Blood Transfusion
- Administration of whole blood or its components into a patient is often necessary for various reasons. Since the only available source of blood is by voluntary human donation, which is scarce, whole blood is separated into its components, namely packed red blood cells, fresh frozen plasma, platelets, and cryoprecipitate.
- Some coagulation factors may be isolated and separately stored (e.g. factor 8 for administration into haemophiliacs).
- Packed red cells are the most common blood products used. Their transfusion increases the oxygen-carrying capacity of blood. Transfusion of one unit of packed cells raises haemoglobin by approximately 1 g% in an adult.
Read And Learn More: Basic Principles Of Surgery Notes
Table of Contents
Indications for Blood Transfusion:
1. Packed red cells are used to replace acute and major blood loss in:
- Haemorrhagic shock
Guidelines: When to Transfuse:
-
- Haemoglobin <7 g%: Transfusion of red cells is likely to be associated with reduced mortality.
- Haemoglobin 7–10 g%: The decision to transfuse blood should be taken, only if there are clinical symptoms and signs due to anaemia.
- Haemoglobin >10 g%: Transfusion is not indicated.
- Major surgery—open heart surgery, gastrectomy
- Extensive burns
2. Packed red cells are also used to treat anaemia due to:
- Extensive burns
- Chronic blood loss—haemorrhoids, bleeding disorders, chronic duodenal ulcer, etc.
- Inadequate production—malignancies, nutritional anaemia
3. Platelet concentrates are administered to replace platelets in thrombocytopenia:
- Fresh frozen plasma is given to replace plasma volume as well as Vit K dependent coagulation factors unresponsive to vitamin K replacement (for example liver disease, to reverse the effects of warfarin).
- Cryoprecipitate is used to replace fibrinogen in patients with disseminated intravascular coagulation.
4. Whole fresh blood administration is described in massive trauma with heavy blood loss, the rationale being that the patient rapidly loses all components of blood, so all components need to be replaced.
- Thus, it makes sense to administer whole blood, rather than individual components. However, many blood banks are reluctant to allow whole blood transfusions.
- This is to ensure that the donated blood can be optimally utilised. In such cases, red blood cells, fresh frozen plasma, and platelets are given in a 1:1:1 ratio.
Blood Products
Packed Red Blood Cells (PRBC):
- When whole blood is centrifuged, red blood cells settle down and the platelet-rich plasma remains supernatant.
- The plasma is transferred to another bag, while a preservative is added to red cells and the bag is sealed. Each bag of packed cells contains approximately 250–300 ml with a red cell concentration of 70%.
- Red cell transfusion is required for patients whose haemoglobin is <7 g% (e.g. patients undergoing chemotherapy, major surgery, delivery, trauma, grossly anaemic neonates and infants, sickle cell anaemia—especially in sickle cell crisis). Each unit of packed cells should increase haemoglobin by 1 g% and haematocrit by 3%.
- The red cells may be leucodepleted (white blood cells removed) for use in patients requiring multiple transfusions to prevent the development of antibodies to leukocytes or in those who are known to react to leucocytes.
- The rate of blood or blood product administration depends on the quantity and speed at which it is lost from the body. Slow transfusion is indicated in those with cardiac disease, renal dysfunction, severe chronic anaemia, and in paediatric patients.
- In semi-emergent situations (for example treatment of low haemoglobin in patients awaiting surgery or delivery), each bag of packed cells may be transfused slowly. The initial 25 ml may be given slowly to check whether the patient tolerates it.
- Thereafter, it can be transfused at a rate of 3–4 ml/kg/h (approximately 1.5 h for a bag of red cells). The red cells must be transfused within 4 hours of removing it from the refrigerator, as bacterial growth may be promoted after that.
- In emergent situations, red cells may be transfused much faster to keep up with the loss. When large amounts are transfused in a short period (see section on massive transfusion), the effects of other components, such as citrate, become significant.
- Packed red blood cells must be both ABO and Rh compatible unless the patient has life-threatening massive bleeding, in which case O –ve packed cells may be transfused.
Platelets:
- The bag containing platelet-rich plasma is again centrifuged to express off the plasma so that the bag with the remaining platelets can be sealed off. Each unit (50 mL) should contain at least 5.5 × 1010 platelets (platelet concentrate), and each unit should elevate the platelet count by 5–10,000 cells/mm3 in a 70 kg person. Platelets may be transfused prophylactically or for therapeutic purposes.
- Prophylactic platelet transfusion may be done when the platelet count is <10,000 cells/mm3 in oncology patients. In patients who are at high risk of alloimmunisation (e.g. leukaemia), the threshold for platelet transfusion is even lower at 5000 cells/mm3, whereas in patients with clinical instability, the threshold may be raised to 20,000 cells/mm3.
- Patients who need to undergo major surgery or require invasive procedures, such as spinal anaesthesia, liver biopsy, etc. generally need their platelet count elevated to >50,000 cells/mm3. However, patients requiring surgery in critical areas, such as neurosurgery or ophthalmic surgery, will need their platelet count raised to >1,00,000 cells/mm3.
- Therapeutic platelet transfusion is required in patients who are known to be thrombocytopenic and are actively bleeding (platelet count <50,000 cells/mm3). The dose of platelets may be calculated as follows: One unit of platelets (60 ml) for every 10 kg body weight.
- Platelets must be transfused within 4 hours of commencing the infusion. ABO compatibility is not required for platelets. Filters of 170–260 µ must be used to transfuse platelets as with other blood components.
Fresh Frozen Plasma (FFP):
- The remaining plasma (200–230 ml) may be stored in a frozen form (called fresh frozen plasma) at –18°C for one year. It needs to be thawed over half an hour before use.
- Fresh frozen plasma transfusion is indicated in patients with prolonged INR >1.5 who are bleeding or require surgery.
- FFP provides coagulation factors to those who are actively bleeding and those on warfarin. It is administered in a dose of 10–20 ml/kg body weight and must be ABO compatible.
- FFP must be transfused as soon as possible after thawing, and definitely within 24 hours. A hanging unit must be transfused within 6 hours of the commencement of transfusion due to the risk of bacterial contamination.
Cryoprecipitate:
- FFP may be further treated to produce cryoprecipitate, which is rich in fibrinogen. Transfusion of cryoprecipitate (each bag contains 20 ml) is indicated in patients with fibrinogen levels <1 g% (for example disseminated intravascular coagulation (DIC)).
- The dose is 0.2 units/kg body weight, but usually, 10 units are transfused initially and repeated as necessary.
Important points to remember before transfusing blood products:
- Always obtain informed consent from patients or immediate relatives (in emergent situations) prior to the transfusion.
- Recheck the patient’s blood group and that of the donor blood, preferably along with one other qualified person, to ensure that the correct bag is being transfused to the patient. The patient’s hospital number and the blood bag number should also be checked. Both people must sign on the transfusion report.
- Visually check the blood bag for any obvious abnormalities, such as very dark blood or visible clots, and if present, do not transfuse, and return it to the blood bank.
- Check the date of collection and date of expiry before transfusion. The storage shelf-life of red blood cells depends on the preservative added.
- Watch for any transfusion reactions.
Storage of Blood:
Preservative – RBC survival (days)
- ACD (acid-citrate-dextrose) – 21
- CPD (citrate-phosphate-dextrose) – 28
- CPDA (citrate-phosphate-dextrose-adenine) – 35
- SAGM (saline-adenine-glucose-mannitol) – 35
Whole blood: Transfusion is not advisable for routine use and is also not available from blood banks. However, there is some evidence in the literature that fresh whole-blood transfusion may be helpful in patients with massive trauma, as all blood components are replaced simultaneously to match the loss.
- However, this is difficult practically, as the blood products still need to be screened for various antigens and infections.
Other blood: Products available are fibrinogen concentrate (high risk of hepatitis), factor 8 and factor 9 concentrates (for use in haemophilia and Christmas disease, respectively) and factor 7 concentrate (for use in disseminated intravascular coagulation—DIC).
Complications Of Blood Transfusion Immune Complications
Haemolytic Reactions:
Major (ABO) Incompatibility Reaction:
- This is the result of mismatched blood transfusion.
- The majority of cases are due to technical errors, such as sampling, labelling, and dispatching.
- This causes intravascular haemolysis.
Complications Of Blood Transfusion Clinical features:
- Haematuria
- Pain in the loins (bilateral)
- Fever with chills and rigors
- Oliguria due to products of mismatched blood transfusion blocking the renal tubules. It results in acute renal tubular necrosis.
Complications Of Blood Transfusion Treatment:
- Stop the blood transfusion. Send the blood bag to the blood bank and recheck.
- Repeat coagulation profile.
- IV fluids, monitor urine output, check urine for Hb.
- Diuresis with furosemide 20–40 mg IV or injection mannitol 20% 100 ml IV to flush the kidney.
- If renal failure does occur, haemodialysis may be required.
- If haemodynamically unstable, cardiovascular support may be required.
Complications Of Blood Transfusion Minor Incompatibility Reaction:
- Occurs due to extravascular haemolysis.
- Usually mild, occurs at 2–21 days.
- Occurs due to antibodies to minor antigens.
- Malaise, jaundice, and fever.
- Treatment is supportive.
Nonhaemolytic Reactions:
Febrile Reaction:
- Occurs due to sensitisation to WBCs or platelets
- Increased temperature—no haemolysis
- Use of a 20–40 mm filter or leucocyte-depleted blood may prevent it.
Allergic Reaction:
- Occurs due to allergy to plasma products.
- Manifests as chills, rigors, and rashes all over.
- Often subsides with antihistamines, such as chlorpheniramine maleate 10 mg IV.
Transfusion-related Acute Lung Injury (TRALI):
- It is a rare complication, that occurs within 6 hours of a transfusion due to the presence of anti leucocyte antibodies in the transfused plasma, which cause the patient’s white cells to aggregate in the pulmonary circulation.
- This leads to degranulation of leucocytes, causing increased capillary permeability and noncardiogenic pulmonary oedema.
- Symptoms may vary from mild dyspnoea to full-blown acute respiratory distress syndrome. Proper supportive therapy will see that it resolves within 24–48 hours.
Congestive Cardiac Failure (CCF):
CCF may occur, if whole blood is transfused rapidly, especially in patients with chronic anaemia due to circulatory overload.
Congestive Cardiac Failure Treatment:
- Slow transfusion, injection of furosemide 20 mg IV.
- Packed cell transfusion is the choice in these patients.
Infectious Complications
- Serum hepatitis, AIDS, malaria, and syphilis are dangerous infectious diseases that may be transmitted from one patient to another through blood transfusion.
- The danger is increased in cases of multiple transfusions and in emergencies. “Prevention is better than cure”. Hence, it is mandatory to screen the blood for these diseases before transfusion.
Other Complications
Citrate toxicity (causing hypocalcaemia), dilutional coagulopathy (thrombocytopenia and dilution of clotting factors) can occur especially in massive transfusion.
Massive Blood Transfusion
Massive Blood Transfusion Definition: Massive blood transfusion has been defined as follows:
- Replacement of >1 blood volume (or >10 units of packed cells) in 24 hours.
- Half the patient’s blood volume in 6 hours.
- >4 RBC units in one hour with ongoing need for transfusion, or
- 500 ml over 5 min with haemodynamic instability and need for transfusion.
Massive blood loss may occur with trauma, postpartum haemorrhage, or during major surgeries.
Massive transfusion protocol: In patients with massive bleeding who are expected to require massive transfusion, the blood bank should be intimated to activate massive transfusion protocol (MTP).
- A blood sample must be sent for cross-matching with an initial request for 2 units of red blood cells. After this, if MTP is requested, the blood bank releases blood products in different ‘boxes’.
- The blood products are obtained box by box as necessary. If the bleeding stops, the blood bank should
Release of blood products on activation of MTP:
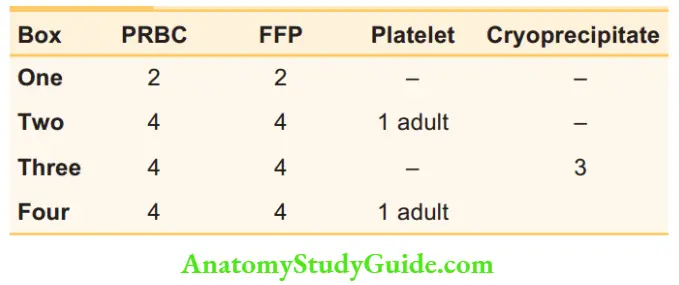
be intimated immediately, so that MTP can be terminated. If bleeding continues, boxes three and four are alternately requested.
- In such patients, regular and half-hourly measurements of acid-base status, haemoglobin, platelets, prothrombin time (PT), activated partial thromboplastin time (aPTT), fibrinogen, and serum calcium are required with an aim to normalise their values.
- The use of thromboelastography or Rotem allows visualisation of the process of clot formation and helps identify specific defects. This is now being made available as ‘point-of-care’ testing.
Disseminated Intravascular Coagulation (DIC):
- It occurs in massive blood transfusion, wherein all factors of coagulation are consumed, resulting in a bleeding disorder (consumption coagulopathy).
- It produces severe afibrinogenemia.
- It is treated by replacement with fibrinogen (cryoprecipitate) and other clotting factors.
Autologous Transfusion
This concept originated to avoid transfusion reactions that may develop when homologous blood is used.
- Here, the patient’s blood is used.
Types of Autologous Transfusion:
1. Predeposit: 2–5 units of blood may be donated over 2–4 weeks before elective surgery.
2. Preoperative haemodilution: For cases like surgery for thyrotoxicosis or abdominopelvic resection, one can expect 1–2 units of blood loss.
- Just before surgery, 1–2 units of blood are removed, stored in blood bags and serially numbered. Normovolaemia is ensured by infusing 2–3 times the volume drawn with crystalloids.
- The blood drawn is transfused in the reverse order after the procedure (the last bag is first and the first bag containing higher haematocrit is given last).
3. Blood salvage: Blood which is lost during surgery is collected, mixed with an anticoagulant solution, washed, and reinfused. This may be done, if the surgery does not involve severe infection, bowel resection, or malignancy, and does not contain materials such as bone cement.
Autologous Transfusion Advantage:
All the risks involved with blood transfusion are avoided.
Autologous Transfusion Disadvantages:
- May not be acceptable to the patient.
- Requires sophisticated equipment.

Blood Products:
Figure 18.1 depicts blood products.
Bleeding Disorders
Haemophilia:
This is the most common bleeding disorder, which occurs due to an X-linked genetic disorder of coagulation.
Bleeding Disorders Types:
- Haemophilia A: This results from a reduction of factor 8 (anti-haemophilic factor) and is carried by a recessive gene.
- Haemophilia B: This results due to a deficiency of factor 9.
1. Haemophilia A:
- ‘Excessive, unusual and unexpected bleeding’ during surgery could be due to haemophilia ‘A’. This must be confirmed with laboratory tests.
- A haemophilic patient’s daughters will be carriers, but all sons will be normal.
- Thus, a carrier woman has 50% chance of producing a haemophiliac male or a female carrier.
- The level of coagulation factor 8 in the blood may be <1% of that in a normal individual.
Bleeding Disorders Clinical Features:
Bleeding into joints (haemarthrosis):
- Large joints, such as knees, elbows, ankles, and wrists are affected.
- Spontaneous bleeding is common. It may also occur due to minor trauma.
- Repeated bleeding may result in permanent damage to the articular surfaces, resulting in deformity of the joints.
Blood changes in haemophilia A and von Willebrand’s disease:

Bleeding into muscles:
Calf muscle and psoas muscle haematomas are common, resulting in contraction and fibrosis of muscle, muscle pain, and weakness of the limb. Intramuscular injections should be avoided.
Investigations:
Bleeding Disorders Treatment:
- Administration of factor 8 concentrate by intravenous infusion is the treatment whenever there is bleeding.
- It should be given twice daily since it has a half-life of 12 hours.
- Any major surgical procedure should be carried out only after raising factor 8:C levels to 100% preoperatively, and it should be maintained >50% until healing occurs.
- Synthetic vasopressin (DDAVP) produces a rise in factor 8:C.
Bleeding Disorders Causes of Death:
- Cerebral haemorrhage used to be the most common cause of death in haemophiliacs. However, HIV infection seems to be the most common cause of death today.
- Hepatocellular carcinoma and cirrhosis due to HIV and HCV are the other causes (due to repeated blood transfusions).
2. Haemophilia B (Christmas Disease):
- Incidence: One in 30,000 males. The inheritance and clinical features are identical to haemophilia A.
- It is caused by a deficiency of factor 9.
- Treatment is with factor 9 concentrates.
von Willebrand’s Disease (vWD):
- In vWD, there is defective platelet function and factor 8:C deficiency due to a deficiency or abnormality of vWF.
- Epistaxis, menorrhagia, and bleeding following minor trauma or surgery are common.
- Haemarthrosis is rare.
- Treatment is similar to mild haemophilia. DDAVP, 4 infusion of factor 8:C or vWF is required before surgery.
- A clinical haematologist must be consulted to help optimise a patient with bleeding disorders for surgery.
Hyperbaric Oxygen
Hyperbaric oxygen is administered to patients when their haemoglobin is unable to carry adequate oxygen or when hyperoxia is required. Hyperbaric O2 is administered using either a monoplace (single patient) or multiple compression chamber.
Indications:
- Carbon monoxide poisoning: Hyperbaric O2 at 2–3 atm is given to increase the O2 dissolved in plasma (5–6 ml/dl at 3 atm, whereas it is 0.3 ml/dl at 1 atm) which is sufficient to meet the O2 requirement of the body, even in the absence of normal haemoglobin (severe CO poisoning).
- Infections such as gas gangrene: Hyperoxia suppresses the growth of anaerobic organisms.
- Cancer therapy to potentiate radiotherapy
- Arterial insufficiency
- Decompression sickness and air embolism: Hyperbaric oxygen reduces the size of air bubbles, and rapidly eliminates nitrogen from the air bubbles.
Contraindications:
Untreated pneumothorax: May expand with hyperbaric O2, resulting in life-threatening tension pneumothorax.
Multiple Choice Questions
Questions 1. Which of the following statements about packed red cells is true?
- It has a haematocrit of 35%
- The volume in one adult bag is 500 mL
- Shelf-life is 35 days with CPDA solution
- It contains no plasma
Answer: 3. Shelf-life is 35 days with CPDA solution
Questions 2. Haemolytic reactions due to mismatched blood transfusion include the following except:
- Haematuria
- Pain in the loins
- Fever with chills and rigours
- Diuresis
Answer: 4. Diuresis
Questions 3. Which of the following is decreased in disseminated intravascular coagulation?
- Prothrombin time
- Partial thromboplastin time
- Bleeding time
- Platelets
Answer: 4. Platelets
Questions 4. The following are true in haemophilia except:
- Prothrombin time is unaffected
- Partial thromboplastin time is unaffected
- Platelet count is unaffected
- Bleeding time is unaffected
Answer: 3. Platelet count is unaffected
Questions 5. The following are true about blood products:
- Fresh-frozen plasma is given in severe liver failure
- Cryoprecipitate is not used during massive transfusion
- Fibrinogen has a high risk of hepatitis
- Factor 8 is used in haemophilia
Answer: 2. Cryoprecipitate is not used during massive transfusion
Questions 6. The following are true about platelet transfusion:
- They remain viable for 3–5 days
- Each unit will raise platelet count by 5–10,000 cells/mm3
- Platelets must be ABO compatible
- Platelets may be low after heparin
Answer: 3. Platelets must be ABO compatible
Leave a Reply