titrated with a standard solution of 0… IN sodium thiosulfate.
⇒ I2 + 2Na2S2O3 → 2 NaI + Na2 S2O4
Saturated fatty acids will not give the halogenation reaction. If the iodine number is between 0-70, it will be a fat and if the value exceeds 70 it is an oil. Starch is used as the indicator for this reaction so that the liberated iodine will react with starch to give purple coloured product and thus the endpoint can be observed.
Iodine Procedure:
- Arrange all the reagent solutions prepared and the requirements on the table.
- Pipette out 10ml of fat sample dissolved in chloroform to an iodination flask labeled as “TEST”.
- Add 20ml of Iodine Monochloride reagent into the flask. Mix the contents in the flask thoroughly.
- Then the flask is allowed to stand for half an hour of incubation in the dark.
- Set up a BLANK in another iodination flask by adding 10ml Chloroform to the flask.
- Add to the BLANK, 20ml of Iodine Monochloride reagent and mix the contents in the flask thoroughly.
- Incubate the BLANK in the dark for 30 minutes.
- . Meanwhile, Take out the TEST from incubation after 30 minutes and add 10 ml of potassium iodide solution into the flask
- Rinse the stopper and the sides of the flask using 50 ml distilled water.
- Titrate the “TEST” against standardized sodium thiosulphate solution until a pale straw colour is observed.
- Add about 1ml starch indicator into the contents in the flask, a purple color is observed. continue the titration until the color ofthe solution in the flask turns colorless.
- The disappearance of the blue color is recorded as the endpoint ofthe titration.
- Similarly, the procedure is repeated for the flask labeled ‘Blank’.
- Record the endpoint values of the BLANK.
- Calculate the iodine number using the equation below:
Volume of sodium thiosulphate used
= (Blank – Test ) ml
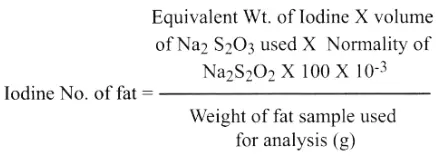
Equivalent Weight ofIodine = 127
Normality of sodium thiosulphate ( Na2S2O3) = 0.1
Iodine Significance:
This denotes the degree of unsaturation of fats. The saturated fat takes up no Iodine so their Iodine value is said to be of value zero. But the unsaturated fats have double or triple bonds which are generally very reactive towards the iodine. With the increase in double or triple bonds in carbon, the reactivity with iodine increases and it consumes more Iodine in reaction and so has a higher iodine value.
Question 6. Explain the mechanism of halogenation of benzene. Halogens are the deactivator group but ortho and para are activators. Give reasons.
Answer:
Halogenation of Benzene
Benzene reacts with halogens in the presence of Lewis acids like FeCl3, and FeBr3 to form aryl halides. This reaction is termed as halogenation of benzene.
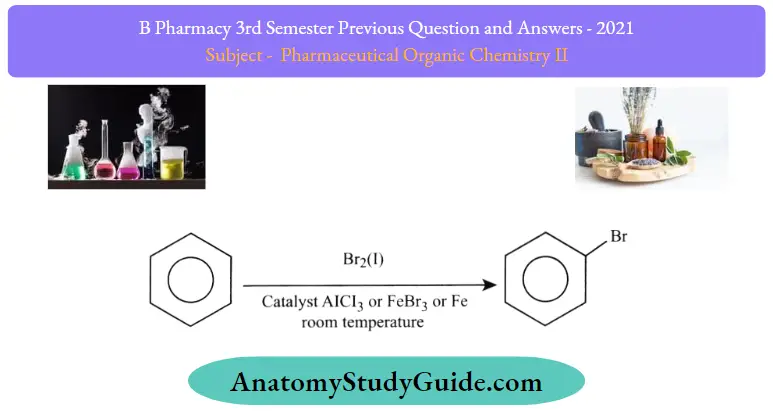
The mechanism for halogenation of benzene:
Step 1: Being a Lewis acid, FeBr3 helps in the generation of electrophile bromine ion by combining with the attacking reagent.
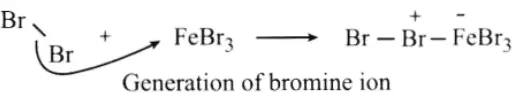
Step 2: The bromine ion acts as an electrophile in the process which further reacts with benzene to form an arenium ion which finally converts to bromobenzene.
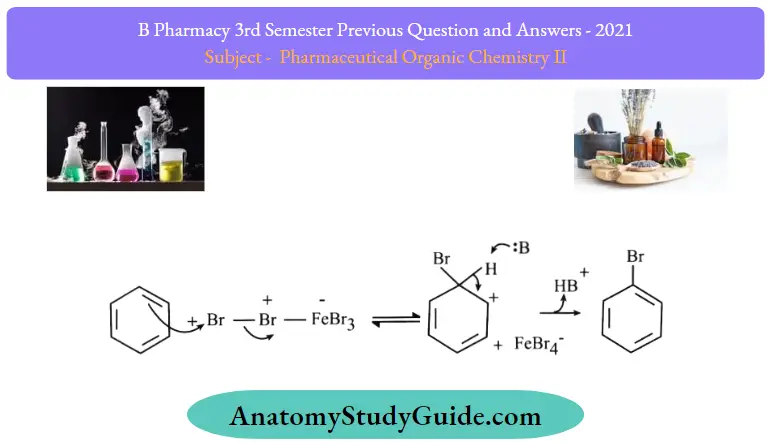
Halogens are deactivator groups but ortho and para are activators:
Like nitrogen and oxygen, the halogens F, Cl, Br, and I, are all more electronegative than carbon and they withdraw the electron density from the ring by the inductive effect. Fluorine donates the nonbonded pair of electrons to the ring by resonance as well. However, it is so electronegative that this effect is shaded, and overall fluorine makes the ring electron-poor and deactivates it towards electrophilic substitutions.
The other halogens are not as electronegative. However, they do not show a strong resonance effect either. The reason for this is the lack of a good overlap between the 2p orbital ofthe carbon and the 4p and 5p orbitals of chlorine and bromine. Remember, the conjugation and bond-making depend on the efficiency ofthe orbital overlapping.
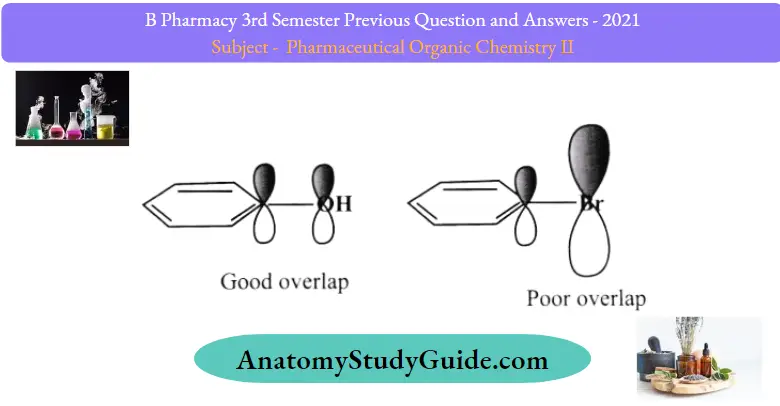
Orbital overlap
Now, as to why they are ortho-para directors, you need to remember that they are still electron-donators by resonance which does stabilize the transition state by an additional resonance contributor whether it is significant or not.
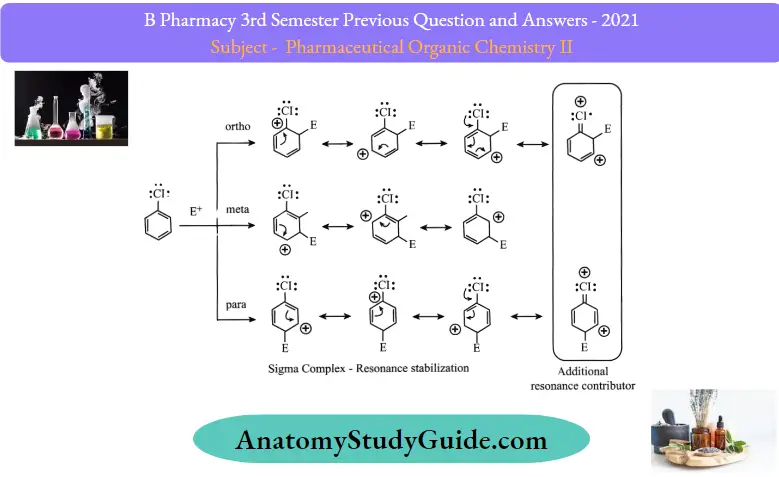
To keep this in a nutshell, remember that halogens deactivate the ring, but they still orient the substitution to the ortho and para positions because of the resonance effect.
Question 7. Enlist the analytical constants of oils and fats. Discuss in detail about acid value and iodine value and give their significance.
Answer:
Analytical constants of oils and fats:
1. Acid value:
The acid value is the number that expresses in milligrams the amount of potassium hydroxide necessary to neutralize the free acids present in one gram of the substance.
Acid value Method:
- Dissolve about 10 gm of substance under examination, accurately weighed in 50ml of a mixture of equal volumes of ethanol and ether, previously neutralized with 0.1M potassium hydroxide to phenolphthalein solution.
- If the sample does not dissolve in the cold solvent, connect the flask with a reflux condenser and warm slowly, with frequent shaking.
- Until the sample dissolves. Add 1 ml of phenolphthalein solution and titrate with 0.1M potassium hydroxide until the solution remains faint after shaking for 30 seconds.
- Calculate the acid value from the suspension.
Acid value = 5.6 n/w
Where n = The number of 0.1ml of 0.1 M potassium hydroxide
W = The weight in grams of the substance.
Note:
If the oil has been saturated with carbon dioxide for preservation, gently reflux the solution ofthe oil in ethanol and ether for lOmin before titration. The oil may be freed from the carbon dioxide by exposing in it to a shallow dish in a vacuum desiccator for 24 hours before weighing the sample.
Acid value Significance:
- The acidity of oil is due to hydrolysis or oxidation of oil by atmospheric moisture leading to the formation of fatty acids
- Lubricant oil with acid values greater than 0.1 corrode metals, and forms gum, and sludge during operation.
2. Iodine value:
The iodine value, also known as the iodine number, is a measure ofthe degree of unsaturation of fat, wax, or oil; it is expressed in gms, the amount of iodine, which is taken up by 100 gms of the fat, wax, or oil.
Iodine value Principle:
- The oil or fat is taken in carbon tetrachloride and is treated with a known excess of iodine monochloride solution in glacial acetic acid.
- The excess ofiodine monochloride is treated with potassium iodide and the liberated iodine is estimated by titration with sodium thiosulphate solution.
Iodine Value Method:
- Wij’s Iodine monochloride solution Carefully break open 1 X 50-g ampoule of Iodine Monochloride (Use Iodine Monochloride ofE.
- Merck or of similar quality) and pour into 155ml of glacial acetic acid. Mix well and store in a stoppered amber-colored bottle.
- This is the stock solution. Dilute 50 ml of the stock to 1000 ml using Glacial acetic acid.
- Mix well and store in a well-stoppered clean and amber colored glass bottle.
- This is the solution to be used for the determination of iodine value.
Iodine value Significance:
- Iodine is one of three trace elements ofthe body that are vitally important for the human body. Whilst the body only contains approximately 14 mg of iodine, it is vital for thyroxine formation, which is a hormone secreted from the thyroid gland in the neck that plays an important part in regulating the metabolic rate ofthe body.
- To meet the requirements of the body, we need only mg per week. Now, most of the salts are fortified by iodine salts which is one of the reasons that goitres are less common than they were once.
- The end-product is transported around the blood bound to the proteins (primarily thyroid, binding globulin).
- It is released to cells slowly in an active form, and from here, it regulates the excitability ofthe nerve fibres to the metabolism rate.
Question 8. What are aromatic amines? Explain the reactions of aromatic amines. Discuss the effect of substituents on the basicity of aromatic amines.
Answer:
1. Aromatic amines
An aromatic amine is an organic compound consisting of an aromatic ring attached to an amine. It is a broad class of compounds that encompasses anilines, but also many more complex aromatic rings and many amine substituents beyond NHr
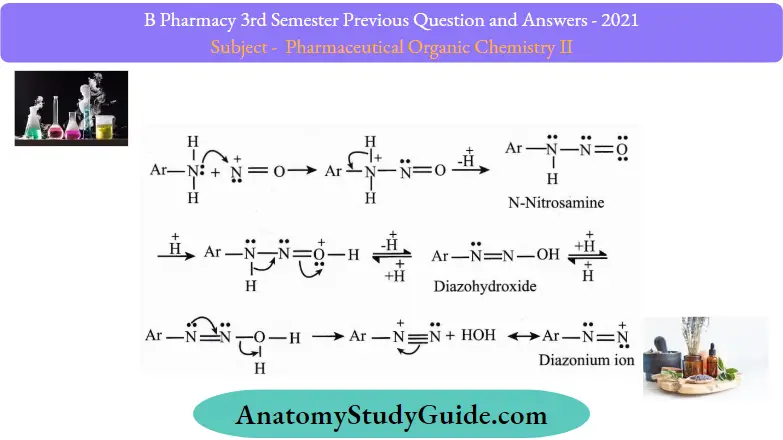
Diazotization reaction:
The chemical process used in converting a primary aromatic amine into the corresponding diazonium salt ofthe amine is commonly referred to as diazotization. This process is also known as ‘diazotization’.
Diazotization Reaction Mechanism:
The diazotization reaction mechanism begins with the reaction of nitrous acid with the other acid to give water and nitrosonium ions.
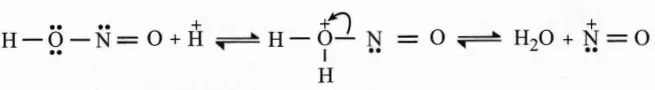
Thus, the required nitrosonium ion is formed. This is now reacted with the aromatic ring to which the NH2 group is attached.
- The positive charge of the nitrosonium ion is now shifted to the nitrogen which is directly attached to the aromatic ring as a new nitrogen-nitrogen bond is formed.
- The subsequent deprotonation gives the n-nitrosamine. In the presence of excess acid, nitrosamine can be converted into a diazohydroxide by protonation and subsequent deprotonation.
- Diazohydroxide is now protonated and water is removed from the compound to give the required aryl diazonium ion (which can easily be converted into a diazonium salt).
The mechanism ofthe diazotization reaction of aniline is illustrated below:
2. Basicity of Amines
Unlike ammonia, amines serve as bases and are reasonably strong (for some examples of Ka conjugate acid values, see the table provided).
The basicity of amines varies by molecule and largely depends on:
- The presence ofthe lone pair of nitrogen electrons
- The electronic properties of the substituent groups attached
- For example: Alkyl groups increase the basicity, aryl groups decrease it, etc.) the degree of solvation of the protonated amine, which mostly depends on the solvent used in the reaction.
- Simple amine water solubility is largely due to the hydrogen bonding capacity that can exist between the protons on the water molecules and these lone electron pairs.
- We usually consider amines as bases, but remember that lo and 2o-amines (not 3o-amines without N-H protons) are also weak acids (the pKa value of ammonia is 34).
- In this regard, it should be noted that, as in the previous section, amine’s acidity is measured by the pKa rather than its conjugate acid.
The relevance of all these acid-base relationships to functional organic chemistry lies in the need for differing-intensity organic bases as reagents suited to the particular reaction requirements.
- In many organic solvents, the common base such as sodium hydroxide is not soluble and is therefore not commonly used as a reagent like organic reactions.
- Alkoxide salts, amines or amide salts are the bulk ofthe base reagents.
- Since alcohols are much stronger acids than amines, they have weaker conjugate bases than amide bases and fill the gap between amines and amide salts in the base strength.
Factors that Affect the Basicity of Amines:
- The presence of an electron-donating group increases the basicity ofthe amines
- Examples: CH3, -CH3 CH3
- The electron-withdrawing group decreases the basicity.
- Example: Nitro group.
Pharmaceutical Organic Chemistry 2 Long Question And Answers 2020
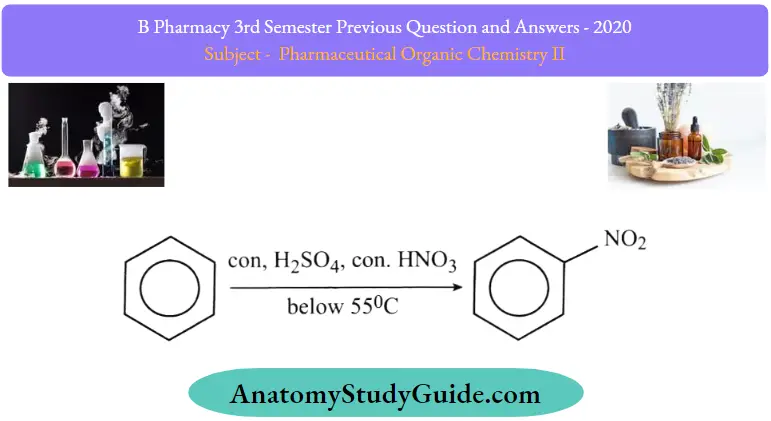
Question 1. What is an electrophilic aromatic substitution reaction? Discuss the mechanism of nitration and Friedelcraft’s alkylation. con, H2SO4, con. HNO3 below 55°C
Answer:
Electrophilic aromatic substitution reactions are organic reactions wherein an electrophile replaces an atom that is attached to an aromatic ring. Commonly, these reactions involve the replacement of a hydrogen atom belonging to a benzene ring with an electrophile.
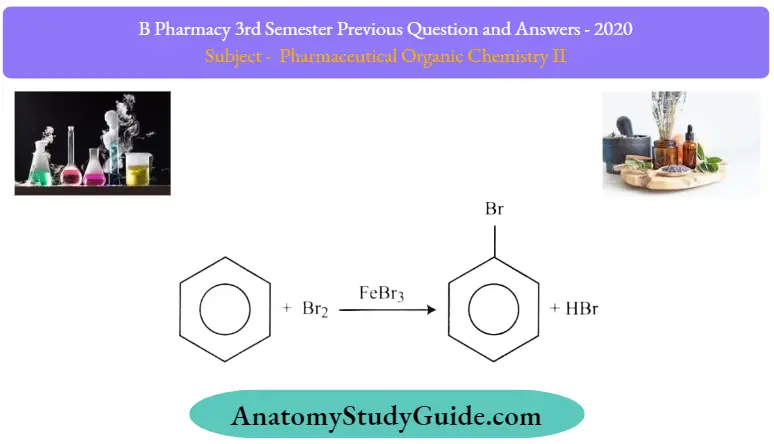
The aromaticity of the aromatic system is preserved in an electrophilic aromatic substitution reaction. For example, when bromobenzene is formed from the reaction between benzene and bromine, the stability of the aromatic ring is not lost. This reaction can be illustrated as follows
Aromatic Nitration:
- Nitration reaction involves the replacement of hydrogen with a nitro (NO2) group.
- Sulfuric acid (H2SO4) is used as a catalyst in this process.
- The acid is used to protonate the nitric acid which leads to the formation of nitronium ion.
- The nitronium ion can then be processed as per the mechanism of electrophilic aromatic substitution reaction.
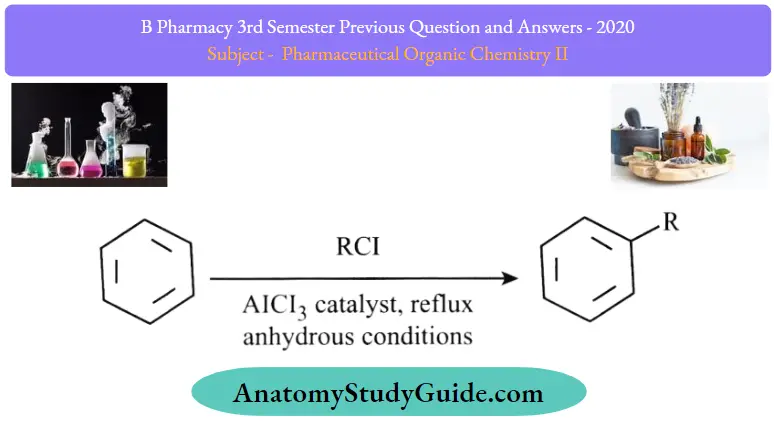
Friedel-Crafts Alkylation:
In Friedel-Crafts Alkylation, the hydrogen is mostly replaced with an alkyl group (R).
- In this reaction, alkyl halide such as CH3CH2Cl is used along with Lewis acids like AlCl3 or FeCl3 amongst others.
- The acid helps to accelerate the reaction by coordinating with the halogen. They further weaken the C-Cl bond and make it a better-leaving group.
- However, one disadvantage of this reaction is that the product is more nucleophilic than the reactant. Moreover, overalkylation can also occur.
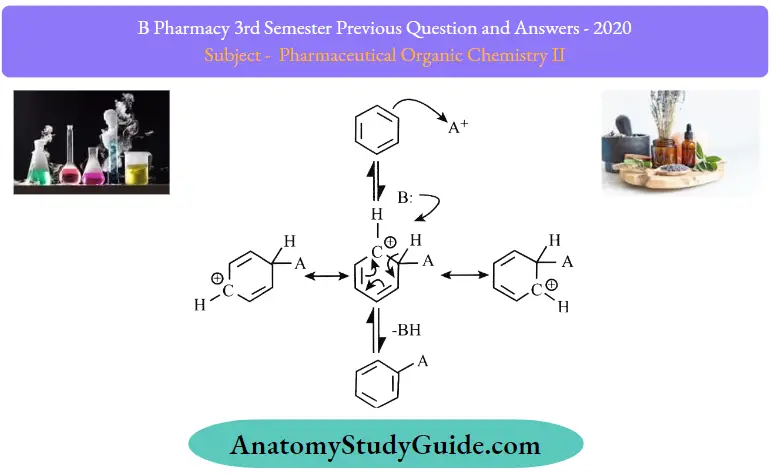
Electrophilic Aromatic Substitution Mechanism
An electrophilic aromatic substitution consists of three main fundamental components:
- During the reaction, a new 6 bond is formed from a C=C in the arena nucleophile.
- The proton is removed by the breaking of the C-H O bond.
- The C = C is reformed which restores the aromaticity.
As for the mechanism ofthe reaction, it usually includes two main steps.
Step 1:
- The reaction begins with the electrophile attacking the pi electrons present in the aromatic benzene ring.
- This results in the formation of positively charged and delocalized cyclohexadienyl cation or a resonance-stabilized carbocation known as an arenium ion
- This ion contains three resonance contributors.
- The electrophilic attacking the aromatic ring generally takes time and is a slow process.
- It is further endergonic and there is a presence of high activation energy due to the loss of aromaticity.
- Some main factors that are used to determine the attack of the electrophile are resonance, probability, and steric hindrance
Step 2:
- This step involves the deprotonation of the arenium ion by a weak base.
- The carbocation intermediate that is formed is attacked by a base that results in the loss of a proton. The electrons are then used to reform a pi bond and aromaticity is yet again restored.
- This is a very fast process and usually exergonic.
- An important thing to remember here is that the carbocation loses a proton as a result of the electrophile attacking the benzene ring
Question 2. What are phenols? Discuss the reactions of phenols.
Answer:
Phenol is an aromatic compound. The chemical formula of this organic compound is C6H6O. It consists of a hydroxyl group and a phenyl group attached. It considerably dissolves in water.
Reactions of phenols:
A hydroxyl group is attached to an aromatic ring and it is strongly activating ortho/para director, phenols possess considerable reactivity at their ortho and para carbons toward electrophilic aromatic substitution.
- Reactions ofthe Aromatic Ring: The -OH group in phenol is ortho and para directing because it increases electron density at ortho and para positions due to resonance. Thus phenol undergoes electrophilic substitution reactions.
- Halogenation: Like the -NH2 group, the -OH group is so activating that it is rather difficult to prevent polysubstitution. If it is required to arrest the reaction at the mono substitution stage, the reaction should be carried out in non-polar solvents like CCl4 and CS2 and at lower temperatures.
C2H6O Uses (Phenol):
- It is used as a precursor in drugs
- It is used as an antiseptic
- It is used in the production of nylon
- It is used to preserve vaccine
- It is used in oral analgesics
- Derivatives of phenol are used in beauty products like
- hair color and sunscreen
- It is used in the synthesis of plastics
- It is used to produce detergents and carbonates
Question 3. Discuss Bayer’s strain theory with limitations
Answer:
Baeyer’s strain theory:
Baeyer, Adolf(1885) proposed a theory to clarify the relative stability of the first few cycloalkanes based on the fact that in tetrahedral geometry, the normal angle between any pair of carbon atom bonds is 109.28′ (or 109.50)(methane molecule).
- In tetrahedral geometry, the bond angle for carbon atoms is 109.28′ (or 109.50)(methane molecule). Baeyer discovered that various cycloalkanes have different bond angles, as well as different properties and stability.
- He proposed angle strain theory based on this.
- The theory describes cycloalkane reactivity and stability.
- The optimum overlap of atomic orbitals, according to Baeyer, is achieved for a bond angle of 109.50. In a nutshell, it’s the best bond angle for alkanes.
- The most efficient and desirable overlap of atomic orbitals results in the highest bond strength and the most stable molecule.
- Rings cause strain when bond angles deviate from the ideal.
- The higher the pressure, the more unstable the system.
- Higher strain results in increased reactivity and combustion heat.
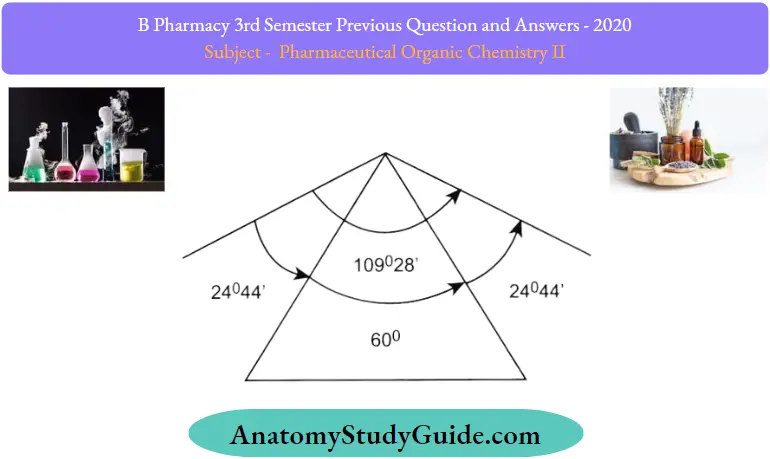
According to Baeyer, “any deviation of bond angle from the ideal bond angle value (109.50) creates a strain in the molecule.” The lower the variance, the less unstable the situation.
Bae yer’s Strain Theory Chemistry is Cycloalkanes are
Founded on the Following Assumptions:
- Planar rings are used in all ring structures. Unstable cycloalkanes arise from deviations from standard tetrahedral angles
- Negative strain is present in large ring structures, but they do not exist.
- Since the carbon rings of cyclohexane and higher cycloalkanes (cycloheptane, cyclooctane, cyclononane, etc.) are puckered rather than planar (flat), their bond angles are not greater than 109.50.
- These assumptions are useful in understanding cycloalkane ring system instability.
Baeyer Strain Theory in Cycloalkanes:
When carbon is bound to two other carbon atoms in an open-chain compound (propane), it is s sp/ hybridized, and these hybrid orbitals are used to form bonds (strong sigma bonds).
- Since the carbon atoms in cyclopropane do not use these hybrid orbitals to form bonds, the bond (bent bond) is weaker than a typical carbon-carbon bond. This is known as angle strain.
- The ring produces strain when bond angles deviate from the ideal.
- Higher strain results in increased volatility, reactivity, and heat of combustion. Simply put, the lower the deviation, the lower the instability.
- Baeyer discovered that various cycloalkanes have different bond angles, as well as different properties and stability.
- He proposed angle strain theory based on this. The theory describes cycloalkane reactivity and stability.
The cyclopropane ring is a triangle. The standard tetrahedral angle between two bonds is compressed to 600 and each ofthe two bonds involved is pulled in by 24.75p so all three angles are 600 instead of 109.50 (normal bond angle for carbon atom). The angle strain, or deviation, of each bond from the usual tetrahedral direction, is defined by the value 24.75p.
- Similarly, cyclobutane is square, with bond angles of 90p instead of109.5p (normal bond angle for carbon atom) to make the ring system square (angle strain 9.75p)
- For cyclopropane and cyclobutane ring systems, a deviation from the usual tetrahedral angle causes ring pressure.
- In comparison to molecules with a tetrahedral bond angle, the ring strain will make them unstable.
- In comparison to cyclobutane, Baeyer believes that cyclopropane should be a highly stressed and unstable compound.
- As a consequence, the triangle ring can be expected to open up at the slightest provocation, releasing the tension within it.
- This is valid since cyclopropane undergoes Br, ring-opening reactions.
Cyclopentane (angle strain 0.75p ) is considered to be the least stressed and the most stable. It is not surprising, then, that it has no ring-opening reactions.
- In cyclohexane, the angle strain is greater than in cyclopentane. If the number of carbon atoms in the ring increases, the strain increases as well.
- Theoretically, cyclohexane and higher cycloalkanes should become more unstable and reactive as time goes on.
- In contrast to this prediction, cyclohexane and its higher members are found to be very stable, undergoing substitution rather than additional reactions.
- As a result, the hypothesis adequately accounts for the first three.
Cyclopentane > Cyclobutane > Cyclopropane
Limitations of Baeyer Strain Theory in Cycloalkanes:
- In larger ring structures, Baeyer was unable to describe the impact of angle pressure.
- According to Baeyer, cyclopentane should be much more stable than cyclohexane, but the situation is the opposite.
- According to Baeyer, larger ring structures are not feasible due to negative pressure, but they do exist and are very stable.
- To remove angle pressure, larger ring structures are wrinkled (puckered) rather than planar
Question 4. Give any four methods of synthesis of cycloalkanes.
Answer:
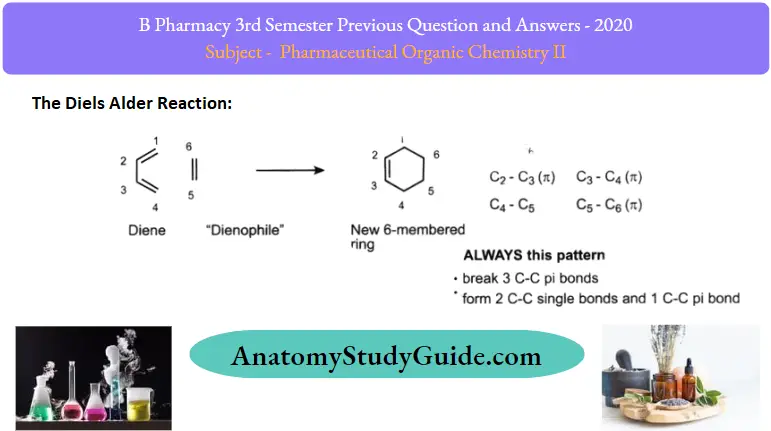
1. Diels Alder reaction:
The addition of conjugated diene to another unsaturated molecule generally called dienophile gives adduct. This reaction is called as diels alder reaction
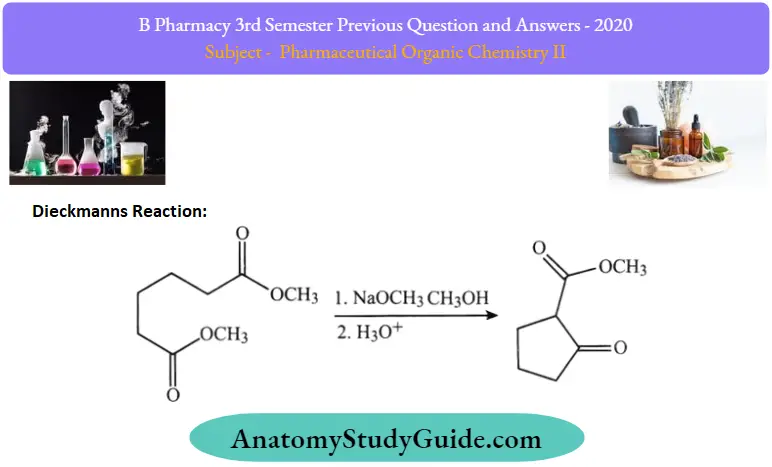
2. Dieckmanns reaction:
Esters ofdicarboxylic acids when heated with sodium followed by hydrolysis cyclic ketone is formed which on Clemmensen reduction gives cycloalkanes. It is an intramolecular Claisen Condensation reaction.
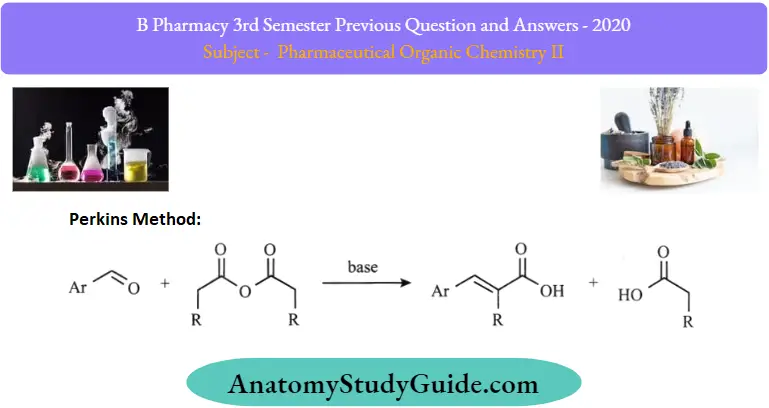
3. Perkins method:
This method is suitable for the synthesis of cycloalkanes up to six six-membered rings and was suggested by Perkin. When terminal dibromide is treated with malonic ester in the presence of sodium ethoxide, a cycloalkane-1,1-dicarboxylic ester is formed which upon hydrolysis followed by heating gives the corresponding cycloalkane.
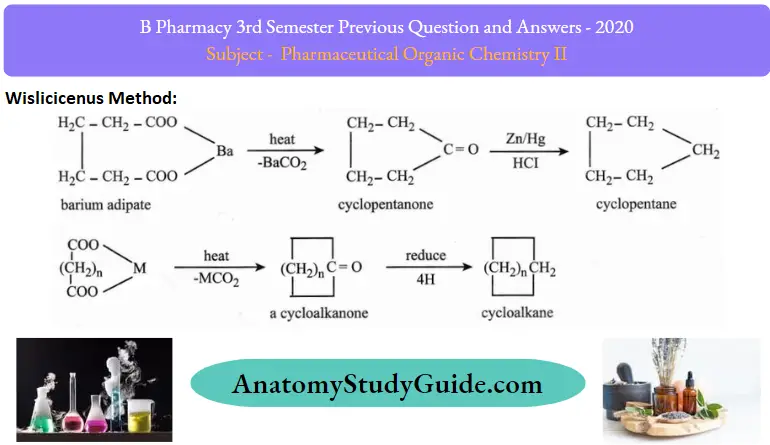
4. Wislicicenus method:
On heating calcium or barium salt of dicarboxylic acids, cyclic ketones is formed which can be further reduced to cycloalkane by Clemmensen reduction using Zn-Hg/ cone HCl.
Pharmaceutical Organic Chemistry 2 Long Question And Answers 2019
Question 1. What is an electrophilic aromatic substitution reaction? Discuss the mechanism of halogenation.
Answer:
Electrophilic aromatic substitution reactions are organic reactions wherein an electrophile replaces an atom that is attached to an aromatic ring. Commonly, these reactions involve the replacement of a hydrogen atom belonging to a benzene ring with an electrophile.
The aromaticity of the aromatic system is preserved in an electrophilic aromatic substitution reaction.
Mechanism of nitration of benzene:
For the formation of the electrophile you are going to substitute an -NO2 group into the ring, then the electrophile must be NO2+. This is called the “nitronium ion” or the “nitryl cation”, and is formed by a reaction between the nitric acid and sulphuric acid.
Note:
If you don’t understand why the electrophile has got to be NO2+, then you really should look at What is electrophilic substitution. before you go on. If you are going to substitute X onto the ring, then the electrophile must be X+. If you are going to insert a NO2 group onto the ring, then the electrophile must be NO2+
The Equation:
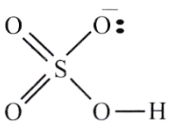
HNO3 + 2H2SO4 → NO2– + 2HSO4+H3O+
The hydrogen sulfate ion, H2SO4, will also be involved in The hydrogen sulfate ion, H2SO4, which will also be involved
The electrophilic substitution mechanism:
Stage one As the NO2+ ion approaches the delocalised electrons in the benzene, those electrons are strongly attracted toward the positive charge
Two electrons from the delocalized system are used to form a new bond with the NO2+ ion. Because those two electrons aren’t a part ofthe delocalized system any longer, the delocalisation is partly broken, and in the process, the ring gains a positive charge
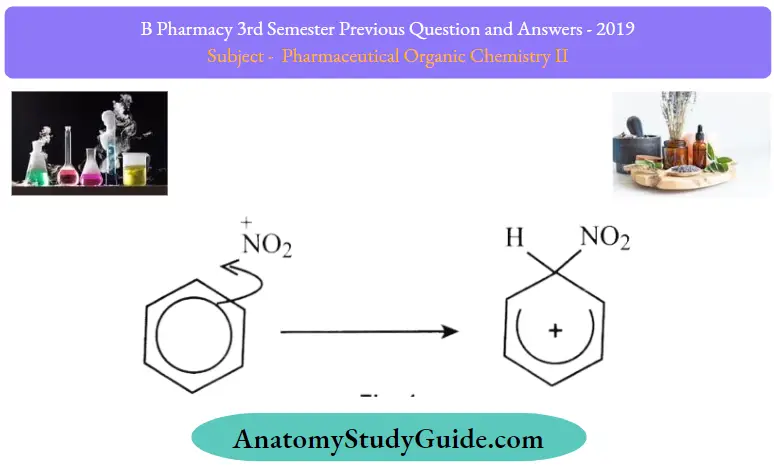
The hydrogen shown on the ring is the one that was already attached to that top carbon atom – it’s nothing new or subtle! We need to show it there because it has to be removed in the second stage.
Stage ftfoThe second stage involves a hydrogen sulfate ion, H2 SO4, which was produced at the same time as the N02+ ion (refer back to the equation showing the formation ofthe electrophile if you’ve forgotten).
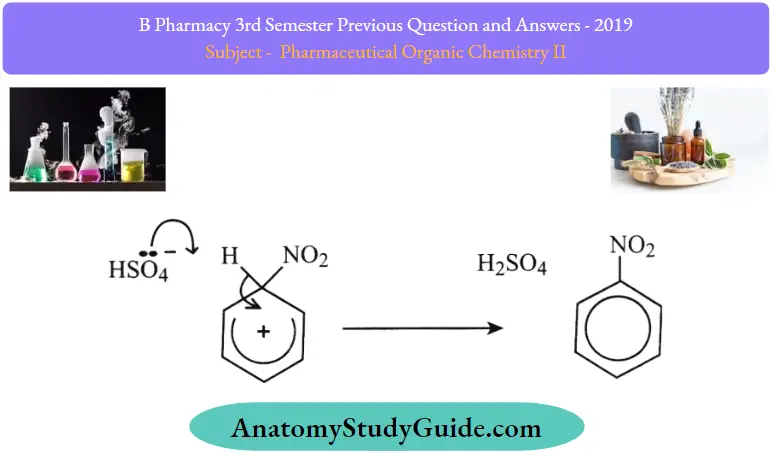
This removes hydrogen from the ring to form sulphuric acid – the catalyst has therefore been regenerated. The electrons that originally joined the hydrogen to the ring are now used to re-establish the delocalized system
Stage two – a sloppy way of writing the same thing:
You will often find the second stage of this reaction simplified in many (or even most) books.
The second stage is shown as:
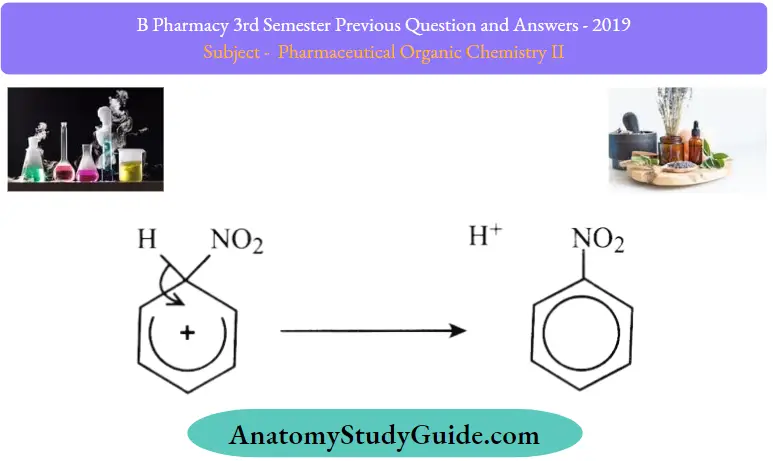
The hydrogen is shown as “falling off’ the ring as a hydrogen ion. This is sloppy and unsatisfactory on two counts:
- Hydrogen ions never exist on their own in a chemical reaction. A hydrogen ion is a raw proton – the most intensely positive thing you can imagine. It will always be attached to something else.
- By not showing the hydrogen sulfate ion, you can’t show that the sulphuric acid catalyst has been regenerated. That’s simply an unsatisfying
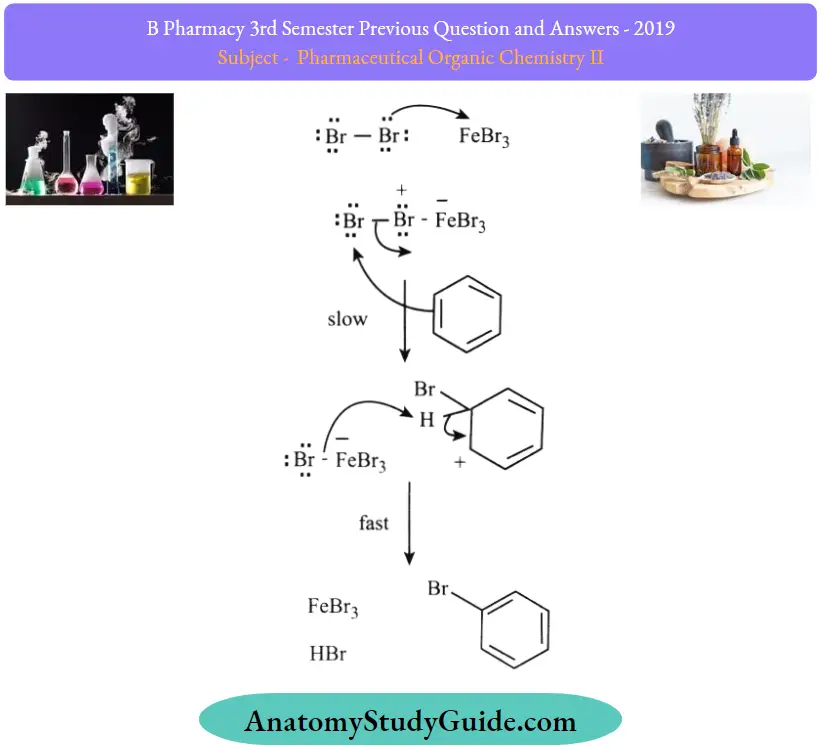
Mechanism of Halogenation of Benzene:
- Step 1: The bromine reacts with Lewis acid to create a complex that makes bromine more electrophilic.
- Step 2: The 5 electrons of aromatic C=C behave as a nucleophile which attacks the electrophilic Br and displaces iron tetrabromide.
- Step 3: The proton is removed from sp³ C and bears the Bromo group that reforms C=C and the aromatic system generates HBr and regulates active catalyst.
Question 2. What are aromatic acids? Explain the effect of substituent on the acidity of aromatic acids. Give the important reactions of benzoic acid.
Answer:
Aromatic acids include compounds that contain a COOH group bonded to an aromatic ring. The simplest aromatic acid is benzoic acid. Aromatic carboxylic acids show not only the acidity and other reactions expected of carboxylic acids (as an acid, benzoic acid is slightly stronger than acetic acid) but, similar to other aromatic compounds, also undergo electrophilic substitution reactions.
Effect of substituents on the acidity of aromatic acids:
- Electron-withdrawing groups: The conjugate base of benzoic acid is stabilized by electron-withdrawing groups. This makes the acid more acidic. Electron withdrawing groups deactivate the benzene ring to electrophilic attack and make benzoic acids more acidic.
- Electron-donating groups: The conjugate base of benzoic acid is destabilized by electron-donating groups. This makes the acid less acidic. Electron donating groups activate the benzene ring to electrophilic attack and make benzoic acids less acidic
Hydroxyl group: Electron-donating or electron-releasing group:
It behaves differently for different systems.
- For aliphatic systems with no conjugation, it behaves as an electron-withdrawing group due to the -I effect.
- For aliphatic systems with conjugation, it behaves as an electron-withdrawing group by the -I effect and an electron-donating group by the mesomeric effect.
- For aromatic systems,
- When it is in the ortho position, it behaves as an electron-withdrawing group by -I effect and an electron-donating group due to the mesomeric effect.
- When it is in the meta position, it behaves as an electron-withdrawing group by -I effect. Since the distance has increased as compared to ortho, its effect decreases.
- When it is in a para position, it behaves as an electron-donating group by mesomeric effect. Since the distance has increased further, the electron-withdrawing nature by -I effect is negligible
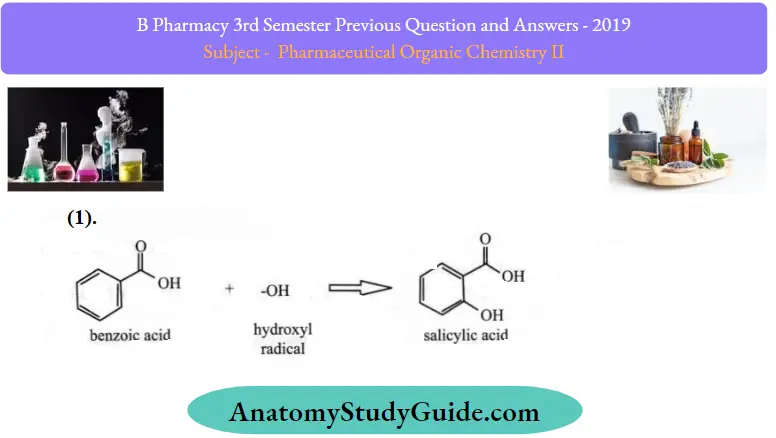
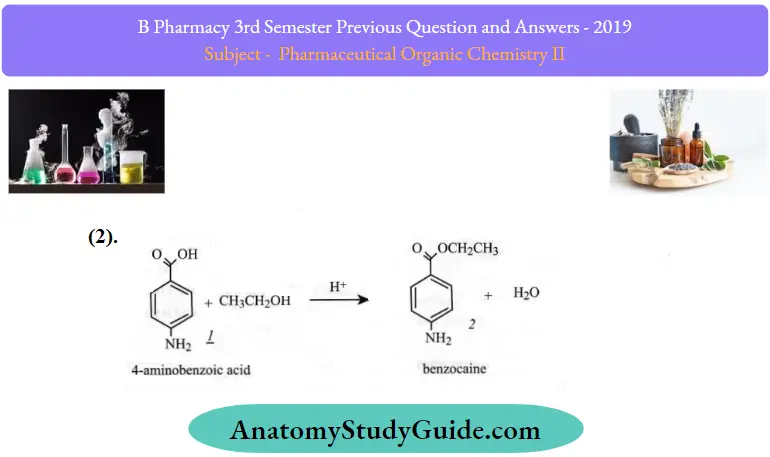
Reactions of benzoic acid:
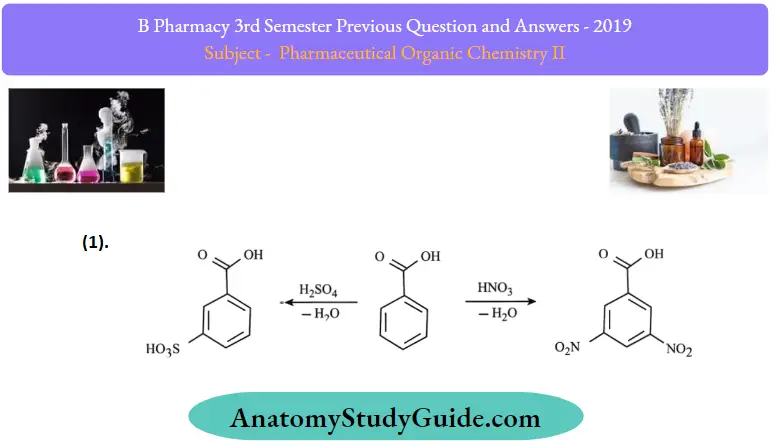
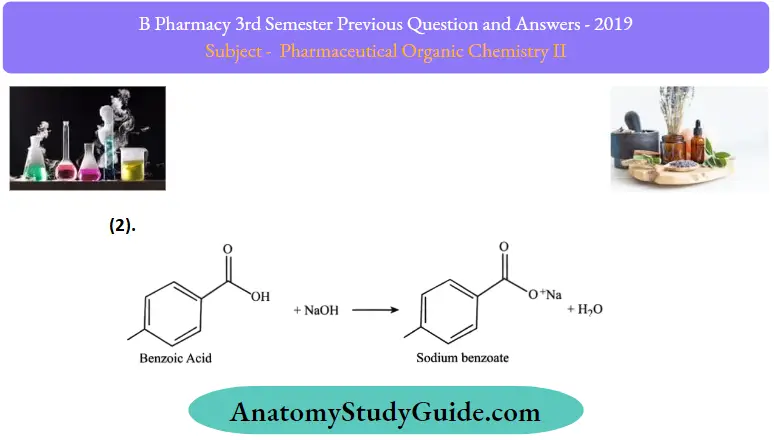
Question 3. Define fats and oils. Give any two reactions of fatty acids.
Answer:
- Fats: Some compounds that are soluble in organic solvents and mostly insoluble in water are called fats.
- Oils: Fats that are liquid at room temperature are called oils.
Reactions of fatty acids:
1. Reaction of the alkaline form of fatty acids with ethylene oxide
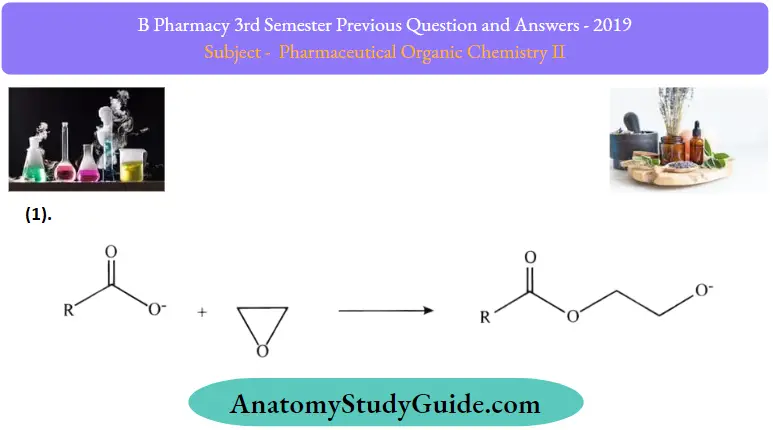
2. Propagation of ethoxylation:
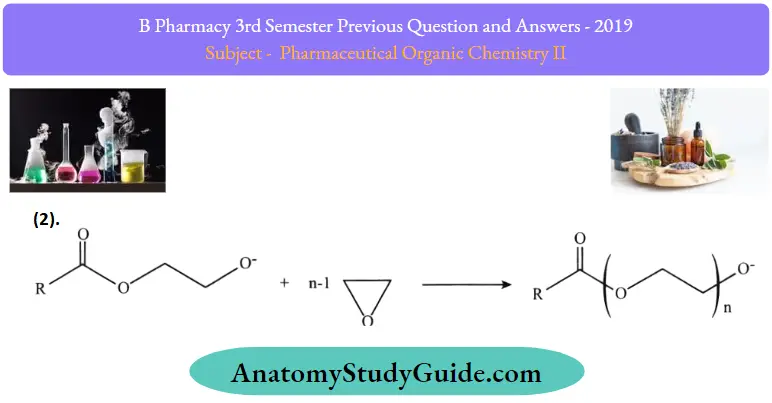
3. Transesterification of PEG monoester, formation of PEG diesters, and free PEG under alkaline conditions.
4.
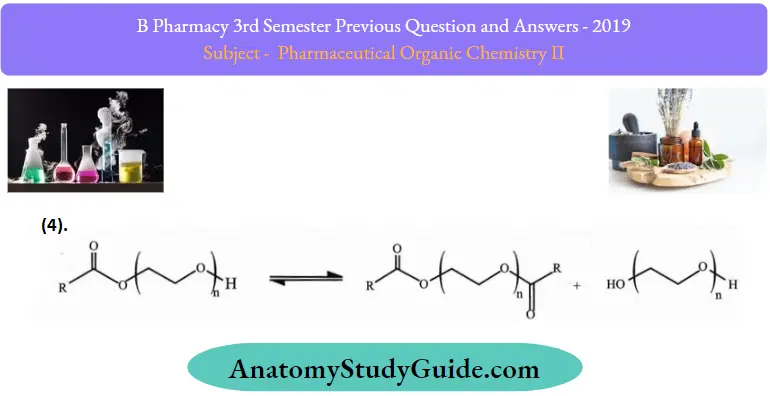
This ethoxylation process; as well as the esterification of fatty acids with PEG, results in a mixture of PEG monoesters, PEG diesters, and free PEG.
Question 4. What are aromatic acids? Give any three chemical reactions of benzoic acid. Add a note on the acidity of aromatic acids
Answer:
An aromatic amino acid (AAA) is an amino acid that includes an aromatic ring. Among the 20 standard amino acids, the following are aromatic: phenylalanine, tryptophan, and tyrosine. However, in addition to being aromatic, tyrosine can be classified as a polar amino acid
Reactions:
1. Acidity of aromatic acids:
Benzoic acid is the simplest of aromatic carboxylic acids. Two factors influence the acidity of substituted aromatic acids. The resonance effect and the inductive effect. Whereas the inductive effect only operates through sigma bonds, the resonance effect operates by electron or charge delocalization through pi bonds.
2. Inductive effect:
- When an aromatic acid has a substituent that does not have a lone pair of electrons or charge that can be delocalized in the aromatic nucleus, then, only the inductive effect can be invoked in explaining the degree of acidity.
- Whereas electron-donating groups suppress the acidity of benzoic acids, electron-withdrawing groups enhance the acidity.
- Halides are usually considered as weakly ring deactivating through the inductive effect. The halobenzoate anions are more stabilized than benzoate anions, hence the higher acidity of all isomeric halobenzoic acids relative to unsubstituted benzoic acid.
- The 2-halo benzoic acids are more acidic than 3- 3-halo benzoic acids, which are more acidic than the 4- 4-halo benzoic acid derivatives
Resonance effect:
- When both resonance and inductive effects apply in a specified substrate, the resonance effect dominates the inductive effect and thus determines the order of activity among isomeric carboxylic acids.
- The carboxylate anion obtained in the ionization of aromatic carboxylic acids is best stabilized when there are electron-withdrawing substituents attached to the aromatic nucleus.
- It is for this reason that the nitro benzoic acid derivatives, with the highly electron-withdrawing nitro group, are stronger acids than benzoic acid.
Leave a Reply