Cardiology Question and Answers
Introduction And Symptomatology
Chest Pain:
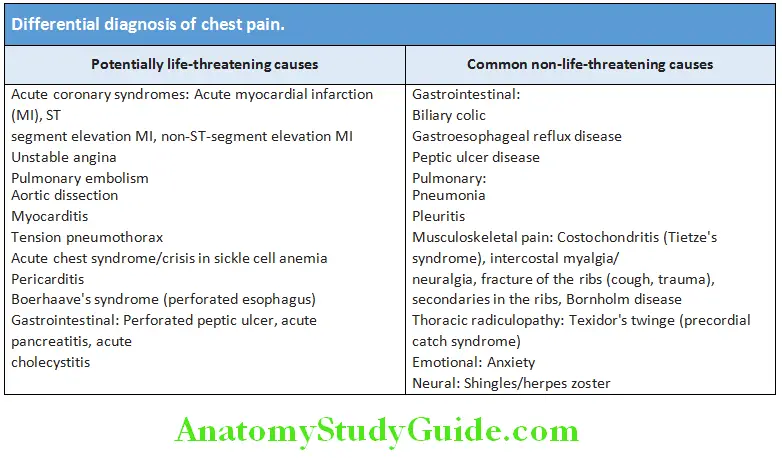
Question 1. Write a short essay on the differential diagnosis of chest pain. List the noncardiac causes of chest pain.
Answer:
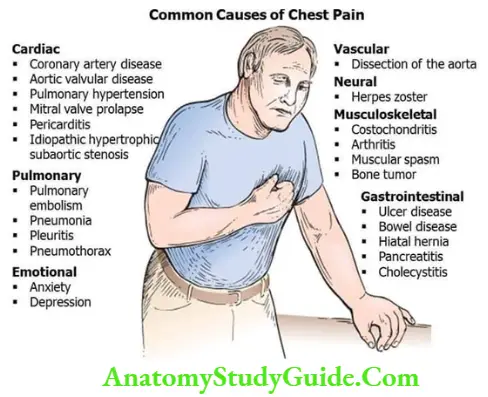
Chest pain is a common symptom of cardiac disease. It can be due to noncardiac causes, such as anxiety or diseases involving the respiratory, musculoskeletal, or gastrointestinal systems.
Read And Learn More: General Medicine Question And Answers
Common Causes of Chest Pain:
Differential Diagnosis of Chest Pain:
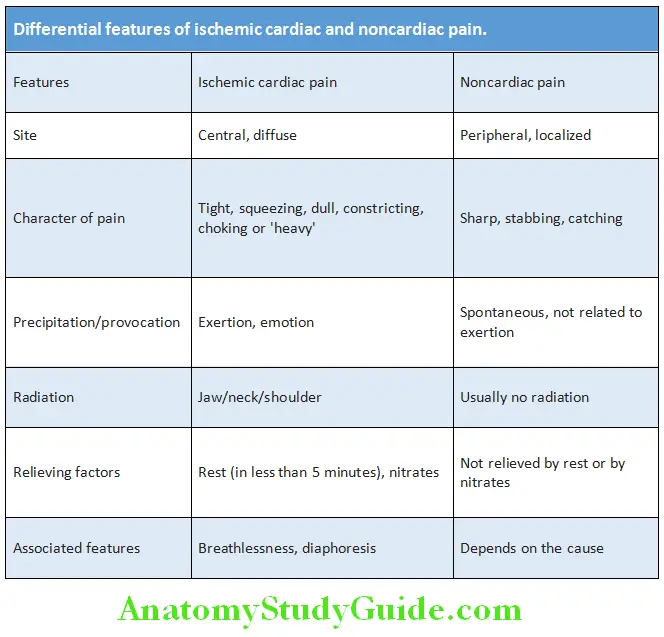
Diffrential Features of Ischemic Cardiac and Noncardiac Pain:
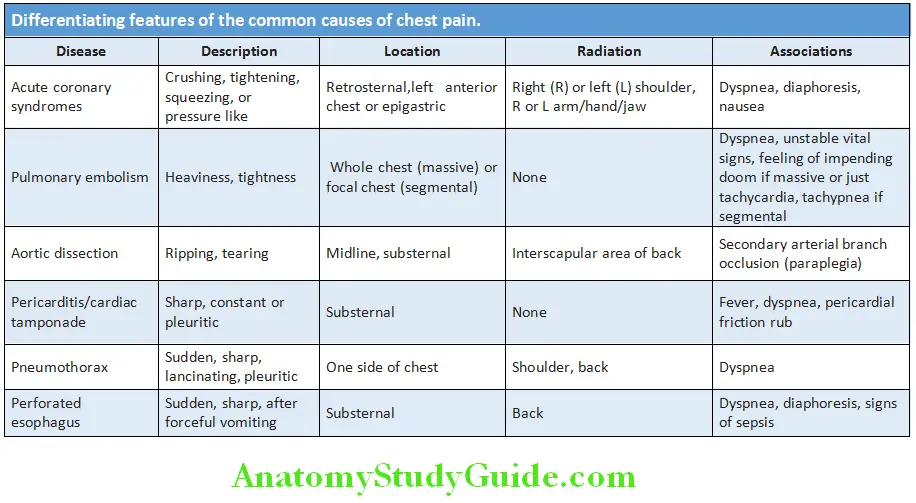
Differentiating Features:
Differentiating features of the common causes of chest pain are shown in Table:
Palpitations:
Question 2. Discuss the approach to a patient with palpitations.
Answer:
- Palpitation is the term used to describe an uncomfortable increased awareness of one’s own heartbeat or the sensation of slow, rapid, or irregular heart rhythms.
- Palpitations do not always indicate the presence of arrhythmia and conversely, an arrhythmia can occur without palpitations. Palpitations are usually noted when the patient is quietly resting.
- Palpitation can be either intermittent or sustained and either regular or irregular. A change in the rate, rhythm, or force of contraction can produce palpitations.
Causes of Palpitations:
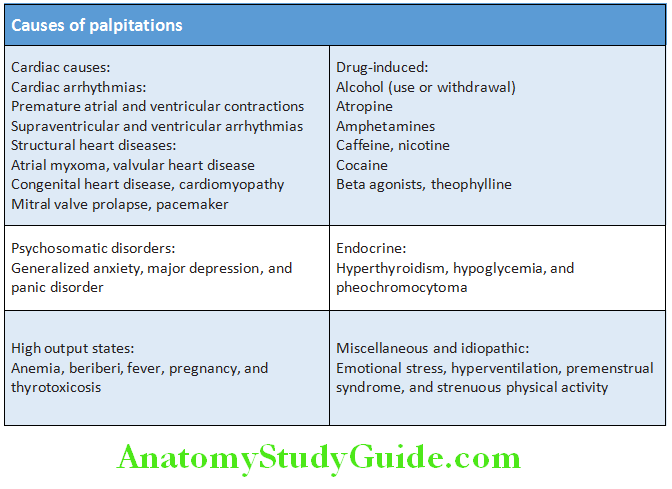
Evaluation of Palpitation:
- Detect and identify any underlying arrhythmia
- Determine presence of any organic heart disease
- Determine any precipitating cause
Clinical presentation:
- Duration and frequency of palpitations:
- Duration may be either short-lasting or persistent
- Note onset and offset of palpitations
- Frequency: It may occur daily, weekly, monthly, or yearly.
- Types of palpitations: They are classified according to the rate, rhythm, and intensity of heartbeat as follows.
- Extrasystolic palpitations: Due to ectopic beats, usually produce feelings of ‘missing/skipping a beat’ and/or a ‘sinking of the heart’ interspersed with periods during which the heart beats normally. Patients report that the heart seems to stop and then start again. It can often be seen in even in young individuals, usually without any disease of the heart, and generally benign.
-
- Tachycardiac palpitations is the rapid fluctuation like “beating wings” in the chest. It may be regular (e.g., in atrioventricular tachycardia, atrial flutter, or ventricular tachycardia) or irregular or arrhythmic (e.g., in atrial fibrillation).
- Anxiety-related palpitations are perceived as a form of anxiety. They begin and end gradually.
- Associated symptoms and circumstances:
- Palpitations developing after sudden changes in posture are usually due to intolerance to orthostatic or to episodes of atrioventricular nodal re-entrant tachycardia.
- Occurrence of syncope or other symptoms, such as severe fatigue, dyspnea, or angina, in addition to palpitations, is more common with structural heart disease.
- Hypersecretion of natriuretic hormone results in polyuria/postpalpitation diuresis in atrial fibrillation.
- Palpitations associated with anxiety or during panic attacks are usually due to sinus tachycardia secondary to the mental disturbance.
- Palpitations may be produced by an increase in the sympathetic drive during physical exercise.
- Typical description:
- Flip-flopping in the chest: Palpitations are sensed as the heart seeming to stop and then start again, producing a pounding or flip-flopping sensation. This type of palpitation is generally caused by supraventricular or ventricular premature contractions.
- Rapid fluttering in the chest: It is due to a sustained ventricular or supraventricular arrhythmia, including sinus tachycardia.
- Pounding in the neck: An irregular pounding feeling in the neck is caused by atrioventricular dissociation, with independent contraction of the atria and ventricles, resulting in occasional atrial contraction against a closed tricuspid and mitral valve. This produces cannon A waves, which are intermittent increases in the “A” wave of the jugular venous pulse. Cannon A waves may be seen with ventricular premature contractions, third-degree or complete heart block, or ventricular tachycardia (VT).
Physical examination:
It may help to confirm or refute the presence of an arrhythmia as a cause for palpitations:
- Measurement of the vital signs
- Assessment of the jugular venous pressure and pulse
- Auscultation of the chest and precordium
Physical examination Investigations:
- Electrocardiogram:
- A resting electrocardiogram can be used to diagnose the arrhythmia.
- If exertion induces arrhythmia and palpitations, exercise electrocardiography can be used to make the diagnosis.
- It may reveal bundle branch block (LBBB), short PR interval, delta waves, prolonged QT interval, ischemia, and enlargement of heart chambers, prior myocardial infarction, or other organic diseases of the heart.
- If the arrhythmia is not frequent, other methods must be used.
Physical examination These include:
- Continuous electrocardiographic (Holter) monitoring or telephonic monitoring.
- Echocardiography: To evaluate any structural heart disease and assessment of left ventricular function.
- An electrophysiology study is an invasive test of the electrical conduction system of the heart.
- Blood tests: Hemoglobin, serum glucose level, serum electrolytes, and thyroid function tests depending on the clinical findings.
Physical examination Management of palpitation:
- Most of the causes do not have serious arrhythmias or underlying structural heart disease.
- In symptomatic patients, occasional benign atrial or ventricular premature contractions can often be treated by beta-blockers.
- Avoid precipitation factors, such as alcohol, tobacco, or illicit drugs.
- If caused by pharmacologic agents: Consider alternative therapies if appropriate or possible.
- Psychiatric causes: By cognitive therapy or pharmacotherapy.
- Reassurance: After all the serious causes have been excluded.
Dyspnea:
Question 3. Write short note on the causes of acute dyspnea.
Answer:
Dyspnea Definition: “Dyspnea” is a term used to characterize a subjective experience of breathing discomfort that is comprised of qualitatively distinct sensations that vary in intensity. The experience derives from interactions among multiple physiological, psychological, social, and environmental factors, and may induce secondary physiological and behavioral responses.
Causes of Dyspnea:
Mechanisms:
- Chemoreceptors:
- Peripheral: Carotid and aortic bodies (sensitive to changes pO2, pCO2, and H+)
- Central: Medulla (sensitive only to changes pCO2, not pO2, change in pH of CSF).
- Increased work of breathing:
- Airflow obstruction: Bronchial asthma, chronic obstructive pulmonary disease (COPD), and tracheal obstruction
- Decreased pulmonary compliance: Pulmonary edema, fibrosis, and allergic alveolitis
- Restricted chest expansion: Ankylosing spondylitis, respiratory paralysis, and kyphoscoliosis.
- Increased ventilatory drive:
- Increased physiological dead space (V/Q mismatch): Consolidation, collapse, pleural effusion (PE), and pulmonary edema
- Hyperventilation due to receptor stimulation
- Chemoreceptors: Acidosis, hypoxia (shock, pneumonia), and hypercapnia
- J receptors at alveolo-capillary junction: Pulmonary edema, pulmonary embolism, and pulmonary congestion (activates Hering-Breuer reflex which terminates inspiratory effort before full inspiration is achieved—rapid and shallow)
- Muscle spindles in intercostal muscles: Tension-length disparity
- Central: Exertion, anxiety, thyrotoxicosis, and pheochromocytoma
- Impaired respiratory muscle function: Poliomyelitis, Guillain-Barré syndrome (GBS), and myasthenia gravis.
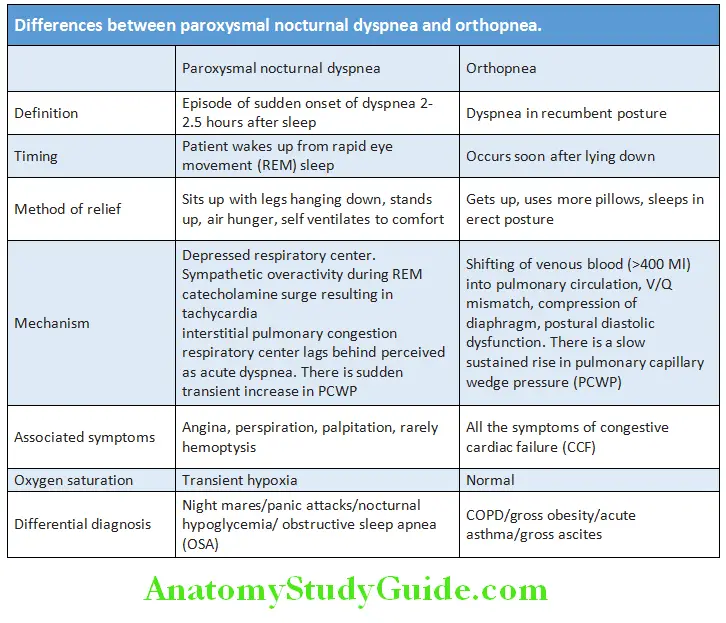
Dyspnea Orthopnea:
Dyspnea develops in recumbent position and is relieved by sitting up or by elevation of the head with pillows.
- Pulmonary congestion during recumbency (cannot be pumped out of LV) seen in congestive heart failure (CHF), COPD, and bronchial asthma.
- Increased venous return
- Diaphragm elevation leading to decreased vital capacity.
Dyspnea Basics: A normal 70 kg person breathes 12–15/minute with a tidal volume of 600 mL. A normal individual is not aware of respiratory effort until ventilation is doubled, and dyspnea is not experienced until ventilation is tripled. Causes of dyspnea in COPD.
Paroxysmal nocturnal dyspnea (PND): Attacks of dyspnea occur at night and awaken the patient from sleep.
It is due to decreased responsiveness of respiratory center in brain during sleep and pulmonary congestion (due to increased sympathetic activity during REM sleep) 2–3 hours after onset of sleep.
Takes 10–30 minutes for recovery after upright posture.
Dyspnea Causes: Specific sign of LV dysfunction and includes ischemic heart disease, aortic valve disease, hypertension, and cardiomyopathy. Differences between orthopnea and paroxysmal nocturnal dyspnea are presented in Table
Dyspnea in COPD:
- Hypoxia and hypercapnia: Chemoreceptors
- Increased airway resistance and hyperinflation
- Deconditioning: Reduced threshold at which respiratory muscles produce lactic acidosis
Trepopnea:
Aggravation of dyspnea when lying on one side and relieved by lying on opposite side.
Its causes are:
- Unilateral lung disease: Uninvolved normal lung receives more blood supply due to gravity.
- Congestive heart failure: Lying on right side enhances venous return and sympathetic activity.
- Lung tumor: Gravity-induced compression of blood vessels or lung.
Platypnea: Dyspnea on sitting or standing and relieved by supine position.
Its causes are:
- Venous to arterial shunting (lung bases)
- Intracardiac shunts (ASD, pneumonectomy)
- Intrapulmonary right to left shunt [hepatopulmonary syndrome, pulmonary embolism (PE), COPD]
- Acute respiratory distress syndrome (ARDS)
Bendopnea:
A newly described symptom in patients with heart failure, is mediated via a further increase in ventricular filling pressures during bending in subjects whose sitting ventricular filling pressures are already high, particularly in patients with low cardiac index.
A patient sits in a chair, bends at the waist, and touches his/her feet. Bendopnea is considered present if dyspnea occurs within 30s of bending.
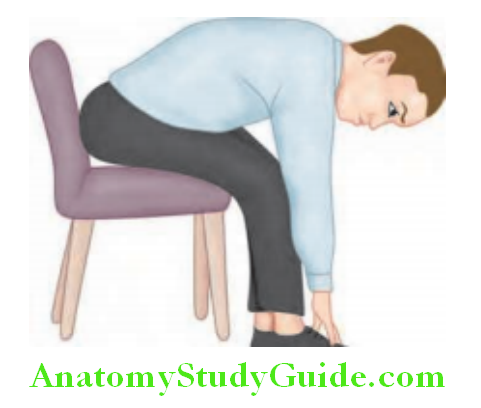
Approach to Dyspnea:
- Onset and duration:
- Minutes to hours (rapid onset): Pneumothorax, acute asthma, PE, pulmonary edema, and foreign body.
- Hours to days (gradual onset): Pneumonia, pleural effusion, anemia, Guillain–Barré syndrome (GBS).
- Months to years (slow onset): Pulmonary tuberculosis (PTB), COPD, carcinoma, and fibrosing alveolitis.
- Severity of dyspnea:
- Aggravating and relieving factors:
- Improves on weekend/holidays: Occupational asthma, extrinsic allergic alveolitis
- Recumbency/sleep: Orthopnea/paroxysmal nocturnal dyspnea (PND)
- Aggravating and relieving factors:
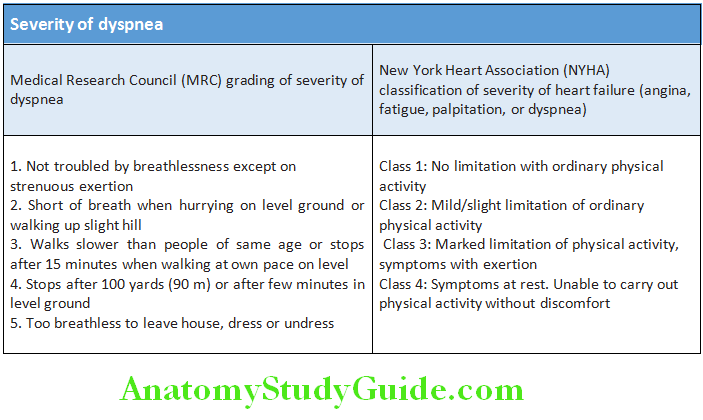
Question 4. NYHA functional classification of cardiac disability.
Answer:
- Associated symptoms:
- Pleuritic chest pain: Pneumonia, pulmonary infarction, rib fracture, and pneumothorax
- Central nonpleuritic chest pain: Myocardial infarction, massive pulmonary embolism
- Cough or wheeze: Asthma, pulmonary embolism, and pneumothorax
Arterial Pulses:
Question 5. Write short essay/note on the clinical value of examination of radial and carotid pulses at bedside.
Answer:
Definition of arterial pulse: It is a pressure distension wave produced by the contraction and relaxation of the left ventricle against a partially filled aorta which is transmitted to peripheries and is felt against bony prominences.
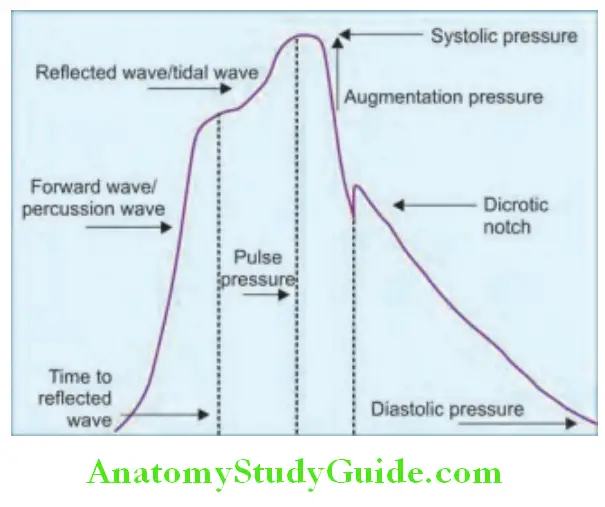
Peripheral arterial pulses: These include radial, brachial, carotid, femoral, popliteal, posterior tibial, and dorsalis pedis pulses. Right radial pulse is the first pulse to be examined during clinical examination. Normal pulse wave.
Examination of the Arterial Pulse:
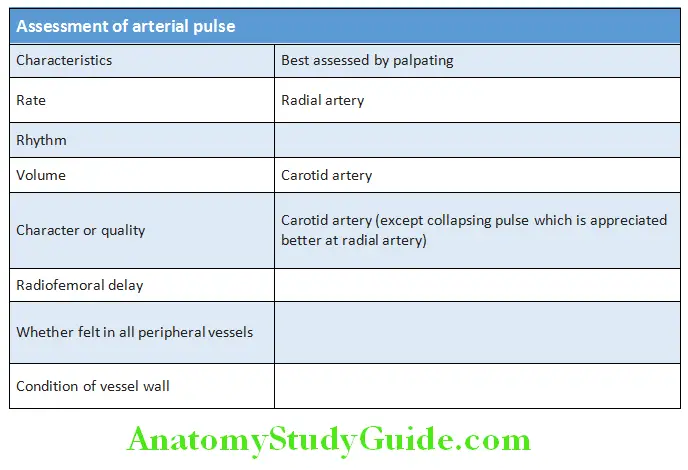
The character of the pulse is determined by stroke volume and arterial compliance, and is best assessed by palpating a major artery, such as the carotid or brachial artery.
Pulse rate:
- Normal (resting) pulse rate in an adult is between 60 and 100 beats/minute (bpm).
- Should be counted for 1 full minute by palpating the radial artery.
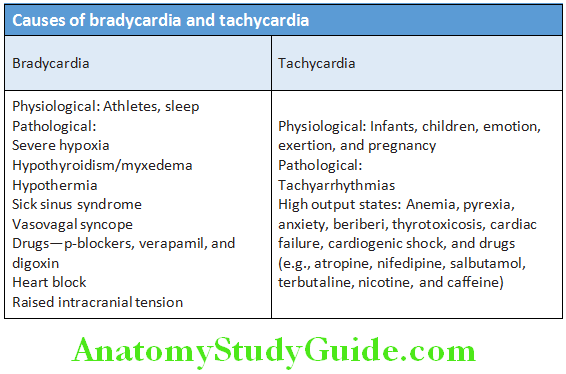
- Sinus bradycardia: Resting pulse rate is less than 60 beats/minute.
- Sinus tachycardia: Resting pulse rate is more than 100 beats/minute.
- Causes of bradycardia and tachycardia.
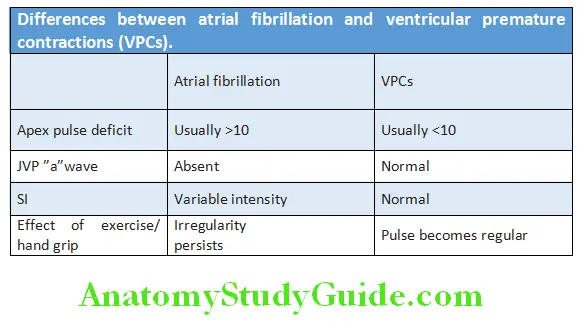
Pulse deficit (Apex-pulse deficit) is the difference between the heart rate (counted by auscultation) and pulse rate when counted simultaneously for 1 full minute.
Arterial Pulses Causes: Pulse defiit of more than 10/minute occurs in atrial firillation and less than 10/minute may be found with ventricular premature beats or slow/controlled atrial firillation (AF).
Rhythm:
- Rhythm is assessed by palpating the radial pulse. The normal rhythm is regular.
- Causes of various types of arterial pulse rhythm abnormalities
Causes of various types of arterial pulse rhythm abnormalities:
Regularly irregular:
- Atrial tachyarrhythmias, sinus arrhythmia, and partial AV blocks
- Ventricular bigeminy, trigeminy
Irregularly irregular:
- Atrial or ventricular ectopics
- Atrial fibrillation
- Atrial tachyarrhythmia with AV blocks
- Frequent extrasystoles
Regular with occasional irregularity:
- Extrasystoles
Question 6. What are the causes for an irregularly irregular pulse?
Answer:
- Ventricular ectopics: These develop as occasional or repeated irregularities superimposed on a regular pulse rhythm. Intermittent heart block also present with occasional beats dropped from an otherwise regular rhythm.
- Atrial fibrillation: It develops an irregularly irregular pulse. This irregular pattern persists when the pulse increases in response to exercise. On the contrary pulse irregularity due to ectopic beats usually disappears with exercise.
Pulse volume:
- Pulse volume is best assessed by palpating the carotid artery.
- However, the pulse pressure (i.e., difference between systolic and diastolic blood pressure) provides an accurate measure of pulse volume.
- Pulse volume is normal when pulse pressure is between 30 and 60 mm Hg, low when it is less than 30 mm Hg and large volume when more than 60 mm Hg.
Pulse character or quality:
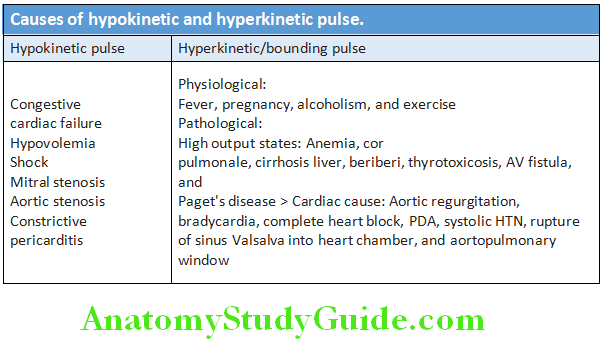
- Differences between atrial fibrillation and ventricular premature contractions (VPCs) are shown in Table Causes of hypokinetic and hyperkinetic pulse are listed in Table.Various types of pulse quality and its causes are mentioned in Table 7.10.
- Grading of pulse: Palpation of pulse is done by the fingertips and intensity of the pulse is graded from 0 to 4 +. 0 = pulse not palpable; 1 + = faint, but detectable pulse; 2 + = slightly more diminished pulse than normal; 3 + = normal pulse; and 4 + = bounding pulse.
Question 7. Write short notes on
Answer:
- Corrigan’s/Water Hammer/Collapsing Pulse And Its Causes.
- Pulsus paradoxus And Its Causes.
- Pulsus Alternans
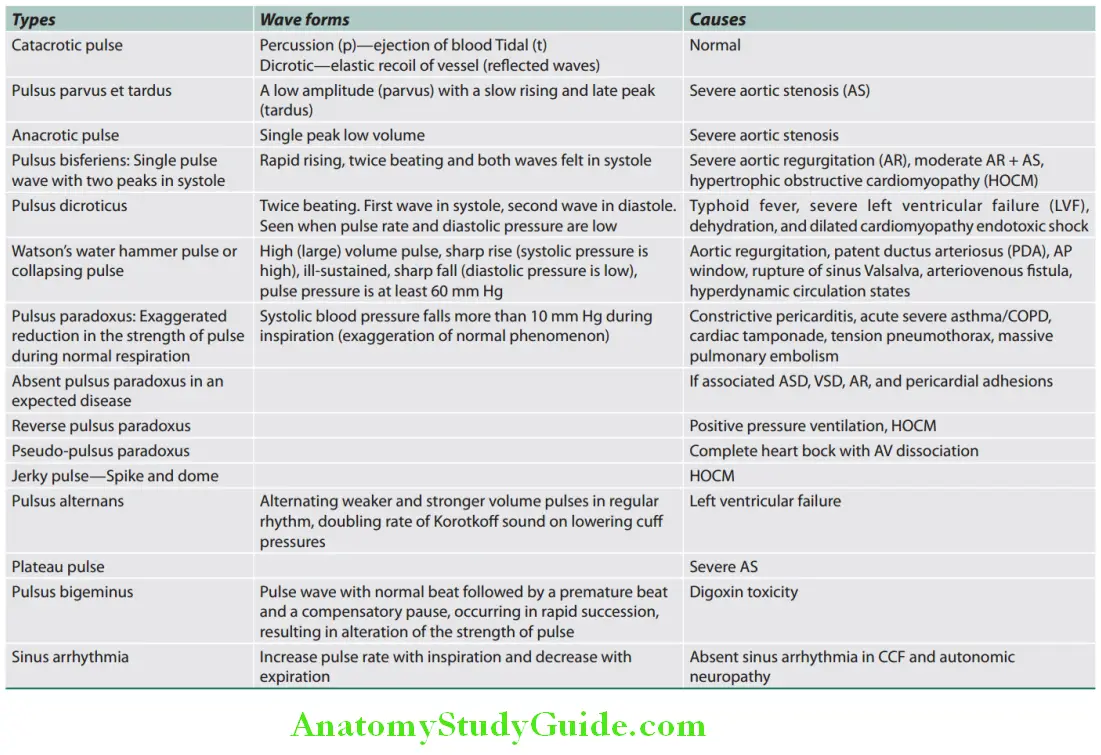
Diagrammatic appearances of various arterial waveforms are presented.
Radiofemoral delay:
- A delayed femoral pulsation compared to the right radial pulse occurs in the coarctation of the aorta.
- Demonstrated by simultaneous palpation of right radial artery and one femoral artery.
- Apico-carotid delay is seen in severe aortic stenosis where there is a delay between apical impulse and the carotid upstroke.
Reduced or absent arterial pulses:
- It indicates impaired blood flow.
- .
- Causes:
- Congenital (E.G., Coarctation Of The Aorta),
- Intrinsic Disease Of Artery (Example, Atherosclerosis, Thrombosis, Arteritis),
- Disorders With Vasospasm (Example., Raynaud’s Phenomenon),
- Extrinsic compression of blood vessels (for example, thoracic outlet syndrome, trauma, and neoplasms).
Jugular Venous Pressure:
Question 8. Write short essay/note on the jugular venous pulse (JVP) and its clinical significance.
Answer:
Definition: Jugular venous pulse is defined as the oscillating top of undulating vertical column of blood in the right internal jugular vein that faithfully reflects the pressure and volumetric changes in the right atrium that varies with all phases of cardiac cycle and respiration. JVP is the vertical height of oscillating column of blood.
Advantages of Internal Jugular Vein (IJV) vs External Jugular Vein (EJV):
- IJV is anatomically closer to and has a direct course to right atrium while EJV does not directly drain into superior vena cava.
- IJV is valveless and pulsations can be seen. EJV has valves and pulsations cannot be seen.
- EJV can become small and barely visible when there is vasoconstriction secondary to hypotension (as in congestive heart failure).
- EJV is superficial and prone to kinking.
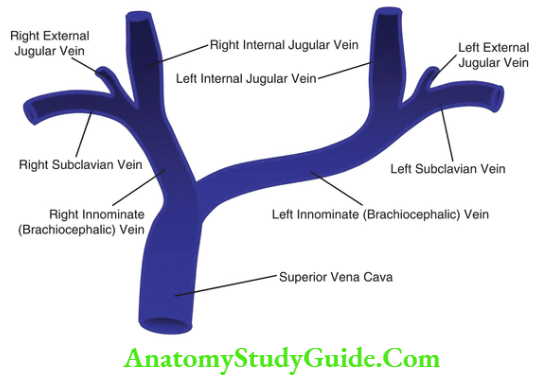
Importance of Right Internal Jugular Vein:
- Right internal jugular veins extend in an almost straight line to superior vena cava, thus favoring transmission of the hemodynamic changes from the right atrium.
- The left innominate vein is not in a straight line and may be kinked or compressed between aortic arch and sternum, by a dilated aorta, or by an aneurysm.
Technique of Measuring JVP:
- Position: Semireclining position with 45° angle between the trunk (not the neck) and the bed turn the head slightly toward left shoulder, so that the neck muscles are relaxed. Tangential light source can be put from opposite side.
- Not in sitting position: Because the upper level of venous column is below the clavicle.
- Not in supine position: Because the whole venous column moves beyond the angle of jaw into the intracranial cavity.
Identify jugular venous pulsation:
- Assure good lighting (can use tangential beam of light through torch).
- Look between the two heads of sternocleidomastoid.
- Note the upper level of pulsation, waveform, and respiratory variation.
- Do not mistake the carotid pulsations for venous pulsations.
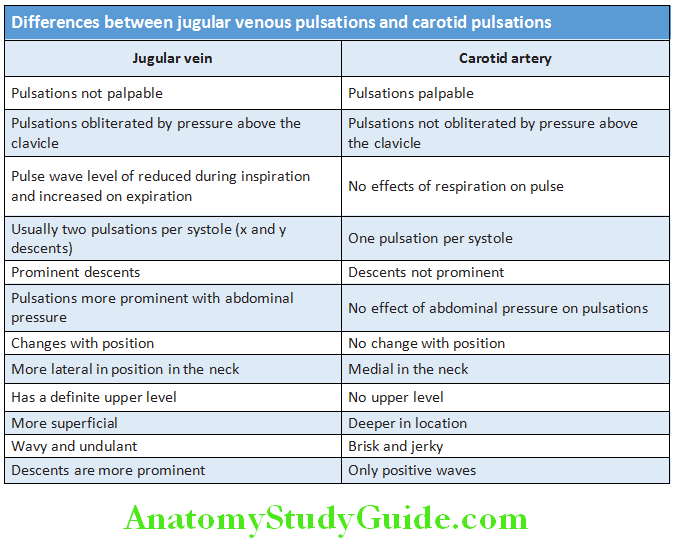
Question 9. List the differences between jugular venous pulse from carotid pulse.
Answer:
- Measurement of JVP: Measure the vertical distance (in cm) between the horizontal lines drawn from the upper level of venous pulsation and the sternal angle.
- This can be done by using two rulers—one placed horizontal to the upper level of pulsation and another taking the vertical distance of that ruler from the sternal angle.
Question 10. Method of calculation of central venous pressure from jugular venous pressure.
Answer:
- Calculate the right atrial pressure (RAP) or central venous pressure (CVP):
- Normally, the center of right atrium is 5 cm below the sternal angle at any position of the patient. Hence, 5 cm is added to the above value to obtain the RAP.
- Conversion: 1.3 cm of H 2O or blood = 1 mm Hg
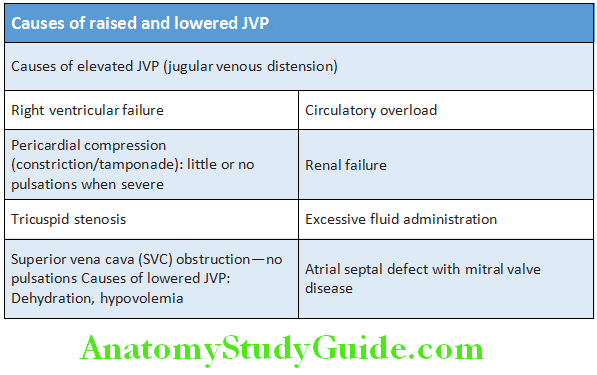
Evaluation/Interpretation of JVP:
Level:
- Normal level of JVP: From sternal angle <4 cm, from center of right atrium <9 cm. In mm Hg <7 mm Hg.
- Causes of elevated and lowered JVP.
Evaluation of jugular venous pulse (JVP):
Level:
- Waveform
- Venous Hum
- Respiratory Variation In Level And Wave Pattern
- Abdominojugular Reflux
- Liver size and pulsations.
Question 11. Write short essay/note on normal wave patterns of JVP and their variations.
Answer:
Wave pattern:
Th normal JVP consists of three ascents or positive waves (a, c and v) and two descents or negative waves (x, x’ and y):
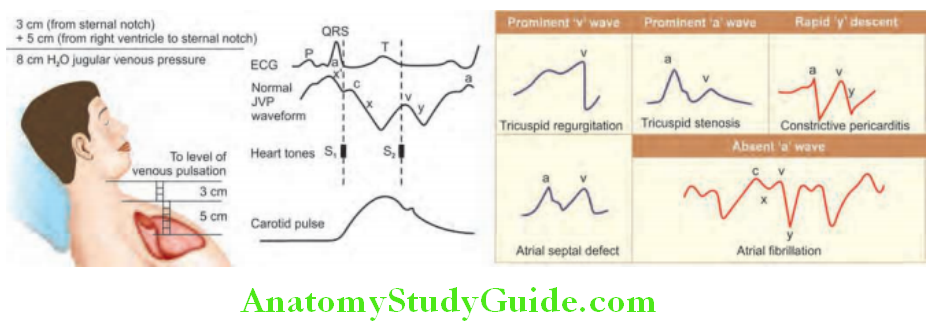
- a wave (ascent): Due to active atrial contraction leading to retrograde blood flow into neck veins.
- x wave (descent): Due to continued atrial relaxation.
- c wave: Due to impact of the carotid artery adjacent to the jugular vein and retrograde transmission of a positive wave in the right atrium. It is produced by the right ventricular systole and the bulging of the tricuspid valve into the right atrium. Not usually seen in humans.
- x’ wave (descent): Due to descent of floor of right atrium (tricuspid valve) during right ventricular systole and continued atrial relaxation.
- v wave (ascent): Due to passive atrial filling (venous filling).
- y wave (descent): Due to opening of tricuspid valve and subsequent rapid inflow of blood from right atrium into the right ventricle leading to a sudden fall in right atrial pressure.
Wave patterns in jugular venous pressure are presented.
Best way to identify the waves (ascents and descents):
Simultaneously auscultate and observe the wave pattern.
- “a” ascent: Clinically corresponds to S1 (though it actually occurs before S1 ); sharper and more prominent than “v” wave.
- “x” descent: Follows S1 ; less prominent than “y” descent.
- “c” ascent: Occurs simultaneously with carotid pulse; but never seen normally.
- “v” ascent: Coincides with S2; less prominent than “a” ascent.
- “y” descent: Follows S2; more prominent than “x” ascent.
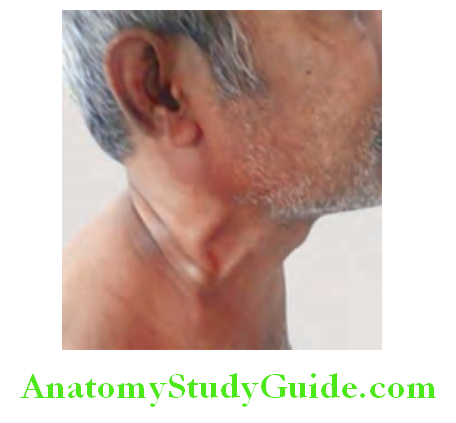
Abnormalities of wave patterns of JVP:
- Prominent “a” waves (giant a-wave or Venous Corrigan):
- Due to resistance to atrial emptying at:
- Tricuspid level: Tricuspid stenosis, right atrial tumors.
- Right ventricular level: Concentric hypertrophy due to severe pulmonary hypertension, right ventricular cardiomyopathy, severe aortic stenosis, acute pulmonary embolism, and acute tricuspid regurgitation.
- Cannon waves: Very prominent “a” waves due to atrial contraction against closed tricuspid valve.
- Regular cannon waves: Junctional rhythm, ventricular tachycardia 1:1 retrograde conduction, and isorhythmic AV dissociation.
- Irregular cannon waves: Complete heart block, ventricular tachycardia, ventricular ectopy, ventricular pacing, and classic AV dissociation.
- Rapid and regular neck pulsations, which are due to prominent and regular A waves, may be seen as a bulging in the neck, sometimes termed as “frog sign” and is most typical of re-entrant supraventricular arrhythmias, particularly AVNRT or atrioventricular re-entrant tachycardia due to a pre-excitation syndrome.
- Absent “a” waves: Atrial fibrillation (AF), post DC conversion of AF, sinoventricular conduction in hyperkalemia.
- Single wave
- “a” and “v” wave merge: Heart rate >120 beats/minute
- Early “v” wave with obliterated “x” wave: Severe chronic tricuspid regurgitation, acute tricuspid regurgitation
- Absent “x” wave (failure of atrial pressure to fall): Atrial fibrillation, severe chronic tricuspid regurgitation, acute tricuspid regurgitation, and constrictive pericarditis.
- Prominent “x” wave: Cardiac tamponade
- Prominent “v” wave: Right ventricular failure, tricuspid regurgitation, atrial septal defect with/without mitral regurgitation.
- Diminished “v” wave: Hypovolemia, venodilators
- Rapid “y” descent: Causes of prominent “v” wave, constrictive pericarditis (Friedrich’s sign).
- Slow “y” descent: Tricuspid stenosis, pericardial tamponade, and tension pneumothorax.
- c-v wave with prominent “y” descent: Tricuspid regurgitation (lateral ear lobe pulsations—Lancisi’s sign).
- Steeply rising “h” wave: Restrictive cardiomyopathy, constrictive pericarditis, and right ventricular infarction.
- Equal “a” and “v” wave (M Pattern): Atrial septal defect.
Venous hum (Pontain’s murmur):
- Continuous bruit over neck veins (normally noiseless) due to increased velocity of blood flow or decreased viscosity of blood.
- Best heard with the bell of the stethoscope over the right supraclavicular fossa/root of neck, patient in standing position.
- Causes:
- Physiological: Children, pregnancy
- Pathological: Hyperkinetic states, anemia (indicates chronic compensated severe anemia), thyrotoxicosis, beriberi, and intracranial AV fistula. Indicates chronic compensated severe anemia.
Respiratory Variation of JVP:
- Normal: Venous column in IJV rises during expiration and falls during inspiration.
- Reason:
- During inspiration venous return to the right side of the heart increases due to increased negative thoracic pressure. However, this is accommodated by the inspiratory decrease in pulmonary vascular resistance. As a result, pulmonary artery, right ventricle, and right atrial pressures fall in spite of increased venous return.
- During expiration, due to increased positive intrathoracic pressure, pulmonary circulation is compressed by the thoracic cage resulting in increased pulmonary resistance and pressure.
Kussmaul’s Sign:
Question 12. Write short essay/note on Kussmaul’s sign
Answer:
- Normally, during inspiration there will be decrease in the height of jugular venous pulsation and drop in jugular venous pressure (respirophasic changes).
- Kussmaul’s sign is paradoxical rise in JVP during inspiration.
- Causes: Constrictive pericarditis, right ventricular infarction, restrictive cardiomyopathy, severe right-sided heart failure, and acute severe asthma. Rarely can be seen in cardiac tamponade, tricuspid stenosis, and severe tricuspid regurgitation.
- Reason: Increased venous return in inspiration cannot be accommodated by the heart in the above-mentioned conditions leading to a translation of the venous blood back through the SVC and presents as distended jugular veins even during inspiration.
Abdominojugular/Hepatojugular Reflx:
Question 13. Write a short note on abdominojugular/hepatojugular reflux test and its clinical significance.
Answer:
Technique: When pressure is applied over the liver by pressing firmly (40 mm Hg) over the abdomen for around 30 seconds, the venous pressure gets exaggerated initially due to increased venous return. Later the myocardium accommodates the extravenous return and the level falls. Normally within 2–3 cardiac cycles, i.e., within 3 seconds.
Normal response: Upper level of jugular venous pulsation moves upward by less than 3 cm and then falls down within 5 seconds even when the pressure is continued.
Abdominojugular Signifiance:
Positive test: Rise in JVP (more than 3 cm) for >10 seconds of firm midabdominal compression.
Early cardiac failure: First sign of right heart failure (RHF).
False positive: Valsalva (abdominal guarding), fluid overload
False negative: SVC/IVC obstruction, Budd–Chiari syndrome where there is no rise in JVP. This test also helps to differentiate venous pulsation from the arterial pulsation.
Square Root Sign:
It is found in constrictive pericarditis on cardiac catheterization to note pressure tracings. A rapid halt of ventricular filling occurs as the ventricular wall is impeded by the abnormal pericardium causing an abrupt rise in LV pressure. This has been described as the square root sign.
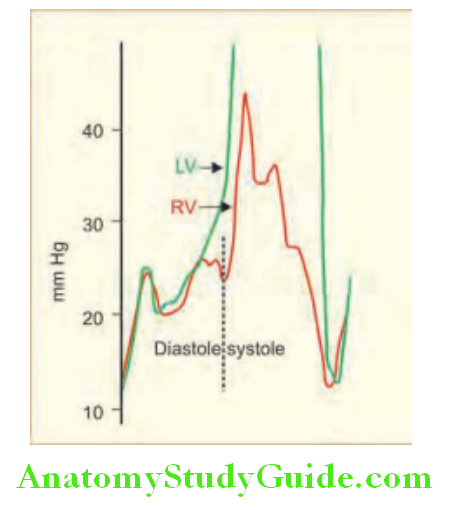
Apical Impulse:
Question 14. Write a short note on apical impulse.
Answer:
Apical Impulse Definition: It is the outermost and lowermost point of maximum impulse (PMI) in early systole, which imparts a perpendicular gentle thrust to a palpating finger, followed by a slight medial retraction in the late systole.
Cause of normal apical impulse: Anterior and counter clock-wise rotation of LV due to isovolumic contraction during early systole and medical retraction due to clock-wise rotation of the LV during late systole. Characteristics of normal apical impulse
Characteristics of normal apical impulse:
- Location: Left 5th ICS at or around half an inch medial to mid-clavicular line and <10 cm from mid-sternal line
- Extent: <2.5 cm in diameter which is one ICS
- Duration: <50% of systole
- Mildly tapping in character
Abnormal Apical Impulse:
Absent: Dilated cardiomyopathy, pericardial effusion, behind the rib, dextrocardia, obesity, COPD, left pleural effusion
Tapping: Mitral stenosis (palpable S1 —closing snap)
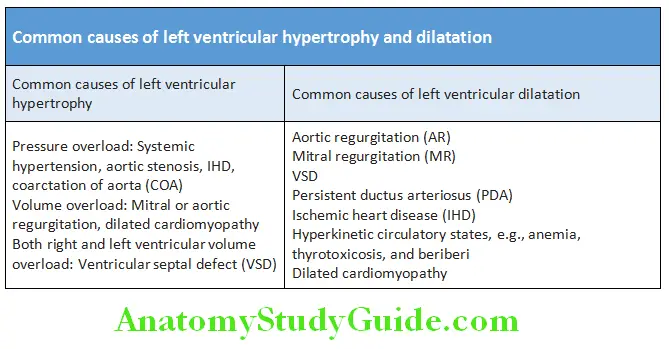
Hyperdynamic: Increased in amplitude, duration is >1/3 to <2/3 of systole, occupies more than one intercostal space. Occurs in LV volume overload [AR (aortic regurgitation), MR (mitral regurgitation), VSD (ventricular septal defect), PDA (patent ductus arteriosus), and high output states]
Heaving: Increase in amplitude, duration is >2/3 of systole and occupies more than one intercostal space. Occurs in LV pressure overload [AS (aortic stenosis), HTN (hypertension), coarctation of aorta]
Diffuse: Occupying more than 1 ICS. Occurs in LV aneurysm, LV dilatation as in AR
Double: Hypertrophic obstructive cardiomyopathy (HOCM), AS with AR, left dyssynergy (LBBB), LV aneurysm
Triple or quadruple: HOCM
Retractile: Severe TR (tricuspid regurgitation)
Medial apical retraction: Left ventricular enlargement
Lateral apical retraction: Right ventricular enlargement
Cheyne–Stokes Breathing:
Question 15. Write short essay on mechanism, causes, consequences, and management of Cheyne–Stokes breathing.
Answer:
Cheyne–Stokes Breathing Definition: This is a cyclical pattern of respiration due to impaired responsiveness of the respiratory center to carbon dioxide.
It is characterized by gradual increases and decreases in respiration. It has two alternative periods, namely
- Rapid Deep Respiration Called Hyperpnea And
- Complete stoppage of respiration called apnea.
Cheyne–Stokes Breathing Mechanism:
- Spontaneous rhythmic activity of breathing is abolished when there is anoxemia. Consequent apnea causes accumulation of carbon dioxide.
- Hypercapnia stimulates respiratory centers → produces hyperventilation → leads to reduced carbon dioxide → causes depression of the respiratory center → resulting in apnea. The cycle is repeated.

Conditions Associated with Cheyne–Stokes Breathing:
- Physiological conditions: During deep sleep, high altitude, and ewborn babies
- Pathological conditions: Severe heart (left ventricular) failure, uremia, chronic hypoxia, diffuse cerebral atherosclerosis, stroke, head injury and hemorrhage, increased intracranial pressure, severe pneumonia, and narcotic drug (e.g., barbiturates, opiates) poisoning.
Heart Sounds:
Relative, brief, auditory vibrations of variable intensity, frequency, and quality.
First Heart Sound (S1):
Question 16. What is the mechanism of first heart sound? Write short note on the variations in first heart sound.
Answer:
First Heart Sound Mechanism:
During systole the atrioventricular (AV), i.e., mitral and tricuspid valves, closes and blood tries to enter the atrium and lead to back bulging of the AV valves into the respective atria. But the taut (stretched or pulled tight) chordae tendineae stop the back bulging and causes the blood to flow forward. This will cause vibration of the valves, blood, and the walls of the ventricles.
TFirst Heart Sound iming:
- Just precedes carotid upstroke.
- S1 will appear to initiate the outward LV thrust of apex beat.
First Heart Sound Characteristics:
- Medium-to-high frequency but lower pitch than S2.
- Q-M1 60 ms, Q-T1 90 ms, M1-T1 30 ms.
- First heart sound has two components:
- Mitral component (M1) due to mitral valve closure followed by
- Tricuspid component (T1) due to tricuspid valve closure.
- S1 is best heard at the apex. It is best heard with the diaphragm of stethoscope.
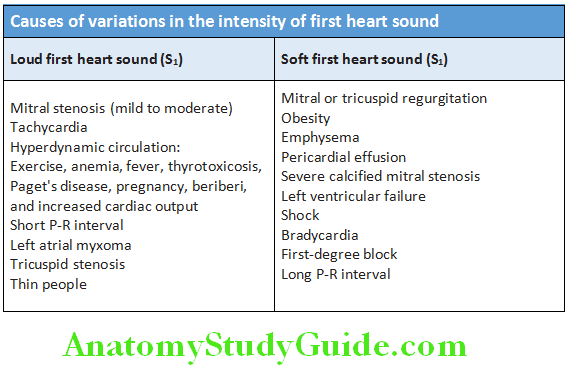
First Heart Sound Intensity:
Determined by structural integrity of mitral valve, position of AV valves at time of ventricular contraction, integrity of isovolumetric systole, heart rate, P-R interval, and myocardial contractility.
Abnormalities of S1:
Variation in the intensity of first heart sound: Atrial fibrillation, complete heart block (CHB), ventricular tachycardia, atrial flutter with varying block, atrial tachycardia with varying block, and ventricular ectopics.
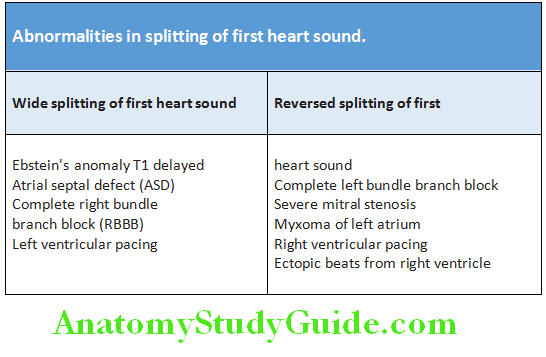
Splitting of S1: Abnormalities in splitting of first heart sound are presented.
Second Heart Sound (S2):
Question 17. What is the mechanism of second heart sound? Write short note on the variations in second heart sound.
Answer:
It is due to the closure of aortic and pulmonary valve.
Second Heart Sound Characteristics:
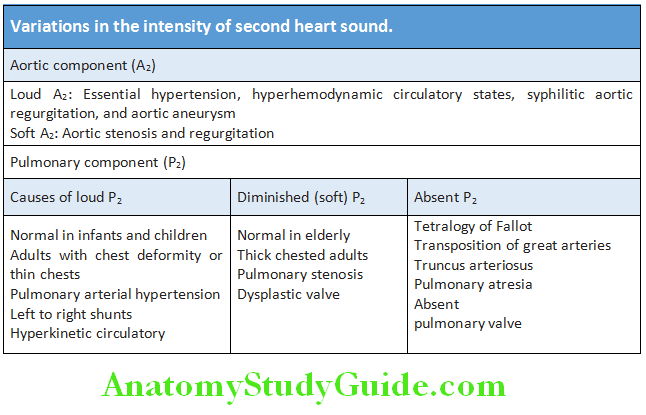
- It has two components: aortic component (A2) due to aortic valve closure, followed by the pulmonary component (P2) due to pulmonary valve closure.
- Loudness of A2 or P2 is directly proportional to the pressures in aorta or pulmonary artery at the beginning of diastole, respectively.
- The second heart sound is medium to high pitched (higher frequency) because:
- The semilunar valves are more taut
- The great elastic coefficient of the taut arteries provides the principle vibrations of the second heart sound. P2 only heard at second left intercostal space. Loudest sound in pulmonary area is A2.
- A2 is audible at region of right second intercostal space, left parasternal space, and apex.
- Normal splitting:
- Splitting during inspiration: During inspiration, the aortic valve closes early than pulmonary valve and produces a physiological inspiratory splitting of second heart sound. Split is audible in second and third intercostal space and amplitude of A2 > P2.
- Normally, during expiration both aortic and pulmonary valves close almost simultaneously and produce a single expiratory second heart sound.
- Frequency of both A2 and P2 components are same.
- Hangout time/interval: The interval between the pressure crossover point and the incisura (the onset of A2 or P2) has been termed “hangout time”. During inspiration, pulmonary vascular impedance declines with a further increase in the pulmonary hangout time, which is the mechanism for inspiratory splitting of S2.
Abnormalities of Second Heart Sound (S2):
- Absent S2: Old age as in calcific aortic stenosis (due to absence of A2) or chronic emphysema.
- Single second heart sound (S2): May be either due to absent A2 (e.g., severe aortic stenosis or atresia) or absent P2 (e.g., severe pulmonary stenosis or atresia) and tetralogy of Fallot.
- Variation in the intensity S2
- Splitting of S2: Physiological splitting of S2 is normally found in children and young adults during inspiration.
- Normal interval is A2-P2 30 ms and A2-OS 30–150 ms.
- Wide split S2: It may be variable or fixed
- ASD with variable split S2: Sinus venosus type of ASD or ASD with atrial fibrillation.
- Reversed (paradoxical) splitting S2: May be due to early P2 or delayed A2
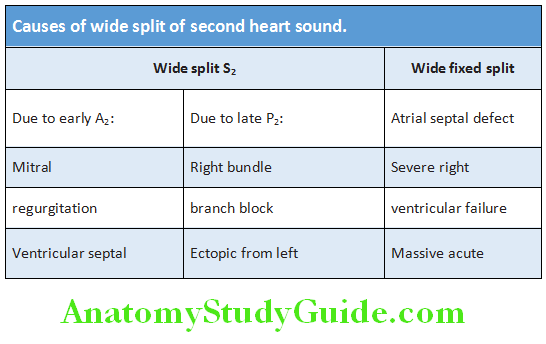
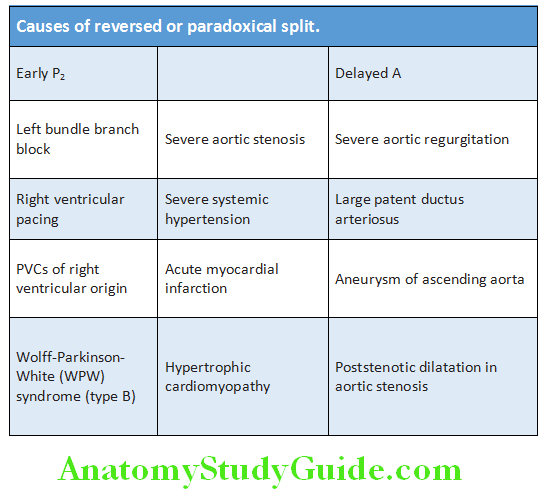
- Pseudo-paradoxical split: Normally S2 split in expiration and inspiration. But during inspiration patients with muffled P2, so that single S2 will be heard in inspiration.
- Narrow splitting: Aging, artifactual muffling of P2, severe pulmonary arterial hypertension, and murmur obscuration.
Third Heart Sound:
Question 18. Write short note on the significance of third heart sound.
Answer:
- Third heart sound (S3) is also called as protodiastolic sound/ventricular gallop.
- S3 is a low-pitched early diastolic sound produced due to rapid ventricular filling immediately after opening of the
atrioventricular valves. - S3 best heard with the bell of stethoscope at the apex. It coincides with rapid ventricular filling. It occurs 0.12–0.18 seconds after S3 and has low frequency and low intensity.
Causes of Third Heart Sound:
- A third heart sound is a normal finding in children, in young adults, and during pregnancy.
- A third heart sound is usually pathological after the age of 40 years. In heart failure S3 occurs with a tachycardia and S1 and S2 are quiet (lub-da-dub).

Fourth Heart Sound:
Question 19. Write short note on significance of fourth heart sound.
Answer:
Fourth heart sound (S4) is less common and is also called presystolic/atrial gallop. It occurs just before the first sound (da-lub-dub). It occurs 0.11 seconds prior to S1.
It is soft and low-pitched, best heard with the bell of the stethoscope at the apex.
It is always pathological and is produced by a rapid (forceful) empting of the atrium into the noncompliant or stiff ventricle. It cannot occur when there is atrial fibrillation.
Third and a fourth heart sound causes a “triple” or “gallop” rhythm. S4 may be confused with spilt S1. Firm pressure by the diaphragm of stethoscope eliminates S4 but not split S1.
Causes of fourth heart sound:
Left sided S4:
- Systemic hypertension with left ventricular hypertrophy
- Hypertrophic cardiomyopathy
- Ischemic heart disease (especially acute myocardial infarction)
- Acute mitral regurgitation
- Anemia, thyrotoxicosis, and AV fistula
Right sided S4:
Right ventricular hypertrophy due to pulmonary hypertension, pulmonary stenosis
Causes of Fourth Heart Sound:
Summation Gallop:
Question 20. Write short note on summation gallop.
Answer:
- Summation is the presence of S1, S2 merging with S3 and S4. It occurs when both S3 and S4 are present in a patient with tachycardia.
- Shortening of diastole causes joining of the two sounds (S3 and S4) and produce a single loud sound.
Pericardial Friction Rub:
Question 21. Write short note on pericardial rub.
Answer:
- It is the sound produced due to sliding (apposition) of the two inflamed layers (visceral and parietal pericardium) of the pericardium.
- Phases:
- It is triphasic:
- Midsystolic
- Mid-Diastolic
- Presystolic.
- Character: It is scratchy, grating, leathery, or creaking in character. Its intensity vary over time, and with the position of the patient.
- Best heard: With diaphragm of stethoscope on the left sternal boarder (3rd and 4th space) leaning over at the end of expiration. It may be audible over any part of the precordium but is often localized.
- Confused with Hamman’s sign in post-open heart surgery (crunch sound from mediastinal air).
- A pleuropericardial rub is a similar sound that occurs in time with the cardiac cycle but is also influenced by respiration and is pleural in origin. Occasionally, a “crunching” noise can be heard caused by air in the pericardium (pneumopericardium).
Cardiac Sounds on Auscultation:
Ejection Sound/Click:
Usually sharp, high frequency sound audible immediately after S1.
Mid-Late Systolic Sounds:
- Click, high frequency sound found in mitral valve prolapse.
- Occurs earlier with Valsalva maneuver or squatting to standing.
Causes of ejection sound/click:
- Aortic valve: Aortic stenosis, bicuspid aortic valve
- Pulmonary valve: Pulmonic stenosis, vary with respirations
- Prosthetic valves: Mechanical, not bioprosthetic
Early Diastolic Sounds:
Question 22. Write short note on opening snap.
Answer:
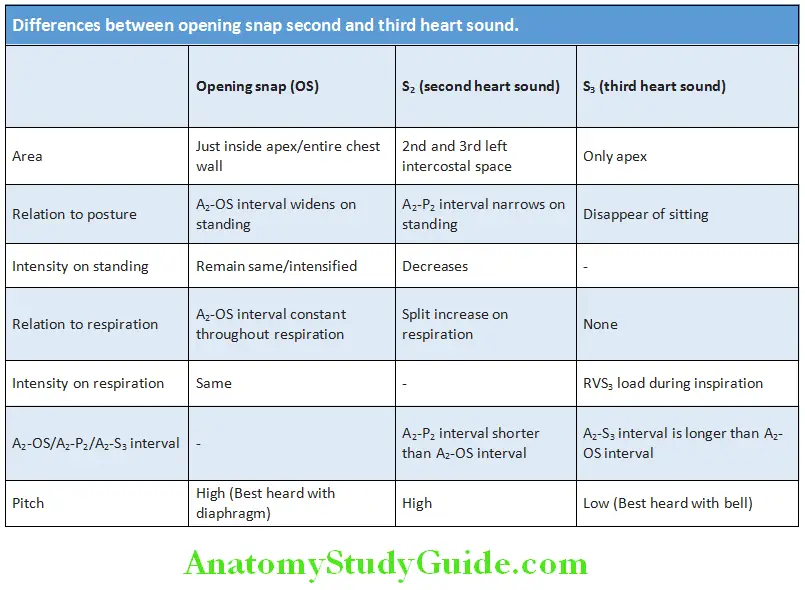
Opening snap of mitral stenosis (MS):
- It is caused by the opening of atrioventricular valve.
- Characteristics: High-pitched sound audible at the apex in left lateral decubitus position. 0.04–0.12 sec after A2 (S3 occurs 0.12 sec after A2). Occurs after S2, before S3.
- Severe mitral stenosis is associated with short A2-OS interval and softer OS or absent OS.
Mechanism of Opening Snap (OS):
- Stenotic anterior mitral valve leaflet suddenly bulging download into the left ventricular cavity like a dome, with a snapping sound when the mitral valve is rapidly opened during diastole. So, OS is audible only if anterior mitral leaflet of mitral valve is mobile.
- OS occurs when movement of AMV suddenly stops, at point when LVP drops below that of LAP.
Differences between opening snap second and third heart sound:
Early Diastolic Sounds Murmurs:
- Sudden deceleration of blood produces heart sounds. Heart murmurs are produced by turbulent flow (Raynold’s number >2,000) across an abnormal valve, septal defect or outflow obstruction, or by increased volume or velocity of flow through a normal valve.
- Murmurs may occur in a healthy heart. These “innocent” murmurs occur when stroke volume is increased, e.g., during pregnancy, and in athletes with resting bradycardia or children with fever
Mechanism of murmurs:
- Blood viscosity: Increased or decreased blood velocity
- Valve: Narrowed or incompetent, organic or relative
- Abnormal connection
- Vibration of loose structure
- Diameter of vessel increased or decreased
Features to be Observed in Murmur:
- Timing: By simultaneous palpation of the carotid arterial pulse and note whether systolic, diastolic, and continuous.
- Shape: Crescendo (grows louder), decrescendo, crescendodecrescendo, and plateau
- Intensity:
- Systolic murmurs are graded on a 6 point scale (Levine and Freeman)
- Diastolic murmurs are usually not graded but can be described as
- Very Soft
- Soft
- Loud with/without thrill.
- Duration:
- Location of maximum intensity depends on the site where the murmur originates.
- Character blowing, harsh, rumbling, and musical
- Pitch high, medium, low depending on the velocity of the jet
- Radiation/conduction:
- Reflects the intensity of the murmur and the direction of blood flow
- Mitral regurgitation murmur (PSM) radiates to axilla
- Aortic stenosis murmur (ESM) conducts to the carotid.
- Variation with respiration/position/other maneuvers
- Best heard with bell or diaphragm of the stethoscope.
Grading of systolic murmurs (Freeman and Levine):
- Grade 1 = very faint
- Grade 2 = quiet but heard immediately
- Grade 3 = moderately loud
- Grade 4 = loud with thrill
- Grade 5 = heard with stethoscope partly off the chest, thrill present
- Grade 6 = no stethoscope needed, thrill present
Systolic Murmurs:
Question 23. Write short essay/note on differential diagnosis of ejection systolic murmurs (ESM). List the causes of systolic murmurs.
Answer:
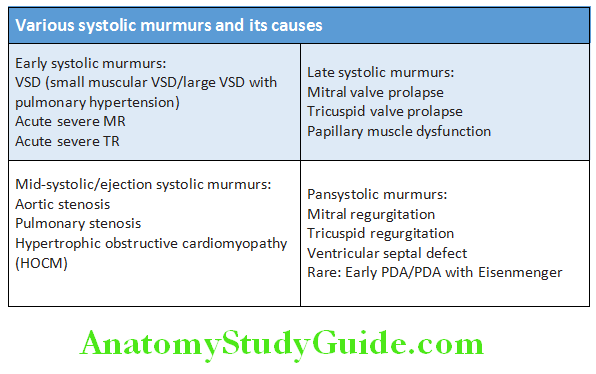
Ejection Systolic (Mid-systolic) Murmurs:
Ejection systolic (mid-systole) murmurs occur when there is ventricular outflow tract obstruction. It begins shortly after the first heart sound (S1) and ends before the second heart sound with a crescendo–decrescendo or diamond-shaped pattern, reflecting the changing velocity of blood flow.
Various Systolic Murmurs and its Causes:
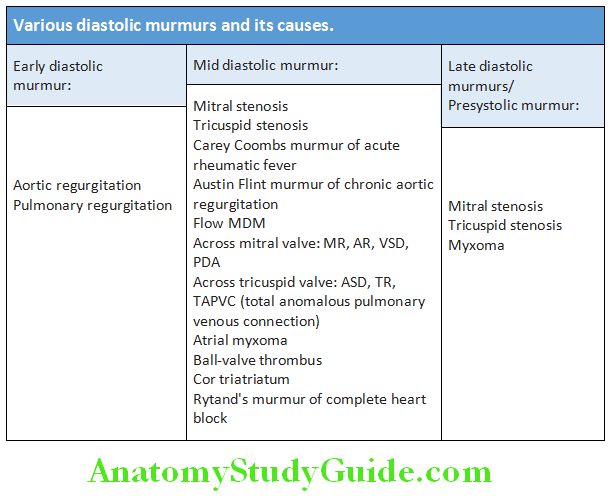
Question 24. Write short note on causes of pansystolic murmur.
(or)
Write short note on causes of mid-diastolic murmurs.
Answer:
Various Diastolic Murmurs and Its Causes:
Continuous Murmurs:
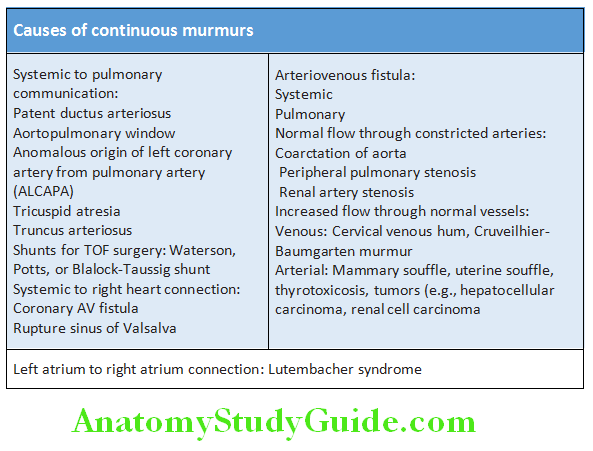
Question 25. Write short note on the common causes of continuous murmurs of heart.
Answer:
The continuous murmur is a murmur that begins in systole and continues without interruption, encompassing the second sound, throughout diastole or part of diastole.
Causes of Continuous Murmurs:
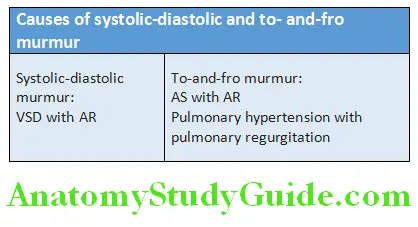
Systolic-Diastolic Murmur/To-and-Fro Murmur:
- The presence of a systolic-diastolic murmur (systolic murmur and diastolic murmur), so called a to-and-fro murmur, is not a continuous murmur.
- It is separating the two murmurs through a small “silence”.
- It involves two components: A systolic one, in which the blood flows in one direction, and a diastolic one in which the blood flows in the opposite direction.
- In contrast to true continuous murmur, the blood flows in the same direction in both systole and diastole.
- To-and-fro murmurs are two murmurs that occur through a single channel.
- Causes of systolic-diastolic murmur are presented in Table
Innocent Murmurs:
Question 26. Write short note on innocent murmurs.
Answer:
Characteristics: Short, systolic (rarely continuous) soft murmur. Normal heart sounds and no hemodynamic abnormalities.
Examples of innocent murmurs:
Venous hum (jugular venous hum; cervical venous hum, Pontian’s murmur).
Examples of innocent murmurs:
Systolic:
- Vibratory systolic murmur (Still’s murmur)
- Pulmonic systolic murmur (pulmonary trunk)
- Mammary soufflé
- Peripheral pulmonic systolic murmur (pulmonary branches)
- Supraclavicular or brachiocephalic systolic murmur
- Aortic systolic murmur
- Still’s murmur: Medium frequency, vibratory, originating from leaflets of pulmonary valve
Continuous:
- Venous hum
- Continuous mammary soufflé
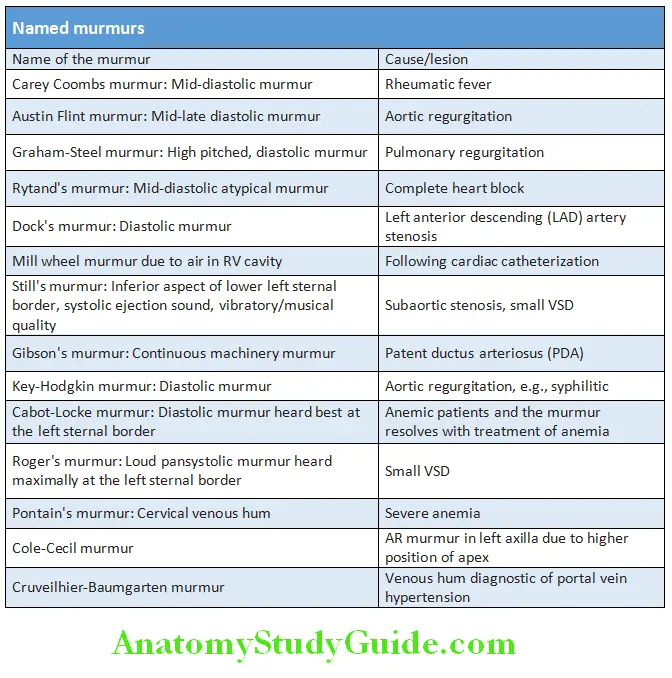
Named murmurs and its causes are listed in Table:
Question 27. Write short note on mammary soufflé.
Answer:
Mammary soufflé is an innocent systolic or continuous cardiac murmur (probably of arterial origin) heard during late pregnancy or in the early postpartum period. It is to be differentiated from pathologic lesions and is unaffected by the Valsalva maneuver.
Changing Murmurs:
Murmurs which change in character or intensity from moment to moment
Changing murmurs:
- Carey Coombs murmur
- Infective endocarditis
- Atrial thrombus
- Atrial myxomas
Dynamic Auscultation:
Question 28. Write short essay/note on dynamic auscultation in cardiac diseases.
(or)
Write short essay/note on maneuvers useful in differentiating murmurs due to various cardiac diseases.
Answer:
This is a technique of altering circulatory dynamics by means of a variety of physiological and pharmacological maneuvers and determining their effects on heart sounds and murmurs.
Interventions most commonly employed are:
- Respiration
- Postural changes
- Valsalva maneuver
- Isometric exercise
- Post-premature ventricular contractions (PVC)
- Vasoactive agents, e.g., amyl nitrite, methoxamine, and phenylephrine.
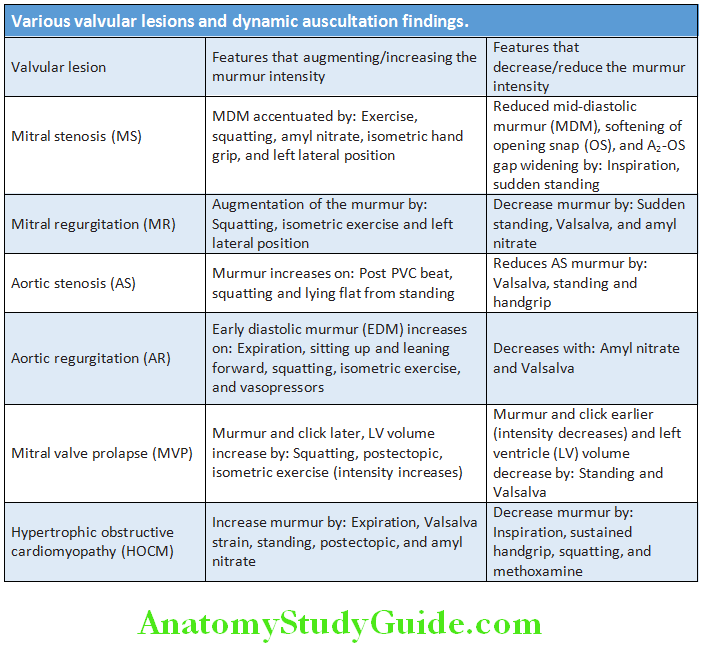
Various valvular lesions and dynamic auscultation findings are presented
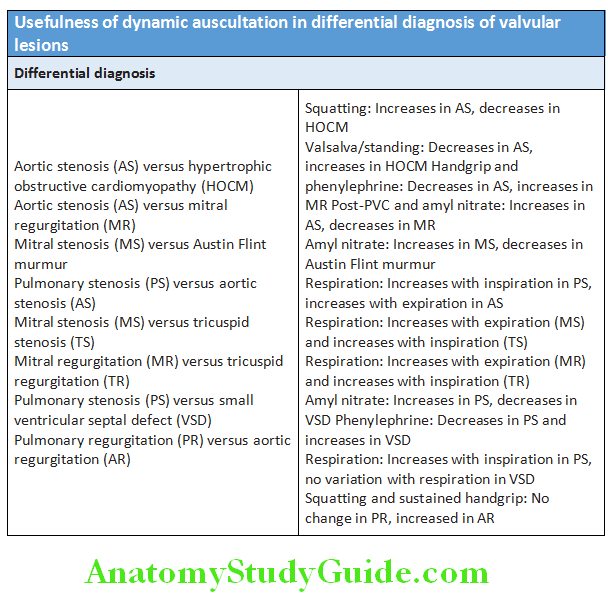
Usefulness of dynamic auscultation in differential diagnosis of valvular lesions
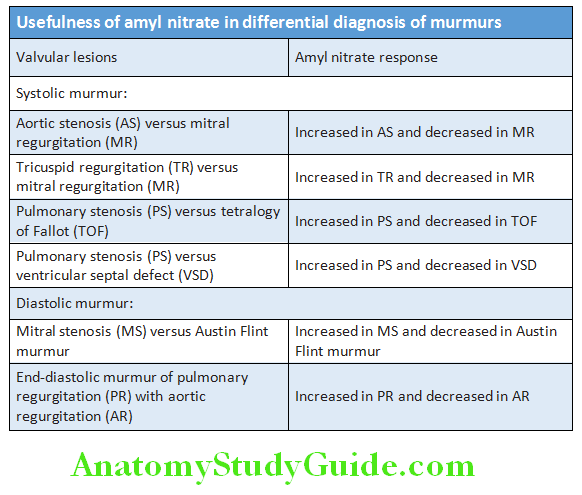
Usefulness of amyl nitrate in differential diagnosis of murmurs is depicted in Table.
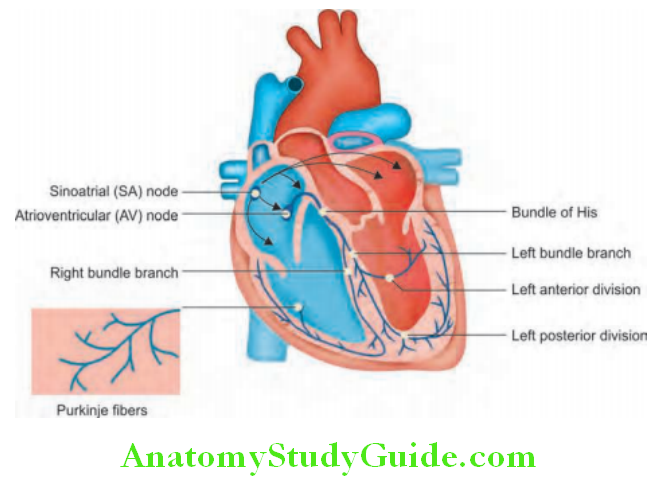
Conduction System Of The Heart:
Question 29. Write short essay/note on the conduction system of the heart.
Answer:
An electrical discharge from the sinoatrial (sinus) node initiates the normal heart beat. It is then sequentially depolarized in the atria followed by ventricles as it passes through specialized conducting tissue.
- Sinus node (SA node): It is located in the lateral and epicardial aspect where the superior vena cava joins the right atrium. It is a natural pacemaker of the heart and controls the rate and rhythm of the heart (rate of 60–100 beats/minute). It has the fastest inherent discharge.
- Atrioventricular (AV) node: The impulse from the SA node spreads through the atrial musculature and down to the atrioventricular (AV) node that is situated above the tricuspid valve. Passage via the AV node is relatively slow, and is responsible for the normal physiological delay in ventricular depolarization. AV node functions as a back-up pacemaker with an intrinsic rateof 40–60 beats/minute.
- His bundle and Purkinje fibers: The impulse then travels downward to the bundle of His and through its branches (right bundle branch and left bundle branch) to the Purkinje network of fibers that convey the impulse to the ventricular endocardium and then epicardium. Potential pacemaking properties also exist in the cells of the AV node, bundle of His, and Purkinje fibers. Ventricular cells also act a back-up pacemaker with an intrinsic rate of 20–45 beats/minute.
Electrocardiogram:
Question 30. Write short essay on analysis of an electrocardiogram (ECG).
(Or)
Write short note on sinus tachycardia.
Answer:
Sinus Rhythm:
Waveforms and Intervals:
Electrocardiogram is the recording of sequential sum of depolarization and repolarization of all myocardial cells. The electrical depolarization of myocardial tissue produces a small dipole current. It can be detected by electrode pairs on the body surface.
These signals are amplified and either printed on special graph paper or displayed on a monitor.
- Depolarization is the sudden change within myocardium, during which it undergoes a dramatic electrical change. Entire myocardium is depolarized in a coordinated manner.
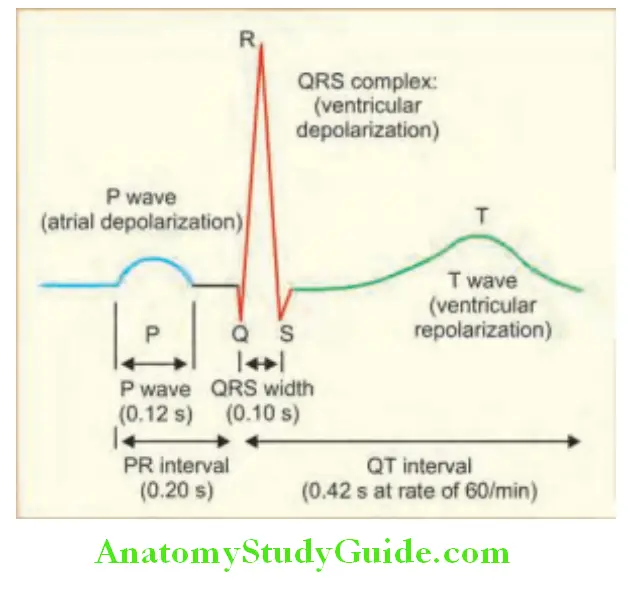
- P wave: The ECG waveforms are labeled alphabetically beginning with the P wave (represents atrial depolarization).
- The SA node triggers atrial depolarization, producing a beginning P wave.
- QRS complex: Depolarization slowly spreads through the AV node, which produces a depolarization wave which is too small to be detected from the body surface. The bundle of His, bundle branches, and Purkinje system are then activated, initiating ventricular myocardial depolarization. During this, QRS complex is produced that represents the duration of ventricular depolarization. Because of the larger size of the ventricular muscle mass than that of the atria, the QRS complex is larger than the P wave. Normal value is 100–110 ms or less. The QRS complex is subdivided into specific deflections or waves.
- Q wave: If the initial QRS depletion in a particular lead is negative, it is termed a Q wave, indicates septal depolarization.
- R wave: The first positive deflection is termed an R wave.
- S wave: A negative deflection after an R wave is an S wave.
- Subsequent positive or negative waves are labeled R′ (R prime) and S′ (S prime), respectively.
- Lowercase letters (qrs) are used for waves of relatively small amplitude. An entirely negative QRS complex is termed a QS wave.
- PR interval: It is the interval between the onset of the P wave and the onset of the QRS complex. Normal value is 120200 ms. It largely reflects the duration of AV nodal conduction between atrial and ventricular depolarization.
- Repolarization is the restoration of the electrical polarity of myocardial muscle.
- Repolarization is a slower process and spreads from the epicardium to the endocardium.
- Atrial repolarization does not produce a detectable signal (too low in amplitude) whereas ventricular repolarization produces the T wave. However, atrial repolarization may become apparent in conditions, such as acute pericarditis and atrial infarction.
- QT interval: It represents the total duration of both ventricular depolarization and repolarization. It varies inversely with the heart rate.
- ST-T-U complex: It consists of ST segment, T wave, and U wave and is due to ventricular repolarization. The J point represents the junction between the end of the QRS complex and the beginning of the ST segment.
- U wave: Small, rounded, upright wave following T wave. Most easily seen with a slow heart rate. Indicates repolarization of Purkinje fibers.
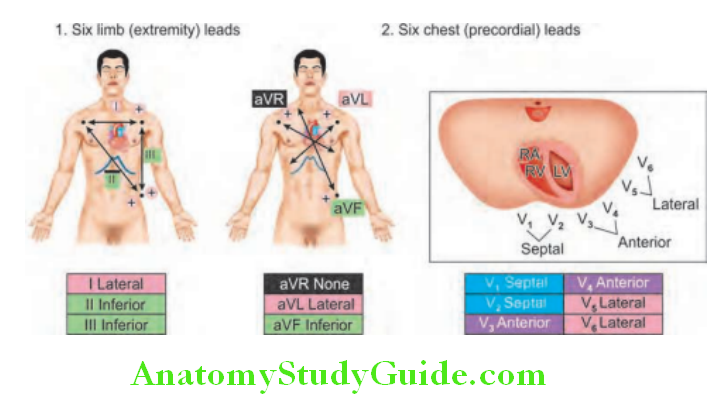
ECG Leads:
The standard 12–lead ECG records the difference in potential between ten physical electrodes placed on the surface of the body. The term twelve “leads” of the ECG is for twelve number of recordings made from pairs or sets of these electrodes.
Type of Leads:
The ECG leads are divided into two groups:
- Six limb (extremity) leads
- Six chest (precordial) leads.
The limb leads record potentials transmitted onto the frontal plane, and the chest leads record potentials transmitted onto the horizontal plane.
Reading 12-lead ECGs:
The best way to read 12-lead ECGs is to develop a step-by-step approach
Steps in reading ECG:
- Calculate rate
- Determine rhythm
- Determine QRS axis
- Check individual waves
- Calculate intervals
- Assess for hypertrophy
- Look for evidence of infarction/dyselectrolytemia/drug effects
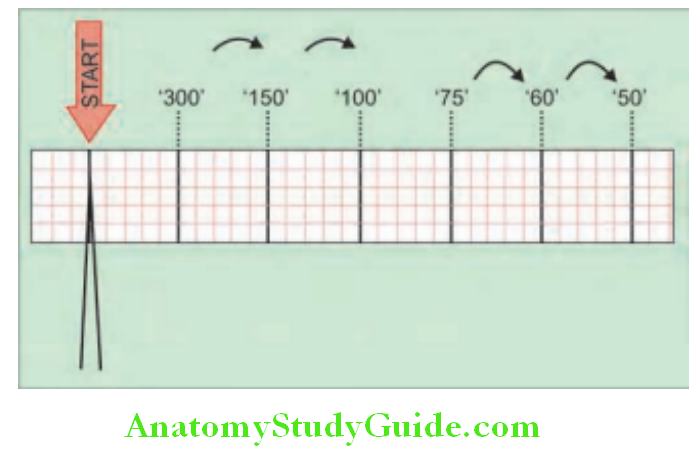
Determining the Heart Rate:
The ECG is normally recorded at a speed of 25 mm/sec. Each small, square, in the graph is 1 mm and represents 0.04 seconds and big boxes with heavier lines represent 0.20 s (200 ms).
Rule of 300/1,500: For regular rhythms, count the number of “big boxes” between two QRS complexes, and divide this into 300. The heart rate (beats per minute) can also be computed readily from the interbeat [R-number of small (0.04 s) units into 1,500].
6-second rule: For irregular rhythms, ECG records 6 seconds of rhythm per page, count the number of beats present on the ECG, multiply by 10.
Variations of heart rate and its causes:

Step 2: Determine Regularity:
- Look at the R-R distances (using a caliper or markings on a paper).
- Regular (are they equidistant apart)? Occasionally irregular? Regularly irregular?
- Irregularly irregular-atrial fibrillation.
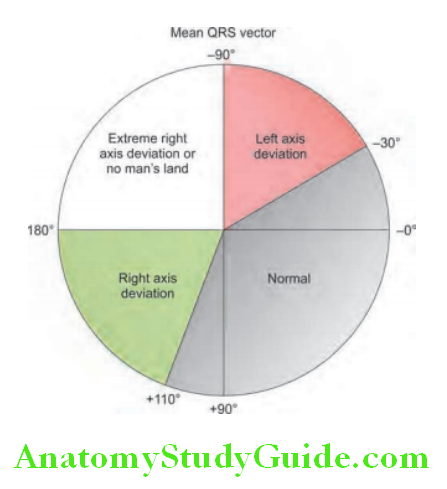
Step 3: Determining the Axis:
- Normal QRS axis from –30° to +110°, –30° to –90° is referred to as a left axis deviation (LAD), +110° to +180° is referred to as a right axis deviation (RAD) and –180° to –90° is referred as north-west axis/extreme axis/axis in no man’s land.
- QRS complex in leads I and aVF determine if they are predominantly positive or negative. The combination should place the axis into one of the 4 quadrants above.
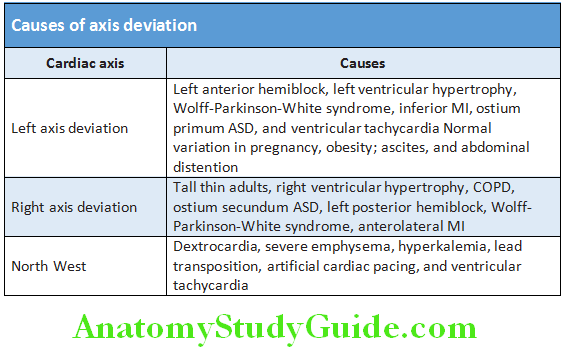
- Various causes of axis deviation.
Step 4: Check Individual Waves:
- Assess P waves
- Normal: Always positive in lead I and II, always negative in lead aVR. Commonly biphasic in lead V1 and best seen in leads <2.5 small squares in duration and <2.5 small squares in amplitude.
Step 5: Calculate Intervals
- PR interval: Normal is 0.12–0.20 seconds.
- Long PR interval may indicate heart block
- Short PR interval: Tachycardia and pre-excitation syndromes (e.g., Lown–Ganong–Levine syndrome, Wolff–ParkinsonWhite syndrome).

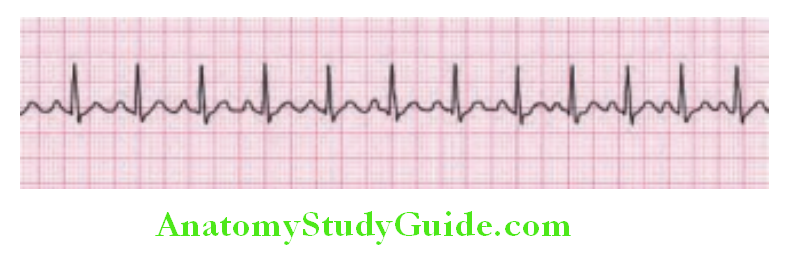
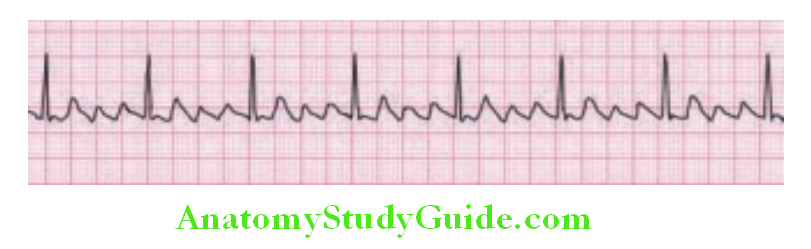

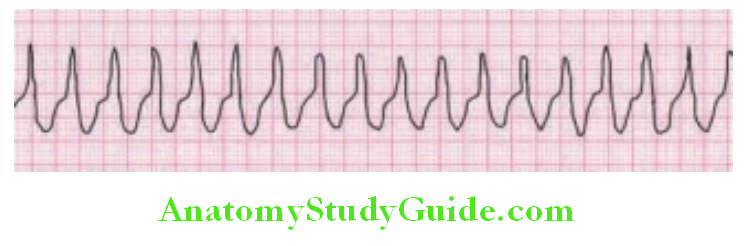



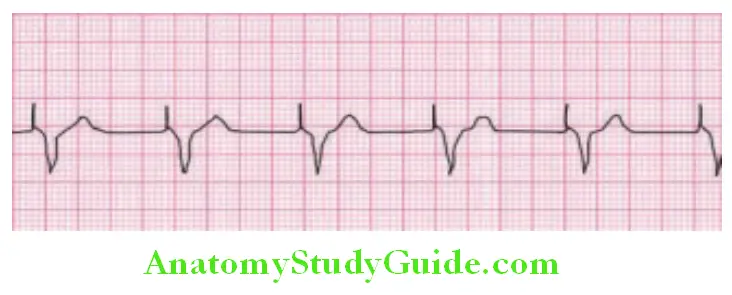
QRS-complex:
- Normal characteristics: Duration is 0.04–0.11 seconds.
- Broad/wide QRS (>0.12s): Ventricular hypertrophy, intraventricular conduction disturbance, bundle branch blocks, aberrant ventricular conduction, ventricular preexcitation, ventricular ectopic or escape pacemaker, and ventricular pacing by cardiac pacemaker
- Height of QRS–Sokolow index (SV2 + RV5) <35 mm (<45 mm for young)
- Increased height: In RV/LV hypertrophy
- Decreased height: Low voltage QRS (<5 mV in limb leads/<10 mV in chest leads): Obese patient, restrictive cardiomyopathy, pericardial effusion, hypothyroidism, hypothermia, and myocarditis.
Q Waves:
- The normal Q wave in lead I is due to septal depolarization. It is small in amplitude (less than 25% of the succeeding R wave, or less than 3 mm). Its duration is <0.04 sec or one small box. It is seen in L1 and sometimes in V5, V6.
- Pathological Q wave of infarction in the respective leads is due to dead muscle. It may also be seen in cardiomyopathies, i.e., hypertrophic (HOCM), infiltrative myocardial disease.
- Absent Q waves in V5-6 is most commonly due to LBBB.
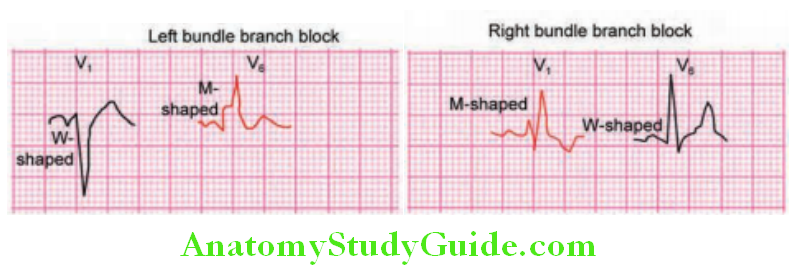
Bundle Branch Blocks:
- Left bundle branch block (LBBB): Indirect activation causes left ventricle to contract later than the right ventricle.
- Right bundle branch block (RBBB): Indirect activation causes right ventricle to contract later than the left ventricle.
Abnormal P waves and its causes:
- Tall (>2.5 mm), pointed P waves (P pulmonale): Suggests right atrial enlargement. Seen in COPD, ASD, TS, Ebstein anomaly (Himalayan P waves)
- Notched/bifid (“M” shaped) P wave (P “mitrale”) in limb leads: Suggests left atrial enlargement. Seen in MS, MR, and systemic hypertension.
- Coarse atrial fibrillation suggests LAE Absent P waves: Atrial fibrillation/flutter
- Inverted P waves in lead II: Dextrocardia
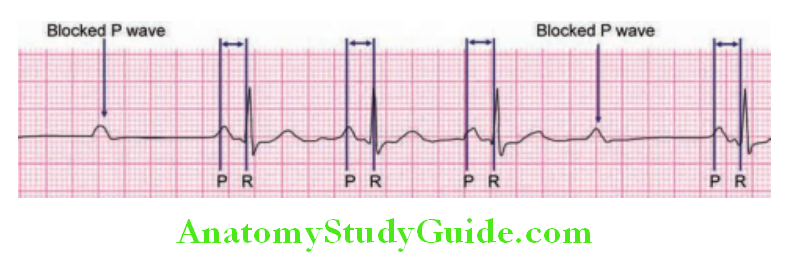
ST Segment:
- ST segment is isoelectric and at the same level as subsequent PR-interval. The length between the end of the S wave (end of ventricular depolarization) and the beginning of repolarization. From J point on the end of QRS complex, to inclination of T wave.
- Causes of ST-segment elevation
T Wave:
- Normally, a repolarization directs from epicardium to endocardium = T wave is concordant with QRS complex
- Causes of T wave inversions
- Tall T waves (height more than 2/3 of neighboring QRS): Hyperkalemia (steeple T waves), hyperacute MI.
QT Interval:
- It represents the time taken for ventricular depolarization and repolarization. The duration of the QT interval is proportionate to the heart rate. The faster the heart beats, the faster the ventricles repolarize so the shorter the QT interval. Therefore, what is a “normal” QT varies with the heart rate. QT interval should be 0.35–0.45 seconds.
- For each heart rate you need to calculate an adjusted QT interval, called the “corrected QT” (QTc)
- QTc = QT/square root of RR interval—Bazett’s formula
- Prolonged QTc (>440 ms): A prolonged QT can be very dangerous. It can predispose an individual to a type of ventricular tachycardia—Torsades de pointes.
- Short QTc (<350 ms): Hypercalcemia, digoxin effect.
U Waves:
- U wave need not be always seen on an electrocardiogram. It is small, round, and symmetrical and follows the T wave and seen positive in lead U waves are due to repolarization of the papillary muscles or Purkinje fibers. It is the same direction as T wave in that lead.
- Prominent U waves: These are seen in hypokalemia, hypercalcemia, thyrotoxicosis, or exposure to digitalis, epinephrine, and Class 1A and 3 antiarrhythmics.
- An inverted U wave may represent myocardial ischemia or left ventricular volume overload.
Causes of ST-segment elevation:
- Ischemia
- Early repolarization
- Acute pericarditis: ST elevation in all leads except aVR
- Pulmonary embolism
- Hypothermia
- Hypertrophic cardiomyopathy
- High potassium
- Cerebrovascular accident (CVA)
- Acute sympathetic stress
- Brugada syndrome
- Cardiac aneurysm
- Left ventricular hypertrophy
- Idioventricular rhythm including paced rhythm
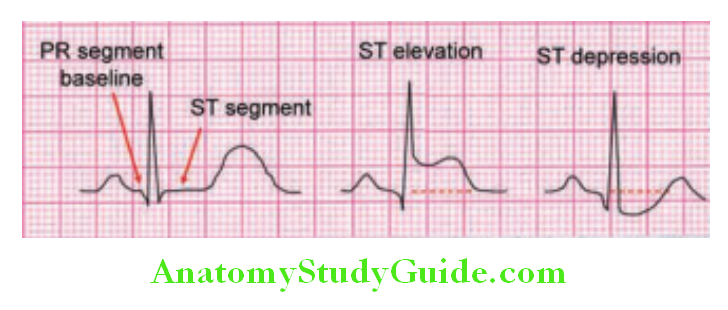
Causes of ST-segment depression:
- Myocardial ischemia/NSTEMI
- Reciprocal change in STEMI
- Posterior MI
- Digoxin effect (Reverse tick mark/‘sagging’ morphology, resembling Salvador Dali’s moustache)
- Hypokalemia
- Bundle branch block
- Ventricular hypertrophy
- Ventricular pacing
Causes of T wave inversions:
- CAD/ischemia
- Cardiomyopathies (hypertrophic), myocarditis, pericarditis, and pulmonary embolism
- Valvular disorders
- Raised intracranial tension, CNS bleed, and ventricular hypertrophy
- Bundle branch block
- Pacing
Causes of prolonged QTc:
- Hypokalemia, hypomagnesemia, and hypocalcemia
- Hypothermia
- Myocardial ischemia
- Raised intracranial pressure
- Congenital long QT syndrome, e.g., Jervell and Lange-Nielsen syndrome, Romano-Ward syndrome
- Drugs, e.g., chlorpromazine, haloperidol, quetiapine, quinidine, and procainamide
Step 6: Assess for Hypertrophy:
Left Ventricular Hypertrophy (LVH):
- Criteria of LVH:
- High QRS voltages in limb leads: Sokolow-Lyon index: S in V1 or V2 + R in V5 or V6 (whichever is larger) ≥35 mm OR R in aVL ≥11 mm
- Deep symmetric T inversion in V4, V5, and V6
- QRS duration >0.09 sec, associated left axis deviation
- Romhilt-Estes scoring system is used for diagnosing LVH.
- Common causes of left ventricular hypertrophy and left ventricular dilatation.
Ventricular Hypertrophy (RVH):
Criteria of right ventricular hypertrophy and its causes
Step 7: Look for Evidence of Infarction/Dyselectrolytemia:
ECG in myocardial infarction (MI): There are two types of MI—ST-elevation myocardial infarction (STEMI) and non-STEMI (NSTEMI).
Romhilt-Estes point score system (≥5 points definite LVH):
- Any limb lead R or S ≥20 mm/SV1 + SV2 ≥30 mm or RV5 + RV6 ≥30 mm—3 points
- ST-T changes with digitalis—1 point, without digitalis—3 points
- LAE—3 points
- Left axis deviation (≤30)—2 points
- QRS duration ≥90 ms—1 point
- Delayed intrinsicoid deflection in V5/V6 ≥50 ms—1 point

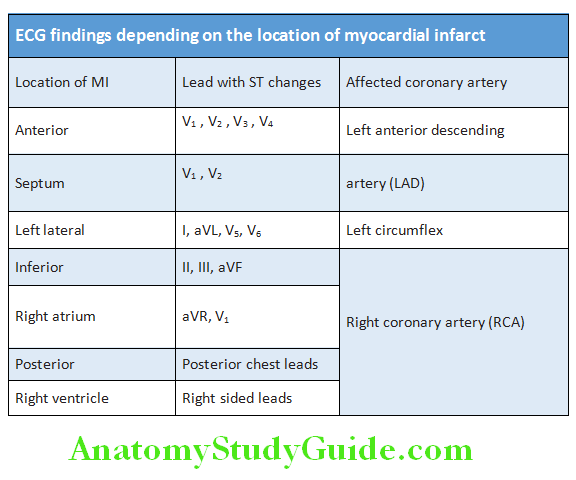
- STEMI criteria:
- ST elevation in >2 chest leads >2 mm elevation
- ST elevation in >2 limb leads >1 mm elevation
- Q wave >0.04s (1 small square).
ECG findings depending on the location of myocardial infarct are presented in Table Sequential ECG changes in STEMI are presented.
- Non-ST-elevation myocardial infarction (NSTEMI):
- NSTEMI is also known as subendocardial or non-Q-wave MIn a patient with acute coronary syndrome (ACS) in which the ECG does not show ST elevation, NSTEMI (subendocardial MI) is suspected if ECG shows T wave inversion (symmetrical, arrowhead) with/without ST depression.
- An ST depression is more suggestive of myocardial ischemia than infarction.
Usefulness of electrocardiogram:
- Cardiac arrhythmias
- Conduction defects
- Hypertrophy of cardiac chamber (atrium or ventricle)
- Electrolyte abnormalities (hypokalemia, hyperkalemia, hypocalcemia, and hypercalcemia)
- Effects of drugs (digitals)
- Hypothermia
- Pericarditis
Ischemic Heart Disease:
Question 31. Discuss the etiology, risk factors, clinical features, investigation, and treatment of ischemic heart disease (IHD).
Answer:
Definition: Ischemic heart disease is a group of heart diseases in which there is an imbalance between myocardial blood supply and its oxygen demand. IHD is the leading cause of death in both males and females.
Etiology:
- Coronary arterial occlusion is the main cause of myocardial ischemia.
- Mostly due to coronary atherosclerosis and its complications. Coronary atherosclerosis narrows one or more of the epicardial coronary arteries → decreases the coronary blood flow in about 90% of cases. Hence, IHD is often known as coronary artery disease (CAD) or coronary heart disease.
- Other rare causes: Emboli, vasculitis, coronary vasospasm, hematologic disorders, such as sickle cell disease, and diminished availability of blood or oxygen (lowered systemic blood pressure as in shock).
Risk Factors for Atherosclerosis:
Question 32. Write short essay/note on
Answer:
- Risk factors for coronary arterial disease
- Risk factors for atherosclerosis
They were identified through several studies most important being the Framingham heart study and atherosclerosis risk in communities study.
Classifiation of risk factors:
The risk factors may be broadly classified as modifiable, nonmodifiable, and additional.

Pathogenesis of Atherosclerosis:
Question 33. Discuss pathogenesis of atherosclerosis.
Answer:
Atherosclerosis is a progressive inflammatory disorder of the arteries characterized by focal deposits of lipids in the intima. It may be clinically silent until they become large enough to reduce tissue perfusion, or until ulceration and disruption of the atheromatous lesion lead to thrombotic occlusion or distal embolization of the vessel.
Early Atherosclerosis:
- Fatty streaks may be the earliest or precursor lesions of atherosclerosis. Endothelial injury and dysfunction leads to adhesion of leukocyte (mainly monocyte) to endothelium, increased vascular permeability, platelet adhesion, and movement of low-density lipoproteins (LDL) across the endothelium into the intima. This initiates the atheroma formation.
- Lipid accumulated in the intima is engulfed by the macrophages and form foam cells. This is followed by migration and proliferation of smooth muscle cells into the intima. Lipids accumulate both intracellularly (within macrophages and smooth muscle cells) and extracellularly. This results in formation of atheromatous plaque.
- Atheromatous plaque:
- It consists of three regions:
-
- Superficial fibrous cap (formed by fibrous tissue synthesized by smooth muscle cells around the lipid core),
- lipid-rich necrotic core (formed by lipid-laden foam cells that have undergone apoptosis) shoulder.
- Some atheromatous plaques bulge into the lumen of the coronary artery and narrow its lumen. This may limit the blood flow, particularly during increased myocardial demand leading to ischemic symptoms. Depending on the structure of plaque, they can be divided into stable and vulnerable (unstable) plaques:
- Stable plaques: They have dense collagenous and thick fibrous caps with minimal inflammation and negligible underlying atheromatous necrotic core. These are less likely to undergo rupture.
- High-risk or vulnerable plaque: They have core with many foam cells and abundant extracellular lipid (large lipid core). The fibrous cap is thin with few smooth muscle cells or groups of inflammatory cells (high density of macrophages and T lymphocytes) and increased inflammation. These are likely to undergo rupture.
- Sequential changes in coronary artery atherosclerosis causing occlusion of lumen in ischemic heart disease
-
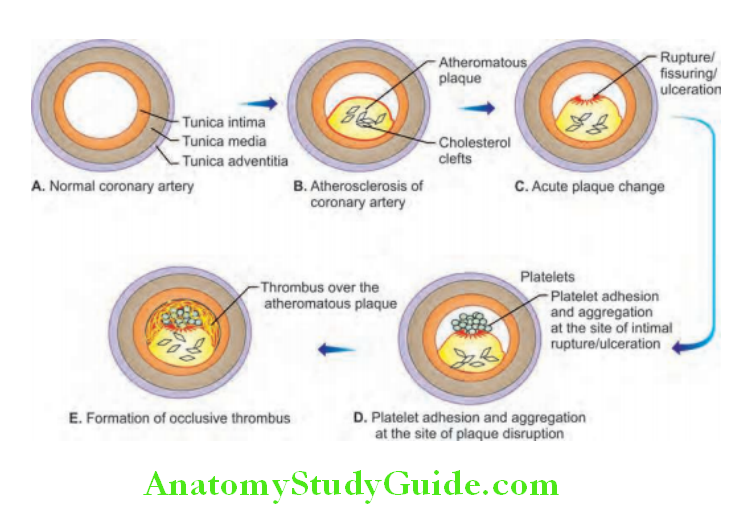
Advanced atherosclerosis/complicated plaques:
Atherosclerotic plaques can undergo clinically important changes
- Rupture, ulceration, or erosion: Plaque protrudes into the lumen and can disturb the blood flow → resulting in turbulent flow of blood → which can damage the endothelium → cause rupture, ulceration, or erosion of the intimal surface of plaques.
- Hemorrhage into a plaque: It may occur due to rupture of the fibrous cap of the plaque or of the thin-walled vessels formed due to neovascularization.
- Thrombosis and embolism: Ulceration/erosion/rupture of endothelial surface→ exposes the blood to highly thrombogenic subendothelial collagen → favors thrombus formation → can partially or completely occlude the lumen (depending on the size of the lumen) → lead to ischemia. The thrombus may become organized or fragment to form thromboemboli.
- Atheroembolism: Plaque rupture → discharge atherosclerotic debris into the bloodstream → results in atheroemboli.
- Aneurysm formation: Atherosclerosis even though an intimal disease may cause pressure or ischemic atrophy of the underlying media. It may also damage the elastic tissue and cause weakening the wall → result in aneurysmal dilation → which may rupture.
- Calcification: It may occur in the central necrotic area of the plaque (dystrophic calcification).
Nonatherosclerotic Causes of ACS:
Complications of atheromatous plaque:
- Rupture, ulceration, or erosion
- Hemorrhage into a plaque
- Thrombosis and embolism
- Atheroembolism
- Aneurysm formation
- Calcification
Nonatherosclerotic Causes of ACS:
The prevalence of nonobstructive coronary artery disease, defined clinically as luminal stenosis <50%, in patients presenting with myocardial infarction (MI) is estimated to be as high as 30% in women and 11% in men.
Important causes include:
- Spontaneous coronary artery dissection (SCAD): It accounts for up to 35% of ACS in women aged <50 years, and has a true prevalence as high as 4% of all cases of ACS. Systemic conditions associated with SCAD, include fibromuscular dysplasia, peripartum status, extreme emotion or exercise, and connective tissue diseases, such as Marfan syndrome, Ehlers–Danlos syndrome, and Loeys–Dietsyndrome.
- Coronary artery embolism—direct, paradoxical, or iatrogenic
- Coronary vasospasm at the epicardial or microvascular level
- Coronary artery bridging—a portion of myocardial muscle overlying a segment of an epicardial coronary artery is defined as a myocardial bridge.
- Stress-induced cardiomyopathy/Takotsubo syndrome (TTS)
Coronary Artery Disease:
Question 34. Discuss etiology, clinical manifestations, investigations, diagnosis, and management of angina pectoris.
Answer:
Angina Pectoris:
Definition: Angina pectoris is a clinical syndrome that presents as paroxysmal and recurrent attacks of substernal or precordial chest discomfort due to transient myocardial ischemia, which falls short of inducing necrosis of myocardial cell.
- Cause:
- Transient myocardial ischemia is due to:
- Obstruction Of Coronary Flow By Atherosclerosis
- Coronary Arterial Spasm
- Thrombosis Of Coronary Artery. Others Include Embolus, Coronary ostial stenosis, and coronary arteritis (e.g., in SLE).
- Transient myocardial ischemia is due to:
- Precipitate factors for angina: These include factors that either increase the oxygen requirement of myocardium or reduce blood supply to the myocardium

Stable Angina Pectoris:
Question 35. Discuss the clinical features, investigations, and treatment of chronic stable angina.
(or)
Write short note on angina decubitus.
(or)
Write short note on microvascular angina.
(or)
Write short note on nocturnal angina.
Answer:
- Coronary autoregulation is modified by coronary atherosclerosis, left ventricular hypertrophy, and alterations in autonomic nerve function and endothelial function.
- Coronary atherosclerosis reduces the lumen of the coronary arteries. It cannot increase in perfusion when the demand for flow is increased (e.g., during exertion or excitement). This leads to a situation where when the demand for blood flow is increased (e.g., during exertion or excitement), there cannot be a corresponding increase in perfusion due to the atheroma.
- Stable angina is due to transient myocardial ischemia. Stable angina shows a fixed reduction of at least 70% in the diameter of coronary arteries which causes reduction in coronary blood flow. Inability to increase oxygen extraction or reduced coronary blood flow, together with increased myocardial demand, leads to angina.
Stable Angina Pectoris Clinical Features:
History:
Diagnosis of angina is mainly depends on the clinical history.
Classical or Stable or Exertional Angina Pectoris:
It is characterized by:
- Chest pain: Constricting discomfort/squeezing/tightening/heaviness/aching in the front of the chest. Pain may radiate to left arm, neck (throat), jaw (chin) or less commonly to right arm, back, and epigastrium. Typical chest pain lasts 2–5 minutes. Levine’s sign (clenched fist held over the chest) may be positive.
- Brought on by physical exertion, such as after meals and in cold, windy weather or by anger or excitement/emotion.
- Relieved (usually within minutes) with rest or sublingual glyceryl trinitrate. Occasionally, it may disappear with continued exertion (“walking through the pain”). Pain seldom lasts more than 20 minutes. Typical angina has all the three features mentioned above. Atypical angina has two out of the three, and non-anginal chest pain one or less of these features. Many patients with angina may have silent episodes of angina, i.e., without any symptoms.

Types of Angina:
- Stable angina (described above)
- Unstable angina
- Refractory angina: Patients with severe coronary disease in whom revascularization is not possible and angina is not controlled by medical therapy.
- Variant (Prinzmetal’s/vasospastic) angina: Pain occurs without exertion and usually at rest. It is due to spasm of coronary artery and more frequent in women. Characteristically, it is associated with transient ST-segment elevation on the ECG during the pain. Provocation tests (e.g., hyperventilation, cold pressure testing, or ergometrine challenge) may be needed for establishing the diagnosis. Prognosis is usually better than those with fixed, significant obstructive lesions. Usually the response to β-blockers may be poor. Calcium channel blockers are used for the treatment.
- Angina decubitus: Pain develops while lying flat (raises end-diastolic left ventricular volume, myocardial wall tension, and hence oxygen demand).
- Nocturnal angina: Unusual form of angina that develops in aortic regurgitation especially syphilitic. It is characterized by paroxysmal, nocturnal angina pains accompanied by nightmares, dyspnea, palpitations, skin flushing, profuse sweating, and wide pulse pressure. It does not relieve by sublingual nitroglycerine.
- Cardiac syndrome X (microvascular angina): These patients have angina-like pain, a positive exercise test and angiographically normal coronary arteries. They form a heterogeneous group. It occurs in patients with metabolic syndrome and is more common in women. This type has a good prognosis. Response to nitrates is less reliable and they are difficult to treat. The myocardium shows an abnormal metabolic response to stress, indicating that the myocardial ischemia probably results from abnormal dilator responses of the coronary microvasculature to stress. About 1% die and 0.6% suffer a stroke within 1 year after their first hospital admission.
Stable Angina Pectoris Others:
- Status anginosus: Frequent, recurrent, sustained angina refractory to usual treatment.
- Walk-through angina: Angina with effort that disappears gradually during activity that is sustained (although usually at reduced intensity) and after which improved exercise tolerance results.
- Second-wind angina: A brief rest after an initial attack results in a markedly improved threshold free from angina. A synonym is “warmup” angina.
- Caudal angina: Angina symptoms occurring in the scalp or head via referred pain.
- Angina equivalents: Symptoms other than pain or discomfort that are ischemic related and serve as angina surrogates, e.g., dyspnea, diaphoresis, fatigue, or light-headedness.
- Silent angina: Objective manifestations of ischemia without symptoms.
Classifiation of Angina:
- Severity of angina is classified by New York Heart Association Classification in class I to class IV (described earlier).
- Canadian Cardiovascular Society functional classification of angina is shown in Table.

Physical Examination:
- Usually no abnormal findings in angina. Occasionally a third/fourth heart sound may be detected during an angina episode,dyskinetic cardiac apex, mitral regurgitation, and even pulmonary edema may be appreciated.
- Physical examination should include a careful search for evidence hyperlipidemia (e.g., xanthelasma, tendon and xanthoma), valve disease (particularly aortic stenosis characterized by slow rising carotid impulse and ejection systolic murmur radiating to the neck), important risk factors (e.g., hypertension and diabetes mellitus), left ventricular dysfunction (cardiomegaly and gallop rhythm), manifestations of arterial disease (carotid bruits and peripheral vascular disease), and unrelated conditions that may exacerbate angina (anemia and thyrotoxicosis), and obesity. Check the blood pressure to identify coexistent hypertension.
- Physical signs of myocardial ischemia: The presence of one or more of these during an attack of pain may be suggestive of myocardial ischemia.
Physical Examination Investigations:
Electrocardiography (ECG):
Question 36. Write a short essay/note on the investigations in a case of suspected angina pectoris.
Answer:
Resting ECG and ECG in between attacks are normal in most patents (even in patients with severe coronary artery). ECG may show evidence of previous MI and there may be T-wave flattening or inversion in some leads, due to myocardial ischemia or damage. The most convincing ECG evidence of myocardial ischemia is the demonstration of reversible ST-segment depression or elevation, with/without T-wave inversion, during the attack of pain (whether spontaneous or induced by exercise testing, such as treadmill testing or bicycle ergometry).
Exercise ECG: An exercise tolerance test (ETT) is usually done by using a standard treadmill or bicycle ergometer protocol (recording of ECG before, during and after exercise). During this process, patient’s ECG, BP, and general condition are monitored.
- Indications:
- Two sets of cardiac enzymes at 4-hour intervals should be normal.
- No significant abnormality in 12-lead ECG at the time of arrival and pre-exercise.
- Absence of ischemic chest pain at the time of exercise testing.
- Contraindications for exercise testing:
- Indications for terminating exercise testing of ECG
- Interpretation: Planar or down-sloping ST-segment depression of 1 mm or more indicates ischemia. Up-sloping ST depression is less specific and often found in normal individuals (modified Bruce protocol is followed).
- Advantages: Exercise testing is useful means of assessing the severity of coronary disease and identifying high-risk individuals (i.e., postinfarct angina, poor effort tolerance, ischemia at low workload, left main or three-vessel disease, and poor LV function).
- Disadvantages: It may produce false-positive results in the presence of digoxin therapy, left ventricular hypertrophy, bundle branch block, or Wolff–Parkinson–White (WPW) syndrome. The accuracy is lower in women than in men. The test is considered inconclusive (rather than negative) if the patient cannot achieve an adequate level of exercise because of locomotor or other noncardiac problems.
Contraindications for exercise testing:
Absolute:
- Acute myocardial infarction (within 2 days)
- High-risk unstable angina
- Uncontrolled cardiac arrhythmias causing symptoms or hemodynamic compromise
- Symptomatic severe aortic stenosis
- Uncontrolled symptomatic heart failure
- Acute pulmonary embolus or pulmonary infarction
- Acute pericarditis or myocarditis
- Acute aortic dissection
Relative:
- Left main coronary stenosis
- Moderate stenotic valvular heart disease
- Electrolyte abnormalities
- Severe arterial hypertension
- Bradyarrhythmias or tachyarrhythmias
- Hypertrophic cardiomyopathy and other forms of outflow
tract obstruction - Mental or physical impairment leading to inability to exercise adequately
- High-degree atrioventricular block
Indications for terminating exercise testing:
- Drop in systolic BP of >10 mm Hg from baseline BP despite an increase in workload, when accompanied by other evidence of ischemia
- Moderate to severe angina
- Increasing nervous system symptoms (e.g., ataxia, dizziness, or near-syncope)
- Signs of poor perfusion (cyanosis or pallor)
- Sustained ventricular tachycardia
- ST elevation (>1.0 mm) in leads without diagnostic Q-waves (other than V1 or aVR)
Other forms of stress testing:
- Myocardial perfusion scanning: It is performed using radioactive isotopes.
- It may be helpful in the:
- Evaluation of patients with an equivocal or uninterpretable exercise test and
- Patients who cannot perform exercise.
- The scintiscans of the myocardium are obtained at rest and during stress (either exercise testing or pharmacological stress, such as a controlled infusion of dipyridamole, adenosine or dobutamine), after the administration of an intravenous radioactive isotope, such as thallium (201thallium) or technetium (99technetium sestamibi). The radioactive isotopes are taken up by viable perfused myocardium. If there is a perfusion defect during stress but not at rest, it indicates evidence of reversible myocardial ischemia, whereas a persistent perfusion defect seen during both phases is usually indicative of previous MI.

- Stress echocardiography: It is an alternative to myocardial perfusion scanning and has similar predictive accuracy. On transthoracic echocardiography the ischemic segments show reversible defects in contractility during exercise or pharmacological stress, whereas infarcted regions do not contract at rest or during stress.
Coronary arteriography: It gives detailed anatomical information about the extent and nature of coronary artery disease. It is
usually done with a view to coronary artery bypass graft surgery or percutaneous coronary intervention (PCI). It is done under local anesthesia and requires specialized radiological equipment, cardiac monitoring, and an experienced team. Indications for coronary angiography
Newer modalities:
These include multiple-slice spiral computed tomographic coronary angiography (CTCA), intravascular ultrasound, magnetic resonance coronary angiography (MRCA), and positron emission tomography (myocardial viability is assessed using glucose metabolism).
Laboratory Tests:
- Fasting lipid profile
- Fasting glucose and/or glycated hemoglobin (HbA1c) level if available; additional oral glucose tolerance test (OGTT) if both are inconclusive.
- Complete blood count (CBC) and hematocrit
- Creatinine level with estimation of glomerular filtration rate (GFR)
- Biochemical markers of myocardial injury (Troponin T or I) if clinical evaluation suggests an acute coronary syndrome (ACS).
- Thyroid function tests
- Liver function tests early after beginning statin therapy.
- Advice to patients with angina is presented in Table

Question 37. Write a short essay/note on
Answer:
- Treatment of angina pectoris.
- Management of chronic stable angina.
- Drugs used in the treatment of chronic stable angina/antianginal drugs.
- Drug treatment of angina pectoris.
Management of angina pectoris:
Management can be discussed under three headings:
- General Measures
- Drug Treatment
- Surgical treatment. Goals of treatment of angina pectoris.
Goals of treatment of angina pectoris:
- Assessment of severity and extent of arterial disease
- Measures to control symptoms
- Identification and control of risk factors (e.g., smoking, hypertension, diabetes, and hyperlipidemia)
- Identification of high-risk patients for treatment and measures to improve life expectancy
General measures:
- Careful explanation about the nature of their condition (disease process).
- Evaluate the risk factors and steps to correct them where possible. Advice to be given to the patient
- Correction of precipitating underlying conditions, e.g., anemia, hyperthyroidism, valvular disease, and arrhythmias should be treated.
- Management of coexistent conditions: Identification and treatment of aggravating conditions, such as aortic stenosis, hypertrophic cardiomyopathy, and control of hypertension and diabetes (ACE inhibitors are useful in these patients).
- Lipid management: Identify hypercholesterolemia (hyperlipidemia) and treat with diet and drugs (with the goal of reducing LDL <100 mg dL; goal of <70 mg/dL in very high-risk patients).
- Lifestyle modification.
Question 38. Write short essay/note on lifestyle modifications in cardiovascular disease.
(or)
Write a short essay/note on antiplatelet drugs and its indications.
Answer:
- Healthy diet: Increase polyunsaturated fatty acid consumption, mainly from oily fish: 2 to 3 servings of oily fish per week may help prevent cardiovascular disease (CVD).
- Saturated fatty acids must comprise less than 10% of the total energy intake. Protein with low saturated fats should replace those high in these harmful fats.
- Limit salt intake to less than 5 grams per day.
- Consume 30 to 45 grams of fiber/day (e.g., wholegrain products, fruits, and vegetables). Avoid simple carbohydrates with high glycemic load.
- Consume 200 grams of vegetables and 200 grams of fruits per day.
- Limit consumption of alcoholic beverages.
Antianginal Drug Treatment:
Antiangina drugs can be divided into five groups. They are used to relieve or prevent the symptoms of angina.
These include:
- Nitrates
- β-Blockers
- Calcium Antagonists
- Potassium Channel Activators
- If channel antagonist.
1. Nitrates:
Short-acting (glyceryl trinitrate (GTN), nitroglycerine) or long-acting (isosorbide dinitrate, isosorbide mononitrate)
Mechanism of action: Nitrates directly act on smooth muscle in the walls of blood vessels and produce dilatation of arteries and veins.
This lowers blood pressure, reduces venous return to heart, and produces dilatation of coronary blood vessels. Nitrates cause reduction in myocardial oxygen demand (lower preload and afterload) as well as an increase in myocardial oxygen supply (coronary vasodilatation) predominantly by perfusing the subendocardial region.
Indications: Prophylaxis and treatment of angina. Prophylactically to use the drug before taking exercise that is liable to produce pain.
Prophylactic use of GTN should be encouraged because physical activity promotes the formation of collateral vessels. For predominant nocturnal angina, long-acting nitrates can be given at the end of the day.
Contraindications: Nitrates should not be given along with phosphodiesterase type 5 (PDE-5) inhibitors (e.g., sildenafil, tadalafil, and vardenafil) within the same 24 hours period because it may produce sever hypotension. Other contraindications include obstructive hypertrophic cardiomyopathy, severe aortic stenosis, constrictive pericarditis, mitral stenosis, and closed-angle glaucoma.
- Glyceryl trinitrate (GTN):
- Preparations:
- Metered-dose aerosol (400 μg per spray)
- As a tablet (300 or 500 μg).
- Preparations:
- Action: Sublingual GTN has a short duration of action and will relieve an attack of angina in 2–3 minutes.
- Isosorbide dinitrate (10–20 mg 2 to 3 times daily) has prolonged action and is given by mouth. Headache is a common side effect but tends to diminish if the patient perseveres with the treatment. Tolerance can develop with continuous nitrate therapy which can be avoided by a 6–8-hour nitrate-free period. Hence, doses are given in the morning and afternoon.
- Isosorbide mononitrate (20–60 mg once or twice daily) can also be given by mouth.
2. β-blockers:
- Mechanism: These drugs lower oxygen demand of myocardium by reducing heart rate, blood pressure, and myocardial contractility. They inhibit apoptosis by inhibiting beta adrenoceptors, and have antioxidant and antiproliferative properties. They also counteract the direct adverse effects of catecholamines and have antiarrhythmic action. They are useful to control tachycardia, hypertension, and continued angina.
- Cardioselective β-blockers: These include slow-release metoprolol 50–200 mg daily, bisoprolol 5–15 mg daily, and atenolol (50–200 mg/day). They have fewer peripheral side effects.
- Non-selective β-blockers: Propranolol is started in a small initial dose (20 mg thrice daily) and gradually increased to 80–120 mg three times daily. They may aggravate coronary vasospasm by blocking the coronary artery β2-adrenoceptors.
- Carvedilol (3.125–25 mg twice a day) has additional advantage of having antiarrhythmic effects.
3. Calcium channel antagonists (calcium channel blockers):
- Dihydropyridine calcium antagonists [e.g., nifedipine, amlodipine (dihydropyridines), felodipine, and nicardipine]. They produce coronary and peripheral arterial dilatation, and negative inotropy. They often cause a reflex tachycardia.
- Nifedipine: It is a powerful coronary and systemic arteriolar dilator. This can cause marked reflex tachycardia. Short-acting nifedipine are not used because it can increase mortality due to myocardial infarction. Long-acting preparations are given usually along with a β-blocker. Dose is 5–20 mg 3 times daily.
- Amlodipine: Dose is 2.5–10 mg daily. Side effects are ankle edema and reflex tachycardia.
- Non-dihydropyridine calcium antagonists, e.g., verapamil (phenylalkylamines) and diltiazem (benzothiazepines). They produce coronary and peripheral arterial dilatation and negative inotropy, and also reduce conductivity. Because of its negative inotropic effect, they should be avoided in patients with impaired ventricular function (uncompensated heart failure).
- Verapamil: Dose is 40–80 mg thrice daily. Useful antiarrhythmic properties. Common adverse effect is constipation.
- Diltiazem: 60–120 mg 3 times daily. Similar antiarrhythmic properties to verapamil.
- β-blockers reduce mortality after myocardial infarction. Hence, it is reasonable to start a β-blocker and then add a calcium channel blocker if needed. However, β-blockers should not be combined with verapamil, because of their synergistic effect on heart rate and myocardial contractility.
4. Second-line antianginal drugs:
- Potassium channel activators/openers: Nicorandil increases potassium ion conductance by opening ATP-sensitive potassium channels resulting in smooth muscle relaxation causing arterial and venous dilatation. It does not develop tolerance as seen with nitrates. They also provide protection of myocardium during ischemia and prevention of intracellular calcium toxicity. It is given in the dose of 10–30 mg twice daily orally.
- If channel antagonist: Ivabradine selectively inhibits inward sodium-potassium current [important pacemaking current in the cells sinus (SA) node]. This slows the rate of diastolic depolarization and induces bradycardia (“bradycardic” drug). In contrast toβ-blockers and ratelimiting calcium antagonists, it does not have other cardiovascular effects. Thus, it does not affect contractility, AV nodal conduction orm hemodynamics. It can be combined with other agents and is safe to use in patients with heart failure. It may produce brightness in visual fields because it also blocks the retinal current and has transient side effect. It is contraindicated in sick sinus syndrome and AV block.
- Ranolazine: It inhibits late sodium channels in cardiac cells. It does not affect heart rate and blood pressure. It is the drug of choice in bradycardic and hypertensive patients. It is metabolized by cytochrome P450 3A4. Dose is 500–1,000 mg twice a day. Side effects include constipation, dizziness, and prolongation of QT interval.
- Trimetazidine: Dose is 20 mg three times daily. It is associated with greater improvements in time to onset of angina, and the mean weekly number of anginal episodes.
- Fasudil: It is an inhibitor of Rho kinase that is involved in the vascular smooth muscle contractile response. It is of benefit in patients with stable and microvascular angina (angina with normal coronary arteries).
- Allopurinol : The drug significantly improves endotheliumdependent vasodilation and completely abolishes oxidative stress.
- Therapeutic angiogenesis for management of refractory angina using VEGF is under trials. None of these groups is more effective than another group. It is conventional to start therapy with low-dose aspirin, a statin, sublingual GTN, and aβ-blocker. If needed later add a calcium channel antagonist or a long-acting nitrate. The goal is the control of angina with minimum side effects and the simplest possible drug regimen. Indications and contraindications of various antianginal drugs are given in Table
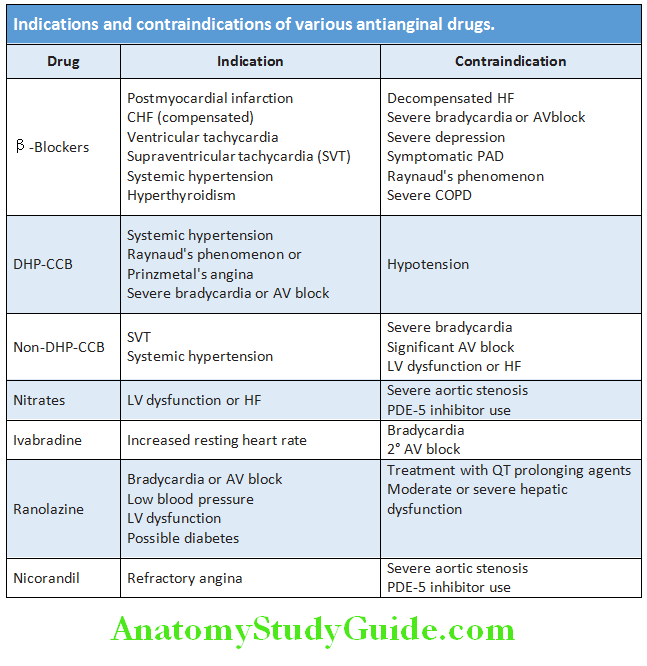
5. Antiplatelet therapy:
Antianginal drugs ameliorate only symptoms but may not reduce mortality. To reduce the risk of adverse events, such as MI, antiplatelet
drugs are given.
Aspirin:
- Aspirin inhibits the synthesis of prostaglandins, namely thromboxane A2, which is a potent vasoconstrictor and platelet activator.
- Dose: Low-dose therapy in the dose of 75–150 mg/day.
P2Y12 antagonists:
Clopidogrel:
- It is a thienopyridine which inhibits ADP-dependent activation of the GPIIb/IIIa complex and prevents platelet aggregation.
- It is an equally effective antiplatelet agent that can be used in patients who cannot tolerate aspirin.
- It may have a synergistic effect when combined with aspirin in patients following acute coronary syndrome or implantation of a drugeluting stent. The benefit of its combination with aspirin was not found chronic stable angina.
- Dose: 75 mg daily
- Prasugrel and ticagrelor are new P2Y12 antagonists and have higher platelet inhibition compared to clopidogrel.
6. Statins:
Irrespective of the LDL or cholesterol levels. Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors confer a mortality benefit to patients with IHD whose LDL levels remain >70 mg/dL despite high-intensity statins.
Invasive (Surgical) Treatment Revascularization:
Percutaneous Coronary Intervention (PCI):
- Percutaneous coronary interventions include angioplasty [percutaneous transluminal coronary angioplasty (PTCA)] or stent placement in the coronary artery. It is the process to maximize and maintain dilatation of a stenosed coronary artery. A coronary stent is a piece of coated metallic “scaffolding” (fine guidewire) that can be deployed on a balloon. In this process, a small inflatable balloon and metallic coronary stent introduced percutaneously into the arterial circulation via an arterial catheter through the femoral, radial, or brachial artery under radiographic control. It is passed across the coronary stenosis and balloon is inflated to dilate the stenosis. Dilatation can be repeated if symptoms recur.
- Types of stents: Two types, namely
- Bare-metal stents
- Drug-eluting stents.
- The drug-eluting stents are coated stents lined
with substances (e.g., sirolimus and pacliltaxel) that prevent neointimal hyperplasia and reduce the risk of coronary artery reocclusion. Recent data suggest both types of stents to be equally effective over long-term follow-up. - Indications for PCI:
- Ideal for single-vessel or two-vessel coronary disease without significant lesions in the proximal left anterior descending artery (LAD), with normal LV function, with high risk on noninvasive testing and a large area of viable myocardium.
- Undergone prior PCI with either recurrence of stenosis or high risk on noninvasive testing.
- Failure of medical therapy and with acceptable risk for PCTreatment of choice for unstable angina (UA) when rest pain recurs in spite of full medical treatment.
- Lesion suitable for PCI
- No diabetes
- Complications of the procedure: Bleeding, hematoma, dissection, and pseudoaneurysm from the arterial puncture site. Serious complications are acute myocardial infarction (2%), stroke (0.4%), and death (1%). Long-term complication is restenosis (33% of cases) which is due to a combination of elastic recoil and smooth muscle proliferation (neointimal hyperplasia). It tends to occur within 3 months.
Coronary Artery Bypass Grafting (CABG):
- In CABG, autologous veins (reversed segments of the patient’s own saphenous vein) or arteries (internal mammary artery/radial artery/gastroepiploic arteries) are anastomosed to the ascending aorta at one end and to the native coronary arteries distal to the area of occlusion/stenosis at the other end.
- Usually done under cardiopulmonary bypass but, in few cases, it can be done in the beating heart (“off-pump” surgery). Aspirin (75–150 mg daily) and clopidogrel (75 mg daily) both improve graft patency, and one or other should be given indefinitely.
- Indications:
- Significant left main coronary disease.
- Triple-vessel disease/two blood vessel disease with reduced left ventricular function (left ventricular ejection fraction is <50%).
- Two-vessel disease with significant proximal left LAD disease and either LVEF <50% or demonstrable ischemic on noninvasive testing.
- Failure of medical therapy and with acceptable risk for CABG.
- Diabetes
- Prior CABG, PCIs (percutaneous coronary interventions) with recurrent rest enosis
- Abnormal stress test
- Risks: Higher risks in elderly, those with poor left ventricular function and those with significant comorbidity, such as renal failure.
- Minimally invasive operative procedures for bypass grafting (MIDCAB): Laparoscopic approaches may be useful in patients with previous CABG and those with coexistent medical conditions which would increase the operative risks of “full” CABG).
Prognosis:
- Critical stenosis (>70%) of coronary arteries and 5 years mortality rate: One artery—2%, two arteries—8%, and three arteries—11%. 50% stenosis of left main coronary artery has a mortality rate of about 15% per year.
- Other poor prognostic factors are advanced age (>75 years), diabetes, morbid obesity, accompanying peripheral vascular and or cerebrovascular disease, previous myocardial infraction, high levels of plasma CRP, and evidence of LV dysfunction.
Novel therapy in angina:
Newer drugs:
- Ivabradine: Selectively blocks I f in a currentdependent fashion in pacemaker cells of the sinoatrial node
- Ranolazine: Partial fatty acid oxidation (pFOX) inhibitor, late sodium current blocker
- Trimetazidine: Inhibitor of partial fatty acid oxidation (pFOX)
- Perhexiline: Inhibits mitochondrial carnitine palmitoyltransferase-1 (CPT-1)
- Etomoxir/Oxfenicine: CPT 1 inhibitor
- Nicorandil: Activation of ATP-sensitive K+channels
- Rho kinase inhibition: Fasudil
- Molsidomine and linsidomine act by releasing nitric oxide
- Others: Testosterone, endothelin receptor blockers (bosentan), allopurinol
TMLR: Transmyocardial laser revascularization
EECP: Enhanced external counter pulsation
Chelation therapy: Intravenous EDTA infusions
Spinal cord stimulation, coronary sinus reducing device, apheresis
Other novel agents:
- Monoclonal antibodies: Inclacumab is a P-selectin monoclonal antibody, in patients with NSTE–ACS undergoing PCIt reduces myocardial damage
- Stem cell therapy: The use of stem cells derived from bone marrow or myocardium to improve cardiac function has been promising
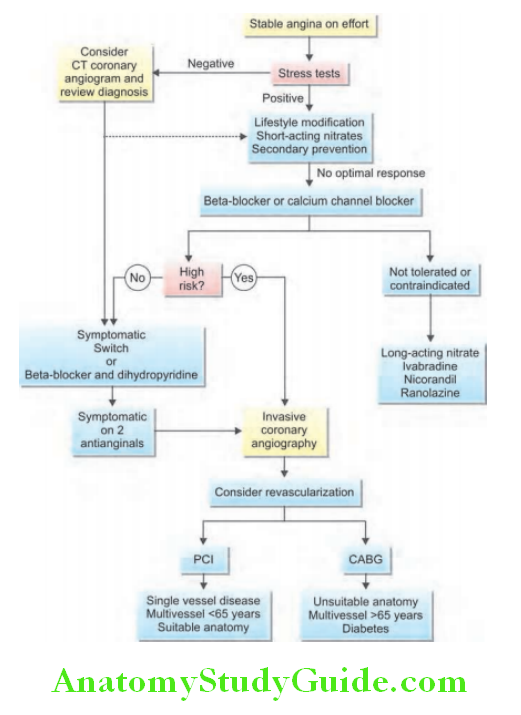
St-Elevation Myocardial Infarction (Stemi):
Question 39. Discuss the etiology (risk factors), clinical features, investigations, complications, and management of acute coronary syndrome/ACS (ST-elevation myocardial infarction—STEMI)/acute myocardial infarction.
(or)
Write short essay/note on risk factors of acute myocardial infarction.
Answer:
- STEMI is due to the formation of an occlusive thrombus at the site of rupture of an atheromatous plaque in a coronary artery. Usually there is minimal prior narrowing of coronary lumen.
- Other causes: Coronary spasm, rarely coronary emboli as well as by ostial narrowing due to aortitis, hypercoagulable state and use of cocaine. Congenital anomalies, such as the origin of the left anterior descending coronary artery from the pulmonary artery may cause myocardial ischemia in infancy, but this cause is very rare in adults.
- Limitation of the ability to increase flow to meet increased myocardial demand occurs with 50% coronary stenosis while 80% coronary stenosis causes myocardial ischemia at rest or with minimal stress.
Types of Myocardial Infarction:
- Type 1: Spontaneous myocardial infarction with ischemia due to a primary coronary event, e.g., plaque erosion/rupture, fissuring or dissection.
- Type 2: Myocardial oxygen demand supply mismatch, i.e., secondary to ischemia due to increased oxygen demand or decreased supply (e.g., coronary spasm, coronary embolism, anemia, arrhythmias, hypertension, or hypotension).
- Type 3: Diagnosis of MI resulting in sudden cardiac death.
- Type 4a: MI after percutaneous coronary intervention (PCI)—post-PCI related.
- Type 4b: Infarction from stent thrombosis.
- Type 5: MI after coronary artery bypass graft—post-coronary artery bypass graft.
Transmural infarct is an infarct that involves the full thickness of myocardium. Subendocardial infarct is an infarct which involves subendocardial region. However, these terms are no longer used. Silent infarct is infarct without symptoms but with only ECG changes.
Non-St-Elevation Myocardial Infarction (Nstemi):
Causes:
- It is most commonly caused by an imbalance between oxygen supply and oxygen demand. This imbalance results from a partially occluding thrombus forming on a disrupted atherothrombotic coronary plaque or on eroded coronary artery endothelium.
- Severe ischemia or myocardial necrosis may develop due to the reduction of coronary blood flow caused by the partially occluding thrombus and by downstream embolization of platelet aggregates and/or atherosclerotic debris.
- Other causes: More than one of the following processes may be involved.
- Dynamic obstruction (e.g., coronary spasm, as in Prinzmetal’s variant angina)
- Severe mechanical obstruction due to progressive coronary atherosclerosis
- Increased myocardial oxygen demand produced by conditions, such as fever, tachycardia, and thyrotoxicosis in the presence of fixed epicardial coronary obstruction.
Diagnosis of NSTEMI:
- Clinical presentation: Diagnosis of NSTEMI is largely based on the clinical presentation. Typically, chest discomfort is severe and has at least one of three features:
- Occurs at rest (or with minimal exertion) lasting for >10 minutes.
- Relatively recent onset (i.e., within the prior 2 weeks) and/or
- Occurs with a crescendo pattern (i.e., distinctly more severe, prolonged, or frequent than previous episodes).
- Evidence of myocardial necrosis: Diagnosis of NSTEMI is established, if a patient with the above clinical features shows the evidence of myocardial necrosis, as reflected in abnormally elevated levels of biomarkers of cardiac necrosis.
Acute Coronary Syndrome:
Question 40. Write short essay on acute coronary syndromes.
Answer:
Ischemic heart disease (IHD) forms a spectrum of diseases and consists of stable angina and acute coronary syndromes (includes STEMI, NSTEMI, and unstable angina).
Acute coronary syndrome (ACS) is a term used for spectrum of clinical presentations due to acute myocardial ischemia.
Acute Coronary Syndrome It includes:
- ST-elevation myocardial infarction (STEMI): Majority of STEMI has Q-wave MI (QwMI).
- Non-ST-elevation myocardial infarction (NSTEMI): A small percentage of STEMI and majority of NSTEMI have non-Q-wave MI (NQwMI, previously known as subendocardial infarction). However, the terms Q-wave or non-Q-wave infarctions are not used at present.
- Unstable angina (UA): It Includes patients with ACS but with normal ECG, without elevation ofcardiac injury markers and no ST elevation in the ECG. Management of unstable angina and NSTEMI is similar.
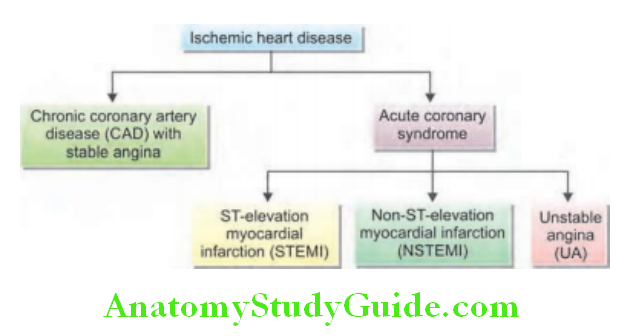
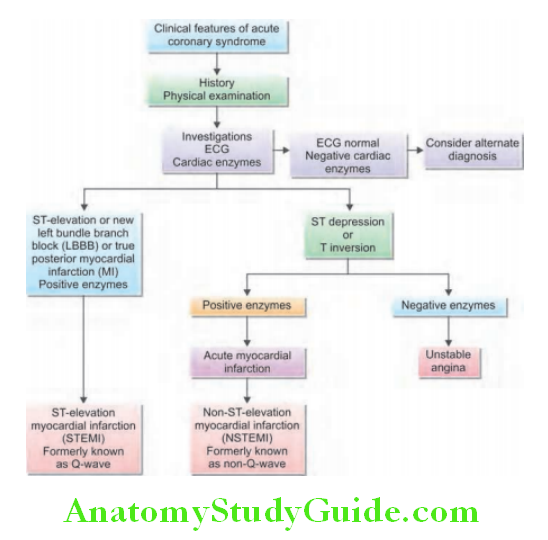
Pathophysiology:
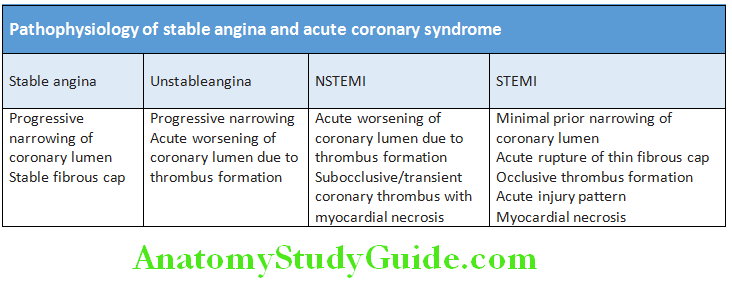
Clinical Features of Acute Coronary Syndrome:
Symptoms:
- Prolonged cardiac pain: Myocardial ischemia causing chest discomfort is termed angina pectoris. Thus, classic manifestation of ischemia is angina, which is usually described as a heavy chest pressure or squeezing, a burning feeling, or difficulty breathing.
- The discomfort often radiates to the left shoulder, neck, or arm. It typically builds in intensity over a period of a few minutes.
- The pain may begin with exercise or psychological stress, but ACS most commonly occurs without obvious precipitating factors.
- Pain may be absent in patients with prior cardiac, prior stroke, age >75 years, and diabetes mellitus. Painless MI is more common in females compared to males.
- Any patient with severe chest pain that lasts for more than 20 minutes may be suffering from a myocardial infarction. This pain usually does not respond to sublingual GTN.
- Other features: Features include anxiety and fear of impending death, nausea and vomiting, breathlessness, and collapse/ syncope.
Physical Signs:
Sometimes infarction occurs without any physical signs.

Complications of Acute Coronary Syndrome:
Major mechanical and structural complications occur only with significant, often transmural, MI
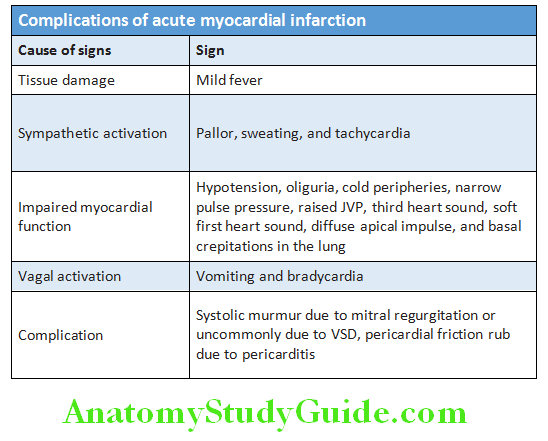
Arrhythmias:
- Many patients with acute coronary syndrome may develop arrhythmia and they are transient in most of cases and are of no hemodynamic or prognostic importance. Relief of pain, rest, and the correction of hypokalemia may prevent arrhythmias.
- Ventricular fibrillation: It develops in 5–10% of patients and appears to be the major cause of death in those who die before receiving medical attention.
- Atrial fibrillation is common but usually transient and does not need emergency treatment. However, if it produces a rapid ventricular rate with hypotension or circulatory collapse, prompt cardioversion by immediate synchronized DC shock is required. In other cases, digoxin or a β-blocker is given.
- Bradycardia: Usually does not need treatment, but if there is hypotension or hemodynamic deterioration, atropine (0.6–1.2 mg IV) may be given. Various arrhythmias in acute coronary syndrome
Various arrhythmias in acute coronary syndrome:
- Atrial fibrillation
- Atrial tachycardia
- Bradycardia (especially after inferior MI) and tachycardia
- Atrioventricular heart blocks
- Ventricular fibrillation
- Ventricular tachycardia
- Accelerated idioventricular rhythm
- Ventricular ectopic beats
Cardiogenic Shock:
- Causes:
- Arrhythmia
- Hypovolemia due to excessive diuretic therapy or recurrent vomiting
- Extensive myocardial damage (has bad prognosis)
- Risk factors: Older age, hypertension, diabetes mellitus, multivessel coronary artery disease, anterior MI, prior MI or angina, prior heart failure, STEMI, and left bundle branch block.
Left Ventricular Failure:
It commonly leads to pulmonary edema.
Mechanical Complications:
- Myocardial rupture: Part of the necrotic muscle in a fresh infarct can result in tear or cardiac rupture. Most frequent during 3 to 7 days after transmural infarcts.
- Rupture of the ventricular free wall: It is most common and result in hemopericardium and cardiac tamponade. It is usually fatal.
- Rupture of the ventricular septum: It is less common and can lead to an acute VSD and left-to-right shunt. It usually presents with sudden hemodynamic deterioration and produces a new loud pansystolic murmur radiating to the right sternal border.
- Rupture of papillary muscle: It can lead to acute severe mitral regurgitation, which presents with a pansystolic murmur and third heart sound.
Embolism:
Thrombus may form on the endocardial surface of freshly infarcted myocardium due to local abnormality in myocardial contractility (causing stasis) and endocardial damage (creating a thrombogenic surface). This can lead to systemic thromboembolism and occasionally causes a stroke. Venous thrombosis and pulmonary embolism may also develop patients on prolonged bed rest, but are now less common with the use of prophylactic anticoagulants and early mobilization.
Ventricular Aneurysm:
After acute transmural infarction, the affected ventricular wall may bulge outward during systole resulting ventricular aneurysm. It develops as a late complication of large transmural infarcts.
Pericarditis:
- Early pericarditis: A transmural myocardial infarct→ can involve the pericardium→ cause fibrinous or fibrinohemorrhagic pericarditis. Usually, develops on second or third day.
- Delayed form of pericarditis (postmyocardial infarction syndrome/Dressler syndrome): It develops 2 to 10 weeks after infarction—probably immunologically mediated reaction to necrotic muscle. It is characterized by fever, pericarditis, and pleurisy. Treatment is with aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs) or corticosteroids.
Investigations:
Question 41. Write short essay/note on ECG changes in acute myocardial infarction.
Answer:
Electrocardiogram (ECG):
The 12-lead ECG is central to confirming the diagnosis and should be done and interpreted within 10 minutes of arrival. The initial ECG may be normal or nondiagnostic in about 30% of cases. Repeated ECGs are needed, especially where the diagnosis is uncertain or the patient has recurrent or persistent symptoms.
Changes in ECG: Characteristic changes are observed in leads that “face” the ischemic or infracted area (e.g., anteroseptal, anterolateral, strict anterior, inferior, and posterior wall infarction).
STEMI: ST-segment deviation is the earliest ECG change. With proximal occlusion of a major coronary artery, ST-segment elevation (or new bundle branch block) is observed initially. Later, there is diminution in the size of the R wave and, in transmural (full-thickness) infarction, there is development of a Q wave. Subsequently, the T wave becomes inverted and persists after the ST segment has returned to normal.
NSTEMI and unstable angina: It is due to partial occlusion of a major vessel or complete occlusion of a minor vessel, causing unstable angina or partial-thickness (subendocardial) MThey usually produce ST-segment depression and T-wave changes. When infarction is present, there may be some loss of R waves in the absence of Q waves. Anatomic site of infarct, ECG lead and location of myocardial infarct are discussed earlier.
Plasma Cardiac Biomarkers (Biochemical Markers of Cardiac Injury):
Question 42. Write a short essay/note on serum markers in acute myocardial infarction.
Answer:
Unstable angina: There is no detectable rise in cardiac biomarkers or enzymes in unstable angina and the initial diagnosis is made from the clinical history and ECG only.
Myocardial infarction: Causes arise in the plasma concentration of enzymes and proteins that are normally concentrated within cardiac cells. These include creatine kinase (CK), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), myoglobin, and troponins (troponin I and troponin T). These markers leak from the necrotic myocardial cells into the blood circulation.
- Cardiac creatine kinase (CK): It is a nonspecific enzyme marker and is present in brain, myocardium, and skeletal muscle. It has two isoforms designated “M” and “B”. MB heterodimers chiefly in cardiac muscle (lesser amounts in skeletal muscle).
- MB form of creatine kinase (CK-MB) is sensitive but not specific, because it is also raised with skeletal muscle injury.
- CK-MB levels rise within 4 to 6 hours of the onset of MI, peaks at 12 hours, and returns to normal within 72 hours.
- Total CK is also raised in diseases of skeletal muscle (e.g., polymyositis and muscular dystrophies), hypothyroidism, and stroke.
- Lactate dehydrogenase: It not specific marker. It starts rising after 24–48 hours. It remains for many days and returns to normal in 7–14 days. An elevated LDH1 (an isoenzyme of LDH) is a more sensitive indicator of myocardial infarction than total LDH.
- Myoglobin: It is an oxygen-carrying respiratory protein found only in skeletal and cardiac muscle. It is an earliest marker of MI, the level rises within 1–3 hours, peaks in about 8–12 hours and return to normal in about 24–36 hours.
- Cardiac troponins: These are proteins involved in heart muscle contraction. Increased plasma levels establish the diagnosis of myocardial infarction. Cardiac-specific proteins are of two types, namely cardiac Troponins I (cTnI) and T (cTnT). They are most sensitive and specific markers of myocardial infarction. Levels begin to rise at 4–6 hours and peak at 48 hours. The elevated troponin levels may remain for 7–10 days after acute MI and therefore, this assay is particular useful in the evaluation of patients who present sufficiently long after their episode of chest pain. Further, about one-third of patients with unstable angina also have elevated cTn, which classifies these groups of patients to nonST-elevation MI.
- Aspartate aminotransferase: Starts to rise by about 12 hours and reaches a peak on the first or second day.
- Other enzymes:
- Ischemia modified albumin,
- N terminal Pro BNP,
- suPAR (soluble urokinase-type plasminogen activator receptor)
- Glycogen phosphorylase isoenzyme BB.
Other Blood Tests:
- Leukocytosis with a peak on first day.
- Erythrocyte sedimentation rate (ESR): Raised and may remain so for days.
- C-reactive protein: Elevated.
- Heart-type fatty acid-binding protein (H-FABP) as a plasma marker for the diagnosis of patients presenting with chest pain suggestive of myocardial infarction, especially in the early hours (within 2 hours) after onset of symptoms. However, their use as a diagnostic tool for MI is limited.
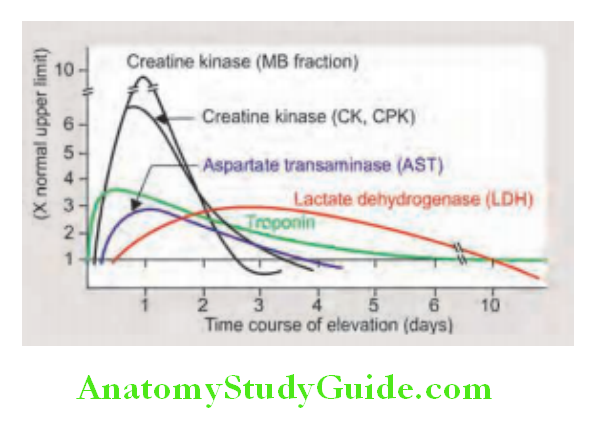
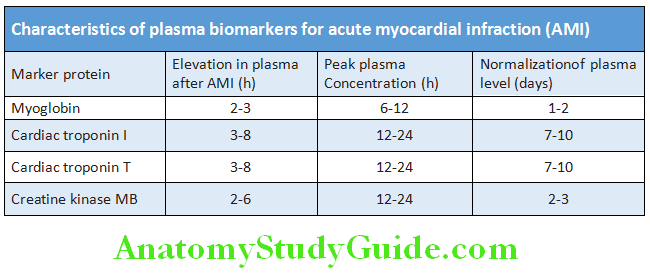
Chest X-ray:
- It may show evidence of pulmonary edema that is not evident on clinical examination.
- Heart size is usually normal but there may be cardiomegaly due to previous myocardial damage or pericardial effusion.
Echocardiography:
It is useful for assessing ventricular function and for detecting complications (e.g., mural thrombus, cardiac rupture, ventricular septal defect, mitral regurgitation, and pericardial effusion).
Radionuclide Scanning:
It helps to detect the site of necrosis and the extent of damage to ventricular function.
Question 43. Write short essay/note on
Answer:
- Management of a case of acute myocardial infarction in the fist 24 hours.
- Management of a case of acute myocardial infarction.
Management:
Immediate management: The first 24–48 hours, the patients should be admitted immediately to hospital. During first 24–48 hours the risk for fatal arrhythmia is highest and as a result, there is a significant risk of death or recurrent myocardial ischemia. Patients are best treated in an intensive coronary care unit.
Management of acute myocardial infarction
Initial treatment:
Admit in intensive coronary care unit, attach a cardiac monitor, and secure an intravenous line.
- General treatment (“M O N A C”):
- Morphine 2–4 mg q 5–10 minute to control chest pain.
- Oxygen 4 L/minute Hypoxemia in uncomplicated MI is usually due to ventilation-perfusion abnormalities and may be exacerbated by CHF. Therefore, oxygen is given to patients suspected of having an acute coronary syndromes and oxygen saturation <90%.
- NTG (nitroglycerine) sublingual or spray, followed by infusion for persistent chest pain
- Aspirin 160–325 mg chew and swallow or/and
- Clopidogrel 300 mg oral
- Confirm diagnosis: By investigations, namely:
- Electrocardiogram (ECG)
- Plasma cardiac biomarkers: Troponin T or I and CK-MB
- Specific therapy:
- Thrombolysis or percutaneous coronary interventions
- β-blockers unless contraindicated
- Treat complications (arrhythmias, congestive failure, and shock)
Control of Pain by Analgesics:
- Proper control of pain is necessary not only to relieve distress but also to lower adrenergic drive which reduces vascular resistance, BP, infarct size, and susceptibility to ventricular arrhythmias.
- Intravenous opiates: Initially, morphine in the dose of 2–4 mg or diamorphine 2.5–5 mg is administered along with antiemetics (metoclopramide 10 mg), and repeated until the patient is comfortable.
- β-blockers, nitroglycerine, and thrombolysis may also help in reducing the pain. Antianginal therapy (refer earlier)
Antiplatelet therapy:
- Aspirin: In acute coronary syndrome, oral aspirin (75–325 mg daily) improves survival and reduce mortality. The first dose (300 mg) should be given orally within the first 12 hours and should be continued indefinitely if there are no side effects.
- Combination therapies: Combination of aspirin and an ADP-receptor blocker (clopidogrel, prasugrel or ticagrelor) is recommended in patients with STEMI who are undergoing primary PCI (for up to 12 months) or (clopidogrel) fibrinolysis and in those who have not undergone reperfusion therapy. In acute coronary syndrome, with/without ST-segment elevation, ticagrelor (180 mg, followed by 90 mg twice daily) is found to be more effective than clopidogrel in reducing vascular death, MI or stroke, and other causes of death.
- Glycoprotein IIb/IIIa receptor antagonists: These are powerful inhibitors of platelet aggregation and prevent thrombus formation (e.g., tirofiban, eptifibatide, and abciximab). Abciximab is a monoclonal antibody which binds tightly and has a long half-life. They are beneficial in patients who undergo PCI, those with recurrent ischemia and those with high risk (e.g., diabetes mellitus and raised troponin concentration). Various antiplatelet agents and their dosage are mentioned.
Anticoagulants (antithrombin therapy):
Prophylactic anticoagulants are given to prevent deep vein thrombosis and pulmonary embolism in patients who do not receive fibrinolytic agents. They reduce the risk of thromboembolic complications, and prevent reinfarction in the absence of reperfusion therapy or after successful thrombolysis.
Preparations:
- Unfractionated heparin: Given as an initial bolus dose of 60 IU/kg (with a maximum dose of 4,000 units) followed by an initial infusion of 12 IU/kg/hour (maximum 1000 units/hour). The dose is adjusted to attain the activated partial thromboplastin time at 1.5–2 times control. Heparin is given before the completion of infusion of rt-PA or tenecteplase or patients receiving STK.
- Low-molecular weight heparin: It is used as an adjunct to thrombolytics. It produces higher reperfusion rate and lower reocclusion rate compared to unfractionated heparin. Dose of 5,000 units is given twice a day subcutaneously.
- Direct thrombin inhibitors: These appear better than the unfractionated heparin in patients undergoing PCThese include hirudin and bivalirudin. Pentasaccharides (subcutaneous fondaparinux 2.5 mg daily) are safe and effective. However, fondaparinux is not used as sole agent and contraindicated if PCI is planned.
- Antithrombin preparations should be continued for at least 48 hours and preferably for 8 days or till discharge or coronary revascularization.
Statins:
High-dose statins are recommended in all patients during the first 24 hours of admission for STEMI, irrespective of the patient’s cholesterol concentration, if there is no contraindications (e.g., allergy, active liver disease). They are recommended during the early phase of therapy up to at least 4 weeks. Patient on statin therapy presenting with STEMI should continue statin.
Statins Advantages:
- Statins lower cholesterol and has direct effects on endothelial function, oxidative stress, inflammation, thrombosis as well as plaque stabilization. High-dose atorvastatin (40–80 mg) or rosuvastatin (20–40 mg) therapy before emergency percutaneous coronary intervention has following advantages:
- Reduce periprocedural inflammatory response
- Reduce myocardial dysfunction
- Prevent contrast-induced nephropathy.
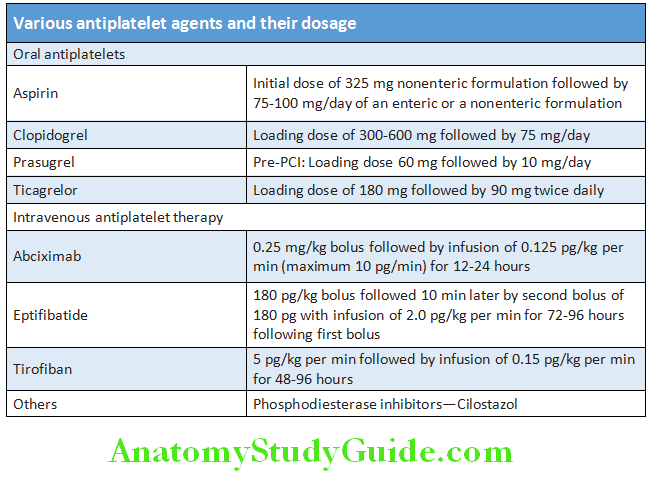
Thrombolytic (or Fibrinolytic) Therapy in Acute Coronary Syndrome:
Question 44. Write short essay/note on indications, contraindications, and therapeutic schedule of thrombolytic therapy in acute myocardial infarction.
Answer:
Indications for Thrombolysis in Acute Myocardial Infarction:
Fibrinolytic therapy should be initiated within 30 minutes (door-to-needle time or first medical contact-to-needle time).
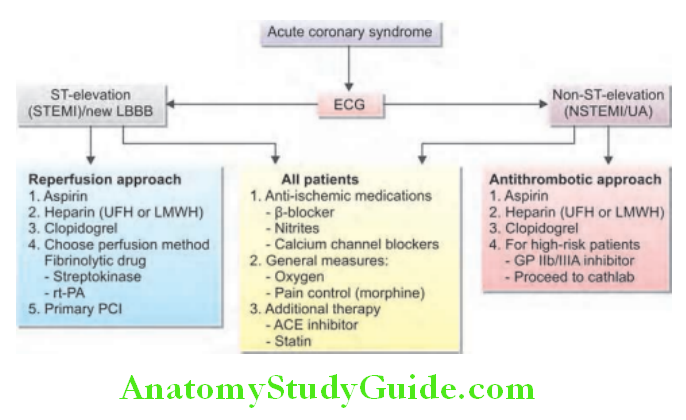
Indications for thrombolysis in acute myocardial infarction:
Thrombolysis Definitely beneficial:
- ST-segment elevation of more than 0.1 mV in two or more contiguous leads, with time to therapy 12 hours or less
- Left bundle branch block (LBBB) obscuring ST-segment analysis and history of acute myocardial infarction for less than 12 hours
Thrombolysis Some benefit:
- ST-segment elevation with time to therapy 12–24 hours
Thrombolysis Not indicated:
- ST-segment depression only (unless leads V1 -V4 show ST depression related to posterior wall myocardial infarction)
- Time to therapy >24 hours

Thrombolytic agents: These include plasminogen activators, i.e., streptokinase (STK), urokinase (UK), human tissue plasminogen activator (tPA-alteplase), recombinant plasminogen activator (rPA-reteplase), tenecteplase, anisoylated plasminogen streptokinase activator complex (AP- SAC, anistreplase), and single-chain urokinase plasminogen activator (scu-PA).
Mechanism of action: Thrombolytic or fibrinolytic agents lyse thrombi/clot to recanalize the occluded vessels (mainly coronary arteries) by the activation of plasminogen to form plasmin. They are curative rather than prophylactic.
- Fibrin-specific fibrinolytics: They generate fibrin-specific fibrinolytics at the site of thrombus/clot. Examples include rPA (reteplase), tenecteplase (TNK), and scu-PA. They have lower mortality rate compared with STK and also lack the
significant acute side effects of hypotension and allergy caused by STK. - Generation plasmin in the systemic circulation: These agents generate plasmin in systemic circulation producing a systemic lytic state. This leads to a reduction in blood viscosity, and produces strong anticoagulant and antiplatelet effects. Examples include streptokinase (STK) and urokinase (UK). STK use is associated with lower incidence of intracranial hemorrhage, especially in older individuals.
Thrombolytic therapy is not recommended for patients with NSTEMI and unstable angina.
Signs of Reperfusion:
Contraindications to Thrombolytic Therapy:
Complications of Thrombolytic Therapy:
- Hemorrhage: It is the major complication. The most common site is in the region of puncture sites, genitourinary system, and intracranial hemorrhage (in about 0.5% of patients).
- Allergic reactions: These may develop with use of STK and APSAC.
- Hypotension: It may develop if STK is infused rapidly.
Thrombolysis in myocardial infarction (TIMI) grading system:
It is a simple qualitative scale to grade the flow in the culprit coronary artery when assessed by angiography. Rescue PCI should be considered in patients with failure of reperfusion (persistent chest pain and ST-segment elevation >90 minutes) with thrombolytic agents.
Signs of reperfusion:
- Immediate relief of chest pain
- Reduction of the initial ST-segment elevation by 50% within 60–90 minutes of fibrinolytic therapy
- Onset of reperfusion arrhythmias (e.g., accelerated idioventricular rhythm and frequent ventricular ectopics)
- Early peaking of CK-MB enzyme
Contraindications to thrombolytic therapy:
Absolute:
- History of cerebrovascular hemorrhage anytime in life
- History of nonhemorrhagic stroke or other cerebrovascular event within the past 1 year
- Uncontrolled marked hypertension (systolic BP >180 mm Hg, diastolic BP >110 mm Hg). However, STK can be given.
- Suspected aortic dissection
- Active internal bleeding (excluding menses)
- Known intracranial aneurysm/AV malformation/neoplasm (primary or metastatic)
- Intracranial/spinal surgery within last 3 months)
Relative:
- Current use of anticoagulants (INR ≥2)
- Recent (<2 weeks) invasive or surgical procedure, prolonged (>10 min) CPR
- Known bleeding diathesis
- Recent trauma (including traumatic resuscitation)
- Pregnancy
- Hemorrhage ophthalmic condition
- Active peptic ulcer disease
- History of severe hypertension that is currently controlled
Thrombolysis in myocardial infarction (TIMI) grading system:
- TIMI 0: Absence of antegrade flow beyond a coronary occlusion
- TIMI 1: Faint antegrade coronary flow beyond the occlusion, although filling of the distal coronary bed is incomplete
- TIMI 2: Flow is delayed or sluggish antegrade flow with complete filling of the distal territory
- TIMI 3: Flow is normal which fills the distal coronary bed completely
Percutaneous Coronary Intervention (PCI):
Percutaneous coronary intervention is the treatment of choice, provided it is performed promptly by a qualified interventional cardiologist in an appropriate facility.
Indications:
Patients with STEMI with following features:
- Symptoms of ischemia of less than 12 hours duration.
- Symptoms of ischemia of less than 12 hours duration who have contraindications to fibrinolytic therapy, irrespective of the time delay from first medical contact.
- Cardiogenic shock or acute severe heart failure (HF), irrespective of time delay from MI onset.
- It may be recommended if there is clinical and/or ECG evidence of ongoing ischemia between 12 and 24 hours after symptom onset. Maximum acceptable delay for primary PCI from presentation to balloon inflation is 60 minutes if a patient presents within 1 hour of symptom of onset or 90 minutes if a patient present later.
Types of PCI: Types include primary PCI, rescue PCI, and facilitated PCI (described below).
Glycoprotein IIb/IIIa inhibitors (e.g., abciximab and tirofiban) may be used in patients undergoing percutaneous interventions.
Reperfusion Options for STEMI:
- Fibrinolysis:
- It is usually preferred in the following situations:
- Early presentation (≤3 hours from symptom of onset)
- In patients where primary PCI cannot done because of the following:
- Catheterization laboratory occupied/unavailable
- Vascular access difficulties
- Lack of access to a skilled PCI laboratory
- Delay to primary PCI:
- Door-to-balloon time minus door-to-needle time is >1 hour.
- Door-to-balloon time is >90 minutes.
- Primary PCI: In which PCI is used solely in acute MIt is indicated in cardiogenic shock, and in patients in whom thrombolytic therapy is contraindicated.
- It is generally preferred with following conditions:
- Skilled PCI laboratory is available with good surgical backup.
- Door-to-balloon time is ≤90 minutes.
- Door-to-balloon time minus door-to-needle time is ≤1 hour.
- High-risk STEMI:
- Cardiogenic shock
- Killip class CHF ≥3
- Contraindications to fibrinolysis including increased risk of bleeding and intracranial hemorrhage.
- Late presentation (>3 hours after symptom of onset).
- Rescue PCI: It is combination of PCI with thrombolytic therapy and PCI is performed within 12 after failed thrombolysis/fibrinolysis for patients with continuing or recurrent myocardial ischemia.
- Indications: STEMI in aged <75 years who received fibrinolytic therapy and have cardiogenic shock, severe congestive heart failure (Killip class III), or hemodynamically compromising ventricular arrhythmias.
- It may be performed in patients with symptoms of persistent ischemia.
- Skilled PCI laboratory is available with good surgical backup.
- Facilitated PCI: In this type PCI is done following initial pharmacological regimen aimed at improving patency of coronary arteries before PCI.
- The pharmacological regimens include GB IIB/IIIa inhibitors, full-dose or reduced-dose of fibrinolytic therapy, and combination of a GP IIb/IIIa inhibitor, and a reduced-dose fibrinolytic/thrombolytic agent. However, this type of reperfusion may be inferior to thrombolysis alone or primary PCI and is usually not recommended in most patients with STEMI.
Coronary Artery Bypass Grafting:
- Recommended in:
- Failed PCI with persistent pain or hemodynamic instability in patients with coronary anatomy suitable for surgery.
- Persistent or recurrent ischemia refractory to medical therapy in patients who have coronary anatomy suitable for surgery, and are not candidates for PCI or fibrinolytic therapy.
- Patients with STEMI at the time of operative repair of mechanical defects.
Management of Complications:
- Ventricular dysfunction:
After STEMI ventricular remodeling occurs, ventricular dysfunction can be prevented by ACE inhibitors and nitrates.
Hemodynamic classification of ventricular dysfunction.
This classification has prognostic value with highest mortality in class IV patients. - Cardiogenic shock:
- This results when there is infarction of ≥40% of left ventricle. The patient has systolic BP <90 mm Hg and pulmonary capillary wedge pressure >18 mm Hg.
- Cardiogenic shock is best managed by invasive means (Primary PCI). Use of intra-aortic balloon pump and IV vasopressors may be beneficial.
- Right ventricular infarction (discussed later):
- Clinically significant right ventricular infarction is rare and occurs in patients with inferior infarction.
Hemodynamic classification of patients with acute myocardial infarction:

Pericarditis:
- Common complication in first week of transmural myocardial infarct.
- Radiation of pain to trapezius muscle is helpful in distinguishing it from ischemia.
- Pericardial rub may be present.
Treatment of pericarditis:
- Aspirin 650 mg qid and withhold anticoagulants.
- Management of other complications:
Treatment of pericarditis These include:
- Thromboembolism,
- Left Ventricular Aneurysm,
- Sinus Bradycardia,
- AV Block
- Ventricular tachycardia
Fibrillation and acute mitral regurgitation, VSR, and tricuspid regurgitation.
Their management is discussed in individual sections.
Aftercare and Rehabilitation:
- Physical activities: To be restricted for 4–6 weeks because replacement of infarct by fibrous tissue takes 4–6 weeks. Advised gradual mobilization and return to work over 6 weeks. Exercise and sexual activity within the limits.
- Complications: Patients who had complications, the regimen depends on the type of complication.
- Lifestyle and risk factor modification: Control of risk factors such as obesity by regular exercises, cessation of smoking, lifestyle modifications, and control of plasma lipids by diets and drugs.
- Secondary prevention drug therapy:
- Aspirin and clopidogrel: Low-dose aspirin (75–150 mg daily) unless there is any contraindication. Clopidogrel (75 mg daily) for up to 12 months, particularly after stent implantation. It may be given as an alternative when aspirin is contraindicated, or in combination with aspirin particularly in patients with unstable angina or recurrent cardiac events.
- β-blocker: Oral β-blockers are continued indefinitely (unless any contraindications). Carvedilol, bisoprolol, or metoprolol (extended release) are given to patients with heart failure. Role ofβ-blockers in the secondary prevention in unstable angina is not known.
- ACE inhibitor is given early after an acute coronary syndrome. Long-term treatment with an ACE inhibitor (e.g., enalapril 10 mg twice daily or ramipril 2.5–5 mg twice daily) is found to counteract ventricular remodeling, prevent the onset of heart failure, and reduce recurrent MI.
- Statin therapy started in the hospital for all patients with coronary artery disease.
- Warfarin after myocardial infarction is given to patients having a high-risk of systemic thromboembolism due to atrial fibrillation, mural thrombus, congestive heart failure, or previous embolization.
- Nitrates: Short-acting nitrates, for chest pain. Long-acting nitrates are given for relief of symptom when β-blocker alone is unsuccessful or is contraindicated.
- Aldosterone antagonist (e.g., eplerenone) is given early after myocardial infarction to patients who have LVEF ≤40%, despite optimum dose of ACE inhibitors and β-blockers, and have either CHF or diabetes.
- Device therapy: Implantable cardiac defibrillators can prevent sudden cardiac death in patients who have severe left ventricular impairment (ejection fraction ≤30%) after MI.
Non-ST-Segment-Elevation Acute Coronary Syndrome (NSTEACS):
Question 45. Briefly discuss unstable angina and non-ST-elevation myocardial infarction.
Answer:
- It includes unstable angina (UA) and non-ST-elevation myocardial infarction (NSTEMI).
- Both are caused by coronary artery spasm, progression of the underlying coronary artery disease (CAD), or hemorrhage into a nonoccluding atheromatous plaque with subsequent thrombosis producing coronary obstruction over a period of few hours. The difference between UA and NSTEMI is that the NSTEMI shows an occluding thrombus, which leads to myocardial necrosis and a rise in serum troponins or CKMB.
Non-ST-elevation MI (NSTEMI):
- It usually shows ST depression and T inversion in the ECG along with elevation in serum troponins or CKMB.
- Myocardial function (as shown by ejection fraction) in NSTEMI is less deranged when compared to STEMI. However, in NSTEMI early as well as late reinfarction rates are higher than in STEMI.
Unstable Angina (UA):
Question 46. Write a short essay/note on unstable angina.
Answer:
- Three principal presentations include:
- Rest angina: Angina occurring at rest and prolonged, usually >20 minutes.
- New-onset angina: New-onset angina of at least CCS Class III severity.
- Increasing angina: Previously diagnosed angina that has become distinctly more frequent, longer in duration, or lower in threshold (i.e., increased by >1 CCS) class to at least CCS Class III severity.
- Normal ECG (no ST elevation), without elevation of cardiac injury markers (normal level of cardiac enzymes).
- Patients with UA have a high risk of developing MI or sudden death when compared to patients with stable angina. Thus, they need aggressive treatment in the hospital.
Classification of risk categories in NSTEMI/UA and its management are mentioned.
Right Ventricular Myocardial Infarction (Rvmi):
- Right ventricular myocardial infarction is seen in patients with an acute inferior MI secondary to complete occlusion of the proximal RCA.
- Right ventricular function is very preload dependent, and frequently, hypotension responds to fluid resuscitation.
- The clinical triad of hypotension, elevated jugular venous pressure, and clear lung fields in the setting of STEMI should prompt an evaluation for RV infarct or massive pulmonary embolism.
- One-mm ST elevations in the V1 or V4R leads are the most sensitive marker of RV involvement.
- Initial therapy is IV fluids. If hypotension persists, inotropic support with dobutamine and/or IABP may be necessary. Right-sided mechanical support devices are available when pharmacologic support fails.
- Management protocol is same as other ACS. Caution should be exercised while using nitrates and beta blockers.
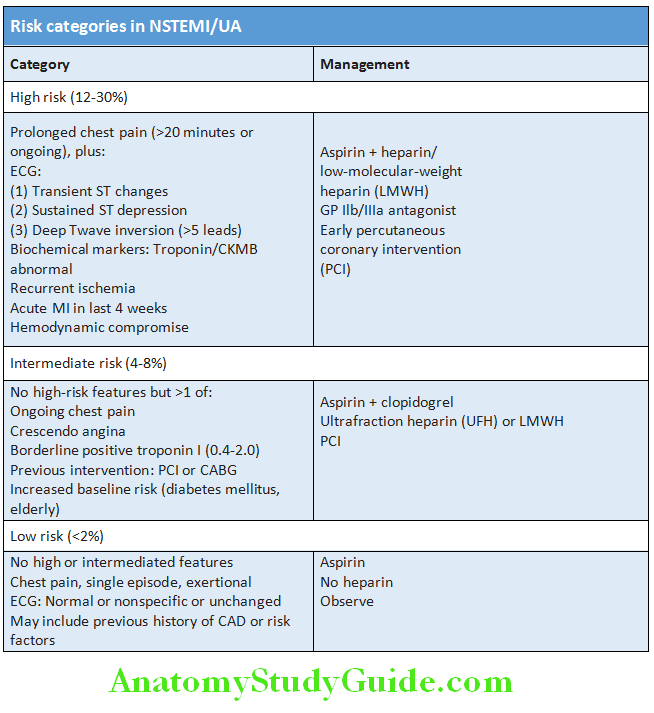
Prinzmetal’s Variant Angina:
Question 47. Write a short essay/note on clinical features and treatment of Prinzmetal’s angina (variant angina).
Answer:
In 1959 Prinzmetal et al. described a syndrome of severe ischemic pain that usually occurs at rest and is associated with transient ST-segment elevation.
Prinzmetal’s Variant Angina Etiology:
- Focal spasm of an epicardial coronary artery is the cause of Prinzmetal’s variant angina (PVA) and leads to severe transient myocardial ischemia and occasionally infarction.
- The cause of the spasm is not well defined, but it may be related to hypercontractility of vascular smooth muscle due to adrenergic vasoconstrictors, leukotrienes, or serotonin.
Prinzmetal’s Variant Angina Clinical Features:
- Pain occurs without exertion and usually at rest. It is more frequent in women.
- Associated with migraine, Raynaud’s phenomenon, and aspirin-induced asthma. Younger patients with history of cigarette smoking.
Prinzmetal’s Variant Angina Investigation:
- Characteristically, it is associated with transient ST-segment elevation on the ECG during the pain.
- Coronary angiography is gold standard for diagnosis. Focal spasm commonly accompanied by stenosis within 1 cm of spasm is the hallmark (most commonly in right coronary artery).
- Provocation tests (e.g., hyperventilation, cold pressor testing, or ergometrine or intracoronary acetylcholine challenge) may be needed for demonstration of focal spasm and establishing the diagnosis.
Prinzmetal’s Variant Angina Management:
- Nitrates and calcium channel blockers are the main therapeutic agents. Aspirin may actually increase the severity of ischemic episodes, possibly as a result of the sensitivity of coronary tone to modest changes in the synthesis of prostacyclin.
- The response to β-blockers is variable but usually poor. Prazosin can be useful.
- Coronary revascularization may be helpful in patients with discrete, flow-limiting, proximal fixed obstructive lesions.
Prinzmetal’s Variant Angina Prognosis:
- Many patients pass through an acute, active phase, with frequent episodes of angina and cardiac events during the first 6 months after presentation.
- Prognosis is better in patients with no or mild fixed coronary obstruction than patients with severe, fixed, significant obstructive lesions. Survival at 5 years is excellent (90–95%).
- Nonfatal MI occurs in up to 20% of patients by 5 years.
- Patients with PVA who develop serious arrhythmias during spontaneous episodes of pain are at a higher risk for sudden cardiac death. In most patients who survive an infarction or the initial 3- to 6-month period of frequent episodes, there is a tendency for symptoms and cardiac events to diminish over time.
Hypertension:
Question 48. Classify hypertension. Discuss the clinical features, baseline investigations, diagnosis, complications, and management of essential hypertension.
(or)
Discuss the causes and investigations of secondary hypertension.
Answer:
Hypertension is a hemodynamic disorder and about 15% of the general population can be regarded as hypertensives.
Hypertension Definition: A well-accepted definition of hypertension was suggested by Evans and Rose:
- “Hypertension should be defined in the terms of blood pressure level above which investigation and treatment do good
more than harm.” - The following diagnostic criteria has been suggested by the 2017 American College of Cardiology/American Heart Association (ACC/AHA) guidelines; meeting one or more of these criteria using ambulatory blood pressure monitoring (ABPM) to qualify as hypertension.
- A 24-hour mean of 125/75 mm Hg or above
- Daytime (awake) mean of 130/80 mm Hg or above
- Nighttime (asleep) mean of 110/65 mm Hg or above.
Hypertension is defined arbitrarily at levels above generally accepted normal (Joint National Committee—JNC 7/8 recommendations) and is presented in Table 7.47A. Other guidelines for classification of hypertension are summarized in Table
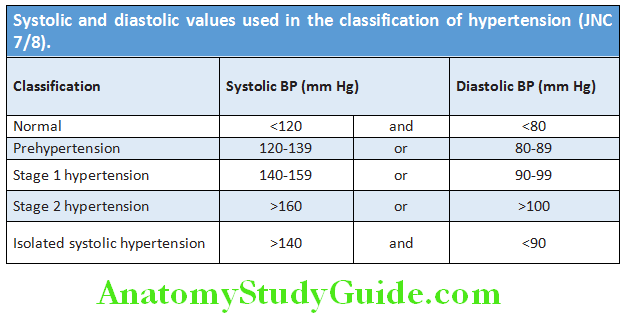
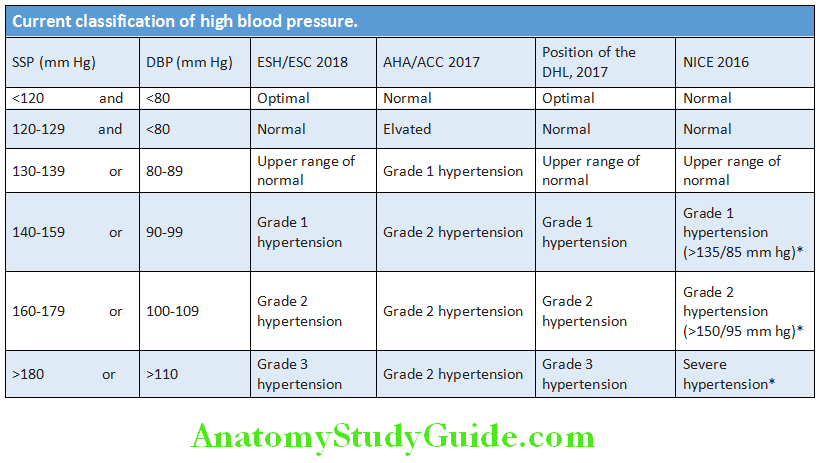
Classifiation and Causes:
Question 49. Write short note on the causes of hypertension.
Answer:
- Primary or essential hypertension: It constitutes about 85% of the cases in which it is not possible to define a specific underlying cause. About 70% of these patients give a positive family history.
- Secondary hypertension: It constitutes remaining 15% of the cases and is due to a specific disease or abnormality.
Question 50. Write short essay or note on secondary hypertension and its causes.
(or)
Write short note on causes of isolated systolic hypertension.
Answer:
Essential Hypertension:
In about 85–95% of cases, a specific underlying cause of hypertension cannot be identified and such hypertension is termed as essential hypertension. The exact pathogenesis is not known. It has multifactorial etiology and many factors may contribute to its development.

Etiology:
Question 51. Write short essay or note on risk factors for hypertension.
Answer:
Genetic factors: Blood pressure tends to run in families and this may be partly due to environmental influences. Environmental factors: Several environmental factors may be involved and these include salt intake, obesity, occupation, alcohol intake, family size, and crowding.
- Obesity: Higher blood pressures are seen in obese individuals compared to thin individuals. Sleep disordered breathing/obstructive sleep apnea often observed with obesity may be an additional risk factor.
- Alcohol intake: There is close relationship between the consumption of alcohol and blood pressure level.
- Sodium intake: Higher sodium intakes may be associated with an increase in blood pressure. A high potassium diet can have protective role against the effects of a high sodium intake. About 60% of hypertensive are salt sensitive. Primary aldosteronism, bilateral renal artery stenosis, renal parenchymal disease, and low-renin essential hypertension are all salt sensitive.
- Stress: Acute pain or stress is associated with raised blood pressure. However, the relationship between chronic stress and blood pressure is not known.
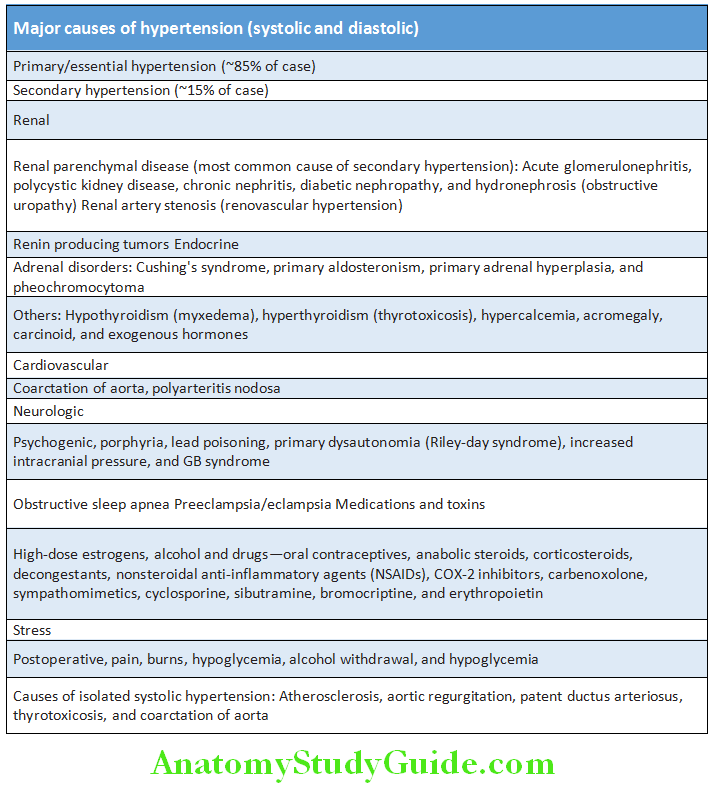
Humoral Mechanisms:
The autonomic nervous system, the renin–angiotensin, natriuretic peptide, and kallikrein kinin system play a role in the physiological regulation of short-term changes in blood pressure. They may also be probably involved in the pathogenesis of essential hypertension.
- A low-renin, salt-sensitive, essential hypertension in which patients have renal sodium and water retention has been found. Low-renin hypertension is more common in elderly and diabetics. These patients are salt-sensitive and diuretic responsive.
- Normal renin hypertension (nonmodulators) is more common in males and postmenopausal females. They are salt sensitive.
- High-renin hypertension is characterized by high plasma renin activity and responsiveness to angiotensin II antagonists.
- Low calcium intake has been associated with an increase in blood pressure in epidemiologic studies. Insulin resistance is responsible for essential hypertension in majority of the patients.
- Metabolic syndrome: It is characterized by hyperinsulinemia, glucose intolerance, reduced levels of HDL cholesterol, hypertriglyceridemia, and central obesity (all related to insulin resistance). Metabolic syndrome is associated with hypertension and is a major risk factor for cardiovascular disease.
Fetal factors:
Impaired intrauterine growth resulting in low-birth weight is associated with subsequent development of high blood pressure.
Approach to Newly Diagnosed Hypertension:
Question 52. Discuss the approach to a case of hypertension in the young.
Answer:
Hypertension is usually asymptomatic and the diagnosis is usually made at routine examination or when a complication arises. A routine BP checkup is necessary every 5 years in adults.
Goals of the initial evaluation with high BP are to:
- Obtain accurate BP measurements
- Identify contributing factors, and risk factors and any underlying cause (secondary hypertension)
- Quantify cardiovascular risk
- Detect any complications (target organ damage)
- Identify comorbidity that may influence the choice of antihypertensive therapy. These are attained by a careful history, clinical examination, and few simple investigations.
Ambulatory Blood Pressure Monitoring (ABPM):
Question 53. Write short note on ambulatory blood pressure monitoring.
Answer:
Ambulatory blood pressure monitoring is the preferred method for confirming the diagnosis of hypertension. High-quality data suggest that ABPM predicts target-organ damage and cardiovascular events better than office blood pressure readings. ABPM records the blood pressure at preset intervals (usually every 15 to 20 minutes during the day and every 30 to 60 minutes during sleep).
Home and Ambulatory BP Recordings:
- A transient rise in BP may occur by exercise, anxiety, discomfort, and unfamiliar surroundings.
- White coat hypertension: It is a transient increase in blood pressure in normal individuals when blood pressure is recorded either in a hospital or in a physician’s clinic.
- Isolated ambulatory or masked hypertension:
- It is reversal of white coat hypertension in which individuals have normal blood pressure (<140/90 mm Hg) in a hospital or in a physician’s clinic but have increased ambulatory or home blood pressure values.
- These individuals have increased prevalence of organ damage, with an increased prevalence of metabolic risk factors.
Uses of ambulatory blood pressure monitoring:
- Confirm white coat and masked hypertension
- Suspected episodic hypertension (e.g., pheochromocytoma)
- Determining therapeutic response (i.e., blood pressure control) in patients who are known to have a substantial white coat effect)
- Hypotensive symptoms while taking antihypertensive medications
- Resistant hypertension
- Autonomic dysfunction
History:
- Record family history, lifestyle (exercise, salt intake, and smoking), other risk factors, history of drug intake or alcohol.
- Symptoms of causes of secondary hypertension (Table 7.49) or complications such as coronary artery disease (e.g., angina and breathlessness).
Clinical Features:
Clinical features of hypertension may be due to hypertension itself and the underlying cause of hypertension.
Clinical features due to hypertension per se:
- Majority of patients are asymptomatic and hypertension is usually detected during routine examination.
- Acute hypertension may produce transient headache and polyuria.
- Long-standing hypertension may cause left ventricular hypertrophy and heaving apical impulse, accentuation of the aortic component of the second heart sound (A2), a fourth heart sound (S4), very short early diastolic murmur, and fundal changes (refer complications).
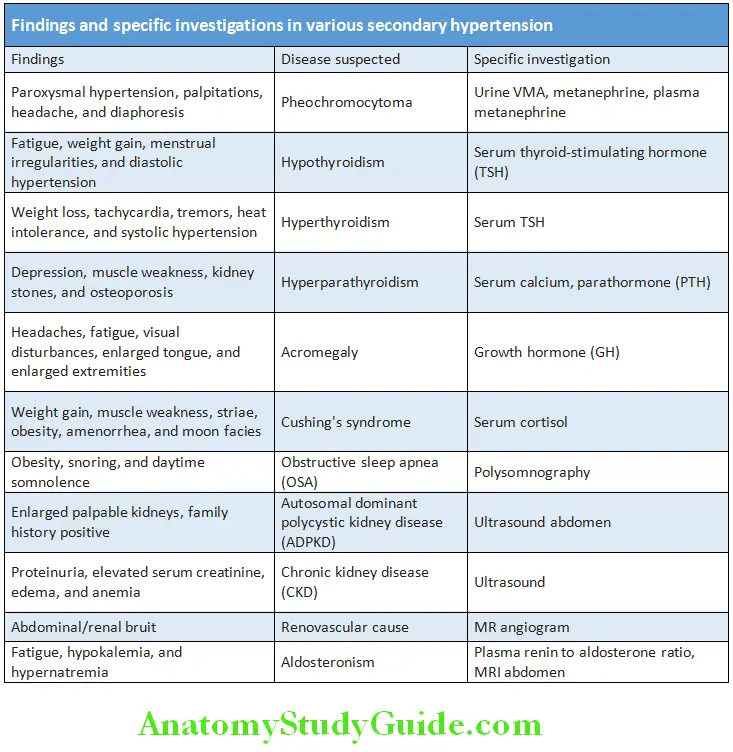
Objectives of clinical examination:
- Identify any underlying cause of hypertension
- Recognize risk factors that lead to complications
- Abnormal signs to detect complications if already developed.
Nonspecific findings: Optic fundi may be abnormal; there may be evidence of generalized atheroma or specific complications, such as aortic aneurysm or peripheral vascular disease. Findings and specific investigations in various secondary hypertension are mentioned.
Question 54. Write short note on causes and investigations of secondary hypertension.
(or)
Write short essay or note on complications of hypertension/target organ damage in systemic arterial hypertension.
Answer:
Target organ damage in hypertension can be clinically detected.
Target Organ Damage (Complications of Hypertension):

Central Nervous System Complications:
- Transient ischemic attacks (TIAs): Carotid atheroma and TIAs are more common in patients with hypertension.
- Cerebrovascular accidents (strokes) are a common complication of hypertension and may be due to cerebral hemorrhage or infarction (due to cerebral atherothrombosis).
- Subarachnoid hemorrhage is also a complication of hypertension.
- Hypertensive encephalopathy is a rare complication characterized by very high blood pressure, neurological manifestations (includes transient disturbances in speech and vision, paresthesias, seizures, disorientation, and loss of consciousness), and papilledema.
Ophthalmic (Retinal) Complications:
Question 55. Write short essay or note on grades of hypertensive retinopathy.
Answer:
Hypertensive retinopathy:
Cardiovascular Complications:
- Coronary artery disease (angina, myocardial infarction) Increased cardiac mortality and morbidity associated with hypertension are chiefly due to a higher incidence of coronary artery disease. High BP may produce left ventricular hypertrophy with a forceful apex beat and fourth heart sound. ECG or echocardiography is useful in risk assessment.
- Left ventricular failure and pulmonary edema: It may develop with severe hypertension.
- Atrial fibrillation is common and may be due to diastolic dysfunction caused by left ventricular hypertrophy or the effects of coronary artery disease.
Hypertensive retinopathy Blood Vessels:
Larger arteries show thickening of the internal elastic lamina, hypertrophy of smooth muscle, and deposition of fibrous tissue. Smaller arteries show hyaline arteriosclerosis in the wall, narrowing of the lumen, and aneurysm. Atherosclerosis may develop in coronary and cerebral blood vessel. Hypertension is a major risk factor involved in the pathogenesis of aneurysm of aorta and dissecting hematoma of aorta (aortic dissection).
Renal Complications:
Long-standing hypertension may cause to damage the renal vasculature and produce
- Proteinuria
- Hematuria
- Progressive renal failure.
Hypertensive retinopathy Investigations:
Basic investigations in all patients:
- Urinalysis: For protein, blood, and glucose.
- Blood urea and creatinine: To assess renal function.
- Serum electrolytes: For hypokalemia (is usually due to diuretic therapy) and alkalosis in hyperaldosteronism
- Fasting and postprandial blood glucose: For hyperglycemia
- Lipid profile: Serum total cholesterol and HDL cholesterol
- Electrocardiogram: 12-lead ECG for left ventricular hypertrophy and coronary artery disease.
Secondary Investigations in Selected Patients:
- Chest radiograph: For detecting the cardiac size, evidence of cardiac failure and aortic dilatation, coarctation of the aorta
- Ambulatory BP recording: To detect borderline, masked hypertension or “white coat” hypertension
- Echocardiogram: To detect or quantify left ventricular hypertrophy and function
- Renal ultrasound: To detect renal disease
- MRI, renal isotope scan, and renal angiography: To detect or confirm renal artery stenosis
- Urinary catecholamines: To detect pheochromocytoma
- Urinary cortisol and dexamethasone suppression test: To detect Cushing’s syndrome
- Thyroid function tests: To detect hypothyroidism or hyperthyroidism
- Serum calcium and parathyroid hormone level: To detect hyperparathyroidism
- Plasma renin activity and aldosterone: To detect possible primary aldosteronism
- Growth hormone: To detect acromegaly
Hypertensive retinopathy Treatment:
- Objective of antihypertensive therapy: To reduce the incidence of adverse cardiovascular events (e.g., coronary artery disease), stroke, and heart failure.
- Target blood pressure: For most patients, a target is <140 mm Hg systolic blood pressure and ≈85 mm Hg diastolic blood pressure. For patients with diabetes, renal impairment, or established cardiovascular disease the target is <130/80 mm Hg.
Management of hypertension can be studied under three headings.
- General measures
- Antihypertensive drug therapy
- Treatment of underlying cause (in secondary hypertension).
General Measures:
Question 56. Write short essay/note on nonpharmacological treatment of hypertension.
Answer:
- Lifestyle modifications: Recommended for all patients with hypertension and prehypertension. A reduction in systolic blood pressure of 5 mm Hg has been associated with about 10% reduction in mortality caused by stroke and heart disease:
- Control ofobesity: Maintain normal body weight (BMI 20–25 kg/m2)
- Diet: Dietary Approaches to Stop Hypertension or DASH eating plan.
- Restrict salt in the diet (<100 mEq sodium or <6 g NaCl or <2.4 g Na/day).
- Reduce intake of fat and saturated fat.
- Increase consumption of diet rich in fruit and vegetables (≥5 portions of fresh fruit and vegetables/day), potassium.
- Limit/reduce alcohol consumption to ≤3 units/day men and ≤2 units/day women.
- Cardiovascular risk reduction: Stop/avoid cigarette smoking and increase intake of oily fish.
- Regular aerobic/physical exercises: Perform ≥30 minute brisk walk most days of the week. Relaxation classes, meditation, and biofeedback.
Question 57. Write short essay or note on various types of antihypertensive drugs.
(or)
Write short note on α-adrenergic blockers.
Answer:
Antihypertensive Drug Therapy:
Diuretics:
- Indications include heart failure and elderly and systolic hypertension
- Thiazide diuretics:
- Adverse drug reactions: These include impotence, postural hypotension, allergic rashes, bone marrow depression, hypokalemia, hyperuricemia, hypercalcemia, and impaired glucose tolerance/diabetes.
- Used as first-line agent in elderly patients with systolic hypertension. If blood pressure is not controlled with a thiazide diuretic, add ACE inhibitors, angiotensin receptor blockers, or calcium channel blockers.
- Loop diuretics: Furosemide or others are used if there is renal impairment or when greater sodium excretion is needed or they are used in conjunction with an ACE inhibitor.
β-adrenoreceptor Antagonists (β-blockers):
Question 58. Write short essay or note on β-blockers, uses, and its side effects.
Answer:
- Uses and contraindications for β-blockers are mentioned. Beta blockers are no longer used as first-line antihypertensive therapy.
They may be useful in following situations in:
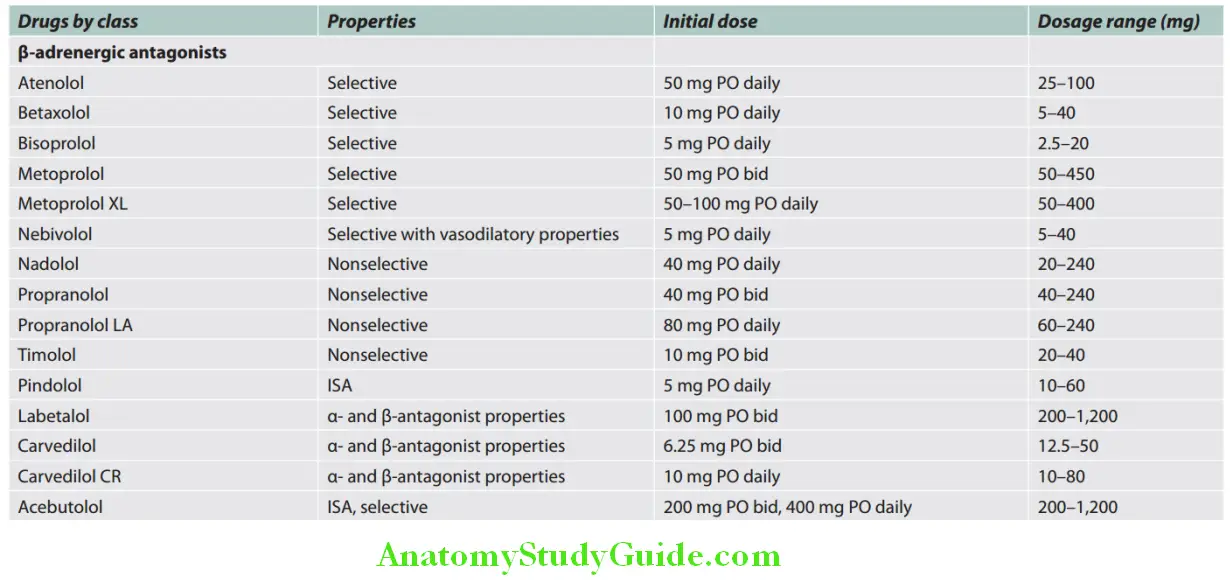
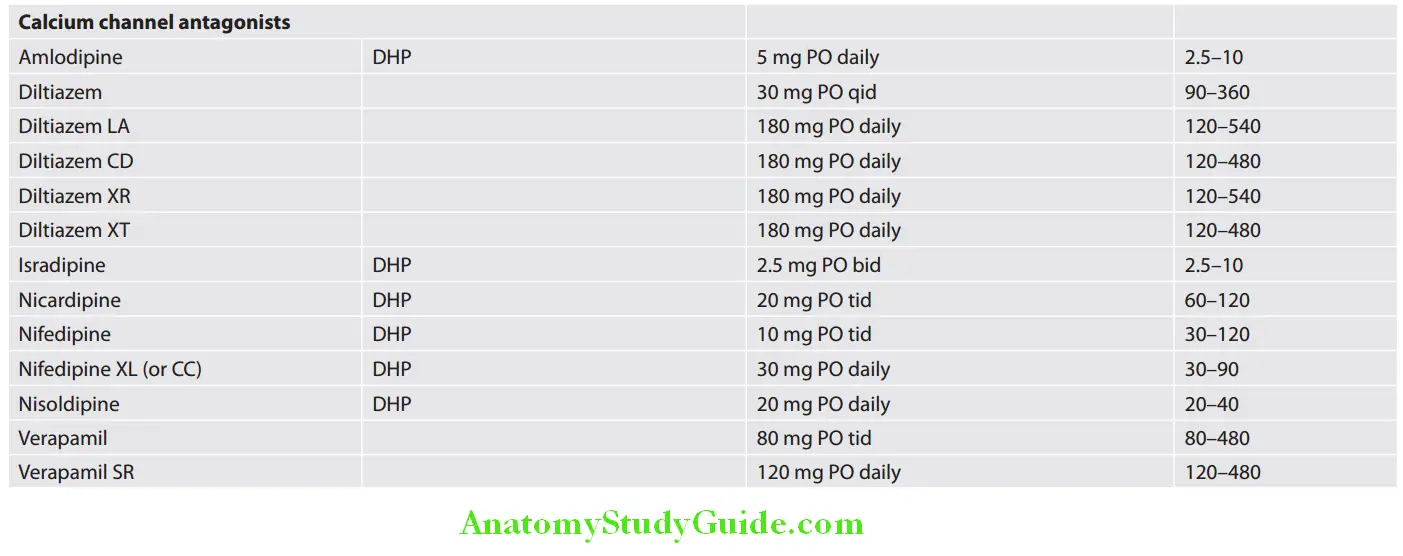
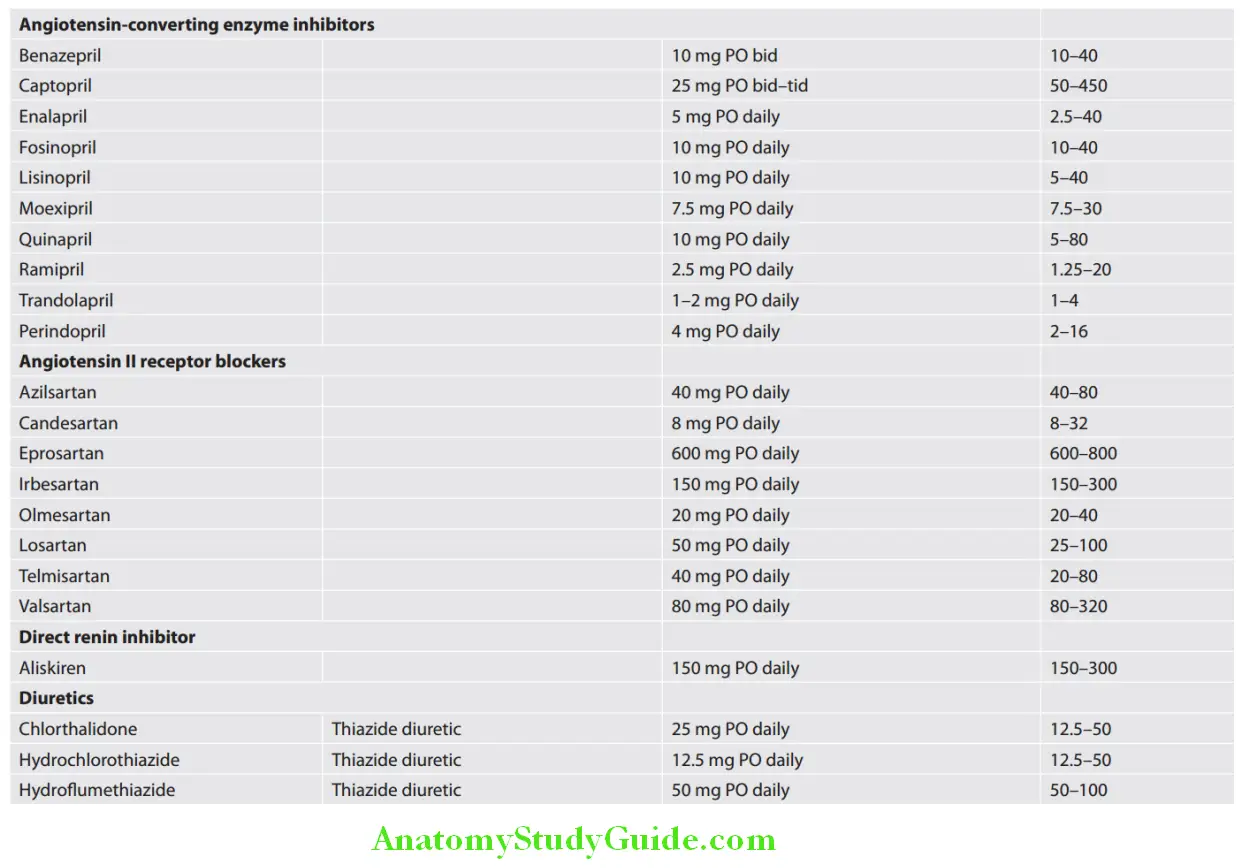

- Younger individuals, particularly who have an intolerance or contraindication to ACE inhibitors and angiotensin II receptor antagonists.
- Women of childbearing potential
- Patients with increased sympathetic drive
- Patients having hypertension and other diseases, such as coronary heart disease (postmyocardial infarction or angina) and heart failure.
Nonselective (β1 and β2):
- Mechanism of action: Sympatholytic effect, antihypertensive effect, and relief of anxiety, palpitation and angina. Large portion of the drug is destroyed during its passage through liver, e.g.:

Individualizing Antihypertensive Therapy:
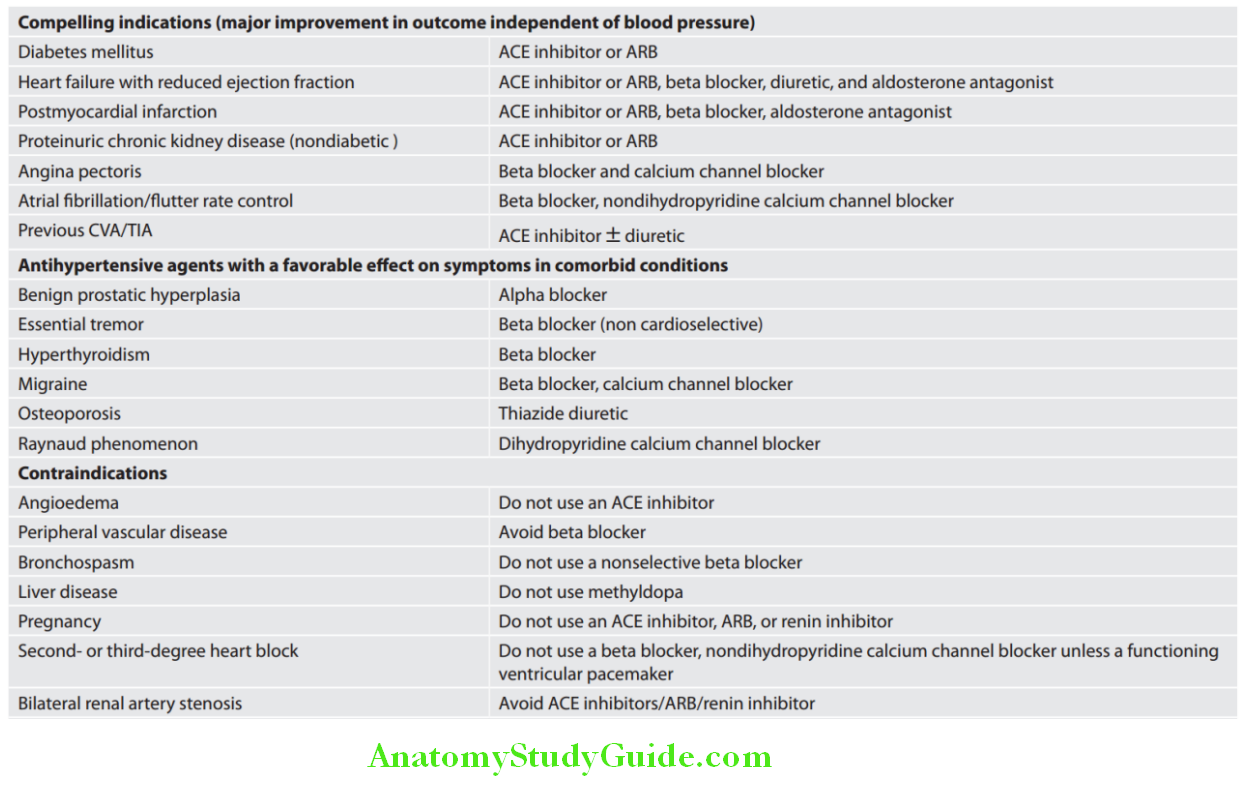
- Dosage: Started with 40 mg twice a day and gradually increased to 160 mg 6 hourly. Slow-release forms are administered as a single daily dose, e.g., propranolol
- Side effects: Gastric disturbances, bronchospasm, bradycardia, cardiac failure, tiredness, bad dreams, hallucinations, cold hands, and muscle weakness.
Cardioselective (β1):
- Mechanisms of action: They act more on the cardiacβ1-adrenoceptors thanβ2-adrenoceptors that mediate vasodilatation and bronchodilatation. Examples: Metaprolol and atenolol
- Dose: Metaprolol 50 mg twice daily to 100 mg thrice daily, atenolol 50–100 mg once daily and bisoprolol 5–10 mg daily. Sustained release preparation of metoprolol is given in the dose of 12.5–100 mg once daily.
- Indications:
- Hypertensive with mild airway obstruction (COPD, asthma), peripheral vascular disease, and type 1 diabetes. However, caution is necessary.
- Ischemic heart disease and supraventricular tachycardia.
- Side effect: Similar to propranolol and also cause hyperkalemia.
Combined α- and β-blockers (Labetalol and Carvedilol):
- Sometimes, these combined β- and α-adrenoceptor antagonists are more effective than pure β-blockers.
- Mechanism of action and side effects: Similar to propranolol.
- Dose: Labetalol 100–200 mg twice daily and carvedilol 6.25–25 mg twice daily. Labetalol can be used as an infusion in the treatment of malignant phase hypertension.
Angiotensin-converting Enzyme (ACE) Inhibitors:
Question 59. Write short essay or note on mechanism of action, indications, and side effects of angiotensin-converting enzyme (ACE) inhibitors.
(or)
Write short note on ramipril.
Answer:
- These include captopril, enalapril (20 mg daily), lisinopril (10–40 mg daily), ramipril (5–10 mg daily), and perindopril.
- Mechanisms of action: They inhibit the conversion of angiotensin I to angiotensin II and produce powerful arteriolar and venous dilatation, and inhibit release of aldosterone.
- Dosage: Start with a small dose (captopril 6.25 mg and enalapril 2.5 mg) and then gradually increase.
- Indication: Used in hypertension, chronic cardiac failure or left ventricular dysfunction, postmyocardial infarction or cardiovascular disease, diabetic nephropathy, and stroke secondary prevention.
- Contraindication: Renal failure (creatinine >3 mg/dL) and bilateral renal artery stenosis because they can reduce the filtration pressure in the glomeruli and precipitate renal failure. Electrolytes and creatinine levels should be checked before and 1–2 weeks after starting therapy. Other contraindications include peripheral vascular disease hyperkalemia and pregnancy.
- Adverse reaction: First-dose postural hypotension, cough, angioedema, skin rashes, blood dyscrasias, hyperkalemia, renal dysfunction neuropathy, and diarrhea.
Peripheral Vasodilators:
- Mechanism of action: Peripheral vasodilators act on arteriolar smooth muscles or smooth muscles of venules and produce peripheral arteriolar and venous dilatation.
- Drugs include hydralazine, prazosin (alpha blocker), diazoxide, and sodium nitroprusside.
Angiotensin Receptor Blockers:
Question 60. Write short essay or note on angiotensin receptor blockers and indications.
Answer:
- Mechanism of action: They block the angiotensin II type I receptor and have similar effects to ACE inhibitors. Reninangiotensin–aldosterone system (RAAS) plays an important role in the regulation of blood pressure.
- Renin catalyzes cleavage of angiotensinogen and produces angiotensin I.
- Angiotensin I is converted to angiotensin II by the action of ACE. Angiotensin II is the most active hormone of the renin–angiotensin system. There are two angiotensin II receptors, namely AT1 (most effective) and AT2 (its function is not established).
- When angiotensin II binds to its AT1 receptor, it produces many effects such as vasoconstriction, cell proliferation, hypertrophy, and aldosterone secretion and provides feedback inhibition of further renin release by the kidney.
- The angiotensin receptor blockers block the action of angiotensin II by blocking AT1 receptors.
- Dose: Losartan 50–100 mg daily, candesartan 2.0–32 mg daily, valsartan 40–320 mg daily, telmisartan 40 mg daily, olmesartan 10–40 mg daily, and irbesartan and azilsartan (40–80 mg daily).
- Side effects: These drugs are better tolerated than ACE inhibitors. Side effects include hypotension, drowsiness, and dizziness. These drugs do not cause cough.
- Indications: ACE inhibitor intolerant hypertension, <55 years old, hypertension with LVH, heart failure or left ventricular dysfunction, myocardial infarction (to reduce myocardial remodeling), diabetic nephropathy (to slow its progression), and chronic renal disease.
Calcium Channel Blockers (Calcium Antagonists):
Question 61. Write short essay or note on uses/indications of calcium channel blockers and its side effects.
Answer:
- They are effective and usually well-tolerated antihypertensive drugs. Various indications of calcium channel blockers.
- Contraindications: Unstable angina, heart failure, hypotension, postinfarct patients, severe aortic stenosis, and second- and third-degree AV block.
- Side effects: Flushing, palpitations, and fluid retention. The main side effect of verapamil is constipation.
Nifedipine and Verapamil:
Dosage: Nifedipine 10–20 mg three to four times daily and verapamil 180–360 mg/day in divided doses. Short-acting nifedipine should be avoided.
Centrally Acting Drugs:
Reserpine: It is a mild antihypertensive with central and peripheral action. It is given in the dose of 0.1–0.5 mg daily. Its side effects include nasal congestion, depression, and Parkinsonism.
α-methyldopa: It is a precursor of dopamine and noradrenaline.
- Mechanism of action: Converted to α-methyl noradrenaline which acts on alpha-2 receptors in brain and causes inhibition of adrenergic discharge in adrenal medulla → fall in peripheral vascular resistance and fall in blood pressure.
- Side effects: Cognitive impairment, postural hypotension, positive Coombs test, etc. Not used therapeutically now except in hypertension during pregnancy.
- Dose: 250–500 mg twice or thrice daily.
Clonidine: Not frequently used because of tolerance and withdrawal hypertension. Side effect is dryness of mouth.
Dose: 0.1–1.0 mg daily.
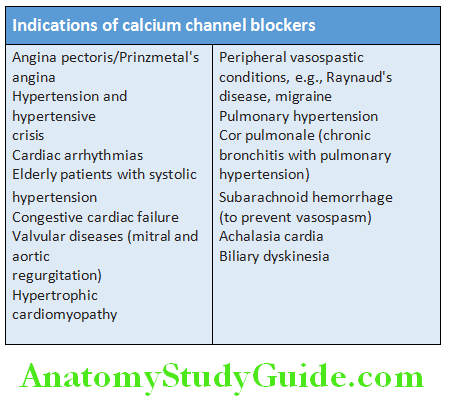
Guidelines for Starting Antihypertensive Agents:
Recommendations in Hypertension Management Suggested by JNC8:
Question 62. Discuss the recommendations in hypertension management suggested by JNC8.
Answer:

Hypertensive Encephalopathy:
Question 63. Discuss briefly about hypertensive crisis/hypertensive emergencies.
Answer:
- It is more generalized term and is not defined by a specific blood pressure reading.
- It develops in previously hypertensive/normotensive patients.
- It is a clinical syndrome that is associated with acute severe or abrupt elevation of blood pressure. Characterized by severe increase in systolic and/or diastolic blood pressure associated with signs or symptoms of acute end-organ damage.
- No blood pressure threshold for diagnosis. Usually, systolic blood pressure >180–220 mm Hg, diastolic blood pressure >120–130 mm Hg, and mean arterial pressure (MAP) >180 mm Hg
- Increased cerebral blood flow→ hyperfiltration→ localized or diffuse cerebral edema → cerebral ischemia resulting from arteriolar spasm.
- Severe headache, vomiting, visual disturbances, confusion, focal or generalized seizures.
- Fundoscopy examination (key role): Papilledema
- It includes:
- Hypertensive Emergency
- Hypertensive urgency.
Hypertensive Emergencies:
Reason includes renovascular disease, pheochromocytoma, nonadherence to antihypertensive medication, hyperaldosteronism, erythropoietin administration, acute glomerular nephropathy, and eclampsia.
Hypertensive Urgency:
- Severe elevation in BP >180/120 mm Hg without symptoms or signs of acute target organ involvement.
- Adequate treatment of these conditions, a BP lowering within 24 hours by administration of oral drugs.
- ICU admission is usually not required.
Hypertensive Encephalopathy Treatment:
Requires an immediate BP reduction in few minutes to hours in an ICU care and brought by IV drugs.
”Malignant” or “Accelerated” Phase Hypertension:
Question 64. Write short essay or a note on accelerated hypertension.
Answer:
- It is a rare condition that may complicate hypertension of any etiology. It occurs when blood pressure rises rapidly with severe hypertension (diastolic blood pressure >120 mm Hg). Histologically, it is characterized by fibrinoid necrosis in the walls of small arteries and arterioles and intravascular thrombosis.
- Diagnosis: Presence of high BP and rapidly progressive end-organ damage, such as retinopathy (grade 3 or 4 with flameshaped hemorrhages, cotton wool spots, hard exudates, and papilledema), renal dysfunction (e.g., proteinuria and hematuria), and/or cerebral edema and hemorrhage with resultant hypertensive encephalopathy. Left ventricular failure may develop.
- If this is untreated, it may lead to death within months from progressive renal failure, heart failure, aortic dissection, or stroke.
Question 65. Write short essay or note on treatment of hypertensive crisis (hypertensive emergencies).
(or)
Discuss the management of accelerated/malignant hypertension.
Answer:
Management of Hypertensive Emergencies:
- Normalization of BP is usually not recommended.
- Sudden fall in BP may cause acute hypoperfusion of vital organs and results in myocardial ischemia or infarction, hemiplegia, or acute renal failure.
- Older patients with long-lasting hypertension and preclinical organ involvement (LVH, atherosclerosis, and arteriolar remodeling) are at risk of these complications as the lower limit of autoregulation shifted to right.
How fast and how much BP to be lowered to be given importance:
- Goal: Reduce mean arterial pressure (MAP) by no more than 20–25%, diastolic blood pressure (DBP) to 100–110 mm Hg within few minutes to 2 hours.
- More aggressive and rapid BP reduction needed in acute pulmonary edema and aortic dissection.
- More slow reduction needed for acute cerebrovascular damages with monitoring of neurological status.
- Constant infusion of intravenous agents required (no intermittent IV boluses/oral/sublingual drugs—drastic BP fall).
Various hypertensive emergencies and their treatment.
Drugs used in hypertensive emergencies and their dosage, action, and adverse effects.
Resistant Hypertension:
Question 66. Write short note on resistant hypertension.
Answer:
Definition: Resistant hypertension is defined as blood pressure above goal (>140/90 mm Hg; >130–139/80–85 mm Hg in patients with diabetes mellitus; >130/80 mm Hg in chronic kidney disease), despite treatment with ≥3 antihypertensive drugs of different classes, including a diuretic, at optimal doses.
- Resistant hypertension is observed in about 10–20% of patients with hypertension.
- Almost 50% of these patients experience an adverse cardiovascular event compared with patients with blood pressure controlled by three or fewer antihypertensive agents. Noncompliance to medication is an important cause.
- Diagnosis requires exclusion of both pseudoresistance and reversible or organic causes.
Treatments for Resistant Hypertension:
- Nonpharmacologic intervention: Reinforce lifestyle changes.
- Drug intervention: Look for drug compliance, optimize doses. Add drugs from other classes.
- Device therapy: Two techniques:
- Percutaneous transluminal radiofrequency sympathetic denervation of the renal arteries.
- Carotid baroreflex activation.
- Medical therapy: Aldosterone antagonist, angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) plus chlorthalidone.
- Percutaneous angioplasty with/without stent placement.
- Surgical revascularization or, nephrectomy in unilateral cases.
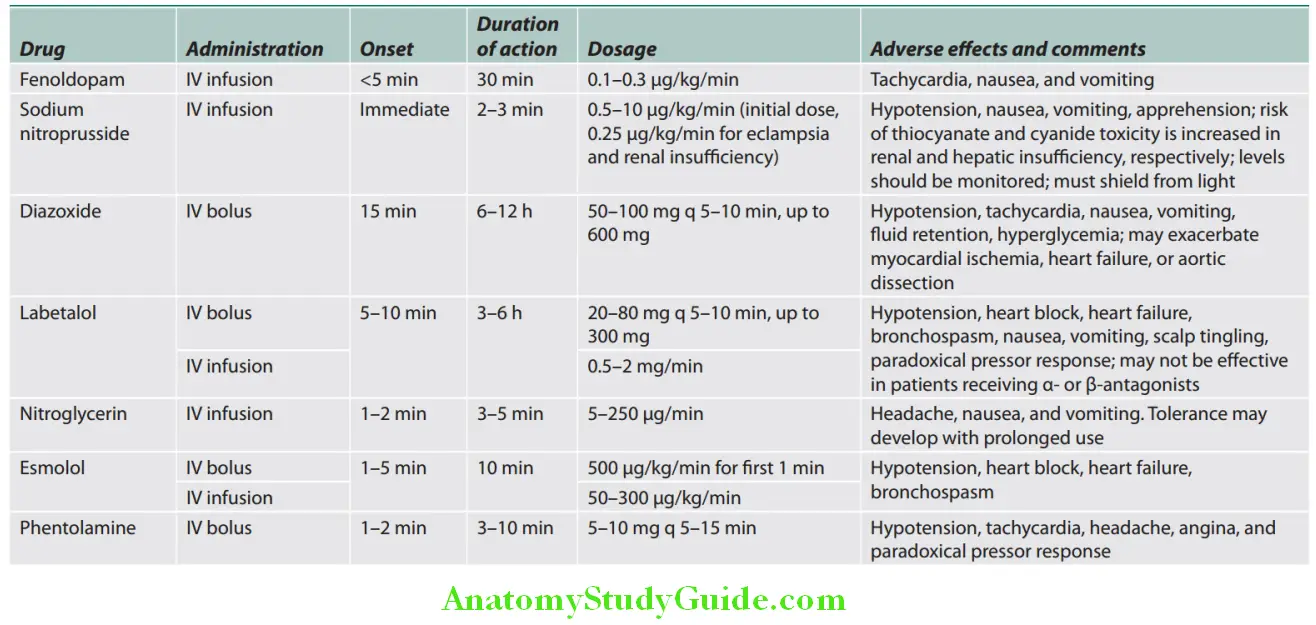
Acute Rheumatic Fever:
Question 67. Define rheumatic fever. Discuss the etiology, risk factors, pathogenesis, clinical features, investigations, diagnosis, complications, and management of rheumatic fever.
Answer:
Acute Rheumatic Fever Definition: Rheumatic fever (RF) is an acute, poststreptococcal, immune-mediated, and multisystem inflammatory disease.
It occurs as a sequel to group A streptococcal pharyngitis.
- Multisystem disease affecting connective tissue particularly of the heart, joints, brain, cutaneous, and subcutaneous tissues.
Acute Rheumatic Fever Phases:
Thre are two major phases:
- Acute rheumatic fever (ARF): It frequently manifests as acute rheumatic carditis.
- Chronic rheumatic heart disease (RHD) is the permanent heart valve damage resulting from one or more attacks of ARF. About 40–60% of patients with ARF will develop RHD. The most common valve affected is the mitral followed by aortic, in that order. However, all four valves can be affected. The deforming fibrotic valvular lesions are the principal/key features of chronic RHD.
Epidemiology and Incidence:
- Age group: Most common in children between 5 and 15 years. It is rare <3 years of age.
- Sex: Both sexes are equally affected. However, certain clinical manifestations, such as mitral stenosis and Sydenham chorea, have a female preponderance after puberty.
- Socioeconomic conditions: Rheumatic fever is a worldwide disease and it is prevalent in regions with poor economic conditions, overcrowding, and substandard housing. Incidence and mortality rate of RF and RHD have markedly decreased over the past century, due to improved socioeconomic conditions and rapid diagnosis and treatment of streptococcal pharyngitis. In India, the annual incidence is 0.18 to 0.3 per 1,000 school children.
- Poor economy and overcrowding: It is a predisposing factor in developing countries. It is a major cause of death and disability in children and adolescents in socioeconomically deprived regions.
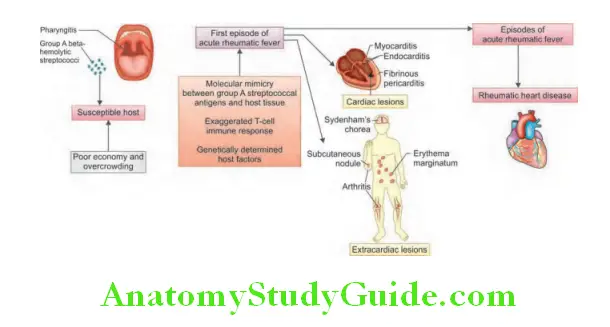
Acute Rheumatic Fever Etiology:
- Acute rheumatic fever is a poststreptococcal disease.
- It develops after a latent period of 2 to 6 weeks after an episode of pharyngitis (sore throat) or tonsillitis by group A β-hemolytic streptococcIt occurs most often in children.
- Rheumatogenic potential of various serotypes of group A streptococci varies. M protein is one of the well-defined determinants of bacterial virulence. M-type 5 is commonly responsible for rheumatic fever and other rheumatogenic serotypes include 1, 3, 6, 14, 18, 19, and 24. M-type 12 is highly prevalent, but usually does cause rheumatic fever.
Recently virus (coxsackie B-4) has been suggested as causative agent with streptococcus acting as conditioning agent.
Pathogenesis:
Immunologically mediated disease: Exact pathogenesis of rheumatic fever is not known. Streptococcal infection introduces the streptococcal antigens into the body may activate both antibody and T cell-mediated reactions against streptococci.
Molecular Mimicry:
- Antibodies may be produced by B-lymphocytes against various antigenic components of the Streptococcus.
- These antibodies cross-react with human tissues because of the antigenic similarity between streptococcal components and human connective tissues (molecular mimicry). One of them produced against the M proteins of streptococci seems to cross-react with certain similar self-antigens in the myocardial cells and glycoproteins of the valves in the heart. This may be the mechanism for pancarditis in acute rheumatic fever.
- Immunologically mediated inflammation and damage (autoimmune) to human tissues which have antigenic similarity with streptococcal components—such as heart, joint, and brain connective tissues.
Streptococcal Super Antigens:
- Super antigens are glycoproteins synthesized by bacteria and viruses. They can bridge class II major histocompatibility complex molecules to specific T-cell receptors, simulating antigen binding and activation of CD4+ T-cells. These T-cells along with antibodies cross-react with self-proteins in the heart. These reactions produce cytokines leading to activation of macrophages, which are seen in lesions of rheumatic fever.
Host Factors:
- Rheumatic fever occurs in a susceptible host and only 0.3–3% of individuals with acute streptococcal pharyngitis develops rheumatic fever.
- In India, HLA-DR3 is associated more frequently in patients with rheumatic fever and rheumatic fever has low frequency of HLA-DR2.
Clinical Manifestations:
Acute rheumatic fever is a multisystem disorder. It usually presents with fever, anorexia, lethargy, and joint pain. Previous history of sore throat: Only two-thirds of patients remember having any upper respiratory symptoms (episode of streptococcal pharyngitis) in the past 2–3 weeks.
Migrating Polyarthritis:
- Migrating polyarthritis is the most common major manifestation. It occurs early when streptococcal antibody titers are high in about 75–90% of patients.
- It is acute, painful, migratory, and asymmetric and is of short duration (fleeting and flitting). Usually >5 joints are affected and mainly involves large joints (knees, ankles, wrists, elbows, and shoulders). The joints are involved in quick succession. Small joints and cervica spine are less commonly involved.
- Involved joints show signs of inflammation (red, swollen, and tender) with or without effusion. Pain and swelling develop quickly and subside within 5–7 days. Arthritis does not progress to chronic disease and over a period of time involved joints heal without any residual deformity (licks the joints).
- Excellent response of high-dose aspirin (salicylates) and NSAIDs.
- Jaccoud’s arthritis is a rare deformity of the metacarpophalangeal joints following repeated attacks of rheumatic fever.
- In children below 5 years, arthritis is usually mild but carditis is more prominent.
Poststreptococcal Reactive Arthritis (PSRA):
- Arthritis developing after an episode of Group A streptococcal pharyngitis without other major criteria of acute rheumatic fever is known as PSRA.
- It develops 10 days after pharyngitis, can involve large joints, small joints or axial skeleton, cumulative and persistent and does not respond to acetylsalicylic acid.
- In contrast, arthritis of rheumatic fever develops 2 to 3 weeks after an episode of Group A streptococcal pharyngitis, migratory and transient, and usually involves only the large joints and responds well to acetylsalicylic acid.
- A small percentage of PSRA patients may subsequently develop valvular heart disease. Hence, patients should be followed up carefully for several months to look for evidence of carditis.
Carditis:
Question 68. Write short essay/note on diagnosis of rheumatic fever.
Answer:
- Early and most serious manifestation that occurs in 60–70% of patients.
- It manifests as pancarditis involving all three layers of the heart (endocardium, myocardium, and pericardium). On microscopic examination, myocardium shows Aschoff body that is pathognomonic of rheumatic myocarditis. Rheumatic endocarditis may involve valvular (valvular endocarditis) or mural endocardium (mural endocarditis). Rheumatic pericarditis produces pericardial effusion and thick fibrinoserous exudates.
- Carditis leaves a sequelae and permanent damage to the organ (bites the heart).
- Valvular damage is the hallmark of RF. Chronic phase is characterized by fibrosis, calcification, and stenosis of heart valves (fish-mouth valves).
- It is more common in younger children, and may be asymptomatic. It is detected only on echocardiograph.
Manifestations of Carditis:
Pancarditis involves the endocardium, myocardium, and pericardium. Incidence of carditis decreases with increasing age.
It ranges from 90% at 3 years to around 30% in adolescence.
- Myocarditis:
- Tachycardia: Disproportionate to fever and persists during sleep
- Features of congestive heart failure: Breathlessness (due to heart failure or pericardial effusion). Cardiac failure may be either caused by dysfunction of myocardium or valvular regurgitation.
- Physical examination may reveal third heart sound (S3), fourth heart sound (S4), or a summation gallop.
- Arrhythmias, prolongation of PR interval being the most common
- Endocarditis: Murmurs are most commonly observed during acute rheumatic fever. It may be new or changed murmurs and include:
- Apical pansystolic murmur is a high-pitched, blowing-quality murmur of mitral regurgitation that radiates to the left axilla.
- Apical soft mid-diastolic murmur (also known as a Carey Coombs murmur) is heard during active carditis due to valvulitis with nodules forming on the mitral valve leaflts. It accompanies severe mitral insuffiency.
- An early diastolic murmur of aortic regurgitation and is high-pitched, blowing, decrescendo, and heard best along the right upper and mid-left sternal border after deep expiration while the patient is leaning forward.
- Pericarditis:
- Chest/pericardial pain (due to pericarditis or pancarditis)
- Pericardial friction rub and precordial tenderness
- Pericardial effsion (uncommon and always small). Rheumatic pericarditis never causes constriction.
Other clinical features of acute rheumatic carditis: These include palpitations, cardiac enlargement (cardiomegaly), and syncope due to conduction defects.
Skin Lesions:
Question 69. Write a short essay/note on skin lesions in acute rheumatic fever.
Answer:
Subcutaneous Nodules:
- Occur in 9–20% of cases and often associated with carditis.
- Appears as a small (0.5–2.0 cm), painless, mobile hard nodules beneath skin appears 4 weeks after onset of RF. Thus, it helps to confirm rather than make the diagnosis.
- Most common along extensor surfaces of joint-knees, elbows, wrists, and also on bony prominences, tendons, dorsum of feet, occipital or cervical spine.
- Delayed manifestation, disappears: Leaves no residual damage.
Erythema Marginatum:
- Occur in <7% and often associated with chronic carditis and are evanescent.
- Unique, transient, serpiginous-looking lesions of 1–2 inches in size.
- Pink/red macules clear centrally, serpiginous spreading edge. More on trunks and limbs, nonitchy and almost never on the face. The resulting red rings or “margins” may coalesce or overlap.
- Worsens with application of heat.
Chorea (Sydenham’s Chorea, Chorea Minor, Saint Vitus Dance) Others:
- Epistaxis, arthralgia, tender lymph nodes, scarlet fever rash, abdominal pain, tonsillar exudates in older children, etc.
- Systemic manifestations are rare and include pleurisy, pleural effusion, and pneumonia.
Duration of Attack of Rheumatic Fever:
- Acute rheumatic fever: Average duration of an untreated acute attack is about 3 months.
- Chronic rheumatic fever: It is defined as persistence of disease for >6 months. It occurs in <5% of patients and may lead to persisting congestive heart failure.
Laboratory Investigations:
Investigations for Evidence of Preceding Streptococcal Infection (Specifi):
- Isolation of group A streptococci/Throat swab culture: Group A β-hemolytic streptococci is usually in only 10–25% of cases. It can be done also in family members and contacts. However, serologic tests are usually done to show the evidence of streptococcal infection.
- Streptococcal antibody tests (serologic tests):
- Serological tests usually confirm a recent group A β-hemolytic streptococcal infection.
- Raised streptococcal antibody levels are found in the early stages of acute rheumatic fever. However, in two situations their levels may be low:
- When the interval between the streptococcal pharyngitis and detection of rheumatic fever in >2 months (e.g., chorea)
- In patients with rheumatic carditis only
- Common serologic tests:
- Antistreptolysin O antibodies (ASO titers): Rising titers, or levels of >200 U (adults) or >300 U (children). This test is positive in 80% of cases. ASO titers are normal in 20% of adult cases of rheumatic fever and most cases of chorea.
- Anti-DNase B
- Antihyaluronidase (AH)
- Antistreptozyme test (ASTZ) is a very sensitive indicator of recent streptococcal infection and is also helpful in
ruling out rheumatic fever. Titers >200 units/mL are considered positive.
The above four tests when combined together help in conforming the diagnosis in 95% of cases.
Investigations for Evidence of a Systemic Illness (Nonspecifi):
- Acute phase reactants: These tests confirm the presence of an inflammatory process, but are nonspecific.
- Erythrocyte sedimentation rate (ESR) is raised.
- Raised C-reactive protein (CRP) in the blood
- Other tests confirming an inflammatory reaction:
- Peripheral blood: Polymorphonuclear leukocytosis and anemia (due to suppression of erythropoiesis)
- Serum: Increase in serum complements, and increase in serum mucoproteins, α2, and γ globulin levels
Investigations for Evidence of Carditis:
- Chest radiography: Chest X-ray may show evidences of cardiac failure, cardiomegaly, and pulmonary congestion.
- Electrocardiogram: ECG changes commonly include:
- Most consistent change is a prolongation of the PR interval and T-wave inversion.
- Other findings are rarely second-degree AV block features of pericarditis and reduction in QRS voltages.
- Echocardiography: It can detect myocardial dysfunction, cardiac dilatation, valvular abnormalities, and pericardial effusion.
Diagnosis of Acute Rheumatic Fever:
Question 70. Write a short essay/note on diagnosis of rheumatic fever/revised Duckett Jones criteria for the diagnosis of rheumatic fever.
Answer:
- Diagnosis of acute rheumatic fever (ARF) is made by the presence of combination of typical clinical features together with evidence of the precipitating group A streptococci (GAS) infection. This uncertainty led Dr T Duckett Jones in 1944 to develop a set of criteria known as Jones Criteria to aid diagnosis.
- Modified Jones criteria: Presently diagnosis is based on modified Jones criteria.
Exceptions to Jones Criteria:
- Chorea alone, if other causes have been excluded.
- Insidious or late-onset carditis (indolent carditis) with no other explanation.
- Patients with documented RHD or prior rheumatic fever, one major criterion, or of fever, arthralgia or high CRP suggests recurrence.
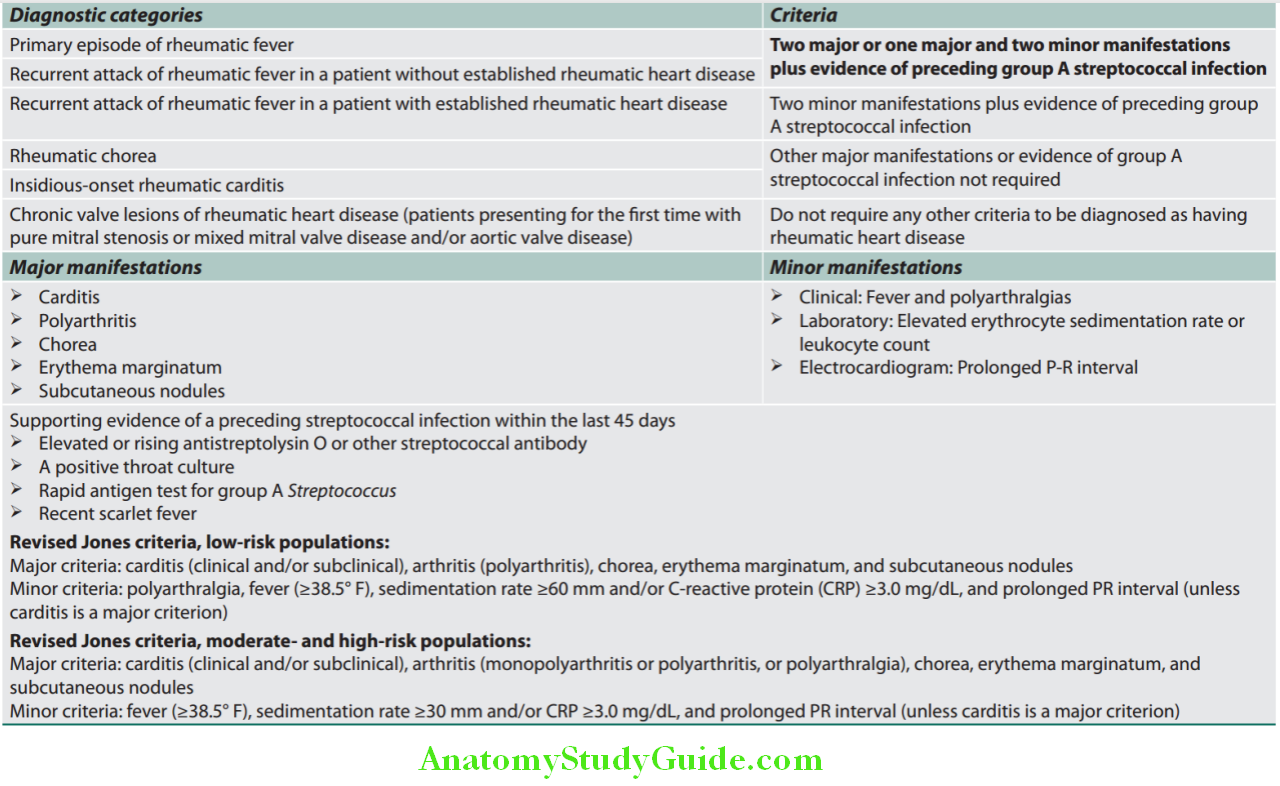
Question 71. Write a short essay/note on treatment of rheumatic fever with carditis.
Answer:
Management of Acute Rheumatic Fever:
- Step 1: Primary prevention (eradication of streptococci)
- Step 2: Anti-inflammatory treatment (aspirin and steroids)
- Step 3: Supportive management and management of complications
- Step 4: Secondary prevention (prevention of recurrent attacks)
Question 72. Write a short essay/note on prophylaxis of rheumatic fever.
Answer:
Step 1: Primary Prevention (Eradication of Streptococci):
- Primary prevention is accurate diagnosis and treatment of group A β-hemolytic streptococcal pharyngeal infection.
- Antistreptococcal therapy/primary prevention are mentioned in Table
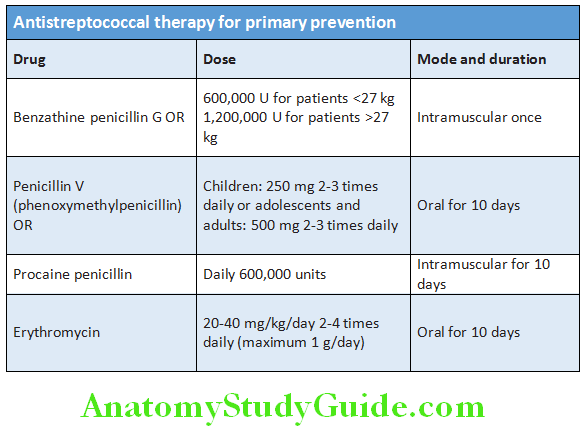
Step 2: Anti-inflammatory Treatment:
- Arthritis only:
- Aspirin usually rapidly relieves the symptoms of arthritis within 24 hours and also helps to confirm the diagnosis. Aspirin is given in the dose of 75–100 mg/kg body weight/day divided into four doses for 6 weeks (attain a body level 20–30 mg/dL). It should be continued till the ESR has fallen. It produces mild toxicity such as nausea, tinnitus, and deafness and serious toxicity as vomiting, tachypnea, and acidosis.
- Carditis or severe arthritis:
- Corticosteroids produce more rapid symptomatic relief compared to aspirin. They are indicated in patients with carditis or severe arthritis. However, their long-term use is not found to be beneficial.
Step 3: Supportive Management and Management of Complications
Bed rest is important, because it reduces joint pain and cardiac workload.
- Patients without carditis: Advice bed rest until temperature and ESR are normal.
- Patients with carditis: Bed rest to be continued for 2–6 weeks after the ESR and temperature has returned to normal. Avoid strenuous exercise in patients who had carditis.
Treatment of congestive cardiac failure: Digitalis and diuretics
- Treatment of chorea: Diazepam or haloperidol
- Rest to joints and supportive splinting
Step 4: Secondary Prevention of Rheumatic Fever (Prevention of Recurrent Attacks)
- Patients with acute rheumatic fever are susceptible to further attacks of rheumatic fever if another streptococcal infection occurs.
- Secondary prevention is directed at preventing acute group A β-hemolytic streptococcal (GABHS) pharyngitis in patients at substantial risk of recurrent acute rheumatic fever by long-term prophylaxis. Duration of prophylaxis is controversial and its broad outlines are provided
- Regimens for secondary prevention of rheumatic fever are mentioned in Table
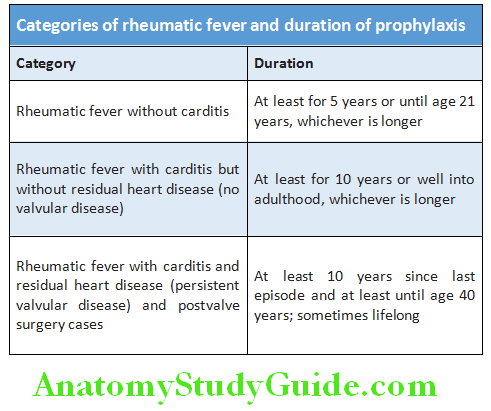

Rheumatic Chorea:
Question 73. Write a short essay/note on clinical features, diagnosis, and treatment of Sydenham’s chorea.
Answer:
It is also known as Sydenham’s chorea, Saint Vitus dance, St.Johannis chorea, chorea minor, or rheumatic chorea.
Rheumatic Chorea Definition: Rheumatic chorea is a syndrome characterized by chorea, muscle weakness, and emotional instability.
Rheumatic Chorea Etiology:
Triggering factor is pharyngeal infection by group A β-hemolytic streptococci.
Rheumatic Chorea Clinical Features:
Age and gender: Occur in 5–10% of cases, mainly in girls of 1–15 years of age.
Onset of chorea: Rheumatic chorea is a late neurological manifestation of acute rheumatic fever. Usually occurs 3–8 months after the triggering infection by A β-hemolytic streptococci when all the other signs may have disappeared. If there is no previous rheumatic manifestation, the term pure chorea is used.
First sign: Emotional lability, difficulty walking, talking, writing which is observed in 30% of patients with acute rheumatic
fever (ARF).
Characterized by spasmodic, brief, purposeless (unintentional), involuntary, jerky movements. Choreiform movements particularly affect the head/face (darting movement of tongue) upper limb hands or feet. Speech may be affected and may be explosive and halting and fidgety. It can be unilateral (hemichorea) or bilateral.
Mild forms may be difficult to diagnose and following signs are helpful in these cases:
- Milkmaid’s grip: When the patient is asked to squeeze the examiner’s fingers, a squeezing and relaxing motion (like milking a cow) occurs. This is described as milkmaid’s grip and is due to inability to maintain muscular contraction.
- Bag-of-worms appearance is due to asynchronous contractions of the lingual muscles.
- Jack-in-the-box sign: When the patient is asked to keep the tongue protruded out, it retracts involuntarily.
- Pronator sign: Holding the arms outstretched may elicit “spooning” (hyperextension of the fingers with dorsiflexion of the wrist).
Severe forms: Patients is unable to get up or sit, and has violent continuous jerks that may cause physical injury. Additional features include hypotonia, pendular knee jerks, and mild generalized muscular weakness.
Rheumatic Chorea Prognosis:
- Usually benign and spontaneously resolves in 2–3 months. Disappears leaving no residual damage.
- About 25% of affected patients develop chronic rheumatic valve disease.
Rheumatic Chorea Management:
- Rule out other causes of chorea such as systemic lupus erythematosus, Huntington’s disease, and Wilson’s disease.
- Rest: Complete mental and physical rest. Keep the patient in a quiet room.
- Padded side-boards for beds to prevent physical injury.
- Drugs: Haloperidol or sodium valproate with diazepam. Steroids may be needed in severe cases.
- Rheumatic fever prophylaxis
Valvular Heart Disease:
Mitral Valve:
It is called so because it resembles the Bishops “miter.”
Mitral Valve Apparatus:
It consists of different components. Normal mitral valve is bicuspid (two leaflets), funnel-shaped valve between left atrium and left ventricle with its apex in the left ventricle. The two valve leaflets are attached by (about 120) chordae tendineae to two papillary muscles. Mitral valve orifice is about 4–6 cm2 (average 5 cm2).
Different components of mitral valve apparatus:
- Mitral annulus
- Mitral leaflets: anterior mitral leaflet (AML), and posterior mitral leaflet (PML)
- Subvalvular apparatus: Chordae tendineae and papillary muscles
- Left ventricular wall where the papillary muscle attaches
- Left atrium
Mitral Stenosis:
Question 74. Discuss the etiology, pathophysiology, clinical features, investigations, complications, and management of mitral stenosis.
(or)
Write a short essay/note on juvenile mitral stenosis and pediatric mitral stenosis.
Answer:
Mitral stenosis (MS) is a valvular heart disease is characterized by the narrowing of the orifice of the mitral valve due to structural abnormality of the mitral valve apparatus.
Mitral Stenosis Etiology and Pathology:
- Rheumatic fever is the most common cause of mitral stenosis. It develops secondary to previous rheumatic fever due to infection with group A β-hemolytic Streptococcus.
- Mitral stenosis is more common in females.
- The latent period from the first attack of rheumatic fever and the development of onset of symptoms due to mitral stenosi is usually as short as 1–2 years in India, compared to long in Western countries. This may be due to repeated attacks of severe carditis in India.
- Clinical manifestation in juvenile mitral stenosis/malignant mitral stenosis develops below the age of 19 years and is common in India. Pediatric mitral stenosis manifests below the age of 12 years. Pin-point mitral valve is seen. Atrial firillation is rare. Valve calcifiation is uncommon.
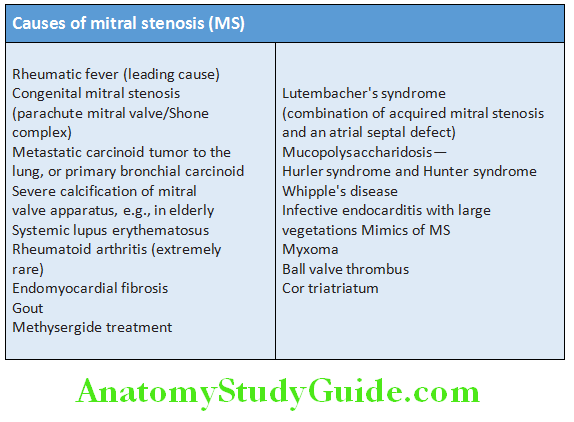
Rheumatic Mitral Stenosis:
In rheumatic MS, chronic inflammation produces:
- Diffuse thickening of the mitral valve leaflets due to fibrosis and/or calcification
- Fusion of commissures and cusp
- Fusion and shortening of the chordae tendineae.
The above morphological changes progress and cause rigidity of mitral valvular cusps which in turn leads to narrowing at the apex of the funnel-shaped (“fish-mouth”) mitral valve → severe narrowing (stenotic) of valve orifice and progressive immobility of the valve cusps.
Salient features of mitral stenosis are presented:
Salient features of mitral stenosis (MS):
- First chamber to fail in MS: Left atrium
- Ventricle to fail in MS: Right ventricle
- Atria that fibrillates in MS: Affects both right and left atria
- Left ventricle in MS: Left ventricular end-diastolic volume
(LVEDV) is reduced in 15%, while it is normal in the rest - The most common complication of MS: Atrial fibrillation (AF)
Pathophysiology and Hemodynamics:
Long latent period: Mitral stenosis clinically manifests after a latent period about 20 years from the first episode of acute rheumatic fever. Its symptoms usually start when the mitral valve surface area reduces to 2.5 cm2.
- Normally, the mitral valve opens during (left ventricular) diastole and allows the flow of blood from left atrium to the left ventricle. During ventricular diastole, the pressures in the left atrium and the left ventricle are equal.
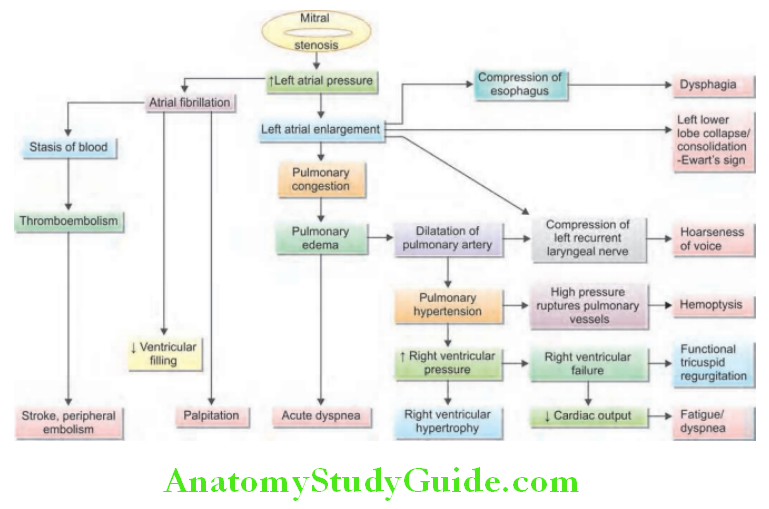
- Mitral stenosis obstructs the blood flow from left atrium to left ventricle and raises the pressure in the left atrium (up to 25 mm Hg in severe stenosis). Initially, this rise in left atrial pressure may occur only during exercise, but later it is raised even during rest.
- Raised left atrial pressure is reflected back in the pulmonary veins that produce pulmonary venous hypertension and subsequently pulmonary arteries which produce pulmonary arterial hypertension. The symptoms consist of episodes of paroxysmal nocturnal dyspnea (PND) evident as pulmonary edema and hemoptysis, i.e., Winter bronchitis.
- Repeated episodes of PND produce arteriolarization of pulmonary capillaries and veins → pulmonary arterial hypertension.
- During this period, there may be resolution of symptoms and signs of pulmonary venous hypertension. Patient develops progressive exertional dyspnea. Patient feels better in terms of symptoms, although the disease has progressed.
- Pulmonary arterial hypertension (pulmonary hypertension) progressively increases in severity over the next 10–15 years till finally the right ventricle fails (early fourth decade). Types of pulmonary hypertension in MS are presented.
- Consequences of chronic pulmonary hypertension:
- Hypertrophy of right ventricle: Which may later undergo dilatation → right ventricular failure
- Functional tricuspid regurgitation due to dilatation tricuspid valve ring secondary to dilatation of right ventricle
- Pulmonary valve incompetence (regurgitation) may develop due to dilatation of pulmonary valve ring
- Consequences of raised left atrial pressure: Causes left atrial dilatation and this enlarged left atrium is prone to:
- Atrial fibrillation
- Stasis of blood with thrombus formation
- Detachment of the thrombus resulting in systemic embolism
- Reduced ejection fraction of left ventricle is found one-third of patients due to:
- Decreased preload due to impaired filing
- Increased afterload secondary to reflex vasoconstriction (secondary to decreased cardiac output)
- Severe mitral stenosis: There is decompensation by tachycardia or high flow. Tachycardia reduces diastolic LV filling time thereby increasing pressure in LA. Hyperkinetic circulatory states increase flow rate across mitral valve leading to increased transmitral gradient leading to increased LA pressure.
Types of pulmonary hypertension in mitral stenosis (MS):
- Passive pulmonary hypertension: Due to passive backward transmission of elevated left atrial pressure by venous and capillary
- Reactive pulmonary hypertension: Due to reflex spasm of pulmonary arterioles in response to elevate pulmonary venous and left atrial pressure
- Obliterative pulmonary hypertension: Due to chronic hypertension producing fibrosis of pulmonary bed
Clinical Features:
Question 75. Write a short essay/note on clinical features of mitral stenosis.
Answer:
Mitral stenosis Symptoms:
- Dyspnea on exertion is due to pulmonary hypertension and is slowly progressive.
- Precipitated by severe exertion, excitement, fever, anemia, sexual intercourse, pregnancy, thyrotoxicosis, and atrial fibrillation.
- Fatigue on exertion: Due to pulmonary hypertension and low cardiac output. It is slowly progressive.
- Hemoptysis:
- Rupture of bronchial veins or of pulmonary vein or bronchial vein collaterals—pulmonary apoplexy
- Rupture of pulmonary capillaries during pulmonary edema
- Pulmonary congestion, embolism, and infarction
- Winter bronchitis
- Pulmonary hemosiderosis due to chronic recurrent pulmonary edema
- Anticoagulant use
- Chest pain: Develops in about 15% patients due to
- Right ventricle enlargement, right ventricular hypertrophy, and pulmonary hypertension
- Coexistent coronary artery disease (CAD)
- Coronary Embolism
- Angina due to decreased cardiac output.
- Palpitations develop when there are atrial fibrillations (AF), chamber enlargement (right ventricle and pulmonary artery).
- Systemic thromboembolism: Paroxysmal AF results in embolism most commonly to the cerebral vessels resulting in stroke, but mesenteric, renal, and peripheral emboli (e.g., ischemic limb) can also develop.
- Syncope: PAH, arrhythmias, and ball valve thrombus
- Hoarseness of voice—La Ortner’s syndrome: Due to compression of the left recurrent laryngeal nerve due to the dilated pulmonary artery or giant left atrium
- Right heart failure
- Orthopnea and paroxysmal nocturnal dyspnea: Due to left atrial failure
- Winter bronchitis: Patients with MS are susceptible to recurrent attacks of bronchitis, especially during the winter.
Physical Signs:
General examination:
- Mitral facies is characterized by cyanotic lips and face, malar flush (dusky pink discoloration over the upper cheeks due to arteriovenous anastomoses and vascular stasis), and mild jaundice. It may develop when MS is very severe with low cardiac output and peripheral vasoconstriction.
- Peripheral edema and ascites when right heart failure develops.
- Pulse:
- Pulse in low volume and peripheral pulse may be absent if embolism develops.
- Pulse rhythm is irregularly irregular and varying volume in atrial fibrillation.
- Blood pressure: May be mildly reduced. Mean of three readings to be taken if atrial fibrillation is present.
- Jugular veins:
- Jugular venous pressure is raised when congestive heart failure develops.
- Jugular venous pulse:
- Prominent a waves (due to vigorous right atrial systole) observed when there is pulmonary hypertension without atrial fibrillation,
- Absence of waves in atrial fibrillation
- Prominent V waves (C-V waves) and rapid y descent when there is development of functional tricuspid regurgitation.
Inspection and Palpation of Precordium:
- Apex beat is not shifted and is tapping character of S1 at apex (closing snap). Apex beat is shifted when there is coexistent of MS with mitral regurgitation (MR)/aortic stenosis (AS)/systemic hypertension/ischemic heart disease (IHD)/myocarditis.
- Diastolic thrill at apex
- Palpable pulmonary component of second heart sound (P2) if there is pulmonary arterial hypertension.
- Left parasternal heave: Present when there is right ventricular hypertrophy or left atrial enlargement.
- Epigastric pulsations of right ventricular type.
- Precordial bulge in juvenile/malignant MS.
Other fidings: Right heart failure is associated with peripheral edema, tender hepatomegaly, and ascites.
Auscultation:
Question 76. Write a short essay/note on auscultatory findings in mitral stenosis.
Answer:
- Loud first heart sound:
- In mitral stenosis, the forces that open and close the mitral valve increase as left atrial pressure increases. Hence, the first heart sound (S1) is loud and can be palpable (tapping apex beat) in mitral stenosis.
- When associated with atrial fibrillation, the intensity of first heart sound varies.
- A low intensity of the first heart sound in MS may be due to
- Calcification of the mitral valve
- Congenital MS
- Dominant associated mitral/aortic regurgitation.
- Loud second heart sound: It is a sign of pulmonary hypertension. Th second heart sound is closely split and the pulmonary component of the second heart sound (P2) is loud.
- Mitral opening snap:
- OS is a sharp, snappy sound heard during early diastole, following the sound of aortic valve closure (A2) by 0.05–0.12 s.
- Opening snap (OS) is produced due to the sudden (abrupt) opening of the dome of the stenosed mitral valve with the force of the increased left atrial pressure during diastole. It is the most important auscultatory sign of valvular involvement in MS. Absent OS indicates the calcification of body of the leaflets.
- The time interval between A2 and OS is inversely proportional to the severity of the MS.
- Best heard: During expiration, just medial to the cardiac apex with the diaphragm of the stethoscope.
- Other conditions with OS: Mitral regurgitation (10%), tricuspid stenosis, and atrial septal defect
- Well’s index: Q-S1 interval minus A2-OS interval and is expressed in units of 0.01 seconds. More than 2 units indicate MVA <1.2 cm2.
- Murmur of mitral stenosis:
- Mid-diastolic/presystolic murmur: Turbulent blood flow through stenosed mitral valve produces the characteristic low pitched, rumbling, mid-diastolic murmur, and sometimes accompanied by a thrill. Murmur is best heard with the bell of the stethoscope held lightly at the apex with the patient lying on the left side.
- Duration of the murmur varies and usually depends on the severity of stenosis. In severe MS, the mid-diastolic murmur is long and merges with the presystolic murmur to produce a holodiastolic murmur.
- To increase intensity of MDM: Left lateral position using bell of stethoscope, while holding expiration auscultate after walking (isotonic exercise), and squatting (increased peripheral resistance in these procedures contribute to the increased murmur).
- In the early phase of mitral stenosis, a presystolic murmur may be the only auscultatory abnormality.
- Mechanism of presystolic murmur:
- Atrial Contraction
- Persistent Atrioventricular Gradient
- Left ventricular contraction in presystole reducing mitral funnel.
- Presystolic accentuation of the murmur: Atrial contraction contributes to increased gradient in presystole.
- Hence, mid-diastolic murmur is accentuated by exercise. In patient with sinus rhythm, the murmur becomes louder during atrial systole and during long R-R interval in atrial fibrillation is termed as presystolic accentuation.
- Absence of presystolic murmur in MS: Atrial fibrillation, mild MS, prolonged PR interval, bradycardia, and elevated left ventricular dysfunction (LVEDP)
- Causes of absent mid-diastolic murmur: Thick chest wall and emphysema, dampened MS (severe pulmonary hypertension throttling the left-sided input), low cardiac output, and marked RV enlargement with RV occupying the apex.
- Systolic murmur: When pulmonary hypertension develops, it will lead to right ventricular hypertrophy and dilatation with secondary tricuspid regurgitation. This produces a systolic murmur and giant “v waves” in the venous pulse.
- If MS coexists with mitral regurgitation, it produces a loud pansystolic murmur that radiates toward the axilla and is heard at the lower left sternal border. Functional tricuspid regurgitation produces a pansystolic murmur. It is accentuated during inspiration (de Carvallo’s sign).
- Murmur of pulmonary regurgitation (Graham Steell murmur): It is a high-pitched early diastolic decrescendo murmur heard along the left sternal border and indicative of severe pulmonary hypertension.
Diagrammatic representation of timing of heart sounds and murmur in mitral stenosis
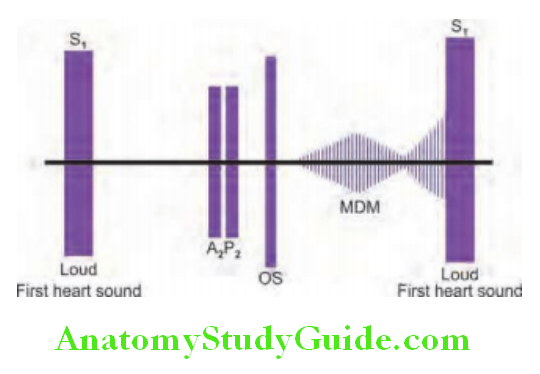
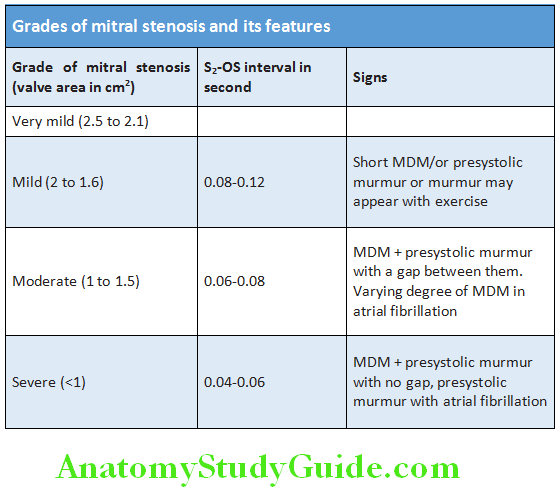
Auscultatory findings in mitral stenosis:
- Loud S1
- Loud P2 and narrow split of S2 if pulmonary arterial hypertension
- Opening snap
- Mid-diastolic murmur at apex with presystolic accentuation
- Tricuspid regurgitation: Pansystolic murmur
- Pulmonary hypertension: Ejection systolic/early diastolic (Graham Steell) murmur
Question 77. Write a short essay/note on grading the severity of mitral stenosis.
Answer:
Clinical judgment of the severity of mitral stenosis:
Following features suggest severe MS:
- Presence of pulmonary hypertension
- More closeness of the opening snap to the second heart sound (short A2 -OS interval)
- Lengthy mid-diastolic murmur
Severity of mitral stenosis Investigations:
Radiological features of mitral stenosis (Chest X-ray)
Question 78. Write a short essay/note on radiological/X-ray chest findings in mitral stenosis.
Answer:
Due to enlargement of left atrium:
- Enlarged left atrial appendage causes filling up of normal concavity between pulmonary artery shadow and the left ventricle.
- Double atrial shadow: Border of enlarged left atrium together with right atrial border gives an appearance like atrium within an atrium.
- Straightening of left heart border: Mitralization of heart Due to left atrial appendage enlargement, large pulmonary artery, hypoplastic aorta, under filled left ventricle.
Due to left atrial appendage enlargement, large pulmonary artery, hypoplastic aorta, under filled left ventricle:
Consequences of left atrial enlargement:
- Pushing of left main bronchus upward causing wide carinal angle (splaying of carina)
- Pushing esophagus backward visible in lateral view of chest X-ray
- Left shift of aorta (Bedford sign)
- Walking man sign (shift of left bronchus forwards)
Pulmonary venous/capillary hypertension:
- Grade 1: Cephalization (prominence of veins of upper lobe of lung) of pulmonary vasculature (pulmonary venous pressure ≤20 mm Hg) (inverted moustache sign/antler’s horn sign).
- Grade 2: Kerley lines (A, B, C) (pulmonary venous pressure 20–25 mm Hg), peribronchial, perivascular cuffing
- Kerley A line: Linear opacities extending from the periphery to hila; they are caused by distension of anastomotic channels between periphery and central lymphatic’s
- Kerley B line: Short horizontal lines situated perpendicularly to the pleural surface at the lung base; they represent edema of interlobar septa.
- Kerley C line: Reticular opacities at lung base, representing Kerley B line en face.
- Grade 3: Batwing opacities (pulmonary venous pressure >25 mm Hg)
Pulmonary arterial hypertension:
Prominent pulmonary outflow tract: Enlarged pulmonary arteries (diameter of right descending pulmonary artery >14 mm in women and >16 mm in men) + pruning of peripheral pulmonary vessels.
Right ventricle:
- Right ventricular hypertrophy: In presence of cardiomegaly, acute angle is observed between apex of enlarged heart and diaphragm.
- Sternal contact sign: Earliest and most sensitive sign in the lateral X-ray is obliteration of Holtzneck’s space, i.e., retrosternal space.
Others:
- Right atrial enlargement
- Calcification of mitral valve/pericardium/MacCallum patch
- Pulmonary hemosiderosis
Electrocardiogram:
It can confirm enlargement of left atrium (“P” mitrale), right ventricular hypertrophy, tall R waves in V1-V3, P mitrale, and atrial fibrillation.
Echocardiogram:
- It can reveal thickening of mitral valve area, valvular leaflet, thickening and shortening chordae tendineae, fusion of commissures, calcification of leaflets and chordae, and diastolic doming (due to commissural fusion).
- Wilkins score (4 to 16): 4 points each for leaflet thickness, leaflet mobility, leaflet calcification, and chordal involvement.
Transesophageal echocardiography (TEE):
To assess mitral regurgitation (MR) severity and to rule out left atrial appendage (LAA) thrombus.
Doppler:
Provides definite evaluation of MS. It shows pressure gradient across mitral valve, pulmonary artery pressure, and left ventricular function.
Cardiac catheterization:
It is not usually needed. It is used to assess coexisting condition such as coronary artery disease, pulmonary artery pressure, mitral stenosis and regurgitation.
Complications of Mitral Stenosis:
Question 79. Write a short essay/note on complications of mitral stenosis.
(or)
Write a short essay/note on indications for mitral valve replacement.
Answer:
- Atrial fibrillation (AF):
- The most common complication of MS prevalence and incidence varies according to age and roughly parallels the age of the patient (e.g., second decade—10% and sixth decade and beyond—80%).
- AF worsens symptoms of MS by:
- Decreasing diastolic filling time—leads to increased LA pressure,
- Loss of atrial contribution to LV filling—leads to increased LA pressure
- LA thrombus leading to systemic embolization.
- Prognosis: 5-year survival of AF without MS is 85% and with MS is 64%
- AF causes decrease in cardiac output by 20% in MS.
- AF worsens symptoms of MS by:
- Systemic embolism:
- Source of emboli: Left atrial thrombus, vegetations of infective endocarditis (rare)
- Factors predisposing to systemic embolism in MS: Atrial fibrillation, spontaneous echo contrast in left atrium, size of left atrium, age, and low cardiac output.
- Clinical features: Depends on the localization of embol
- Cerebral (strokes, abscess), coronary (leads to chest pain), and renal (leads to hypertension)
- Leriche syndrome: Claudication of thigh (due to embolism at branching of common iliac artery) + impotence
- Ball valve or free floating thrombus may produce syncope in specific body position, variability in physical findings, and requires urgent surgery.
- Infective endocarditis: May develop infrequently with isolated mitral stenosis.
- Ortner’s syndrome is a very rare complication of severe pulmonary hypertension secondary to mitral stenosis. It is characterized by paralysis of left recurrent laryngeal nerve due to compression between the enlarged tense pulmonary artery and the aorta at ligamentum arteriosum. It produces hoarseness of voice.
- Damped MS: Development of pulmonary hypertension diminishes the cardiac output (throttle effect) and results in temporary symptom-free period (period of illusion). The MDM may not be audible, only OS will be present.
Complications of Mitral Stenosis Management:
Mild mitral stenosis may not require any treatment other than treatment of attacks of bronchitis.
- Medical management: MS with minor symptoms are treated medically.
- Rheumatic fever prophylaxis to be given. However, infective endocarditis prophylaxis is not necessary.
- Indications for anticoagulation:
- Atrial fibrillation (persistent or paroxysmal),
- Embolic Events,
- Left Trial Thrombus,
- Left Atrial Diameter >55 Mm
- Spontaneous echo contrast.
- Restrict/decrease sodium intake.
- Diuretics: Early symptom such as mild dyspnea (due to pulmonary congestion) is usually treated with low doses of diuretics.
- Beta-blockers or non-dihydropyridines (DHP) calcium channel blockers (e.g., verapamil or diltiazem) to reduce heart rate (even in sinus rhythm, more useful in atrial fibrillation)
- Digoxin if atrial fibrillation with right heart failure. Atrial fibrillation also needs anticoagulation to prevent atrial thrombus and systemic embolization.
Surgical management:
Four operative measures are available.
1. Trans-septal balloon mitral valvotomy (BMV):
- Also known as percutaneous balloon valvuloplasty (PBV) is the treatment of choice.
- Procedure: Under local anesthesia, a catheter is passed through the femoral vein into the right atrium. The interatrial septum is punctured and the catheter is passed into the left atrium and across the mitral valve. A balloon is passed over the catheter across the valve, and briefly inflated to split the valve commissures.
- Indications: Pliable mitral valves with little involvement of the subvalvular apparatus and minimal mitral regurgitation.
- Contraindications: Moderate or severe mitral regurgitation, severe calcification, severe subvalvular fibrosis, thrombus in left atrium or ventricle, recent embolism, bleeding disorders, and interatrial septal thickness >3 mm (relative contraindication).
- Complications: Mitral regurgitation may be severe enough to need surgery (2%), mortality (1–2%), cardiac perforation (1%), and cerebral embolism (1%).
2. Closed mitral valvotomy (CMV):
- Indication: Mobile, noncalcified mitral valves without regurgitation
- Contraindication: Left atrial thrombus, mitral valvular calcification, severe subvalvular disease, or moderate or severe mitral regurgitation
- Advantages: Cardiopulmonary bypass is not required and good result is obtained for 10 years or more.
- Disadvantage: The valve cusps may refuse necessitating another operation.
3. Open mitral valvotomy (OMV):
- It is usually performed and is preferred over closed valvotomy or mitral valve replacement.
- Procedure: Under direct view, the valvular cusps are carefully separated from each other and commissures are incised.
- Advantage: Less chances of traumatic mitral regurgitation, concurrent annuloplasty can be done for mitral regurgitation. Removal of LA thrombus (if present), calcium in leaflets, amputation of LA appendage, and separation of fused chordae can also done along with this surgical procedure.
- Disadvantage: Needs cardiopulmonary bypass.
4. Mitral valve replacement (MVR):
Indications:
- Mitral stenosis associated with mitral regurgitation
- Severely damaged or severely calcifid stenotic valve which cannot be reopened without producing signifiant mitral regurgitation
- Moderate or severe mitral stenosis and presence of thrombus in the left atrium even after anticoagulation therapy
- Type of prosthesis: Mechanical prosthesis if age is <65 years and bioprosthesis if age is >65 years.
- Artificial valves usually work successfully for >20 years and anticoagulants are usually given postoperatively to prevent thrombus formation and its embolization.
Mitral Regurgitation:
Question 80. Discuss the etiology, pathophysiology, clinical features, investigations, complications, and management of chronic mitral regurgitation.
(or)
Write a short essay/note on causes of acute mitral regurgitation/mitral regurgitation.
Answer:
- Management of acute severe mitral regurgitation in pulmonary edema.
Mitral regurgitation (MR) or mitral insufficiency (MI) or mitral incompetence is a disorder of the heart in which the mitral valve does not close properly.
Mitral Regurgitation Etiology:
Lesion in any one of five components of mitral valve apparatus namely:
- Valve Leaflets
- The Annulus
- The Chordae Tendineae
- Papillary Muscles
- The left ventricle can produce mitral regurgitation (MR).
Mitral regurgitation may be acute or chronic.
Causes of MR in IE: Leaflet perforation, vegetations preventing leaflet function, rupture of chordate tendineae, and annular abscess
Causes of MR in RHD: Rigid and retracted leaflet and shortening of chordae tendineae.
Causes of MR in CAD: Regional wall motion abnormalities, ischemia of papillary muscle, and LV failure.
Pathophysiology:
- Acute mitral regurgitation: During this, the normal compliance of the left atrium does not permit much dilatation and the pressure of left atrium rises. This markedly increases the left atrial pressure and pulmonary venous pressure and cause pulmonary edema. Since part of the stroke volume is regurgitated, the stroke volume increases (to a lesser degree than it does in chronic MR) to maintain the forward cardiac output. This may result in the enlargement of the left ventricle.
- Chronic mitral regurgitation: Causes gradual dilatation of left atrium but little increase in left atrial pressure because the regurgitant flow is accommodated by the large left atrium. In longstanding cases, the left ventricle slowly dilates and the left ventricular diastolic and left atrial pressures gradually increase due to chronic volume overload of the left ventricle. MR begets MR.
Causes of acute mitral regurgitation:
- Infective endocarditis, rupture of a papillary muscle (e.g., acute myocardial infarction and mitral valve prolapse), chest trauma, cardiac surgery, acute rheumatic carditis, and dysfunction of prosthetic valve.
Causes of chronic mitral regurgitation:
- Damage to valve leaflets: Rheumatic heart disease, myxomatous degeneration, mitral valve prolapse (MVP), infective endocarditis, and SLE.
- Damage to annulus: Abscess (IE), annular calcification, and dilated cardiomyopathy
- Damage to chordae tendineae: Myxomatous degeneration (MVP, Marfan syndrome, Ehlers–Danlos syndrome), infective endocarditis, and acute rheumatic fever
- Damage to papillary muscles: Coronary artery disease [ischemia, myocardial infarction, rupture (MI), dilated cardiomyopathy]
- Damage to left ventricle: Ischemia and dilated cardiomyopathy
Clinical Features:
Symptoms:
- Symptoms depend on the rapidity of development of the mitral regurgitation.
- Acute mitral regurgitation: Usually presents dyspnea due to acute pulmonary edema.
- Chronic mitral regurgitation: It may be asymptomatic for many years. It may become symptomatic only after the onset of irreversible LV dysfunction.
- Palpitation is the most common symptom due to increased stroke volume or atrial fibrillation.
- Dyspnea and orthopnea: Due to pulmonary venous hypertension and left ventricular failure occur late in the course of mitral regurgitation
- Fatigue and lethargy: Due to reduced cardiac output
- Symptoms of right heart failure: Develop in the late stages and lead to congestive cardiac failure
- Cardiac cachexia
- Thromboembolism: Less common than in mitral stenosis. However, subacute infective endocarditis is more common.
- Other symptoms and complications are similar to mitral stenosis.
Signs:
Pulse: Volume is high and in severe mitral regurgitation, it may be mildly collapsing.
- Irregular rhythm and varying volume if there is atrial fibrillation.
Jugular veins:
- Uncomplicated mitral regurgitation: Jugular venous pressure (JVP) is normal.
- With atrial fibrillation: Disappearance of a waves
- With pulmonary hypertension: Prominent a waves
- With right ventricular failure and functional tricuspid regurgitation: Jugular venous pressure is raised and very prominent v waves.
Blood pressure In severe mitral regurgitation: wide pulse pressure. Three recordings are necessary if patient has atrial fibrillation (AF). Pulsus alternans in acute MR.
Inspection and palpation:
- Hyperdynamic precordium
- Apex beat: Shifted to the left (due to left ventricular dilatation), forceful (feels active and rocking), and diffuse (hyperdynamic) in character due to left ventricular volume overload
- Cardiomegaly: In chronic MR. Acute MR does not produce cardiomegaly.
- Systolic thrill (if MI is severe) at the apex
- Left parasternal heave and palpable P2
- Epigastric pulsations of right ventricular type
Auscultation:
- Soft first heart sound (S1): Because of the incomplete apposition of the mitral valve cusps and partial closure of these valve cusps when ventricular systole begins. It is loud if there is coexistent MS, MVP-MR, or papillary muscle dysfunction MR.
- Widely split second heart sound (S2) is due to aortic valve closure (A1) occurring early but the split is mobile.
- Pulmonary component (P2) of S2 is loud and palpable in pulmonary hypertension and also due to anterior displacement of pulmonary artery caused by dilated LA.
- Left ventricular third heart sound (S3): Indicates severe mitral regurgitation
- S4 is a sign of acute MR as left atrium is not dilated in acute MR.
- Pulmonary ejection sound in pulmonary hypertension

Murmur of mitral regurgitation:
- Apical pansystolic murmur:
- The typical features of murmur in mitral regurgitation are:
- High-pitched, blowing, and usually holosystolic/pansystolic loudest at the apex
- Plateau-shaped, best heard with diaphragm of stethoscope
- Commonly radiates widely over the precordium and into the axilla and left interscapular area (if anterior leaflets involved as in rheumatic) or radiating to base (if posterior leaflets involved)
- It is produced due to the mitral regurgitant jet occurring throughout the whole of systole.
- It may be accompanied by a thrill.
- Character of murmur depends on the underlying pathology in mitral regurgitation.
- Severe MR with soft or no murmur (silent MR): It is observed with left ventricular dilation, acute mitral regurgitation (MR), paraprosthetic MR, COPD, obesity, dampened MR.
- Dynamic auscultation:
- Non-MVP MR murmur is distinguished from MVP murmur by increase with squatting and decrease with standing (opposite with MVP).
- MR murmur is distinguished from AS/HCM murmur by increase with isometric handgrip (opposite for AS/HCM).
- Short mid-diastolic flow murmur: Sometimes a short, rumbling mid-diastolic murmur may be detected at the apex in severe cases due to an increased flow across the mitral valve. It may follow the third heart sound.
- Other murmurs:
- Ejection systolic murmur or early diastolic murmur at pulmonary area when there is pulmonary hypertension.
- Pansystolic murmur at lower left sternal border when there is functional tricuspid regurgitation.
- Opening snap can be heard in 10% patients with MR.

Signs indicating severity of mitral regurgitation:
Question 81. Write a short note on clinical assessment of the severity of mitral regurgitation.
Answer:
Other signs in MR: These include signs related to:
- Atrial fibrillation/flutter
- Pulmonary venous congestion, e.g., crepitations, pulmonary edema, and effusions
- Pulmonary hypertension and right heart failure
- Left and right heart failure
Signs indicating severity of mitral regurgitation (MR):
- Mild MR: Only murmur
- Moderate MR: Murmur + thrill and cardiomegaly
- Severe MR: Murmur + thrill, cardiomegaly, LV S3, flow mid-diastolic murmur, and pulmonary hypertension
Signs in acute mitral regurgitation:
- Normal apical impulse since there is no ventricular dilatation
- Third and/or fourth heart sound
- An early systolic or pansystolic murmur
Differences between acute and chronic mitral regurgitation are listed in Table:
Differences between mitral regurgitation due to mitral valve prolapsed and that due to rheumatic heart disease are listed in Table:
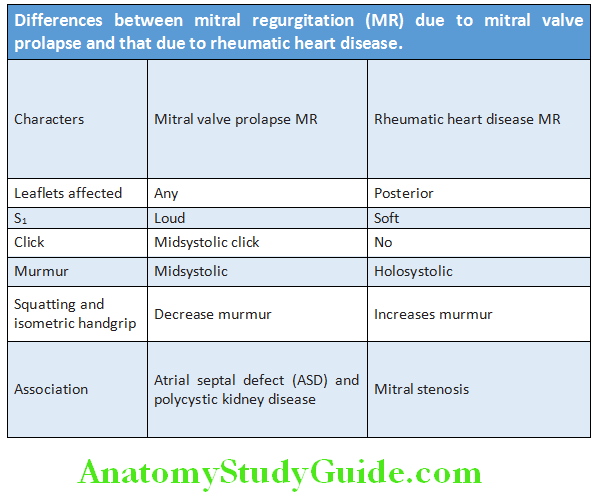
Mitral regurgitation Investigations:
- Electrocardiogram (ECG) It can reveal:
- Enlargement/hypertrophy of left atrium (if not in atrial fibrillation)
- Dilatation and hypertrophy of left ventricle
- Hypertrophy of both left and right ventricle in pulmonary hypertension
- Atrial fibrillation
- Chest X-ray May show the following abnormalities All signs as described for MS:
- Enlargement of the left atrium (more than in MS) and the left ventricle
- Pulmonary venous congestion
- Pulmonary edema: Interstitial edema in acute MR, chronic decompensated MR, or with coexistent MS
- Annular calcium appears as a C-shaped opacity in posterior third of heart in lateral or RAO (right anterior oblique) view.
- Echocardiogram:
- Shows a dilated left atrium and left ventricle
- Structural abnormalities of mitral valve, e.g., prolapsed, chordal, or papillary muscle ruptures, if present
- Doppler echocardiogram:
- Detects and assesses the severity of regurgitation (quantification of regurgitation)
- Mitral annular calcification between mitral valve apparatus and posterior wall
- Transesophageal echocardiography (TEE): Useful to identify structural valve abnormalities and can be helpful before surgery, especially in MVP. Intraoperative TEE helps in the assessment of the efficacy of valve repair. MVP is defined as >2 mm systolic displacement of mitral leaflet into the left atrium (LA).
- Cardiac catheterization: This can show dilated left atrium and left ventricle mitral regurgitation, pulmonary hypertension, and coexisting coronary artery disease (if present).
Complications of mitral regurgitation:
- Progressive heart failure is the most common cause of death.
- Less frequent: Sudden death, stroke, and fatal endocarditis
- Atrial fibrillation, infective endocarditis, and left ventricular failure
- Pulmonary hypertension (late) and right ventricular failure (very late)
- Rarely systemic embolism
Mitral regurgitation Management:
- Asymptomatic mild mitral regurgitation can be managed conservatively.
- Moderate mitral regurgitation can be treated medically.
Medical Management:
- Acute MR: Afterload reduction with nitroprusside
- Chronic MR: In mitral regurgitation, high afterload may worsen the degree of regurgitation, and hypertension is treated with vasodilators, e.g., ACE inhibitors and nifedipine are used.
- Diuretics
- Treatment of AF: Digoxin and anticoagulants
- Anticoagulation
- Infective endocarditis prophylaxis
- Rheumatic fever prophylaxis
Surgery Mitral Valve Replacement:

Conditions in which repair of mitral valve is possible:
- Mitral valve prolapse (MVP)
- Papillary muscle dysfunction
- Leaflet perforation due to infective endocarditis (IE)
- Chordal rupture
- Annular dilation
- Rheumatic mitral regurgitation (MR) in the young
Combined Mitral Stenosis and Mitral Regurgitation:
Clinical assessment of the dominance of lesions:
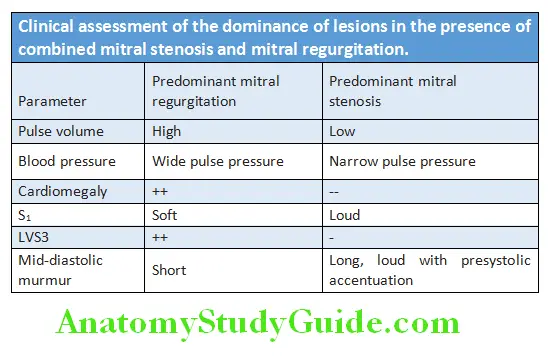
Mitral Valve Prolapse (MVP):
Question 82. Write a short essay/note on mitral valve prolapse (MVP) or Barlow’s syndrome.
Answer:
- Mitral valve prolapse (MVP) is an abnormal movement of one or both of the mitral valve leaflets >2 mm beyond annular plan into the left atrium during systole with or without mitral regurgitation.
- Barlow’s syndrome: Involves the posterior leaflet and cusps. Reads syndrome/floppy valve syndrome is considered as a variant of Barlow that affects both mitral cusps as well-other valves like aortic valve.
- Cobb’s syndrome selectively affects the anterior mitral cusp.
- MVP syndrome is also called as systolic click-murmur syndrome.
- MVP is one of the more common causes of mild mitral regurgitation.
Pathogenesis:
- It is caused by congenital anomalies or degenerative myxomatous changes (e.g., Marfan syndrome and cystic medical necrosis) that are associated with excessive or redundant mitral leaflet tissue.
- Familial incidence with an autosomal dominant mode of inheritance.
- Prolapse of the cusp occurs in systole → tensing of the chordae → pulling of papillary muscles which sometimes may interfere with blood supply.
- Conditions associated with MVP: Atrial septal defect (20%), polycystic kidney disease, chronic rheumatic heart disease, ischemic heart disease, and cardiomyopathies.
Mitral Valve Prolapse Clinical Features:
Symptoms:
- Age and gender: More common in females between 15 and 30 years. But, severe mitral regurgitation caused by prolapsed mitral valve is more common in older males compared to young females.
- May be asymptomatic or present with anxiety neurosis
- Symptoms include atypical chest pain (precordial stabbing), palpitation, syncope or presyncope, and fatigue.
- Rarely: Symptoms of left ventricular failure (exertional dyspnea, orthopnea, and paroxysmal nocturnal dyspnea) in patients with mitral regurgitation, sudden death, and transient ischemic attacks.
Mitral Valve Prolapse Signs:
- Asthenic built, straight back/pectus excavatum
- Mid-systolic click: In mildest forms, the valve is competent but bulges back into the atrium during systole, causing a midsystolic click to occur >0.14 seconds after S1 without any murmur.
- Late systolic murmur: When there is mitral regurgitation, the click is followed by a late systolic murmur apical murmur (rarely “whooping” or “honking”). Its length increases as the regurgitation becomes more severe. The systolic murmur of mitral valve prolapse increases during standing and Valsalva maneuver, but decreases during squatting and isometric exercise.
Mitral Valve Prolapse Investigations:
- Electrocardiogram (ECG): Nonspecific ST-T changes or inverted T waves in leads II, III, and aVF
- Echocardiography: Used for confirmation of the diagnosis. Color Doppler is used to assess the degree of mitral regurgitation.
Mitral Valve Prolapse Complications:
- These include arrhythmias, sudden rupture of the chordate, progressive mitral regurgitation, and infective endocarditis.
- Rare complications include transient cerebral ischemic attacks, embolism, acute severe mitral regurgitation, and sudden death.
Management:
- Medical:
- Reassurance for patients without symptoms
- β-blockers: For atypical chest pain
- Antiarrhythmic drugs: For the treatment of arrhythmias
- Transient ischemic attacks: Aspirin, dipyridamole, or anticoagulants
- No need for infective endocarditis prophylaxis
- Surgical: Mitral valve repair or replacement in severe mitral regurgitation
Prognosis: Long-term prognosis is good.
Aortic Stenosis:
Question 83. Write a long essay on the classification, causes/etiology, clinical features, investigations, and management of valvular aortic stenosis.
Answer:
Aortic stenosis is a chronic progressive disease and characterized by obstruction to the left ventricular stroke volume.
Normal aortic valve area 3–4 cm² without any gradient across it. Critical aortic stenosis develops when an aortic valve area becomes <0.8 cm² or a gradient of >50 mm Hg.
Classifiation and Etiology of Aortic Stenosis:
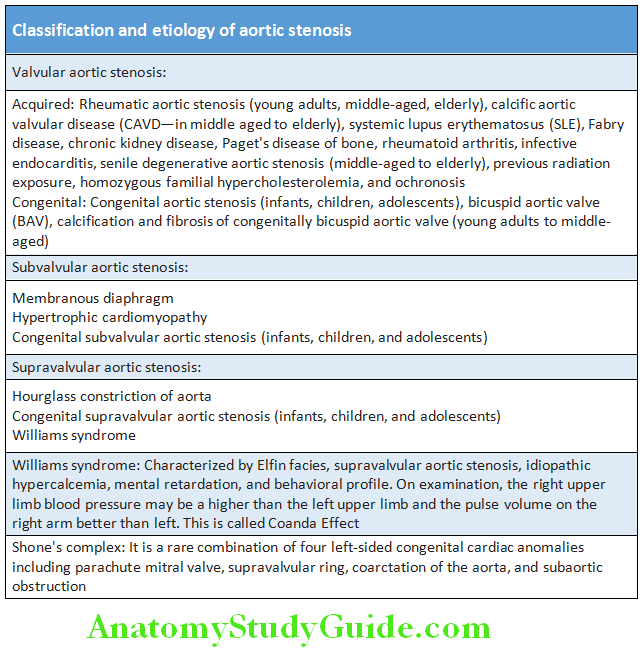
Aortic Stenosis Pathophysiology:
- Normal aortic valve area is 2.5–3.5 sq. cm.
- Hemodynamically significant AS occurs when the aortic valve is reduced by 60–75%.
- Critical AS when valve area <0.8 cm2/gradient >80%.
- Initially, cardiac output is maintained. Pathological hallmark is fixed outflow obstruction to the left ventricle. It limits the increase in cardiac output required during exercise. The coronary blood flow may be inadequate and patients may develop angina even in the absence of concomitant coronary disease. Obstruction to the outflow leads to left ventricular hypertrophy (LVH). Wall stress due to outflow obstruction manifests in two forms.
- Development of concentric LVH: Wall stress is not allowed to rise by development of concentric LVH. This type is more likely in females.
- Ventricular dilation and eccentric hypertrophy: Rise in wall stress and systolic dysfunction are features of this type. This type is more likely in males.
- Later the left ventricle cannot overcome the outflow tract obstruction and pulmonary edema develops. In aortic stenosis, pulmonary arterial hypertension is due to increased LV diastolic pressure.
Aortic Stenosis Clinical Features:
Usually, asymptomatic aortic stenosis is moderately severe (aortic orifice reduced to one-third of its normal size). It is commonly diagnosed in asymptomatic patients during routine clinical examination.
Symptoms:
Question 84. Write a short note on symptomatic triad of aortic stenosis.
Answer:
- Age of onset of symptoms: Bicuspid aortic valve between 50 and 70 years, calcific aortic stenosis >70 years, and rheumatic aortic stenosis between 40 and 50 years of age.
- Symptomatic triad of aortic stenosis: Three cardinal symptoms are
- Breathlessness (Dyspnea)
- Angina
- Syncope.
- Exercise intolerance: Most common initial presentation. Dyspnea with exertion due to cardiac decompensation or fatigue with exertion due to inadequate rise of cardiac output with exertion.
- Angina is typical exertional and is due to mismatch in myocardial oxygen demand-supply ratio, or coexistent coronary artery disease (CAD).
- Exertional syncope (or presyncope): Due to failure of cardiac output to rise to meet demand, leading to a fall in BP.
- Heart failure: Orthopnea and paroxysmal nocturnal dyspnea (PND).
- The average survival after the onset of symptoms were 5, 3, and 2 years respectively for angina, syncope, and dyspnea.
- Infective endocarditis
- Embolism
- Gastrointestinal bleed due to angiodysplasias (Heyde syndrome), acquired von Willebrand syndrome.
If untreated, the approximate time interval from the onset of symptoms to death is 1.5–2 years for heart failure (dyspnea, PND), 3 years for syncope, and 5 years for angina.
Symptoms Signs:
- Appearance: Severe AS produces “Dresden China” look-asthenic appearance with pale skin.
- Pulse:
- Parvus and tardus pulse: Slow rising, late peaking, and low amplitude
- Pulsus bisferiens if AS is associated aortic regurgitation (AR)
- Anacrotic pulse and apicocarotid delay in severe AS
- Coanda effect in supravalvular AS
- Normal pulse if there is coexistent AR, hypertension (HTN)
- Blood pressure:
- Low systolic and pulse pressures. Systolic decapitation (SBP <120 mm Hg)
- Normal BP if there is coexistent AR and HTN
- Carotid shudder: Thrill in carotids
- Jugular venous pressure (JVP): Prominent a waves (Bernheim effect)
- Apex beat: Thrusting/heaving LV type apex beat due to LV pressure overload. It is not usually displaced because hypertrophy (as opposed to dilatation) does not produce significant cardiomegaly.
- Palpable LV S4
Symptoms Auscultation:
- S1 is normal. If loud suspect coexistent MS.
- S2-A2: Soft (single S2) in rheumatic AS, loud in BAV, normal in sub/supravalvular AS. In severe AS, paradoxical (reversed) split of second heart sound A2 (splitting on expiration)
- Systolic ejection click in bicuspid aortic valve (BAV):
- Ejection mid-systolic murmur: Harsh/rough, radiates to the neck (to carotids arteries) and also the precordium with late peaking in severe AS. Best heard in the base of heart (aortic area). Murmur is likened to a saw cutting wood and especially in older patients may have a musical quality like the “mew” of a seagull. It is diamondshaped (crescendo decrescendo). The murmur is usually longer in severe AS. The intensity of the murmur should not be correlated with the severity of AS because it is less intense when the cardiac output is reduced. In severe AS, it may not be audible. In calcific AS, high-frequency components radiate to apex producing a long systolic murmur at apex—Gallavardin phenomenon/hour-glass conduction due to periodic wake phenomenon. With LV failure, murmur intensity decreases and murmur may disappear.
Differences between aortic stenosis and mitral regurgitation presented in Table:
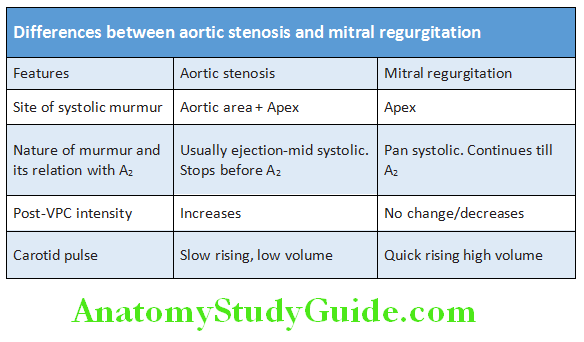
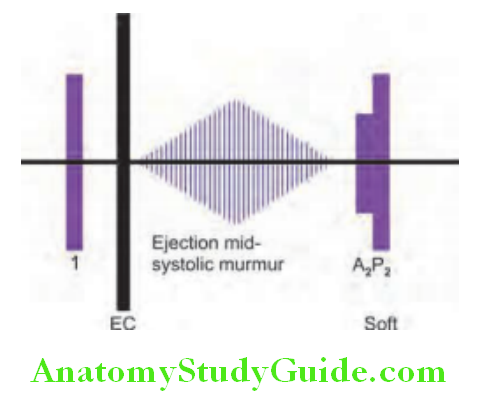
Severity of aortic stenosis:
Signs indicating severity of aortic stenosis:
- Mild: Only murmur (early peaking)
- Moderate: Low volume pulse, mid-systolic peaking murmur, pulsus parvus et tardus
- Severe: Anacrotic pulse, paradoxical split S2, late peaking murmur, palpable S4 apicocarotid delay
Severity of aortic stenosis Investigations:
- Chest X-ray:
- Rounding of apex due to concentric left ventricular hypertrophy with normal heart size or a relatively small heart
- Prominent dilation of ascending aorta (more prominent if bicuspid aortic valve) due to turbulent blood flow above the stenosed aortic valve causing poststenotic dilatation
- Calcification of aortic valve (if present)
- Electrocardiogram (ECG):
- Left ventricular hypertrophy correlation between QRS voltage
- Atrial fibrillation in 10–15%
- AV conduction defects and IVCD in 5% of calcific AS due to extension of calcium to conduction system
- Echocardiography:
- Helps in evaluation of severity and gradient across the aortic valve
- Dobutamine stress echo:
- Severe AS: Increase in gradient. No change in valve area
- Mild AS: Increase in valve area
- Also assesses contractile reserve and predicts improvement in LV function after surgery
- Doppler: Useful for detecting the severity of stenosis and associated aortic regurgitation
- Cardiac magnetic resonance and cardiac CT: They may be required for assessing the presence of aneurysm, dissection, or coarctation of thoracic aorta.
- Cardiac catheterization is rarely needed because since all these information can be obtained from noninvasive with echocardiography and CMR. Coronary angiography is needed before aortic valve surgery and for detecting any associated coronary artery disease.
Severity of aortic stenosis Treatment:
Asymptomatic patients: Irrespective of the severity of AS, asymptomatic patients have a good immediate prognosis. Hence, should be managed conservatively with regular review for assessment of symptoms and echocardiography.
Medical treatment:
- Avoid vigorous physical activity in patients with severe AS.
- Diuretics decrease dyspnea but may also reduce cardiac output.
- ACE inhibitors: To be used with caution and given only if there is LV failure
- Avoid beta-blockers: Because they produce LV failure.
- Vasodilators for other purposes such as angina: Be careful in titration as there will be no compensatory increase in cardiac output.
- Atrial fibrillation: Cardioversion can be tried.
- Infective endocarditis prophylaxis for those who have undergone valvular replacement
- Rheumatic fever prophylaxis
Surgical treatment:
Indications:
- Symptomatic patients: Symptoms are a good index of severity in aortic stenosis, and all symptomatic patients should undergo aortic valve replacement.
- Asymptomatic patients: Surgical intervention for severe aortic stenosis is recommended in patients with:
- Symptoms during an exercise test or with a fall of blood pressure
- Left ventricular ejection fraction of <50%
- Moderate-to-severe aortic stenosis undergoing CABG, surgery of the ascending aorta, or other cardiac valve.
Surgical procedures:
- Balloon dilatation (valvuloplasty) may be tried in adults (especially elderly) as an alternative to surgery. But results are poor and they should be reserved for patients unfit for surgery or as a “bridge” to surgery (as systolic function will often improve).
- Aortic valve replacement: The above procedure causes only temporary relief from the obstruction and aortic valve replacement will be usually needed after few years.
- Percutaneous valve replacement: Transcatheter implantation with a balloon expandable stent valve may be done for patients unsuitable for surgical aortic valve replacement.
- ROSS procedure: Replacement of deceased aortic valve by pulmonic valve and implantation of homograft instead of native pulmonic valve.
- Transcatheter aortic valve replacement (TAVR) and transcatheter aortic valve implantation (TAVI) are procedures for select patients with severe symptomatic aortic stenosis.
Aortic Sclerosis:
Question 85. Write a short note on aortic sclerosis.
Answer:
- Aortic sclerosis is characterized by irregular thickening of the aortic valve leaflets seen on echocardiography but without significant aortic obstruction. It may produce a systolic ejection murmur without any symptoms. Pulse and BP will be normal.
- About 25% patients are above the age 65 years and 40% over 85 years.
- Risk factors: Dyslipidemia, diabetes, hypertension, and smoking. Higher prevalence in Paget disease and end-stage renal disease (ESRD)
- There may be coexisting mitral annular calcification. Even if there is no AS, calcific aortic sclerosis increases cardiovascular death and MI by 50%.
- Rate of progression is variable so difficult to predict in an individual patient. The valve area declines 0.1–0.3 cm2 per year; the systolic pressure gradient across the valve can increase by as much as 10–15 mm Hg per year.
Aortic Sclerosis Treatment:
- No medical treatment available.
- Rosuvastatin has been shown to decrease progression of less severe AS to severe AS. Other drugs tried include ACE inhibitors.
Bicuspid Aortic Valve (BAV) Disease:
Question 86. Write a short note on bicuspid aortic valve disease.
Answer:
- Bicuspid aortic valve is the most common congenital heart disease found in 1–2% of live births.
- Male-to-female ratio is 3:1.
- Some cases are inherited as autosomal dominant and NOTCH1 gene mutation are found in some cases.
- Associated conditions: Other congenital cardiac diseases (e.g., dilatation of proximal ascending aorta secondary to abnormalities of the aortic media), coarctation of aorta, ventricular septal defect (VSD), and atrial septal defect (ASD).
- Calcification is common in adults with bicuspid aortic valve.
- Auscultatory findings: Ejection click best heard at the apex and may be associated murmurs of aortic stenosis or aortic regurgitation.
- Diagnosis: By echocardiography
Bicuspid Aortic Valve Complications:
- Aortic regurgitation: About 20% develop severe AR
- Aortic stenosis: Due to calcification and severe AS occurs after 50 years of age
- Infective endocarditis
- Ascending aortic dilation: Due to medial degeneration and is not related to severity of aortic stenosis
- Aortic dissection: Risk is increased by five to nine times.
Bicuspid Aortic Valve Treatment:
- Medical management: Control of hypertension and use of β-blockers.
- Surgical treatment: Aortic valve replacement and may also require aortic root surgery.
Bicuspid Aortic Valve Prognosis:
Life-expectancy is usually not shortened.
Aortic Regurgitation:
Question 87. Discuss the etiology, clinical features, complications, investigations, and treatment of aortic regurgitation.
Answer:
Aortic regurgitation (AR) is incompetency of the aortic valve which causes backflow (reflux) of blood from the aorta through the aortic valve into the left ventricle during diastole.
Aortic Regurgitation Etiology:
- Aortic regurgitation may be caused by diseases of the aortic valve (e.g., endocarditis) or diseases of the aortic root (e.g., Marfan syndrome) causing dilatation of the aortic root.
- Aortic regurgitation can be acute or chronic.
- Acute aortic regurgitation of significant severity leads to sudden increased blood volume in the left ventricle during diastole. The left ventricle does not have sufficient time to dilate in response to the sudden increase in volume. This causes rapid increase in end-diastolic pressure of left ventricle → leading to an increase in pulmonary venous pressure. As pressure increases throughout the pulmonary circulation, the patient develops dyspnea and pulmonary edema. In severe cases, left ventricular failure with cardiogenic shock may develop. Decreased myocardial perfusion may lead to myocardial ischemia.
- Chronic aortic regurgitation: Produces increased blood volume in the left ventricle during diastole. To maintain the net cardiac output, the total volume of blood pumped from the left ventricle into the aorta increases, and consequently the left ventricle enlarges. AR begets AR. There is fall in the diastolic blood pressure and decreased coronary perfusion.
- Myocardial ischemia occurs due to:
- Increased myocardial oxygen demand due to increased afterload, increased LV ejection time, and increased LV mass
- Decreased myocardial oxygen supply due to decreased aortic diastolic pressure, decreased diastolic filling time due to increased LV ejection time, and decreased effective stroke volume.
Causes of aortic regurgitation:
Acute aortic regurgitation:
- Infective endocarditis
- Aortic dissection
- Acute rheumatic fever
- Ruptured sinus of Valsalva aneurysm
- Failure of prosthetic valve
- Trauma
Chronic aortic regurgitation:
- Congenital: Bicuspid aortic valve or disproportionate cusps
- Acquired:
- Rheumatic heart disease
- Infective endocarditis
- Trauma
- Aortic dilatation, e.g., aneurysm and dissection
- Arthritides, e.g., ankylosing spondylitis
- Rheumatoid arthritis
- Syphilitic aortitis
- Hypertension
- Connective tissue disorders, e.g., Marfan syndrome, Ehlers–Danlos syndrome
- Osteogenesis imperfecta and methysergide
Aortic Regurgitation Clinical Features:
Symptoms:
- Age and gender: About three-fourths of patients with pure or predominant valvular AR are males. Females with primary valvular AR have associated rheumatic mitral valve disease. Chronic AR usually begins during late 50s. Usually, the prevalence and severity of AR increase with age.
- Acute AR:
- Sudden, severe shortness of breath
- Chest pain if myocardial perfusion pressure is decreased or an aortic dissection is present
- Rapidly developing heart failure, pulmonary edema, and cardiogenic shock
- Chronic AR:
- Relatively asymptomatic for 10–15 years and significant symptoms occur late. Symptoms may not develop until left ventricular failure occurs.
- Palpitations, especially on lying down, may persist for many years. Sinus tachycardia, during exertion or with emotion, may produce palpitations as well as head pounding.
- Exertional dyspnea: Usually, the first symptom of diminished cardiac reserve. It depends on the extent of left ventricular dilatation and dysfunction. It is followed by orthopnea, paroxysmal nocturnal dyspnea, and excessive diaphoresis.
- Angina pectoris: Anginal chest pain develops even in the absence of coronary artery disease (CAD) at rest as well as during exertion. Nocturnal angina may be accompanied by marked diaphoresis. These episodes of angina can be prolonged and usually do not respond satisfactorily to sublingual nitroglycerine.
- Mechanisms of angina in AR:
- Associated CAD
- Low aortic diastolic pressure leading to decreased myocardial perfusion (because coronary flow occurs mainly in diastole)
- Increased myocardial oxygen demand due to myocardial hypertrophy
- Congestive heart failure
- Sudden cardiac death
- Arrhythmias are not common.
Signs:
Question 88. Write a short essay/note on peripheral signs of aortic regurgitation.
(or)
Write a short note on Austin Flint murmur.
Answer:
Peripheral signs of aortic regurgitation:
Pulses:
- Peripheral pulses: Prominent, large-volume (bounding), or collapsing
- Low-diastolic and increased/wide pulse pressure: Pulse pressure is the difference between the systolic and diastolic blood pressures. In aortic regurgitation, there is increased/wide pulse pressure. Shows list of (rare) signs of wide pulse pressure that indicates a hyperdynamic circulation in aortic regurgitation.
Inspection and palpation:
- Prominent neck pulsations and thrill in the carotids
- Apex beat: Displaced inferiorly and toward the axilla. The point of maximal impulse may be diffuse or hyperdynamic.
- Pre-systolic impulse
- Diastolic thrill: May be palpable along the left sternal border in patients with thin-chest (Erb’s maneuver)
- Prominent systolic thrill: May be palpable in the suprasternal notch and transmitted upward along the carotid arteries
Auscultation:
- First heart sound (S1): May be soft
- A2 component of second heart sound (S2): Soft in rheumatic aortic regurgitation and loud and “tambour” like in syphilitic AR. Narrowly split, single, or paradoxically split second heart sound.
- S3 gallop, third heart sound (S3), if left ventricular dysfunction is present.
- Fourth heart sound (S4) prominent in left ventricular hypertrophy
- Murmurs:
- Early-diastolic murmur: Early-holodiastolic murmur, immediately after A2, usually as a high-pitched blowing sound that is loudest at the left sternal border, and decrescendo. Best heard in at the end of expiration, in sitting and leaning forward position. The duration of the murmur correlates better with the severity of AR than does the loudness of the murmur.
- Cole-Cecil murmur: It is the term used for the diastolic murmur of AR when well or predominantly heard in the left axilla.
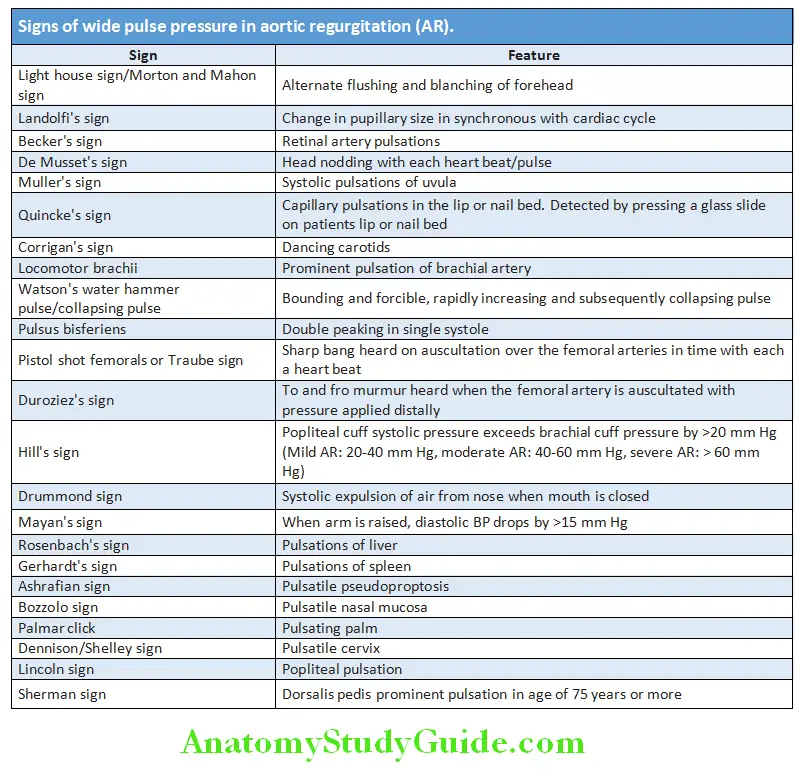
-
- If the murmur is musical (cooing dove murmur), it signifies eversion or perforation of an aortic cusp.
- Harvey sign: When regurgitation is caused by primary valvular disease, the diastolic murmur is heard best along the left sternal border in the 3rd and 4th intercostal space. When murmur is caused mainly by dilation of the ascending aorta, the murmur is more readily audible along the right sternal border.
- A functional systolic flow murmur may also be present because of increased stroke volume, although concurrent AS may also be present.
- Austin Flint murmur: It may be audible at the cardiac apex in severe AR. It is a low-pitched, mid-diastolic rumbling murmur caused due to:
- AR jet impinges on anterior mitral leaflet (AML) forces it down and reduces the mitral orifice.
- Turbulence produced when AR jet meets mitral inflow jet
- AML fluttering due to AR jet
- LV endocardial vibrations due to AR jet
- The auscultatory features of AR are intensified by strenuous and sustained handgrip (increases systemic vascular resistance).
- As heart failure develops, peripheral vasoconstriction may occur and arterial diastolic pressure may rise, even though severe AR is present. Thus, pulse pressure can be normal or narrow in severe AR.
Causes of AR with normal/low pulse pressure:
- Acute AR
- AR with AS or severe MS
- AR with CHF
- AR
Indicators of severity of aortic regurgitation:
- Duration of murmur: If murmur > twothirds of diastole, murmur becomes holodiastolic and rough in quality
- Bisferiens pulse
- S3
- Positive Hill’s sign >60 mm Hg
- Cardiomegaly: Apical impulse displaced down and out
- Austin Flint murmur
- A2 soft
- Marked peripheral signs
Acute Aortic Regurgitation:
- Clinical presentation: Sudden in onset with acute left ventricular failure
- Signs:
- Pulse pressure: Normal and near normal systolic and diastolic pressures
- S1: Soft or absent
- P2: Normal or increased
- S3: Usually audible
- Murmur:
- Early diastolic murmur is short and functional systolic murmur across aortic valve is less loud.
- Austin Flint murmur is usually absent.
- Investigations of acute aortic regurgitation:
- ECG and chest X-ray: Do not show evidence of left ventricular enlargement. Chest X-ray may show redistribution of upper lobe vessels due to pulmonary venous hypertension.
Investigations of Chronic Aortic Regurgitation:
- Chest X-ray: It shows cardiomegaly due to the enlargement/dilatation of the left ventricle in an inferior and leftward direction. The ascending aorta (and the aortic arch or knob) is also severely dilated. Left atrial enlargement is not found unless there is significant left ventricular dysfunction. Calcification of the ascending aortic wall and the aortic valve may be observed in syphilitic aortic regurgitation. Later, it may show features of left heart failure.
- Electrocardiography (ECG): It shows the adaptive changes in the left ventricle due to the volume overload.
- These include:
- LV hypertrophy: Tall R waves and deeply inverted T waves in the left-sided chest leads,
- Left Axis Deviation,
- Left atrial enlargement,
- LV volume overload pattern (prominent Q waves in leads I, aVL, and V3 to V6 and relatively small r waves in V1)
- LV conduction defects (typically late in the disease process).
- Echocardiography:
- Aortic valve structure and morphology (e.g., bileaflet versus trileaflet, flail, thickening).
- Presence of vegetations or nodules (may require transesophageal echocardiography in selected cases)
- Quantitative measurements of regurgitant volume, fraction and orifice area assessed
- Fluttering anterior mitral leaflet
- Regurgitant volume, fraction, and orifice area
- Associated lesions of the aorta, e.g., dilation, aneurysm, dissection, or ectasia
- LV structure and function
- Doppler echocardiography: To assess the severity. Transesophageal echocardiography is not usually needed.
- Cardiac magnetic resonance and cardiac CT: To assess the thoracic aorta for the aneurysm, dissection or coarctation but are rarely needed. Cardiac MR can quantify regurgitant volume.
- Cardiac catheterization
- Coronary angiography: Indicated for the assessment of coronary anatomy prior to aortic valve surgery in patients with risk factors for coronary artery disease
- Other tests:
- VDRL and TPHA: If syphilitic cause is suspected
- RA factor, ANA, ESR and CRP: To exclude connective tissue disorders.
Question 89. Write a short essay/note on the management of acute severe aortic regurgitation in pulmonary edema.
Answer:
Pulmonary edema Treatment:
- Underlying cause of AR-like dissection, endocarditis, syphilis has to be treated.
- Prophylaxis: Rheumatic fever prophylaxis is needed if due to rheumatic. Infective endocarditis prophylaxis is not required.
- Acute severe AR: Surgical intervention is usually needed, but the patient may be medically supported with dobutamine to augment cardiac output and shorten diastole and sodium nitroprusside to reduce afterload in hypertensive patients.
- Chronic severe AR
Medical treatment:
- Vasodilator therapy may be used in selected conditions to reduce afterload in patients with systolic hypertension to reduce wall stress and optimize LV function. In normotensive patients, vasodilator therapy may not be useful because it does not reduce regurgitant volume (preload) significantly.
- The acute administration of sodium nitroprusside, hydralazine, nifedipine, or felodipine reduced PVR and results in an immediate augmentation in forward cardiac output and a reduction in regurgitant volume. Nitroprusside and hydralazine induced acute hemodynamic changes lead to a consistent decrease in end-diastolic volume (EDV) and an increase in ejection fraction (EF).
Surgical treatment:
- AR usually requires replacement of the diseased valve with a prosthetic valve, although valve-sparing repair is available like transcatheter aortic valve replacement/implantation (TAVR/TAVI).
- Indications for aortic valve surgery
- Types of valves: For patients undergoing AV replacement, careful consideration should be given to the relative risks and benefits of mechanical versus bioprosthetic valves.
- Mechanical valves: More durable but require long-term anticoagulation with warfarin due to increased risk of thrombosis
- Bioprosthetic (tissue) valves carry a greater risk of long-term deterioration (e.g., rapid calcification and degeneration of the valves) and risk of reoperation but avoid the need for long-term warfarin. Thus, they are preferred in the elderly and when anticoagulants must be avoided. They are contraindicated in children and young adults.
- Aortic root dilation causing AR: For example, Marfan syndrome. Treated by encircling suture or subcommissural annuloplasty, or aortic graft with prosthetic valve (coronaries need to be reimplanted) or aortic graft alone.
Indications for aortic valve surgery:
- Patient is symptomatic (dyspnea, NYHA class IIIB, angina) with chronic severe aortic regurgitation
- Patient is asymptomatic, with a resting left ventricular ejection fraction (LVEF) of <55%
- Patient is asymptomatic, with left ventricular (LV) ejection fraction >55% but with left ventricle dilation (LV end-diastolic dimension >70 mm or end-systolic dimension >55 mm)
- Fractional shortening (FS) <0.27
- Acute severe aortic regurgitation, e.g., endocarditis
- If undergoing CABG, surgery of the ascending aorta or other cardiac valve
Difference between rheumatic and syphilitic aortic regurgitation:
Question 90. What are the differences between rheumatic and syphilitic aortic regurgitation?
Answer:
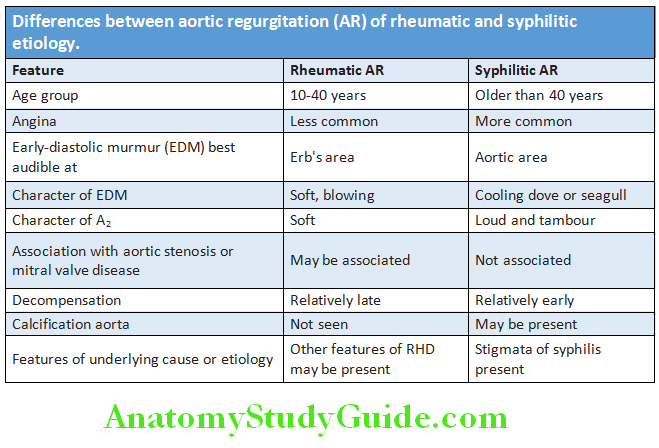
Diffrence between Acute and Chronic Aortic Regurgitation:
Question 91. What are the differences between acute and chronic aortic regurgitation?
(or)
What are the differences between Austin Flint murmur and the murmur of mitral stenosis (MS)?
Answer:
Difference between Austin Flint murmurs from the murmur of mitral stenosis:
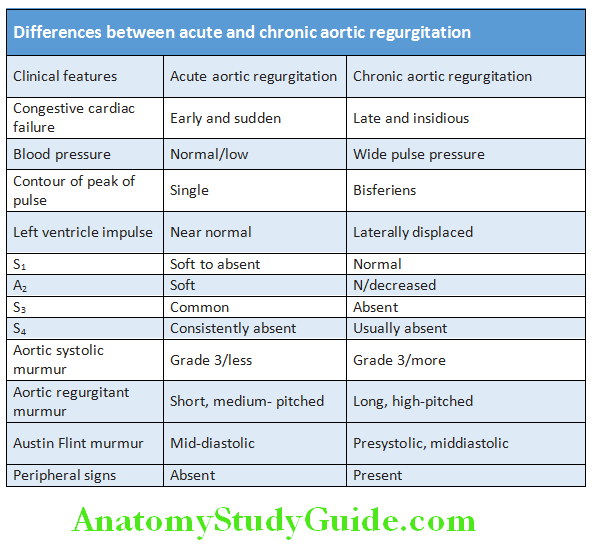
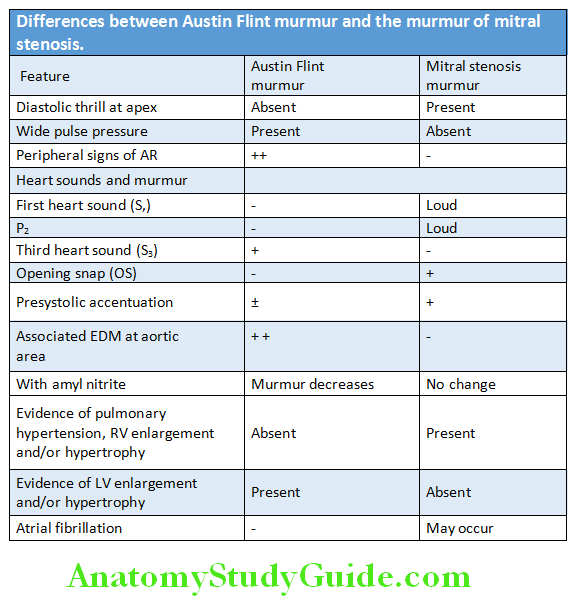
Tricuspid Stenosis (TS):
Question 92. Write a short essay/note on tricuspid stenosis (TS).
Answer:
Tricuspid stenosis is a narrowing of the tricuspid valve opening. Uncommon valve lesion that is more common in females than males.
Etiology:
Tricuspid Stenosis Pathophysiology:
Tricuspid valve stenosis reduces the cardiac output, which is restored to normal when the pressure of right atrium increases—results in systemic venous congestion—hepatomegaly, ascites, and dependent edema.
Causes of tricuspid stenosis:
- Rheumatic (frequently associated with mitral and/or aortic valve disease)
- Carcinoid syndrome: Tricuspid stenosis and regurgitation are found in carcinoid syndrome
- Congenital
- Infective endocarditis
- Anorectics: Fenfluramine
- Fabry disease
Tricuspid Stenosis Clinical Features:
Symptoms:
- Usually, symptoms are due to associated left-sided rheumatic valve lesions (e.g., mitral stenosis)
- Tricuspid stenosis may produce symptoms of right heart failure: Abdominal pain/hepatic discomfort (due to hepatomegaly), peripheral edema, and ascites
- Little or no dyspnea and fatigue is common.
Signs:
- Jugular venous pressure (JVP): JVP is raised with prominent a waves (giant a waves) and slowly descent due to the loss of normal rapid right ventricular filling.
- Mid-diastolic murmur: Loud first heart sound and a rumbling mid-diastolic murmur (higher-pitched than the murmur of mitral stenosis) with presystolic accentuation, best heard at the lower left sternal border and increases/louder during inspiration (De Carvallo’s sign).
- Tricuspid opening snap (OS): Occasionally heard
- Right heart failure: Hepatomegaly with presystolic pulsation felt over the liver, ascites, and peripheral edem
Tricuspid Stenosis Investigations:
- Chest X-ray: May show a prominent right atrial bulge
- ECG: Enlarged right atrium
- Echocardiogram: May reveal thickened and immobile tricuspid valve.
Tricuspid Stenosis Treatment:
- Medical management: Diuretic therapy and salt and fluid restriction
- Surgical treatment: Tricuspid valve replacement.
Tricuspid Regurgitation:
Question 93. Write a short essay/note on tricuspid regurgitation. What is De Carvallo’s sign?
Answer:
In tricuspid regurgitation, tricuspid valve does not close properly, causing blood to flow backward (leak) into the right atrium.
Tricuspid Regurgitation Etiology:
Tricuspid regurgitation is common. Most commonly, it is functional secondary to right ventricular dilatation.
Causes of Tricuspid Regurgitation:
- Primary/organic: Rheumatic heart disease, endocarditis (IV drug abuse), Ebstein anomaly and carcinoid syndrome
- Secondary:
- Functional tricuspid regurgitation: Right ventricular dilatation due to chronic left heart failure
- Right ventricular infarction, inferior wall infarction
- Pulmonary hypertension (e.g., cor pulmonale)
- Cardiomyopathy
Tricuspid Regurgitation Clinical Features:
- Symptoms are usually nonspecific. Tiredness due to reduced forward flow.
- Valvular regurgitation increases the right atrial and systemic venous pressure. Patients may develop of right heart failure, i.e., edema, ascites, and hepatomegaly with systolic pulsations due to venous congestion.
- JVP: Raised with most prominent “giant” v wave in the jugular venous pulse (a cv wave replaces the normal x descent)
- Earlobe pulsations (Lancisi’s sign)
- Blowing pansystolic murmur: At the lower-left sternal border that is increased during inspiration and reduced during expiration (De Carvallo’s sign).
- P1 may be loud (due to pulmonary hypertension) and RV S3 is heard.
- Severe TR: Right jugular venous thrill and an RV impulse at the left lower sternal border. Long-standing severe TR may lead to RV dysfunction-induced heart failure and atrial fibrillation (AF)
- Echocardiogram: It shows dilated right ventricle and thickened valve.
Tricuspid Regurgitation Treatment:
Functional tricuspid regurgitation due to right ventricular dilatation usually disappears with treatment of the underlying cause of right ventricular overload. Treatment consists of with diuretic and vasodilator treatment of congestive cardiac failure.
Organic tricuspid regurgitation: TR is usually well tolerated when there is normal pulmonary artery pressure. Severe organic tricuspid regurgitation may require operative repair of the tricuspid valve by annuloplasty or plication or valve repair, or occasionally tricuspid valve replacement. In drug addicts with infective endocarditis of the tricuspid valve, surgical removal of the valve is recommended to eradicate the infection.
Pulmonary Stenosis:
Question 94. Write a short essay/note on pulmonary stenosis.
Answer:
Pulmonary stenosis (PS) is characterized by obstruction to blood flow from the right ventricle to the pulmonary artery.
Pulmonary Stenosis Etiology:
- Congenital: Usually, PS is a congenital lesion. It may be associated with rubella during pregnancy. May be an isolated lesion or associated with a ventricular septal defect (Fallot’s tetralogy).
- Acquired:
- Rheumatic (very uncommon)
- Carcinoid syndrome
Pulmonary stenosis may be classified as valvular, subvalvular (infundibular), or supravalvular.
Pulmonary Stenosis Clinical Features:
Symptoms:
- Mild pulmonary stenosis may be asymptomatic.
- Fatigue, syncope, dyspnea, and the symptoms of right heart failure. Angina or syncope indicates severe pulmonary stenosis.
Signs:
- Systolic thrill: The ejection systolic murmur (described below) is often associated with a thrill. It is best felt when patient sits up, learns forward, and breathes out.
- Right ventricular heave (sustained impulse) and epigastric pulsation may be felt.
- Pulmonary closure sound (P3) is soft and delayed.
- Wide splitting of second heart sound: Due to delay in right ventricular ejection
- Ejection systolic murmur: It is a principal physical sign. It is best heard on inspiration at the left upper sternum in the second intercostal space. It radiates toward the left shoulder. With progressive severity, the murmur gets harsh/louder longer and peaks later in systole. This murmur is usually preceded by an ejection sound (click).
- Ejection click: Mild stenosis is characterized by a systolic ejection click which is often more prominent in expiration. This seemingly paradoxical behavior of the pulmonary ejection click is explained by an inspiratory increase in RV end-diastolic pressure, which opens the valve in late diastole and hence absence of systolic ejection click during inspiratory phase.
- High-pitched diastolic murmur after a prominent P2 may be evident in patients with pulmonary regurgitation secondary to pulmonary hypertension (Graham Steell murmur).
- If stenosis is valvular, pulmonary ejection sound/click will be heard.
- Moderately severe stenosis is associated with a right ventricular fourth sound and a prominent jugular venous a wave.
- Features of severe pulmonary stenosis:
- Loud harsh murmur,
- Inaudible Pulmonary Closure Sound (P2)
- Increased Right Ventricular Heave
- Prominent A Waves In The Jugular Pulse
- Right Ventricular Hypertrophy In Ecg
- Chest X-ray shows poststenotic dilatation in the pulmonary artery.
Pulmonary Stenosis Investigations:
- ECG: Shows both right ventricular and right atrial hypertrophy
- Chest X-ray: Shows a prominent pulmonary artery (extending into the left pulmonary artery branch) owing to poststenotic dilatation
- Doppler echocardiography is the definitive investigation.
- Cardiac catheterization
Pulmonary Stenosis Treatment:
- Mild and even moderate pulmonary valve stenosis and regurgitation do not result in RV overload and may require no specific treatment other than prophylaxis for infective endocarditis.
- Moderately severe and severe pulmonary valve stenosis (resting gradient >50 mm Hg with a normal cardiac output) is currently treated by percutaneous balloon valvotomy or, if this is not available, by surgical valvotomy. Postoperatively pulmonary regurgitation may develop which is benign in nature.
Pulmonary Stenosis Prognosis:
Long-term prognosis is very good.
Infective Endocarditis:
Question 95. Discuss the etiology, pathology, clinical features, complications, investigations, management/treatment of subacute bacterial endocarditis (SBE).
Answer:
Infective endocarditis (IE) is an infection of the endocardium of heart valves or the mural endocardium.
Infective Endocarditis Defiition:
Infective endocarditis is defined as an endovascular infection of cardiovascular structures by microbes. These structures include heart valves, atrial and ventricular endocardium, large intrathoracic vessels, and intracardiac foreign bodies (e.g., prosthetic valves, pacemaker leads, and surgical conduits).
The causative organism may be bacterium, fungus, chlamydia, or rickettsia. Majority of cases are due to infection by streptococci and staphylococci.
Consequences of infective endocarditis: Valvular dysfunction, localized or generalized sepsis, and source for embolism.
Infective Endocarditis Classifiation:
- According to the clinical course: Acute, subacute, and chronic
- According to the causative organism: Bacterial, viral, rickettsial, or fungal
- According to the nature of valve or device: Native or prosthetic
- According to the side affected: Left-sided or right-sided infective endocarditis
- According to the side affected and nature of valve/device:
- Left-sided infective endocarditis
- Left-Sided Prosthetic Infective Endocarditis
- Right-Sided Infective Endocarditis
- Device-related (permanent pacemaker, implantablem cardioverter defibrillator) infective endocarditis.
Differences between acute endocarditis and subacute endocarditis:
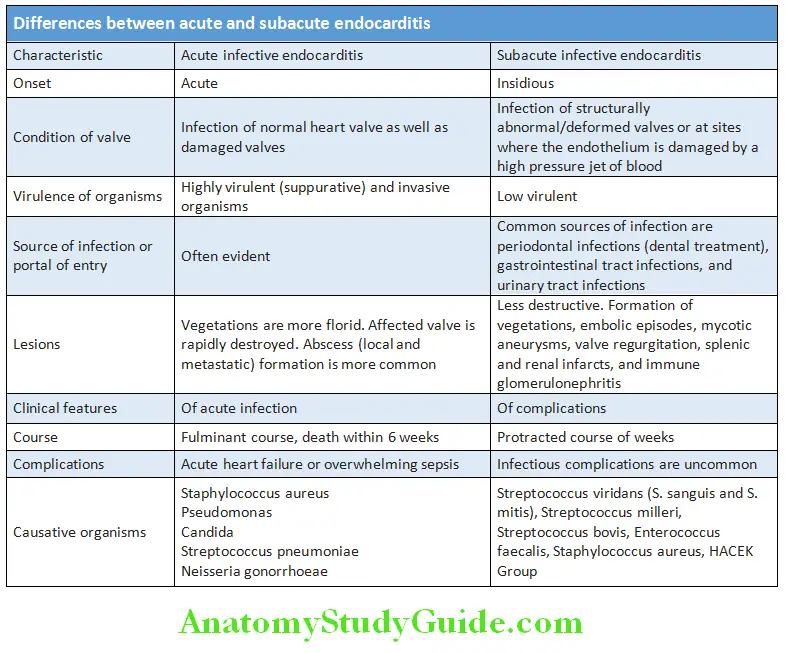
Predisposing Factors:
- Underlying heart disease:
- About 72% of patients have a pre-existing structural cardiac abnormality.
- Congenital isolated aortic valvular stenosis is most often associated with infective endocarditis (IE). Other congenital heart diseases include ventricular septal defect (VSD), tetralogy of Fallot (TOF), idiopathic subaortic stenosis, and atrial septal defect (uncommon).
- IE commonly involves mitral valve followed by aortic valve and uncommonly tricuspid valve (1%).
- Major risks for IE are cardiac prosthetic valves and parenteral narcotic drug abuse.
- Impaired host defense mechanism: It may occur in diabetes mellitus, malignancies (e.g., leukemias and lymphomas), cytotoxic therapy, and neutropenia.
- Conditions with bacteremia: Most important predisposing factors to the development of endocarditis are conditions that lead to bacteremia. Transient bacteremia from any cause may lead to infective endocarditis and include the following surgical procedures from different sources:
- Respiratory tract: Tonsillectomy and/or adenoidectomy, surgical operations (incision or biopsy) involving respiratory mucosa, and any invasive procedure of the respiratory tract used to treat an established infection (e.g., drainage of an abscess or empyema)
- Oral cavity: All dental procedures in which there is handling of gingival tissue or the periapical region of teeth or perforation of the oral mucosa (e.g., dental extractions, suture removal, placement of orthodontic band, root canal treatment).
- Gastrointestinal tract: Procedures done in an established GIT infection
- Genitourinary tract: Procedures performed in an established infection (e.g., cystoscopy during known enterococcal urinary tract infection—UTI)
- Skin and musculoskeletal system: Procedures involving infected tissue.
Common Organisms for Infective Endocarditis:
Question 96. Write a short essay/note on common causative agents for infective endocarditis.
Answer:
More than 75% of infective endocarditis is caused by streptococci or staphylococci.
Postoperative Endocarditis or Prosthetic Valve Endocarditis:
- It develops after cardiac surgery and may affect native or prosthetic valves and other prosthetic materials.
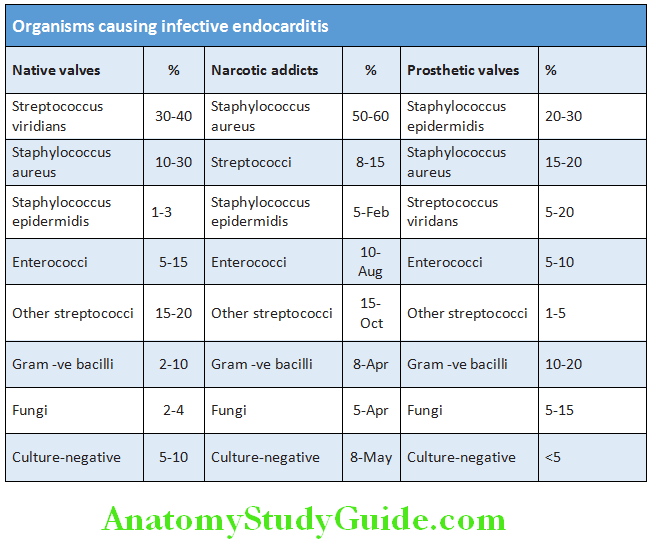
- Depending on the time of development of endocarditis following surgery, it can be divided into early, intermediate, and late endocarditis.
- Early (within 60 days of surgery) endocarditis is caused by intraoperative or hospital-acquired infections. The most common organism is a coagulase-negative Staphylococcus (S. epidermidis) which is a normal commensal in the skin and followed by fungi (Candida, Aspergillus).
- Late (after 1 year) endocarditis is caused by infection with community-acquired organisms. The causative organism is the same with those causing acute and subacute endocarditis of native valve.
- Endocarditis developing between 60 days and 1 year is due to a mixture of hospital-acquired episodes caused by less virulent organisms community-acquired episodes. The organisms include Streptococcus viridans and Enterococci.
- Types of prosthetic valves:
- Aortic prosthetic valve is more prone to endocarditis than mitral prosthetic valve.
- During first 3 months after surgery mechanical heart valves develop endocarditis more frequently than bioprosthetic ones. However, later on the rates of infection for the two valve types are same and are comparable at 5 years.
Right-sided endocarditis: Develops in intravenous drug users and are mainly caused by organisms found on the skin (e.g., Staphylococcus aureus, Candida). It usually presents with acute endocarditis and affects mainly tricuspid valve.
Culture-negative Endocarditis:
- It is defined as endocarditis without etiology following inoculation of at least three independent blood samples in a standard blood culture system with negative cultures after 7 days of incubation and subculturing.
- They constitute 5–10% of cases of endocarditis.
Causes of culture negative endocarditis:
- It is usually due to prior antibiotic therapy
- Inadequate quantity of blood taken for culture
- Anaerobic infection
- Some may be due to a variety of fastidious organisms that does not grow in normal blood cultures. These include Coxiella burnetii (cause of Q fever), Chlamydia species, Bartonella species (cause trench fever and cat scratch disease), and Legionella
- Right-sided endocarditis
- Noninfective endocarditis
Pathogenesis:
Mechanism by which virulent organisms infect apparently normal valves is poorly understood.
The probable sequence of events, which occur with the infection of a damaged valve by less-virulent organisms is as follows:
- Endocardial damage/injury and denudation
- Formation of sterile thrombus
- Adherence of the microorganisms: Transient bacteremia → microorganisms gain access to the circulation and adhere and gets deposited to the sterile vegetations (infection of thrombi)
- Proliferation of microorganisms within vegetations
- Formation of emboli: The vegetation may get detached and form infective emboli → cause spread of infection to visceral organs such as kidney, spleen, and brain. They may result in infarction or abscess. Septic emboli cause arteritis with weakness of arterial wall leading to mycotic aneurysms.
- Deposition of immune complexes: Antigen and antibody may form immune complexes and can produce focal glomerulonephritis and microscopic hematuria, diffuse glomerulonephritis, vasculitis of cerebral vessels leading to cerebrovascular accidents, and perisplenitis.
Clinical Features:
Question 97. Write a short essay/note on important signs of infective endocarditis.
Answer:
- The clinical features of infective endocarditis depend on the causative organism and the presence of predisposing cardiac conditions.
- Endocarditis may be acute or a more insidious “subacute” form. However, there is overlap between the two because the clinical pattern depends not only on the causative organism, but also depends on the site of infection, prior antibiotic therapy, and the presence of a valve or shunt prosthesis.
Subacute Endocarditis (SBE):
Clinical suspicion of subacute endocarditis is necessary when a patient with congenital or valvular heart disease develops certain features. These include:
Evidence of infection:
- Vague symptoms such as unusual tiredness, fatigue, lassitude, loss of appetite, loss of weight
- Persistent or intermittent low-grade or high-grade fever (84%) with night sweats, chills, and rigors
- Clubbing in fingers is a late sign and is found only in 10–20% of patients.
- Splenomegaly: Spleen is frequently palpable and the liver may also be enlarged.
- Brownish pigmentation of face and limbs
Evidence of new valve lesion (regurgitant)/murmurs or change of murmur:
- New murmur: Particularly, a diastolic murmur is the diagnostic feature. It is detected in about 15% of patients during initial period. However, most patients develop a murmur during the course of the disease.
- Changing murmurs: Apart from integrity of valves, factors such as change in cardiac output, temperature, and hematocrit may be responsible for change of murmurs. However, new regurgitant murmur developing in acute sepsis is almost diagnostic of endocarditis.
Evidence of embolism:
- Cutaneous manifestations: These include the following:
- Purpura and petechiae (20–40%), subconjunctival splinter, or subungual splinter hemorrhages due to microthromboemboli
- Janeway lesions: Erythematous, nontender nodules, on palms or soles
- Ocular manifestations: Roth spot are flame-shaped hemorrhages which may occasionally appear like cotton wool spots.
- Septic infarcts/abscess: Spleen (painful splenomegaly), kidney (loin pain, hematuria, and renal failure), brain (convulsions, hemiplegia, aphasia, loss of vision, and cerebellar disturbances), peripheral arteries (claudication, absence of peripheral pulses, and gangrene), or lungs (pulmonary infarction, pleurisy, and pleural effusion due to right-sided endocarditis)
- Mycotic aneurysms
Evidence of immunological phenomena:
- Osler nodes: Tender, small, painful, swollen, purplish/erythematous subcutaneous papules/nodules in pulp of distal fingers due to hypersensitive angiitis. Cultures are negative and persist for hours to several days.
- Roth spots are circular retinal hemorrhages with white or pale centers spots composed of fibrin.
- Focal segmental glomerulonephritis: Develop due to deposition of antigen-antibody complexes in glomeruli. Grossly, the outer surface of kidney develops a flea-bitten appearance due to patchy hemorrhagic foci involving the glomeruli. Patents develop microscopic hematuria.
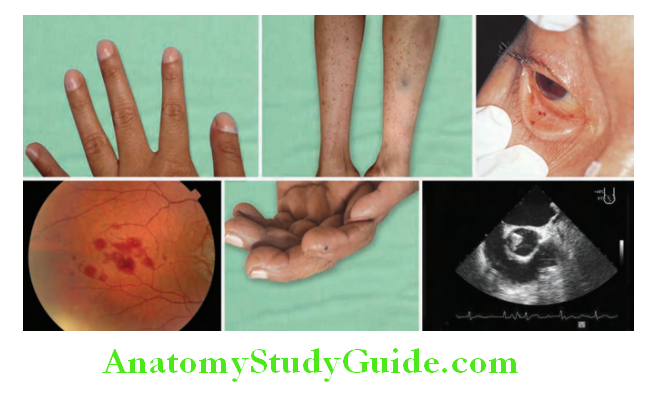
Investigations:
Aim of investigations:
- To confirm the diagnosis of infective endocarditis
- To identify the causative organism for appropriate therapy
- To assess the patient’s response to therapy.
Question 98. Write a short essay/note on tests for infective endocarditis.
Answer:
Microbiological Investigations:
- Blood cultures are the crucial investigations in infective endocarditis
- To identify the causative microorganism
- Guide antibiotic therapy. At least three sets of blood samples for cultures should be taken from different venipuncture sites over 24 hours before commencing therapy. Aseptic technique should be followed and the risk of contaminants should be minimized. Blood samples for cultures should not be obtained from an in-dwelling line. Both aerobic and anaerobic cultures are needed.
- Serological tests are useful if there is suspicion of diagnosis and the cultures are negative. Culture-negative cases may be due to organisms which will not grow in blood cultures such as Coxiella, Bartonella, Legionella, and Chlamydia.
- Electrocardiogram: ECG will help to detect complications such as conduction abnormalities (AV block) and occasionally myocardial infarction due to emboli. PR prolongation/heart block is associated with aortic root abscess.
- Chest X-ray: It may show evidence of cardiac failure, cardiomegaly, and pulmonary edema in left-sided disease, pulmonary emboli/abscess in right-sided disease.
- Echocardiography: It plays a key role to identify the presence and size of vegetations, assess valve damage, abscess formation, detect intracardiac complications, and assess cardiac function. However, if vegetations are not detected, the diagnosis of endocarditis cannot be excluded.
- Transthoracic echocardiography (TTE): It is a first-line noninvasive imaging test with sensitivity of 60–75% and high specificity for demonstrating vegetations, valvular dysfunction, ventricular function, and abscesses. Vegetations a small as 2–4 mm can be detected.
- Transesophageal echocardiography (TOE): It is a second-line invasive imaging test with greater sensitivity (>90%) and specificity than TTE. It can detect vegetations even smaller ones (1–1.5 mm), aortic root abscess, and prosthetic valve endocarditis.
- Complete blood counts: May show normocytic normochromic anemia (reduced hemoglobin) and increased WBC counts and increased or reduced platelets
- Urea and creatinine: May be increased due to glomerulonephritis
- Liver biochemistry: Serum alkaline phosphatase may be increased.
- Inflammatory markers: Increased erythrocyte sedimentation rate (ESR) and C-reactive protein are observed. CRP also helps in monitoring response to therapy; it is reduced in response to therapy and increased with relapse.
- Urine: Proteinuria and hematuria occur frequently.
Modifid Duke Criteria for the Diagnosis of Infective Endocarditis:
- Diagnosis by these guidelines requires either pathologic or clinical criteria
- If clinical criteria are used:
- Definite endocarditis, two major, or one major + three minor, or five minor criteria are required for diagnosis
- Possible endocarditis = One major and one minor, or three minor criteria
Complications:
Question 99. Write a short essay/note on complications of infective endocarditis.
Answer:
- Cardiac complications: These are due to direct valvular damage and consequences of local invasion. The infection may spread locally from valve into the valve ring, adjacent mural endocardium, or chordae tendineae.
- Ring abscess: Vegetations may erode the underlying myocardium and produce an abscess which is known as ring abscess.
- Perforation and rupture: May involve valve leaflets, aorta, or interventricular septa (depending on the site of infection)
- Myocardial abscess usually due to S. aureus
- Suppurative pericarditis
- Valvular dysfunction and heart failure: Stenosis or insufficiency. Heart failure is most common and is due to valvular regurgitation.
- Prosthetic dehiscence
- Valvular distortion/destruction chordal rupture
- Conduction abnormalities
- Purulent pericarditis

- Renal:
- At least in four forms:
- Prerenal Due To Low Cardiac Output,
- Microabscess Formation Secondary To Septic Emboli
- Glomerular Dysfunction As A Result Of Circulating Immune Complexes
- Renal failure as a result of antibiotics.
- Embolic events (discussed under evidence of embolism—subacute endocarditis)
- Vegetations are usually friable and likely to be break, detach, and cause embolism
- Embolic complications (due to septic emboli): Emboli contain large numbers of virulent organisms → abscesses develop at the sites of arrest of the emboli. Septic emboli from the left side of the heart: They enter systemic circulation and its consequences are:
- Septic infarcts (e.g., spleen, kidney, or brain)
- Mycotic aneurysms
- Small emboli (microthromboemboli) may produce:
- Splinter or subungual hemorrhages
- Janeway lesions
- Septic emboli from the right side of the heart: They enter pulmonary circulation → lead to pulmonary abscess.
- Immunological phenomena:
- Focal segmental glomerulonephritis: Develop due to deposition of antigen–antibody complexes in glomeruli. Grossly, the outer surface of kidney develops a flea-bitten appearance due to patchy hemorrhagic foci involving the glomeruli.
- Osler nodes: They are small, tender subcutaneous nodules in the pulp of the digits and persist for hours to several days.
- Roth spots are retinal hemorrhages with white or pale centers composed of fibrin.
Question 100. Write a short essay/note on management of infective endocarditis/subacute infective endocarditis.
Answer:
Subacute infective endocarditis Management:
Medical treatment:
- Blood should be collected for cultures before starting the empirical antibiotic therapy. However, this should not delay therapy in unstable patients.
- I f source of infection is identified, it should be removed as soon as possible (e.g., tooth with an apical abscess should be extracted).
- Empirical treatment regimen: Penicillins are fundamental to the therapy of bacterial endocarditis. Empirical treatment regimen depends on the mode of presentation, the suspected organism, and whether the patient has a prosthetic valve or penicillin allergy.

Subacute infective endocarditis Prognosis: It is fatal in about 20% patients and higher in those with prosthetic valve endocarditis and those infected with antibiotic-resistant organisms.
Surgical treatment:
Decisions to carry out surgical intervention in patients with infective endocarditis should take into account the patient specific features such as age, noncardiac morbidities, presence of prosthetic material or cardiac failure, and infective endocarditis features such as causative organism, size of vegetation, presence of perivalvular infection, and systemic embolization.
Cardiac surgery consists of debridement of infected material and valve replacement. Antimicrobial therapy should be starte before surgery.
Indications for surgical treatment in infective endocarditis:
- Endocarditis of prosthetic valve
- Large vegetations
- Left-sided large vegetation (10 mm) with an episode of embolization
- Very large (15 mm) and mobile vegetation (high-risk of embolism)
- Progressive cardiac failure due to valvular damage
- Active infection persisting, i.e., fever and evidence of bacteremia for >7–10 days in spite of adequate antibiotic treatment
- Abscess formation, perivalvular involvement, S. aureus, and fungal endocarditis
Bacterial Endocarditis Prophylaxis:
Question 101. Write a short essay/note on antibiotic prophylaxis against infective endocarditis during dental procedure/during urogenital surgery.
(or)
Write a short essay/note on prophylaxis of infective endocarditis.
Answer:
Prophylaxis is not needed for routine local anesthetic injections through noninfected tissue, placement of removable prosthodontic or orthodontic appliances, shedding of deciduous teeth, vaginal delivery, and hysterectomy or tattooing, bronchoscopy, laryngoscopy, endotracheal intubation, cystoscopy, colonoscopy, or skin suturing.
Indications:
Question 102. Write a short note on infective endocarditis/subacute infective endocarditis (SBE) prophylaxis.
Answer:
- Procedures: Endocarditis prophylaxis is advised in patient at high or moderate risk for endocarditis.
- These include following procedures:
- Dental procedures: Extraction, periodontal procedures, implant placement, root canal instrumentation, and intraligamentary injections (anesthetic)
- Respiratory procedure: Bronchoscopy with rigid bronchoscope operations involving the mucosa
- Gastrointestinal procedures: Sclerotherapy of esophageal varices, stricture dilation, ERCP, biliary tract surgery, and surgery involving mucosa
- Genitourinary procedures: Urethral dilation, prostate or urethral surgery, or cystoscopy
Infective endocarditis prophylaxis:
High-risk group:
- Prosthetic heart valves
- Prior bacterial endocarditis
- Complex cyanotic congenital heart disease
- Surgically constructed systemic-pulmonary shunts
- Repaired shunts within 6 months
- Valvulopathy after cardiac transplantation
Moderate-risk group:
- Congenital cardiac malformations (other than those listed in other two groups)
- Acquired valvular dysfunction (e.g., rheumatic heart disease)
- Hypertrophic cardiomyopathy
- Mitral valve prolapse with valvular regurgitation, thickened leaflets, or both
Negligible-risk group:
- Isolated secundum atrial septum defect
- Surgical repair of atrial septal defect, ventricular septal defect, or patent ductus arteriosus
- Previous coronary artery bypass graft surgery
- Mitral valve prolapse without valvular regurgitation
- Physiologic, functional, or innocent heart murmurs
- Previous Kawasaki syndrome without valvular dysfunction
- Previous rheumatic fever without valvular dysfunction
- Cardiac pacemakers (intravascular and epicardial) and implanted defibrillators
Antibiotic Regiment for Prophylaxis of Endocarditis in Adults at Moderate- or High-risk:
- Oral cavity, respiratory tract, or esophageal procedures
- Standard regimen: Amoxicillin 2.0 g PO 1 hour before procedure
- Inability to take oral medication: Ampicillin 2.0 g IV or IM within 30 minutes of procedure
- Penicillin allergy:
- Clarithromycin 500 mg PO 1 hour before procedure
- Cephalexin or cefadroxil 2.0 g PO 1 hour before procedure
- Clindamycin 600 mg PO 1 hour before procedure or IV 30 minutes before procedure
- Inability to take oral medication: Cefazolin 1.0 g IV or IM 30 minutes before procedure
Genitourinary and Gastrointestinal Tract Procedure:
- High-risk patients: Ampicillin 2.0 g IV or IM plus gentamicin 1.5 mg/kg IV or IM within 30 minutes of procedure. Repeat ampicillin, 1.0 g IV or IM or amoxicillin 1.0 g PO 6 hours later
- High-risk penicillin-allergic patients: Vancomycin 1.0 g IV or 1M over 1–2 hours plus gentamicin 1.5 m/kg IV or IM within 30 minutes before procedure, no second dose recommended
- Moderate-risk patients: Amoxicillin 2.0 g PO 1 hour before procedure or ampicillin 2.0 g IV or IM 30 minutes before procedure
- Moderate-risk, penicillin-allergic patients: Vancomycin 1.0 g IV inferred over 1–2 hours and completed within 30 minutes of procedures.
Culture-Negative Infective Endocarditis:
- Culture-negative infective endocarditis (IE) is defined as endocarditis without etiology following inoculation of three blood samples in a standard blood culture system (e.g., negative cultures after 7 days).
- Reasons for culture-negative in infective endocarditis:
- Administration of antimicrobial agents prior to blood culture incubation
- Inadequate microbiological techniques
- Infection with highly fastidious bacteria or nonbacterial pathogens (e.g., fungi)
Noninfective Endocarditis:
Question 103. Write a short essay/note on noninfective endocarditis.
Answer:
It is characterized by the formation of sterile platelet and fibrin thrombi on cardiac valves and adjacent endocardium.
- It develops in response to trauma, circulating immune complexes, vasculitis, or a hypercoagulable state
- Symptoms are caused due to systemic arterial embolism.
- Diagnosis is by echocardiography and negative blood cultures.
Marantic Endocarditis (Nonbacterial Thrombotic Endocarditis—NBTE):
Noninfective Endocarditis Etiology:
- NBTE is often encountered in a number of conditions. These include debilitated patients with cancer or sepsis, hence previously termed marantic endocarditis (root word marasmus, relating to malnutrition). It frequently develops concomitantly with deep venous thrombosis, pulmonary emboli, or underlying systemic hypercoagulable state and advanced malignancy. There is a striking association with mucinous adenocarcinomas, potentially relating to the procoagulant effects of tumor-derived mucin or tissue.
- Other less common causes include systemic lupus erythematosus, antiphospholipid syndrome, rheumatic heart disease, rheumatoid arthritis, and burns.
Vegetations are small, bland, and usually attached to the line of closure of valve leaflets.
Noninfective Endocarditis Treatment: Consists of systemic anticoagulant therapy and treatment of the underlying malignancy or associated condition.
Libman–Sacks Endocarditis:
- Libman–Sacks endocarditis (otherwise known as verrucous endocarditis) is characteristic cardiac manifestation of the autoimmune disease such as systemic lupus erythematosus.
- Pathogenesis: It produced due to circulating immune complexes.
- Endocarditis: It is characterized by small- or medium-sized vegetations on either side of the valve leaflets. They appear mulberry-like clusters of verrucae and consist of accumulations of immune complexes and mononuclear cells.
Heart Failure:
Question 104. Define heart failure. Discuss the types, common cause, pathophysiology, clinical features, and management of heart failure.
(or)
Describe the etiology, clinical features, and management of chronic heart failure. Mention the factors which will acutely decompensate a patient with chronic stable heart failure.
(or)
Discuss the symptoms and signs of heart failure. Outline the rational management.
(or)
Describe the etiology, classification, clinical features, diagnosis, and management of congestive cardiac failure (heart failure).
(or)
Outline the clinical features, investigations, and treatment of congestive cardiac failure with hypertensive heart disease.
Answer:
Heart Failure Defiition:
Heart failure or cardiac failure is the pathophysiological process in which the heart as a pump is unable to meet the metabolic requirements of the tissue for oxygen and substrates despite the venous return to heart is either normal or increased.
- It is a complex syndrome that can result from any structural or functional cardiac disorder. In heart failure, heart is unable to pump blood at a rate of sufficient to meet the metabolic demands of the tissues or can do so only at an elevated filling pressure.
- Older term congestive heart failure (CHF) should be avoided because not all patients with heart failure have volume overload. Hence, the term heart failure is preferred.
- Heart failure is the common end stage of many forms of chronic heart disease and most common reason for hospitalization in adults >65 years of age.
- Heart failure usually develops insidiously from the cumulative effects of chronic work overload (e.g., in valve disease or hypertension) or ischemic heart disease (e.g., following myocardial infarction with heart damage). However, acute hemodynamic stresses, such as fluid overload, sudden valvular dysfunction, or myocardial infarction, can cause sudden heart failure.
Pathophysiology:
Question 105. Write a short essay/note on pathophysiology of heart failure.
Answer:
Cardiac output is determined by preload (the volume and pressure of blood in the ventricles at the end of diastole), afterload (the volume and pressure of blood in the ventricles during systole), and myocardial contractility.
Myocardial contractility (inotropic state): It depends on the adrenergic nervous activity and the levels of circulating catecholamines.
Heart Failure:
- In the intact heart, myocardial failure causes reduction of the volume of blood ejected with each heartbeat and an increase in the volume of blood remaining after systole. This increased diastolic volume stretches the myocardial fibers and restores the myocardial contraction. However, the failing myocardium results in myocardial depression (of the ventricles).
- Heart failure is a progressive disorder that is initiated after an index event that either damages the heart muscle (with loss of functioning cardiac myocytes) or disturbs the myocardial ability to generate force (prevents the heart from contracting normally). The common feature of these events is a decrease in the pumping capacity of the heart. The compensatory mechanisms include tachycardia, increased myocardial contractility, and activation of neurohumoral systems.
If the heart failure progresses, the compensatory mechanisms are overwhelmed and become pathological. Thus, there is pathological peripheral vasoconstriction and sodium retention. Heart failure is the common end stage of many forms of chronic heart disease.
Heart failure usually develops insidiously from the cumulative effects of chronic work overload (e.g., in valve disease or hypertension) or ischemic heart disease (e.g., following myocardial infarction with heart damage). However, acute hemodynamic stresses, such as fluid overload, sudden valvular dysfunction, or myocardial infarction, can cause sudden heart failure.
Question 106. Write a short note on ventricular remodeling/cardiac remodeling.
Answer:
Ventricular remodeling/cardiac remodeling: Refers to the changes in size, shape, structure, and physiology of the heart after injury to the myocardium.
- Left ventricular remodeling is the process by which mechanical, neurohormonal, and possibly genetic factors alter ventricular size, shape, and function.
- Remodeling occurs in several clinical conditions, including myocardial infarction, hypertension, cardiomyopathy, and valvular heart disease.
- It is characterized by hypertrophy, loss of myocytes, and increased interstitial fibrosis.
Mechanism of heart failure is depicted in Flowchart:
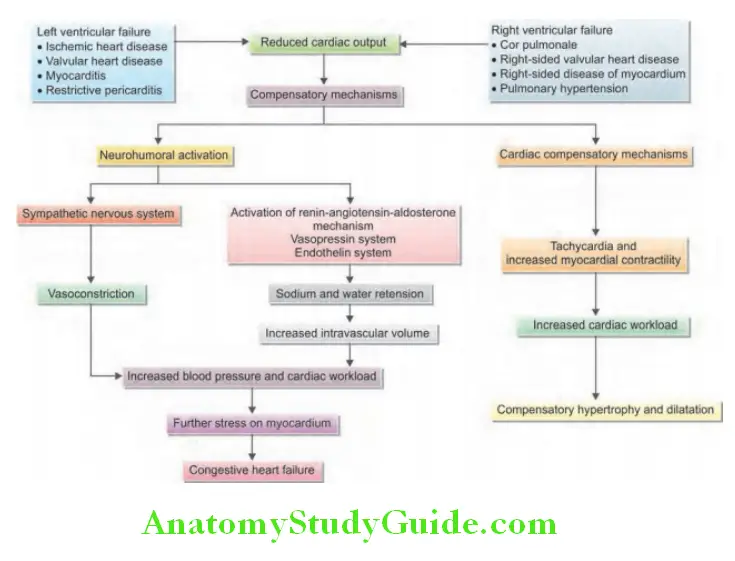
Types of Heart Failure:
Question 107. Write a short note on types of heart failure.
Answer:
Depending on Output:
- Low-output heart failure:
- Systolic heart failure: Characterized by decreased cardiac output and decreased left ventricular ejection fraction
- Diastolic heart failure: Characterized by elevated left and right ventricular end-diastolic pressures and may have normal left ventricular ejection fraction
- High-output heart failure: Characterized by failure of the heart to maintain sufficient circulation despite an increased cardiac output (defined as cardiac output >8 L/minute or a cardiac index >3.9 L/minute/m2). Examples include cardiac failure associated with hyperthyroidism, anemia, pregnancy, arteriovenous fistulae, beriberi, and Paget’s disease.
Diastolic and Systolic Dysfunction:
- Systolic dysfunction: Systolic heart failure is characterized by an abnormality of ventricular contraction. The ejection fraction is usually below 40%. Causes include coronary artery disease, hypertension, and valvular heart disease.
- Diastolic dysfunction: Characterized by an impaired ventricular relaxation and increased ventricular stiffness resulting in reduced filling (diastolic dysfunction). Causes include hypertension, coronary artery disease, hypertrophic obstructive cardiomyopathy (HOCM), and restrictive cardiomyopathy.
Acute and Chronic Heart Failure:
- Acute heart failure is characterized by sudden development of heart failure. This suddenly reduces cardiac output and systemic hypotension without peripheral edema. Examples include acute myocardial infarction and rupture of a cardiac valve. Acute left heart failure may develop either de novo or as an acute decompensated episode, on a background of chronic heart failure (acute-on chronic heart failure).
- Chronic heart failure is characterized by gradual development of heart failure and systemic arterial pressure is well maintained, but edema develops. Examples include dilated cardiomyopathy and multivalvular disease.
- Compensated heart failure is the term used to describe the condition of those with impaired cardiac function, in whom adaptive/compensatory changes have prevented the development of overt heart failure. Severe overt or acute heart failure may be precipitated by minor insult such as an infection or development of atrial fibrillation.
Left-sided, Right-sided, and Biventricular Heart Failure:
The left side of the heart consists of the functional unit of the left atrium, left ventricle, and mitral and aortic valves. The right side of the heart consists of the right atrium, right ventricle, and tricuspid and pulmonary valves.
- Left-sided (left ventricular) heart failure is characterized by:
- Reduction in left ventricular output
- Increase in left atrial and pulmonary venous pressure: An acute increase in left atrial pressure produces pulmonary congestion or pulmonary edema (e.g., myocardial infarction). A more gradual increase in left atrial pressure leads to reflex pulmonary vasoconstriction, which prevents the development pulmonary edema (e.g., in mitral stenosis and aortic stenosis). This increases pulmonary vascular resistance and leads to pulmonary hypertension, which in turn can impair the function of right ventricle.
- Right-sided (right ventricular) heart failure is characterized by:
- Reduction in right ventricular output
- Increase in right atrial and systemic venous pressure
- Causes of isolated right heart failure, e.g., chronic lung disease (cor pulmonale), multiple pulmonary embolism, and pulmonary valvular stenosis
- Biventricular heart failure is characterized by failure of the ventricles of the left and the right heart.
- Dilated cardiomyopathy or ischemic heart disease, affects both ventricles
- Disease of the left heart leads to chronic elevation of the left atrial pressure, pulmonary hypertension, and leading to right heart failure.
Forward and Backward Heart Failure:
- Forward heart failure is characterized by decreased cardiac output, inadequate perfusion of organs that causes poor tissue perfusion. Reduced renal perfusion activates renin–angiotensin–aldosterone system resulting in excessive absorption of sodium by renal tubules.
- Backward heart failure is characterized by a normal cardiac output, severe salt and water retention, and venous congestion in the pulmonary and systemic circulation.
Question 108. Write a short note on causes of heart failure.
(or)
Write a short essay/note on factors that precipitate heart failure.
Answer:
In practice, the most common causes of heart failure are ischemic heart disease and hypertensive heart diseases.
Factors that may precipitate or aggravate heart failure in preexisting heart disease:
Risk factors for heart failure: Hypertension, diabetes mellitus, use of cardiotoxic substances (e.g., alcohol, tobacco, and cocaine), hyperlipidemia, and coronary artery disease
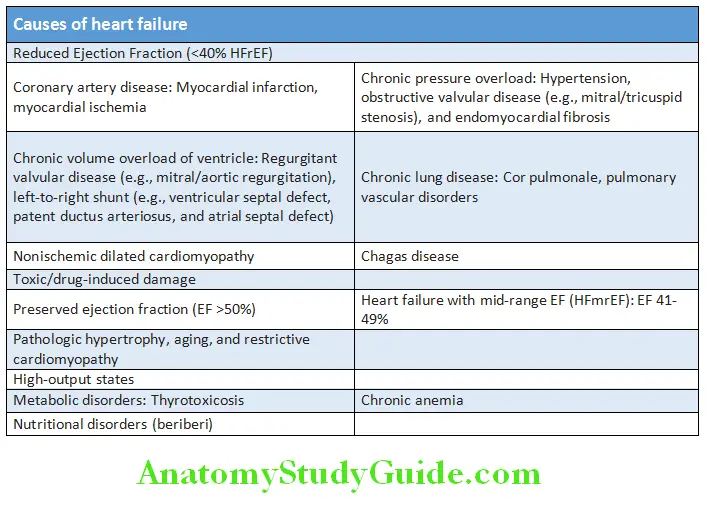
Precipitating factors for heart failure:
- Intercurrent illness, e.g., infection
- Supervening heart diseases, e.g., myocardial ischemia or infarction, myocarditis, and infective endocarditis
- Cardiac arrhythmia, e.g., atrial fibrillation
- Poor compliance with therapy
- Administration of a drug with negative inotropic (β-blocker), disopyramide, or fluid-retaining properties [nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids]
- Pulmonary embolism
- Conditions associated with increased metabolic demand, e.g., anemia pregnancy and thyrotoxicosis
- IV fluid overload, e.g., postoperative IV infusion
- Systemic hypertension
- Excess salt intake
- Physical and emotional stress
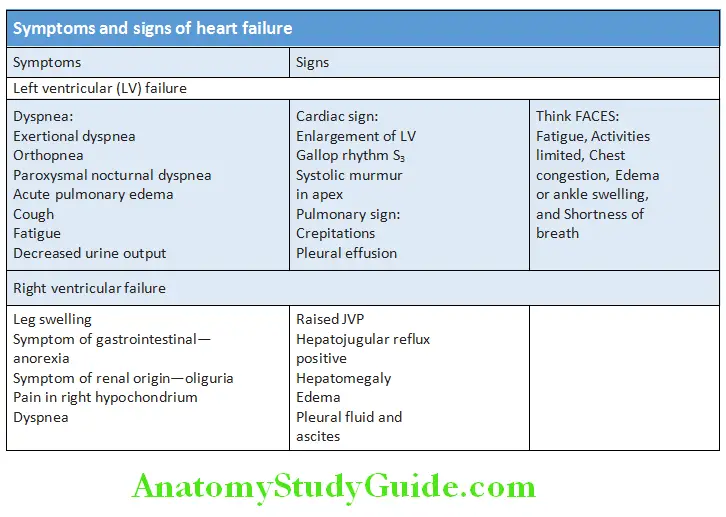
Clinical Manifestations of Heart Failure:
Question 109. Write a short essay/note on clinical features (symptoms and signs) of heart failure and acute left ventricular failure.
Answer:
Symptoms:
Dyspnea:
- Exertional dyspnea
- Orthopnea, bendopnea
- Paroxysmal nocturnal dyspnea (PND)
- Cardiac asthma is closely related to PND and nocturnal cough.
- It is characterized by wheezing (secondary to bronchospasm) which is most prominent at night. Acute pulmonary edema represents a severe form of cardiac asthma.
- Acute pulmonary edema
- Cheyne-Stokes respiration.
Nocturia:
It is observed during early heart failure. It is because renal perfusion and diuresis is better at night when the patient is supine. Cerebral symptoms includes confusion, difficulty in concentration, impaired memory, headache, insomnia, and anxiety. These symptoms may be produced due to arterial hypoxemia and reduced cerebral perfusion.
Nonspecific symptoms:
- Fatigue due to low cardiac output and weakness due to decreased perfusion of skeletal muscles
- Low-grade fever due to reduction of cutaneous blood flow
- Anorexia, nausea, and abdominal fullness/pain due to congestion of liver and portal venous system.
Signs:
Dependent/cardiac edema:
- Dependent/cardiac edema: Due to gravity, fluid accumulates over dependent parts of the body. In ambulant patients, it is found symmetrically in the both legs, particularly in the pretibial region and around the ankles. It is less in the morning and become more toward the evening. In bedridden patients, it is observed in the sacral region.
- Anasarca: In advanced heart failure, the fluid accumulating throughout the body and is severe termed as anasarca. Face and upper limbs are usually spared until the terminal stages.
Cyanosis:
It is usually observed in lips and nail beds. The extremities appear cold and pale due to reduced blood flow.
Pulse:
Pulse volume is reduced and pulsus alternans is a sign of severe heart failure.
Blood pressure:
- Reduced pulse pressure is due to reduced stroke volume.
- Occasionally, there may be mild elevation of diastolic blood pressure due to generalized vasoconstriction.
- Hypotension is prominent in acute heart failure.
Jugular venous pressure (JVP):
- JVP is raised because of elevated systemic venous pressure.
- JVP may not be raised at rest during early stages of heart failure. It may be demonstrated:
- During and immediately after exercise
- With sustained pressure on the abdomen (positive hepatojugular or abdominojugular reflux).
Third and fourth heart sound:
- Presence of a third heart sound in highly suggestive of heart failure
- Triple/quadruple/summation gallop is seen.
Respiratory system:
Percussion: Dull percussion notes over the bases of lungs (infrascapular, infra-axillary areas)
Auscultation: Inspiratory crepitations over the bases of lungs. If there is pulmonary edema, coarse crepitations are heard widely over both lung fields associated with expiratory rhonchi.
Liver (congestive hepatomegaly):
- Hypoglycemia can develop in long-standing cases due to depletion of glycogen stores in liver and increased production of lactic acid from glucose, induced by hypoxia.
- Jaundice: Hyperbilirubinemia of both conjugated and unconjugated type may be a late feature. In acute hepatic congestion, jaundice is severe and liver enzymes are markedly elevated.
Effsion:
- Pleural effusion develops more commonly on the right side and is due to raised pleural capillary pressure and transudation of fluid into pleural space.
- Ascites is due to transudation secondary to raised pressure in the hepatic vein, portal veins, and veins draining the peritoneum.
- Pericardial effusion: Rare.
Renal system:
- Poor renal perfusion leads to oliguria and prerenal azotemia (uremia). Blood urea is usually raised out of proportion to serum creatinine.
- Urine: Specific gravity of urine is high, mild proteinuria is seen, and urinary sodium is low.
Cardiac cachexia:
Chronic heart failure is sometimes associated with severe anorexia, marked weight loss (cardiac cachexia), impaired absorption due to gastrointestinal congestion, and skeletal muscle atrophy due to immobility.
Framingham Criteria for Diagnosis of Heart Failure:
The diagnosis of heart failure should not be made on history and clinical findings alone. It requires evidence of cardiac dysfunction (by investigation) and identification of the underlying cause of heart failure in all cases.

Criteria for diagnosis of heart failure one major + two minor:
Investigations in Heart Failure:
- Chest X-ray: It may reveal cardiomegaly. Other findings include phantom tumor (fluid in horizontal or oblique fissures of lungs which disappears after treatment with diuretics), bat’s wing appearance [hazy opacification spreading from the hilar regions on both sides, and pleural effusion (bilateral or unilateral).
- Electrocardiography: It may reveal previous MI, active ischemia, ventricular hypertrophy (e.g., due to hypertension), atria abnormality, arrhythmias, and conduction abnormalities (e.g., arrhythmia).
- Echocardiography is very useful to:
- Determine the etiology
- Detect any unsuspected valvular heart disease (e.g., occult mitral stenosis)
- identify patients who will benefit from long-term drug therapy (e.g., ACE inhibitors)
- Assess cardiac chamber dimension (size and shape), ejection fraction, valvular functions, cardiomyopathies, and regional wall motion abnormalities
- Differentiate between systolic and diastolic heart failure.
- Stress echocardiography: It helps in assessing the viability in dysfunctional myocardium. Dobutamine stress identifies contractile reserve in stunned or hibernating myocardium.
- Nuclear cardiology: Radionucleotide angiography (RNA) is useful for quantify ventricular ejection fraction, single photon emission computed tomography (SPECT), or positron emission tomography (PET) can reveal myocardial ischemia and viability in dysfunctional myocardium.
- CMR (cardiac MRI): It helps to assess the viability in dysfunctional myocardium with the use of dobutamine for contractile reserve or with gadolinium for delayed enhancement (“infarct imaging”).
- Cardiac catheterization: It is useful in the diagnosis of ischemic heart failure (and suitability for revascularization), measurement of pulmonary artery pressure, left atrial (wedge) pressure, and left ventricular end-diastolic pressure.
- Cardiac biopsy: It is useful in the diagnosis of cardiomyopathies (e.g., amyloid) and follow-up of cardiac transplanted patients to assess rejection.
- Cardiopulmonary exercise testing: Peak oxygen consumption (VO) is useful in predicting hospital admission and death in heart failure.
- Ambulatory 24-hour ECG monitoring (Holter): May be necessary in patients with suspected arrhythmia. It may be useful in severe heart failure or inherited cardiomyopathy to decide the necessity of defibrillator.
- Brain natriuretic peptide (BNP) or N-terminal portion of proBNF (NPproBNP) is elevated in heart failure and highly sensitive for the diagnosis of its diagnosis. It is a marker of risk (>100 pg/mL) and is useful in the investigation of patients with breathlessness or peripheral edema (to differentiate cardiac from respiratory cause of acute dyspnea).
- Blood tests: Full blood count, liver function tests, serum urea, creatinine and electrolytes, cardiac enzymes in acute heart failure, thyroid function may help to establish the nature and severity of the underlying heart disease and detect any complications.
- Invasive hemodynamic monitoring: Useful in selected patients with acute heart failure who have persistent symptoms is spite of empiric standard therapies.
Complications in Advanced Heart Failure:
- Renal failure: Cardiorenal syndrome (poor renal perfusion caused by low cardiac output) may be worsened by therapy [e.g., diuretics, angiotensin-converting enzyme (ACE) inhibitors, and angiotensin receptor blockers].
- Hypokalemia: Due to the result of treatment with potassium-losing diuretics or hyperaldosteronism produced by activation of the renin–angiotensin system and impaired aldosterone metabolism due to congestion of liver.
- Hyperkalemia: Due to the effects of drugs which promote renal resorption of potassium (e.g., combination of ACE inhibitors or angiotensin receptor blockers, and mineralocorticoid receptor antagonists).
- Hyponatremia: May develop in severe heart failure and is a poor prognostic sign. It may be due to diuretics, inappropriate retention of water (due to high ADH secretion), or failure of the cell membrane ion pump.
- Hepatic dysfunction: Due to hepatic venous congestion and poor arterial perfusion.
- Thromboembolism: Deep vein thrombosis and pulmonary embolism may develop due to the effects of a low cardiac output and immobility. Systemic emboli may develop in patients with atrial fibrillation or flutter, or with intracardiac thrombus complicating conditions such as mitral stenosis, MI, or left ventricular aneurysm.
- Atrial and ventricular arrhythmias: Very common and include atrial fibrillation (20%), sudden death (50%) due to a ventricular arrhythmia, ventricular ectopic beats, and nonsustained ventricular tachycardia. They may be due to electrolyte changes (e.g., hypokalemia and hypomagnesemia), the underlying heart disease, and the proarrhythmic effects of sympathetic activation.
Question 110. Write a short essay on management of congestive heart failure.
Answer:
Management of Heart Failure:
Aim of treatment:
- Relief of symptoms
- Prevent and control of disease causing cardiac dysfunction and heart failure
- Retard disease progression
- Improve quality and length of life.
1. General Lifestyle Advice/Measures:
- Education of patients and their relatives: Explanation of nature of disease, about the causes and treatment of heart failure.
- Measures to prevent heart failure: Cessation of smoking and illicit drugs, control of hypertension, diabetes and hypercholesterolemia, and pharmacological treatment following myocardial infarction. Identify and treat any factor that aggravates the heart failure.
- Treatment of the underlying cause of heart failure (e.g., coronary artery disease): Wherever possible to prevent progression to heart failure
- Dietary modifications: Good general nutrition and maintain desired weight and body mass index. Avoid large meals, foods rich in salt, or added salt. Diet low in fat, rich in fruit and vegetables, and increase fiber. Fluid restriction (limited to 1.5 L) is needed only when heart failure is severe. Alcohol has a negative inotropic effect and should be avoided. Omega-3 polyunsaturated fatty acids reduce mortality and admission.
- Physical activity, exercise, and emotional rest:
- Physical activity and exercise: Regular low level endurance exercise (e.g., 20–30 minutes walking three or five times/ week at 70–80% of peak heart rate) reverses “deconditioning” of peripheral muscle metabolism and is advisable in patients with compensated heart failure. Avoid strenuous isometric activity.
- Bed rest: Reduces the demands on the heart. Bed rest for a few days is for patients with exacerbations of congestive cardiac failure. However, prolonged bed rest may predispose to deep vein thrombosis. This can be avoided by daily leg exercises, low-dose subcutaneous heparin, and elastic support stockings.
2. Drug Therapy/Management:
Function of heart can be improved by increasing contractility, reducing preload or afterload. Drugs that reduce preload are used in patients with high end-diastolic filling pressures and pulmonary or systemic venous congestion. Drugs that reduce afterload or increase myocardial contractility are used in patients with signs and symptoms of a low cardiac output.
- Diuretic therapy:
- In heart failure, diuretics act by increasing the urinary excretion of salt (sodium) and water leading to reduction in blood and plasma volume. Diuretics reduce preload and improves pulmonary and systemic venous congestion. They may also reduce afterload and ventricular volume and increase cardiac efficiency.
- Loop diuretics (e.g., furosemide 20–40 mg once or twice) and thiazide diuretics (e.g., hydrochlorothiazide
25 mg once or twice, or metolazone 2.5–5 mg OD) are given to patients with fluid overload. - In severe heart failure, the combination of a loop and thiazide diuretic may be needed. Regular monitoring of serum electrolytes and renal function is necessary because of risk of hypokalemia and hypomagnesemia.
- Mineralocorticoid receptor antagonists, such as spironolactone (12.5–25 once or twice), are potassium-sparing diuretics and are beneficial in patients having heart failure with severe left ventricular systolic dysfunction.
- Angiotensin-converting enzyme inhibitors (ACEI) therapy:
- Mechanism of action: They prevent the conversion of angiotensin I to angiotensin II. This in turn prevents peripheral vasoconstriction, activation of the sympathetic nervous system, and salt and water retention due to aldosterone release. Thus, they interrupt the vicious circle of neurohumoral activation that is characteristic of moderate and severe heart failure. They also prevent the undesirable activation of the renin–angiotensin system caused by diuretic therapy.
- Uses: ACEIs improve survival in patients in all functional classes (NYHA I IV) and are given to all patients at risk of developing heart failure. They improve effort tolerance and mortality. They can also improve outcome, prevent the onset of overt heart failure in patients with asymptomatic heart failure following myocardial infarction.
- Initiation: Start low dose; if tolerated then gradual increase in few days to weeks to target dose or maximum tolerable dose with regular blood pressure monitoring. Serum creatinine should be measured concomitantly and potassiumsparing diuretics should be discontinued.
- Drugs and dosage: Captopril (6.25 mg thrice till 50 mg thrice a day), enalapril (2.5 mg twice to 10–20 mg twice a day), lisinopril (2.5–5 mg once to 20–40 mg once a day), and ramipril (1.25–2.5 mg once till 10 mg once a day).
- Angiotensin II receptor antagonists (ARA)/blockers therapy:
- Indications: ARAs are indicated as second-line therapy in patients intolerant of ACEI or alternative to ACEI.
- Drugs and dosage: Losartan (25–50 mg once till 50–150 mg once a day), valsartan, and telmisartan. Olmesartan (20–40
mg twice till 160 mg twice). - Same initiation and monitoring as ACEI and titration by doubling the dose.
- Beta-adrenoceptor blocker therapy:
- Indications: To all patients with current or prior HF and a LVEF ≤40% (HFrEF) in the absence of a contraindication
- Start low and increase gradually over a 12-week period even during hospitalization.
- Drugs and dosage bisoprolol (1.25–2.5 mg once till 10 mg once), carvedilol (3.125 twice till 50 mg twice), and metoprolol succinate (12.5 once till 200 mg once)
- Aldosterone receptor antagonists:
- Indications: NYHA II-IV, EF <35%, no contraindication (GFR >30, creatinine: 2.5 mg/dL male and 2.0 mg/dL female, K<5 mg/dL). They improve survival in patients with heart failure.
- Dose: Spironolactone 12.5–25 mg once till 50 mg daily
- Monitoring: Stop all K+ supplements, check K+, and creatinine 2–3 days after starting then 1 week and every month for 3 months and every 3 months and when clinically indicated.
- Side effects: Increase K+ (10–15%), gynecomastia, or breast pain
- Digoxin (Cardiac glycoside):
- Digoxin is a cardiac glycoside that is used in patients in atrial fibrillation with heart failure. It can be used as add on therapy in symptomatic heart failure patients already receiving ACEI and beta-blockers. No mortality benefit, only decrease in frequency of hospitalizations.
- No loading required. Usual dose 0.125–0.25 mg daily (low dose 0.125 mg alternate day if >70 years, chronic kidney disease, low lean body mass). Maintain 5–0.9 ng/dL plasma concentration (narrow therapeutic range).
- Vasodilators and nitrates (Hydralazine Nitrate Combination):
- The combination of hydralazine and nitrates reduces afterload and preload. Their use is limited by pharmacological tolerance and hypotension.
- Indication: African-American origin, NYHA III-IV, low EF on ACEI and BB, patients intolerant or contraindication of ACEI or ARA (e.g., in severe renal failure)
- Dose: 37.5 mg hydralazine and 20 mg and isosorbide dinitrate start one tab TID to increase till two tabs TID.
- Ivabradine:
- Ivabradine acts on the inward current if, in the SA node and reduces the heart rate. It reduces hospital admission and mortality rates in patients with heart.
- It is best given to patients who cannot take β-blockers or in whom the heart rate remains high despite β-blockade. It is not useful in patients with atrial fibrillation.
- Anticoagulation therapy is indicated in patients with heart failure who are at risk for thromboembolism. These include patients with atrial fibrillation, valvular heart disease, documented left ventricular thrombus, or a history of embolic stroke.
3. Nonpharmacological treatment of heart failure:
Question 111. Write a short note on implantable cardioverter defibrillator (ICD).
Answer:
- Device therapy:
- Implantable cardioverter defibrillator (ICD): Patients with symptomatic ventricular arrhythmias and heart failure have a very bad prognosis. Irrespective of their response to antiarrhythmic drug therapy, implantation of a cardiac defibrillator improves survival of all these patients. It is indicated in nonischemic or ischemic heart disease (at least 40 days post-MI) with LVEF of <35% with NYHA class II or III symptoms or NYHA 1 with EF <30% on chronic medical therapy, who have reasonable expectation of meaningful survival for >1 year.
- Cardiac resynchronization therapy (CRT):
- Indicated for patients who have LVEF of 35% or less, sinus rhythm, left bundle-branch block (LBBB) with a QRS duration of 150 ms or greater, and NYHA class II, III, or ambulatory IV symptoms
- In this, both the LV and RV are paced simultaneously to generate a more coordinated left ventricular contraction and improve cardiac output. It improves symptoms and survival.
- Coronary revascularization: Coronary artery disease is the most common cause of heart failure. Patients with angina and left ventricular dysfunction have a higher mortality from surgery (10–20%), but most patients’ symptoms and prognosis are improved. Coronary artery bypass surgery or percutaneous coronary intervention may improve function in region of the myocardium that is “hibernating” because of inadequate blood supply. The “hibernating” myocardium can be identified by stress echocardiography and specialized nuclear or MR imaging. Before recommending for surgery factors such as age, symptoms and evidence for reversible myocardial ischemia must be considered.
- Hibernating myocardium and myocardial stunning:
- Hibernating myocardium is the reversible left ventricular dysfunction with decreased myocardial perfusion, which is just sufficient to maintain viability of the heart muscle. It is due to an underlying chronic coronary artery disease. Myocardial hibernation is produced due to repetitive episodes of cardiac stunning which may occur, e.g., with repeated exercise in a patient with coronary artery disease. It responds positively to inotropic stress and indicates the presence of viable heart muscle that may recover after revascularization.
- Myocardial stunning is reversible ventricular dysfunction that persists following an episode of ischemia when the blood flow has returned to normal. This is due to a mismatch between flow and function.
- Hibernating myocardium and myocardial stunning:
- Heart (Cardiac) transplantation:
- Cardiac transplantation is an established and successful treatment of choice for younger patients with severe intractable heart failure, whose life expectancy is <6 months.
- Indications: Usually, reserved for young patients with severe symptoms despite optimal therapy. Most common indications are coronary artery disease and dilated cardiomyopathy.
- Contraindication: Patients with pulmonary vascular disease due to long-standing left heart failure, complex congenital
heart disease (e.g., Eisenmenger’s syndrome), or primary pulmonary hypertension
- Ventricular assist devices (VADs): There is limited supply of donor organs and VADs take over pumping for the ventricles. Hence, VADs are used as
- A bridge to cardiac transplantation
- Potential long-term therapy
- Short-term restoration therapy following a potentially reversible insult (e.g., viral myocarditis).
Newer Agents in Heart Failure Management:
Question 112. Discuss newer agents in heart failure management.
Answer:
- Nesiritide (recombinant analog of BNP):
- Actions:
- Increase natriuresis, diuresis, and cardiac index.
- Reduce pulmonary capillary wedge pressure, pulmonary artery pressure, pulmonary vascular resistance, and systemic blood pressure in a dose-dependent manner.
- Reversal of the deleterious neurohormonal response associated with heart failure.
- Reduce levels of endothelin 1, aldosterone, and norepinephrine.
- Advantages: Does not require ICU admission or invasive monitoring, lower incidence of tachycardia and proarrhythmic effects, and lessen the need for supportive therapies, such as diuretics.
- Actions:
- Endopeptidase inhibitor (ACE + neutral peptidases), e.g., omapatrilat
- Calcium sensitizer: For example, levosimendan. Levosimendan is a novel agent with inotropic properties developed specifically for the management of ADHF (acute decompensated heart failure). It acts by sensitizing troponin C to calcium.
- Endothelin receptor antagonist: For example, bosentan and tezosentan. Effective in acute coronary syndromes, acute renal failure, and acute heart failure. Indirectly improve contractility while decreasing pulmonary capillary wedge pressure.
- Vasopressin antagonists (V2 RA): For example, tolvaptan, lixivaptan, and conivaptan. It can be used as adjuvant to diuretic in advanced heart failure.
- Enoximone: Type 3 phosphodiesterase inhibitor
- Angiotensin receptor-neprilysin inhibitor: Sacubitril-valsartan for NYHA class II to IV heart failure with reduced ejection fraction.
Management of heart failure based on symptoms, cardiac output, and pulmonary capillary wedge pressure is depicted

Refractory Heart Failure:
Question 113. Write a short note on refractory heart failure and its management.
Answer:
- Optimal “triple therapy” for patients with chronic congestive heart failure (CHF) includes diuretics, digoxin, and either angiotensin-converting enzyme inhibitors or hydralazine plus nitrates.
- Refractory CHF is defined as symptoms of CHF at rest or repeated exacerbations of CHF despite “optimal” triple-drug therapy.
Management: It is managed as end-stage heart failure, i.e., stage D. Novel agents and nonpharmacological agents can be tried.

Brain Natriuretic Peptide:
Question 114. Write a short essay on brain natriuretic peptide (BNP).
Answer:
- It is called so because it was first discovered in porcine brain. It is a 32-amino acid peptide predominantly secreted by the left ventricles (as a response to left ventricular stretching or wall tension); along with an inactive 76-amino acid N-terminal fragment (NT-proBNP).
- It is activated only after a prolonged period of volume overload. It has an action similar to that of ANP but greater diagnostic and prognostic value as it has a longer half-life.
- Utility: Normal values of BNP: <50 pg/mL or <50 ng/L
- Diagnostic and prognostic utility: BNP levels are raised in conditions associated with left ventricular systolic dysfunction. It is used in the diagnosis of congestive heart failure and useful in patients presenting in the emergency room when the clinical diagnosis of heart failure is uncertain. BNP measurement is used to assess prognosis and monitor response to therapy in patients with heart failure. Raised levels of BNP in heart failure may be associated with an increased risk of death or cardiovascular events.
- Other conditions with raised BNP levels: Renal failure (due to decreased clearance), pulmonary embolism, pulmonary hypertension, chronic hypoxia, atrial fibrillations, acute myocardial infarction, obese patients, and sepsis.
Pulmonary Edema:
Question 115. Describe the clinical features, diagnosis and management of acute pulmonary edema.
(or)
Define pulmonary edema. Enumerate the causes of pulmonary edema.
(or)
Discuss the etiology, clinical presentation, investigations and management of cardiogenic pulmonary edema. Add a note on the pathophysiology.
(or)
Discuss the etiology/causes, clinical features, and management of acute left ventricular failure.
(or)
Define pulmonary edema.
Answer:
Pulmonary Edema Defiition:
Pulmonary edema is a condition characterized by accumulation of excess fluid in interstitium and alveoli of the lung as a result of an alteration in one or more of starling forces.
Classifiation and Causes of Pulmonary Edema:

Question 116. Write a short essay/note on noncardiac causes of pulmonary edema.
Answer:
Pulmonary edema can result from hemodynamic disturbances (hemodynamic or cardiogenic pulmonary edema) or from direct increases in capillary permeability (noncardiogenic pulmonary edema due to microvascular injury).
Based on underlying cause it can be classified as:
- Cardiogenic pulmonary edema is defined as a high pulmonary capillary hydrostatic pressure (as estimated clinically from the pulmonary capillary wedge pressure—PCWP) is responsible for abnormal fluid accumulation in alveoli of the lung.
- Noncardiogenic pulmonary edema is caused by various disorders in which factors other than elevated pulmonary capillary pressure are responsible for protein and fluid accumulation in the alveoli.
Stages in Development:
- Interstitial edema: This is an early stage where edema is limited to interstitium of lung.
- Alveolar edema: This occurs later, and characterized by the movement of edema fluid, macromolecules, and red blood cells into the alveoli.
Clinical Features of Acute Pulmonary Edema:
Cardiogenic pulmonary edema (CPE):
- Clinical features of left heart (acute ventricular) failure: Extreme breathlessness, shortness of breath orthopnea, and paroxysmal nocturnal dyspnea, anxiety, and feelings of drowning. Cough, pink frothy sputum.
- Physical findings:
- Tachypnea and tachycardia
- Hypertension
- Cool extremities may indicate low cardiac output
- Auscultation reveals fine, crepitations, or wheezes
- CVS findings: S3, accentuation of pulmonic component of S2, and jugular venous distention
- Patients with (RV) failure may present with hepatomegaly, sustained hepatojugular reflux, and peripheral edema. Change in mental status, caused by hypoxia or hypercapnia.
Diffrences between CPE and NCPE:
- CPE: A history of an acute cardiac event is usually present. Physical examination shows an S3 gallop, jugular venous
distention, and crackles on auscultation. - NCPE: They have a warm periphery, a bounding pulse, and no S3 gallop or jugular venous distention. Definite differentiation is based on pulmonary capillary wedge pressure (PCWP) measurements. The PCWP is generally >18 mm Hg in CPE and <18 mm Hg in NCPE.
Unusual Type Pulmonary Edema:
- Neurogenic pulmonary edema: Seen in patients with central nervous system disorders and without apparent preexisting LV dysfunction
- Re-expansion pulmonary edema: Develops after removal of air or fluid that has been in pleural space for some time, post-thoracocentesis. Patients may develop hypotension or oliguria resulting from rapid fluid shifts into lung.
- High altitude pulmonary edema: Occurs in young individuals who have quickly ascended to altitudes above 2,700 meters (8,000 feet) and who then engage in strenuous physical exercise at that altitude, before they have become acclimatized. Reversible in <48 hours.
Pulmonary edema Investigations:
Same as heart/cardiac failure and ARDS.
Question 117. Write a short essay/note on treatment of acute pulmonary edema/acute left ventricular failure.
Answer:
Treatment of Acute Cardiogenic Pulmonary Edema:
- Initial management: ABCs of resuscitation, i.e., airway, breathing, and circulation
- Medical treatment of CPE focuses on three main goals:
- Reduction of pulmonary venous return (preload reduction)
- Reduction of systemic vascular resistance (afterload reduction)
- Inotropic support
- Oxygenation: Oxygen should be administered to all patients to keep oxygen saturation at greater than 90%. Methods of oxygen delivery include the use of a face mask [noninvasive ventilation which includes (BiPAP) and (CPAP)], and intubation and mechanical ventilation. Oxygen corrects hypoxia and positive pressure raises intra-alveolar pressure reducing transudation of fluid.
- Preload reduction decreases pulmonary capillary hydrostatic pressure and reduces fluid transudation into the pulmonary interstitium and alveoli. Nitroglycerin oral or I/V 10–100 μg/min.
- Diuretics: They reduce the circulating blood volume and hasten the relief of pulmonary edema. Intravenous furosemide has a venodilator action by which it reduces venous return. This effect occurs within a few minutes while diuresis may take 30 minutes.
- Morphine sulfate: Morphine 2–5 mg intravenously slowly, and repeated if necessary, reduces anxiety and reduces venous return.
- Afterload reduction increases cardiac output and improves renal perfusion, which allows for diuresis in the patient with fluid overload.
- ACE inhibitors: Enalapril 1.25 mg IV or captopril 25 mg sublingually
- Angiotensin II receptor blockers
- Nitroprusside for 3–4 μg/kg/min IV infusion
- Inotropic agents: Dobutamine and dopamine
- Intra-aortic balloon pumping (IABP):
- The IABP is inserted percutaneously through the femoral artery to descending aorta using a modified technique. Fluoroscopy may be used for correct positioning of the balloon, and Helium gas is used to inflate the balloon.
- The IABP decreases afterload as the pump deflates; during diastole the pump inflates to improve coronary blood flow.
- Ultrafiltration is a fluid removal procedure that is particularly useful in patients with renal dysfunction and expected diuretic resistance.
- Correction of precipitating causes, e.g., infection or arrhythmias
- Treatment of underlying cause.
Cardiac Arrhythmias:
Question 118. Write a short note on definition of cardiac arrhythmias.
Answer:
Cardiac Arrhythmias Definition:
An abnormality (disturbance) of either rate or electrical rhythm of contraction of the heart is called a cardiac arrhythmia.
- Arrhythmias are usually due to structural disease of the heart but may also occur because of abnormal conduction or depolarization in an otherwise healthy heart.
Main Types of Arrhythmia:
- Bradyarrhythmia: The heart rate is slow and <60/min during the day or <50/min at night.
- Tachyarrhythmia: The heart rate is fast and >100/min.
Sinus Arrhythmia:
Question 119. Write a short essay on sinus arrhythmia.
Answer:
- Sinus arrhythmia is a normal physiological phenomenon and involves cyclic changes in the heart rate during breathing.
- Normal variation (phasic changes) of the heart rate in relation to breathing (during respiration) may occur due to fluctuations of normal parasympathetic nervous system (autonomic) activity/tone. During inspiration, parasympathetic tone falls and the heart rate increases, and on expiration, the heart rate decreases. This variation is normal, especially in children and young adults.
- The nonrespiratory form may occur in diseased hearts and sometimes confused with sinus arrest (also known as “sinus pause”).
- Typically, sinus arrhythmia results in predictable irregularities of the pulse.
- Significance: It is the most common arrhythmia and is manifestation of normal autonomic nervous activity. Absence of this normal variation in heart rate with breathing (sinus arrhythmia) or with changes in posture may be seen in autonomic neuropathy and cardiac failure.
Treatment is not usually needed unless symptomatic bradycardia is present.
Ectopic Beats (Extrasystoles; Premature Beats):
Question 120. Write a short essay/note on ectopic beats (extrasystoles; premature beats).
Answer:
- Definition: Ectopic beat is a heartbeat occurring as a result of an impulse originating in an area other than the sinoatrial (SA) node.
- Classification of ectopic beats is based on the site of origin of the impulse (ectopic focus).
- Supraventricular: Atrial ectopics, atrial premature beats arising from atrium
- Junctional: Arising from AV junction
- Ventricular: Arising from ventricular muscle
- Symptoms: Extra beats, thumping beats, or missed beats
- Signs: Irregularity in rhythm, missing of beats, postectopic bounding beat, and cannon waves on JVP.
Supraventricular Ectopics (Atrial Ectopics; Atrial Premature Beats):
Question 121. Write a short essay/note on supraventricular ectopics (atrial ectopics; atrial premature beats).
Answer:
- Atrial premature complexes (APCs) can be found on 24-hour Holter monitoring in over 60% of normal adults. They are usually asymptomatic and benign.
- Causes: Idiopathic in healthy individuals, anxiety, excessive coffee, tea or tobacco, ischemic and valvular heart disease
- Electrocardiogram (ECG) shows early abnormal p waves with a morphology that differs from the sinus p waves. It may be inverted if it originates near the AV node. R-R interval preceding and following the ectopic beat is less than twice the basal R-R interval (i.e., pause following the ectopic is not fully compensated).
- Supraventricular ectopics sometimes may precipitate atrial tachycardia, flutter, and fibrillation.
Supraventricular Ectopics Treatment: Treatment is useful only if they cause palpitations. Treatment of the underlying cause. Trigger for paroxysmal supraventricular tachycardias (e.g., alcohol, tobacco, or adrenergic stimulants) should be identified and eliminated. In their absence, mild sedation or betablocker may be tried.
Ventricular Premature Complexes (VPCs)/Ventricular Premature Beats (VPB)/Ventricular Ectopics:
Question 122. Write a short essay/note on ventricular ectopics [ventricular extrasystoles; ventricular premature beats, premature ventricular contractions (PVC)].
Answer:
- Of adult males, >60% will exhibit VPCs during a 24-hour Holter monitoring.
- In patients with previous MI, if frequent (>10 per hour) or complex VPCs are present, they are associated with increased mortality.
- Causes: Idiopathic in healthy individuals, excessive tea, coffee and alcohol, acute myocardial infarction, myocardial ischemia, myocarditis, digitals toxicity, hypokalemia, and mitral valve prolapse
- Electrocardiogram:
- These premature beats have wide, bizarre QRS complex, >0.12 seconds in duration and usually without a preceding P wave. They arise from an abnormal (ectopic) site in the ventricular myocardium.
- Usually, following a premature beat, there is complete compensatory pause because the AV node or ventricle is refractory
to the next sinus impulse. - The pause is fully compensated so that the sum R-R intervals preceding and following the ectopic QRS equals double the normal sinus R-R interval.
- When a VPC depolarizes the ventricles at a similar time as a conducted atrial beat, a fusion beat is observed.
- The term bigeminy refers to a normal complex followed by premature complex; trigeminy indicates a premature complex that follows two normal beats; a premature complex that occurs after three normal beats is called quadrigeminy. Two successive VPCs are known as a couplet, whereas three successive VPCs are called a triplet. Arbitrarily, three or more successive PVCs are termed Salvos/ventricular tachycardia. VPCs having different contours are often called multifocal.
Ventricular Ectopics Treatment:
- In the absence of cardiac disease, isolated asymptomatic VPCs, regardless of configuration and frequency, does not require any treatment.
- Beta-blockers are useful in symptomatic VPCs that occur mainly in the daytime or under stressful situations and in specific settings such as thyrotoxicosis and mitral valve prolapse.
- Implantable cardiac defibrillator (ICD) placement improves prognosis in patients with inducible VT and LV dysfunction
Tachycardia:
Question 123. Write a short note on tachycardia/narrow-complex and wide-complex tachycardia. Write a note on tachycardia.
Answer:
Tachycardia Definition: Tachycardia is defined as a heart rate of >100/minute.
Tachycardia Classifiation:
- Depending on origin:
- Supraventricular tachycardia is a tachycardia which utilizes atrial or AV nodal tissue as part of its mechanism (i.e., originating above the bundle of His).
- Ventricular tachycardia: Originates below the level of bundle of His
- Depending on QRS complex:
- Narrow-complex (QRS complex <0.12 seconds) tachycardia: Usually, tachycardia that originates from or above AV node generally produces narrow QRS complexes.
- Wide-complex (QRS >0.12 seconds) tachycardia: Usually, tachycardias that originate below the AV node produce wide QRS complexes.
Supraventricular Tachycardia:
Question 124. What do you understand by the term supraventricular tachycardia? Name various supraventricular tachycardia.
(or)
Write a short note on paroxysmal supraventricular tachycardia (PSVT).
Answer:
- Supraventricular tachycardia (SVT) is an arrhythmia and the term commonly used to describe regular tachycardia (heart rate exceeding 100 beats/minute) that arises from the atrium or the atrioventricular junction. Its conduction is via the His–Purkinje system. Hence, on ECG, the QRS shape during tachycardia is usually similar to that seen in the same patient during baseline rhythm. Various causes of supraventricular tachycardia are presented.
- These are usually associated with a narrow QRS complex and are characterized by a re-entry circuit or automatic focus involving the atria. The term SVT is misleading, as, in many cases, the ventricles also form part of the re-entry circuit, such as in patients with AV re-entrant tachycardia.
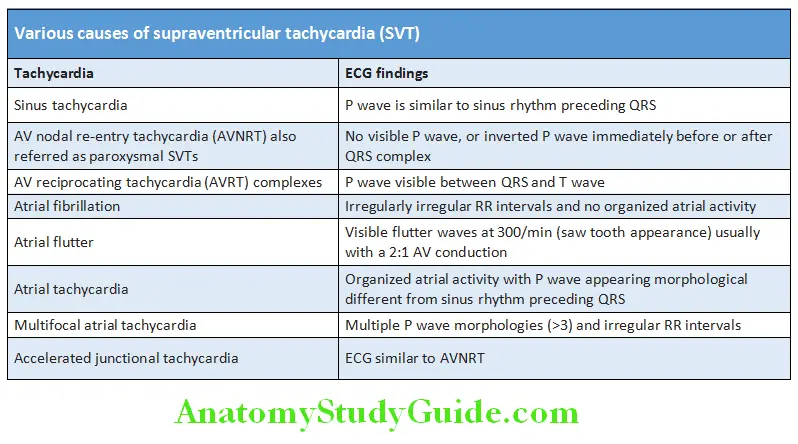
Sinus Tachycardia:
Question 125. Write a short note on sinus tachycardia.
Answer:
Sinus Tachycardia Defiition:
It is characterized by a heart rate of >100/ minute with normal P waves, P-R interval, and QRS complexes.
- If rate-dependent blocks appear or an accessory path is present, there may be widened QRS complexes.
- Causes: It is usually due to an increase in sympathetic activity associated with exercise, emotion, or pregnancy. Pathological causes include anemia, thyrotoxicosis, anxiety, fever, heart failure, pheochromocytoma, drugs, e.g.,β-agonists (bronchodilators) embolism and shock.
Sinus Tachycardia Treatment:
- Identification and treatment of the underlying cause
- If the tachycardia produces myocardial ischemia, treat with β-blockers.
AV Node Re-entry Tachycardia:
AV Node Re-entry Tachycardia (AVNRT) is the most common cause of paroxysmal supraventricular tachycardia (PSVT). AVNRT produces a regular tachycardia with a rate of 120–240/min.
Mechanism: AVNRT is due to re-entry in a circuit involving the AV node and there are two functionally and anatomically different right atrial input pathways.
- Superior “fast” pathway has a longer effective refractory period and conducts faster.
- Inferior “slow” pathway characterized by a short effective refractory period and slow conduction
If the atrial impulse occurs early (e.g., an atrial premature beat) when the fast pathway is still refractory, the slow pathway takes over in propagating this atrial impulse to the ventricles. It then travels back through the fast pathway which has already recovered its excitability. Thus, initiating the most common “slow, fast, or typical,” AVNRT.
Sinus Tachycardia Clinical features:
- It is common in women than men (F:M = 2:1).
- It usually occurs suddenly without any structural heart disease or obvious provocation. However, exertion, coffee, tea, and alcohol may aggravate or induce the arrhythmia.
- Episodes/attack may last from a few seconds to many hours and may stop spontaneously or may continue indefinitely until medical intervention.
- During the attack, the patient is usually aware of a rapid, very forceful, regular heartbeat, and may experience chest discomfort, lightheadedness, or breathlessness. Sometimes, polyuria (due to the release of atrial natriuretic peptide) may develop.
ECG: Usually shows a tachycardia at a rate of 140–240/minute with normal regular QRS complexes. Occasionally, the QRS complexes will show typical bundle branch block.
Sinus Tachycardia Management:
- Treatment is not always required.
- Episode/attack may be terminated in patients without hypotension by carotid sinus pressure or by the Valsalva maneuver.
- If these maneuvers are unsuccessful, adenosine (3–12 mg rapidly IV in incremental doses until tachycardia stops) or verapamil (5 mg IV over 1 min) will restore sinus rhythm in most of the patients. Intravenous β-blocker or flecainide may be helpful.
- Rarely, when there is severe hemodynamic compromise, the tachycardia should be terminated by DC cardioversion.
- Recurrent SVT: Catheter ablation will permanently prevent SVT in >90% of cases.
Atrial Fibrillation:
Question 126. Discuss the etiology, pathophysiology, clinical features, complications, diagnosis, and management of atrial fibrillation.
Answer:
Atrial Fibrillation Definition:
- Atrial fibrillation (AF) is an arrhythmia characterized by disorganized atria and produces multiple atrial foci fire impulses at a rate of 350–600/minute. There is no atrial contraction but only fibrillation. The ventricles respond at irregular intervals, usually at a rate of 100–140/minute.
- It is the most common cause of sustained cardiac arrhythmia.
Atrial Fibrillation Etiology:
Any condition that raises the atrial pressure, increases atrial muscle mass, causes atrial fibrosis, or inflammation and infiltration of the atrium, may produce atrial fibrillation. Many genetic (gene defects linked to chromosomes 10, 6, 5, and 4) and systemic causes also may be responsible for atrial fibrillation.
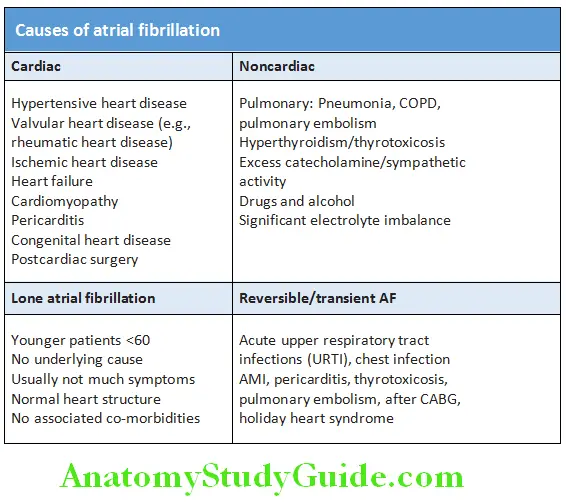
Mechanisms of Atrial Fibrillation:
- AF is a complex arrhythmia. It is characterized by
- Abnormal automatic firing at a rate of 350–600/minute
- The presence of multiple interacting re-entry circuits looping around the atria.
- Episodes of atrial fibrillation are initiated by rapid bursts of depolarizing automatic ectopic beats arising from conducting tissue in the pulmonary veins or from diseased atrial tissue.
- The atria respond electrically at this rapid rate. Many of them reach the AV node during its refractory period, and hence not conducted.
- AF becomes maintained by continuous, rapid (300–600/min) activation of the atria by multiple re-entrant conduction within the atria or sometimes because of continuous ectopic firing. Re-entry occurs in enlarged atria or in which conduction is slow (occurs in many forms of heart disease).
Atrial Fibrillation Clinical Features:
Symptoms:
Usual symptoms include palpitations, fatigue, syncope, angina, and symptoms of cardiac failure and thromboembolism.
Cardinal signs:
- Irregularly irregular pulse, varying volumes of pulse, pulse deficit (apex pulse deficit)
- Blood pressure: Hypotension, mean of three recordings to be taken
- Absence of a waves on JVP
- Varying intensity of the first heart sound, disappearance of the fourth heart sound, and disappearance of the PSA of the mid-diastolic murmur of mitral stenosis (MS) in a few.
Investigations:
Question 127. Write a short note on ECG changes in atrial fibrillation.
Answer:
- Electrocardiogram
- 2D ECHO to look for LA size, thrombus, LV function
- Holter monitoring: For paroxysmal AF
- Exercise stress test: Ischemic heart disease (IHD)
- Catheterization: Before ablation
- Thyroid function tests and serum electrolytes in all patients
- Chest X-ray
- Fasting lipid profile.
ECG changes in atrial fibrillation:
- No clear P waves
- Fine very irregular, disorganized atrial fibrillatory or F waves, coarse fibrillary waves indicate LAE
- F waves: Several independent re-entrant wavelets within atria, which may be fine or coarse
- Atrial rate: 350–500 bpm
- Ventricular rate: Irregularly irregular, conduction of AV node
- RS complexes: Rhythm is rapid and irregular, narrow/broad. Broad-BBB, aberrant conduction, and pre-excitation
- Long R-R interval is followed by short R-R intervals. Atrial impulse may find the RBBB still refractory. Ashman phenomenon is characterized by wide QRS complexes with a short cycle following long cycle commonly seen in ASD.
Question 128. Write a short note on complications of atrial fibrillation. Complications: Thromboembolism, precipitation/worsening of cardiac failure, syncope, hypotension, angina, precipitation of pulmonary edema in mitral stenosis.
Answer:
Management/Treatment:
Goals of management:
- Hemodynamic stabilization
- Heart rate control or restoration of sinus rhythm
- Minimize symptoms associated with excessive heart rates
- Prevent tachycardia-associated cardiomyopathy
- Prevention of recurrent AF
- Reduction of the risk of thromboembolism: Anticoagulation prevents embolic complications.
- Treatment of underlying cause: When atrial fibrillation develops as a complication of acute precipitating event (e.g., alcohol toxicity, chest infection, pulmonary embolism, or hyperthyroidism), the provoking primary disorder/cause should be effectively treated. It will often restore sinus rhythm.
Strategies for the acute management of AF:
- Ventricular rate control is achieved by drugs which block the AV node.
- Cardioversion (± anticoagulation): Cardioversion is a procedure by which an abnormally fast heart rate (tachycardia) or cardiac arrhythmia is converted to normal rhythm using electricity or drugs. Cardioversion is achieved electrically by DC shock or medically antiarrhythmic drug either by intravenous infusion (e.g., flecainide, propafenone, vernakalant, or amiodarone) or orally (flecainide or propafenone). Oral agent is administered to a particular patient who is previously tested in hospital and found to be safe (“pill in pocket” approach).
Long-term management of atrial fibrillation:
Clinical classification of atrial fibrillation is presented. Long-term management of persistent atrial fibrillation has two options:
1. Rhythm control is an attempt to restore and maintain normal sinus rhythm by antiarrhythmic drugs plus DC cardioversion plus oral anticoagulation.
- Pharmacological cardioversion: By quinidine, ibutilide, flecainide, propafenone, or amiodarone. The dose of amiodarone 5–7 mg/kg intravenously over 1 hour followed by 1.2–1.8 g/24-hour infusion.
- Electric cardioversion: If there is failure of medical cardioversion, electric cardioversion is done after 3 weeks of warfarin therapy which is continued further for another 4 weeks after cardioversion. Anticoagulation is to be given to these patients.
2. Rate control: If sinus rhythm cannot be restored, treatment is aimed to control the ventricular rate to <100/minute and to prevent embolic complications. This is achieved by AV nodal slowing agents plus oral anticoagulation.
- Calcium channel blockers (calcium antagonists):
- Verapamil: 5–10 mg intravenously over 2 minutes. If required repeat in 30 minutes.
- Diltiazem: 10 mg intravenously over 2 minutes. If required repeat same dose in 15 minutes. An infusion at 10–15 mg/hour is started to maintain ventricular rate below 100/minute.
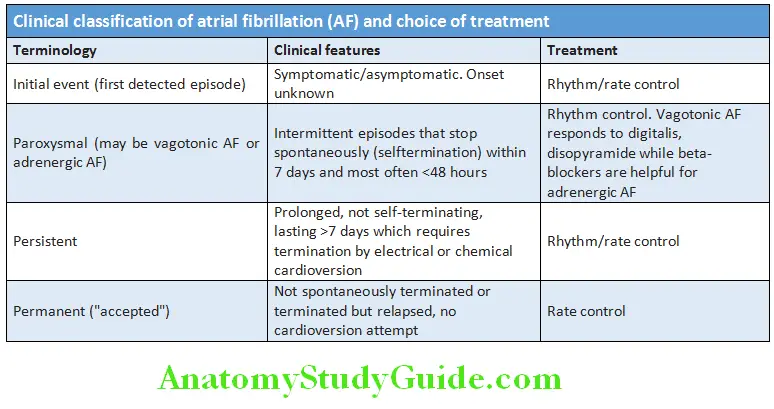
- β-blockers: Propranolol 1 mg intravenously over 2 minutes. Dose is repeated every 5 minutes up to a maximum of 5 mg.
- Digoxin: 0.25–0.5 mg intravenously, then 0.25 mg after 4–6 hours and another after 12 hours. At present less commonly used.
- Amiodarone: 150 mg over 10 minutes followed by 1 mg/minute over 6 hours and then 0.5 mg/minutes for next 18 hours. Adverse effects include: hepatic toxicity (hepatitis that can progress to cirrhosis), pulmonary toxicity (cough and dyspnea), thyroid dysfunction (hypothyroidism, hyperthyroidism), sun sensitivity, and ocular symptoms.
Prevention of recurrence:
- After restoring the sinus rhythm (either by electric or pharmacological cardioversion), recurrence is prevented by quinidine, amiodarone, or dronedarone (safer than amiodarone).
- When cardioversion is unsuccessful or if atrial fibrillation is likely to recur, following treatment is given:
- Allow the patient to remain in atrial fibrillation but reduce the ventricular rate by digitals, diltiazem, verapamil, or propranolol.
- Chronic anticoagulation
- Reduction in stroke risk; by warfarin or antiplatelet agents.
Reduction of the risk (prevention) of thromboembolism (anticoagulation):
One of the complications of atrial fibrillation is embolism. First step is to determine the need for anticoagulation. A scoring system known as CHA2DS2-VASc is used to assess the risk of embolism.
Indications: Atrial fibrillation in patients with rheumatic mitral stenosis or with mechanical prosthetic heart valve.
Other indications include:
- Transesophageal echocardiography should be done in patients with atrial fibrillation to exclude an atrial thrombus.
- If no atrial thrombi: Heparin is given before cardioversion (if conversion to sinus rhythm) and followed by warfarin for 4 weeks.
- If atrial thrombi present: Warfarin is given for 3 weeks prior to cardioversion and is continued for another 4 weeks after cardioversion.
- If cardioversion is unsuccessful and patient remains in atrial fibrillation, long-term warfarin should be given.
Anticoagulants: With warfarin, the INR should be maintained between 2.0 and 3.0 (2.5 and 3.5 in case of underlying valvular lesion). Newer oral anticoagulants include dabigatran (a direct thrombin inhibitor in the dose of 150 mg BID), apixaban (5 mg BID), and rivaroxaban (20 mg OD).
Aspirin: In the dose of 325 mg/day may be used as an alternative to warfarin when patient is allergic to warfarin or is contraindicated, patient <75 years of age with no previous stroke or transient ischemic attack without hypertension, diabetes, or heart failure.
Radiofrequency ablation (RF) ablation therapy:
- Indications are:
- Very symptomatic patients who refuse antiarrhythmic drug therapy
- Young patients where only effective antiarrhythmic drug is amiodarone
- Patients with significant bradycardia for whom antiarrhythmic drug therapy will require pacemaker
- Surgical procedure for AF is the “cut-and-sew” maze procedure
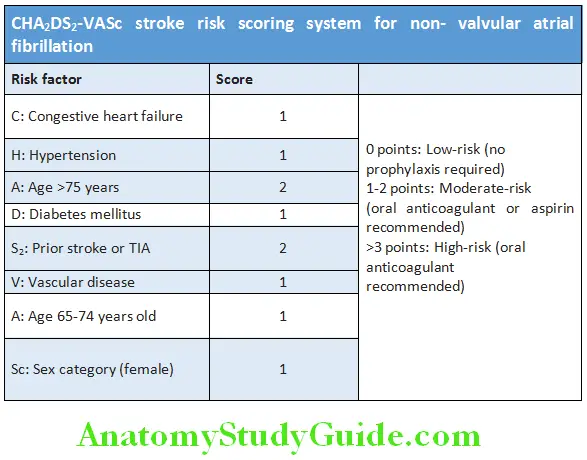
Atrial Flutter:
Question 129. Define atrial flutter. Discuss the causes, electrocardiographic features, and management of atrial flutter.
Answer:
Atrial Flutter Defiition:
Atrial flutter is a cardiac arrhythmia usually characterized by an organized, regular, rapid atrial rate between 250 and 350/ minute. The ventricles respond to every second, third, or fourth beat (2:1, 3:1, or 4:1 AV block).
Atrial Flutter Common Causes:
- Cardiac causes: Organic heart diseases (e.g., ischemic, rheumatic, congenital), pericarditis, following open heart surgery (1st week)
- Acute respiratory failure
ECG: Characteristic saw-toothed flutter waves (“F” waves) between QRS complexes. QRS complexes are regular.
Atrial Flutter Management:
- Restoration to sinus rhythm: Treatment of a symptomatic acute paroxysm is direct electrical cardioversion. Atrial flutter of >1–2 days should be treated similar to atrial fibrillation and anticoagulated for 3 weeks prior to cardioversion.
- Control of ventricular rate: By using digoxin, β-blockers, or verapamil/diltiazem followed by conversion to sinus rhythm using quinidine, amiodarone, disopyramide, or flecainide
- Prevention of recurrences: Recurrent paroxysms can be prevented by class III antiarrhythmic agents such as quinidine, amiodarone, disopyramide, or flecainide. Treatment of choice for patients with recurrent atrial flutter is catheter ablation.
- Prevention of stroke: Risk of stroke is similar to that of atrial fibrillation and the management is almost identical but anticoagulants may be stopped earlier after successful ablation.
WolffParkinson–White Syndrome (WPW Syndrome):
Question 130. Write a short essay on Wolff–Parkinson–White (WPW) syndrome.
Answer:
Wolff–Parkinson–White (WPW) syndrome is an extra electrical (accessory) pathway (Bundle of Kent) between atria and ventricles and causes tachycardia.
Accessory pathways: Extra electrical pathway consists of an abnormal band of rapidly conducting fibers which connects the atria and ventricles. These conducting fibers resemble Purkinje tissue, in that they conduct very rapidly and are rich in sodium channels. Accessory pathways may be of two types.
1. Concealed accessory pathway: About 50% of cases, this pathway only conducts in the retrograde direction (from ventricles to atria) and thus does not change the appearance of the ECG in sinus rhythm. It is called as a concealed accessory pathway.
2. Manifest accessory pathway: In remaining 50%, the pathway conducts anterograde direction (from atria to ventricles). Thus, AV conduction in sinus rhythm is mediated via both the AV node and the accessory pathway and distorts the QRS complex. Premature ventricular activation via this pathway shortens the PR interval and produces a “slurred” initial deflection of the QRS complex, and is called a delta wave. This manifests as an accessory pathway. Because AV node and accessory pathway have different conduction speeds and refractory periods, a re-entry circuit can develop, producing tachycardia. When this is associated with symptoms, it is termed as Wolff–Parkinson–White (WPW) syndrome.
ECG: During tachycardia is indistinguishable from that of AVNRT.
- Short PR interval (<0.12 second)
- Slurred upstroke of the QRS complex (Delta wave)
- Wide QRS complex
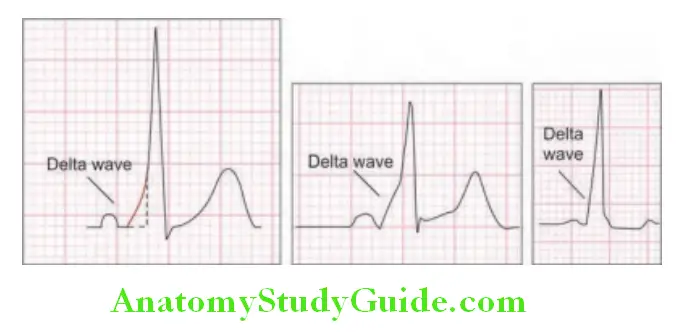
Complications: Atrial fltter, atrial firillation, supraventricular tachycardia, ventricular tachycardia, ventricular firillation, and death
WolffParkinson–White Syndrome Treatment:
- Carotid sinus pressure or intravenous adenosine used for termination of the tachycardia
- Acute episodes of PSVT in Wolff–Parkinson–White (WPW) syndrome are treated similar to that of PSVT.
- Life-threatening atrial fibrillation is treated as an emergency with DC cardioversion.
- In nonlife-threatening atrial fibrillation (AF), lidocaine (3–5 mg/kg) or procainamide (15 mg/kg) administered IV over 15–20 minutes will usually show the ventricular response. Ibutilide can also be used.
- Avoid IV verapamil and digitalis in WPW and AF, because they can increase ventricular response (by shortening the refractory period of accessory pathway). Beta-blockers are of no use in controlling ventricular response during AF when conduction proceeds over the bypass tract.
- Catheter ablation of bypass tracts is possible in >90% of the patients and is the treatment of choice in patients with symptomatic arrhythmias. Surgical ablation may be required in an occasional patient in whom catheter ablation fails.
Sick Sinus Syndrome:
Question 131. Write a short essay on sick sinus syndrome.
Answer:
- It comprises a number of abnormalities namely sinus bradycardia, sinus arrest, combinations of sinoatrial and AV blocks, tachycardia-bradycardia, and supraventricular tachycardia.
- It is characterized by sinus node dysfunction with an atrial rate inappropriate for physiologic requirements.
- Cause: Usually due to ischemia, fibrosis, drug-induced, or autonomic dysfunction.
Sick Sinus Syndrome Clinical Features:
- Majority are asymptomatic
- Clinical features are due to cerebral hypoperfusion and reduced cardiac output. These include syncope, palpitations, and dizziness. There may be symptoms caused due to worsening of underlying conditions (e.g., congestive heart failure, angina pectoris, and cerebrovascular accident).
- A slow heart rate, fever, left ventricular failure, or pulmonary edema suggest sick sinus syndrome.
Treatment of recurrent symptomatic bradycardia or prolonged pauses by implantation of a permanent pacemaker.
Ventricular Tachycardia (VT):
Question 132. Write a short essay on ventricular tachycardia.
Answer:
- Most common cause of wide complex tachycardia (80%)
- VT heart rate is 100–200 bpm and if more than rate >220 bpm, it is ventricular fibrillation (VF).
- Major cause of morbidity and mortality in patients with structural heart disease
- Major cause of sudden cardiac death—60% cases on Holter monitoring
- Relatively organized tachyarrhythmias with discrete QRS complexes
- Recurrence is more common in <1 year of onset
- The occurrence of three or more VPC complexes with a rate of >120 bpm in succession is called as VT.
Types of VT:
- Nonsustained VT is termination of VT by self <30 seconds.
- Sustained VT is presence of VT for >30 seconds or hemodynamically unstable but terminated in <30 seconds.
- Slow VT: Heart rate >100 or <120 bpm.
- Pulseless VT: VT with hemodynamic collapse that requires DC cardioversion
- Refractory VT: VT that does not revert to sinus rhythm on medication use or use of three shocks
- VT storm: Repeated VT episodes requiring the DC shocks/ICD shocks
Ventricular Tachycardia Etiology:
Re-entry is the most common mechanism.

Ventricular Tachycardia Clinical Features:
Ventricular Tachycardia Diagnosis:
- ECG: 12 lead with long rhythm strip of lead II
- 24-hour Holter monitoring in case of transient episode
- 2D echo for the etiology
- Routine investigations, serum electrolytes, calcium, magnesium, and ABG
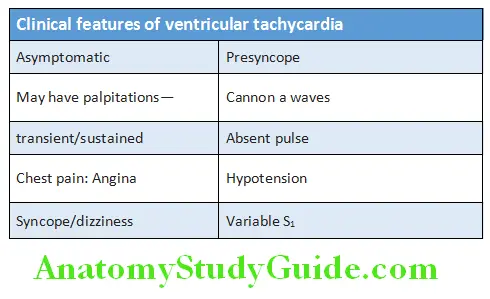
Ventricular Tachycardia Treatment:
- Prompt restoration of sinus rhythm followed by prophylactic therapy.
- Hemodynamically unstable patients: Synchronized DC cardioversion is the treatment of choice (if systolic BP is below 90 mm Hg).
- If the patient is stable and arrhythmia is well tolerated: With preserved left ventricular function IV procainamide 50 mg/min. In presence of left ventricular dysfunction, IV 150 mg amiodarone given as a bolus, followed by a continuous infusion. IV lidocaine can be used but may depress left ventricular function, causing hypotension, or acute heart failure.
- Correction of electrolyte or acid–base imbalance, e.g., hypokalemia, hypomagnesemia, acidosis, and hypoxemia
- Prevention of VT: Beta-blockers reducing ventricular automaticity and amiodarone can be added if additional control is needed. Do not use class Ic antiarrhythmic drugs in patients with coronary artery disease or heart failure because they depress myocardial function and can be proarrhythmic (increases the possibility of a fatal arrhythmia).
- Implantable cardiac defibrillator is the absolute indication in the presence of LVEF <30% and is recommended for patients with high risk of arrhythmic death (e.g., poor left ventricular function, or associated with hemodynamic compromise). Rarely, surgery (e.g., aneurysm resection) or catheter ablation may be required for patients with VT associated with a myocardial infarct scar.
Torsades De Pointes:
- A special type of ventricular tachycardia (VT) characterized by polymorphic QRS complexes that change in amplitude and cycle length, giving the appearance of oscillations around the baseline. This is associated with QT prolongation.
- QT prolongation may result from electrolyte disturbances (particularly hypokalemia and hypomagnesemia), antiarrhythmic drugs (quinidine), phenothiazines, tricyclic agents, and bradyarrhythmias. It may be due to congenital long QT syndrome (Andersen–Tawil syndrome, Romano–Ward syndrome, Jervell and Lange-Nielsen syndrome).
- The electrocardiographic hallmark is polymorphic VT preceded by marked QT prolongation, often in excess of 0.60 second.
Torsades De Pointes Treatment:
- Correct electrolyte abnormalities or underlying cause
- Intravenous magnesium (8 mmol over 15 minutes, then 72 mmol over 24 hours) should be given in all patients.
- Avoid class IA and class III antiarrhythmic agents.
- Lidocaine does not prolong QT interval. Isoproterenol bridge before temporary pacemaker. Phenytoin 100 mg every 5 minutes maximum dose of 500 mg.
- Temporary pacemaker
- Beta-blockers (BBs) to be used to prevent syncope in patients with congenital QTc prolongation syndrome
Brugada Syndrome:
- Named by the Spanish cardiologists, Pedro Brugada and Josep Brugada
- Major cause of sudden unexpected death syndrome (SUDS), and is the most common cause of sudden death in young en without known underlying cardiac disease in Thailand and Laos.
- Genetic abnormality of the cardiac sodium channels, leading to ventricular tachycardia and/or sudden cardiac death.
- Hereditary: 60% of Brugada patients have a family history of SCD. Mutations of SCN5a genes resulting in diminished inward sodium current in the region of the RV outflow tract epicardium appear responsible for this syndrome.
- The large potential difference between the normal endocardium and rapidly depolarized RV outflow epicardium gives rise to ST segment elevation in V1 to V3 in sinus rhythm and predisposes to local re-entry and life-threatening ventricular arrhythmias in the absence of structural heart disease.
- Arrhythmias frequently occur during sleep. Affects relatively young and predominant in males.
Brugada Syndrome Treatment:
- No benefit from beta-blockers
- Implantable cardiac defibrillator (ICD) treatment to manage recurrences. A history of syncope spontaneous ST segment elevation and inducibility of VT with programmed stimulation.
Ventricular Fibrillation:
Question 133. Write a short note/essay on causes, diagnosis, and treatment of ventricular fibrillation.
Answer:
- It is characterized by very rapid, irregular, and uncoordinated movement of the ventricles with no mechanical effect.
- Patient is pulseless and becomes rapidly unconscious, and respiration ceases (cardiac arrest).
- ECG: Shows shapeless/bizarre, rapid oscillations
- Ventricular fibrillation rarely reverses spontaneously.
Ventricular Fibrillation Treatment: Electrical defibrillation.
Atrioventricular Blocks:
Question 134. Discuss the causes, clinical features, and management of atrioventricular blocks and AV conduction disturbances.
Answer:
Heart block or conduction block may develop at any level in the conducting system. Block in either the AV node or the His bundle produces AV block, whereas block lower in the conduction system results in bundle branch block.
Atrioventricular Blocks Types:
Atrioventricular block consists of three forms:
- First-Degree Av Block
- Second-Degree Av Block
- Thirddegree (complete) AV block.
First-degree Atrioventricular (AV) Block:
- In this type of block, AV conduction is delayed.
- ECG shows simple prolongation of the PR interval to >0.22 seconds. Every atrial depolarization is followed by conduction to the ventricles but occurs with delay. So, all the P waves are conducted and the QRS is normal as the delay is most often in the AV mode.
Second-degree Atrioventricular (AV) Block:
In this type of AV block, dropped beats occur because some impulses from the atria fail to conduct to the ventricles.
Second-degree Atrioventricular Types:
- Mobittype I second-degree AV block (Wenckebach block phenomenon):
- It is characterized by progressive slowing of AV conduction until it is totally blocked. ECG shows progressive lengthening of successive PR intervals until a P wave fails to conduct (culminating in a dropped beat).
- Mobittype II second-degree AV block: In this type, the PR interval remains constant but some atrial impulses (P waves) are not conducted. It occurs when a dropped QRS complex is not preceded by progressive PR interval prolongation. The QRS complex is usually wide (>0.12 second).
- 2:1 or 3:1 (advanced) block: It occurs when every second or third P wave conducts to the ventricles. The block is defined by a ratio in which the first digit represents the total number of P waves, and the second digit represents the number of P waves conducted (i.e., the number of QRS complexes). This form of second-degree block is neither MobitI nor II.
Third-degree (Complete) Atrioventricular Block:
Question 135. Write a short essay on complete heart block.
Answer:
Complete heart block occurs when all the atrial activities fail to conduct (AV conduction fails completely) to ventricles and both the atria and ventricles beat independently (AV dissociation). In this ventricular activity (and the life) is maintained by spontaneous escape rhythm arising in the AV node or bundle of His (narrow QRS complexes) or the distal Purkinje tissues (broad QRS complexes). Distal escape rhythms are slower and less reliable. Causes of complete heart block.
Clinical features of complete heart block:
- Regular (except in congenital complete AV block), high volume slow pulse (25–50/minute) which does not vary with exercise.
- Usually, there is a compensatory increase in stroke volume and produces a large-volume pulse.
- Irregular cannon waves on JVP in the neck
- Stokes–Adams attacks
- Varying intensity of first heart sound
ECG: Constant P-P and R-R intervals, complete AV dissociation (i.e., the atria and ventricles beat independently) and there is no relation between the P waves and the QRS complexes.
Causes of heart block:
Congenital:
Acquired:
- Myocardial ischemia or infarction
- Idiopathic fibrosis
- Inflammation:
- Acute (e.g., aortic root abscess in infective endocarditis)
- Chronic (e.g., sarcoidosis and Chagas disease)
- Trauma (e.g., intracardiac surgery)
- Drugs (e.g., digoxin intoxication and β-blocker)
- Tumors and infections involving the conducting system
- Infections: Rheumatic autoimmune disease and neuromuscular diseases
- Lev’s disease
- Lenegre’s disease
Adams–Stokes Attacks (Stokes–Adams–Morgagni Attacks):
Question 136. Write a short essay on Adams–Stokes attacks (Stokes–Adams–Morgagni attacks) and their clinical features.
Answer:
Stokes–Adams attack is characterized by a recurrent episode of sudden loss of consciousness unrelated to posture with or without convulsions due to a disorder of heart rhythm in which there is bradycardia or absent pulse.
Adams–Stokes Attacks Etiology:
Episodes of ventricular asystole may complicate intermittent high-grade AV block (Mobittype II or complete heart block), profound bradycardia, or ventricular standstill. Sinoatrial disease and neurocardiogenic syncope may produce similar symptoms.
Adams–Stokes Attacks Clinical Features:
- Prodrome preceding the attack may be observed in few patients.
- Sudden loss of consciousness: Patient may fall to the ground with sudden loss of consciousness without warning and results in collapse. During the attack, patient is pale with a death-like appearance and deeply unconscious. The pulse is usually very slow or absent. Recovery is rapid and after a few seconds, the patient recovers consciousness with characteristic flush as the heart starts beating again and pulse quickens.
- Occasionally, brief anoxic generalized convulsions/seizures and death may occur if there is prolonged asystole or severe prolonged bradycardia (>10 seconds) causing cerebral hypoxia/ischemia. Prolonged bradycardia may produce cyanosis.
- Usually there are no sequelae, but patients may injure themselves due to the sudden fall.
Treatment of heart block:
- Removal of offending agent,
- Injection atropine/injection isoprenaline if symptomatic
- Pacemaker insertion.
Therapeutic Procedures:
Defirillation and Cardioversion:
Question 137. Write a short note on defibrillation and cardioversion.
Answer:
Defirillation and Cardioversion Introduction:
- By the passage of sufficiently large electrical current (from an external source) through the heart, it can be completely depolarized. This will interrupt any arrhythmia and produce a short period of asystole and is usually followed by the resumption of normal sinus rhythm.
- Cardioversion is a procedure by which an abnormally fast heart rate (tachycardia) or cardiac arrhythmia is converted to a normal rhythm using electricity (defibrillation) or drugs.
Defirillation and Cardioversion Defirillation:
- In this technique, ventricular fibrillation is converted to sinus rhythm by defibrillators which deliver direct current (DC). When the defibrillator is discharged, a high-voltage field electric shock of short duration is delivered to the heart.
- This electric shock envelopes the heart and depolarizes the myocardium which causes an organized heart rhythm to emerge.
- Method: Defibrillators deliver a DC, high-electrical energy, short-duration shock through two large electrodes or paddles coated with conducting jelly or a gel pad. One electrode is positioned over the upper right sternal edge and the other over the cardiac apex. Present-day modern units deliver a biphasic shock. Advantage is that the shock polarity is reversed during mid-shock and this decreases the total shock energy needed to depolarize the heart.
Direct current cardioversion (DCC):
- Transthoracic electric shock: It is used to convert sinus rhythm in tachyarrhythmias which do not respond to medical treatment or that are associated with hemodynamic compromise (e.g., hypotension and worsening heart failure).
- Precautions:
- A short-acting general anesthetic or powerful sedation is used in elective cardioversion.
- Withdraw digitalis therapy at least 36 hours before cardioversion
- Patients with long-standing (>48 hours) atrial fibrillation or flutter should be anticoagulated adequately for at least 3 weeks before cardioversion to reduce the risk of embolization.
- Levels of cardiac enzyme may rise after a cardioversion.
- Difference between defibrillation and cardioversion: A nonsynchronize shock is used to defibrillate and accidental defibrillation (in patient who do not need it) may itself precipitate ventricular fibrillation. A synchronized shock (i.e., one delivered during the QRS complex) is used for all cardioversions except for very rapid ventricular tachyarrhythmias, such as ventricular flutter or VF.
- Indications for DCC: Ventricular fibrillation, sustained ventricular tachycardia, atrial fibrillation/flutter, and supraventricular tachycardia.
Cardiac Pacemakers:
Question 138. Write a short note on types of cardiac pacemakers and its indications.
Answer:
Temporary pacing:
- It involves delivery of an electrical impulse into the heart to initiate depolarization and to trigger cardiac contraction.
- Indication: Transient AV block, arrhythmias complicating acute MI or cardiac surgery, to maintain the rhythm in other situations of reversible bradycardia (i.e., due to metabolic disturbance or drug overdose), or as a bridge to permanent pacing
Transvenous pacing: It is done in patients with symptomatic bradycardias. In this procedure under the guidance of cardiac fluoroscopic imaging, a thin (French gauge 5 or 6), bipolar pacing electrode wire is inserted via an internal jugular vein, a femoral vein, or a subclavian vein and is positioned at the apex of the right ventricle. The electrode is connected to an external pacemaker with an adjustable energy output and pacing rate usually about 60–80/minute.
Complications: Pneumothorax, brachial plexus or subclavian artery injury, local infection or septicemia (usually Staphylococcus, aureus), and pericarditis.
Transcutaneous pacing is performed in selected patients with asymptomatic bradycardia or conduction abnormalities.
It may be lifesaving for patients with cardiac arrest precipitated by bradycardia. This method consists of depolarizing the myocardium by current flow between two large adhesive electrodes positioned anteriorly and posteriorly on the chest wall.
Permanent pacing:
Pacing electrodes (leads) can be placed via the subclavian or cephalic veins and are designed to both pace and sense either the ventricles or the atria, or more commonly, both chambers.
Indication for permanent pacing:
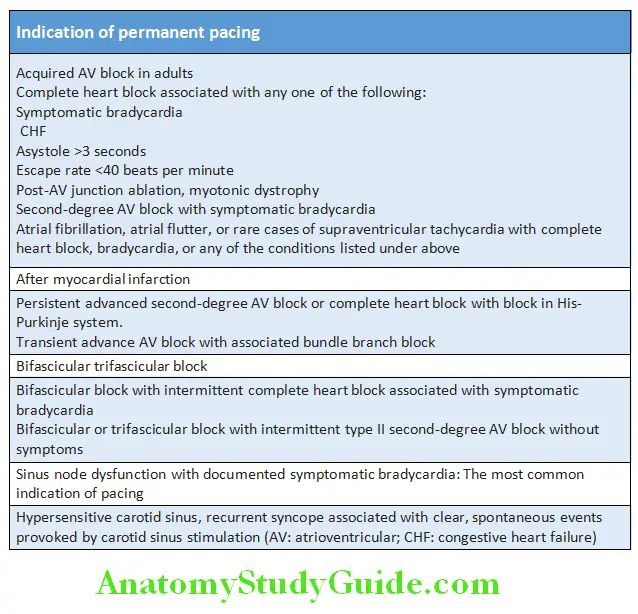
Antiarrhythmic Drugs:
Question 139. Write a short essay on antiarrhythmic drugs.
Answer:
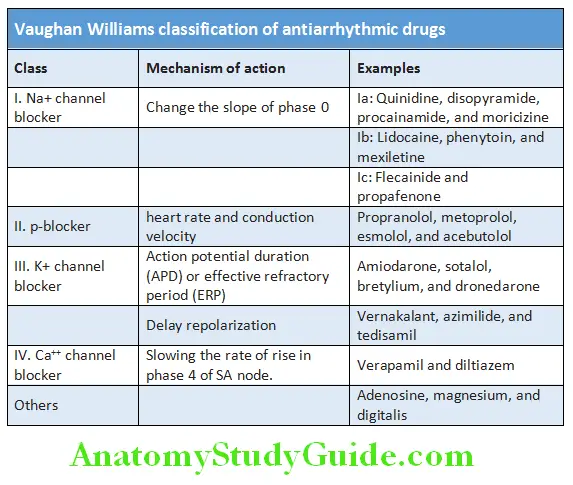
Therapy of Various Arrhythmias:
Amiodarone:
Question 140. Write a short note on amiodarone and its use in clinical practice.
Answer:
Amiodarone is an unusual iodine containing highly lipophilic long-acting antiarrhythmic drug.
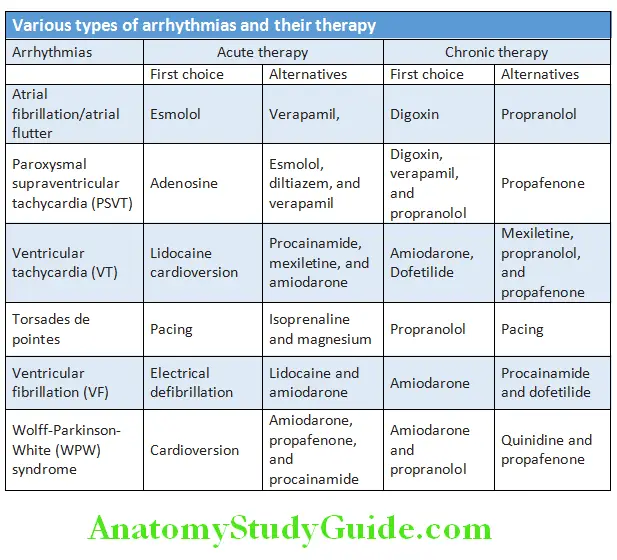
Indications: Useful in wide range of ventricular and supraventricular arrhythmias.
- Resistant ventricular tachycardia/pulseless VT
- Recurrent ventricular fibrillation
- To maintain sinus rhythm in atrial flutter when other drugs have failed. For patients with heart failure or left ventricular hypertrophy, only amiodarone is recommended.
Duration of action: Long. Hence suitable for long-term prophylactic therapy
Adverse reactions: These are dose-related and increase with duration of therapy. These reactions include fall in blood pressure, bradycardia, and myocardial depression on IV injection and on drug cumulation. Nausea, gastrointestinal upset with oral medication, photosensitization, and bluish skin discoloration pigmentation may develop in about 10% of patients.
Pulmonary alveolitis and fibrosis are serious adverse reactions. Cirrhosis occurs uncommonly. Neurologic dysfunction, and hyperthyroidism (1–2%) or hypothyroidism (2–4%) can be seen.
Dose:
- Oral 400–600 mg/day for few weeks, followed by 100–200 mg for maintenance therapy
- Slow IV injection of 100–300 mg (5 mg/kg) over 30–60 minutes
Diseases Of The Myocardium:
Myocarditis:
Question 141. Write a short essay/note on acute myocarditis.
Answer:
- Acute myocarditis is an acute inflammatory disease of the myocardium (heart muscle).
- Myocarditis may present with a wide range of symptoms, ranging from mild dyspnea or chest pain that resolves without specific therapy to cardiogenic shock and death.
- Major long-term sequelae of myocarditis is DCM (dilated cardiomyopathy) with chronic heart failure.
- Three distinct forms of inflammatory cardiomyopathy (myocarditis associated with cardiac dysfunction) are recognized: Idiopathic, autoimmune, and infectious.
Etiology:
Question 142. Write a short essay/note on causes of myocarditis.
Answer:
Clinical Features of Myocarditis:
Range from an asymptomatic or present with fatigue, palpitations, chest pain, dyspnea, and fulminant congestive cardiac failure depending on the type of myocarditis.
Classification: Depending on the clinical presentations, myocarditis can be classified into four groups:
1. Fulminant myocarditis: Has abrupt onset and follows a viral prodrome (fever, chills, myalgia, and constitutional symptoms) or influenza-like illness, and produces severe acute heart failure or cardiogenic shock. Prognosis is good.
2. Acute myocarditis: It presents with heart failure, acute myocardial infarction, or sudden cardiac death. It may progress to dilated cardiomyopathy and chronic heart failure. More common in children/teenagers.
3. Chronic active myocarditis: It is rare and have insidious onset. It is seen in older adults and microscopically shows
chronic inflammation of the myocardium.
4. Chronic persistent myocarditis: It shows focal myocardial infiltrates and cause chest pain and arrhythmia without causing ventricular dysfunction.
Physical examination may be normal or present with soft muffled heart sounds, a prominent third sound, inappropriate tachycardia, arrhythmias including conduction blocks, signs of congestive heart failure, and pericardial friction rub (associated with pericarditis).
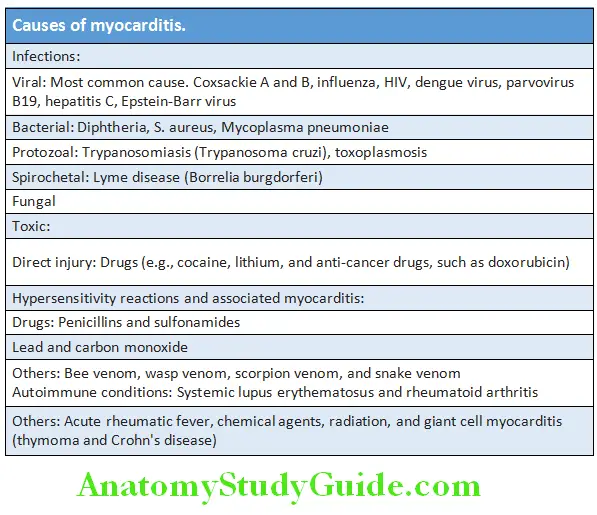
Myocarditis Investigations:
- Chest X-ray: May show mild cardiac enlargement
- ECG changes are common but nonspecific. May show standard T wave abnormalities and arrhythmias. Heart block may develop in diphtheritic myocarditis, Lyme disease, and Chagas disease.
- Cardiac enzymes: Biochemical markers of myocardial injury such as troponin I and T, creatine kinase may be raised during the early phases.
- Echocardiography: May show left ventricular dysfunction, and global hypokinesia with or without pericardial effusion
- Cardiac magnetic resonance imaging: MRI may show myocardial inflammation or infiltration. May show increased myocardial T2 signal on inversion recovery sequence and delayed contrast enhancement after gadolinium-DTPA infusion.
- Viral antibody titers: May be increased
- Endomyocardial biopsy is sometimes useful to confirm the diagnosis and may show acute inflammation. Although controversial, still the current gold-standard test for diagnosis. It should be considered when there is suspicious of giant cell myocarditis, hypersensitivity/eosinophilic myocarditis, and cardiac involvement in a systemic disease.
- Viral RNA, DNA: Genome by polymerase chain reaction (PCR) or in situ hybridization.
- Blood: Leukocytosis, elevated ESR, or eosinophilia
Complications: Ventricular arrhythmia, heart block, congestive heart failure, acute pericarditis, progression to chronic myocarditis, or chronic dilated cardiomyopathy (e.g., Chagas disease).
Myocarditis Management:
- Identify, treat, eliminate, or avoid the underlying cause.
- Prolonged bed rest during the acute phase of the illness and restriction of physical activities for 6 months (till ECG is normal). Because physical activities can induce potentially fatal ventricular arrhythmias.
- Treatment of heart failure with diuretics, ACE inhibitors/all receptor antagonists, β-blockers, spironolactone ± digoxin. Arrhythmias are treated by amiodarone and β-blockers. Digoxin should be used with caution.
- Antibiotics: Specific antimicrobial therapy if a causative organism has been identified.
- NSAIDs should not be given in the acute phase but may be given in the late phase.
- Corticosteroids and immunosuppressive agents use is controversial.
- Immunoglobulin: High-dose intravenous immunoglobulin appears to hasten the resolution of the left ventricular dysfunction and improved survival.
- Refractory patients may rarely need cardiac transplantation or temporary circulatory support (with a mechanical ventricular assist device).
Cardiomyopathy:
Question 143. Define and classify cardiomyopathy.
Answer:
Cardiomyopathy Defiition:
Cardiomyopathies are the heterogeneous group of diseases of the myocardium that affect the mechanical or electrical function of the heart.
- The term cardiomyopathy should be restricted to the conditions which primarily affect the myocardium. It does not include myocardial involvement due to congenital, acquired valvular, hypertensive, and coronary arterial or pericardial abnormalities.
- Etiology: They can be genetic/inherited or have infective, toxic causes, or idiopathic.
Cardiomyopathy Classifiation:
Cardiomyopathies may be classified according to a variety of criteria, including the underlying genetic basis of dysfunction.
Two fundamental forms of cardiomyopathy are:
1.Primary cardiomyopathy: Consists of heart muscle disease predominantly involving the myocardium and/or of
unknown cause.
2.Secondary cardiomyopathy: Consists of myocardial disease of unknown cause or cardiomyopathy associated with systemic disease (e.g., chronic alcohol use, amyloidosis).
Etiologic classification of cardiomyopathy:
Primary cardiomyopathy:
- Idiopathic (D, R, H)
- Familial (D, R, H)
- Eosinophilic endomyocardial fibrosis (R)
- Endomyocardial fibrosis (R)
Secondary cardiomyopathy:
- Infective (D): Viral, bacterial, fungal, protozoal, metazoal, rickettsial, spirochetal myocarditis.
- Metabolic (D): Familial storage disease (D, R): Glycogen
storage disease, mucopolysaccharidosis, hemochromatosis,
Fabry’s disease - Deficiency (D): Electrolytes, nutritional
- Autoimmune disease: Systemic lupus erythematosus,
polyarteritis nodosa, rheumatoid arthritis - Infiltrations and granulomas diseases (R, D): Amyloidosis, sarcoidosis, malignancy
- Neuromuscular: Muscular dystrophy, myotonic dystrophy, Friedrich’s ataxia (H, D)
- Sensitivity and toxic reaction (D): Alcohol, drugs, radiation
- Peripartum heart disease Takotsubo (stress) cardiomyopathy
Clinical Classifiation of Cardiomyopathy:
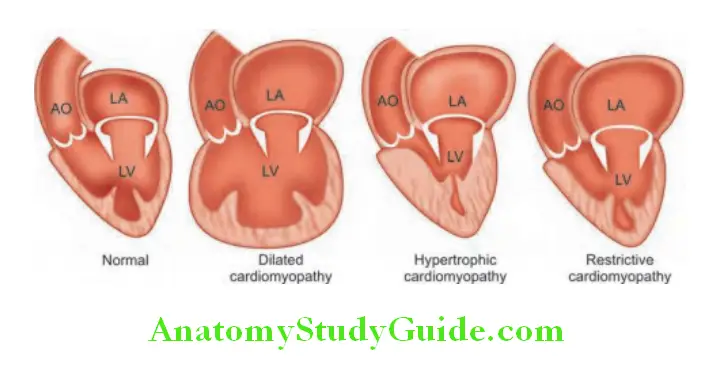
Dilated (Congestive) Cardiomyopathy (DCM):
Question 144. Write short essay/note on dilated cardiomyopathy (congestive cardiomyopathy).
Answer:
- DCM is characterized by dilatation/enlargement of the ventricular chambers and systolic dysfunction (impaired contraction of left and/or right ventricular) with preserved wall thickness.
- Left ventricular mass is increased but thickness of wall is normal or reduced. Dilatation of the valve rings may cause functional incompetence of mitral and tricuspid valves.
- Most common of all cardiomyopathies.
- Responsible for about one in three cases of heart failure and third most common cause of heart failure.
Dilated (Congestive) Cardiomyopathy Etiology:
- Familial DCM: One-fifth to one-third of patients has familial forms of DCM. It is inherited as an autosomal dominant disorder and is associated with more than 20 varieties of single-gene mutations. Most of these mutations involve genes encoding the cytoskeleton of the myocyte (dystrophin, lamin A and C, emerin, and metavinculin). Most of the X-linked inherited skeletal muscular dystrophies (e.g., Becker and Duchenne) are associated with cardiomyopathy.
- Sporadic DCM Causes include:
- Myocarditis: Coxsackie, adenoviruses, HIV, bacteria, fungal, mycobacteria, parasitic (Chagas disease)
- Toxins: Alcohol, chemotherapy (e.g., adriamycin, trastuzumab, cyclophosphamide), metals (cobalt, lead, mercury, arsenic)
- Autoimmune diseases (SLE, systemic sclerosis, dermatomyositis)
- Endocrine: Diabetes mellitus, thyrotoxicosis, hypothyroidism
- Neuromuscular: Muscular dystrophies, Friedrich’s ataxia, myotonic dystrophy
Dilated (Congestive) Cardiomyopathy Clinical Features:
- It is three times more common in men than women and the peak incidence is in middle age.
- Symptoms may be gradual in onset or may cause sudden cardiac death due to arrhythmias. Thromboembolism, conduction defects and sporadic chest pain may be present.
- Symptoms/signs of heart failure
- Fatigue and weakness
- Pulmonary congestion (left heart failure), dyspnea (rest, exertional, nocturnal), orthopnea
- Systemic congestion (right heart failure), edema, nausea, abdominal pain, nocturia
- Low cardiac output
- Hypotension, tachycardia, tachypnea
- Narrow pulse pressure
- Elevated jugular venous pressure (JVP)
- Arrhythmia: Atrial fibrillation, conduction delays, complex PVCs, sudden death
Dilated (Congestive) Cardiomyopathy Investigations:
- Chest X-ray: Generalized enlargement of heart, CHF
- Electrocardiogram: ECG shows tachycardia, conduction abnormalities (AV block, LBBB), diffuse nonspecific ST segment and T wave changes, arrhythmias (i.e., atrial fibrillation, ventricular premature contractions or ventricular tachycardia)
- 24-hour Holter monitor: If lightheadedness, palpitation, syncope
- Echocardiogram, CTI, CMRI: Show left ventricular dilation with normal or minimally thickened or, thinned walls, global hypokinesia, low EF
- Elevated BNP
- Cardiac catheterization to rule out coronary artery disease
- Myocardial biopsy, rarely necessary.
Dilated (Congestive) Cardiomyopathy Prognosis: Majority particularly >50 years die within 4 years of onset. Spontaneous improvement or stabilization occurs in 25% of cases. Death is due to progressive heart failure, ventricular tachycardia. Sudden cardiac death (SCD) is a constant threat.
Alcoholic Cardiomyopathy:
- Individuals who consumes >90 g/day of alcohol for many years.
- Clinical picture resembling idiopathic or familial DCM.
- Partially genetically predetermined (ALDH2).
- Abstention may halt the progression or may even reverse.
Peripartum Cardiomyopathy:
- Cardiac dilatation with CHF develops during last trimester or within 6 months of delivery.
- Typically present in multiparous of age >30 years.
- Unknown cause
- Inflammatory myocarditis, immune activation, multiple gestations have been incriminated.
- Symptoms, signs and management are that of IDCM
- Further pregnancy should be discouraged.
Dilated (Congestive) Cardiomyopathy Treatment:
- Control and conventional management of heart failure: Salt restriction, fluid restriction, and initiate standard treatment of heart failure. Medical therapy consists of ACE inhibitors, diuretics, digoxin and hydralazine/nitrate combination. L-carnitine, antioxidants and chelating agents have been tried.
- Anticoagulation prophylaxis
- Implantation of a cardiac defibrillator and/or cardiac
resynchronization therapy in some patients. - Cardiac transplantation in certain patients.
Hypertrophic Cardiomyopathy:
Question 145. Write short essay/note on hypertrophic cardiomyopathy (HCM).
Answer:
- HCM is characterized by left ventricular hypertrophy, typically of a nondilated chamber, without any obvious cause.
- Two significant features are:
- Asymmetric myocardial hypertrophy of the left ventricle with greater hypertrophy of the interventricular septum.
- A dynamic left ventricular outflow tract pressure gradient, related to narrowing of the sub aortic area which may produce LV outflow tract obstruction.
- It is familial and transmitted as an autosomal dominant trait.
Hypertrophic Cardiomyopathy Clinical Features:
- Asymptomatic: Echocardiographic finding only and family history may be positive.
- Symptomatic: Dyspnea in 90%. Other symptoms include effort-related such as angina and breathlessness, arrhythmia, and sudden death.
Signs:
- Rapidly rising carotid pulse (“jerky” “spike and dome”)/bisferiens pulse (two systolic peaks).
- Double apical impulse
- Reversed pulsus paradoxus on arterial pulse with Bernheim a wave in JVP.
- Harsh ejection systolic murmur best heard at the lower-left sternal border as well as the apex due to left ventricular outflow tract obstruction. This murmur increases during standing and Valsalva maneuver (which reduce ventricular preload) but decreases during squatting and sustained hand grip (which increase afterload), and also by leg rising (which increases preload).
- Pansystolic murmur at mitral area due to mitral regurgitation.
Hypertrophic Cardiomyopathy Investigations:
- Electrocardiogram: Abnormal in 85–90% of cases. Shows left ventricular hypertrophy, abnormal ST-Ts, giant T-wave inversions, abnormal Q’ waves, bundle branch block (BBB).
- Chest X-ray: Normal or shows mild-to-moderate cardiac enlargement.
- Echocardiography is diagnostic. Increased LV wall thickness ≥15 mm, systolic anterior motion (SAM) of the mitral valve and dynamic LV outflow tract obstruction.
- Genetic testing for evaluation of family members.
- Apical nonobstructive cardiac hypertrophy (Yamaguchi syndrome) is a relatively rare form of hypertrophic cardiomyopathy.
- Typical features of apical HCM include giant T-wave negativity on the electrocardiogram, especially in the left precordial leads, a “spade-like” configuration of the left ventricular cavity at end-diastole on left ventriculography.
Hypertrophic Cardiomyopathy Treatment:
- Avoid: Dehydration, digitalis, diuretics, dihydropins, and vasodilators.
- Drug therapy: Beta-adrenergic blockers, calcium-channel blockers (verapamil, diltiazem, etc.), disopyramide and antiarrhythmics (amiodarone).
- Implantable cardiac defibrillator (ICD).
- Myectomy (partial surgical resection of septum) may improve outflow tract obstruction.
- Iatrogenic infarction of the basal septum (septal ablation) using a catheter delivered alcohol solution.
- Transplantation.
Hypertrophic Cardiomyopathy Prognosis:
- Risk of SCD higher in children. Clinical deterioration usually is slow.
- Poor prognosis in males, young age of onset, family history of SCD, history of syncope, exercise induced hypotension (worst).
- Progression to DCM occurs in 10–15%.
Restrictive (Obliterative) Cardiomyopathy:
Question 146. Write short essay/note on restrictive cardiomyopathy (obliterative cardiomyopathy).
Answer:
- Rare condition in which ventricular filling is impaired because of the stiff ventricles. Hallmark is an abnormal diastolic function.
- It resemblances constrictive pericarditis, and it is important to differentiate because constrictive pericarditis is an operable disease.
- Much less common than DCM or HCM outside the tropics, but frequent cause of death in Africa, India, South and Central America and Asia primarily because of the high incidence of endomyocardial fibrosis in those regions.
Restrictive (Obliterative) Cardiomyopathy Causes:
- Idiopathic:
- Myocardial:
- Noninfiltrative: Idiopathic, scleroderma
- Infiltrative: Amyloidosis, sarcoidosis, Gaucher’s disease, Hurler disease
- Storage disease: Hemochromatosis, Fabry disease, glycogen storage diseases
- Endomyocardial: Tropical endomyocardial fibrosis, hypereosinophilic syndrome, carcinoid, metastatic malignancies, radiation, anthracycline drugs.
Restrictive (Obliterative) Cardiomyopathy Clinical Features:
- Symptoms of right and left heart failure. Exercise intolerance, dyspnea, peripheral edema, ascites, and enlarged tender liver.
- Systemic embolism may develop in about 25% cases.
- Jugular venous pressure may be raised with diastolic collapse and positive Kussmaul’s sign.
- Mild cardiac enlargement, the cardiac apex is easily palpable, and a mitral regurgitation murmur may be heard (not found in constrictive pericarditis).
- Heart sounds are soft and third and fourth heart sounds may be heard.
Restrictive (Obliterative) Cardiomyopathy Investigations:
- Electrocardiogram: Shows nonspecific ST-T wave changes, low voltage, arrhythmias
- Chest X-ray: Shows mild cardiomegaly.
- Doppler echocardiography: Abnormal mitral inflow pattern, symmetrically thickened LV walls and systolic dysfunction, prominent E wave (rapid diastolic filling), reduced deceleration time (increased left atrial pressure).
- Cardiac MRI and CT: Show symmetrically thickening of left ventricle wall, and normal or slightly reduced ventricular volumes and systolic function. These findings are useful in differentiating it from constrictive pericarditis.
- Cardiac catheterization: Shows increased ventricular filling pressures with dip-and-plateau pattern.
Restrictive (Obliterative) Cardiomyopathy Treatment:
- No satisfactory medical therapy.
- Drug therapy must be used with caution:
- Diuretics for extremely high-filling pressures.
- Chronic anticoagulation is often recommended.
- Vasodilators may decrease filling pressure.
- Calcium-channel blockers to improve diastolic compliance.
- Digitalis and other inotropic agents are not indicated.
- Transplantation may be indicated.
Stress Cardiomyopathy:
- Stress cardiomyopathy is a clinical syndrome characterized by an acute and transient (<21 days) left ventricular (LV) systolic (and diastolic) dysfunction often related to an emotional or physical stressful event, most often identified in the preceding days (1–5 days).
- First described in Japan in 1990; also called as Takotsubo cardiomyopathy, broken heart syndrome, apical ballooning syndrome.
- Prevalence is currently estimated at 1 to 2% of patients with suspected acute coronary syndrome.
- Exact pathophysiology is unknown and most accepted hypothesis is norepinephrine and NPY are stored in the presynaptic terminations of the post-ganglionic sympathetic system.
- Risk factors include female sex, menopausal, psychiatric illness, diabetes mellitus , obstructive airway disease, substance abuse disorder.
Clinical Presentation:
- Clinical presentation of typical patient with stress cardiomyopathy include postmenopausal woman who presents with acute or subacute onset of chest pain (>75%) and/or shortness of breath (approximately 50%), often with dizziness (>25%) and occasional syncope (5–10%)
- In majority of the cases, the patient has experienced an emotional or physical stress that he/she may not share with the healthcare provider unless asked.
- Physical examination reveals features of acute decompensated left-sided heart failure and a systolic murmur when there is accompanying LV outflow tract obstruction.
- Diagnosis is based on RWMA beyond territory supplied by single coronary artery with reversible in due course and associated with physical/emotional stress.
- Electrocardiogram is abnormal in >95% patients with ischemic ST segment and T wave changes. T wave inversion are usually deep and wide associated with prolonged QTc likely occurring after 24-48 hours of the precipitating factor.
Clinical Presentation Treatment:
- Management is mainly supportive therapy and prevention of complications until recovery. Treatment of heart failure with or without cardiogenic shock is as per guideline directed heart failure management.
- Anticoagulation is considered for apical thrombus (for upto 3 months) or large area of akinesis (until LVEF improves)
- Recurrence is seen in 2–4% of patients and in hospital mortality is seen upto 5% of patients.
- Recovery of LVEF is seen from 48 hours up to 6 weeks.
Congenital Heart Diseases:
Question 147. Write short note on.
Answer:
- Common congenital heart diseases seen in adults.
- Enumerate cyanotic heart diseases.
Congenital heart disease (CHD) is the most common group of structural malformations in children. CHD occurs in 8 per 1000 infants. About 1 in 10 stillborn infants have a cardiac anomaly.
Classifiation of Congenital Heart Diseases:
Classification of congenital heart diseases is summarized.
Acyanotic Congenital Heart Diseases:
Persistent Ductus Arteriosus (Patent Ductus Arteriosus):
Question 148. Write short essay on persistent ductus arteriosus (patent ductus arteriosus).
Answer:
Persistent Ductus Arteriosus Etiology:
Rubella infection during the fist trimester of pregnancy and fetal valproate syndrome is associated with a high incidence of patent ductus arteriosus (PDA). Patent (persistent) ductus arteriosus is a congenital anomaly in which the ductus arteriosus remains open after birth. Ths produces a persistent communication between the proximal left pulmonary artery and the descending aorta. Since the pressure in the aorta is higher than that in the pulmonary artery, it produces a continuous arteriovenous left-to-right shunt the volume of which depends on the size of the ductus. About 50% of the left ventricular output is recirculated through the lungs, with a consequent increase in the work of the heart. PDAs may occur as an isolated anomaly (about 90%), or associated with other abnormalities, such as ventricular septal defect (VSD), coarctation of the aorta, or pulmonary or aortic valve stenosis.
Persistent Ductus Arteriosus Clinical features:
Symptoms:
- More common in females with female-to-male ratio of about 2:1.
- When the shunt is small, then it may be asymptomatic for years.
- If the shunt is moderate-to-large, there is retardation of growth and development. It produces left heart volume overload.
Cardiac failure may develop producing dyspnea. In some cases, it may raise pulmonary artery pressure resulting in pulmonary hypertension and Eisenmenger’s syndrome.
Question 149. Write short essay on differential cyanosis.
Answer:
Persistent ductus with reversed shunting: When the pulmonary vascular resistance increases, pulmonary artery pressure may rise until it equals or exceeds aortic pressure. Then the shunt through the patent ductus arteriosus may reverse causing Eisenmenger’s syndrome. Patients with Eisenmenger’s syndrome are cyanotic and may have differential cyanosis. It is characterized by cyanosis and clubbing of the toes but not the fingers because the right-to-left ductal shunting is distal to the subclavian arteries.
Signs:
Question 150. Write short essay on cardiac findings in patent ductus arteriosus.
Answer:
- Bounding pulse: Pulses are increased in volume.
- Apex beat is shifted down and out and hyperdynamic.
- PDA produces a characteristic continuous harsh murmur known as “machinery-like”/Gibson’s murmur. It is heard with late systolic accentuation and maximal in the first left intercostal space below the clavicle and frequently accompanied by a continuous thrill at the upper-left sternal edge. However, in a large PDA when pulmonary hypertension develops the murmur becomes softer.
- First heart sound is loud (due to loud mitral component).
- Graham Steell murmur of pulmonary hypertension will be present.
Patent ductus arteriosus Investigations:
- Chest X-ray shows enlargement of the pulmonary artery with increased vascular markings (plethoric fields).
- ECG is usually normal with smaller ductal shunts. It may demonstrate left atrial enlargement, left ventricular hypertrophy, sinus tachycardia or atrial fibrillation in patients with moderate or large shunts.
- Echocardiogram and color Doppler shows PDA and the amount of blood flow through the ductus arteriosus.
- Magnetic resonance imaging and computed tomography: It can assess the degree of calcification, which is important, if surgical therapy is considered.
Complications: Cardiac failure, hypertensive pulmonary vascular disease, endarteritis, paradoxical embolism.
patent ductus arteriosus Management:
- Small ductus arteriosus may predispose to endarteritis and ductus closure should be done unless clinically silent.
- Ductus closure is indicated for any child or adult who is symptomatic from significant left-to-right shunting through the PDA.
- Transcatheter-occluding devices (e.g., coils, buttons and umbrellas) are increasingly used.
- Video-assisted thoracoscopic clip closure.
- Surgical ligation or division of the PDA remains the treatment of choice for the rare very large ductus arteriosus.
- Symptomatic patients with PDA usually improve with a medical regimen of diuretics, digoxin and angiotensin-converting enzyme inhibition, antidysrhythmic medications with anticoagulation may be useful in patients with atrial fibrillation or flutter.
- Pharmacological treatment in the neonatal period: In the first week of life, if the ductus is patent; a prostaglandin synthetase inhibitor (e.g., indomethacin or ibuprofen) may be used to induce its closure. However, if there is an impaired lung perfusion (e.g., severe pulmonary stenosis and left-to right shunt through the ductus), the ductus must be kept open with prostaglandin treatment to improve oxygenation.
Coarctation of Aorta:
Question 151. Write short essay on coarctation of aorta.
Answer:
Coarctation of the aorta (COA) is narrowing of the lumen of the aorta at the region or just distal to the insertion of the ductus arteriosus distal to (just below) the origin of the left subclavian artery.
Coarctation of Aorta Etiology: It is congenital heart disease associated with other abnormalities, such as bicuspid aortic valve (80% of cases) and ‘berry” aneurysms in the circle of Willis, Turner’s syndrome. Other lesions that may be associated include patent ductus arteriosus, ventricular septal defect and patent ductus arteriosus, mitral stenosis or regurgitation.
Types: Two classic forms.
1. Infantile (preductal) form: Characterized by tubular hypoplasia of the aortic arch proximal to a patent ductus arteriosus. It produces symptoms in early childhood.
2. Adult (postductal) form: Shows narrowing of the aorta, opposite the closed ductus arteriosus (ligamentum arteriosum) distal to the arch vessels.
Coarctation of Aorta Clinical features:
Symptoms:
- It is twice common in men compared to women.
- It may present as cardiac failure in the newborn but often asymptomatic for many years when detected in older children or adults.
- May present with headaches and epistaxis/nosebleeds due to hypertension proximal to the coarctation.
- Occasionally present with weakness or cramps/claudication in the legs and cold legs due to decreased blood flow to the lower part of the body limbs.
Signs:
Question 152. Write short note on radiofemoral delay and Suzman’s sign.
Answer:
- Pulse: Th femoral pulses and pulses in the lower limbs are weak, delayed (radiofemoral delay) in comparison with the radial pulse. If coarctation is proximal to the left subclavian artery, asynchronous radial pulses in right and left arms are observed. Prominent pulsations in the neck. In severe coarctation, pulses are poor.
- BP: It is raised in the upper limbs, but normal/low in the legs (diffrence >20 mm Hg).
- Heart sounds and murmurs:
- Ejection systolic murmur (ESM): Systolic murmur is heard posteriorly, over the spine due to coarctation.
- Systolic or continuous murmurs over lateral thoracic wall due to collaterals.
- An ejection click and systolic murmur in the aortic area, if associated with bicuspid aortic valve. Heaving apical impulse.
- Bruits: Aortic narrowing causes formation of collaterals mainly in the periscapular, internal mammary and intercostal arteries, and may produce localized bruits.
- Suzman’s sign: It is characterized by dilated, tortuous, pulsatile arteries around the scapulae and intercostals regions in the back. It is seen better when the patient bends forwards with hands hanging down.
Corkscrew-shaped retinal arteries. Absence of papilledema despite high BP.
Suzman’s sign Investigations:
- Chest X-ray: In early childhood, it is often normal. Later, it may show post-stenotic dilatation of aorta and indentation of the descending aorta at the site of the coarctation. This produces an aorta shaped like a ‘figure 3’ (due to combination of dilated left subclavian artery above, stenosed, coarcted segment in the middle and dilated poststenotic aorta below in the upper right mediastinum. Tortuous and dilated collaterals may erode the undersurfaces of the ribs producing notching of the under-surfaces of the posterior ribs (“rib notching” or “Dock’ sign”) extending from third to ninth ribs. It may be unilateral or bilateral, and found only after 6 years of age.
- CT, MRI, and CMR scanning is the best imaging method to accurately demonstrate the coarctation and quantify flow.
- ECG: May demonstrate left ventricular hypertrophy.
- Echocardiography sometimes shows the coarctation and other associated anomalies. It confirms left ventricular hypertrophy.
Complications: Hypertension, left ventricular/congestive heart failure, infective endocarditis (at the site of coarctation, bicuspid aortic valve or collateral channels), cerebral hemorrhage due to rupture of Berry aneurysm and rupture or dissection of aorta.
Suzman’s sign Treatment:
- If untreated, death may occur due to complications (e.g., left ventricular failure, dissection of the aorta or cerebral hemorrhage).
- Treat hypertension and congestive heart failure. Avoid ACE inhibitors and angiotensin II receptor antagonists because they may lead to inadequate perfusion of lower-body and may precipitate renal failure.
- Surgical correction: Intervention is needed if there is a peak–peak gradient across the coarctation of >20 mm Hg and/or proximal hypertension. In neonates surgical repair is required. In older children and adults, balloon dilatation and stenting or surgery is advisable. Recurrence of stenosis may occur as the child grows. A balloon dilatation is preferred for recoarctation (sometimes stenting).
Atrial Septal Defect:
Question 153. Write short essay on atrial septal defect.
Answer:
An atrial septal defect (ASD) is an abnormal, fixed opening in the atrial septum. It is due to incomplete formation of the atrial septum.
Types of ASD:
- Ostium secundum defects (75–85% of ASDs) are located in the region of the midseptum (fossa ovalis).
- Ostium primum (atrioventricular septal) defects (10–15%) are located in the lower portion of the atrial septum.
- Sinus venosus defects:
- Superior sinus venosus type defect (5–10%): Defects are located in the superior part of the septum near the orifice of the superior vena cava (SVC).
- Inferior sinus venosus (IVC) type defect (1%): Defects are located on the inferior part of the septum near the IVC entry point.
- Coronary sinus (1%) septal defect (in which a defect between the coronary sinus and the left atrium allows a left-to-right shunt to occur through an ‘unroofed’ coronary sinus).
Hemodynamics:
In ASD flow of blood is between the left and right atria. Normal RV is more compliant than the LV and initially a large volume of blood shunts through the atrial defect from the left atrium to right atrium and then to right ventricle. This increasesright ventricular output and markedly increased pulmonary blood flow through the pulmonary arteries. As a result, there is progressive enlargement of right atrium, right ventricle and pulmonary arteries. Eventually, pulmonary hypertension develops and sometimes reversal of the shunt from right to left (tend to occur later in life).
Atrial Septal Defect Clinical Features:
Symptoms:
- ASDs are often asymptomatic till adulthood.
- Two to three times more common in women than in men.
- Symptoms include easy fatigability, recurrent chest infection, exertional dyspnea, palpitations related to arrhythmias (especially atrial fibrillation), platypnea-orthodeoxia-dyspnea with standing, relieved by rest and cardiac failure.
- After 40 years: Deterioration due to atrial fibrillation, increased left to right shunt due to hypertension and coronary artery disease (CAD) which decrease left ventricular (LV) compliance.
Atrial Septal Defect Signs:
- Pulse: No variation in rate or volume with Valsalva, irregularly irregular pulse with AF.
- JVP:
- A and v waves have equal height that is reflection of LA waves
- A wave becomes taller when pulmonary hypertension develops or associated mitral stenosis (MS).
- Systolic pulsations in second and third left intercostals space due to dilated pulmonary artery.
- Characteristic physical signs due to the volume overload of the right ventricle:
- S2– wide fixed split: Wide, fixed splitting of the second heart sound (S2).
- Wide due to
- Increased RV ejection time (delay in right ventricular ejection) and
- increased pulmonary hangout interval.
- Fixed because the atrial septal defect equalizes left and right atrial pressures throughout the respiratory cycle.
- A systolic flow murmur over the pulmonary valve not due to atrial septal defect.
- Diastolic flow murmur over the tricuspid valve may be heard in children with a large shunt.
- S2– wide fixed split: Wide, fixed splitting of the second heart sound (S2).
- S1-loud and P2 is loud (due to increased recoil of the dilated PA and close proximity of dilated PA to chest wall).
- Ventricular heave/parasternal impulse present.
Atrial Septal Defect Investigations:
- Chest X-ray: It shows enlargement of the heart (right atrium, right ventricle) and prominent pulmonary arteries (Jug handle appearance) with pulmonary plethora. Hilar dance on fluoroscopy is characteristic.
- ECG: Incomplete right bundle branch block (RBBB due to right ventricular depolarization) and right axis deviation (due to dilatation of the right ventricle).
- Ostium primum defects may show left axis deviation.
- Echocardiography: May show hypertrophy and dilatation of the right heart and pulmonary arteries.
- Subcostal views with 2D and color Doppler demonstrates the ASD and helps in
calculation of the left right shunt (QP: QS ratio). - CMR and CT helps to assess for anomalous pulmonary venous drainage.
- MRI: Can be used to identify size and location of defect, A major advantage of MRI is the ability to quantify right ventricular size, volume, and function along with the ability to identify the systemic and pulmonary venous return.
- Cardiac catheterization: In nonrestrictive ASD, pressure gradient between atria is less than 3 mm Hg.
Atrial Septal Defect Complications: Congestive heart failure (in neonates), paradoxical embolism, atrial fibrillation. Others include pulmonary hypertension (late), Eisenmenger’s syndrome (very late), and very rarely infective endocarditis.
Atrial Septal Defect Management:
- Prompt treatment of respiratory infections.
- Indications for intervention
- ASD with significant left to right shunting resulting in right atrial/ventricular enlargement irrespective of symptoms.
- Atrial septal defects in which pulmonary flow is increased 50% above systemic flow (i.e., flow ratio of 1.5:1).
- Thromboembolic events.
- Contraindications to surgery: Severe pulmonary hypertension and shunt reversal.
- Surgical options available are:
- Median sternotomy with direct closure of small to moderate defect.
- Larger defects closed with autologous pericardium or synthetic patches like polyester polymer (Dacron) or polytetrafluoroethylene (PTFE).
- Surgical closure of the defect is done in patients above 3 years of age, provided there are no signs of pulmonary hypertension and the pulmonary flow is 50% more than the systemic blood flow (Qp:Qs>1.5/1).
- A transcatheter septal clamshell device closure may be used for most secundum ASDs (if suitable size).
- Uncorrected ASD does not require antibiotic prophylaxis for endocarditis unless other high-risk conditions are present.
Prognosis: Long-term prognosis thereafter is excellent after surgical intervention.
Patent Foramen Ovale:
- It may be found in >25% of adult population and is hemodynamically insignificant.
- Usually asymptomatic but may be associated with paradoxical emboli and increased incidence of embolic stroke.
Ventricular Septal Defect:
Question 154. Write short essay on ventricular septal defect (VSD). VSD is the most common congenital heart disease (2 per 1,000 live births).
Answer:
Ventricular Septal Defect Etiology:
- Congenital ventricular septal defect are due to incomplete septation of the ventricles. Embryologically, the interventricular septum has two portions, namely
- A membranous
- A muscular portion (which is further divided into inflow
- Trabecular and outflow portions). Most congenital ventricular defects are “perimembranous,” i.e., at the junction of the membranous and muscular portions. Congenital ventricular septal defect may be isolated or may be associated with other congenital heart disease.
- Causes of acquired ventricular septal defect: Ventricular septal rupture as a complication of acute MI, infective endocarditis or rarely from trauma/cardiac catheterization.
Hemodynamic Consequences:
Left ventricular pressure is higher than right ventricle. Therefore, blood flows from left-to-right ventricle and this causes a volume overload of the right ventricle and increased pulmonary blood flow.
The shunt volume in a VSD depends on the shunt size of thedefect and the pulmonary vascular resistance. If the defect is large (at a later stage), large volumes of blood flows through the pulmonary vasculature leading to pulmonary hypertension. When the right ventricular pressure becomes higher than left, the blood starts to shunt from right to left leading to Eisenmenger’s complex. It is characterized by cyanosis.
Ventricular Septal Defect Clinical Features:
Symptoms:
- Symptoms depend on the size of the defect and severity the shunt.
- In patients with large VSD, symptoms develop soon after birth. It includes dyspnea, repeated pulmonary infection, hepatomegaly, sweating, and failure to thrive. Irreversible pulmonary vascular disease after 1–2 years of age.
- Some children with isolated VSD develop subpulmonic stenosis: Patient not at risk of pulmonary vascular disease.
Signs:
Question 155. Write short note on Maladie de Roger.
Answer:
- Pulse pressure is relatively wide.
- Murmur:
- Blood flow from the high-pressure left ventricle to the low-pressure right ventricle during systole produces a pansystolic murmur (PSM). It is usually heard best at the left sternal edge (3rd, 4th and 5th intercostal space) and radiates all over the precordium. S1and S2 are masked by a pansystolic murmur.
- A small defect usually produces a loud PSM murmur (Maladie de Roger) presenting in older children when there is no other hemodynamic disturbance.
- Large defect produces a softer murmur, especially when the right ventricular pressure is raised. This can be detected immediately after birth, while pulmonary vascular resistance is high, or when the shunt is reversed as in Eisenmenger’s syndrome.
- Precordium is hyperkinetic with a systolic thrill at left sternal border (LSB).
- Prominent parasternal pulsation, tachypnea and indrawing of the lower ribs on inspiration.
- Mortality—27% by 20 years and 69% by 60 years.
Maladie de Roger Investigations:
- Chest X-ray: Shows cardiomegaly proportional to the volume overload. Mainly LV, LA, and RV enlargement. Increased pulmonary blood flow, PAH.
- Electrocardiogram: May show right/left or combined ventricular hypertrophy. RBBB is common. KatWachtel sign, i.e., large equiphasic QRS in V2-V4 suggestive of biventricular hypertrophy.
- Doppler echocardiography is useful to determine location of VSD, morphology of LV outflow, aortic valve involvement.
- Cardiac catheterization to assess pulmonary vascular resistance in complicated VSD.
- Complications: These include congestive heart failure, pulmonary hypertension, Eisenmenger’s syndrome, right ventricular outflow tract obstruction, infective endocarditis.
Natural History:
- Spontaneous closure occurs in about 40% of cases.
- Development of RVOT obstruction/infundibular stenosis (Gasul’s transformation) may occur in 5% of the defects. Though patient requires surgery, it prevents development of pulmonary vascular obstructive disease.
- Aortic insufficiency develops in approximately 5% of patients. This may either be related to prolapse of an aortic valve cusp into VSD or lack of support to the aortic root.
Maladie de Roger Management:
- Small VSD: No medication or surgery needed if asymptomatic. About 75–80% closes by 2 years and it needs observation.
- Cardiac failure in infancy: It is initially managed with digoxin and diuretics. If failure persists surgical repair of the defect should be performed.
- Moderate/large VSD: Repair by intervention
- Large hemodynamically significant VSD: L to R shunting with Qp/Qs ≥ 2:1, even if asymptomatic, ideally before 1 year.
- Growth failure, unresponsive to medical therapy is an indication for surgery.
- Surgical closure is contraindicated in fully developed Eisenmenger’s syndrome. These patients may be treated by heart–lung transplantation.
Eisenmenger’s Syndrome:
Question 156. Write short essay on Eisenmenger’s syndrome.
Answer:
Eisenmenger syndrome characterized by an untreated congenital cardiac defect with intracardiac communication that leads to pulmonary hypertension, reversal of flow, and cyanosis.
- It develops as a consequence of the reversal of a left-to-right shunt to a right-to-left shunt. It develops in patients with congenital heart disease, such as PDA, VSD, and ASD.
- VSD Eisenmenger is called as Eisenmenger complex.
Eisenmenger’s Syndrome Clinical Features:
Symptoms:
Dyspnea, fatigue, dizziness, and syncope.
Signs:
- Central cyanosis and clubbing: It develops due to the mixing of deoxygenated blood with oxygenated blood. It is generalized in ASD and VSD reversal, whereas it is differential (only lower limbs) in PDA with reversal.
- Signs of pulmonary hypertension and its sequelae (refer ‘pulmonary hypertension’).
- Heart sounds: S2 is loud with palpable P2.
- In ASD with reversal: S2 fixed but narrowly split
- In VSD with reversal: S2 single
- In PDA with reversal: S2 mobile but narrowly split
- Right ventricular S4 and pulmonary ejection click appear.
- Murmurs: The murmurs of the underlying cause (ASD, VSD, PDA) decrease in intensity, duration and finally disappear with the development of Eisenmenger’s syndrome. Early diastolic murmur at pulmonary area and pansystolic murmur of TR appear.
Complications: Right heart failure, infective endocarditis, pulmonary infections, severe hemoptysis, secondary polycythemia, pulmonary thrombosis with infarction, brain abscess, cerebral stroke, and ventricular arrhythmias.
Eisenmenger’s Syndrome Treatment:
- Surgical correction of underlying defect (refer treatment of ASD, VSD and PDA).
- Do not advise vasodilator therapy using calcium channel blockers. It causes systemic vasodilatation and increases right-to-left shunt.
- Long-term oxygen inhalation may be useful to relieve symptoms.
- Phlebotomy when hyperviscosity syndrome develops due to polycythemia.
- Heart-lung transplantation is the only curative treatment.
- Relief of symptoms: By drugs such as prostanoids (e.g., epoprostenol, iloprost, treprostinil), endothelin receptor antagonists (e.g., bosentan) and phosphodiesteraase-5 inhibitors (e.g., sildenafil, tadalafil).
Cyanotic Congenital Heart Diseases:
Tetralogy of Fallot:
Question 157. Write short essay on Fallot’s tetralogy or Tetralogy of Fallot (TOF).
Answer:
It is the most common congenital cyanotic heart disease in adults (75%). It consists of four features:
- Ventricular septal defect usually large and similar in aperture to the aortic orifice.
- Pulmonary stenosis: Right ventricular outflow tract obstruction mostly subvalvular (infundibular) but may be valvular, supravalvular or a combination of these.
- Overriding of dextroposed aorta
- Right ventricular hypertrophy
Presence of ASD along with TOF is known as pentalogy of Fallot.
Tetralogy of Fallot Etiology:
- It occurs in about 1 in 2,000 births and is the most common cause of cyanosis in infancy after the first year of life.
- It is due to abnormal development of the bulbar septum that separates the ascending aorta from the pulmonary artery.
Tetralogy of Fallot Pathophysiology:
- When RV and LV pressures become identical, there is little or no left to right shunt. Hence, VSD is silent.
- Right ventricle empties into pulmonary artery across pulmonic stenosis producing ejection systolic murmur. Hence, the more severe the pulmonary stenosis, more is the left-to-right shunt and less is the flow into the pulmonary artery and the ejection systolic murmur is shorter.
- When tetralogy causes elevation of right ventricular pressure, right-to-left shunting of cyanotic blood across the ventricular septal defect occurs.
- Congestive failure usually does not develop because right ventricle is effectively decompressed by ventricular septal defect.
Tetralogy of Fallot Clinical Features:
Symptoms:
- Symptoms depend on the severity of pulmonary stenosis and may present any time after birth.
- Paroxysmal attacks of dyspnea, anoxic spells, predominantly after waking up. Child cries becomes blue (cyanosis due to increased right sided pressures, resulting in a right to left shunt), lose consciousness and may develop convulsion. Frequency varies from once a few days to many attack every day.
- Squatting is common
- Fallot’s spell (“tet spell”): It is lethal, unpredictable, episodes in which the child suddenly becomes increasingly cyanosed usually during feeding, crying, fever or exercise and may become apneic and unconscious. This is because of spasm of the subpulmonary muscle (infundibular septum), systemic vasodilatation producing increased right-to-left shunting across
- VSD and acute increase right ventricular outlet obstruction.
- Progression of spell may lead to metabolic acidosis which further reduces systemic resistance and increases pulmonary vascular resistance. This can cause sudden death.
- Fallot’s spell can be relieved by increasing systemic/peripheral resistance. This diminishes (reduces) right-to-left shunting, increases systemic venous return and increases pulmonary blood flow. Example of postural maneuver that increases peripheral/systemic resistance is sitting posture/squatting (Fallot sign).
- Adults have growth retardation, fatigue and dyspnea onexertion but cyanotic spells are not usual. Secondary polycythemia due to chronic hypoxia and can produce thrombotic strokes.
Signs:
- Combination of cyanosis with a loud ejection systolic murmur: In the pulmonary area (as for pulmonary stenosis) is the most characteristic feature. The ejection systolic murmur is heard in the second and third left intercostal spaces and is due to pulmonary outflow obstruction. Intensity and duration of ejection systolic murmur is inversely proportional to severity of right ventricular outflow tract obstruction. However, cyanosis may not be seen in the newborn or in patients with only mild right ventricular outflow obstruction (“acyanotic tetralogy of Fallot”).
- Second heart sound: Pulmonary obstruction results in delayed P2 and P2 become soft or inaudible (too soft to be heard). Since P2 is inaudible, hence S2 = 2 (S2 is single loud second heart sound).
- Aorta is displaced anteriorly too, A2 becomes loud. Ascending aorta in TOF is large, results in an aortic ejection click.
- Other signs include clubbing, parasternal heave, systolic thrill.
Tetralogy of Fallot Investigations:
- Chest X-ray: Shows an abnormally small pulmonary artery, large right ventricle and a ‘boot-shaped’ heart (“Coeur en Sabot”).
- ECG: Shows right ventricular hypertrophy.
- Echocardiography: It is diagnostic and highly sensitive. It shows that the aorta is not continuous with the anterior ventricular septum.
- Cardiac catheterization is rarely required.
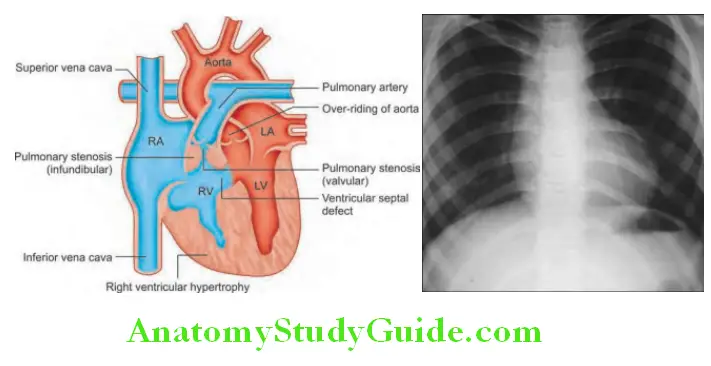
Complications These include:
- Intravascular thrombosis, cerebrovascular accidents and embolism secondary to polycythemia
- Brain abscess
- Infective endocarditis.
Ebstein Anomaly:
- Ebstein’s anomaly is a cyanotic congenital heart disease that is characterized by abnormalities of the tricuspid valve and atrialization of right ventricle. It is associated with maternal lithium consumption.
- Clinical features: Patients present with episodic tachyarrhythmia’s (associated WPW syndrome) and cyanosis. They are prone to develop paradoxical embolism, brain abscess and sudden cardiac death.
- On examination, there is central cyanosis and clubbing, prominent a waves in JVP and hepatomegaly. On auscultation, patient will have a split first heart sound, split second heart sound, S3 and S4. Pansystolic murmur of TR is present.
- ECG reveals right axis deviation, “Himalayan” giant P waves, RBBB, preexcitation pattern. CXR reveals gross cardiomegaly often called as wall to wall heart or the box heart.
- Doppler echocardiography is diagnostic.
Prognosis: After total surgical correction is good (especially if the operation is performed in childhood). About 66% live to 1 year, 49% to 3 years, and 24% to 10 years.
Tetralogy of Fallot Management:
Medical management include treatment of arrhythmias, embolism and nitric oxide. Surgical repair includes tricuspid replacement, selective plication of the atrialized right ventricle and closure of intracardiac shunt (Danielson repair, Carpentier technique)
Diseases Of The Pericardium:
Acute Pericarditis:
Question 158. Write short essay/note on
(OR)
Write short note on causes of pericarditis.
Answer:
- Acute pericarditis.
- Causes and management of pericarditis.
Acute Pericarditis Definition: Acute pericarditis is defined as an acute inflammation of the pericardium.
Acute Pericarditis Etiology:
Classifiation:
- Depending on the duration: Pericarditis is classified as acute (<6 weeks), subacute (6 weeks to 6 months) and chronic (>6 months).
- Depending on the nature of inflammation: Pericarditis may be associated with pericardial effusion and also may coexist with myocarditis. Depending on the nature of pericardial inflammation, it may be classified as serous, hemorrhagic or purulent pericarditis.
- Fibrinous pericarditis: Fibrinous exudate may heal and form varying degrees of adhesion formation.
- Serous pericarditis: Produces a large effusion of turbid, straw colored fluid with high protein content.
- Hemorrhagic pericarditis is usually due to malignant disease (e.g., carcinoma of the breast or bronchus, and lymphoma).
- Purulent pericarditis: May develop as a complication of septicemia, by direct spread from an intrathoracic infection, or from a penetrating injury.
Common causes of pericarditis:
- Idiopathic
- Infections
- Viral (Coxsackie A and B, Echovirus, HIV)
- Pyogenic (Pneumococcus, Staphylococcus, Legionella)
- Tuberculosis: Most common cause in India
- Fungal (Histoplasmosis, Candida)
- Syphilis, parasitic
- Acute myocardial infarction
- Metastatic neoplasm
- Hypothyroidism
- Radiation therapy (up to 20 years earlier)
- Chronic renal failure
- Connective tissue disorder (RA, SLE)
- Drug reaction (Procainamide, hydralazine)
- Autoimmune following heart surgery and MI (Dressler’s syndrome)
- Trauma
Acute Pericarditis Clinical Features:
Pericardial pain:
- Pericardial inflammation produces characteristic pericardial pain. It is sharp, retrosternal (central) chest pain which radiates to the shoulders and neck. Radiation of pain to trapezius muscle ridges is a feature that favors pericardial pain.
- Pain may be steady and constrictive. Typically aggravated by deep breathing, movement, and a change of position/lying down, coughing exercise and swallowing.
- Typically relieved by sitting up and leaning forward.
Nonspecifi symptoms:
- Low-grade fever, and malaise. Myocarditis may be associated with pericarditis.
- Large pericardial effusion can compress adjacent bronchi and lung and produce dyspnea (Ewart’s sign).
Pericardial friction rub:
- Pericardial friction rub is a high-pitched superficial scratching or crunching sound, produced by movement of the inflamed pericardium. It is diagnostic of pericarditis.
- Best heard by applying firm pressure with the diaphragm of stethoscope at the lower left sternal edge at the end of expiration with the patients sitting upright and leaning forward. Usually heard in systole but may also be audible in diastole.
- It has a “to-and-fro,” leathery quality. It is usually transitory, repeatedly disappearing and re-appearing.
- Three phase: Classical pericardial friction rub has three phases corresponding to movement of heart during three phases of cardiac cycle, namely
- Atrial systole
- Ventricular systole
- Ventricular diastole (rapid ventricular filling during early diastole).
- However, in some it may be heard as only one (monophasic) or two (biphasic) rubs.
Acute Pericarditis Other features:
- Pulsus paradoxus (paradoxical pulse) is characterized by weakness/disappearance of arterial pulse during inspiration.
- Cardiac tamponade: Characterized by the accumulation of pericardial fluid under pressure.
- Constrictive pericarditis: Results from scarring and consequent loss of elasticity of the pericardial sac. Typically pericardium is thicker than normal and shows chronic inflammation, sometimes with calcification.
- Effusive-constrictive pericarditis: Characterized by constrictive physiology with a coexisting pericardial effusion, usually with tamponade.
Acute Pericarditis Investigations:
- Blood: Leukocytosis (bacterial pericarditis) or lymphocytosis (viral pericarditis) and raised erythrocyte sedimentation rate (ESR).
- Cardiac enzymes (CPK-MB, troponin T): Normal unless associated with myocarditis.
- Electrocardiogram: ECG is diagnostic and changes may be found over the affected area, which may be widespread.
- These are as follows:
- Widespread concave upwards (saddle shaped/smiling face) ST elevation in multiple leads (particularly leads I, II, aVL, aVF, and V1-V3) and reciprocal ST depression in leads aVR and V.
- PR segment depression is a characteristic feature of acute pericarditis.
- The above changes evolve over time and later followed by resolution of the ST elevation, T wave flattening/inversion (due to myocarditis) and finally T wave normalization.
- Finally ECG becomes normal.
- When there pericardial effusion QRS voltage is reduced. Electrical alternans is seen with effusion.
Diffrential diagnosis:
- Myocardial infarction: The early ECG changes observed in pericarditis should be differentiated from the ST elevation observed in myocardial infarction (limited to the infarcted area, e.g., anterior or inferior). Most reliable feature may be the ratio of ST segment elevation (in millimeters) to T-wave amplitude in lead V6; ratio >0.24.
- Chest X-ray:
- May be normal in pericarditis without effusion. Stenciled borders of heart.
- Rapid increase in the size of the cardiac shadow (cardiomegaly-pear-shaped) may be seen in pericarditis with effusion. (money bag/water-bottle appearance).
- Oreo cookie sign/epicardial fat pad sign can be seen on lateral X-ray chest.
- Echocardiography used for confirmation of the pericardial effusion.
- CT and cardiac MR may be useful when there is thickening (>4 mm) or inflammation of pericardium and myocardium.
- Paracentesis: Diagnostic paracentesis is performed when there is pericardial effusion.
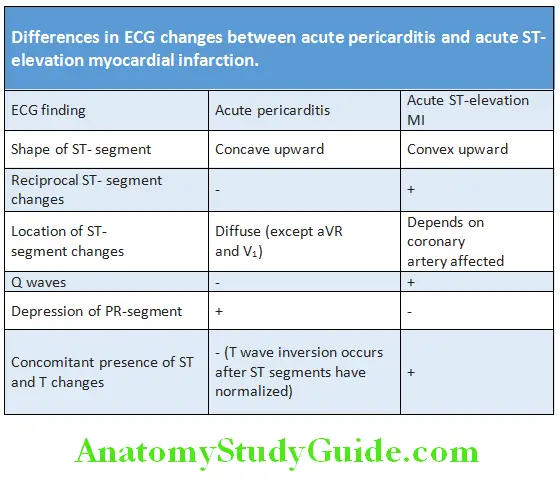
Question 159. Write short note on management of pericarditis.
Answer:
Pericarditis Treatment:
- Treatment of the underlying cause (e.g., tuberculosis, uremia, autoimmune disease).
- Bed rest and avoid physical activity.
- Analgesics: The pain is usually relieved by oral NSAIDs (high dose aspirin indomethacin or ibuprofen). It also decreases the inflammation in idiopathic or viral pericarditis.
- Aspirin in the dose of 600 mg 4 hourly is the drug of choice for patients with a recent myocardial infarction. Ibuprofen 300–800 mg 3 times daily may also be used. They can be given till pain and pericardial effusion disappears and ESR, CRP values return normal.
- It usually takes 7 to 10 days.
- Indomethacin is a more potent anti-inflammatory drug and can be given in the dose of 25 mg 3 times daily. It should be avoided in elderly patients because it can decrease the coronary blood flow.
- Colchicine 0.5 mg twice a day is also effective in combination with NSAIDs.
- Corticosteroids (10–30 mg/day for 2–4 weeks) should be reserved for patients with an immune cause or patients who do not respond to NSAIDs. Because their use is associated with an increased rate of recurrence. Colchicine or corticosteroids reduce the symptoms but does not hasten the cure.
- Purulent pericarditis: Antimicrobial therapy, pericardiocentesis and surgical drainage (if required).
- Indications for hospitalization: Fever (38°C), leukocytosis, large pericardial effusion, lack of response to NSAIDs after 1 week of therapy acute trauma, cardiac tamponade, immunosuppressed state, recurrent pericarditis.
Pericardial Effsion:
Question 160. Write short essay/note on pericardial effusion and its causes.
Answer:
Pericardial Effsion Definition: Pericardial effusion is an accumulation of fluid within the potential space of the serous pericardial sac.
- Slowly developing pericardial effusions may be asymptomatic. If it develops over a short period, and volume of effusion is large, it may lead to compromisation of ventricular filling leading to embarrassment of the circulation. This is known as cardiac tamponade.
- Type: Pericardial effusion may be transudate (hydropericardium), exudate (pyopericardium) or hemopericardium.
- Etiology: It commonly develops during an episode of acute pericarditis.
Pericardial Effsion Clinical Signs:
Symptoms commonly reflect the underlying pericarditis. Sometimes, a sensation of retrosternal oppression may be present.
Signs:
- Cardiovascular examination is normal except if the effusion is large, apex beat/impulse is not palpable (sometimes palpable medial to the left border of cardiac dullness).
- Increase in cardiac dullness on percussion.
- Heart sounds are faint, soft, distant or muffled.
- A pericardial friction rub due to pericarditis may be audible in the early stages. It becomes quieter as fluid accumulates and pushes the layers of the pericardium apart.
- Ewart’s sign: Rarely, large effusion may compress the base of the left lung. It produces an area of dullness to percussion and tubular breath sounds on auscultation in the left axilla or left base (below the angle of the left scapula) and termed as Ewart’s sign.
Investigations:
Question 161. Write short note on radiological findings in pericardial effusion.
Answer:
- Chest X-ray: Shows increased size of the cardiac silhouette, large globular or pear-shaped heart or water-bottle appearance with sharp outlines and lucent pericardial fat lines. Typically, the pulmonary veins are not distended Oreo cookie sign/ Epicardial fat pad sign.
- Electrocardiogram: ECG often shows low voltage QRS complexes (<0.5 mV in limb leads) in the presence of large effusions with sinus tachycardia. Electric alternans characterized by alternation of QRS amplitude or axis between beats) due to a to-and-fro motion of the heart within the fluid-filled pericardial sac may be observed.
- Echocardiography is the most useful investigation for demonstrating the pericardial effusion. It is also useful to monitor the size of the effusion and its effect on cardiac function.
- Cardiac CT or MRI is advisable if loculated pericardial effusions are suspected (postcardiac surgery).
- Pericardial aspiration (pericardiocentesis): It is aspiration of pericardial fluid effusion with aseptic technique under echocardiographic guidance. A needle is inserted medial to the cardiac apex or below the xiphoid process, directed upwards toward the left shoulder. It is indicated for diagnostic purposes (e.g., tuberculous, malignant or purulent effusion) or for the treatment of cardiac tamponade. Complications of pericardiocentesis include arrhythmias, damage to myocardium and coronary vessels, air embolism, and pneumothorax. The pericardial fluid is can be sent for investigations, such as:
- Cell counts, protein, glucose and LDH to differentiate exudates from transudates, Gram’s stains and AFB stain.
- Cytology for malignant cells
- Mycobacterium culture
- Pericardial biopsy may be advisable if when tuberculosis is suspected and pericardiocentesis is not diagnostic.
- Other tests: Depending on the underlying causes, e.g., blood cultures, autoantibody screen.
Pericardial effusion Treatment:
- Treat the underlying cause if possible.
- Anti-inflammatory drugs (aspirin or indomethacin). Most effusions resolve spontaneously, but rapid effusion may produce cardiac tamponade.
- Therapeutic pericardiocentesis: Indicated to relieve the pressure and a pigtail catheter drain may be left in temporarily to allow sufficient release of fluid.
- Pericardial fenestration: If pericardial effusions accumulate (e.g., malignancy), it may be treated by pericardial fenestration. This procedure consists of creating a window in the pericardium to allow the slow release of fluid into the surrounding tissues. It may be performed either transcutaneously under local anesthetic or by a conventional surgical approach.
- Intrapericardial instillation of chemotherapeutic agents may be useful in malignant effusion.
Cardiac Tamponade:
Question 162. Write short essay/note on clinical features, signs and management of pericardial tamponade.
Answer:
- Cardiac tamponade is the term used for acute heart failure that results from large or rapidly developing pericardial effusion which compresses the heart and impair diastolic filling.
- Minimum amount of pericardial fluid necessary for the development of cardiac tamponade depends on the speed of its accumulation. About 250 mL in rapidly developing effusions and more than 2000 mL in slowly developing effusions.
Cardiac Tamponade Etiology: Pericardial effusion.
Cardiac Tamponade Clinical Features:
Symptoms:
- Due to reduced cardiac output: Dyspnea, orthopnea, substernal chest discomfort radiating to neck and jaw.
- Due to systemic venous congestion: Pain in the right upper quadrant and pedal edema in slowly developing cardiac tamponade (subacute tamponade).
Signs:
- Friedreich’s sign: Markedly raised jugular venous pressure with sharp rise and y descent.
- Pulsus paradoxus or paradoxical pulse: An exaggeration in the normal variation in pulse pressure seen with inspiration. There is drop in systolic blood pressure of ≥10 mm Hg. Pulsus paradoxus is the hallmark of cardiac tamponade.
- Kussmaul’s sign: Rise in JVP/increased neck vein distension during inspiration.
- Others: Reduced cardiac output, hypotension, tachycardia, and oliguria.
- Beck’s triad of hypotension, muffled heart sounds, and elevated jugular venous pressure remains a useful clue to the presence of severe tamponade.
Cardiac Tamponade Investigations:
- Chest X-ray: Discussed earlier in pericarditis.
- Electrocardiogram: May show sinus tachycardia, reduction in QRS voltage, nonspecific ST-T changes and electrical alternans (alternation of QRS complex amplitude or axis between beats).
- Echocardiography: A pericardial effusion appears as a lucent separation between parietal and visceral pericardium and shows separation visceral and parietal layers for the entire cardiac cycle.
- Small effusions are first evident over the posterobasal left ventricle.
- Early diastolic collapse of the right ventricle and collapse of the right atrium (which occurs during ventricular diastole) are sensitive and specific signs.
- Right atrial collapse is considered more sensitive.
- RV collapse more specific for tamponade.
Cardiac Tamponade Management:
- Emergency pericardiocentesis is necessary.
- If patient is hypotensive, as a temporary measure expansion of blood volume by saline, blood, plasma, and dextran.
- Avoid positive pressure mechanic ventilation in acute tamponade because it reduces cardiac filling further.
- Treatment of underlying cause.
Chronic Constrictive Pericarditis:
Question 163. Write short essay/note on chronic constrictive pericarditis.
Answer:
- Constrictive pericarditis is an end stage inflammatory process involving pericardium.
- It is characterized by progressive thickening, fibrosis, and calcification of the pericardium and visceral and parietal pericardium usually becomes adherent resulting in obliteration of the pericardial space. In some cases, the constricting process is formed by the visceral pericardium (epicardium) alone.
Pathophysiology: The heart is encased in a solid shell and cannot fill properly. The calcification may extend into the myocardium and may impair myocardial contraction. Following features develop:
- Restricted diastolic filling of the heart by fibrotic pericardium.
- Limitation of venous return to the heart → reduced ventricular filling → inability to maintain adequate preload.
- Filling pressures of the heart tend to become equal in both the ventricles and the atria.
- Systolic function is rarely affected until late in the course of the disease.
- Preservation of myocardial function in early diastole aids in distinguishing constrictive pericarditis from restrictive cardiomyopathy.
Chronic Constrictive Pericarditis Etiology Clinical Features:
The symptoms and signs are due to:
- Reduced ventricular filling which is similar to cardiac tamponade (i.e., Kussmaul’s sign, Friedreich’s sign, pulsus paradoxus.
- Systemic venous congestion: Symptoms consistent with CCF (especially rightsided heart failure), such as ascites, dependent edema, hepatomegaly and raised JVP.
- Reduced cardiac output: Inability of the heart to increase stroke volume produces fatigue, hypotension, and reflex tachycardia.
- Rapid ventricular filling: Pericardial knock (occurs 0.09–0.12 second after A2) can be heard in early diastole at the lower left sternal border.
- Rarely pulmonary venous congestion causing dyspnea, cough, orthopnea.
Causes of constrictive pericarditis:
- Idiopathic
- Infectious:
- Tuberculosis
- Viral especially Coxsackie B
- Bacterial
- Fungal
- Parasitic
- Postradiotherapy
- Postcardiac surgery
- Post-traumatic
- Neoplastic
- Connective tissue diseases (e.g., rheumatoid arthritis and SLE)
- Toxic/metabolic: Uremia, chylous pericardium, methysergide
- Post-myocardial infarction
- Familial
Chronic Constrictive Pericarditis Investigations:
- ECG: Rarely normal, nonspecific and highly variable. Atrial arrhythmias are frequent, with atrial fibrillation occurring late in course. Low voltage (<50% cases) and LA enlargement (19–37%).
- Chest X-ray: Pulmonary venous congestion and pleural effusions (late in the disease). Calcified pericardium is highly suggestive of constrictive pericarditis when present in a patient with constrictive/ restrictive physiology.
- Echocardiography: Pericardial thickening better seen through transesophageal echo, normal RV and LV chamber size, LA and RA enlargement, abnormal septal and posterior wall motion, paradoxical septal motion, premature opening of the pulmonic valve and dilated IVC without respiratory variation.
- Magnetic resonance imaging/CT scan can confirm pericardial thickening. Pericardial thickening >4 mm assists in differentiating constrictive disease from restrictive cardiomyopathy, and thickening >6 mm adds even more specific for constriction. Normal pericardial thickness does not exclude pericardial constriction, and the clinical situation must always be taken in account.
- Cardiac catheterization: Ventricular pressure initially decreases rapidly (steep y descent on RA pressure waveform tracings) and then increases abruptly to a level that is sustained until systole (the “dip-and-plateau waveform” or “square root sign” seen on RV or LV pressure waveform tracings).
Chronic Constrictive Pericarditis Treatment:
- Pericardiectomy (resection of the pericardium) is the only definitive treatment.
- Pharmacologic therapy
- Steroids for subacute constrictive pericarditis (before pericardial fibrosis occurs).
- Diuretics to relieve congestion and optimize clinical volume status (may decrease preload to the point of reducing cardiac output).
- Treatment of the causative disease (e.g., antituberculosis medication).
- Avoid beta-blockers and calcium channel blockers (CCBs).
Pulmonary Hypertension:
Question 164. Write short essay/note on pulmonary hypertension.
Answer:
Pulmonary Hypertension Definition (WHO):
- Pulmonary hypertension (PH) is defined as an increase in blood pressure in pulmonary circulation (either in the arteries, or both in arteries and veins).
- Normal pressure as measured at right heart catheterization is 14–18 mm Hg at rest and 20–25 mm Hg on exercise. Hemodynamically it is defined as an increase in mean pulmonary arterial pressure to 25 mm Hg at rest.
- Definition may be refined by giving consideration of the pulmonary wedge pressure; the cardiac output and the transpulmonary pressure gradient (mean PAP mean PWP).
WHO Classifiations of Pulmonary Hypertension:
WHO classification of pulmonary hypertension has been shown in Table:
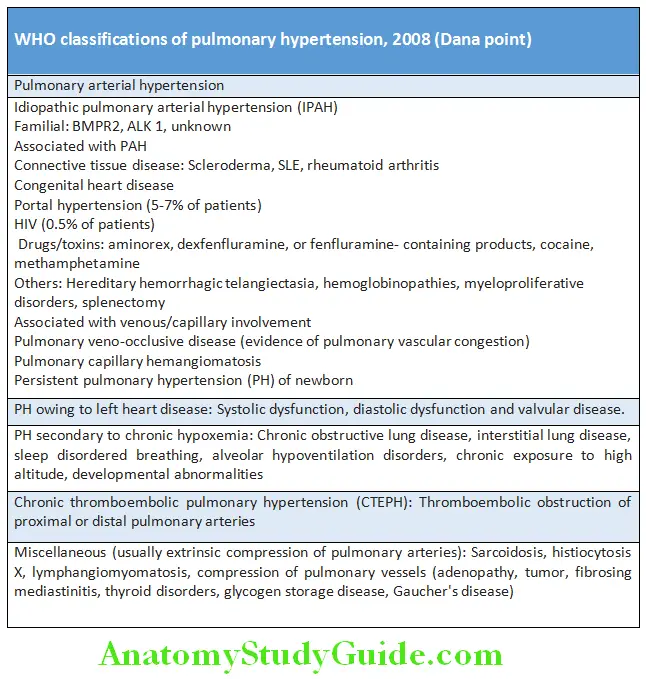
Question 165. Write short essay/note on causes of pulmonary hypertension.
Answer:
Pulmonary hypertension Clinical Features:
Symptoms: Insidious in onset and is usually diagnosed late. Usual symptoms include exertional breathlessness/dyspnea (60%), fatigue (19%), palpitation and near syncope/syncope (13%), chest pain (7%), palpitations (5%), LE edema (3%), and hoarseness of voice (2%).
Signs:
Pulmonary hypertension These include:
- Cold extremities, peripheral edema, cyanosis, and rarely clubbing
- Raised jugular venous pulse, prominent V wave if tricuspid regurgitation is present
- Left parasternal lift/heave due to right ventricular hypertrophy.
- Large pulsatile liver
- Auscultatory signs in pulmonary hypertension
- Signs of underlying cause, e.g., interstitial lung disease or cardiac, liver, or connective tissue disease.
Pulmonary hypertension Investigations:
- Chest X-ray
- Enlargement of the pulmonary artery and its main branches
- Peripheral “pruning” of vascular shadows
- Enlarged right atrium, and right ventricle
- Findings of underlying lung or cardiac pathology
- Electrocardiogram: Shows right axis deviation, R/S wave ratio greater than one in lead V1, right bundle branch block (incomplete or complete) and increased P wave amplitude in lead II (due to right atrial enlargement).
- Echocardiography: Most useful for detecting pulmonary hypertension and excluding cardiac disease. It shows tricuspid regurgitation, right atrial and ventricular hypertrophy, flattening of interventricular septum, small LV dimension, dilated pulmonary artery, and secondary causes (if any).
- Transthoracic echocardiography: Doppler assessment of the tricuspid regurgitant jet is a noninvasive method of determining the pressure of pulmonary artery.
- Right heart catheterization: Mean PAP pressure at rest >25 mm Hg, with exercise >30 mm Hg, wedge pressure <15 mm Hg.
- Other tests:
- Complete blood counts, prothrombin time, partial thromboplastin time, and liver function tests.
- Autoantibodies if autoimmune disease is suspected.
- HIV-ELISA if the patient has risk factors.
- Arterial blood gas to exclude hypoxia and acidosis as contributors to pulmonary hypertension.
- Sleep studies if sleep apnea suspected.
- Pulmonary function tests to establish airflow obstruction or restrictive lung disease.
- High-resolution computed tomography of chest to exclude occult interstitial lung disease.
- Helical CT to detect pulmonary thromboembolism.
- Ventilation-perfusion scanning to differentiate chronic thromboembolism from primary pulmonary hypertension.
Auscultatory signs in pulmonary hypertension:
- Pulmonary ejection sound (ES)
- Abnormal second heart sound (S2): Loud P2, narrow splitting
- Right atrial fourth heart sound (S4)
- Right ventricular third heart sound (S3)
- Pulmonary ejection systolic murmur (ESM)
- Pulmonary early diastolic murmur (EDM): Graham Steel murmur
- Tricuspid pansystolic murmur (PSM)
Pulmonary hypertension Treatment:
- Early recognition and treatment of the underlying cause.
- Medical: Diuretics mainly to treat edema from right heart failure, digoxin (for atrial tachyarrhythmias), oxygen (keep oxygen saturation ≥90%) and anticoagulants (IPAH) to keep INR 2.0–2.5.
- PAH-specific therapy:
- Calcium channel blockers: Indicated in patients who respond to vasodilators during catheterization. High doses required, e.g., nifedipine 240 mg/day, or amlodipine, 20 mg/day.
- Oral endothelin receptor antagonists (ERAs): For example, bosentan, sitaxsentan, ambrisentan. Liver function be monitored
monthly throughout the duration of use.- Bosentan initiated at 62.5 mg BD for first month and increased to 125 mg BD.
- Ambrisentan initiated as 5 mg OD and can be increased to 10 mg daily.
- Phosphodiesterase type 5 inhibitors (PDE 5-I): For example, sildenafil, tadalafil, vardenafil. The most common side effect is headache. Effective dose for sildenafil is 20–80 mg TID and for tadalafil is 40 mg OD.
- Prostaglandins: For example, epoprostenol (prostacyclin), treprostinil, iloprost, prostacyclin 1P receptor antagonist (Selexipag).
- Guanylate cyclase stimulant: For example, riociguat.
- Pneumococcal and influenza vaccination.
- Surgical therapy:
- Atrial septostomy (the creation of a right-to-left shunt).
- Lung transplantation
- Pulmonary thromboendarterectomy for chronic proximal thromboembolic pulmonary hypertension.
Sudden Cardiac Death:
Question 166. Write a short note on sudden cardiac death.
Answer:
Sudden cardiac arrest (SCA) and SCD refer to the sudden cessation of cardiac activity with hemodynamic collapse.
- Sudden cardiac death is defined as an unexpected, nontraumatic, natural death due to cardiac causes occurring in a short period (within 1 hour of symptom onset) in an individual with or without any previously identified heart disease but in whom the time and mode of death are unexpected.
- It has four temporal elements:
- Prodromes
- Onset Of Terminal Event
- Cardiac Arrest
- Biologic death.
- Bimodal age distribution with one peak between birth and 6 months of age and another after 65 years of age. It is a male preponderance.
Sudden Cardiac Death Causes:
Sudden Cardiac Death Treatment:
- Identifying individuals at high risk of SCD: Combination of factors more useful, most important parameter is left ventricular ejection fraction (LVEF).
- Pharmacological agents
- Beta blockers, ACEI, amiodarone
- Revascularization
- ICD/CRT.
Causes of sudden cardiac death:
- Coronary artery disease: Coronary atherosclerosis, developmental anomalies, coronary artery embolism, others (e.g., vasculitis, dissection)
- Myocardial diseases: Cardiomyopathies, myocarditis and other infiltrative processes, right ventricular dysplasia
- Valvular diseases: Mitral valve prolapse, aortic stenosis and other forms of left ventricular outflow obstruction, endocarditis
- Conduction system abnormalities: Wolff-Parkinson-White (WPW) syndrome, Brugada syndrome, long QT syndromes
Cardiac Arrest:
Cardiac Arrest Definition: Cardiac arrest is a sudden loss of cardiac pump function which can reversed by a prompt intervention, without which it lead to death.
Causes of Cardiac Arrest:
Question 167. Write short note on causes of cardiac arrest.
Answer:
Diagnosis of cardiac arrest (TRIAD):
- Loss of consciousness
- Loss of apical and central pulsations (carotid, femoral)
- Apnea.
ECG: Three basic patterns
1. Ventricular tachyarrhythmia: Ventricular fibrillation (VF)/sustained type of pulseless ventricular tachycardia.
2. Ventricular asystole or a bradyasystolic rhythm with an extremely slow rate.
3. Pulseless electrical activity (PEA) previously referred to as electromechanical dissociation.
Causes of cardiac arrest (6 H and 4 T):
- Hypoxia
- Hypotension
- Hypothermia
- Hypoglycemia
- Acidosis (H +)
- Hyperkalemia (electrolyte disturbance)
- Cardiac Tamponade
- Tension pneumothorax
- Thromboembolism (pulmonary, coronary)
- Toxicity (e.g., digoxin, local anesthetics, insecticides)
Question 168. Write short essay/note on management of cardiac arrest.
Answer:
Cardiac arrest Management:
Chain of Survival:
It refers to the sequence of events that is required to maximize the chances of survival in a patient with cardiac arrest.
Survival is most likely if all links in the chain are strong. The chain of survival consists of following links, namely
- Immediate identification of cardiac arrest and activation of the emergency response system (ERS) by a trained individual.
- Immediate cardiopulmonary resuscitation (CPR) with chest compressions.
- Quick defibrillation.
- Effective advanced life support (ALS).
- Integrated post-cardiac arrest care.
Immediate Identification and Activation of ERS:
- Immediate identification of cardiac arrest: Assessment is of crucial importance.
- It includes:
- Unresponsiveness (Check The Individual For A Response, Gently Shake Shoulders And Ask ‘Are You All Right?’),
- No Breathing Or No Normal Breathing (I.E. Only Gasping) And
- No pulse felt within 10 seconds.
Activation of ERS: After activation of the ERS, all rescuers should immediately begin CPR.
Question 169. Write short essay/note on cardiopulmonary resuscitation (CPR).
Answer:
CPR:
It provides artificial ventilation and perfusion to the vital organs, particularly heart and brain until spontaneous cardiopulmonary function is restored. It consists of both basic life supports (BLS) and advanced life support (ALS). BLS provides adequate oxygen and perfusion to vital organs (brain and heart) until advanced cardiac life support is available

BLS consists of maneuver purpose of which is to maintain a low level of circulation until more definitive treatment with advanced life support can be provided.
- Change from A-B-C to C-A-B: CPR includes four sequential: Circulation, Airway and Breathing (CAB) and defibrillation. Previously, the sequence used to be Airway, Breathing and Circulation (ABC).
- Circulation: The brain cannot survive for more than 3 minutes without circulation. Hence, start chest compressions immediately for a patient without central pulsations.
- Chest compressions (cardiac massage):
- Place the patient on a hard surface (wooden board).
- The palm of one hand is placed in the concavity of the lower half of the sternum 2 figers above the xiphoid process (avoid xiphisternal junction → fracture and injury). The other hand is placed over the hand on the sternum.
- Shoulders should be positioned directly over the hands with the elbows locked straight and arms extended. Use your upper body weight to compress.
- Sternum must be depressed at least 5 cm in adults, and 2–4 cm in children, 1–2 cm in infants.
- Push hard and push fast. Must be performed at a rate of 100–120/min.
- During CPR the ratio of chest compressions to ventilation should be: single rescuer = 30:2 and in the presence of 2 rescuers, chest compressions must not be interrupted for ventilation.
- Chest compressions must be continued for 2 minutes before reassessment of cardiac rhythm (2 minutes = equivalent to 5 cycles 30:2).
- Assessment of the adequacy of chest compressions: Systolic BP = 60–80 mm Hg, diastolic BP (>40 mm Hg) and COP = 30% of normal.
- Complications of chest compressions: Fractures of rib/sternum, rib separation, pneumothorax, hemothorax, contusions of lung, lacerations of liver, fat emboli.
- Airway: Loss of consciousness usually produces obstruction of airway due to loss of the muscle tone in the airway and falling back of the tongue. Hence, clear the airway. Basic techniques for airway patency:
- Head tilt, chin lift: Place one hand on the forehead and the other on the chin. The head is tilted upwards to displace the tongue anteriorly.
- Jaw thrust method: In this angles of mandible are grasped with both hands and the mandible is lifted forward.
- Finger sweep: Sweep out foreign body in the mouth by index finger in unconscious patients and not in a conscious or convulsing patient.
- Heimlich maneuver: Useful to remove the foreign body in a conscious patient. It is done while the patient is standing up or lying down. In this subdiaphragmatic abdominal thrust elevates the diaphragm and expels a blast of air from the lungs that displaces the foreign body. In infants this is performed by a series of blows on the back and chest thrusts.
- Breathing: Rescue breathing can be mouth-to-mouth breathing or mouth-to-nose breathing (if there is serious injury of the mouth or it cannot be opened). With the airway open (using the head-tilt, chin-lift maneuver), pinch the nostrils shut for mouth-to-mouth breathing and cover the person’s mouth with rescuer making a seal.
- Mouth-to-mouth breathing: With the airway held open, pinch the nostrils closed, take a deep breath and seal your lips over the patients mouth. Blow steadily into the patient’s mouth watching the chest rise as if the patient was taking a deep breath. Volume of each rescue breath should produce visible chest rise.
- Mouth-to-nose breathing: Seal the mouth shut and breathe steadily through the nose.
- Mouth-to-mouth and nose: It is used in infants and small children.
- Assessment of restoration of breathing and circulation: Contraction of pupil, improved color of the skin, free movement of the chest wall, swallowing attempts and struggling movements.
- Indications for termination of BLS: Pulse and respiration returns, emergency medical help arrives, physician declared patient is deceased, in a nonhealth setting, another indication to stop BLS would be that the rescuer was exhausted and physically unable to continue to perform BLS.
- ALS: The purpose is to restore normal cardiac rhythm by defibrillation when the cause is tachyarrhythmia, or to restore cardiac output by correcting other reversible causes of cardiac arrest.
- It includes:
- Circulation by cardiac massage.
- Airway management by equipment.
- Breathing by advanced techniques.
- Defibrillation by manual defibrillator.
- Drugs.
Advanced techniques for airway patency:
- Face mask,
- Oropharyngeal Airway
- Nasopharyngeal Airway
- Laryngeal Mask Airway
- Endotracheal Intubation
- Combitube
- Cricothyrotomy
- Tracheostomy.
Advanced breathing: Expired air contains 16% O2 so supplemental 100% O2 should be used as soon as possible. Successful breathing is achieved by delivery of a tidal volume of 800–1,200 mL in adults at a rate of 10–12 breaths/min in adults.
Advanced techniques include:
- Self-inflating resuscitation bag (Ambu bag), Mechanical ventilator or in ICU. Advanced circulation
- It consists of continuation of chest compression and establishing an intravenous access, attaching a cardiac monitor/defibrillator, assessing the rhythm, defibrillation and administering appropriate drugs for rhythm as well as the condition.
- Continuous chest compression is performed at the rate of 100/minute and ventilation is provided at 8–10 breaths/minute (1 breath/6–8 seconds).
Rhythm in cardiac arrest It may be:
- Shockable rhythm (ventricular tachycardia/ventricular fibrillation)
- Nonshockable rhythm (asystole and pulseless electrical activity).
Rhythm checks should be performed done only after 2 minutes of CPR and not immediately following a defibrillation attempt.
Shockable rhythms: Ventricular fibrillation or pulseless ventricular tachycardia (VT).
Defibrillation: Completely depolarize all myocardial cells so SA node can re-establish as pacemaker. Voltage of electricity discharge High from 150 J to 360 J (biphasic) 360 J (monophasic). Continue CPR for 2 minutes and briefly check the monitor for rhythm. If VT/VF persists give second shock and immediately resume CPR and continue for 2 minutes, and repeat this cycle.
Drugs:
- Adrenaline: Given as a vasopressor α-1 effect (not as an inotrope). Dose is 1 mg (0.01 mg/kg) IV every 4 minutes (alternating cycles) while continuing CPR.
- Given:
- Immediately in nonshockable rhythm (non-VT/VF),
- In VF or VT given after the 3rd shock.
- Repeated in alternate cycles (every 4 minutes).
- Once adrenaline → always adrenaline.
- Given:
- Amiodarone: Given in shockable rhythm after the 3rd shock. If unavailable give lidocaine 100 mg IV (1–1.5 mg/kg). Dose is 300 mg IV bolus (5 mg/kg).
- Vasopressin (ADH): 40 IU single dose once.
- Magnesium: Given
- VF/ VT with hypomagnesemia
- Torsade de pointes
- Digoxin toxicity. Dose is 2 g IV.
- Calcium: Dose is 10 mL of 10% calcium chloride IV.
- Indications: PEA caused by hyperkalemia, hypocalcemia, hypermagnesemia, and overdose of calcium channel blockers.
- Calcium solutions and NaHCO3 should not be given simultaneously by the same route.
- Thrmbolytics: Fibrinolytic therapy is considered when cardiac arrest is caused by proven or suspected acute pulmonary embolism. If a fibrinolytic drug is used in these circumstances consider performing CPR for at least 60–90 minutes before termination of resuscitation attempts. Example: Alteplase, tenecteplase (old generation streptokinase).
- Sodium bicarbonate: Used in
- Severe Metabolic Acidosis (Ph < 7.1)
- Life-Threatening Hyperkalemia
- Tricyclic antidepressant overdose.
- Dose: (Half correction) 1/2 base deficit × 1/3 body weight
- Adverse drug reactions:
- Increases CO2 load,
- Inhibits Release Of O2 To Tissues,
- Impairs myocardial contractility
- Causes hypernatremia.
- Atropine: Its routine use in pulseless electrical activity (PEA) and asystole is not useful. Indicated in sinus bradycardia or AV block causing hemodynamic instability. Dose is 0.5 mg IV. Repeated up to a maximum of 3 mg (full atropinization).
Nonshockable rhythms: Pulseless electrical activity (PEA) and asystole.
- PEA is characterized by cardiac electrical activity in the absence of any palpable pulse. They usually have some very weak mechanical myocardial contractions and does not produce a detectable pulse.
- Start CPR. Begin with chest compressions, and continue for 2 minutes before the rhythm check is repeated.
- Give 1 mg adrenaline IV immediately and re-check rhythm after 2 minutes of CPR.
- If PEA or asystole persists, continue CPR and recheck rhythm every 2 minutes. Administer adrenaline every 3–5 minutes. Do not give atropine.
- Check rhythm. If it shows change, check for pulse. If pulse appears, start post-resuscitation care. If there is still no pulse, continue CPR with rhythm check every 2 minutes and adrenaline every 3–5 minutes. If the rhythm develops into VF/VT, defibrillate the patient.
IV fluids: Infuse fluids rapidly if hypovolemia is suspected. Use normal saline (0.9% NaCl) or Ringer’s solution. Avoid dextrose which is redistributed away from the intravascular space rapidly and causes hyperglycemia which may worsen neurological outcome after cardiac arrest. Dextrose is indicated only if there is documented hypoglycemia.
Postresuscitation care:
- Maintain adequate airway and support breathing.
- Continue cardiac monitoring.
- Vasoactive medications (norepinephrine, dobutamine and epinephrine) and IV fluids to support circulation.
- Avoid hyperthermia, hyperglycemia (maintain blood sugar <200 mg/dL).
- Treating the precipitating cause of cardiac arrest
Syncope:
Question 170. Write short essay/note on cardiac syncope/syncope.
Answer:
Cardiac syncope Definition: Syncope is defined as a transient loss of consciousness due to inadequate cerebral blood flow with loss of postural tone.
- It is associated with loss of postural tone, with spontaneous return to baseline neurologic function without any resuscitative efforts.
- Presyncope is the term used for lightheadedness in which the individual thinks he/she may black out.
- Classical vasovagal syncope: Syncope triggered by emotional or orthostatic stress such as venipuncture (experienced or witnessed), painful or noxious stimuli, fear of bodily injury, prolonged standing, heat exposure, or exertion.
Mechanism: Global hypoperfusion of cerebral cortices or focal
hypoperfusion of the reticular activating system.
- About one-third of individuals may develop a syncopal episode during their lifetime.
- Its incidence increases with age (sharp rise at age 70 years).
- Cardiac syncope has a high incidence (about 24%) of subsequent cardiac arrest.
Causes of Syncope:
Causes of nonsyncopal attacks: Episodes that may be confused with syncope include disorders without impairment of consciousness and disorders with partial or complete loss of consciousness.
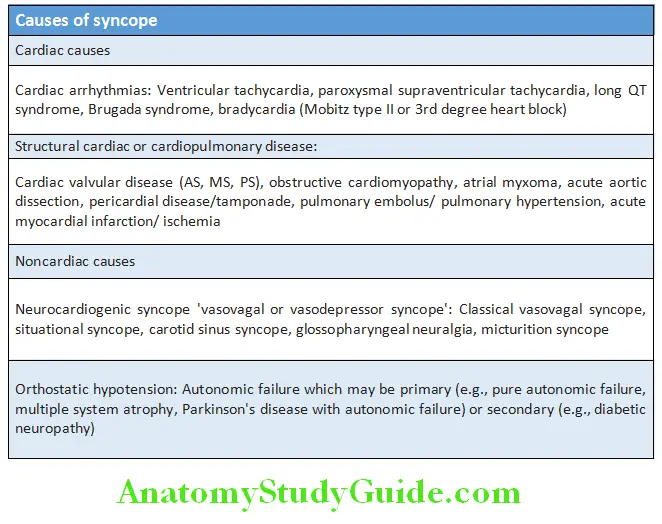
Causes of nonsyncopal attacks:
- Seizures
- Metabolic or toxic abnormalities: Hypoglycemia and encephalitis rarely syncope.
- Neurologic syncope: Subarachnoid hemorrhage, transient ischemic attack, complex migraine headache.
- Psychiatric syncope
- Drug-induced loss of consciousness: Drugs of abuse and alcohol.
Cardiac syncope Investigations:
- ECG: Factors that suggests arrhythmia-induced syncope are prolonged intervals (QRS, QTc), severe bradycardia, pre-excitation, evidence of myocardial infarction.
- Holter monitoring
- Echocardiography
- Neurological: CT brain and MRI, EEG
- Laboratory evaluation: Full blood count (FBC) and hematocrit, random blood sugar (RBS) test, electrolytes, pregnancy test.
- Head up tilt table test.
Cor Pulmonale:
Question 171. Discuss the etiology, pathogenesis, clinical features, investigations, and management of chronic cor pulmonale.
Answer:
- Cor pulmonale is a Latin word means “pulmonary heart” and is defined as symptoms and signs of fluid overload secondary to lung disease.
- Cor pulmonale is a disease of the right ventricle characterized by its hypertrophy and dilation with or without failure secondary to diseases directly affecting the lung parenchyma, pulmonary vasculature, chest bellows or central. This excludes pulmonary alterations produced due to diseases that primarily affect the left side of the heart (e.g., congenital heart diseases).
Types of Cor Pulmonale:
Question 172. Write short essay/note on types and causes of cor pulmonale.
(or)
Write short essay/note on acute cor pulmonale.
Answer:
According to rate of development: Acute or chronic.
- Acute cor pulmonale develops due to a sudden increase in right ventricular pressure. It usually follows acute massive pulmonary embolism that is sufficient enough to obstruct more than 60% of pulmonary circulation. It leads to acute pulmonary hypertension, acute right ventricular dilatation and failure. Other example is acute respiratory distress syndrome.
- Chronic cor pulmonale: It can be further divided hypoxic type or vascular obliterans type.
- Hypoxic subtype: Chronic obstructive pulmonary disease (COPD) is the most common disease associated with hypoxic subtype.
- Obliterans subtype: Most common process associated with this subtype is pulmonary thromboembolic disease.
Types of Cor Pulmonale Etiology and Clinical Features:
Usually the signs and symptoms are minimal except in the advanced stage. Mostly the clinicians focus on the disease producing cor pulmonale rather than on cor pulmonale itself.
Causes of cor pulmonale:
- Diseases of lung: Chronic obstructive pulmonary disease (COPD, e.g., chronic bronchitis and emphysema), pulmonary tuberculosis, interstitial lung disease, high altitude dwelling, cystic fibrosis, pleural fibrosis
- Diseases of pulmonary circulation: Recurrent pulmonary thromboembolism, primary pulmonary hypertension, collagen vascular diseases, chronic liver disease
- Diseases of thorax: Kyphoscoliosis, neuromuscular diseases, sleep apnea syndrome, obesity
- Diseases of respiratory control: Brainstem lesions, central sleep apnea—Ondine’s curse.
Symptoms:
Similar to those of right side heart failure: Fatigability, dyspnea on exertion, syncope, chest pain, palpitation, abdominal distension, edema of the lower extremity, exercise-induced peripheral cyanosis and excessive daytime somnolence.
Signs:
- Pedal edema
- Accentuated A wave of the jugular venous pulsations and prominent jugular V wave, indicating the presence of tricuspid regurgitation.
- Palpable left parasternal lift.
- Accentuated pulmonic component of the second heart sound, right sided S4 heart sound, ejection systolic murmur in the pulmonary area. In addition there may be pansystolic murmur of tricuspid regurgitation or early-diastolic murmur of pulmonary regurgitation.
- Overt right side heart failure In a patient with chronic cor pulmonale reveal following signs:
- Increasing peripheral edema
- Jugular venous pressure: Raised and hepatojugular reflux positive
- Tender hepatomegaly and enlargement of heart
- Right ventricular third heart sound and a gallop rhythm, and right sided fourth heart sound.
Management of Chronic Cor Pulmonale:
These include:
- General Measures
- Additional Measures In Cor Pulmonale Due To Copd
- Surgical treatment.
- General measures
- Nonpharmacological treatment:
- Oxygen therapy.
- Phlebotomy.
- Noninvasive positive pressure ventilation (NIPPV).
- Pharmacological treatment:
- Diuretics.
- Anticoagulation.
- Vasodilators
- Treatment to decrease pulmonary hypertension
- Treatment of underlying disease.
- Oxygen therapy is the most important treatment for reducing pulmonary hypertension. The long-term oxygen therapy retards its progression of pulmonary hypertension and oxygen therapy should be started if the arterial oxygen tension is 55 mm Hg or less.
- Treatment of right heart failure
- Restriction of salt intake
- Digoxin: Role not clear
- Diuretics
- Chronic anticoagulation with Warfarin may benefit patients with cor pulmonale due to thrombo-occlusive pulmonary disease.
- Surgical treatment
- Pulmonary embolectomy: If pulmonary emboli are not resolved.
- Heart lung transplantation: For primary pulmonary hypertension
Types of Cor Pulmonale Investigations:
- Chest X-ray:
- Enlarged pulmonary artery and right ventricle, distended azygous or other central vein.
- In pulmonary embolism Wester mark sign “oligemia of lung lobe or entire lung.” Hampton’s hump ‘wedge-shaped opacity’
- In COPD anterior-posterior diameter increased, diaphragm flattened, honeycombing and hyperlucency of lung.
- Electrocardiogram: Right axis deviation, P pulmonale (large P wave) in the inferior and anterior leads ‘right atrial enlargement’, right bundle branch block, right precordial T-wave inversions, delayed intrinsicoid deflection of right precordial leads, RVH, S1 Q3 T3 pattern and QR pattern in lead V1 or V3R.
- Arterial blood gas analysis: Hypoxia, hypercapnia.
- BNP: Used as biomarker for pulmonary hypertension in COPD patients.
- Echocardiography: Hypertrophy and dilatation of right ventricle.
- Others: Contrast-enhanced computed tomography/high-resolution computed tomography (CECT/HRCT) chest, nuclear scan for detecting pulmonary embolism, rarely right heart catheterization.
Diseases Of Vessels:
Aortic Aneurysms:
Question 173. Write short note on aneurysm of aorta.
(or)
Write short note on classification of aortic aneurysms and its common causes.
Answer:
Definition of aneurysm: It is an abnormal permanent focal dilatation of a blood vessel/artery to 1.5 times its normal diameter.
Classifiation of Aortic Aneurysm:
- Depending on the gross appearance:
- Fusiform aneurysm
- Saccular Aneurysm
- Cylindrical
- Arterial dissection.
- Depending on the sites/region involved:
- Thoracic part of aorta
- Ascending Aorta
- Arch Of Aorta
- Descending Aorta
- Abdominal aorta (commonest site is infrarenal portion).
- Depending on the etiology: Atherosclerosis (commonest cause), cystic medial necrosis, syphilis (aneurysm of ascending aorta), rheumatic aortitis, and trauma.
Aortic Dissection:
Question 174. Write short note on aortic dissection.
Answer:
Aortic Dissection Definition: Aortic dissection develops when blood from the aortic lumen enters into the aortic wall through a breach in the integrity of the aortic wall.
- Blood enters the media, split it into two layers and travels along the layers of the media creating a “false blood-filled lumen” alongside the existing or “true lumen.”
- Usually, the false lumen re-enters the true lumen, producing a double-barreled aorta. However, it may rupture into the surrounding structures (e.g., left pleural cavity, pericardium) with fatal consequences.
- Dissection may damage aortic valve and compromise the branches of the aorta.
- Affects males more than females during 5th to 7th decades. Men/Female ratio 2:1 to 5:1. Proximal dissection peak age is 50–55 years and distal dissection peak age is 60–70 years.
- Dissection in younger patients is usually associated with Marfan’s syndrome, pregnancy or trauma.
Predisposing Factors and Classifiation:
Predisposing factors for aortic dissection Stanford anatomical classification
Type A dissections (66%): Dissection involving either both the ascending and descending aorta or only the ascending aorta (types I and II of the DeBakey classification). It is more common but dangerous.
Type B dissections: They have dissection limited to descending aorta (usually begin distal to the subclavian artery) and do not involve the ascending part and (DeBakey type III).
Predisposing factors for aortic dissection:
- Systemic hypertension (~80%)
- Aortic diseases: Aortic atherosclerosis, dilatation, nonspecific aneurysm, arteritis, coarctation, bicuspid aortic valve. Previous aortic surgery (e.g., coronary artery bypass grafting (CABG), aortic valve replacement)
- Trauma
- Direct iatrogenic, e.g., cardiac catheterization, intra-aortic balloon pumping
- Indirect trauma, e.g., sudden deceleration
- Hereditary connective tissue diseases, e.g., Marfan syndrome, Ehlers-Danlos syndrome
- Chromosomal aberrations: Turner’s syndrome, Noonan’s syndrome
- Others: Third trimester of pregnancy or first stage of labor, giant cell arteritis, Behçet’s disease, and syphilis
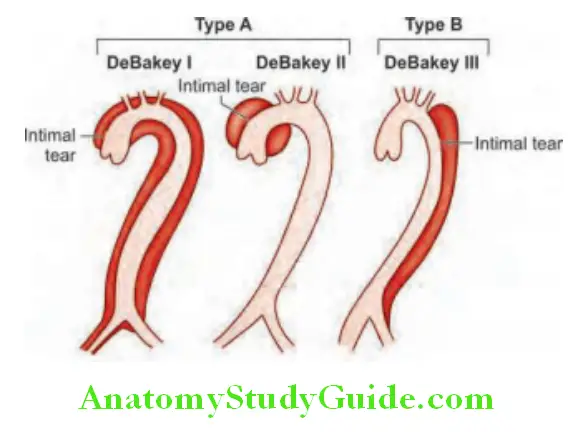
- DeBakey classification:
- Type 1: Involves ascending to descending aorta
- Type 2: Limited to ascending or arch of aorta
- Type 3: Involves descending aorta only.
- According to the timing of diagnosis from the initial onset of symptoms:
- Acute diagnosis is made <2 weeks of symptoms
- Subacute 2–8 weeks of symptoms
- Chronic >8 weeks of symptoms.
Aortic Dissection Clinical Features:
Symptoms:
- Sudden (abrupt) onset of severe, tearing, migratory pain, beginning in the anterior chest, radiating to the back between the scapulae (interscapular area), and moving downward as the dissection progresses. Pain may be misdiagnosed as that of myocardial infarction. Usually anterior chest pain develops when there is involvement of the ascending aorta, pain is intrascapular when descending aorta is involved. Collapse is common.
- Others: Syncope, dyspnea, weakness, hypertension or hypotension.
Signs:
Asymmetry of pulses and blood pressure: It may produce asymmetry of the brachial, carotid or femoral pulses and blood pressure.
- Signs of aortic regurgitation.
- Signs of occlusion of branches of aorta: It may occlude orifices of branches of aorta and cause MI (coronary), stroke (carotid) paraplegia (spinal), mesenteric infarction with a acute abdomen (celiac and superior mesenteric), renal failure (renal) paraplegia (spinal cord) and ischemia of limb (usually leg).
- Others: Pulmonary edema, hemopericardium, cardiac tamponade.
Aortic Dissection Investigations:
- Chest X-ray: May show widening/broadening of superior mediastinum and distortion of the aortic “knuckle” but these may be absent in 10% of cases. A small left-sided pleural effusion may be found.
- Electrocardiogram: ECG may show left ventricular hypertrophy in patients with hypertension (33% of cases). ECG is necessary to rule out any ischemic changes or MI. Rarely changes of acute MI (usually inferior) may be evident.
- Doppler echocardiography: It may reveal aortic regurgitation, and dilated aortic root.
- Transesophageal echocardiography (TEE): May be useful since it can show images of the first 3–4 cm of the ascending aorta (sensitivity 98–99% and specificity 94–95%).
- CT and MRI angiography: Highly (98–100%) specific and sensitive MRI is the gold standard for diagnosis.
Aortic Dissection Management/Treatment:
- Mortality rate of acute aortic dissection if untreated is 1–5% /hour and 90% within 3 months. Hence urgent treatment is needed.
- Goal: Reduction of systolic blood pressure (100–120) and diminution of dp/dt (reflects force of LV ejection) through use of a beta blocker. Therapy is targeted to halt the progression of the dissection. The course of the tear rather than the tear itself leads to compromise of vasculature or rupture.
- Initial medical management consists of control of pain and antihypertensive treatment. About 50% of patients are hypertensive and they are given urgent antihypertensive medication to reduce systolic blood pressure to below 120 mm Hg. This may be achieved by
- Intravenous beta-blockers (e.g., labetalol, metoprolol) and vasodilators (GTN).
- Intravenous calcium channel blockers (e.g., verapamil or diltiazem) are used if β-blockers are contraindicated.
- If the above drugs fail to control BP adequately, sodium nitroprusside may be given.
- Type A dissections need emergency surgery to replace the ascending aorta (arch replacement) if fit enough, because medical management has a high mortality (50% within 2 weeks).
- Better prognosis with type B dissections and have a survival of 89% at 1 month. They are initially treated medically unless they develop complications such as actual or impending external rupture, or vital organ (gut, kidneys) or limb ischemia. In these patients very high morbidity and mortality rate associated with surgery.
- Surgical therapy: Percutaneous or minimal access endoluminal repair and involves either “fenestrating” (perforating) the intimal flap or endovascular implantation of a stent graft placed from the femoral artery.
Cause of death: Acute aortic regurgitation, major branch vessel obstruction, aortic rupture (into pericardium, left pleural cavity, or mediastinum).
Marfan’s Syndrome:
Question 175. Write short note on Marfan’s syndrome and its clinical features.
Answer:
It is an autosomal dominant disorder of connective tissue characterized by mutations in the fibrillin gene on chromosome 15.
Systems Affcted:
- Cardiovascular system: Weakening of the media of the aortic root cause dilatation (Fig. 7.55D), regurgitation and dissection. Mitral valve prolapse, mitral regurgitation.
- Skeletal system: Tall stature, arachnodactyly, wrist, joint hypermobility, scoliosis, chest deformity, and high arched palate
- Eyes: Dislocation of the lens (superior and temporal).
Marfan’s Syndrome Investigations: Chest X-ray (Fig. 7.55D), echocardiography, MRI or CT may detect aortic dilatation.
Marfan’s Syndrome Treatment:
β-blockers reduces the rate of aortic dilatation and risk of rupture. Elective replacement of the ascending aorta: in patients with progressive aortic dilatation.
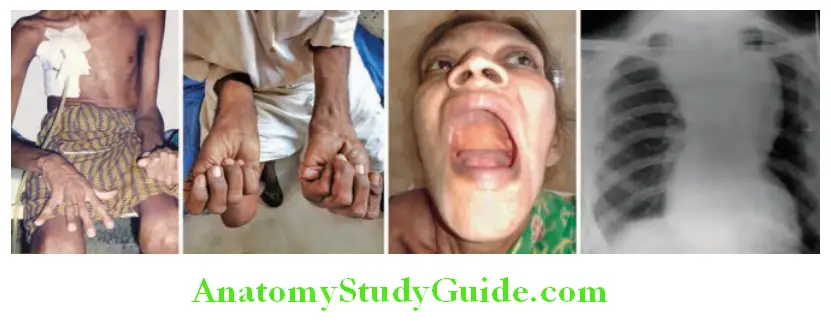
Raynaud’S Phenomenon And Raynaud’S Disease:
Question 176. Write short note on Raynaud’s phenomenon, its causes and Raynaud’s disease.
Answer:
- Raynaud’s phenomenon: Chronic episodic attacks of digital ischemia due to spasm of the digital arteries usually precipitated by exposure to cold or emotional stress and relieved by heat. An underlying disease or cause is present and most commonly connective tissue disease, such as systemic sclerosis. If there is no underlying cause, it is termed Raynaud’s disease (primary or idiopathic Raynaud’s phenomenon).
- Affects 5–10% of population.
Causes of Raynaud’s Phenomenon and Clinical Features
- Characterized by sequential color changes: White (pallor due to vasospasm), >blue (cyanosis due to deoxygenated blood) > and red (rubor due to reactive hyperemia). Pallor is essential for diagnosis.
- Age of onset and gender: Usually less than 40 years of age, but it may occur over this age. F:M = 5:1. However, in secondary cases it depends on the underlying disease, and is not restricted to female sex.
- Raynaud’s phenomenon usually involves the hands and less common in the feet. Rarely, involves nose, ear lobe, tongue, etc.
- Typical attack is characterized by sudden attack of pallor in one or more digits, followed after a few minutes by cyanosis or/and sometimes by erythema. Attacks are usually precipitated by cold (either local or of the whole body), by pressure or by psychological stimuli. Episodes may occur infrequently or many times each day.
- Primary Raynaud’s: Usually bilateral, symmetrical and involves several digits (fingers).
- Secondary Raynaud’s: Involves only one or a few digits and asymmetry is not unusual.
- Severe cases:
- Usually seen in secondary type and may be complicated by:
- Telangiectasias of the nail fold
- Thinning and ridging of the nail
- Atrophy or sclerosis of the fingers (sclerodactyly).
- Skin necrosis: Rare in primary Raynaud’s phenomenon but not uncommon in secondary Raynaud’s phenomenon and may lead to destruction of the digits.
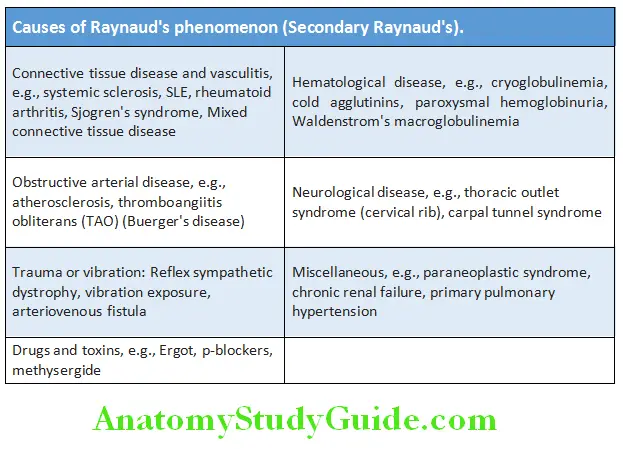
Raynaud’S Phenomenon Investigations:
- Blood test:
- Complete blood count, ESR, urinalysis
- Patient with ANA, specific antigen like Sm antigen or topoisomerase or centromere or DsDNA to rule out SLE or scleroderma.
- Chest X-ray: To look for cervical rib or evidence of ILD.
- Specific test: Nail fold capillary microscopy procedure differentiates primary from secondary Raynaud phenomenon. Patients with connective tissue diseases may have enlarged deformed capillary loops surrounded by avascular areas.
Raynaud’S Phenomenon Treatment:
General measures:
- Lifestyle modifications to minimize exposure to cold.
- Patient’s education to recognize and terminate attacks promptly by returning to a warmer environment and applying local heat to hands (e.g., by placing their hands in warm water or by using a hair dryer).
- Cessation of smoking because nicotine induces cutaneous vasoconstriction.
- Stress modification and social support to minimize vasoconstriction induced by hyperactivity of sympathetic nervous system.
- Treatment of underlying cause.
- Drugs: Calcium channel blockers (e.g., nifedipine, amlodipine), sildenafil, prostaglandin analogues (e.g., ilaprost), bosentan, losartan, topical glyceryl trinitrate.
- Sympathectomy: Patients with refractory, disabling attacks or with an acutely ischemic digit that is unresponsive to other measure.
Circulatory Failure Shock:
Question 177. Write short note/essay on
- Septic shock, SIRS and sepsis.
- Anaphylactic shock and neurogenic shock.
- Management of septic shock.
(or)
Define acute circulatory failure/shock.
Answer:
Circulatory Failure Shock Defiition:
- Shock (acute circulatory failure, low-output state) is defined as a state with impaired cardiac pump, circulatory system, and/or volume that can lead to compromised blood flow to tissues.
- Kumar and Parrillo (1995): Shock is a state in which profound and widespread reduction of effective tissue perfusion leads first to reversible, and then if prolonged, to irreversible cellular injury.
Classifiation and Causes:
Question 178. Write short note/essay on classification and causes of shock.
(or)
Causes of cardiogenic shock.
Answer:
Stages of Shock:
Stages of shock have been shown in Table:
Pathogenesis of Various Types of Shock:
Pathogenesis of various types of shock has been shown in Flowchart:
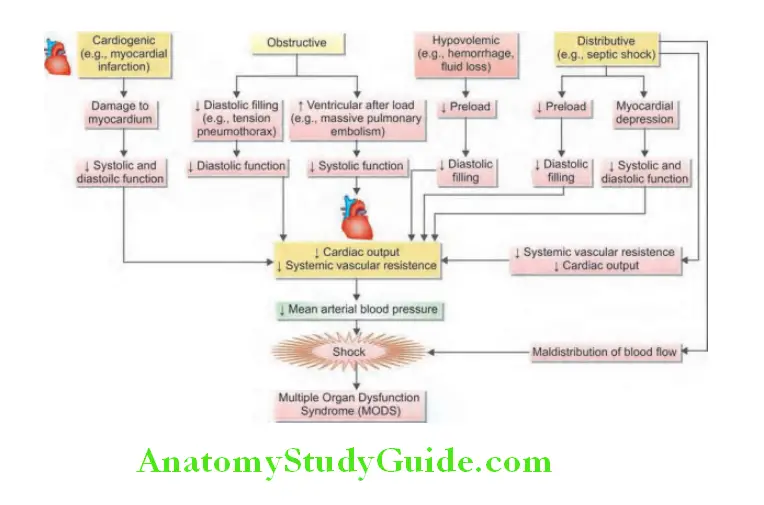
Shock Clinical Features:
Clinical features of various types of shock have been shown in Table:
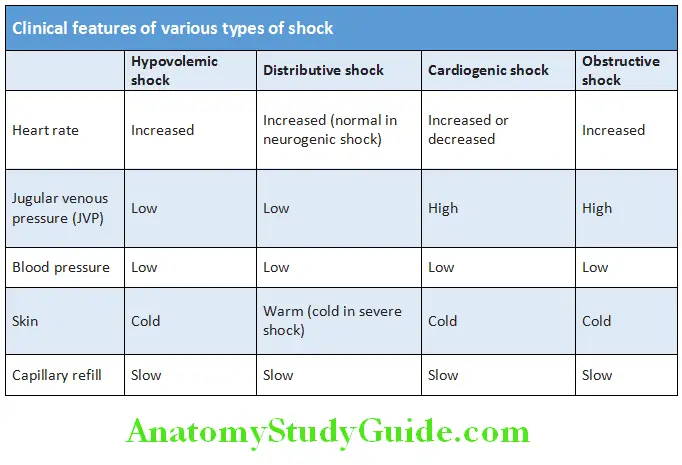
Question 179. Write short notes on hypovolemic shock.
Answer:
Types of Shock:
- Neurogenic shock:
- Neurogenic is the rarest form of shock. Caused by the loss of sympathetic control of resistance vessels resulting in massive dilatation of arterioles and venules.
- A type of distributive shock that results from the loss or suppression of sympathetic tone.
- Causes massive vasodilatation in the venous vasculature → venous return to heart → cardiac output.
- Most common cause is spinal cord injury above T6.
- Sepsis:
- Sepsis syndrome: SIRS with confirmed infectious process associated with organ failure or hypotension
- Two phases:
- ‘Warm’ shock-early phase: Hyperdynamic response, vasodilation
- “Cold” shock-late phase: Hypodynamic response
- Decompensated state
- Anaphylactic shock:
- A type of distributive shock that results from widespread systemic allergic reaction to an antigen.
- Differences between anaphylactic shock and anaphylactoid shock.
- This hypersensitive reaction is life-threatening.
- Cardiogenic shock:
- The impaired ability of the heart to pump blood. Pump failure of the right or left ventricle.
- Most common cause is left ventricular myocardial infarction (anterior). Occurs when >40% of ventricular mass damage.
- Mortality rate is 80% or more.
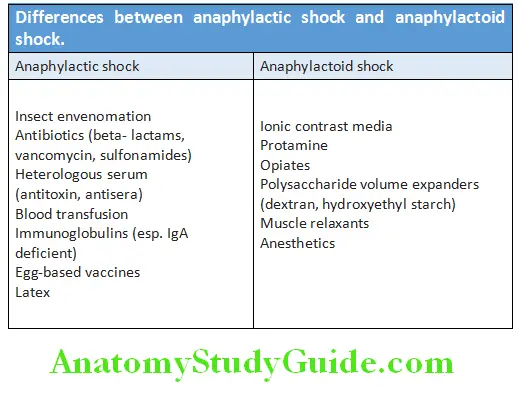
Question 180. Write short note/essay on management of shock.
Answer:
Management of Shock:
Goal of management:
- Treat reversible causes
- Protect Ischemic Myocardium
- Improve tissue perfusion.
Patient Monitoring:
- Initial assessment (ABC)
- Airway:
- Does patient have mental status to protect airway?
- Glasgow Coma Scale (GCS) less than “eight” means “intubate”
- Airway is compromised in anaphylaxis.
- Breathing: If patient is conversing with you, airway (A) and breathing (B) are fine, place patient on oxygen.
- Circulation: Vitals (heart rate and blood pressure), 2 large bore IV, start fluids (careful if cardiogenic shock), put on continuous monitor.
- In a trauma, perform ABCDE, not just ABC.
- Deficit or disability:
- Assess for obvious neurologic deficit.
- Moving all four extremities—Yes/No Pupils-Reaction
- Glasgow Coma Scale (M6, V5, E4).
- Exposure: Remove all clothing on trauma patients.
General Measures:
- Optimize oxygen content
- Hemoglobin: Check if appears pale or anemic, check coagulation status.
- SaO2: Just a pulse oximeter tells you the SpO2. Check SaO2 on ABG.
- Optimize cardiac output
- Cardiac output (CO) = Stroke volume (SV) x Heart rate (HR)
- Stroke volume depends on preload, contractility and afterload
- Preload: Look at response to fluid bolus.
- If improves BP could be suggestive of decreased preload (volume) and a reasonable contractility.
- If no improvement or worsening BP could be suggestive of a contractility problem or excess preload (volume) situation.
- Look at CVP
- Contractility
- Check any history suggestive of ischemic disease or CCF.
- Check ECHO and ECG.
- A high systolic pressure could be suggestive of good contractility.
- Afterload
- Check ECHO if suggestive of any obstructive features.
- If peripheries cold could indicate increased vascular resistance.
- If peripheries warm could indicate vasodilation and decreased vascular resistance.
- A low diastolic blood pressure could indicate low vascular resistance.
- A high diastolic blood pressure could indicate increased vascular resistance.
- Preload: Look at response to fluid bolus.
- Heart rate:
- If low, 2 possible interventions, namely electric pacing or pharmacological intervention.
- If high, 2 possible interventions, namely electric cardioversion/defibrillation or pharmacological intervention.
Optimize Blood Pressure:
Question 181. Write short note/essay on sympathomimetic amines and vasopressor agents in shock.
Answer:
- Vasopressors and inotropes (sympathomimetic amines and vasopressin)
- Dobutamine alone is useful to augment cardiac output if arterial pressure is near-normal. Otherwise, a combination of dopamine and dobutamine is preferred initial sympathomimetic agents.
- Vasopressin
- Vasopressin constricts vascular smooth muscle directly via V1 receptors, and also increases responsiveness of the vasculature to catecholamines.
- Vasopressin may also increase blood pressure by inhibition of vascular smooth muscle nitric oxide production.
- Milrinone can be used to increase cardiac contractility.
- Steroids can be used in specific types as septic shock.
Treatment of Underlying Cause:
- Hypovolemic shock
- Rapid replacement of blood, colloid, or crystalloid.
- Identify source of blood or fluid loss and treat it. Endoscopy/ colonoscopy or angiography
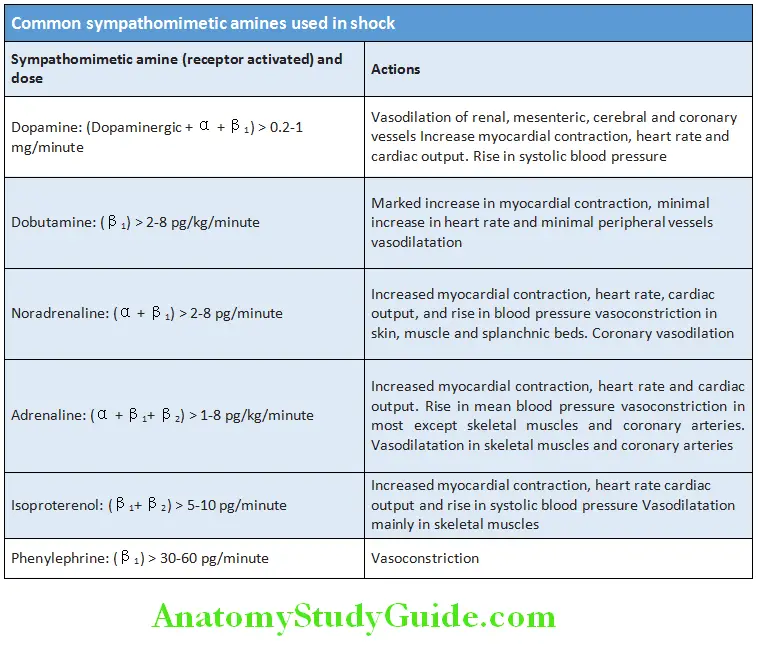
Question 182. Write short note on management of cardiogenic shock.
Answer:
Cardiogenic Shock:
Extracardiac obstructive shock:
- Pericardial tamponade: Pericardiocentesis and surgical drainage (if needed).
- Pulmonary embolism
- Heparin
- Ventilation/perfusion lung scan
- Pulmonary angiography
- Consider: Thrombolytic therapy, embolectomy at surgery.
Septic shock:
- Identify site of infection and drain, if possible.
- Antimicrobial agents (key rules).
- ICU monitoring and support with fluids, vasopressors, and inotropic agents.
Management of neurogenic shock:
Hypovolemia: Treat with careful fluid replacement for BP<90 mm Hg, UO <30 cc/hr.
- Observe closely for fluid overload
- Vasopressors may be needed.
- Hypothermia: Warming. Avoid large swings in patient’s body temperature.
- Treat hypoxia
- Maintain ventilatory support.
- Alpha agonist to augment tone if perfusion still inadequate
- Dopamine (>10 mg/kg per min)
- Ephedrine (12.5–25 mg IV every 3–4 hour)
- Treat bradycardia with atropine 0.5–1 mg doses to maximum 3 mg, may need transcutaneous or transvenous pacing temporarily.
Management of cardiogenic shock:
Left ventricular infarction:
- Intra-aortic balloon pump (IABP)
- Cardiac angiography
- Revascularization: Angioplasty, coronary bypass
Right ventricular infarction:
- Fluid and inotropes with PA catheter monitoring
Mechanical abnormality:
- Echocardiography
- Corrective surgery
Pulmonary Embolism And Venous Thrombosis:
Pulmonary Embolism (PE):
Question 183. Discuss the etiology, clinical features, investigations, diagnosis and management of pulmonary thromboembolism and pulmonary infarction.
Answer:
Pulmonary Embolism Defiition:
- Pulmonary embolism (pulmonary thromboembolism—PTE) is defined as an embolism in which emboli occlude pulmonary arterial tree (pulmonary artery or its branches).
- Pulmonary embolism is the second most common cause of unexpected death in most age groups. It accounts for about 15% of all postoperative deaths. Present in 60–80% of patients with DVT, more than 50% of them are asymptomatic.
- Pulmonary infarction: It is ischemic necrosis of lung tissue following embolic occlusion. It develops in less than 10% of cases of pulmonary embolism. They are more common in patients with an underlying cardiac or pulmonary disease.
Source of Emboli:
Pulmonary embolism usually results from dislodgement of venous thrombi and the source of these emboli are as follows:
- Thromboemboli:
- Deep leg veins: Deep venous thromboses (DVTs) are the source in more than 95% of cases of pulmonary emboli. Deep leg veins include popliteal, femoral or iliac veins. Other source of thromboemboli is from pelvic veins, abdominal veins and vena cava.
- Other sites: Rarely, source of emboli may be right atrium or right ventricle.
- Other nonthrombotic emboli: They are rare and include septic emboli (from endocarditis involving the tricuspid or pulmonary valves), tumor emboli (especially choriocarcinoma), fat (long bone fractures), air, amniotic fluid emboli, and foreign material during IV drug use.
Causes and Risk Factors:
Causes and risk factors (predisposing factors) for venous thrombosis and embolism have been shown in Table

Question 184. Write short essay/note on the predisposing causes of pulmonary embolism.
(or)
Write short note on Virchow’s triad.
Answer:
Virchow’s triad: It consists of three factors that predispose to thrombus formation and includes
- Endothelial Injury,
- Altered Blood Flow And
- Hypercoagulability. These factors favor the adhesion of platelets at the site of endothelial damage.
These platelets undergo aggregation and release factors which generate thrombin generation. Venous thrombi form and flourish in the presence of stasis and hypercoagulability.
Pathological Consequences:
Embolization: The deep venous thrombi may detach from their site of formation. These emboli pass to the vena cava, right atrium and right ventricle, and finally lodge in the pulmonary arterial circulation. In the lung, they produce acute pulmonary embolism.
- Acute massive pulmonary embolism: When emboli lodges in the main pulmonary artery (may result in death). Massive pulmonary embolism shows 60% or more obstruction of the pulmonary circulation. Massive PE accounts for 5–10% of cases.
- Pulmonary infarction: It results from embolism to smaller pulmonary arteries. Most (about 75%) small pulmonary emboli do not produce infarcts. However, in the patients with congestive heart failure or chronic lung disease an embolus can cause infarction.
- Pulmonary hemorrhage: Obstruction of medium-sized pulmonary arteries by emboli and subsequent rupture of these vessels can result in pulmonary hemorrhage.
- Pulmonary hypertension: Multiple recurrent pulmonary emboli → may cause mechanical blockage of the arterial bed → result in chronic pulmonary hypertension → right ventricular failure.
Clinical Features of Pulmonary Embolism and Pulmonary Infarction:
- Dyspnea (75–85%), pleuritic chest pain (57–87%), cough (40–53%), hemoptysis, syncope, similar to obstructive lung disease (chronic).
- Massive pulmonary embolism: Rarer condition characterized by sudden collapse because of an acute obstruction of the right ventricular outflow tract. Symptoms include collapse/hypotension, unexplained hypoxia, engorged neck veins, and acute right heart failure (acute cor pulmonale).
Physical fidings:
- Patient may be normal.
- There may be tachypnea (respiratory rate >16/min) in 96% of cases, rales (58%), accentuated second heart sound (53%), tachycardia (heart rate >100/min), fever (temperature >37.8 °C), diaphoresis, S3 or S4 gallop.
- Other clinical signs and symptoms suggesting thrombophlebitis, lower extremity edema, cardiac murmur, cyanosis and clinical evidence of DVT may be present.
Wells Scoring System:
Wells scoring system for the diagnosis of pulmonary embolism (PE):
- Clinically suspected DVT: 3.0 points
- Alternative diagnosis is less likely than PE: 3.0 points
- Tachycardia (heart rate >100): 1.5 points
- Immobilization (≥3d)/surgery in previous four weeks: 1.5 points
- History of DVT or PE: 1.5 points
- Hemoptysis: 1.0 point
- Malignancy (with treatment within six months) or palliative: 1.0 point Score >4-PE likely. Consider diagnostic imaging and score 4 or less-PE unlikely.
Diagnosis:
Question 185. Write short essay/note on diagnosis of pulmonary embolism.
Answer:
Pulmonary embolism is the Great Masquerader. Its symptoms and signs are nonspecific and the diagnosis is difficult. Not all leg pain is due to deep vein thrombosis and not all dyspnea is due to pulmonary embolism. Pulmonary embolism should be considered if a patient has pleuritic chest pain, hemoptysis or dyspnea that is out of proportion to the size of pleural effusion.
- Nonimaging diagnostic modalities Blood tests:
- Pulmonary infarction: It results in polymorphonuclear leukocytosis, raised ESR and raised lactate dehydrogenase levels in the serum.
- D-dimer assay: D-dimer is a specific degradation product produced during the breakdown of endogenous fibrin (fibrinolysis) by plasmin. It is best screening test for PE or DVT.
- Its significance are:
- Raised D-dimer: The quantitative raise in plasma D-dimer [measured by enzyme-linked immunosorbent assay (ELISA)] indicates the presence of DVT or PE or clinically ineffective thrombolysis. The sensitivity of the D-dimer is >80% for DVT (less sensitive than for PE because the size of DVT thrombus is smaller) and >95% for PE. However, raised D-dimer is not specific, because it may be also raised in other conditions (e.g., myocardial infarction, pneumonia, sepsis, cancer, postoperative state and during second or third trimester of pregnancy). It has no role in hospitalized patients because levels are usually raised due to systemic illness. A positive test or normal test in high-risk patients requires further specific diagnostic tests, such as Doppler of legs for deep vein thrombosis, spiral CT pulmonary angiography (CT-PA), perfusion scanning of the lungs and pulmonary angiography.
- Normal D-dimer: If D-dimer is normal, it rules out the diagnosis of embolism (unless patient has a high-risk of PTE clinically).
- ECG is often normal but helps in excluding other differential diagnoses (e.g., acute myocardial infarction and pericarditis). It may show sinus tachycardia, changes of acute pulmonary hypertension and right ventricular strain. Other abnormalities include atrial fibrillation; ECG changes include the Sl Q3T3 sign: an S wave in lead 1, a Q wave in lead III, and an inverted T wave in lead III.
- Arterial blood gases: Show a hypoxemia (reduced PaO2) and a normal or hypocapnia (low PaCO2) i.e. type I respiratory failure pattern and an increased alveolar–arterial oxygen gradient.
- Noninvasive Imaging Modalities
- Ultrasound scanning: For the detection of clots in deep veins, pelvic or iliofemoral veins.
- Venous ultrasonography: Color Doppler ultrasound of the leg veins examines the venous flow dynamics and is the investigation of choice in suspected DVT. Normally, manual compression of calf increases the Doppler flow pattern. Loss of normal respiratory variation is seen when there is occluding DVT or any obstruction within the pelvis.
- Chest X-ray
- Pulmonary embolism
- Normal or nearly normal
- Increased radiolucency in lung zones (due to decreased or absent blood flow)
- Small infiltrative shadows (due to linear atelectasis)
- Elevated hemidiaphragm
- Difference in the diameter of pulmonary arteries and their main branches on either side.
- Abrupt “cut-off” of a blood vessel
- Enlarged right descending pulmonary artery (Palla’s sign).
- Pulmonary infarction
- May be normal during early stages
- Parenchymal, peripheral, wedge-shaped (pulmonary infarct) density above the diaphragm (Hampton’s hump). Previous infarcts may appear as opaque linear scars.
- Translucent under perfused distal zone, infiltrative shadow abutting against the pleura (usually appears 12–36 hours later)
(Westmark’s sign). - Abrupt cutoff of a pulmonary artery
- Blunting of a costophrenic angle (due to a small pleural effusion).
- Chest CT: CT of the chest with intravenous contrast is the main imaging test for the diagnosis of PE.
- Lung scanning: It is the second line diagnostic test for PE and performed in patients who cannot tolerate intravenous contrast. The perfusion scan defect favors absent or decreased blood flow possibly due to PE whereas a normal perfusion scan excludes significant pulmonary embolism. However, an abnormal perfusion scan may be due to lung pathology (e.g., asthma and chronic obstructive pulmonary disease). In such patients, a ventilation-perfusion scan is needed though it may still give ambiguous results.
- Magnetic resonance (MR) (contrast-enhanced) imaging: It is useful mainly in detecting large proximal PE but is not reliable for smaller segmental and subsegmental PE.
- Echocardiography is not useful for diagnosis of acute PE, but is very useful for detecting conditions that mimic PE
(e.g., acute myocardial infarction, pericardial tamponade, and aortic dissection). - Radionuclide ventilation/perfusion scanning (V/Qscan): It uses less radiation and contrast and is a good diagnostic tool. Pulmonary Tc scintigraphy shows under perfused regions indicative of pulmonary infarcts. However, this test should be interpreted in consideration of history, examination and other investigations.
- Pulmonary embolism
- Invasive diagnostic modalities:
- Pulmonary angiography: Chest CT with contrast has replaced invasive pulmonary angiography as a diagnostic test. It is performed in patients planned for interventional procedure (e.g., catheter-directed thrombolysis). Features of PE includes visualization of a filling defect in the lumen, abrupt blockage (“cutoff”) of vessels, segmental oligemia or avascularity, a prolonged arterial phase with slow filling, and tortuous, tapering peripheral vessels.
Question 186. Write short essay/note on management of pulmonary embolism.
Answer:
Pulmonary embolism Management:
Supportive measures:
- Prompt recognition and treatment.
- Bed rest in acute stage.
- Oxygen should be given to hypoxemic patients to maintain arterial oxygen saturation above 90%.
- Intravenous saline and/or noradrenaline may be necessary to maintain venous pressure in massive embolism.
- Avoid diuretics and vasodilators because they will reduce cardiac output.
- Analgesics: Opiates may be needed to relieve pain and distress.
- External cardiac massage in the moribund patient may dislodge and breaking up a large central embolus.
Anticoagulation:
- Unfractionated heparin: Initial dose is 5,000–10,000 units IV, followed by maintenance.
- Maintenance: By any one of the three regimens namely
- Continuous Intravenous
- Intermittent Intravenous
- Intermittent subcutaneous.
- Dosage and monitoring of anticoagulant therapy.
- After initiating heparin therapy, repeat APTT every 6 hour for first 24 hours and then every 24 hours when therapeutic APTT is achieved.
- Warfarin 5 mg/d can be started on day 1 of therapy; there is no benefit from higher starting doses.
- Platelet count should be monitored at least every 3 day during initial heparin therapy.
- Therapeutic APTT should correspond to plasma heparin level of 0.2–0.4 IU/mL.
- Heparin is usually continued for 5–7 days. Heparin can be stopped after 4–5 d of warfarin therapy when INR is in 2.0–3.0 range.
- Novel oral anticoagulants.
- Rivaroxaban is an oral factor Xa inhibitor approved by the FDA in November 2012 for the treatment of DVT or PE. Apixaban was approved for treatment of PE in August 2014.
- Direct thrombin inhibitors: Argatroban, ximelagatran.
- Anticoagulation is maintained for at least 6 months.
Thrombolytic therapy:
- Indications: Hemodynamic instability, hypoxia on 100% oxygen, and right ventricular dysfunction by echocardiography.
- Approved thrombolytics for pulmonary embolism
- Streptokinase: 250,000 IU as loading dose over 30 minutes, followed by 100,000 U/h for 24 hours.
- Urokinase: 4400 IU/kg as a loading dose over 10 minutes, followed by 4400 IU/kg/h for 12–24 hours.
- Recombinant tissue-plasminogen activator:100 mg as a continuous peripheral intravenous infusion administered over 2 hours
- Contraindications:
- Absolute: Active internal bleeding
- Relative: Recent surgery, bleeding disorder (thrombocytopenia, renal failure, liver failure), hypertension >200 mm Hg systolic or 110 mm Hg diastolic, hypertensive retinopathy with hemorrhages or exudates.
- Surgical therapy: Include surgical pulmonary embolectomy, inferior vena caval filters (to prevent recurrent emboli) and venous interruption.
- Prophylaxis: To prevent pulmonary embolism (refer prophylaxis for venous thrombosis).
Venous Thrombosis:
Question 187. Discuss the risk factors, clinical features, investigations, diagnosis, and treatment of venous thrombosis [deep venous thrombosis (DVT)].
Answer:
Venous thrombosis is the formation of thrombus in the venous circulation namely veins or right side of the heart.
Sites of Venous Thrombosis:
Most commonly deep or superficial veins of the leg are involved.
- DVT:
- Usually develops in the lower extremity, starting at the larger calf vein level and progressing proximally to involve popliteal, femoral, or iliac veins. In about 80–90% pulmonary embolism, the source is deep vein thrombus. Lower extremity DVTs are found in associated with venous stasis and hypercoagulable states.
- More prone to embolization into the lungs and produce pulmonary infarction. About 50% of DVTs are asymptomatic and are detected after embolization.
- Other systemic veins, e.g., pelvic veins
- Right side of the heart: Atrium (e.g., in patients with atrial fibrillation and cardiac failure) and right ventricle.
- Superficial venous thrombi: They develop in the varicosities involving saphenous veins. It can cause local congestion, swelling (edema), pain, and tenderness. Embolization is very rare.
Risk Factors of Venous Thrombosis:
Clinical Features of Deep Venous Thrombosis:
- Often DVT is asymptomatic with minimal symptoms and signs in about 50% of patients. It may present with clinical features of pulmonary embolism.
- Usually unilateral (only one leg) but may be bilateral (both legs), and thrombus may extend proximally into the inferior vena cava. Bilateral DVT develops more commonly associated with underlying malignancy.
- Major presenting features are: Pain in the calf or tenderness, or both, swelling (ankle edema), redness, an increase in temperature (affected calf is warmer) and dilatation or engorgement of the superficial veins.
- Homan’s sign: Pain in the posterior calf on forced dorsiflexion of the foot while the knee is fully extended. It can be usually demonstrated, but is not diagnostic and may be observed with all lesions of the calf. This test should not be done, because it has the possibility to dislodge the DVT and development of pulmonary embolism.
- Moses test is tenderness over calf muscles on squeezing the muscles from side to side. It is also not done because of possibility of pulmonary embolism.
- Thrombosis in the iliofemoral region: Severe pain, swelling of the thigh and/or ankle edema.
- Complete occlusion of a large vein: Cyanosis of the limb, severe edema, and very rarely lead to venous gangrene.
Diagnosis of DVT:
Question 188. Write short essay/note on Wells probability score.
Answer:
Wells clinical prediction guide for diagnosis of DVT is presented in Table 7.107. It incorporates risk factors, clinical signs, and the presence or absence of alternative diagnoses. It helps in quantifying the pretest probability of DVT namely high score is 3 or more, a moderate score 1−2 and a low score 0. It groups the patients into high, moderate, or low-risk categories.
Complications:
- Pulmonary embolism
- Post-thrombotic syndrome: Consists of edema, skin pigmentation, venous eczema/dermatitis, ulceration around the medial malleolus, venous claudication, nocturnal cramping, and lifelong pain in the involved limb.
- Venous gangrene: Due to complete obstruction of large veins.
DVT Investigations:
- D-Dimer: Raised (>500 ng/mL) but is not specific. May be raised in trauma, recent surgery, hemorrhage, cancer, etc. In DVT, it remains raised for 7 days.
- Negative D-dimer assay: Rules out DVT in patients with low-to-moderate risk and a Wells score ≤1. No further investigations needed.
- Positive D-dimer assay: With a moderate-to-high risk of DVT (Wells score ≥2) need diagnostic investigation, e.g., Doppler ultrasonography.
- B mode venous compression, ultrasonography or Doppler (duplex) ultrasonography: Compression ultrasound is the imaging investigation of choice and has sensitivity for proximal DVT (of the popliteal vein or above) of 99.5%. Venous thrombosis is associated with failure to compress the vascular lumen.
- Contrast venography (phlebography) can reliably detect below knee thromboses, but is now rarely by venography with noninvasive.
- Impedance plethysmography (IPG) also detects above knee thrombi.
- Radiofibrinogen scanning is very accurate in calf vein and lower thigh thrombi.
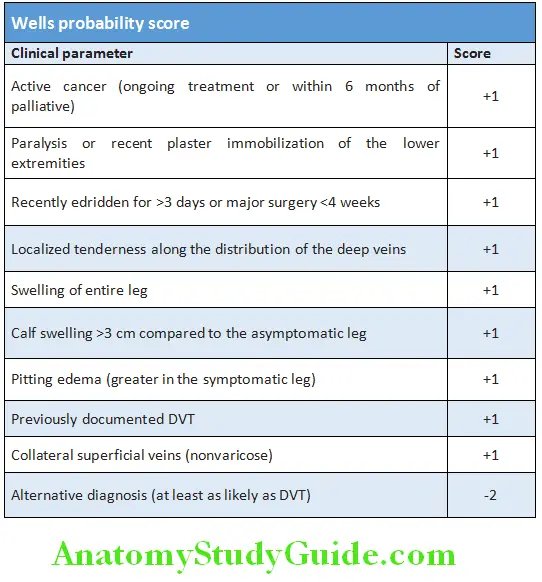
Treatment of DVT:
- Bed rest till the patient is fully anticoagulated.
- Legs: Elevation by 15° and physiotherapy.
Prophylaxis:
Question 189. Write short essay/note on prophylaxis of venous thrombosis (deep venous thrombosis-DVT).
Answer:
- Prophylaxis for DVT and pulmonary embolism is indicated in at high-risk patients.
Prophylaxis of DVT:
- Nonpharmacological measures:
- Mechanical VTE (antiembolism stockings) thigh or knee length.
- Foot impulse devices.
- Physiotherapy
- Intermittent pneumatic compression devices (thigh or knee length): Compress the calf intermittently (usually once a minute).
- Pharmacological prophylaxis
- Fondaparinux or low-molecular weight heparin or unfractionated heparin are given before operation or on admission to hospital, continued until the patient is ambulatory.
- When risk factors are present treatment with warfarin to be given for a prolonged time.
High-risk factors for deep vein thrombosis and pulmonary thromboembolism:
- Patients undergoing major surgery, e.g., abdominal, thoracic or gynecological
- Fractures: Involving pelvis or extremities
- Significant medical comorbidities, e.g., heart disease, metabolic, endocrine or respiratory diseases pathologies, acute infectious diseases, inflammatory conditions
- Patients admitted with major medical diseases:
- Acute coronary syndrome, acute congestive failure [New York Heart Association (NYHA) classes III, IV]
- Acute respiratory diseases (respiratory failure with or without mechanical ventilation), chronic respiratory disease (e.g., chronic obstructive pulmonary disease)
- Active cancer or cancer treatment
- Inflammatory bowel disease
- Acute arthritis of lower extremities
- Sepsis, stroke, paraplegia, obesity
- Varicose veins with phlebitis
Disorders Of Blood Lipids And Lipoproteins:
Question 190. Write short essay/note on structure of lipoproteins and their transport in body.
Answer:
Classes of lipid: Most important are:
- Cholesterol: It is composed of hydrocarbon rings.
- Triglycerides (TG): These are esters composed of glycerol linked to three long-chain fatty acids.
- Phospholipids: Composed of a hydrophobic “tail” consisting of two long-chain fatty acids connected to a hydrophilic head containing a phosphate group by glycerol. Phospholipids are present in cell membranes and constitute important signaling molecules.
Both plasma cholesterol and TG are clinically important because they are major treatable risk factors for atherosclerosis.
Lipoproteins:
Lipids are poorly water soluble, but are absorbed from the gastrointestinal tract and transported throughout the body. This is achieved by lipids by forming complexes known as lipoproteins.
Lipoproteins are spherical or disc-shaped and composed of:
- Hydrophobic core: Consisting of TG and cholesterol ester which is enveloped by a less hydrophobic surface coat
(mentioned below). - Less hydrophobic surface coat: Consisting of phospholipids, unesterified cholesterol and special proteins called apolipoproteins. Some apolipoproteins make these lipoproteins to act as enzyme co-factors or cell receptor ligands.
Types of Lipoprotein:
- Chylomicrons
- Very low-density lipoprotein (VLDL)
- Intermediate-density lipoprotein (IDL)
- Low-density lipoprotein (LDL): Lipoprotein [Lp(a)] is a type of lipoprotein similar to LDL with the addition of apolipoprotein (a)/Lp(a) links lipid metabolism with blood coagulation.
- High-density lipoprotein (HDL).
VLDL particles containing more cholesterol ester are more atherogenic than LDL particles.
Transport and Storage of Lipids:
Three main pathways involved in metabolism of lipid are:
1. Exogenous (dietary) lipid pathway:
- Dietary lipids are digested and absorbed in the small intestine and form chylomicrons in the epithelial cells of the intestines. Chylomicrons are the largest lipoprotein particle containing TG and cholesterol ester.
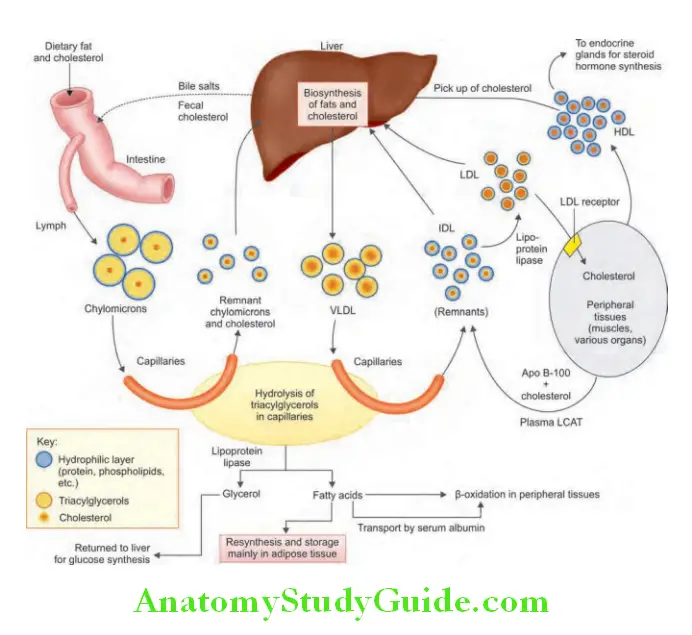
- Chylomicrons enter the blood circulation and are hydrolyzed by lipoprotein lipase (LPL) located on the endothelium of tissue capillary beds and release fatty acids. These fatty acids are utilized locally for energy production or stored as TG in muscle or fat.
2. Endogenous pathway:
- During fasting, the liver is the main source of plasma lipids and constitute endogenous pathway.
- TGs and cholesterol ester are produced by the liver and these lipids are secreted by the liver into the circulation as very low-density lipoproteins (VLDL). VLDL is also rich in TG but differ from chylomicrons in that they contain fulllength Apo B100.
- VLDL secreted into the circulation undergoes metabolic processing similar to that of chylomicrons. Hydrolysis of VLDL TG by capillary LPL releases fatty acids to tissues (utilized by fat and muscle) and remaining lipid portion of VLDL is converted into ‘remnant’ particles termed as intermediate-density lipoproteins (IDL).
- Major part of IDL is taken up and quickly cleared by LDL receptors in the liver. Some of IDL is processed by hepatic lipase, which converts the IDL to LDL by removing TG.
- LDL is an atherogenic lipoprotein. LDL particles carry the majority of the cholesterol in the blood and are a source of cholesterol for cells and tissues.
- Regulation of plasma LDL concentration: LDL cholesterol is taken up by receptor-mediated endocytosis through the LDL receptor in the peripheral cells or liver.
- Controls the rate of cholesterol synthesis: The supply of cholesterol through this pathway down-regulates further expression of the LDL receptor gene. This in turn decreases the synthesis and activity of the rate-limiting enzyme for cholesterol synthesis, HMGCoA reductase. This negative feedback loop, along with the modulation of cholesterol esterification, controls the intracellular free cholesterol level.
3. Reverse cholesterol transport:
- It is the process in which cholesterol is removed from the peripheral tissues and is returned to the liver. This protects against excessive cholesterol accumulation in peripheral tissues.
- High-density lipoproteins (HDL) are the main lipoprotein along with lipid-poor Apo A1 (derived from the liver, intestine and the outer layer of chylomicrons and VLDL) involved in his reverse cholesterol transport process. HDL is produced and catabolized in the liver and intestines.
- HDL is able to accept more free cholesterol from atherogenic lipoproteins and peripheral tissues to the liver. A circulating enzyme called lecithin cholesterol acyltransferase (LCAT) esterifies these HDL.
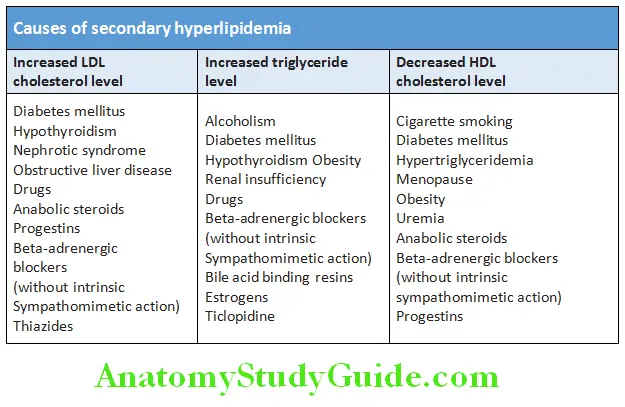
Hyperlipidemia:
Question 191. Write short essay/note on hyperlipidemias, their causes, and management.
Answer:
Hyperlipidemia is characterized by an abnormality in the lipid profile, consisting of a variety of disorders with raised total cholesterol, LDL or TG, or conversely, lower levels of HDL.
Classifiation and Causes:
- Primary hyperlipidemia: It is a genetic disorder of lipid metabolism (e.g., familial hypercholesterolemia). It is characterized by a genetic mutation, which causes impaired clearance of LDL from the circulation due to absence of LDL receptors.
- Secondar y hyperlipidemia: These are characterized by hyperlipidemia secondary to a disease/disorder other than the genetic defect
Management Strategies:
- Lipid profile of an individual should be interpreted along with the risk factors for CAD.
- Major modifiable risk factors:
- Hypercholesterolemia
- Smoking
- Diabetes Mellitus
- Hypertension as per
- Framingham Risk Score (FRS). It provides the risk of an individual and developing a cardiac event in the next 10 years.
- Emerging risk factors: That may contribute to the risk of CAD include:
- Apolipoprotein B
- Lipoprotein (a)
- C-reactive protein
- Hyperhomocysteinemia
- Small, dense LDL particles
- CAD equivalents: These include:
- Diabetes mellitus
- Patients with FRS ≥20%
- In these patients, the goals of treatment are same as those for patients with established CAD.
- LDL cholesterol goals in different categories.
- Therapeutic drug classes, their effect on lipid profile.
Newer agents:
- Torcetrapib is a cholesteryl ester transfer protein (CETP) inhibitors.
Coronary heart disease risk based on risk factors other than the LDL level:
Positive risk factors:
- Sex: Male ≥45 years and female ≥55 years or postmenopausal without
estrogen replacement therapy - Family history of premature coronary heart disease: Myocardial infarction or sudden death before age 55 in father or other male first-degree relative or before age 65 in mother or other female first-degree relative)
- Current cigarette smoking
- Hypertension: Blood pressure ≥140/90 mm Hg or patient is receiving antihypertensive drug therapy
- HDL cholesterol level <35 mg per dL (<0.90 mmol per L)
- Diabetes mellitus
- Negative risk factor: High HDL cholesterol level (≥60 mg per dL [≥1.60 mmol per L])
Hyperlipidemia Management:
- Therapeutic lifestyle changes.
- Patients with normal-weight, dyslipidemia [body mass index (BMI) 18.5–24.9 kg/m ²]: Focus on healthy eating and regular exercise.
- Overweight and obese patients (BMI ≥25 kg/m 2): Reduce caloric intake from fats, simple carbohydrate and ≥30 mins physical activity most days.
- Intake of soluble fiber to be increased (10–25 g/day).
- Diet (rich fruits, veg, nuts, whole grains, monounsaturated oils; low red meat, animal fat) reduces LDL 5–15% (ATP III TLC diet).
- Aerobic exercise: Running, walking, cycling, swimming enhance weight reduction facilitates achieving optimum lipid levels.
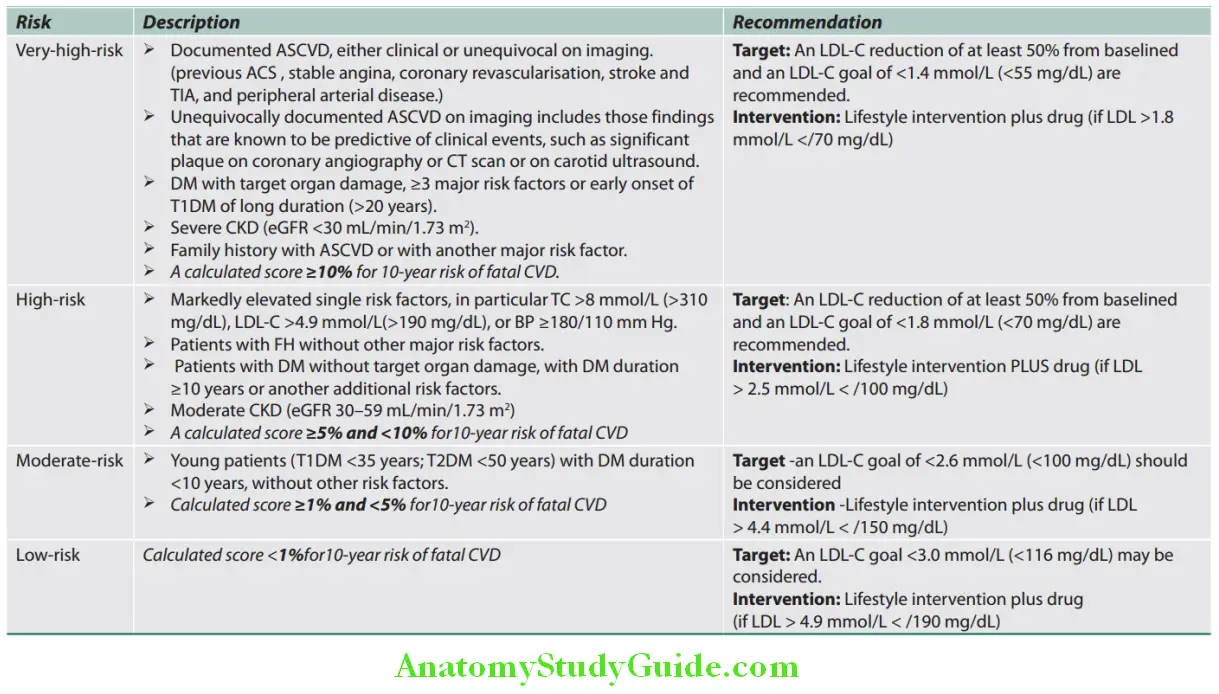
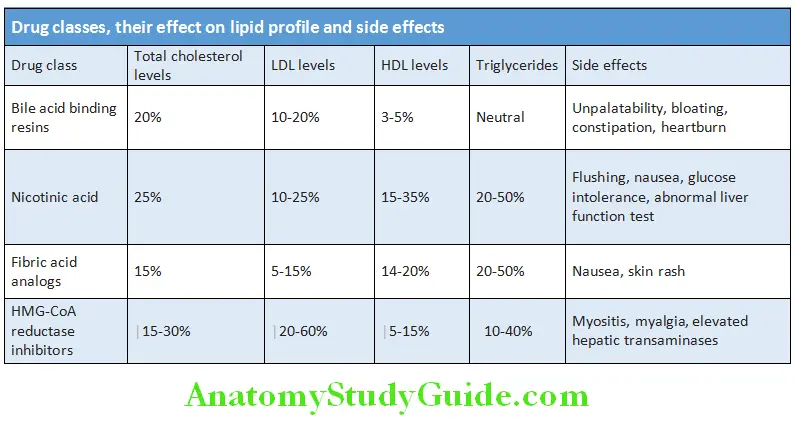
- PCSK9 inhibitors:
- Monoclonal antibodies have been developed that lower LDL-C. They work by inhibiting the Proprotein convertase subtilisin/kexin type 9 (PCSK9) enzyme, which is involved in breaking down the LDL-C receptor.
- Two PCSK9 inhibitors are approved. Evolocumab is dosed at 140 mg subcutaneously every 2 weeks or 420 mg sc every 4 weeks. Alirocumab is dosed at 75 or 150 mg every 2 weeks or 300 mg every 4 weeks.

ASCVD Risk:
ASCVD stands for atherosclerotic cardiovascular disease, defined as a nonfatal myocardial infarction (heart attack), coronary heart disease death, or stroke. It estimates the risk of ASCVD within a 10-year period among patients without pre-existing cardiovascular disease who are between 40 and 79 years of age who have never had one of these events in the past. This risk estimate considers age, sex, race, cholesterol levels, blood pressure, medication use, diabetic status, and smoking status.
Moderate- or high-intensity statin therapy is recommended for
- Individuals with clinical ASCVD
- Individuals with primary elevations of LDL ≥190 mg/dL
- Individuals 40 to 75 years of age with diabetes and an LDL 70 to 189 mg/dL without clinical ASCVD
- Individuals without clinical ASCVD or diabetes who are 40 to 75 years of age with LDL 70 to 189 mg/dL and a 10-year ASCVD risk of 7.5% or higher.
Lipodystrophy:
Question 192. Write a short note on lipodystrophy.
Answer:
Lipodystrophy is changes in the distribution body fat. Lipoatrophy implies partial or complete decrease of fat. However, the terms lipoatrophy and lipodystrophy are used interchangeably in clinical practice.
Classifiation of Lipodystrophies:
- Total lipodystrophy: Two types namely congenital and acquired.
- Berardinelli-Seip (congenital): Autosomal recessive, hypermetabolic state.
- Seip-Lawrence (acquired): Begins <15 years old, often <5 years
- Partial lipodystrophy:
- Kobberling-Dunnigan syndrome: Inherited form, autosomal dominant
- Progressive partial lipodystrophy: Acquired forms
- Centrifugal: One type is seen primarily in infants from Japan. 90% are under 5 years. Centrifugally spreading loss of abdominal fat over 3–8 years with regional lymphadenopathy. Resolves completely after progression stops.
- Semicircular: Adult women affected with single or multiple, asymptomatic, symmetric depressions of anterolateral thigh. Often after trauma, resolves in several years.
- Annular atrophic panniculitis: 10 cm band of atrophy, bilaterally, around the ankles of children and young adults. Rare.
- Lipoatrophia annularis: Bracelet-like constrictions of upper extremities 1–2 cm wide on women following a period of swelling and erythema of extremity. Arthralgias and pain of affected extremity. Persist up to 20 years.
- Localized lipodystrophy: Secondary to injection of medications especially insulin.
- HIV-associated lipodystrophy:
- Occurs in effectively trebated AIDS patients with reverse transcriptase inhibitors and protease inhibitors.
- Fat redistribution from face, buccal, buttocks and limbs is lost to neck, upper back and inter abdominal areas.
- Peripheral lipodystrophy, central fat accumulation and lipomatosis.
Question 193. Write short note on indications for low-dose aspirin.
Answer:
Indications for low-dose aspirin:
- Secondary prevention of cardiovascular disease: CAD (coronary artery disease, stroke, post-CABG (coronary artery bypass grafting)
- Primary prevention of ischemic heart disease
- Transient ischemic attacks (TIA)
- Antiphospholipid antibody (APLA) syndrome
- Pre-eclampsia
- Essential thrombocytosis, polycythemia vera
- Venous thromboembolism—prophylaxis
Straight Back Syndrome:
Question 194. Write short essay/note on straight back syndrome (SBS).
Answer:
- In straight back syndrome, there is loss of normal curvature upper thoracic spinal which reduces the anteroposterior diameter of the chest. This deformity compresses or “pancakes” the heart (cardiomegaly) and great vessels so as to appear enlarged. There is displacement of the heart toward left, resulting in cardiac murmurs.
- Clinical features: Chest pain, tracheal compression and mitral valve prolapse (MVP).
- Chest X-ray: Lateral chest X-ray reveals significant reduction of distance from the middle of the anterior border T8 to a vertical line connecting T4 and T12. A value less 1.2 cm is diagnostic.
- ECG often shows right bundle branch block.
Cardiac Tumors:
Myxoma:
- Most common type of primary cardiac tumor in adults seen in third to sixth decade of life.
- Most common in females.
- Most are sporadic, but some are familial (autosomal dominant)–10%.
- Most are solitary and located in left atrium arising from inter atrial septum near fossa ovalis.
- Its presentation most commonly mimics that of mitral valve disease.
- “Tumor plop” is a characteristic low-pitched sound which may be audible during early or mid-diastole and is thought to result from the tumor abruptly stopping as it strikes the ventricular wall.
- Symptoms and signs: Fever, weight loss, cachexia, malaise arthralgia, rash, clubbing, and Raynaud’s phenomenon. Peripheral or pulmonary embolization can develop.
- Investigations:
- Anemia, polycythemia, elevated ESR, thrombocytopenia or thrombocytosis.
- 2-D echocardiography: Useful screening tool
- CT and MRI are investigations of choice
- Treatment is surgical excision using cardiopulmonary bypass. Rhabdomyomas are the commonest cardiac tumor in children.
Digoxin:
Question 195. Write short essay/note on digoxin, manifestations of digoxin/digitalis toxicity, and its management.
Answer:
Digoxin is a purified glycoside derived from Digitalis lanata having cardiac inotropic property.
Pharmacological Actions:
Force of myocardial contraction is increased by a direct action of digitalis.
- Heart rate: Decreased and bradycardia is more marked in CHF.
- Electrophysiological properties:
- Prolongs the refractory period of V node → slows the ventricular rate.
- Reflex vasodilation in CHF.
Pharmacokinetics:
- Has a good oral absorption.
- Digoxin concentration in the heart is 30 times more than in the plasma.
- The elimination half-life for digoxin is 36–48 hours in patients with normal or near-normal renal function. This permits once-a-day dosing; near steady-state blood levels are achieved one week after initiation of maintenance therapy.
- About 85% of digoxin is excreted through the kidneys and the remaining 15% through the stool.
Indications:
- Cardiac arrhythmias: Supraventricular tachycardia, tachyarrhythmias and atrial fibrillation with a fast ventricular rate.
- Heart failure with reduced ejection fraction (HFrEF)
- Heart failure accompanied by atrial fibrillation or flutter with a rapid ventricular rate.
Dosage and Route of Administration:
The dosage and route is determined based on the desired action.
- Rapid digitalizing (loading dose) regimen:
- Intravenously: Initial loading dose of 0.25–0.5 mg followed by 0.25 mg every 6 hour. Careful monitoring of clinical response and toxicity should be performed before each dose.
- Orally: Initial loading dose is 0.5–1 mg followed by 0.25 mg 6 hourly. Careful monitoring of clinical response and toxicity before each dose.
- Slow digitalization: Maintenance dose (0.125–0.25 mg/day) given from the beginning. Dose may be increased every 2 weeks depending on clinical response, serum levels of the drug, and toxicity.
- As per ACCF/AHA guidelines, a loading dose to initiate digoxin therapy in patients with heart failure is not required.
Digoxin Toxicity and its Management:
Digitalis has high toxicity low margin of safety. Definite toxicity defined as nausea/vomiting, with cardiac effects and resolution of side effects on discontinuation. Serum digoxin concentration (SDC) is not a necessary criterion but SDC >2.0 ng/mL helpful.
Manifestations of Digoxin Toxicity:
- Extracardiac manifestations
- Nonspecific but more common
- Gastrointestinal (60–80%): Nausea/vomiting, anorexia, abdominal pain, diarrhea
- Malaise (30–40%), lethargy, fatigue
- Neurological (20–30%): Dizziness, confusion, headache, visual changes (flashing lights, halos, color disturbances in green-yellow spectrum, blurred vision)
- Cardiac side effects
- More specific but less common
- Almost any permutations and combinations of heart block (partial to complete), brady and tachydysrhythmias can be produced.
- Classical: Paroxysmal atrial tachycardia, ventricular bigeminy, bidirectional ventricular tachycardia, nodal and ventricular extrasystoles.
- ECG changes of digoxin toxicity: Inverse check mark with proximal ST segment depressed, in leads other than those with tall R waves, with T wave not rising above baseline (Inverted T), with shortened QTc.
Management of Digoxin Toxicity:
- Stop digoxin.
- Blood test: Urea, electrolytes and plasma digoxin concentration.
- Correction of hypokalemia: Intravenous supplementation of no more than 20 mmol/h of potassium. More rapid infusion may cause asystole.
- Heart blocks: Atropine and temporary cardiac pacing.
- Malignant ventricular arrhythmias
- Phenytoin (100 mg intravenously, repeated after 5 minutes if required) opposes digitalis binding and may improve atrioventricular conduction by its anticholinergic properties.
- Ultra-short-acting beta blockers, e.g., esmolol.
- Avoid cardioversion wherever possible, due to the risk of precipitating asystole.
- Digoxin-specific antibody fragments: (Digibind) are the most effective but expensive.
- Hence, reserved for treatment of serious toxicity, especially in the presence of malignant cardiac dysrhythmias.
Valsalva Maneuver:
Question 196. What is the Valsalva maneuver?
Answer:
The Valsalva maneuver is a forceful attempted exhalation against a closed glottis.
Valsalva Maneuver Method:
- Instruction: Take a deep breath, close your mouth and pinch your nose with the thumb and index finger and attempt to breathe out gently, keeping your cheek muscles tight, not allowing the air to escape by keeping the lips pursed.
- “Standard” or “quantitative:” Blowing out with an open glottis into a tube of a sphygmomanometer against the pressure of 40 mm Hg.
Phases of Valsalva Maneuver:
Physiological effects on blood pressure heart rate and phases of Valsalva maneuver are presented.
Phase 1: The onset of blowing. The pressure within the chest and abdomen increases and presses upon the arteries in the chest, which results in an increase in mean arterial blood pressure. This activates the baroreceptor reflex, which results in an increase in parasympathetic (vagal) activity and hence in a drop in heart rate. The increased intrathoracic pressure also reduces the amount of blood that comes into the right atrium (decreased venous return or preload).
Phase 2: A decrease of venous return results in a lower amount of blood that is ejected from the heart, which results in a decrease of central venous pressure and consequently in a decrease of mean arterial blood pressure. This activates the baroreflex, which results in a decrease of the parasympathetic (vagal) activity and consequent increase of the heart rate, and in an increase in sympathetic activity, which constrict the arteries (an increase of peripheral resistance), which results in a slight rise of the blood pressure at the end of phase 2 (2b).
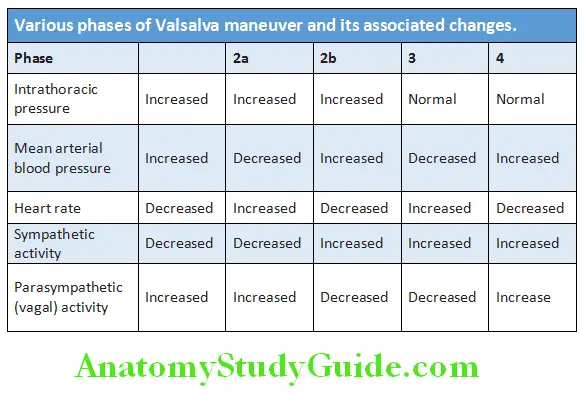
Phase 3: Relaxation—the end of the maneuver. The intrathoracic pressure decreases, so the intrathoracic arteries widen, which results in a brief drop in blood pressure. At the same time, the venous blood fills the heart.
Phase 4: The heart ejects the blood into the arterial system against increased peripheral resistance (which has developed in phase 2), so the blood pressure rises again (blood pressure overshoot). This activates the baroreflex, which results in a drop in heart rate (bradycardia). Eventually, both the blood pressure and heart rate normalize.
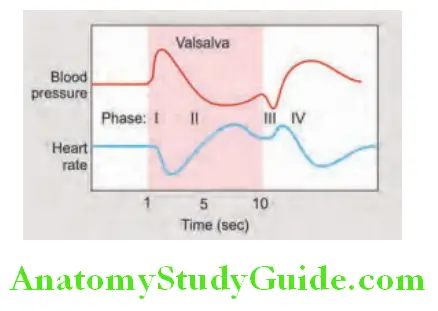
Valsalva Maneuver Uses:
- Eustachian tube dysfunction
- Heart murmurs: Valsalva increases murmurs in hypertrophic cardiomyopathy and mitral valve prolapse and decreases them in atrial septal defects and aortic stenosis.
- Congestive heart failure: Valsalva responses lost
- Function of the autonomous nervous system: An abnormal blood pressure response (for example, an absence of the blood pressure rise in phase 4) suggests an abnormality of the sympathetic system.
- An abnormal heart rate response suggests an abnormality of the parasympathetic Valsalva maneuver can be used as a provocative test to check for:
- Neurogenic orthostatic hypotension
- Chiari malformation, the Valsalva maneuver (coughing) triggers a headache at the back of the head.
- Diagnosis of inguinal hernia, prolapse of the uterus, bladder or vagina, varicocele and intrinsic sphincter deficiency in stress urinary incontinence system.
- Valsalva maneuver can help: Equalize the pressure between the middle ear and the ambient pressure during scuba diving, driving from a steep hill, elevator descending, parachuting or plane landing or in individuals with Eustachian tube dysfunction.
Modified Valsalva maneuver: Modified Valsalva maneuver is used to terminate an attack of supraventricular tachycardia (SVT); it includes blowing against a closed glottis followed by lying down face up and raising legs with the help of an assistant, may be effective in 19–54% of cases.
Reversed Valsalva—Müller’s maneuver: Müller’s maneuver is the opposite of the Valsalva maneuver and includes forced exhalation followed by an attempted forceful inhalation with a closed mouth and nose or just with a closed glottis. The test can be used to evaluate weakness of the soft palate and throat walls in individuals with obstructive sleep apnea.
Cardiorenal Syndrome:
Question 197. List types of cardiorenal syndrome.
Answer:
There are five subtypes of cardiorenal syndrome:
Type 1: A sharp decline in cardiac function that results in an acute decrease in renal function, e.g., cardiogenic shock.
Type 2: Chronic cardiac dysfunction that results in a sustained reduction in renal function, e.g., chronic heart failure resulting in nephropathy.
Type 3: A sharp decline in renal function that results in an acute reduction in cardiac function, e.g., acute kidney injury.
Type 4: Chronic decline in kidney function that results in chronic cardiac dysfunction, e.g., chronic kidney disease.
Type 5: Systemic diseases that result in both cardiac and renal dysfunction, e.g., diabetes, hypertension.
Leave a Reply