Biofilm In Endodontics Notes
Biofilm Introduction
The persistence of microorganisms has been shown to be the most important cause for endodontic failure. Root canal infections are biofilm mediated. The complexity and variability of the root canal system, along with the multi-species nature of biofilms, make disinfection of this system extremely challenging.
Table of Contents
Due to increased concern about biofilms, endodontic research has focused on the characterization of root canal biofilms and the clinical methods to interrupt the biofilms along with killing microorganisms.
Read And Learn More: Endodontics Notes
Biofilm In Endodontics
Endodontic infections are biofilm mediated. The complexity of the root canal system, along with the multi-species nature of biofilms, makes the disinfection of this system extremely challenging. Microbial persistence is the most important factor for root canal failure.
Biofilm removal can be done by a chemo-mechanical process, using specific instruments and disinfecting chemicals in the form of irritants and/or intracanal medicaments. Endodontic research has focused on the characterization of root canal biofilms and the clinical methods to disturb the biofilms along with killing microbes.
The smear layer is a surface accumulation of debris formed on the dentine during root canal instrumentation. It consists of organic and inorganic components and forms both a superficially and deeper layers. It can prevent the penetration of intracanal medicaments into dentinal tubules and influence the adaptation of filing materials to canal walls. Whether it should be removed or not is still a debatable issue.
Biofim Definition
Biofim can be defined as a sessile multicellular microbial community characterized by cells that are firmly attached to a surface and enmeshed in a self-produced matrix of extracellular polymeric substances.
Bacterial biofilms are very prevalent in the apical root canals of teeth with primary and post-treatment apical periodontitis. These bacterial endodontic communities are often found adhered to or at least associated with the dentinal canal walls.
Characteristics Of Biofilm
Microorganisms present in a biofilm must have the following four basic criteria to survive:
- Autopoiesis: Ability to self-organize.
- Homeostasis: Resist environmental perturbations.
- Synergy: Effective in association than in isolation.
- Communality: Respond to environmental changes as a unit rather than as single individuals.
- Quorum sensing:
Cell-to-cell signaling is known as quorum sensing. In this, bacterial cells communicate with each other by releasing, sensing, and responding to small diffusible signal molecules. The ability of bacteria to communicate and behave as a group for interactions like a multicellular organism shows many benefits to bacteria in host colonization, formation of biofilms, defense against competitors, and adaptation to changing environments.
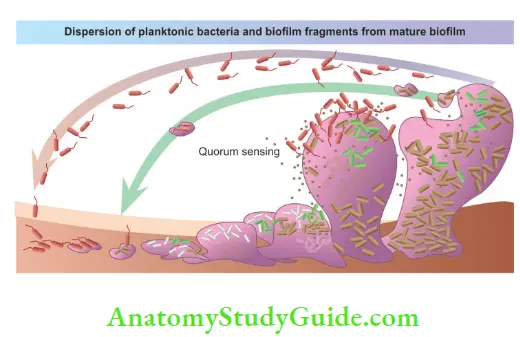
Microorganisms function less effectively as individuals compared to coherent groups. Quorum sensing allows bacteria to monitor the environment for other bacteria and allow alteration of one’s behavior in a population-wide scale. Pooling the activity of a quorum of cells can improve the successful persistence of bacteria.
Stages Of Biofilm Formation
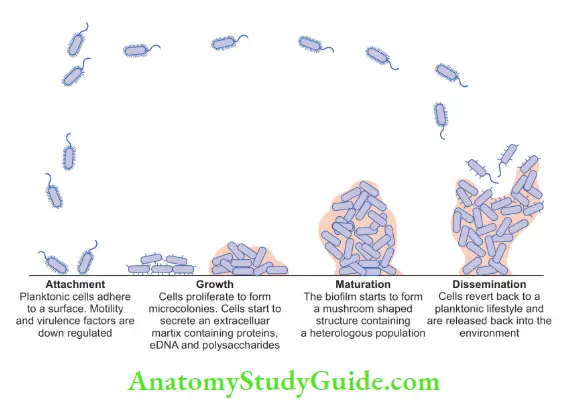
The formation of biofilm occurs in the following developmental stages.
1. Attachment:
It is the first stage of biofilm formation which involves the adsorption of planktonic cells to the surface. Film on tooth surface consists of proteins, extracellular or DNA, and glycoproteins from saliva and
gingival crevicular fluid and microbial products.
2. Growth:
It is the second stage which involves the proliferation of microorganisms to form microcolonies. In this, cells start to secrete an extracellular matrix containing proteins and polysaccharides.
3. Maturation:
It is the third stage which involves the formation of a structurally organized mixed microbial community. Biofilm forms a mushroom-shaped structure with a heterogeneous population.
4. Dissemination:
It is the fourth stage which involves the detachment of biofilm microorganisms. It is assumed that enzymes released from monolayers of bacteria result in their detachment. Detachment of microorganisms results in spreading and colonization to newer sites.
Types Of Endodontic Biofilm
Types of endodontic biofilm
- Intracanal microbial biofims
- Extraradicular microbial biofilms
- Periapical microbial biofilms
- Biomaterial-centered infections
Intracanal Microbial Biofims:
- This microbial biofilm formed on the root canal dentin of the infected tooth.
- The first identification of biofilm was earlier reported by Nair in 1987 under transmission electron microscopy.
- Mainly found bacteria in this biofilm are loose collections of cocci, rods, filaments, and spirochetes. Many other types of bacteria may also be found in these biofilms, but the ability of Enterococcus faecalis to form calcified biofilm on root canal dentin can be a main factor that contributes to its persistence and resistance to the treatment.
Extraradicular Microbial Biofilms:
- Also termed as root surface biofilms, formed on the root surface (cementum) adjacent to the root apex of endodontically infected teeth
- Sites
- Teeth with asymptomatic periapical periodontitis
- A chronic periapical abscess associated with sinus tract
- In these biofilms, mainly found organisms are cocci and rods
- Filamentous and fibrillar forms have also been observed in these. In some cases, calcified biofilms have also been observed on the apical root surface of teeth with lesions refractory to root canal treatment.
Periapical Microbial Biofilms:
- Periapical microbial biofilms are isolated biofilms found in the periapical region of an endodontically infected tooth, asymptomatic periapical lesions refractory to endodontic treatment.
- Microorganism involved are
- Actinomyces
- Propionibacterium propionicum
Fimbriae present on Actinomyces cells help them to adhere to the root canal wall and to dentinal debris pushed out in the periapical region during treatment and to adhere to other bacteria or host cells as they advance into periapical areas. This results in the formation of aggregates of cohesive colonies of tangled filaments.
Biomaterial-Centered Infections:
- Biomaterial-centered infection (BCI) occurs when bacteria adhere to an artificial biomaterial surface such as root canal obturating materials and form biofilms. It can be intraarticular or extraarticular depending on whether the obturating material is within the root canal space or it has extruded beyond the root apex.
- The presence of biomaterial in close proximity to the host immune system can increase the susceptibility to BCI.
- Staphylococcus, Staphylococcus aureus, enterococci, streptococci, Pseudomonas aeruginosa, and E. faecalis form biofilm on Gutta-Percha points where as Fusobacterium nucleatum, Propionibacterium acnes, Porphyromonas gingivalis, and Prevotella intermedia do not form biofilm on Gutta-Percha points.
- Bacterial adhesion to biomaterial surface can be explained in the following three phases:
- Phase 1: Transport of bacteria to the biomaterial surface
- Phase 2: Initial, nonspecific adhesion phase
- Phase 3: Specific adhesion phase.
Ultrastructure Of Biofilm
A fully formed biofilm is labeled as a heterogeneous arrangement of microbial cells on a solid surface.
Microcolonies:
- Microcolonies or cell clusters form the basic structural unit of a biofilm. These are formed by the adherence of bacterial cells on the surface
- These are isolated units of densely packed bacterial cell aggregates consisting of one or many species.
- There is a 3-D distribution of bacterial cells of different physiological and metabolic states within a biofilm.
Glycocalyx Matrix:
- A glycocalyx matrix, made up of EPS (extracellular polysaccharides)surrounds the microcolonies and holds the bacterial cell to the substrate
- Biofilm consists of matrix material 85% and cells 15%
- A fresh biofilm matrix is made of biopolymers, such as polysaccharides, proteins, nucleic acids, and salts.
- The structure and composition of mature biofilm get modified according to environmental conditions like nutritional availability, nature of fluid movements, physicochemical properties of the substrate, etc.
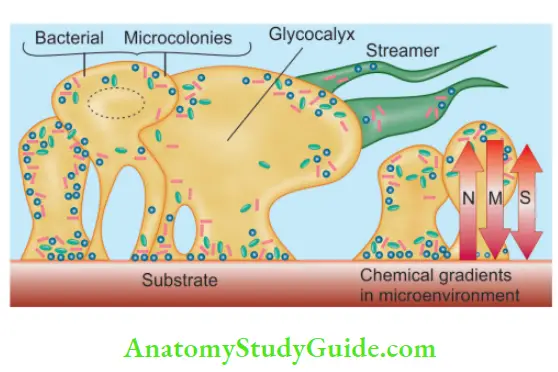
Water Channels:
- Present in biofilm facilitate efficient exchange of materials between bacterial cells and fluid, which also helps to coordinate functions in a biofilm community. A fully hydrated biofilm appears as a tower- or mushroom-shaped structure adherent to a substrate.
Microbes In Endodontic Biofilms
Methods to isolate microbes
- Culture
- Microscopy
- Immunological methods
- Molecular biology methods
Microorganisms involved in biofilm formation
- E. faecalis
- Coagulase–negative Staphylococcus
- Streptococci
- Actinomyces species
- P. propionicum
- Others: P. aeruginosa, fungi, F. nucleatum, Porphyromonas gingivalis, Tannerella forsythensis, Actinomyces species, and P. propionicum
About 40%–55% of the endodontic microbiota in primary infections is composed of species still uncultivated.
Methods To Eradicate Biofilms
Sodium Hypochlorite:
It is effective against biofilms containing P. intermedia, Peptostreptococcus micros, Streptococcus intermedius, F. nucleatum, and E. faecalis as it disrupts oxidative phosphorylation and inhibits DNA synthesis of bacteria. Many studies have shown that NaOCl is the only irrigant that has a significant effect on biofilm viability and architecture.
Chlorhexidine Digluconate:
It is effective against both Gram-positive and Gram-negative bacteria due to its ability to denature the bacterial cell wall while forming pores in the membrane. Williamson et al. showed that 2% of chlorhexidine was less effective against E. faecalis biofilm when compared to 6% NaOCl.
Shen et al. showed that the combined use of chlorhexidine along with mechanical instrumentation showed a more noticeable antimicrobial effect against the biofilms. It has been shown that bacteria in mature biofilms and nutrient-limited biofilms are more resistant to chlorhexidine than in young biofilms.
QMix:
QMix consists of EDTA, chlorhexidine, and detergent. It is as effective as 6% NaOCl in killing 1-day-old E. faecalis but slightly less effective against bacteria in the 3-week-old biofilm.
Iodine:
It is bactericidal, fungicidal, tuberculocidal, virucidal, and sporicidal as it penetrates into microorganisms and attacks proteins, nucleotides, and fatty acids resulting in cell death. Studies have shown that iodine and NaOCl caused 100% bacteria elimination after 1-hour incubation for all used strains.
However, 10% povidone-iodine has shown to be less effective against E. faecalis biofilm as compared to NaOCl.
EDTA:
It has little antibacterial activity. On direct exposure for an extended time, EDTA extracts bacterial surface proteins by combining with metal ions from the cell envelope, which can eventually lead to bacterial death.
MTAD (Mixture of a Tetracycline Isomer, an Acid, and a Detergent):
It has been shown that Biopure MTAD does not disintegrate and remove bacterial biofilm. It is not able to remove E. faecalis biofilm.
Tetraclean:
It is a mixture of antibiotic, acid, and a detergent like MTAD but with different concentrations. Papen et al. (2010) found that tetra-clean is more effective than MTAD against E. faecalis in planktonic culture.
Calcium Hydroxide (CH):
A commonly used intracanal medicament has been shown to be ineffective in killing E. faecalis on its own, especially when a high pH is not maintained. However, a combination of calcium hydroxide and camphorated paramonochlorophenol completely eliminates E. faecalis. 2% chlorhexidine gel when combined with calcium hydroxide achieves a pH of 12.8 and can completely eliminate E. faecalis within dentinal tubules.
Ultrasonically Activated Irrigation:
Bhuva et al. (2010) found that use of ultrasonically activated irrigation using 1% sodium hypochlorite, followed by root canal cleaning and shaping improves canal and isthmus cleanliness in terms of necrotic debris/biofilm removal.
Ozone/Ozonated Water:
Viera et al. (1999) reported that Ozone in 0.1 ppm concentration is able to completely kill bacteria after 15 or 30 min of contact time. But Hems et al. showed that NaOCl is superior to Ozonated water in killing E. faecalis in biofilm.
Lasers:
Lasers induce a thermal effect producing an alteration in the bacterial cell wall leading to changes in the osmotic gradients and cell death. Noiri et al. found that Er:YAG irradiation reduces the number of viable cells in most of the biofilms of A. naeslundii, E. faecalis, Lactobacillus casei, P. acnes, F. nucleatum, P. gingivalis, and Prevotella nigrescens.
Plasma Dental Probe:
It is effective for tooth disinfection. Plasma emission spectroscopy identifies atomic oxygen as one of the likely active agents for the bactericidal effect.
Photoactivated Disinfection:
It is a combination of a photosensitizer solution and low-power laser light. Photodynamic therapy/Light-activated therapy destroys an endodontic biofilm when a photosensitizer selectively accumulated in the target cell is activated by a visible light of appropriate wavelength.
Antibacterial Nanoparticles:
Antibacterial nanoparticles bind to negatively charged surfaces and have excellent antimicrobial and antifungal activities. Studies have also shown that the treatment of root dentin with ZnO nanoparticles, chitosan-layer–ZnO nanoparticles, or chitosan nanoparticles produces an 80%–95% reduction in the adherence of E. faecalis to dentin.
Endoactivator System:
It is able to debride lateral canals, remove the smear layer, and
dislodge simulated biofilm clumps within the curved canals.
Herbal Irrigants:
Herbs like green tea, Triphala, German chamomile, and tea tree oil can be used as irrigants because of their anti-inflammatory antioxidant, and radical scavenging activity. But when compared, studies have shown 5% NaOCl to be more effective against biofilm.
Smear Layer In Endodontics
The smear layer in instrumented root canals was first researched
by McComb and Smith. It contains remnants of odontoblastic processes, pulp tissue, bacteria, and dentin.
AAE (American Association of Endodontists) defined a smear layer as a “surface film of debris retained on dentin or other tooth surfaces like enamel, cementum after instrumentation with either rotary instruments or endodontic files.”
Structure of Smear Layer:
It consists of two separate layers, a superficial layer and a layer loosely, attached to the underlying dentin. It is composed of organic and inorganic components. The organic component consists of heated coagulated proteins, necrotic or viable pulp tissues, odontoblastic processes, saliva, blood cells, and microorganisms.
The inorganic component is made up of tooth structure and some nonspecific inorganic constituents. The smear layer on deep dentin contains more organic material than superficial dentin. It is because of a greater number of proteoglycans lining the tubules or the greater number of odontoblastic processes near the pulp.
Dentin debris enters the tubules and forms smear plugs to occlude the ends of tubules. The smear layer is 2–5 µm thick with particles ranging in size from 0.5 to 1.5 µm. Depth entering the dentinal tubules varies from a few micrometers to 40 µm.
Its thickness depends upon the type and sharpness of cutting instruments, the amount and type of irrigating solution used, and whether dentin is wet or dry when cut. There does not seem to be a consensus on removing of smear layer. It has both advantages and disadvantages that remain controversial.
- Motorized preparation with Gates Glidden or post drills produces a greater volume of smear layer as compared to hand filing due to increased centrifugal forces resulting from the proximity of the instrument to the dentin wall. Also, filling a canal without irrigation will produce a thicker smear layer.
- Post-space preparation results in a thicker smear layer and also consists of fragments of sealer and gutta-percha. So when posts are cemented without removal of the smear layer it will bond to the loosely adherent smear layer and ultimately bond will break
Advantages:
- It provides a natural barrier to bacteria/bacterial products in the dentinal tubules.
- Removal of the smear layer may lead to bacterial invasion of dentinal tubules if the seal fails.
- Reduction of dentin permeability to bacterial toxins and oral fluids.
- Reduction of diffusion of flids and prevents wetness of cut dentin surface.
Disadvantages:
- It acts as a barrier against the action of irrigants and disinfectants during the chemo-mechanical preparation of the root canal.
- It may harbor bacteria which multiply by taking nourishment from the smear layer or dentinal fluid
- Bacteria may penetrate deeper into the dentinal tubules.
- It is loosely adherent and so may lead to microleakage between root canal filing and dentinal wall.
- It doesn’t allow root canal sealer to penetrate into the dentinal tubules and hence jeopardize the fluid-tight seal.
- The smear layer when exposed to periapical exudate dissolves that leads to leakage and void between the dentin and obturating materials.
- The smear layer itself is infected.
To Keep or Remove the Smear Layer?
Drake et al. showed that the removal of the smear layer opened the dentinal tubules allowing bacteria to colonize in the tubules to a much higher degree compared with roots with an intact smear layer. Removal of the smear layer facilitates passive penetration of bacteria.
It is not practical to remove the smear layer completely due to the anatomical
complexity of the root canal. Also, a 95% success rate has been achieved in root canal treatment even without the removal of the smear layer. On the other hand, the removal of the smear layer improved the fluid-tight seal of the root canal system by opening up the tubules and exposing the surface collagen for covalent linkage while the firing technique/sealer doesn’t make much
difference.
After removing the smear layer thermoplasticized gutta-percha adapts better with/without sealer. Also, sealers/obturating material/cement for post-cementation require the removal of the smear layer. Removal of the smear layer results in sealer penetration from 40 to 60 µm which increases the interface between the obturating material and the dentin.
However, ZOE (Zinc oxide eugenol) sealers are the most commonly used. But their particle size is >1 µm. The diameter of dentinal tubules is <1 µm near the apex. So, the removal of the smear layer seal at the apex is questionable.
Agents Used for Removal of Smear Layer:
1. Sodium hypochlorite:
Dissolves organic tissues. Hence, it will dissolve organic components of the smear layer. It’s ability is increased with increased temperature.
2. Chelating agents
- EDTA:
It forms chelates with calcium of dentin. It decalcifis dentin to a depth of 20–30 µm in 5 min. However, the effect was negligible in apical one-third of root canals. Paste-type EDTA is less effective in removing the smear layer than liquid.
Some EDTA preparations contain wax which leaves a residue on walls and prevents the formation of flid tight seal. The optimal working time of EDTAC in root canal is 15 min and no further chelating action occurs after that.
- Salvizol:
0.5% BDAC (bis-dequalinium-acetate) removes organic as well as inorganic smear layer throughout the root canal. It has better cleaning than EDTAC and removal of the organic portion is comparable to 5.25% NaOCl.
- EGTA [Ethylene glycol-bis (β-aminoethyl ether)-N,N,ntetraacetic acid]:
EDTA was more effective in removing the smear layer than EGTA which was much less effective in the apical third. But EDTA also leads to erosion of
peritubular and intertubular dentin
- Tetracycline:
Tetracycline hydrochloride, minocycline, and doxycycline Thy have low pH and can act as chelator. Barkhordar et al. reported that doxycycline hydrochloride was effective in removing the smear layer from instrumented canals and root end cavity preparations.
- MTAD:
Removes smear layer and also leads to disinfection of root canal.
- Citric acid:
10%, 25%, and 50% solutions can be used. But 25% citric acid was less effective than the 17% EDTA– NaOCl combination. Also, citric acid left precipitates on the root canal wall which might be disadvantageous to root canal obturation.
- 3% and 20% polyacrylic acid, lactic acid, and 25% tannic acid may be used. Berry et al. reported 40% polyacrylic acid to be very effective in the removal of the smear layer.
- 5.2% NaOCl and 17% EDTA:
Alternate use can remove both inorganic and organic portions of the smear layer.
3. Chlorhexidine:
Does not dissolve or remove the smear layer.
4. Ultrasonics:
Final irrigation with ultrasonics for 3–5 min produced smear layer-free canals. When used during instrumentation direct contact of fie with canal walls decreases acoustic streaming. Acoustic streaming is maximized when a small file as No. 15 is used which increases microstreaming for debris removal.
Other investigations have shown that it is not effective and it is unnecessary to combine 15% EDTAC with distilled water or 1% NaOCl to remove the smear layer.
5. Lasers:
Vaporize tissues in main root canal and remove smear layer
- Nd: YAG laser, CO 2 laser, argon fluoride excimer laser: Have shown variable results from no effect, disruption of smear layer to actual melting, and recrystallization of dentin.
- Er:YAG: Removal of smear layer without melting/ charring or recrystallization. But destruction of peritubular dentin has been observed However, lasers have large probes and hence cannot be used in small canals.
6. Micro end brush:
Invented by Ruddle. They can be used with hand, rotary or sonic or ultrasonic handpieces. The brush is made of a braided/twisted wire base which is flexible to reach all irregularities of the root canal. Bristles are made of nylon that radiates from a wire base
It can be used in slow-speed handpieces at a speed of 150–300 rpm.
7. Endoactivator
8. Maleic acid: 5%–7% can be used
QMix: Contains a mixture of biguanide [antimicrobial agents, polyamino carboxylic acid (as calcium chelating agent)]. It removes the smear layer and can be used as a final rinse instead of EDTA followed by CHX.
Modification of Smear Layer:
A percentage of 4% of titanium fluoride and 30% potassium oxalate have been used to modify the smear layer. Titanium is a polyvalent metal and has a remarkable capacity to bind to the organic material of dentin forming a tenacious coating of titanium dioxide which is resistant to dissolution by KOH washing/HCl treatment.
Conclusion-
The most common cause of endodontic infection is the surface-associated growth of microorganisms. To understand the pathogenesis and disinfection of root canal microflora, the biofilm concept should be applied. One must understand the formation and mechanism of resistant endodontic biofilm. Contrary to the vulnerable planktonic state, bacteria are protected from the antibacterial agents in biofilm.
To date, with the advent of scientific approaches along with the coordination of newer clinical, laboratory, chemical, and research methods, it is possible to imagine overcoming biofilm problems.
The problem of the smear layer is still controversial. However, growing evidence supports its removal. If it is to be removed 5% NaOCl followed by 10 mL of EDTA for 1 min should be used. NaOCl should not be used after EDTA as it leads to remarkable erosion of dentin. CHX can be used in the end to disinfect dentin. Alternatively, a final rinse with MTAD/ QMix or ultrasonic may be used.
Sealers with bonding ability like AH Plus show greater bond strength when the smear layer has been removed. However, some studies show decreased bond strength, but completely reversed by the application of 10% ascorbic acid/10% sodium ascorbate.
With the advent of adhesive dentistry for endodontics use of medicated and resin-based sealers and obturating materials like Resilon and Active GP (Gutta-percha) definitely mandates the removal of smear layer for producing secondary and tertiary monoblocks for adequate bonding and obtaining optimal bond strength for a fluid-tight seal.
Bibliography
- Ajeti MSN, Hoxha SV, Elezi SX, et al. Demineralization of root
canal dentine with EDTA and citric acid in different concentrations, pH, and application times. ILIRIA Int Rev 2011;1(2). - Buyukyilmaz T, Ogaard B, Rolla G. Th resistance of titanium tetrafluoride treated human enamel to strong hydrochloric acid. Eur J Oral Sci 1997;105:473–7.
- Chavez de Paz LE. Redefining the persistent infection in root canals: Possible role of biofilm communities. J Endod 2007;33:652–62.
- Chávez de Paz LE, Bergenholtz G, Svensäter G. Th effcts of antimicrobials on endodontic biofim bacteria. J Endod 2010;36:70–7.
- Distel JW, Hatton JF, Gillespie MJ. Biofilm formation in medicated root canals. J Endod 2002;28:689–93.
- Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev 2002;15:167–93.
- Donlan RM, Costerton JW. Biofilms: Survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev 2002;15:167–93.
- Galvan DA, Ciarlone AE, Pashley DH, et al. Effect of smear layer removal on the diffusion permeability of human roots. J Endod 1994;20(2):83–6.
- Gencoglu N, Samani S, Gunday M. Dentinal wall adaptation of thermoplasticized gutta-percha in the absence or presence of smear layer: a scanning electron microscopic study. J Endod 1933;19:558–62.
- Gencoglu N, Samani S, Gunday M. Evaluation of sealing properties of Thrmafi and Ultrafi techniques in the absence or presence of smear layer. J Endod 1992;19:599–603.
- Hargreaves KM, Cohen S. Pathways of the pulp. 10th ed.
Mosby: Elsevier; 2012. - Ingle JI, Bakland LK, Decker BC. Endodontics. 6th ed.
Elsevier; 2008. - Kishen A, Haapasalo M. Biofim models and methods of biofilm assessment. Endod Topics 2010;22:58–78.
- Kishen A. Advanced therapeutic options for endodontic biofilms. Endod Topics 2010;22:99–123.
- Kishen A. Advanced therapeutic options for endodontic
biofilms. Endo Topics 2012;22:99–123. - Lester KS, Boyde A. Scanning electron microscopy of instrumented, irrigated, filed root canals. Brit Dent J 1977;43:359.
- Mader CL, Baumgartner JC, Donald DP. Scanning electron microscope investigations of the smeared layer on root canal walls. J Endod 1984;10:477–83.
- Sahar-Helft S, Stabholtz A. Removing smear layer during endodontic treatment by different techniques – a in vitro study. A clinical case – Endodontic treatment with Er: YAG Laser. Stoma Edu J 2016;3(2):162–7.
- Serper A, Calt S. Th demineralizing effects of EDTA at different concentrations and pH. J Endod 2002;28(7):501–2.
- Shahravan A, Haghdoost AA, Adl A, et al. Effect of smear layer on sealing ability of canal obturation: A systematic review and meta-analysis. J Endod 2007;33(2):96–105.
- Shehadat SA. Smear layer in endodontics: role and management. J Clin Dent Oral Health 2017;1:1–2.
- Siqueira JF, Jr, Rôças IN. Exploiting molecular methods to explore endodontic infections: Part 2-Redefiing the endodontic microbiota. J Endod. 2005;31:488–98.
- Svensater G, Bergerholtz G. Biofims in endodontic infections. Endo Topics 2004;9:27–36.
- Usha HL, Kaiwar A, Mehta D. Biofilm in endodontics: New understanding to an old problem. Int J Contem Dent 2010;1:44–51.
- Vallabhaneni K, Kakarla P, Avula SSJ, et al. Comparative analyses of smear layer removal using four different irrigant solutions in the primary root canals–A scanning electron microscopic study. J Clin Diagn Res 2017;11(4): ZC64.
- Violich DR, Chandler NP. The smear layer in endodontics – A review. Int Endod J 2010;43(1):2–15.
- White RR, Goldman M, Lin RS. The influence of the smeared layer upon dentinal tubule penetration by plastic filling materials. J Endod 1984;10:558–62.
Leave a Reply