Physical Pharmaceutics 1 Short Question And Answers
Question 1. Define the term conjugated solution. Explain the phenol water system.
Answer:
Conjugate solution- a mixture of two partially miscible liquids A and B produces two conjugate solutions: one of A in B and another of B in A.
Phenol Water System Theory:
- Two liquids when mixed may be completely miscible, completely immiscible, or partially miscible.
- One will have a definite solubility in the other. An example of such cases are phenol water system. If we add phenol to water it will be soluble up to a certain solubility value will increase with the increase in temperatures.
- Similarly, if we add water to phenol same phenomenon will be observed. The number of homogenous, mechanically separable, and physically distinct parts of a heterogenous system is known as the number of phases, P of the system.
- Each phase is separated from other phases by a physical boundary. When an equilibrium exists between several phases under external controlling conditions such as temperature, pressure, and concentration,
The following will hold well:
F = C-P+2 (phase rule)
Where,
- P = number of phases in equilibrium
- C = number of components in the system
- F = number of degrees of freedom
When 2 partially miscible liquids are mixed and shaken together, we get 2 solutions of different compositions. Above a particular temperature, such solutions are completely miscible in all proportions. Such a temperature is known as the critical solution temperature.
Phenol Water System Procedure:
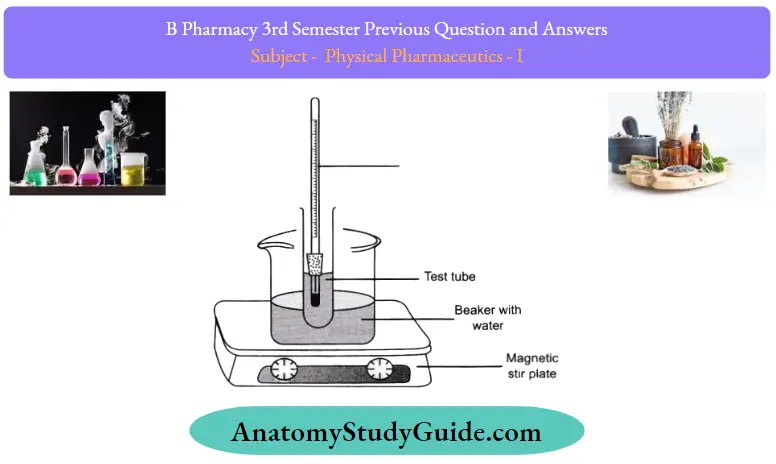
- Weigh about 5gm of phenol in a dry boiling tube.
- Add 2 ml ofdistilled water. The solution is stirred
- Heat the solution in a water bath, with continuous stirring.
- At a certain temperature, the mixture becomes clear.
- Note this temperature.
- Remove the tube from the water bath, and allow the solution to cool down slowly. Note the temperature at which the turbidity reappears.
- Repeat steps 2-6 after each addition of 2ml of the solution, followed by heating and subsequent cooling, note the temperature of the disappearance of turbidity, and the temperature ofthe reappearance of turbidity.
- The observation is that the temperature of complete miscibility rises, reaches a maximum value, and then decreases
Question 2. Explain organic molecular complexes with examples.
Answer:
These types of complexes are formed by noncovalent interaction between ligand and substrate and held together by weaker forces or hydrogen bonding.
Organic Molecular Complexes Classification
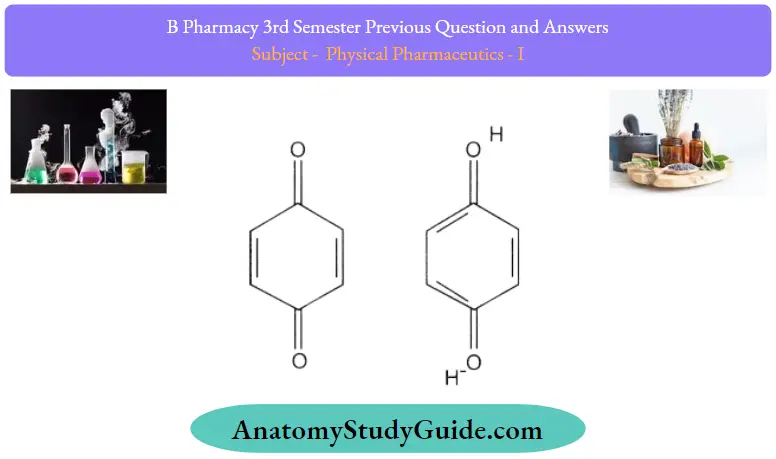
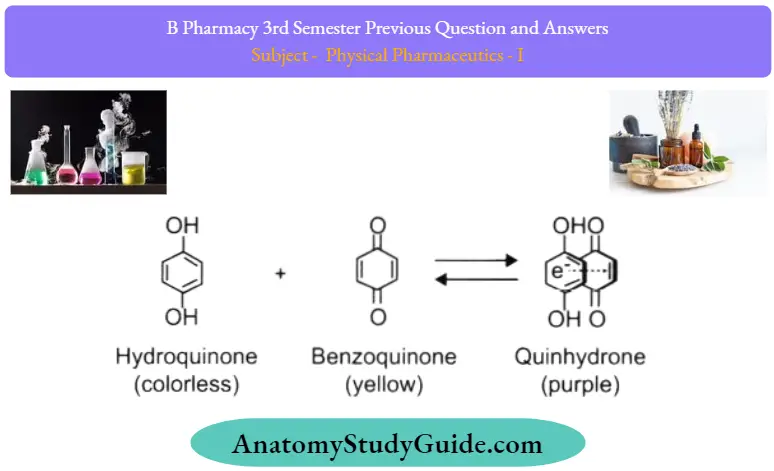
1. Quinhydrone type:
- They are formed when the alcohol solution of benzoquinone and the alcoholic solution of hydroquinone is mixed in equimolar concentration.
- The complex is formed by overlapping a pi cloud of electron-deficient benzoquinone with a pi cloud of electron-rich hydroquinone. These are used as an electrode to determine pH.
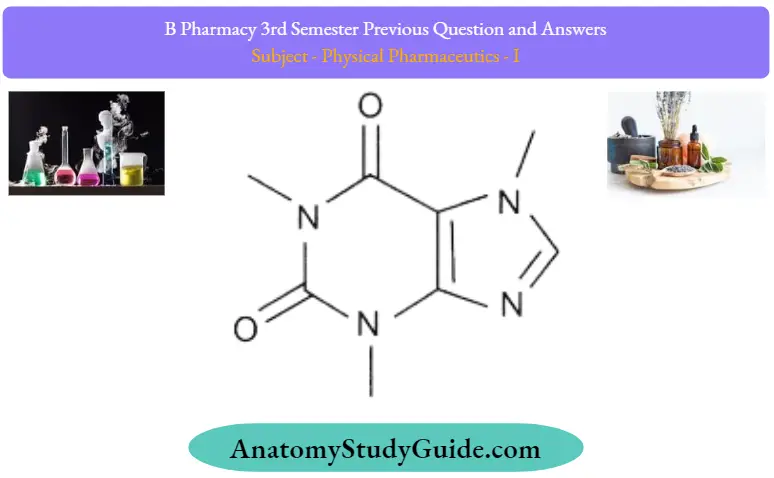
2. Drug and caffeine complex:
- Several acidic drugs are known to form complexes with caffeine. Drugs such as benzocaine, and procaine form complexes with caffeine.
- • The mechanism behind this is hydrogen bonding between the polarized carbonyl group of caffeine and hydrogen atom of the acidic drug. Induced dipole-dipole force between carboxy oxygen of benzocaine and electrophilic nitrogen of caffeine
Caffeine drug complex:
- May enhance or inhibit solubility
- Also, these complexes are used to mask the taste of dmg
- Improve stability of drug
- Improve absorption and bioavailability of drugs
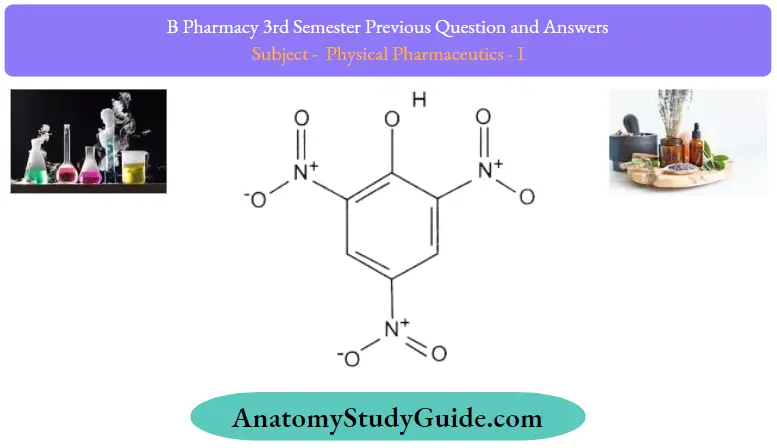
3. Picric acid complexes:
- Picric acid is a strong acid from organic molecular complexes with weak bases.
- The complex is an indication of the magnitude of carcinogenic activity.
4. Polymer complexes:
Polymeric materials such as polyethylene glycols, and carbowaxes contain nucleophilic oxygen and form complexes with various drugs.
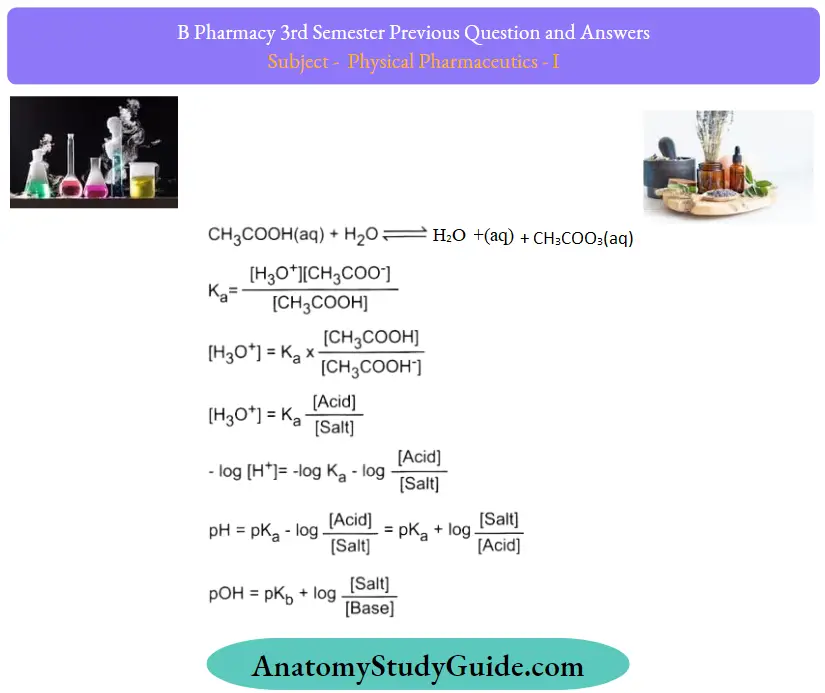
Question 3. What are buffers? Derive an equation for weak acid and its salts.
Answer:
A buffer is an aqueous solution of either a weak acid and its conjugate base or a weak base and its conjugate acid. A buffer may also be called a pH buffer, hydrogen ion buffer, or buffer solution.
Question 4. Explain the different methods to determine the HLB of a surfactant.
Answer:
HLB is a hydrophilic-lipophilic balance consists a laboratory scale in which values are assigned to different surfactants according to their nature. This system was introduced by Griffin. A value of 1-20 on the HLB scale represents the lipophilic and hydrophilic parts. A higher number indicates that the agent is hydrophilic while a low HLB value indicates the agent is lipophilic.
- HLB value of 1 indicates surfactants is soluble in oil
- HLB value of 20 indicates surfactants are soluble in water
- HLB value 1.8-8.6 indicates surfactants are lipophilic
- HLB value is 9.6-16.7 indicates the surfactant is hydrophilic
- The HLB value of surfactants containing glycerin monostearate is calculated by
HLB = 20 × (1-S/A)
HLB value of beeswax and lanolin derivatives are calculated by
HLB = E + P / S
Where,
- E is the percentage by weight of ethylene oxide
- P is the percentage by weight of the polyhydric alcohol group
In another method, the component groups of surfactants are assigned group members. By adding these numbers
HLB value is calculated.
HLB = hydrophilic group number – lipophilic group number + 7
Question 5. Enumerate different methods of analysis of complexes. Explain the solubility method of analysis.
Answer:
1. Analysis Complexation Methods
- Job’s Method of Continuous Variation
- As per the Job, the species possess several characteristics that are
- Dielectric constant.
- Refractive Index.
- Spectrophotometric extinction coefficient.
Analysis of complexes Principle:
When there is no complexation between the species, the value of the property is additive.
- On complexation, these properties change but additive rule do not hold good.
- The change in the characteristics proves that the complexation has taken place.
- Let’s take two species A and B whose individual dielectric constant in solid form and Absorbance in solution form were measured.
- Then two species in both forms were mixed. The dielectric constant and absorbance were determined
- The individual values are subtracted with mixed additive values and the result was found out.
- If the result is zero then no complexity and if the result is not zero then there is complexity.
2. pH Titration Method
pH Titration Principle:
- This method is applicable for that complex that produces the changes in pH on interaction. The significant change in pH will determine that complexation has taken place.
- Let us take 75 ml ofglycine solution and it is titrated with strong alkali NaOH solution. The pH was recorded. A graph was drawn between pH and volume of NaOH added.
- In another test, a complex solution of glycine and copper salt is titrated. The change in pH with increments of NaOH solution was also recorded. A graph was drawn between pH and volume of NaOH added.
- The two plots are compared and it is seen that the plot of glycine with copper is well below that ofthe pure glycine, which indicates that complexation is obtained throughout the titration range.
3. Distribution Method
- The method of distributing a solute between two immiscible solvents can be used to determine the stability constant for certain complexes.
- The distribution behavior of a solute between two immiscible liquids is expressed by distribution or partition co-efficient.
Distribution Method Principle:
When a solute complexes with an added substance, the solute distribution pattern changes depending on the nature ofthe complex.
The complexation of iodine by potassium iodide.
I2 + K+ I- ————- K+ I3 –
The Equilibrium stability constant,
K = [K+ I3 – ]/ [I2] [K+ I – ] 9
The distribution coefficient of iodine between disulfide and water is 625.
The K value iodine-potassium iodide complex is 954.
This change in the distribution coefficient proves that the complexation has taken place
4. Solubility Method
Solubility Method Principle:
- When the components in a mixture produce a complex, the solubility of one of the components may be increased or decreased. The change in solubility is a sign of complexity.
- The experimental data can be used to analyze complexes in terms of donor-acceptor ratio and equilibrium stability constant.
- Example – PABA and Caffeine and Paracetamol Caffeine.
Spectroscopy Method:
The study of donor-acceptor (D-A) or charge transfer complexation is generally undertaken with absorption spectroscopy in the visible and UV regions of the spectrum.
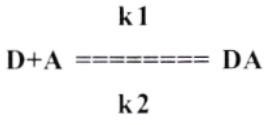
Where, D and A represent electron donor and acceptor, kl and k2 are interaction rate constants. K = kl/ k2 = Equilibrium or stability constant for complexation.
- The absorbance A of the charge transfer band is measured at a definite wavelength and the constant K is obtained from the Benesi-Hildebrand equation.
A0/A = (l/ε) + (1/Kε) (1/D0)
- Where AO and D0 are the initial concentrations of acceptor and donor species in mole/liter.
- Is the molar absorptivity of the charge-transfer complex at its particular wavelength and K is the stability constant in liter/mole.
- A plot of A0/A versus 1/D0 results in a straight line with a slope of 1/Kε and an intercept of l/ε.
- The spectrometric method was used to investigate the interaction of nucleic acid bases with catechol, epinephrine, and isoproterenol.
Question 6. What is pH? Explain Sorenson’s pH scale. pH is defined as the negative logarithm of H+ ion concentration. Hence the meaning of the name pH is justified as the power of hydrogen
Answer:
Sorensen pH scale:
- A system of grading pH levels and different concentrations of H3O+ ions has been devised and named after Sorenson, who was the original developer.
- Having a pH value of zero indicates a hydrogen ion concentration of 1 pH levels of this level are indicative of highly acidic solutions. This indicated the strongly alkaline properties.
- pH 14 means hydrogen ion concentration is between 10-14. The central point of pH in the scale is 7, meaning there is a concentration of 10-7 hydrogen ions.
- It represents neutrality as 7. Generally, a pH value under 7 indicates an acidic region and a pH value above 7 indicates an alkaline area. By measuring the pH levels of colloidal and non-aqueous solutions, the activity scale has little correlation.
Question 7. State and explain Nernest distribution law along with its limitations.
Answer:
“ If a solute “X” distributes between two non-miscible solvents ‘a’ and ‘b’ at a constant temperature and “X” is in the same molecular for in both of them, then the ratio of the concentrations of “X” in the two solvents is a constant quantity ”

The constant K is called the partition coefficient and is also denoted by Kd (distribution constant)

For example, Iodine a violet cyrstalline solid when added to a mixture of two immiscible liquids water and carbon tetrachloride distributes in both of them. The value ofK in this case is 85 at room temperature i.e. 298 K.
Limitations of the Nernst Distribution Law:
The law is valid when the molecular state ofthe solute is the same in both the solvents. If the solute undergoes dissociation or association in any one ofthe solvents, then in such cases the distribution law no longer holds good
Question 8. Define diffusion and dissolution.
Answer:
Diffusion:
“Diffusion is the movement of molecules from a region of higher concentration to a region of lower concentration down the concentration gradient.”
Dissolution:
Dissolution refers to a process by which dissolved components, i.e. solutes, form a solution in a solvent. In this process, a solution of the gas, liquid or solid in the original solvent is formed.
Question 9. Two applications of buffers.
Answer:
- Buffers’ resistance to changes in pH makes these solutions very useful for chemical manufacturing and essential for many biochemical processes. The ideal buffer for a pH has a pKa equal to the pH desired since a solution of this buffer would contain equal amounts of acid and base and be in the middle ofthe range of buffer capacity.
- Buffer solutions are necessary to keep the correct pH for enzymes in many organisms to live. Many enzymes work only under very precise conditions; if the pH is too far, the enzymes slow or stop working and can denature, thus permanently disabling its catalytic activity. A buffer of carbonic acid (H2CO3) and bicarbonate (HCO–3) present in blood plasma, help to maintain a pH between 7.35 and 7.45. Pepsin is another example that shows maximum activity at pH 1.5.
- Industrially, buffer solutions are used in fermentation processes.
- Buffers can also be used to maintain the drug in its ionized as well as unionized form. The ionized form of a drug is more water-soluble than the unionized form. Buffers can be used to maintain a drug in its ionized (salt) form for aqueous solutions. The unionized form of the drug is more lipid-soluble than the ionized form. The unionized form, therefore, penetrates biological membranes much more efficiently than the ionized form.
Question 10. Define the spreading coefficient.
Answer:
The spreading coefficient or parameter is a measure of the tendency ofa liquid phase to spread (complete wetting) on a second, liquid, or solid phase.
The spreading coefficient S is the difference between the work of adhesion W1,2 between the phases and the work of cohesion W1,1of the phase under consideration:
S = W1,2 – W1,1
Equally, the spreading coefficient can be expressed as the difference between the surface tensions σ1 and σ2 and the interfacial tension σ1,2
S = σ2 – σ1 – σ1,2
If the spreading coefficient is positive, the phase under consideration spreads; if it is negative, wetting is not complete.
Question 11. Write a note on inclusion complexes.
Answer:
The inclusion compounds are of two general types, the cage or clathrates (Greek word meaning “lock”) and the channel compounds. These compounds have one underlying principle in common: their formation depends on the spatial “fitting in” of the guest molecules into the crystal lattice cavities or the “holes” of liquid aggregates of host molecules. There is on chemical affinity between the host and guest molecules in the usual sense,
For example: Inclusion compounds of even inert gases have been formed.
Inclusion compound formation depends upon the size and the share of guest molecules and the reciprocal size of the cavities created by the host molecules.
Question 12. Write any four examples of pharmaceutical buffers.
Answer:
- Acetic acid/ sodium acetate
- Phosphate acid/ sodium phosphate
- Citric acid/ sodium citrate
- Boric acid/ sodium borate
Question 13. What are clathrates?
Answer:
A clathrate is a chemical substance consisting of a lattice that traps or contains molecules. Most clathrate compounds are polymeric and completely envelop the guest molecule, but in modern usage, clathrates also include host-guest complexes and inclusion compounds. Clathrates are inclusion compounds “in which the guest molecule is in a cage formed by the host molecule or by a lattice of the host molecule
Question 14. Define vapor pressure.
Answer:
Vapor pressure, also known as vapor equilibrium pressure, can be defined as the pressure exerted (in a system featuring thermodynamic equilibrium) by a vapor with its condensed phases (solid or liquid) in a closed system at a given temperature. The equilibrium vapor pressure is known to serve as an indicator of the evaporation rate of a liquid.
A material that, at normal temperatures, has a high vapor pressure is generally referred to as a volatile material. It can be noted that the pressure exhibited above a liquid surface by the vapor is called vapor pressure
Question 15. Define polymorphism with examples.
Answer:
Polymorphism is the ability of solid materials to exist in two or more crystalline forms with different arrangements or conformations ofthe constituents in the crystal lattice.
- These polymorphic forms of a drug differ in physicochemical properties like dissolution and solubility chemical and physical stability, flowability, and hygroscopicity.
- These forms also differ in various important drug outcomes like drug efficacy, bioavailability, and even toxicity.
- Polymorphic studies are important as a particular polymorph can be responsible for a particular property that might not be exhibited by any other form.
Question 16. What are quinhydrone complexes? Give example.
Answer:
This molecular complex is formed by mixing equimolar quantities of benzoquinone with hydroquinone. Complex formation is due to the overlapping of the w-framework of the electron-deficient benzoquinone with the framework of the electron-rich hydroquinone (charge transfer).
Question 17. Define isotonic and paratonic solutions.
Answer:
Isotonic solutions:
Isotonic solutions are those that have equal osmotic pressure and water potential as both the solutions have an equal concentration of water molecules. E.g., blood serum is isotonic to the physiological salt solution. Solutions with the same tonicity do not allow water to pass through the cell membrane
Paratonic Solutions:
Paratonic solutions are the solutions with different osmotic pressures. Paratonic solutions when instilled into eyes or nose may cause irritation especially ifthe quantity instilled is large
Question 18. Describe the solubilization process.
Answer:
Mechanism of Solubilization Polar solvents:
- Owing to their high dielectric constant, polar solvents reduce the force of attraction between oppositely charged ions in crystals
- Polar solvents break covalent bonds of potentially strong electrolytes by acid-base reactions
- Polar solvents are capable of solvating molecules and ions through dipole interaction forces, particularly hydrogen-bond formation, which leads to the solubility ofcompound.
However the nonpolar solvents won’t obey the above mechanism of solubilization, so they are unable to dissolve the ionic and polar solutes. Nonpolar solvents can dissolve the nonpolar solute with similar internal pressure through induced dipole interactions.
In short, solubilization takes place by consideration of:
- Polarity
- Dielectric constant,
- Association,
- Salvation,
- Internal pressure,
- Acid-base reaction
Question 19. What are eutectic mixtures? Explain with examples.
Answer:
A eutectic system is a homogeneous, solid mixture of two or more substances that form a super-lattice; the mixture either melts or solidifies at a lower temperature than the melting point of any of the individual substances. The phrase most commonly refers to a mixture of alloys. A eutectic system only forms when there is a specific ratio between the components.
Examples of Eutectic Systems:
Several examples of eutectic systems or eutectoids exist, in metallurgy and various other fields.
These mixtures typically have useful properties that are not possessed by any single constituent substance:
- Sodium chloride and water form a eutectoid when the mixture is 23.3% salt by mass with a eutectic point at -21.2 degrees Celsius. The system is used to make ice cream and to melt ice and snow.
- The eutectic point ofthe mixture of ethanol and water is nearly pure ethanol. The value means there is a maximum proof or purity of alcohol that can be obtained using distillation.
- Eutectic alloys are often used for soldering. A typical composition is 63% tin and 37% lead by mass.
- Eutectoid glassy metals exhibit extreme corrosion resistance and strength.
- Inkjet printer ink is a eutectic mixture, permitting printing at a relatively low temperature.
- Galinstan is a liquid metal alloy (composed of gallium, indium, and tin) used as a low-toxicity replacement for mercury
Question 20. Explain in detail diffusion principles in biological systems.
Answer:
Diffusion:
Molecules move in mass, usually due to gradients of concentration when they are subjected to random motion.
Diffusion It involves:
- Dissolving a drug from its dose form.
- There are four types of dialysis:
- Ultrafiltration
- Microfiltration
- Dialysis, and
- Hemodialysis.
- Drug distribution and phenomenon in biological tissue.
- Determination of polymer molecular weight.
- Predicting absorption and elimination ofdrugs.
Diffusion Types:
- Passive diffusion: Moving from a high-low concentration zone, materials are being moved.
- Diffusion continues until the gradient has disappeared
- Facilitated diffusion: Cellular membrane molecules are transported across the membrane by special transport proteins.
- Active transport: A progression of molecules along a membrane against a gradient of concentration from a region of low concentration to high concentration.
- Filtration: Influence of hydraulic pressure on the movement of the solvent molecule by the following curve.
Diffusion Laws:
Fick’s first theory of diffusion
If steady-state diffusion is assumed, diffusion flux has a direct relationship with concentration gradients.
1. J = -D dc/dx:
Where,
- J = diffusion flux
- Whenever D is given, it is the diffusion co-efficient = change in concentration of material
- Distance change (cm) = dx
- Mass is transferred across unit cross-section area per unit by diffusion
2. J = dM/S dt:
Where,
- Second law of diffusion according to Fick
- Changes in concentration in one region with time are proportional to the increase or decrease in concentration gradient at that particular point in the system.
3. Dc/dt = -dJ/dx
Question 21. What are buffered isotonic solutions? Explain
Answer:
Buffered Isotonic Solutions Pharmaceutical solutions that are meant for application to delicate membranes of the body should also be adjusted to approximately the same osmotic pressure as that of the body fluids.
Isotonic solutions cause no swelling or contraction ofthe tissues with which they come in contact and produce no discomfort when instilled in the eye, nasal tract, blood, or other body tissues. Isotonic sodium chloride is a familiar pharmaceutical example of such a preparation.
The need to achieve isotonic conditions with solutions to be applied to delicate membranes is dramatically illustrated by mixing a small quantity of blood with aqueous sodium chloride solutions of varying tonicity.
- Isotonic: If a small quantity of blood is mixed with a solution containing 0.9 g of NaCl per 1 00 mL, the cells retain their normal size. The solution has essentially the same salt concentration and hence the same osmotic pressure as the red blood cell contents
- Hypertonic: If the red blood cells are suspended in a 2.0% NaCl solution, the water within the cells passes through the cell membrane in an attempt to dilute the surrounding salt solution. This outward passage of water causes the cells to shrink and become wrinkled or crenated.
- Hypotonic: If the blood is mixed with 0.2% NaCl solution or with distilled water, water enters the blood cells, causing them to swell and finally burst, with the liberation of hemoglobin. The salt solution in this instance is said to concern the blood cell contents. Finally, This phenomenon is known as hemolysis.
Isosmotic solutions:
The red blood cell membrane is not impermeable to all drugs; that is, it is not a perfect semipermeable membrane. Thus, it will permit the passage of not only water molecules but also solutes such as urea, ammonium chloride, alcohol, and boric acid.
- These solutes are regarded as solvents and they do not exert an osmotic pressure on the membrane (the solutions are isosmotic but not isotonic).
- A 2.0% solution of boric acid has the same osmotic pressure as the blood cell contents when determined by the freezing point method and is therefore said to be isosmotic with blood.
- The molecules of boric acid pass freely through the erythrocyte membrane, however, regardless of concentration.
- As a result, this solution acts essentially as water when in contact with blood cells. Because it is extremely hypotonic concerning the blood, boric acid solution brings about rapid hemolysis.
- Therefore, a solution containing a quantity of drug calculated to be isosmotic with blood is isotonic.
- Accordingly, a 2.0% boric acid solution serves as an isotonic ophthalmic preparation. Tonicity
Osmolality and osmolarity:
These are colligative properties that measure the concentration of the solutes independently of their ability to cross a cell membrane.
- Tonicity:
- This is the concentration of only the solutes that cannot cross the membrane since these solutes exert an osmotic pressure on that membrane.
- Tonicity is not the difference between the two osmolarities on opposing sides ofthe membrane.
- The solution might be hypertonic, isotonic, or hypotonic relative to another solution. A solution containing a quantity of drug calculated to be isosmotic with blood is isotonic only when the blood cells are impermeable to the solute (drug) molecules and permeable to the solvent, water.
Question 22. Write a note on critical solution temperature and its applications.
Answer:
Critical solution temperature:
- If temperatures are raised or lowered, the temperature where complete miscibility is reached for partially miscible liquids that are not miscible in ordinary circumstances.
- A mixture will become at a certain temperature if all of its components become soluble. The temperature of the upper critical solution is called the upper solution temperature or upper critical solution temperature. Partially miscible liquids become more stable at a certain temperature before becoming completely miscible.
- Temperatures lower than this are known as critical solution temperatures or solution temperatures. If there is an impurity added, the temperature above the phase may change.
- Lower critical temperature- lower than the temperature at which a two liquid phases are completely mixed and behave like a single phase.
Critical solution Applications:
- Critical solution temperature can be used to determine the purity of a substance. If there are small amounts of impurities present in CST values, the value will be impacted very strongly. It is usually a linear relationship between CST value and impurity concentration.
- The CST values of alcohol cyclohexane systems enable us to estimate the amount of water present in the alcohol
- For example, phenolic disinfectants used in water are formulated using this principle.
- A very small amount of sodium oleate reduced the CST value ofthe phenol water system considerably as given by cresol with the soap solution.
- Characterization of polymer mixtures is done by determining the lower critical solution temperature.
- With the addition of the third substance, the liquids can be separated using this method. A third substance is added to the extraction to extract drugs for analysis.
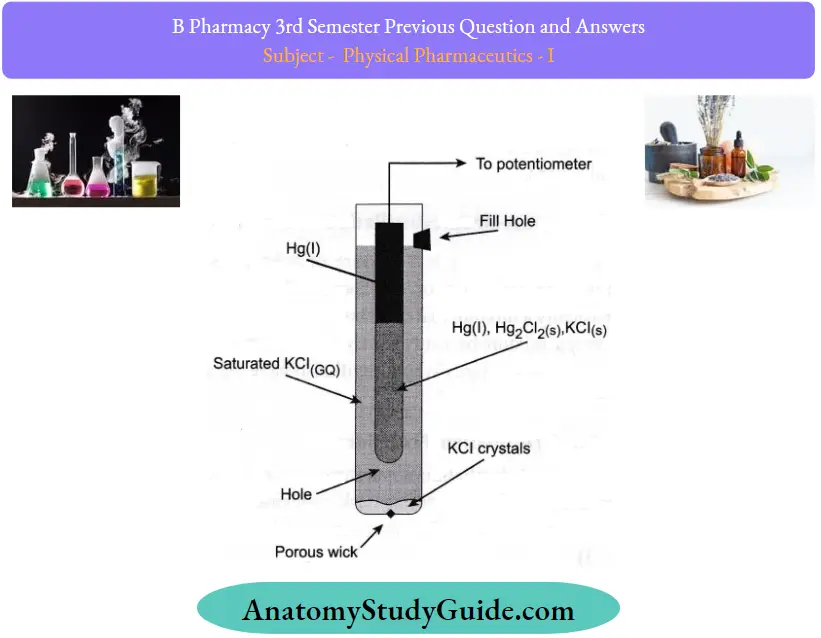
Question 23. Explain any one method for the determination of pH.
Answer:
The electrometric method is the most accurate ofthe methods. It measures accurately to 0.1 to 0.001 pH.
pH Principle:
The basic principle involved in electrometric pH determination is to determine the activity ofthe hydrogen ion by potentiometric measurement using a standard hydrogen electrode and a reference electrode.
The electronic pH measurement system consists of:
- Measuring electrode
- Reference electrode
1. Measuring electrode:
- It consist of the glass envelope having pH pH-sensitive glass membrane at the bottom which contains a constant pH buffer solution.
- A glass electrode is made of a thin glass membrane of special composition.
- This electrode is dipped in the measuring solution so that the potential is developed at the platinum electrode which is proportional to the pH of the measuring solution.
- This potential is measured against a standard calomel electrode.
2. Reference electrode:
The calomel electrode can be used as a reference electrode having a glass envelope containing a glass tube calomel along with platinum wire submerged there.
- This tube is surrounded by a saturated KCl solution that diffuses slowly into the process liquid through the liquid junction provided by the asbestos fiber.
- Because of this, the reference electrode develops a constant potential. A pH meter is composed of a probe, which is formed by two electrodes.
- This probe passes electrical signals to a meter that displays the reading in pH units. The measuring and reference electrodes together form an electrolyte cell.
- A voltage difference occurs due to the differences in the electron mobility produced by the two electrodes. This net voltage is recorded and calibrated as a function ofpH ofthe measuring liquid.
- A pH meter essentially measures the electrochemical potential between a known liquid inside the glass electrode and an unknown liquid on the outside.
- Since the operation of the electrode depends on the electrical resistivity of the glass, the temperature change can cause an error in the pH reading.
- To compensate for changes in the temperature of the ensuring solution, a temperature compensation resistance is included in the circuit that is immersed in the solution.
pH Advantages:
- It is more precise than the colorimetric method.
- Readings do not fluctuate by the natural color or opacity of a solution.
- Results can be easily read
pH Disadvantages
- Chances of damaging the electrode.
- Expensive equipment.
- Some chemicals other than H+ may cause spurious readings
Question 24. Write four applications of complexation in pharmacy.
Answer:
- Complexation formation has been used to alter the physicochemical & biopharmaceutical properties of drugs.
- In various types of poisonings
- In chug absorption & bioavailability from various dosage forms.
- Complexation is used in solubilization.
- Stability of product.
- Therapeutic agents such as.
- In diagnosis, therapeutic tool, Assay of drug
- Development of novel drug delivery system.
- Partition coefficient.
Question 25. Define steady-state diffusion and sink condition.
Answer:
Steady-state diffusion is a form of diffusion that takes place at a constant rate. Here, the number of moles of particles that cross a given interface is constant with time. Therefore, throughout the system, the rate of change of the concentration with distance (dc/dx) is a constant value and the change of concentration with time is zero (dc/dt).
Question 26. Two applications of amphiphiles in pharmacy.
Answer:
Question 27. Define isotonic solutions and give two examples
Answer:
Isotonic is a kind of solution that consists ofthe same salt concentration as the cells and the blood.
- Isotonic Solution Examples
- Some examples are as follows:
Alcohol Proof:
- We know that a shot of beer produces fewer effects on us. Whereas, a shot of whiskey causes less effect on us. This happens because the amount ofwhiskey in alcohol is more than that in the beer.
- Scientifically, whiskey has more ethanol. It is the molecule that is responsible for the effect of alcohol in comparison to beer.
Relieving Dehydration:
A saline solution contains water mixed with sodium chloride i.e. NaCl. Saline solutions are useful in the hospital. Useful for patients experiencing severe dehydration.
- However, it is also useful for patients suffering from the problem of fluid intake through traditional consumption.
- Moreover, unlike hypertonic and hypotonic saline solutions,
these solutions have the same concentration of NaCl as a cellular fluid in the body of a human being
Question 28. Define latent heat.
Answer:
Heat absorbed or released as the result of a phase change is called latent heat. There is no temperature change during a phase change, thus there is no change in the kinetic energy ofthe particles in the material.
Equation:
L = Q/M
Where,
- L = Specific latent heat of a substance
- Q = Amount of heat
- M = Mass of the substance
Question 29. Define buffers and buffer capacity
Or
Write a note buffer capacity
Answer:
Buffers:
It is an aqueous solution that consists of a mixture of a weak acid and its salt or a weak base with its salt. Its pH changes very little when a small amount of strong acid or base is added to it and is thus used to prevent a solution’s pH change.
Buffer capacity:
Buffer capacity can be defined as the ability of a solution to resist rapid changes in pH. The solution either absorbs or removes H+ and OH– ions. To elaborate, if we take acid or base and add it to a buffer system there will be a change in the pH.
- The change can be either large or small. This will mainly depend on two main factors such as the initial pH ofthe solution and the extent to which the buffer can resist the change in pH
- We look at the buffer it will resist change in pH after the addition of an acid or base, which leads to the consumption of the buffer.
- Till the buffer is completely absorbed, the pH will not change drastically. As the buffer is depleted it will become less resistant to change.
- In other words, we can also simply describe buffer capacity as a quantitative measure of resistance to pH change when H+ or OH– ions are added to the buffer solution.
- Buffer capacity is also known as acid neutralizing or alkalinity capacity.
- That being said, mathematically we can define buffer capacity (P) as the moles or gram equivalent of an acid or base that is required to bring a change in the pH of a solution by 1, divided by the pH change caused by the addition of base or acid. It can be represented as;
β = AB / ΔpH
Buffer capacity usually depends on the concentrations of the species in the buffered solution. If the buffer solution is highly concentrated it will have a greater buffer capacity. It is usually determined through a process known as titration.
Buffer capacity can be ‘used in the analysis of water samples to determine the water quality and maintain stable pH levels.
Question 30. What do you mean by polarization?
Answer:
It is defined as a phenomenon caused due to the wave nature of electromagnetic radiation. Sunlight travels through the vacuum to reach the Earth, which is an example of an electromagnetic wave. These waves are called electromagnetic waves because they form when an electric field interacts with a magnetic field. In this article, you will learn about two types of waves, transverse waves, and longitudinal waves.
Question 31. Define and classify complexes.
Answer:
A complex is a molecular entity formed by a loose association involving two or more component molecular entities (ionic or uncharged), or the corresponding chemical species. The bonding between the components is normally weaker than in a covalent bond
Complexes Types:
- Metal ion complexes
- Inorganic type
- Chelates
- Olefin type
- Aromatic type
Organic molecular complexes:
- Quinhydrone type
- Picric acid type
- Caffeine and other drug type
- Polymer type
Inclusion compounds:
- Channel lattice type
- Layer type
- Clathrates
- Monomoleculartype
- Macromolecular type
The ions or molecules (For example Cl–, NH3, HO2, Br–, I–, CN–, etc.) directly bound with the metal are called ligands. The interaction between the metal and the ligand represents a Lewis acid-base reaction in which the metal ion (Lewis acid) combines with a ligand (Lewis base) by accepting a pair of electrons from the ligand to form the coordinate covalent or electrostatic forces:
CO3+ + 6:NH3 < [Co NH3 6] 3+
Metal ion Complexes Inorganic Complexes:
- The number of ligands bound to the metal ion is defined as the coordination number. The coordination number of cobalt is six since it is complex with six NH3 groups. Coordination numbers usually determine the geometry ofthe complex
- A compound (For example NH3 ) that has a single pair ofelectrons for bonding with the metal ion, is called an unidentate ligand.
- Ligands with two or three groups are known as bidentate or tridentate respectively.
- Ethylenediaminetetraacetic acid (EDTA) has six points for attachment (two nitrogen and four oxygen donor groups) and is called hexadentate.
Metal ion Complexes Chelates:
Chelation is the formation of two or more coordinate bonds between a multidentate ligand (an organic compound called the chelating agent) and a single central atom. The bonds in the chelate may be ionic, primary covalent, or coordinate type.
Organic Molecular Complexes:
- Organic molecular complexes are formed as a result of noncovalent interactions between a ligand and a substrate.
- The interactions can occur through Vanderwaals forces, charge transfer, hydrogen bonding, or hydrophobic effects.
- Many organic complexes are so weak that they cannot be separated from their solutions as definite compounds, and they are often difficult to detect by chemical and physical means.
Organic Molecular Complexes:
Complexation differs from the formation of organic compounds in the forces between the constituents:
For example: Dimethylaniline and 2,4,6- trinitroanisole react in the cold to give a molecular complex. However, at elevated temperatures, they react to yield a salt, in which the molecules are held together by primary valence bonds.
Charge transfer complex:
- A charge transfer complex is an association of two or more molecules in which a fraction of electronic charge is transferred between the molecular entities.
- The molecules from which the charge is transferred are called the electron donor and the receiving species is called the electron acceptor
- Attraction in charge-transfer complexes is weaker than in covalent forces.
- Usually, these complexes are formed by sharing of w-electrons
For example:
- The complex between n benzene and trinitro benzene (1:1 type), (polar nitro group of trinitro benzene induces a dipole in the readily polarizable benzene molecules, resulting in electrostatic attraction).
- The difference between a donor-acceptor and a charge transfer complex is that in the latter type, resonance makes the main contribution to complexation, whereas in the former,
- London dispersion forces contribute more to the stability ofthe complex
Question 32. Define protein binding.
Answer:
Protein binding describes the ability of proteins to form bonds with other substances, and most commonly refers to the bonding of drugs to these molecules in blood plasma, red blood cells, other components ofthe blood, and to tissue membranes.
- It refers to other chemicals that enter the bloodstream. In the context of pharmacology, the amount of drug that is bound determines how effective it is in the body.
- The bound drug is kept in the bloodstream while the unbound component may be metabolized or excreted, making it the active part. So, if a drug is 95% bound to a binding protein and 5% is free, that means that 5% is active in the system and causing pharmacological effects
Question 33. Define protein binding.
Answer:
Protein binding describes the ability of proteins to form bonds with other substances, and most commonly refers to the bonding of drugs to these molecules in blood plasma, red blood cells, other components ofthe blood, and to tissue membranes. It refers to other chemicals that enter the bloodstream. In the context of pharmacology, the amount of drug that is bound determines how effective it is in the body.
The bound drug is kept in the bloodstream while the unbound component may be metabolized or excreted, making it the active part. So, if a drug is 95% bound to a binding protein and 5% is free, that means that 5% is active in the system and causing pharmacological effects
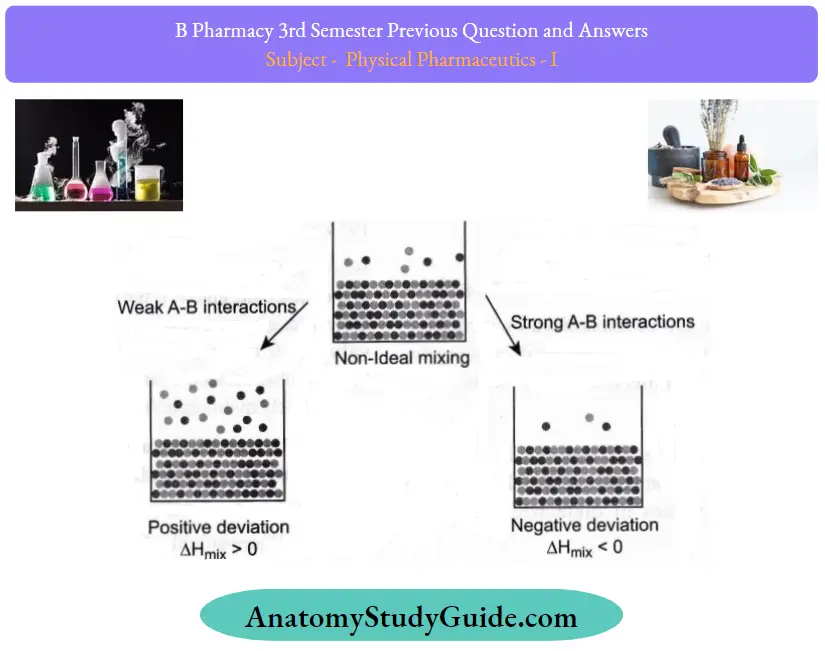
Question 34. Explain real solutions with examples.
Answer:
Real solutions are called non-ideal solutions:
- The solutions that do not follow Raoult’s Law at every concentration range and at all temperatures and deviate from it are called Non-Ideal Solutions.
- The resulting solution obtained by mixing two liquids, A (solvent) and B (solute), will experience several intermolecular forces of attraction as summarised below:
- A-A intermolecular forces of attraction (solvent solvent)
- B-B intermolecular forces of attraction (solute solute)
- A-B intermolecular forces of attraction (solvent-solute)
- In a non-ideal solution, the intermolecular forces of attraction between A – A, B – B, and A are not equal,
i.e., A – A ≠ B – B≠ A – B.
Non-ideal solutions depict characteristics as follows:
- Non-ideal solutions do not abide by Raoult’s Law. The partial pressure of components A and B in a non¬ideal solution will be PAAXAPA0 and PB*XBPB° where PA° and PB° are respective vapor pressures in pure form. XA and XB are respective mole fractions of components A and B
- The solute-solute (B-B) and solvent-solvent (A-A) interactions are different from that of the solute-solvent (A-B) interaction.
- The enthalpy of mixing is AmixFML This means that heat might have been released if the enthalpy of mixing is negative(AmixH<0), or the heat might have been absorbed if the enthalpy of mixing is positive. (AmixH>0).
- The volume of mixing is AmixWO. This depicts that there will be some expansion(?mixV>0) or contraction (ΔmixV < 0) on the dissolution of liquids. nonideal solution can occur in two ways
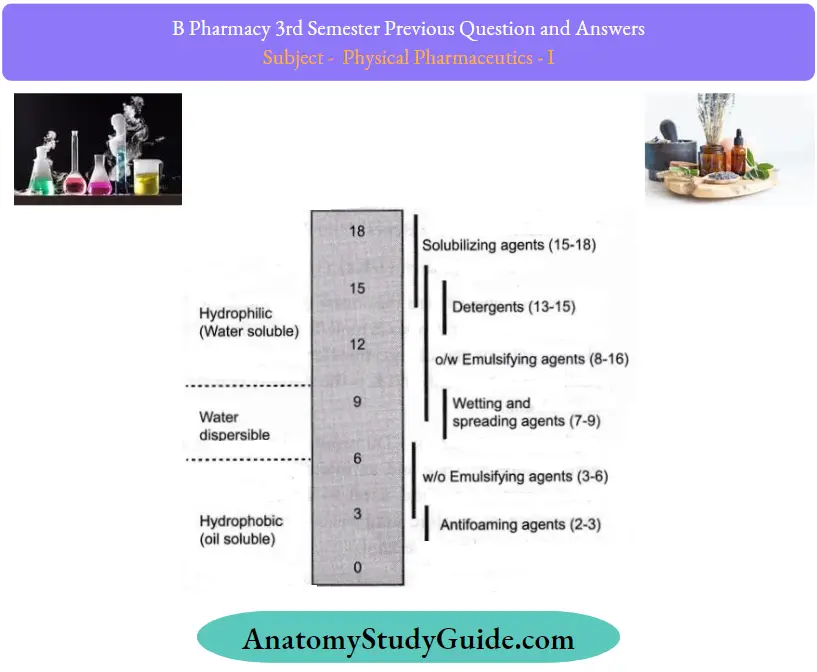
Question 35. Describe Griffin’s HLB scale.
Answer:
The hydrophilic-lipophilic balance of a surfactant is a measure of the degree to which it is hydrophilic or lipophilic, determined by calculating values for the different regions ofthe molecule
Griffin’s method for non-ionic surfactants as described in 1954 works as follows:
- Where is the molecular mass of the hydrophilic portion of the molecule, and M is the molecular mass ofthe whole molecule, giving a result on a scale of 0 to 20.
- An HLB value of 0 corresponds to a completely lipophilic/ hydrophobic molecule, and a value of 20 corresponds to a completely hydrophilic/lipophobic molecule.
The HLB value can be used to predict the surfactant properties of a molecule:
- < 10: Lipid-soluble (water-insoluble)
- > 10: Water-soluble (lipid-insoluble)
- 1 to 3: Anti-foaming agent
- 3 to 6: W/O (water in oil) emulsifier
- 7 to 9: Wetting and spreading agent
- 13 to 16: detergent
- 8 to 16: O/W (oil in water) emulsifier
- 16 to 18: Solubiliser or hydrotrope
Question 36. Write a note on complexation.
Answer:
Complexes are compounds that result from donor-acceptor mechanisms between two or more chemical species.
Complexes can be divided broadly into three classes depending on the type of the acceptor substance:
- Metal ion complexes
- Organic molecular complexes
- Inclusion complexes
Intermolecular forces involved in the formation of complexes:
- Vander Waals forces.
- Hydrogen bonds (important in molecular complexes).
- Coordinate covalence (important in metal complexes).
- Charge transfer.
- Hydrophobic interaction
Question 37. Describe in detail the electrometric determination of pH.
Answer:
The electrometric method is the most accurate method for the determination of the pH of a solution. This method measures the electromotive force setup due to the existence of H+ and OH in the solution. The instrument that measures pH in this manner is known as a pH meter.
Electrometric Method of pH Determination:
The switch of the instrument is put on and after a few minutes when the instrument is warm enough, the pointer is adjusted to 0 mV or 7.0 pH position by set zero control. A standard buffer solution is taken in a clean glass or polythene beaker.
- Electrodes are lowered so that they are immersed in the solution to a depth of about one inch.
- The temperature of the buffer solution is measured by a thermometer and the instrument is again adjusted by temperature compensation control to this value. The pointer is again set at 7.0 pH using set control.
- The Selector switch is now turned to the proper range, i.e. 0- 7 or 7-14.
- The pointer will now move to show the pH of the buffer and with the help of set buffer control, the pointer is set to the exact pH value of the buffer.
Now the pH meter is standardized and is ready for determination of pH of any given solution.
- To measure the pH of a given solution, the selector is brought back to Zero position and the electrodes are cleaned with distilled water.
- The given solution is taken in a clean beaker and the electrodes are immersed in it.
- The selector switch is now put on the expected pH range and the reading ofthe pointer on the pH scale is noted. This value is the pH ofthe given solution.
- Principle of Determination of pH When equal volumes ofthe unknown solution and a series of standard buffer solutions with closely varying pH values are treated with a suitable indicator, the colour of the unknown solution will be found to match with one of the standard buffer solutions.
- The pH of the unknown is, therefore, the same as the pH of the buffer with which its color matches.
A permanent standard made from glass discs of the appropriate color and shade for each indicator may also be used in place of the standard buffer and indicator.
Hellige’s comparator with its permanent standard discs made of coloured glass is a very convenient instrument for this purpose.
- pH Determination of a Colourless Solution First determine the approximate pH of the solution with the help of a universal indicator and color chart.
- Take a small piece of universal indicator pamper and just dip its one end in the given solution.
- After about 30 seconds compare the color of the paper with the standard color chart provided and know the approximate pH.
- If a universal indicator solution is to be used in place of paper, put a drop of it in about 2 ml ofthe solution and compare the color ofthe solution with that of the color chart.
After having known the approximate pH ofthe solution. choose from the set of indicators an indicator that more closely covers the pH range of the solution under examination. Then add a drop of this indicator to approximately 2 ml ofthe solution, and match the shade of the color produced against the series of colored standard buffers.
- The standard color solutions are prepared by adding one drop of the same indicator to 2 ml of each of the buffer solutions (chosen in the pH range of the unknown).
- The pH ofthe unknown solution is the pH ofthe buffer with which the color matches.
- In case, the color of the unknown is intermediate between two colors produced by two consecutive buffers, the pH of the unknown lies between the two known standards. The accuracy of this method can be increased by employing a set of buffer solutions with closely differing pH values.
pH Determination of Coloured Solution:
Place a test tube containing plain urine of colored solution in front of the colored buffer standards. View the two test tubes together and then match this against the urine or colored solution containing the same indicator in front of which a test tube containing water is to be placed. The use of a comparator makes the matching easier, otherwise, a double-rowed test tube rack is also convenient
Question 38. Write the solubility expressions for the solubility of solids in liquids.
Answer:
- Percent by mass,
- Percent by volume,
- Molality (m), molarity (M),
- Mole fraction (x),
- And parts per million (ppm)
Question 39. What do you mean by glassy states?
Answer:
It is a state in which “Glass is a non-equilibrium, non¬ crystalline state of matter that appears solid on a short time scale but continuously relaxes towards the liquid state
Question 40. What do you mean by glassy states?
Answer:
It is a state in which “Glass is a non-equilibrium, non¬ crystalline state of matter that appears solid on a short time scale but continuously relaxes towards the liquid state
Question 41. What are real and ideal solutions?
Answer:
Ideal solution:
There are some solutions whose components follow Raoult’s law quite closely. An example of such a solution is one composed of hexane C6H)4 and heptane C?H16. The total vapor pressure of this solution varies in a straight-line manner with the mole fraction composition of the mixture. An ideal solution is one whose vapor pressure follows Raoult’s law throughout its range of compositions.
Real solution:
Real solutions are more like real societies, in which some members are “more equal than others.” If like molecules of each kind are more attracted to each other than to unlike ones, then the molecules that happen to be close to their own kind will be stabilized.
Question 42. Define the term isotonicity with examples.
Answer:
Isotonicity is used to describe the property of a solution in which the concentration of solute is equivalent to the solute concentration of another solution. Thus, isotonic solutions are those that have equal osmotic pressure and water potential as both solutions have an equal concentration of water molecules.
For Example : Blood serum is isotonic to the physiological salt solution. Solutions with the same tonicity do not allow water to pass through the cell membrane
Question 43. Write any two applications of buffered isotonic solutions in pharmacy.
Answer:
The preparation of such dosage forms as injections and ophthalmic solutions, which are placed directly into pH-sensitive body fluids;
- The manufacture of formulations in which the pH must be maintained at a relatively constant level to ensure maximum product stability;
- Pharmaceutical tests and assays requiring adjustment to or maintenance of a specific pH for analytic purposes
Question 44. What is Henderson Hassalbach’s equation? Give its applications in Pharmacy.
Answer:
Handerson-Hasselbalch Equation
Preparation of acid Buffer
Consider an acid buffer solution, containing a weak acid
(HA) and its salt (KA) with a strong base (KOH).
Weak acid HA ionizes, and the equilibrium can be written as
HA +H2O ⇆ H+ + A–
Acid dissociation constant = Ka = [H+] [A– ]/HA
Taking, the negative log of RHS and LHS
– log Ka = -log [H+] – log([A–]/[HA])
pKa = pH – log ([salt]/[acid])
pH of acid buffer = pKa + ([salt]/[acid]):
Preparation of Base Buffer
Consider a base buffer solution, containing a weak base and its salt (BA) with strong acid.
pOH, can be derived as above
- pOH of a basic buffer = pKb + log ([salt]/[acid])
- pH of a basic buffer = pKa- log ([salt]/[acid])
Henderson Hassalbach’s Applications:
- Calculating pH ofthe solution using pKa.
- Calculating the ionized/unionized concentration of chemicals.
- Calculating the pKa ofthe molecule using pH
Question 45. Mention the applications of optical rotation in pharmacy.
Answer:
- Optical rotation is a function of and it is used to determine kinetic reactions
- This is also used to plot optical rotatory dispersion curves for a various range of wavelengths this helps in analyzing molecular structure
- The optical rotation is measured on a layer of suitable thickness at the wavelength specified in the monograph.
- If the specific rotation of a sample is known its concentration in the solution can be estimated.
- If the concentration of the material in the sample is known then its specific rotation can be determined.
- The technique may be extended to the determination of optical substances in the presence of optically inactive species.
Question 46. Explain ideal solutions with examples.
Answer:
An ideal solution is a mixture in which the molecules of different species are distinguishable, however, unlike the ideal gas, the molecules in the ideal solution exert forces on one another. When those forces are the same for all molecules independent of species then a solution is said to be ideal
If we take the simplest definition of an ideal solution, then it is described as a homogeneous solution where the interaction between molecules of components (solute and solvents) is the same to the interactions between the molecules of each component itself. These types of solutions follow Raoult’s Law at almost all levels of concentration and temperatures.
Characteristics of Ideal Solution:
An ideal solution can be obtained by mixing a solute and a solvent which consist of similar molecular structure and size. If we take two substances X and Y and mix them we will see that several intermolecular forces exist between them
For example:
- X and X experience intermolecular forces of attraction.
- Y and Y experience intermolecular forces of attraction.
- X and Y experience intramolecular forces of attraction.
Here, when the intermolecular forces of attraction are the same or equal, it gives us an ideal solution. Understanding the concept of an ideal solution is important, especially when we are studying chemical thermodynamics and topics like colligative properties.
Raoult’s Law:
Raoult’s law states that “the relative lowering of the vapor pressure of a solvent by dissolving a non-volatile electrolyte is equal to the mole fraction ofthe solute”. Thus ΔP/ΔAo = No. of moles of solute/ Total moles of solute and solvent
Thus
ΔP / PA = P°A- PA / PoA = XB = n/n+N
ΔP/PA = P°A- /P°A = XB = n/n+N
ΔP / PA = P°A- /P°A = XB = n/n+N
Where n and N are the numbers of moles of solute and solvent respectively.
In a dilute solution
N>> n
Hence,
XB = n/N = w/m/W/M
Where w and W are the weights of solute and solvent and m and M are their molecular weights respectively.
Therefore
PoA- PA/PoA = P/PoA = wM/Wm
Properties of an Ideal Solution:
An ideal solution most ofthe time has physical properties that are closely related to the properties of the pure components. Some of its properties are;
- The enthalpy ofthe solution is zero. The enthalpy of the solution gets closer to zero it is more likely to show an ideal behavior A mix H = 0
- The volume of mixing is also zero A mix V = 0
Examples of Ideal Solutions:
Getting a well-balanced ideal solution can be rare but some solutions can sometimes exhibit ideal behavior.
Some examples include:
- Toluene and Benzene.
- Ethyl Iodide and Ethyl Bromide.
- Bromobenzene and Chlorobenzene.
- N-heptane and n-hexane.
Notably, when we talk about the concentrations of the solutions most of the dilute solutions also have or show characteristics of the ideal solution
Question 47. Define dissociation constant. Write a note on its application in pharmacy.
Answer:
It is the tendency of a substance Ax By to reversibly dissociate in a solution into smaller components and B is as shown below
Ax By ⇆ xA+yB
Dissociation constant (Kd) = [A]x [B]y / [Ax By]
Where
[A] = Molar concentrations of A
[B] = Is molar concentrations of B
[Ax By] = Is the molar concentrations of AxBy
The dissociation constant is defined as the tendency of a particular substance in solution is dissociate into ions. it is equal to the product of the respective ion concentrations divided by the concentration of the ionic molecule, the acid dissociation constant is a quantitative measure ofthe strength of acid in the solution. The relationship between pH, pKa, and the extent of ionization is described by the Henderson-Hasselbalch equation.
For acid: pH = pKa + log Cu / Ci
For base: pH = pKa + log Ci / Cu
Where:
Cu is the concentration of a unionized form of drug
Ci is the concentration of the ionized form of the drug
pKa is the dissociation constant of the drug
The larger the value of pKa, the smaller the extent of dissociation percent ionization is defined at a particular concentration as the amount of a weak acid that exists as ions, the percent ionization of weak acid is
% ionized = I /1 + U × 100
Where
It is a concentration of ionic form
U is the concentration of the undissociated form
Dissociation constant Applications:
- The acid-base dissociation constant of a drug is a key physicochemical parameter influencing many pharmaceutical characteristics.
- The knowledge ofthe pKa values is important for the quantitative evaluation of systems involving acid-base equilibria in solution
- The dissociation constant is also essential for working with buffers, the design of these solutions depends on a knowledge ofthe pKa values of their components.
- Another application of ka is with pH indicators. A pH indicator is a weak acid or a weak base that changes color in the transition pH range, ie about pKa ± 1.
- The dissociation constant is also essential for working with buffers, the design of these solutions depends on a knowledge ofthe pKa values of their components.
- Another application of Ka is with pH indicators. A pH indicator is a weak acid or a weak base that changes color in the transition pH range, ie about pKa ± 1.
Question 48. State Raoults law.
Answer:
The partial vapor pressure (Pj) of each component in a solution is directly related to the mole fraction (X.) of the component in the liquid and the vapor pressure of the pure component ( P.*)
Pi = Xi Pi*
Question 49. What are chelates?
Answer:
Chelation is the process of formation of two or more separate coordinate bonds between a polydentate ligand and a single central atom
For example: EDTA
Question 50. What are sandwich complexes?
Answer:
Bimetallic complexes are coordinated molecules that contain two metals coordinated to a Dinucleating ligand. There is a new trend in the recent past to use bimetallic complexes due to their promising bioactivities. In a bimetallic complex, the presence of a second metal center may show an increase in the antimicrobial properties of the complex as compared to its monometallic counterpart.
The two metal centers will each contribute according to their chemical properties. The main mode of action by a majority ofthe complexes is via DNA cleavage or damage. The review also highlights a few synthetic routes used by researchers to synthesize either homo or heterobimetallic complexes
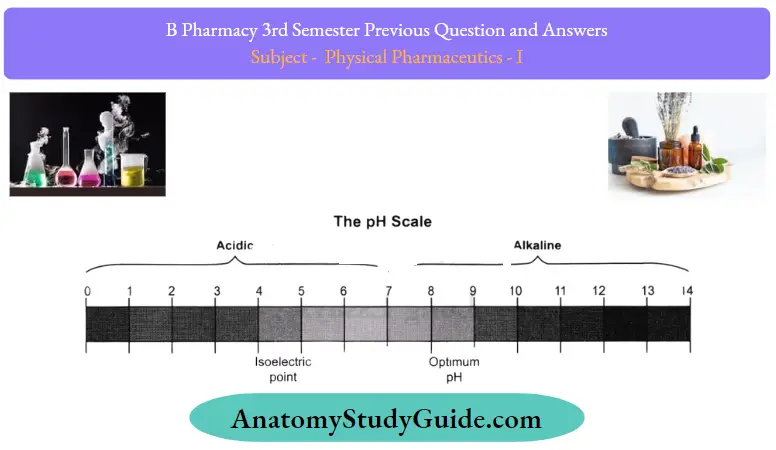
Question 51. What is the pH soreness scale?
Answer:
It is a system of grading pH levels and different concentrations of H30+ ions have devised and named after Sorenson. Having a pH value of 0 indicates a hydrogen ion concentration of 1 (or 100). pH level of this level is indicative of highly acidic solutions. This indicates strong alkaline properties. pH 14 means hydrogen ion concentration between 10-14 which means the solution has a higher pH.
As hydrogen ion is equal to hydroxyl ion the centre point of pH in scale is 7 meaning there is a concentration of 10-7 hydrogen ions. It represents neutrality as 7. Generally, a pH under 7 indicates an acidic area and a pH above 7 indicates an alkaline area. By measuring the levels of colloidal and non-aqueous solutions, the activity scale has little correlation
The pH Scale:
Question 52. What is buffer capacity?
Answer:
Buffer capacity can be defined as the ability of a solution to resist rapid changes in pH. The solution either absorbs or removes H+ and OH– ions. To elaborate, if we take acid or base and add it to a buffer system there will be a change in the pH. The change can be either large or small. This will mainly depend on two main factors such as the initial pH of the solution and the extent to which the buffer can resist the change in pH.
Mention the applications of buffers in pharmacy:
- Improves purity
- Increases the stability
- Enhanced solubility
- Optimizing biological activity
Question 53. What are channel-type complexes? Give examples.
Answer:
Channel-type complexes are in which channels are formed by crystallization of the host molecules, the guest component is usually limited to long, unbranched straight-chain compounds
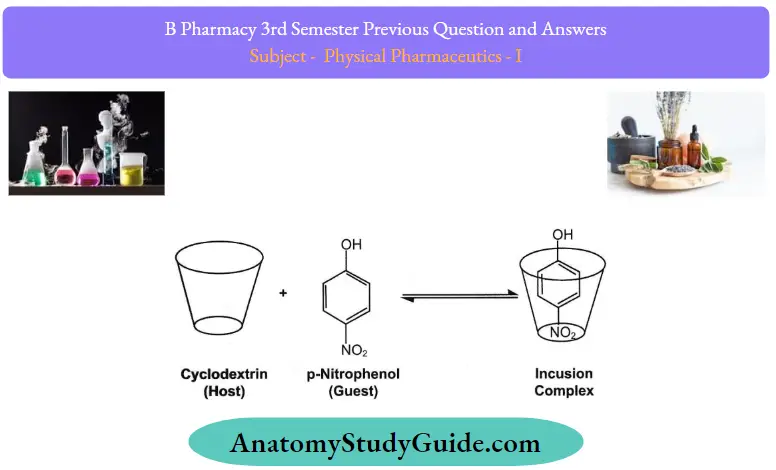
Question 54. Write the solubility expressions for the solubility of solids in liquids.
Answer:
Solubility of solids in liquids is expressed as mgC and mol L-1
Question 55. What are aerosols?
Answer:
An aerosol is a colloid of fine solid particles or liquid droplets, in the air or another gas. Aerosols can be natural or artificial and they can be found in the air over oceans, deserts, mountains, forests, ice, and every ecosystem in between. Examples of natural aerosols are fog and geyser steam.
Leave a Reply