Asepsis In Endodontics Notes
Dental professionals are exposed to a wide variety of microorganisms in the blood and saliva of patients, making infection control procedures important. A common goal of infection control is to eliminate or reduce the number of microbes being transferred from one person to another.
Table of Contents
Rationale For Infection Control
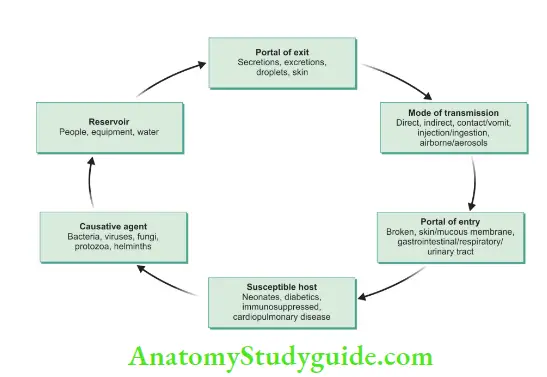
The deposition of organisms in the tissues and their growth resulting in a host reaction is called an infection. The number of organisms required to cause an infection is termed as the infective dose.
- Factors affecting infective dose are
- Virulence of the organism
- Susceptibility of the host
- Age, drug therapy, pre-existing disease, etc.
Read And Learn More: Endodontics Notes
Microorganisms can spread from one person to another via direct contact (by touching soft tissues or teeth of patients), indirect contact (injuries with contaminated sharp instruments, needlestick injuries, or contact with contaminated equipment and surfaces), and droplet infection (by large particle droplets spatter which is transmitted by close contact).
Cross Infection
Cross-infection is the transmission of infectious agents among patients and staff within a clinical environment.
Different Routes of Spread of Infection:
- Patient to dental health care worker
- Dental health care worker to the patient
- Patient-to-patient
- Dental office to the community
- Community to the patient (community to patient infection
occurs through the water supply of the dental unit. These microorganisms colonize inside the water lines, and thereby form a biofilm which is responsible for causing the infection)
Modes of Transmission:
- Direct contact through a break in the skin or direct contact with mucous membrane
- Indirect contact via sharp cutting instruments and needlestick injuries
- Droplet infection by spatter produced during dental procedures
Objective Of Infection Control
The main objective of infection control is the elimination or reduction in the spread of infection from all types of microorganisms.
Universal Precautions
It is always recommended to follow some basic infection control procedures for all patients, termed “universal precautions.”
These are as follows:
- Immunization: All members of the dental team (who are exposed to blood or blood-contaminated articles) should be vaccinated against hepatitis B
- Use of personal protective barrier techniques, that is, use of protective gown, face mask, protective eyewear, gloves, etc. These reduce the risk of exposure to infectious material and injury from sharp instruments
- Maintaining hand hygiene
Personal Protection Equipment:
- Barrier Technique:
The use of barrier technique is very important, which includes gown, face mask, protective eyewear, and gloves (Fig. 10.1). Protective clothing should be made of flid-resistant material and should not be worn out of the office for any reason. These should be washed in hot water (70–158°F) or cool water containing 50–150 ppm of chlorine.
- Protective gown:
The protective gown should be worn to prevent contamination of normal clothing and protect the skin of the clinician from exposure to blood and body substances.
-
- The gown can be reusable or disposable for use. It should have a high neck and long sleeves to protect the arms from splash and spatter
- Protective clothing must be removed before leaving the workplace
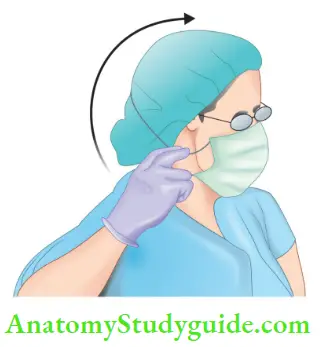
- Facemask:
A surgical mask that covers both the nose and mouth should be worn by the clinician during procedures. Though facemasks do not provide complete microbiological protection, they prevent the splatter from contaminating the face. Mask with 95% filtration efficiency for particles 3–5 µm in diameter should be worn.
-
- The outer surface of the mask can get contaminated with infectious droplets from spray or from touching the mask with contaminated fingers, so should not be reused
- The maximum time for wearing masks should not be more than one hour since it becomes dampened from respiration, resulting in its degradation
- To remove the mask, grasp it only by its strings not by the mask itself

- Head cap:
Hairs should be properly tied and covered with a head cap.
- Protective eyewear:
Eyewear protects the eyes from injury and from microbes such as hepatitis B virus, which can be transmitted through conjunctiva. Eyewear should be clear, antifog, distortion-free, close-fitting, and shielded.
- Face shield:
chin-length plastic face shield can be worn as an alternative to protective eyewear
- Gloves:
Gloves should be worn to prevent contamination of hands when touching mucous membranes, blood, and saliva, and to reduce the chances of transmission of infected microorganisms from clinician to patient. Gloves should be of good quality and well-fitted. Gloves should be disposed of after the activity for which they were used.
Some persons can show allergic reactions to gloves due to latex (polyisoprene) or antioxidants such as mercaptobenzothiazole. Ensure that latex-free equipment and nonlatex gloves (polyurethane or vinyl gloves) are used on patients who have a latex allergy. Overgloves or paper towels should be used for opening drawers, cabinets, etc.
Hand Hygiene:
Hand hygiene significantly reduces potential pathogens on the hands and is considered the single most critical measure for reducing the risk of transmitting organisms to patients and dentists. Flora present on the skin can be:
Transient flares, which colonize the superficial layers of the skin, are easier to remove by routine handwashing. These are acquired by direct contact with patients or contaminated environmental surfaces
Resident flora, attached to deeper layers of the skin, is more resistant to removal and less likely to be associated with such infections
The purpose of surgical hand antisepsis is to eliminate transient flora and reduce resident flra for the duration of a procedure to prevent the introduction of organisms in the operative wound if gloves become punctured or torn.
Hand cleansers containing mild antiseptics like 3% PCMX (parachlorometaxylenol), triclosan, or chlorhexidine control transient pathogens and suppress the overgrowth of skin bacteria.
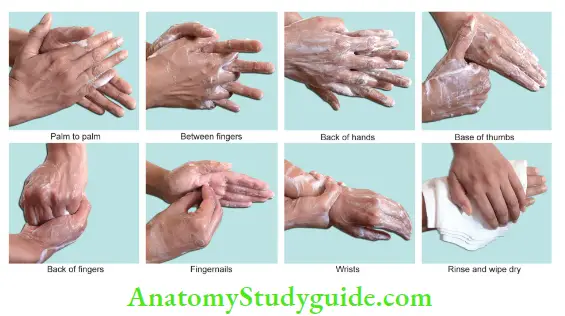
Hand washing instructions:
- Wet hands with warm water.
- Apply an adequate amount of soap to achieve lather.
- Rub vigorously for a minimum of 15 seconds, covering all surfaces of hands and fingers. Pay particular attention to fingertips, between fingers, backs of hands, and base of thumbs, which are the most commonly missed areas.
- Rinse well with running water.
- Dry thoroughly with a disposable paper towel.
Indications for hand hygiene
- At the beginning and after the completion of the case.
- Before putting on and after the removal of gloves.
- Before touching the eyes, nose, face, or mouth
- Before eating and drinking
- After barehanded contact with contaminated equipment or surfaces and before leaving treatment areas
- At the end of the day
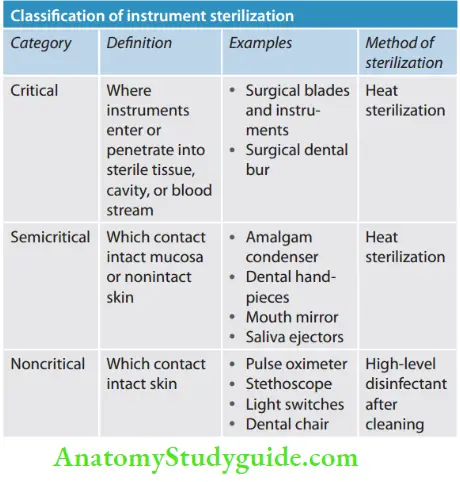
Classifiation Of Instruments
Centers for Disease Control and Prevention (CDC) classified the instrument into critical, semicritical, and noncritical depending on the potential risk of infection during the use of these instruments. These categories are also referred to as Spaulding classification (given by Spaulding in 1968).
Definitions:
- Cleaning:
It is the process that physically removes contamination but does not necessarily destroy microorganisms. It is a prerequisite before decontamination by disinfection or sterilization of instruments since organic material prevents contact with microbes and inactivates disinfectants.
- Disinfection:
It is the process of using an agent that destroys germs or other harmful microbes or inactivates them, usually referred to as chemicals that kill the growing forms (vegetative forms) but not the resistant spores of bacteria.
- Antisepsis:
It is the destruction of pathogenic microorganisms existing in their vegetative state on living tissue.
- Sterilization:
Sterilization involves any process, physical, or chemical that will destroy all forms of life, including bacteria, fungi, spores, and viruses.
- Aseptic technique:
It is the method that prevents contamination of wounds and other sites by ensuring that only sterile objects and fluids come into contact with them and that the risks of airborne contamination are minimized.
- Antiseptic:
It is a chemical applied to living tissues, such as skin or mucous membranes to reduce the number of microorganisms present, by inhibiting their activity or by destruction.
- Disinfectant:
It is a chemical substance that causes disinfection. It is used on nonvital objects to kill surface vegetative pathogenic organisms, but not necessarily spore forms or viruses.
Instrument Processing Procedures Or Decontamination Cycle
Instrument processing is the collection of procedures which prepare the contaminated instruments for reuse. For a complete sterilization process, instruments should be processed correctly and carefully.
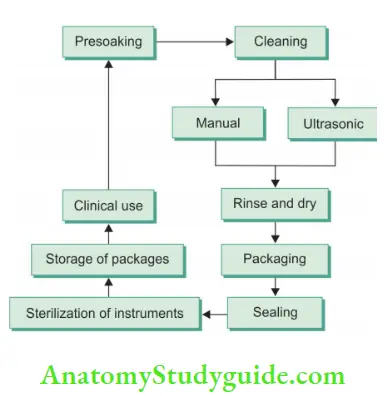
Steps of instrument processing:
- Presoaking (holding)
- Cleaning
- Corrosion control
- Packaging
- Sterilization
- Monitoring of sterilization
- Handling the processed instrument
Presoaking (Holding):
It facilitates the cleaning process by preventing debris from
drying.
- Procedure:
Wear puncture-resistant heavy utility gloves and personnel protective equipment. Place loose instruments in a perforated cleaning basket and then place the basket into the holding solution. A perforated cleaning basket reduces the direct handling of instruments.
So, the chances of contamination are decreased. Holding solutions for instruments can be neutral pH detergents, water, etc.
- Cleaning:
Cleaning reduces the bioburden, that is, microorganisms, blood, saliva, and other materials. Methods used for cleaning:
- Manual scrubbing:
It is one of the most effective methods for removing debris if performed properly. Brush delicately all the surfaces of instruments while submerged in cleaning solution using a long-handled stiff nylon brush to keep the scrubbing hand away from sharp instrument surfaces. Use neutral pH detergents while cleaning.
- Ultrasonic cleaning:
It is an excellent cleaning method as it reduces the direct handling of instruments
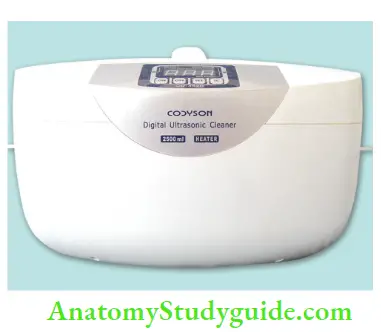
- Mechanism of action:
Ultrasonic energy generated in the ultrasonic cleaner produces billions of tiny bubbles which, further collapse and create high turbulence at the surface of the instrument. This turbulence dislodges the debris.
- Usually, the time ranges vary from 4 to 16 min but the time for cleaning varies due to
- Nature of instrument
- Amount of debris
- The efficiency of the ultrasonic unit
- Mechanical–instrument washer:
These are designed to clean instruments in a hospital setup. An instrument washer also has the advantage that it reduces the direct handling of the instrument.
Control of Corrosion by Lubrication:
For rust-prone instruments, use dry hot air oven/chemical vapor sterilization instead of the autoclave. Use rust inhibitor(sodium nitrite) spray on the instruments.
Packaging:
It maintains the sterility of instruments after sterilization. Packaging materials can be self-sealing, paper–plastic, and peel-pouches. Instruments are kept wrapped until ready for use and sterilized packs should be allowed to cool before storage; otherwise, condensation will occur inside the packs. Sterile packs should be stored in an ultraviolet (UV) chamber or drums which can be locked.
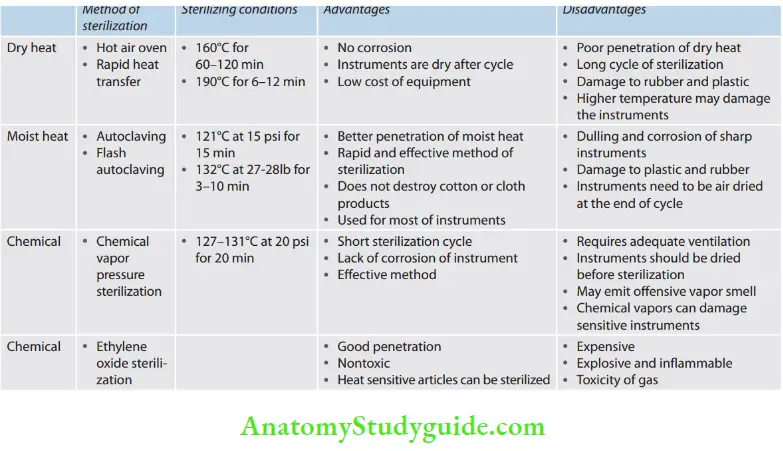
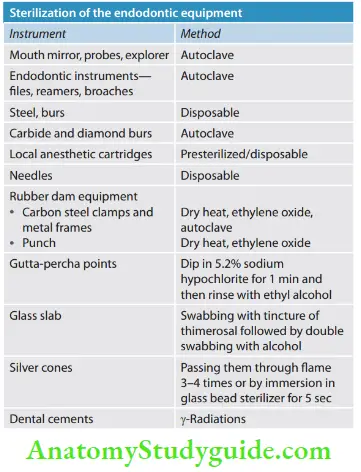
Methods of Sterilization:
Sterilization is the process by which an object, surface, or medium is freed of all microorganisms either in the vegetative or spore state.

Though there are many ways of sterilization, the following four methods are accepted methods in dental practice:
- Moist/steam heat sterilization
- Dry heat sterilization
- Chemical vapor pressure sterilization
- Ethylene oxide (ETOX) sterilization
Moist or Steam Heat Sterilization:
- Autoclave:
- Autoclave provides the most efficient and reliable method of sterilization for all dental instruments
- It involves heating water to generate steam in a closed chamber resulting in moist heat that rapidly kills microorganisms
- Saturated steam under pressure is the most efficient, quickest, safest, and most effective method of sterilization because
- It has high penetrating power
- It gives up a large amount of heat (latent heat) to the surface with which it comes into contact and on which it condenses as water
- Packaging of Instruments for Autoclaving:
-
- For wrapping, closed containers such as closed metal trays, glass vials, and aluminum foils should not be used since they stop the steam from reaching the inner part of the packs
- For packaging of autoclaving instruments, one should use a porous covering to permit steam to penetrate through and reach the instruments
- Materials used for packaging can be fabric or sealed paper or cloth pouches and paper-wrapped cassettes
- If instruments are to be stored and not used shortly after sterilization, the autoclave cycle should end with a drying phase to avoid tarnish or corrosion of the instruments
- Pressure, temperature, and time
-
- The higher the temperature and pressure, the shorter is the time required for sterilization
- At 15 psi pressure and temperature of 121°C, the time required is 15 min
- At 126°C, the time required is 10 min
- At 132°C, the time required is 3 min at 27–28 lbs—Flash sterilization
Advantages of autoclaving
- Time efficient
- Good penetration
- The results are consistently good and reliable
- The instruments can be wrapped prior to sterilization
Disadvantages of autoclaving
- Blunting and corrosion of sharp instruments
- Damage to rubber goods
Dry Heat Sterilization:
Dry heat utilizes the hot air which has very little or no water
vapors in it to sterilize the instruments.
- Conventional Hot Air Oven:
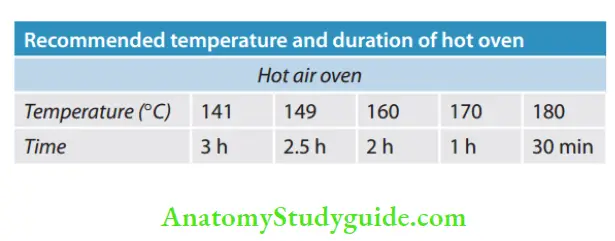
Hot air oven utilizes radiating dry heat for sterilization as this type of energy does not penetrate materials easily. So, long periods of exposure to high temperatures are usually required. In a conventional type of hot air oven, air circulates by gravity flow, thus it is also known as Gravity convection.
- Packaging of Instruments for Dry Heat:
Dry heat ovens usually achieve temperatures above 320°F (160°C). The packaging or wrapping material used should be able to withstand high temperatures; otherwise, it may get charred. Acceptable materials for packaging are paper and plastic bags, wrapped cassettes, and aluminum foil.
Unacceptable materials are plastic and paper bags which are not able to withstand dry heat temperature. Packs of instruments should be placed ≥1 cm apart for air to circulate in the chamber.
- Mechanism of Action:
- Dry heat kills microorganisms by protein denaturation, coagulation, and oxidation
- Instruments that can be sterilized in a dry hot oven are glassware such as pipettes, flasks, scissors, glass syringes, carbon steel instruments, and burs. Dry heat does not corrode sharp instrument surfaces. Also, it does not erode glassware surfaces
- Before placing in the oven, the glassware must be dried. The oven must be allowed to cool slowly for about 2 h as the glassware may crack due to sudden or uneven cooling.
- Rapid heat transfer (forced air type):
In this type of sterilizer, a fan or blower circulates the heated air throughout the chamber at a high velocity which, in turn, permits a more rapid transfer of heat energy from the air to instruments, thereby reducing the time.
- Temperature/cycle recommended:
- 370–375°F—12 min for wrapped instruments
- 370–375°F—16 min for unwrapped instruments
Advantages of dry heat sterilization:
- No corrosion is seen in carbon steel instruments and burs
- Maintains the sharpness of cutting instruments
- Effective and safe for sterilization of metal instruments and mirrors
- Low cost of equipment
- Instruments are dry after the cycle
- Industrial forced draft types usually provide a larger capacity at a reasonable price
- Rapid cycles are possible at higher temperatures
Disadvantages of dry heat sterilization:
- Poor penetrating capacity of dry heat
- A long cycle is required because of poor heat conduction and poor penetrating capacity
- High temperatures may damage heat-sensitive items such as rubber or plastic goods
- Instruments must be thoroughly dried before placing them for sterilization
- Inaccurate calibration and lack of attention to proper settings often lead to errors in sterilization
- Generally not suitable for handpieces
- Cannot sterilize liquids
- May discolor and char fabric
Chemical Vapor Sterilization/Chemiclave:
Sterilization by chemical vapor under pressure is known as chemical vapor sterilization. Here, formaldehyde and alcohol formulation is heated in a closed chamber, producing hot vapors that kill microorganisms. The temperature, pressure, and time required for the completion of one cycle is 270°F (132°C) at 20 lb for 30 min.
Chemical solution contents:
- Active ingredient—0.23% formaldehyde
- Other ingredient—72.38% ethanol + acetone + water and other alcohols
Mechanism of Action:
- Coagulation of protein
- Cell membrane disruption
- Removal of free sulfhydryl groups
- Substrate competition
Mechanism of Action Advantage:
- Eliminates corrosion of carbon steel instruments, burs, and pliers.
Mechanism of Action Disadvantages:
- The instruments or items which are sensitive to elevated temperature are damaged
- Sterilization of linen, textiles, fabric, or paper towels is not recommended
- Dry instruments should be loaded in the chamber
Ethylene Oxide (ETOX) Sterilization:
This sterilization method is best used for sterilizing complex
instruments and delicate materials. ETOX is a highly penetrative, noncorrosive gas above 10.8°C with a civil action against bacteria, spores, and viruses.
Mechanism of Action:
It destroys microorganisms by alkylation and causes the denaturation of nucleic acids of microorganisms. The duration that the gas should be in contact with the material to be sterilized depends on temperature, humidity, pressure, and the amount of material.
Ethylene Oxide (ETOX) Sterilization Advantages:
- It leaves no residue
- It is a deodorizer
- Good penetration power
- It can be used at a low temperature
- Suited for heat-sensitive articles, for example, plastic, rubber
Ethylene Oxide (ETOX) Sterilization Disadvantages:
- High cost of the equipment
- Toxicity of the gas
- Explosive and inflammable
Irradiation:
Ionizing Radiation (X-rays, γ-rays, and High-speed Electrons):
Ionizing radiations are effective for heat-labile items. They are commonly used by the industry to sterilize disposable materials such as needles, syringes, culture plates, suture material, cannulas, and pharmaceuticals sensitive to heat. High-energy γ-rays from cobalt-60 are used to sterilize such articles.
Nonionizing Radiation (Ultraviolet Light and Infrared Light):
- Ultraviolet rays:
- UV rays are absorbed by proteins and nucleic acids and kill microorganisms by the chemical reactions
- Their main application is air purification in operating rooms to reduce the bacteria in air, water, and on the contaminated surfaces
- Infrared:
- It is used for sterilizing a large number of syringes sealed in metal containers in a short period of time It is used to purify air in the operating room
Glass Bead Sterilizer:
It is a rapid method of sterilization which is used for sterilization of instruments. It uses table salt which consists approximately of 1% sodium silicoaluminate, sodium carbonate, or magnesium carbonate. So it can be poured more readily and does not fuse under heat.
Salt can be replaced by glass beads provided the beads are smaller than 1 mm in diameter because larger beads are not efficient in transferring the heat to endodontic instruments due to the presence of large air spaces between the beads.
Instruments can be sterilized in 5–15 seconds at a temperature of 437–465°F (260°C) even when inoculated with spores.
Glass Bead Sterilizer Advantages:
- Commonly used salt is table salt which is easily available and cheap
- Salt does not clog the root canal. If it is carried into the canal, it can be readily removed by irrigation
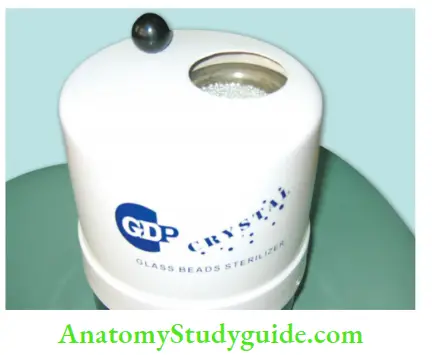
Glass Bead Sterilizer Disadvantage:
The handle portion is not sterilized, therefore instruments are not entirely “sterile”.
Monitoring of Sterilization:
Monitoring of instruments must be conducted through a combination of mechanical, chemical, and biological means, which evaluate both the sterilizing conditions and the procedure’s effectiveness.
- Mechanical indicators are the gauges on the sterilizer for cycle time, temperature, and pressure. Mechanical indicators must be checked and recorded for each load.
- Chemical indicators use sensitive chemicals to assess physical conditions during the sterilization process. For example, when a heat-sensitive tape is applied to the outside of a package, a rapid change in color indicates that the package has undergone a sterilization cycle, although it does not guarantee that sterilization has been achieved.
- Biological indicators are the most accepted means for monitoring of sterilization because they assess the effctiveness of sterilization in killing the most resistant micro-organisms. In these, an inactivated BI signifies that other potential pathogens in the load have been killed.
Storage of Processed Instruments :
After sterilization, instruments should be stored in an enclosed space like closed or covered cabinets. The date should be mentioned on storage packets because dating helps in assessing the shelf life of sterilization.
Packages containing sterile instruments should be inspected before use to verify barrier integrity and dryness. If packaging is compromised, the instruments should be cleaned, packaged, and sterilized again.
Disinfection
It is the term used for the destruction of all pathogenic organisms, such as vegetative forms of bacteria, mycobacteria, fungi, and viruses, but not bacterial endospores.
Methods of Disinfection:
- Disinfection by Cleaning: Cleaning with a detergent and clean hot water removes almost all pathogens including bacterial spores.
- Disinfection by Heat: Heat is a simple and reliable disinfectant for almost anything except living tissues. Mechanical cleaning with hot water provides an excellent quality of disinfection for a wide variety of purposes.
- Low-Temperature Steam: Most vegetative microorganisms and viruses are killed when exposed to steam at a temperature of 73°C for 20 min below atmospheric pressure. This makes it a useful procedure to leave spoiled instruments safe to handle prior to sterilization.
- Disinfection by Chemical Agents: They are used to disinfect the skin of a patient prior to surgery and to disinfect the hands of the operator.
Disadvantages of using chemicals:
- No chemical solution sterilizes the instruments immersed in it
- There is a risk of producing tissue damage if the residual solution is carried into the wound
Levels of Disinfectant :
- Alcohols—Low-Level Disinfectant
- Ethanol and isopropyl alcohols are commonly used as antiseptics
- Possess some antibacterial activity, but they are not effective against spores and viruses
- Act by denaturing proteins
- To have maximum effctiveness, alcohol must have a 10-min contact with the organisms
- Instruments made of carbon steel should not be soaked in alcoholic solutions, as they are corrosive to carbon steel
- Rubber instruments absorb alcohol, thus their prolonged soaking can cause a reaction when material comes in contact with living tissue
- Phenolic Compounds—Intermediate Level, Broad-Spectrum Disinfectant
- The phenolic compounds were developed to reduce their side effects but are still toxic to living tissues
- At high concentrations, these compounds are protoplasmic poison and act by precipitating the proteins and destroying the cell wall
- These compounds are used for disinfection of inanimate objects such as walls, floors, and furniture
- They may cause damage to some plastics and they do not corrode certain metals such as brass, aluminum, and carbon steel
- Aldehyde Compounds—High-Level Disinfectant:
- Formaldehyde:
- Broad-spectrum antimicrobial agent
- Flammable and irritant to the eye, skin, and respiratory tract
- Has limited sporicidal activity
- Used for large heat-sensitive equipment such as ventilators
and suction pumps excluding rubber and some plastics - Not preferred due to its pungent odor and because 18–30 h of contact is necessary for cidal action
- Glutaraldehyde
- Toxic, irritant, and allergenic
- A high-level disinfectant
- Active against most vegetative bacteria, fungi, and bacterial spores
- Frequently used for heat-sensitive material
- A solution of 2% glutaraldehyde (Cidex) requires immersion of 20 min for disinfection and 6–10 h of immersion for sterilization
- Safely used on metal instruments, rubber, plastics, and
porcelain - Activated by addition of sodium bicarbonate, but in its activated form, it remains potent only for 14 days
- Formaldehyde:
Antiseptics
Antiseptic is a chemical disinfectant that can be diluted sufficiently to be safe for application to living tissues like intact skin, mucous membranes, and wounds.
- Alcohols:
- Two types of alcohols are used—ethyl alcohol and isopropyl alcohol
- Used for skin antisepsis
- Their benefit is derived primarily in their cleansing action
- The alcohols must have prolonged contact with the organisms to have an antibacterial effect
- Ethyl alcohol is used in a concentration of 70% as a Skin antiseptic
- Isopropyl alcohol is used in a concentration of 60%–70% for disinfection of skin
- Aqueous Quaternary Ammonium Compounds:
- Benzalkonium chloride (Zephiran) is the most commonly used antiseptic
- It is well tolerated by living tissues
- Iodophor Compounds:
- Used for surgical scrub, soaps, and surface antisepsis
- Usually effective within 5–10 min
- Discolor surfaces and clothes
- Iodine is complexed with organic surface-active agents such as polyvinylpyrrolidone (Betadine, Isodine). Their activity is dependent on the release of iodine from the complex
- Concentrated solutions have less free iodine. Iodine is released as the solution is diluted
- These compounds are effctive against most bacteria, spores, viruses, and fungi
- Chloride Compounds:
- Commonly used are sodium hypochlorite and chlorine dioxide
- Sodium hypochlorite has rapid action
- A solution of one part of 5% sodium hypochlorite with nine parts of water is used
- Chlorous acid and chlorine dioxide provides disinfection in 3 min
- Diguanides:
- Chlorhexidine is active against many bacteria
- Gets inactivated in the presence of soap, pus, plastics, etc.
- Mainly used for cleaning the skin and mucous membrane
- As a 0.2% aqueous solution or 1% gel, it can be used for the suppression of plaque and postoperative infection
Infection Control Checklist
- Infection Control During the Pretreatment Period:
- Use disposable items whenever possible
- Ensure before treatment that all equipment have been sterilized properly
- Remove avoidable items from the operatory area to facilitate a thorough cleaning following each patient
- Identify those items that will become contaminated during treatment, for example, light handles, X-ray unit heads, and tray tables. Disinfect them when the procedure is complete
- Review patient records before initiating treatment
- Preplan the materials needed during treatment to avoid opening of the cabinets and drawers once the work is started
- Use separate sterilized bur blocks for each procedure to eliminate the contamination of other, unneeded burs
- Always keep the rubber dam kit ready in the tray
- Follow the manufacturer’s directions for care of dental unit water lines (DUWL)
- The clinician should be prepared before initiating the procedure; this includes the use of personal protective equipment (gown, eyewear, masks, and gloves) and hand hygiene
- Update patient’s medical history
- Chairside Infection Control:
- Treat all patients as potentially infectious
- Take special precautions while handling syringes and needles
- Use a rubber dam whenever possible
- Use high-volume aspiration
- Ensure good ventilation of the operatory area
- Be careful while receiving, handling, or passing sharp instruments
- Do not touch unprotected switches, handles, and other equipment once gloves have been contaminated
- Avoid touching drawers or cabinets, once gloves have been contaminated. When it becomes necessary to do so, ask your assistant to do this or use another barrier, such as over glove to grasp the handle or remove the contaminated gloves and wash hands before touching the drawer and then resolve for patient treatment
Infection Control during the Post-treatment Period:
- Remove the contaminated gloves used during treatment, wash your hands and put on a pair of utility gloves before beginning the cleanup
- Continue to wear protective eyewear, mask, and gown during cleanup
- Dispose of blood and suctioned flids which have been collected in the collection bottles during treatment
- After disposing of blood and suctioned flids, use 0.5% chlorine solution to disinfect the dental unit collection bottle. Keep the solution in the bottle for ≥10 min
- Clean the operatory area and disinfect all the items not protected by barriers
- Remove the tray with all instruments to the sterilization area separate from the operatory area
- Never pick up instruments in bulk because this increases the risk of cuts or punctures. Clean the instruments manually or in an ultrasonic cleaner
- Sterilize the handpieces whenever possible. In general, the handpiece should be autoclaved, but the handpiece that cannot be heat sterilized should be disinfected by the use of chemicals. Clean the handpiece with a detergent and water to remove any debris and sterilize it
- Waste that is contaminated with blood or saliva should be placed in sturdy leak proof bags
- Handle sharps items carefully
- Remove personal protective equipment after cleanup. Utility gloves should be washed with soap before removal
- At the end, thoroughly wash your hands
Waste Disposal
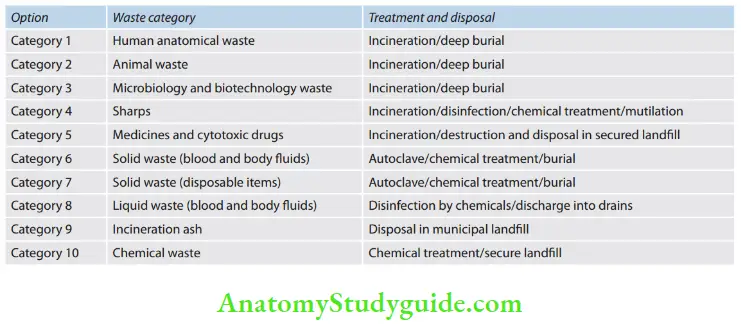
Biomedical waste: Waste generated during the diagnosis, treatment, or immunization of human beings or animals or research activities pertaining to, or production or testing of biological or in health camps.
Types of waste:
- Human Anatomical Waste:
Human tissues, organs, body parts, etc. Animal Anatomical Waste
Experimental animal carcasses, body parts, organs, and tissues, including the waste generated from animals used in experiments.
- Soiled Waste:
Items contaminated with blood, body fluids like dressings, plaster casts, cotton swabs, and bags containing residual or discarded blood and blood components.
- Expired or Discarded Medicines:
Pharmaceutical waste like antibiotics and cytotoxic drugs including all items contaminated with cytotoxic drugs along with glass or plastic ampoules, vials, etc.
- Chemical Waste:
Chemicals used in the production of biological and used or discarded disinfectants.
- Chemical Liquid Waste:
Liquid waste is generated due to the use of chemicals in the production of biological and used or discarded disinfectants.
- Microbiology, Biotechnology and Other Clinical Laboratory Waste:
It include blood bags, cultures, specimens of microorganisms, vaccines, and human and animal cell cultures used in research and industrial sectors.
Leave a Reply