Kidney
Functional Anatomy Of Kidney
Question 1. Describe the nephron with the help of a diagram. What are the functions of the kidney?
Answer:
Table of Contents
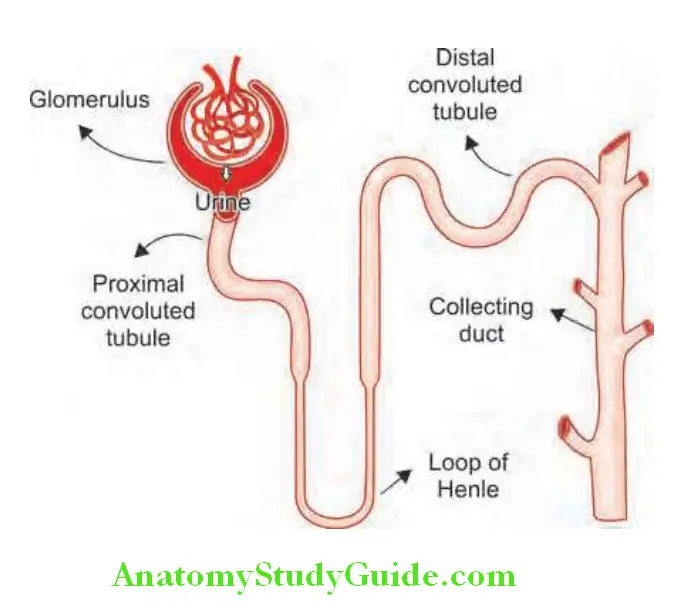
Unit of Kidney:
The nephron is the structural and functional unit of the kidney. It consists of the glomerulus and its tubule, and the common collecting system. Each kidney consists of about 1 million nephrons.
Read And Learn More: General Medicine Questions and Answers
Different Components Of A Nephron:
Functions Of Kidney:
- Excretion of many metabolic breakdown products (including ammonia, urea, and creatinine from protein, and uric acid from nucleic acids), drugs, and toxins
- Regulation of water and electrolyte balance
- Maintenance of acid-base balance
- Reabsorption of essential substances
- Secretion of hormones such as erythropoietin and renin
- Metabolism of vitamin D
- Regulation of blood pressure
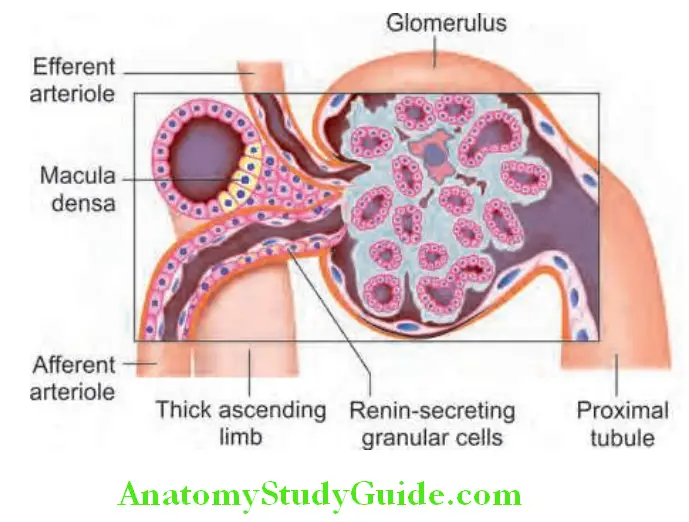
Juxtaglomerular Apparatus:
Question 2. Write a short note on the juxtaglomerular apparatus.
Answer:
Components of juxtaglomerular apparatus: It consists of macula densa, the extraglomerular mesangium, the terminal portion of the afferent glomerular arteriole (contains renin-producing granular cells), and the proximal portion of the efferent arteriole.
- Macula densa: The afferent arterioles and the thick ascending limb of loop of Henle are in contact for a short distance. The macula densa is a plaque of tall and columnar cells within this thick ascending limb of the loop of Henle and contains large, tightly packed cell nuclei (hence termed macula densa).
- This anatomical arrangement allows changes in the renal tubule to influence the behavior of the adjacent glomerulus (tubuloglomerular feedback).
- Extraglomerular mesangium.
- Terminal portion of the afferent glomerular arteriole: Shows thickening due to specialized myoepithelioid cells juxtaglomerular cells) that contain large secretory granules of renin.
- The proximal portion of the efferent arteriole.
Renin:
Renin is secreted and stored in the juxtaglomerular cells.
Factors Controlling the Release Of Renin:
- Pressure changes in the afferent arteriole
- Sympathetic tone
- Sodium, chloride, and osmotic concentration of the fluid in the distal convoluted tubule at the macula densa
- Local prostaglandin and nitric oxide release.
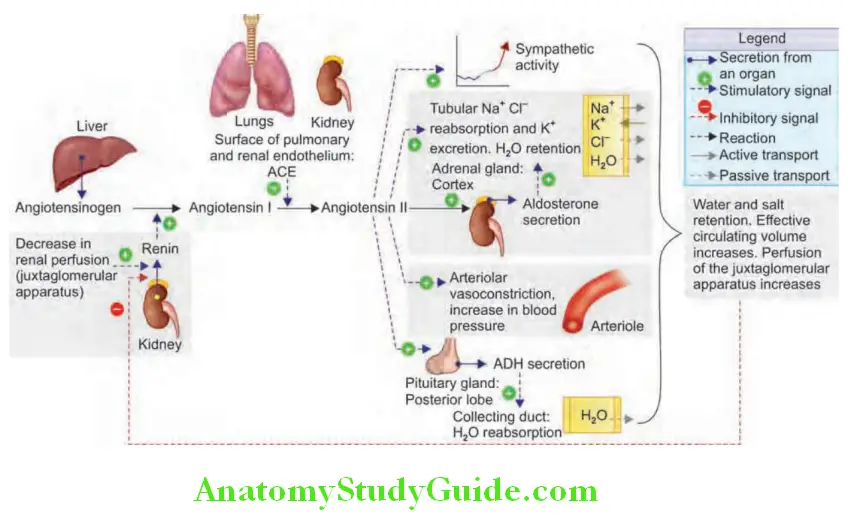
Mechanism of action of renin:
- Renin acts on angiotensinogen in the blood and converts angiotensinogen to angiotensin I.
- Angiotensin I (decapeptide) is converted to angiotensin II (octapeptide) by angiotensin-converting enzyme (ACE), which is present in the lung, luminal border of endothelial cells, glomeruli, and other organs.
- Angiotensin II has two major systemic effects: Systemic vasoconstriction and sodium and water retention by release of aldosterone from the adrenal cortex.
- Aldosterone produces constriction of the efferent arteriole of the glomerulus and thereby increases glomerular filtration pressure.
- Consequences of renin action
- By the above-mentioned mechanisms, the kidneys “defend” circulating blood volume, blood pressure, and glomerular filtration during circulatory shock.
- However, the same mechanisms can lead to systemic hypertension in renal ischemia.
Renin-angiotensin-aldosterone system (RAAS) is presented:
Approach To Renal Diseases:
Azotemia:
Azotemia Definition: It is a biochemical abnormality characterized by an elevation of the blood urea nitrogen (BUN) and creatinine levels. It is mainly due to a decreased glomerular filtration rate (GFR).
Azotemia Causes: Azotemia can be divided into prerenal, renal, and postrenal (discussed later under the AKI section).
Glomerular Filtration Rate (GFR):
- Measurement of the GFR is required to know the exact level of renal function. It is necessary to calculate GFR when the serum (plasma) urea or creatinine is within the normal range.
- The glomerular filtration rate for an average adult is about 125 mL/minute. The formula for the calculation of GFR is presented.
- The most common methods utilized to estimate the GFR are—measurement of the creatinine clearance and estimation equations based upon serum creatinine such as the Cockcroft–Gault equation, and the Modification of Diet in Renal Disease (MDRD) study equations.
Azotemia Measurement:
- Renal clearance = [latex]\frac{UV}{P}[/latex]
- C = renal clearance, U = urinary concentration of any substance, P = plasma concentration of the same substance,
- V = minute volume of urine.
Inulin Clearance:
- It is the gold standard but is not practical.
- Inulin is a polysaccharide that passes freely through the glomerular capillary wall. It is neither absorbed nor excreted by the tubules and hence, the quantity of inulin excreted in urine (UV) is identical to the amount filtered by the glomeruli. Therefore, the renal clearance of inulin can be used to measure GFR.
Creatinine Clearance:
Question 3. Write a short note on creatinine clearance and mention the formula for its calculation.
Answer:
- The measurement of creatinine clearance, which approximates to that of inulin is the most commonly used for measurement of GFR. Other substances that are used are iohexol, iothalamate, 51Cr-EDTA
- Principle: Creatinine clearance is based on the fact that daily production of creatinine (mainly from muscle cells) is remarkably constant and little affected by protein intake. Thus, serum creatinine and urinary output vary very little throughout the day and renal creatinine clearance given an estimate of the glomerular function of the kidney.
- Cockroft–Gault equation: If serum creatinine level is a table, creatinine clearance (hence GFR) can also be calculated by using the Cockroft–Gault formula
Formulas used to estimate eGFR/creatinine clearance (CrCl):

Cystatin C:
Cystatin C is filtered at the glomerulus and not reabsorbed. The serum cystatin C concentration may correlate more closely with the GFR than the serum creatinine concentration.
Renal Function Tests:

Examination of the Urine:
Question 4. Write a short essay on routine examination of urine.
(or)
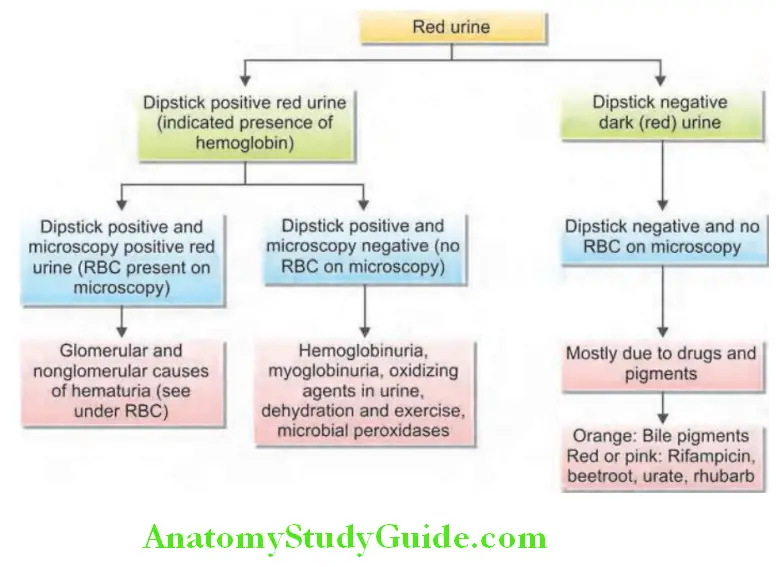
Write a short note on the causes of red-colored urine.
Answer:
Physical Examination
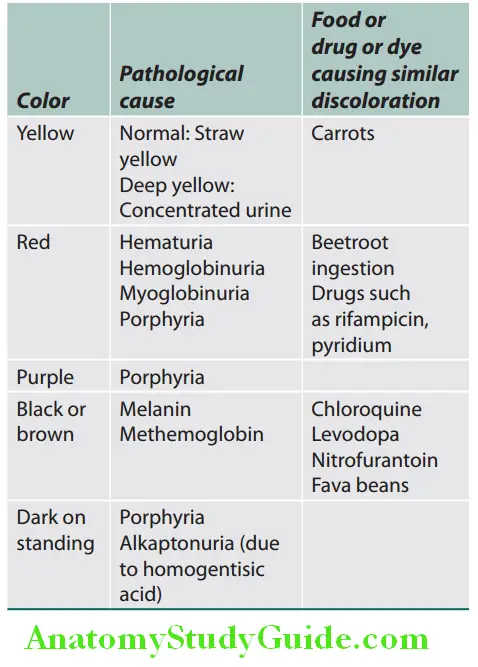
Appearance: Color and clarity
- Normal urine is straw to amber colored due to the presence of urochrome pigment, excretion of which is generally proportional to the metabolic rate.
- Urine color interpretation .
- Approach to red urine
- Clarity
- Cloudy or turbid urine is most commonly associated with urinary tract infections (UTIs). Turbid white urine is sometimes called as albinuria.
Causes of turbid urine or albinuria:
- Chyluria
- Filariasis, schistosomiasis, postsurgery, malignancy
- Hyperuricosuria
- Phosphaturia
- Hyperoxaluria
- Proteinuria
- Pyuria
- Lipiduria
- Caseous material from renal tuberculosis
- Congenital malformations of the lymphatic vessels
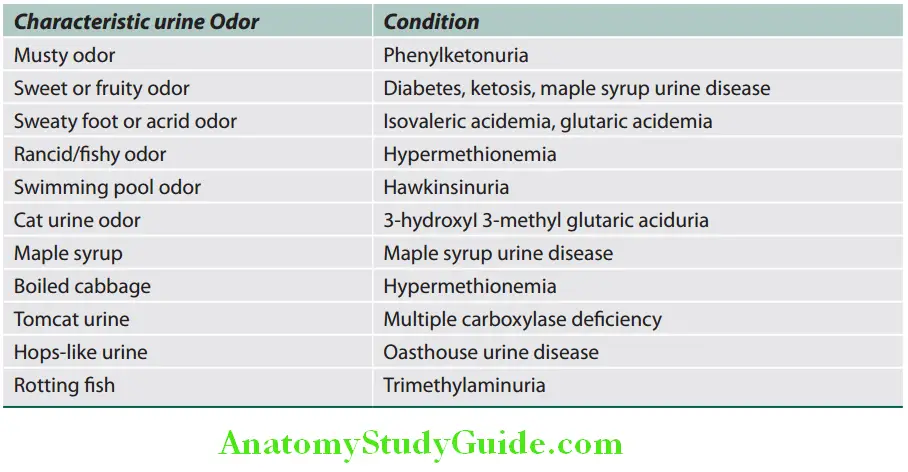
Odor:
The characteristic urine odor and condition.
Urine Volume:
A healthy adult excretes about 600–2,000 mL of urine in 24 hours. Volume is measured by collecting 24-hour urine samples in a measuring cylinder.
Question 5. Define oliguria, anuria, and polyuria. List the causes.
Answer:
- Oliguria: Decreased production of urine usually <400 mL of urine per day or less than 17 mL/hour. On an average diet, about 300–500 mL urine/day is required to excrete the solute load at maximum concentration.
- Causes of oliguria
- Anuria: Anuria is defined as urine output that is less than 100 mL/24 h or 0 mL/12 h. Anuria more commonly suggests reduced production of urine or obstruction to urine flow from both kidneys (until proved otherwise). Bladder outflow obstruction must always be considered first.
- Causes of anuria.
- Polyuria: Polyuria is defined as persistent large increase in urine volume of >3 L/day and 2 L/m2 in children. This term should exclude normal individuals who take large amount offluid and therefore, form large volumes of urine. Polyuria may be either due to increased urinary solute excretion (osmotic/ solute diuresis) or pure water diuresis
Causes of anuria:
- Obstruction:
- Bilateral ureteric obstruction
- Prostatic or urethral obstruction
- Renal stones
- Tumors
- Renal ischemia: Bilateral renal arterial or venous occlusion
- All causes of oliguria can lead to anuria
Causes of polyuria:
- Pathological polyuria:
- Increased excretion of solute (osmotic diuresis):
- Glucosuria—hyperglycemia, administration of mannitol, and hypercalcemia. Urea diuresis and sodium diuresis.
- Defective renal concentrating ability: Diabetes insipidus, papillary necrosis, and diuretic phase of ATN
- Failure of production of ADH: Idiopathic (50%), mass lesion, trauma, and infection
- Drugs/toxins: Diuretics, lithium, and alcohol
- Primary (or psychogenic) polydipsia: Excess fluid intake
Specific Gravity and Osmolality:
Urine specific gravity is used as a measure of the concentrating power of the kidney. It is a measure of the weight of dissolved particles in urine. Urine osmolality reflects the number of dissolved particles.
Normal specific gravity of a 24-hour urine sample is 1.003–1.035, average being 1.016.
Oliguria, Anuria Uses:
- Specific gravity provides information about the renal status and hydration.
- It is used only in the differential diagnosis of oliguric renal failure or the investigation of polyuria or inappropriate ADH secretion.
- Fixed specific gravity: When specific gravity is fixed at 1.010, this is known as isosthenuria. It is indicative of severe renal damage [chronic renal failure (CRF)/chronic kidney disease (CKD)] or acute tubular necrosis (ATN) with disturbance of both the concentrating and diluting abilities of the kidney.
Urinary pH:
- Normal urine is usually acidic with pH varying from 4.6 to Measurement of urinary pH is not necessary except in the investigation and treatment of renal tubular acidosis (RTA).
Chemical Examination:
Urine should be examined for the presence of protein, blood and sugar in all patients suspected of having renal disease.
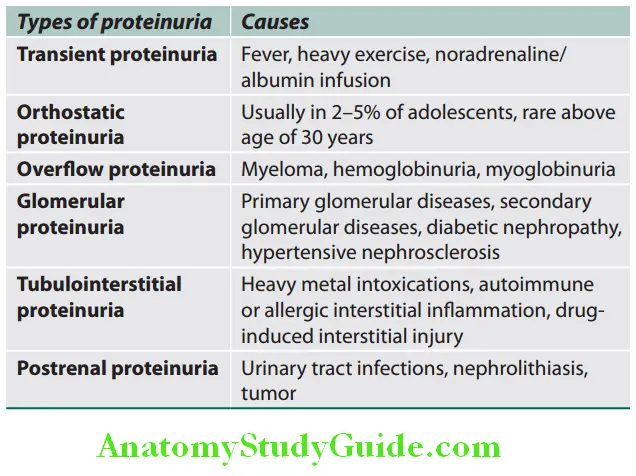
Proteinuria:
Question 6. Write a short note on proteinuria and microalbuminuria and their causes.
Answer:
- Healthy adults may daily excrete <150 mg of total proteins and <30 mg of albumin.
- Proteinuria is defined as the urinary excretion of >150 mg of protein/day. It is one of the most common signs of renal disease. Pyrexia, exercise, and adoption of the upright posture (postural proteinuria) may also increase urinary protein output.
The types of proteinuria and causes are described:
- Algorithm approach to proteinuria is presented.
- Amount of pathological proteinuria: It may be “mild” (<1.0 g/day), “moderate” (1.0–3.5 g/day) or “massive”/“heavy” (>3.5 g/day).
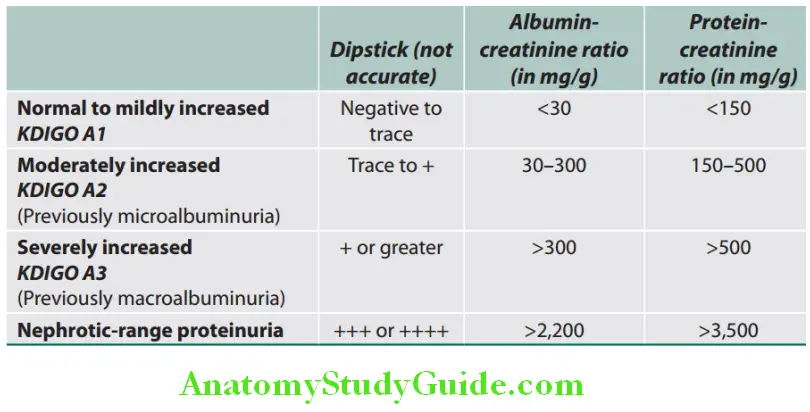
Microalbuminuria:
Normal urine contains <30 mg of albumin/day (<20 μg of albumin per minute).
Microalbuminuria Definition: Microalbuminuria is the presence of albumin (small amounts) in urine about >30 to <300 mg/day. It is defined as the persistent elevation of the urinary albumin excretion of 30–200 mg/L (or 20–200 mg/min) in an early morning urine sample. It indicates early and possibly reversible glomerular damage.
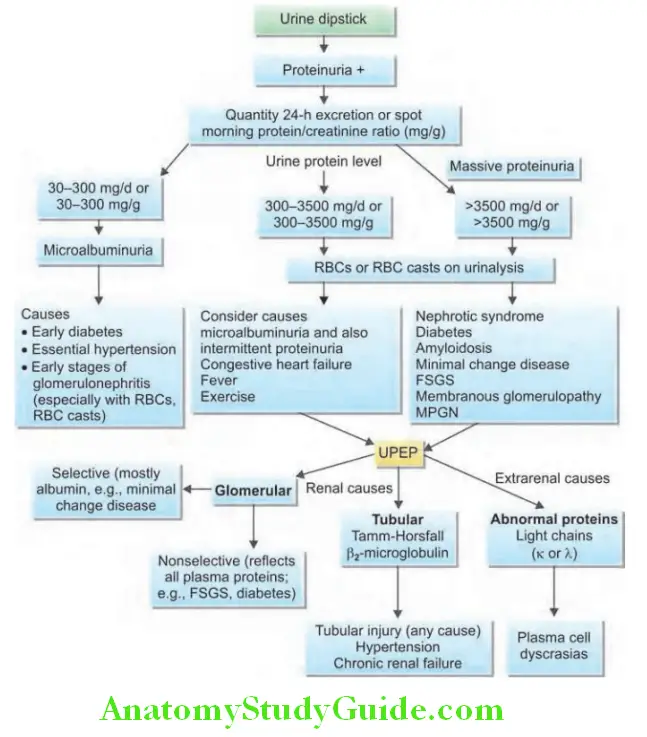
- It is so named because conventional dipsticks cannot detect albumin levels of 30–300 mg/day (if urine volume is normal).
- An increase in albumin excretion between these two levels is called microalbuminuria.
- Significance: The presence of albumin in the urine is a sign of glomerular abnormality.
- Diabetes mellitus:
- Microalbuminuria is an early indicator of diabetic glomerular disease. It is widely used as a predictor of the development of nephropathy in diabetics (raised fractional excretion of magnesium is a more sensitive marker than microalbuminuria in detecting early diabetic nephropathy).
- In diabetic patients, the presence of microalbuminuria is associated with increased cardiovascular mortality.
- Essential hypertension: In hypertensive patients, microalbuminuria predicts cardiovascular morbidity and mortality.
- Normotensive individuals:
- The risk marker for the presence of cardiovascular disease predicts the progression of nephropathy when it increases to frank albuminuria (>300 mg/day).
- Atherosclerosis: Persistent microalbuminuria is also associated with an increased risk of atherosclerosis and cardiovascular mortality.
Microalbuminuria Treatment:
- Microalbuminuria can be reduced, and its progress to overt proteinuria can be prevented or retarded by aggressive reduction of blood pressure (especially with ACE inhibitors or angiotensin receptor blockers), and control of diabetes mellitus.
- Blood pressure should be maintained at or below 130/80 mm Hg in patients with diabetes or kidney disease.
Albumin-Creatinine Ratio:
Measurement of 24-hour urinary excretion rates provides the most precise measure of microalbuminuria. However, it is often difficult to obtain 24-hour urine, it is more convenient to measure urinary albumin: creatinine ratio in a random urine sample and generally albumin-creatinine ratio (ACR) of 2.5–20 corresponds to albuminuria of 30–300 mg daily respectively. The adequacy of the collection can be estimated by quantifying the 24-hour urine creatinine and comparing this value to the expected urine creatinine.
Tamm–Horsfall Mucoprotein (Uromodulin):
Question 7. Write a short note on Tamm–Horsfall mucoprotein.
Answer:
- It is a protein present in normal urine produced in the thick ascending limb of the loop of Henle. It is excreted at a rate of 25 mg/day.
- The function is not known. Probably it may have some immunomodulatory activity and may protect against UTI.
- It is a constituent of all types of urinary casts. It is involved in the pathogenesis of cast nephropathy (observed in renal failure associated with multiple myeloma) in which intratubular casts occlude the flow of urine.
Hematuria:
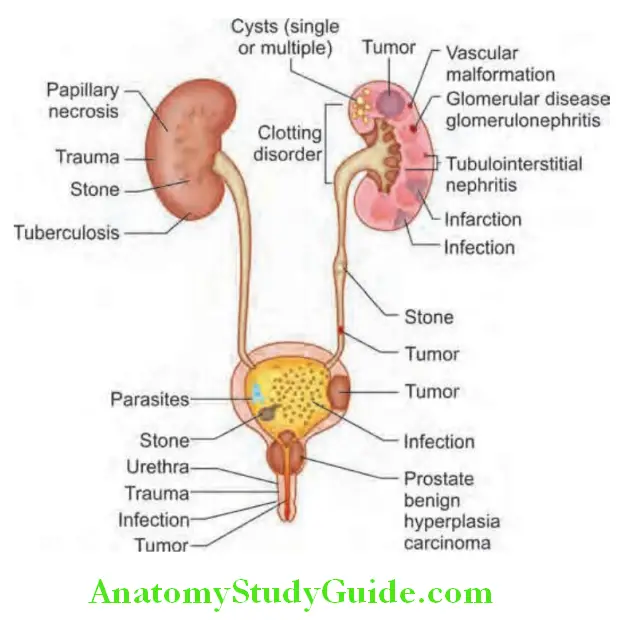
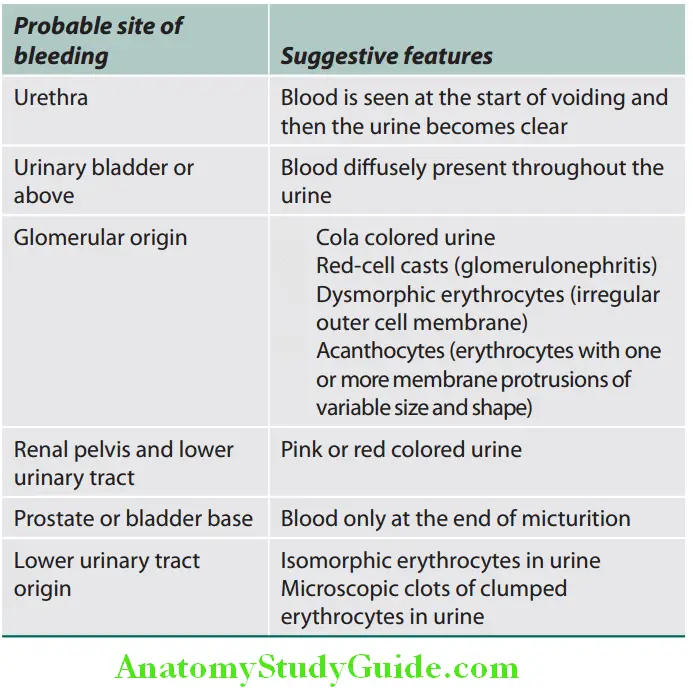
Question 8. Enumerate the common causes of hematuria/the causes of painless hematuria. How will you clinically localize and evaluate the site of bleeding?
Answer:
- Hematuria may be visible on gross examination and reported by the patient as bloody urine (macroscopic/overt hematuria), or invisible and detected on dipstick/chemical testing of urine [microscopic hematuria—three or more red blood cells (RBCs) per high-power field].
- Site of bleeding: Bleeding may occur at any site within the urinary tract and common causes of hematuria are illustrated. Features that may help to localize the site of bleeding in the urinary tract are mentioned.
Probable site of bleeding and its features:
Causes of Painless Hematuria:

Glycosuria:
- Blood glucose level varies between 70 and 120 mg/dL. This may increase to 120–160 mg/dL after a meal. Normally, all the glucose in the blood is filtered through the glomerulus and reabsorbed at the proximal tubules.
- If the renal threshold (the lowest blood glucose level that will result in glycosuria) is exceeded (usually >180–200 mg/dL), the excess glucose will not be reabsorbed into the blood and will be eliminated in the urine as in cases of diabetes mellitus. The presence of detectable amounts of glucose in urine is termed glycosuria.
Bacteriuria:
Dipstick tests used for the detection of bacteriuria detect nitrite produced from the reduction of urinary nitrate by bacteria. It is also for the detection of leukocyte esterase, an enzyme specific for neutrophils. Enterobacteriaceae species elaborate the enzyme nitrate reductase, which confers the ability to convert urinary nitrate to nitrite. So, nitrite-positive urine may indicate bacteriuria.
Microscopy:
Microscopic examination of urine should be done in all patients suspected of having renal disease, on a “clean” midstream sample.
Cells:
They are expressed as several cells per low-power or high-power field.
- Red blood cells: The presence of RBCs (more than 3/hpf) in the urine indicates bleeding at any point in the urinary system from the glomerulus to the urethra. In glomerular diseases, the urine show red cells with cellular protrusions or fragmentation and are named as dysmorphic (distorted morphology) RBCs.
- White blood cells: Increased number of WBCs (mainly neutrophils more than 5/hpf) in urine is known as pyuria. It is indicative of an inflammatory reaction within urinary tract such as UTI, stones, tubulointerstitial nephritis (TIN), papillary necrosis, tuberculosis, and interstitial cystitis. The causative organism of infection may be identified by bacteriological examination. When accompanied by leukocyte casts or mixed leukocyteepithelial cell casts, increased urinary leukocytes are considered to be of renal origin.
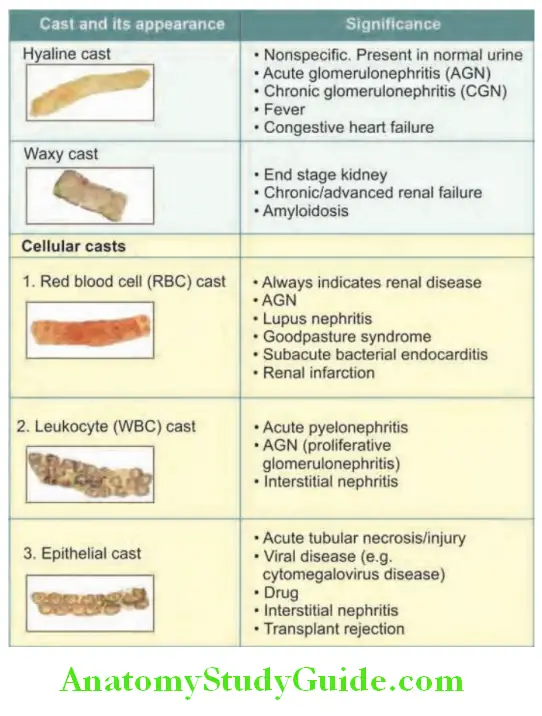
Casts:
Casts are one of the organized elements which are formed only in the kidney and are indicative of a renal disease. They are cylindrical bodies, molded in the shape of the distal tubular lumen formed by solidification of Tamm–Horsfall protein, a glycoprotein secreted in the distal convoluted tubules and collecting tubules. These proteins form a fibrillar meshwork (basic matrix) and can trap any elements including cells, cell fragments or granular material.
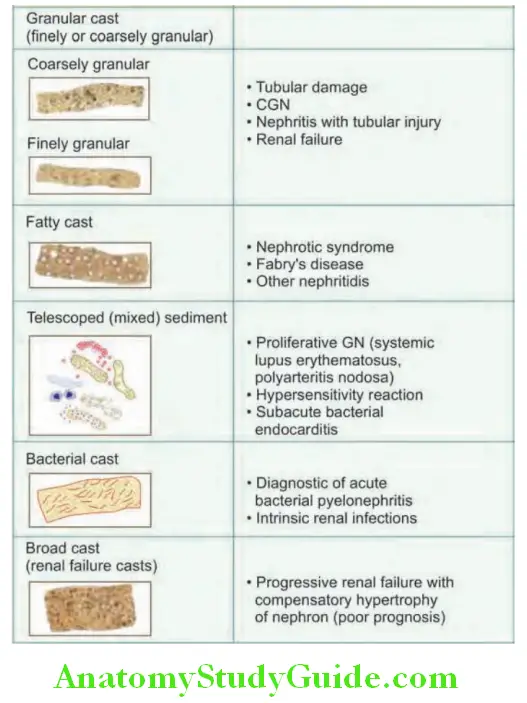
Types of urinary casts and their signifiance:
Hematuria Crystals:
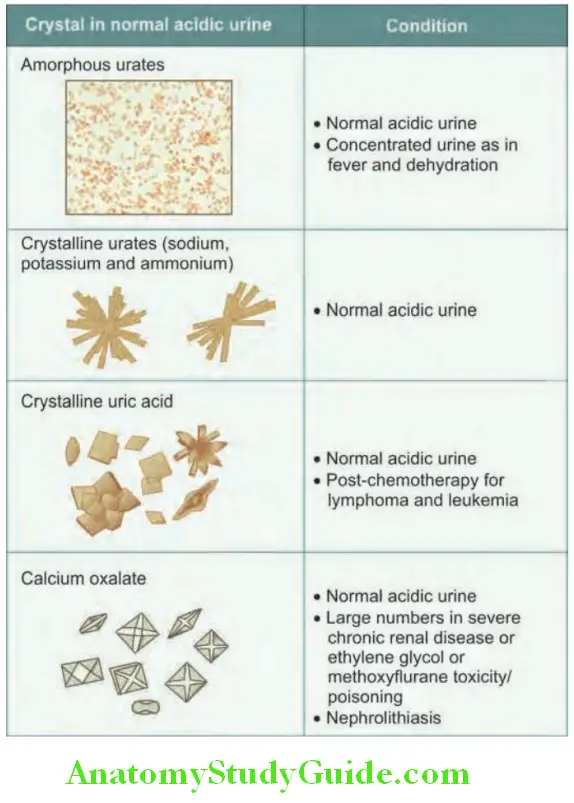
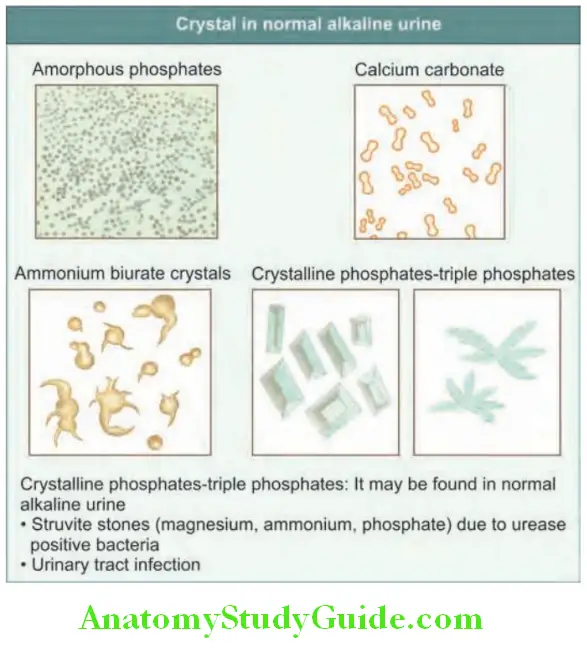

They may be found in patients with renal calculi. Calcium oxalate and urate crystals can be found in normal urine that has been left to stand.
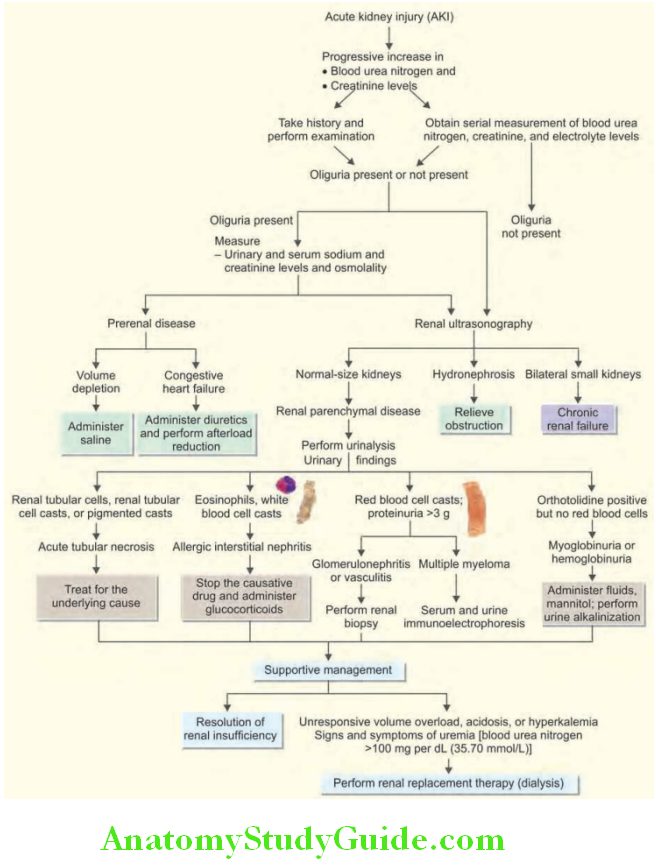
Acute Kidney Injury (Acute Renal Failure):
Question 9. Discuss the classification, causes, pathogenesis, clinical features, diagnosis, investigations, and management of acute kidney injury/acute renal failure.
Answer:
Renal failure is the failure of renal excretory function due to reduced GFR. Acute kidney injury (AKI) was previously known as acute renal failure (ARF). The other term used for this is azotemia.
Acute Kidney Injury Definition:
Acute kidney injury is a clinical syndrome, defined as an abrupt, deterioration of kidney function, which is usually, but not invariably, reversible over a period of days or rarely over a few weeks. The deterioration in renal function is sufficiently severe to result in the retention of nitrogenous wastes in the body (uremia) and other waste products normally cleared by the kidneys.
- It is usually but not invariably accompanied by oliguria.
- Conventionally the term ARF is often used in reference to the subset of patients with a need for acute dialysis support.
Classification of Acute Kidney Injury:
Acute kidney injury is a medical emergency. It may produce sudden, life-threatening biochemical disturbances. AKI includes an increase in serum creatinine by ≥0.3 mg/dL (27 µmol/L) within 48 hours or an increase to ≥1.5 times the presumed baseline value that is known or presumed to have occurred within the prior 7 days, or a decrease in urine volume to <3 mL/ kg over 6 hours [Kidney Disease: Improving Global Outcomes (KDIGO)-AKI].
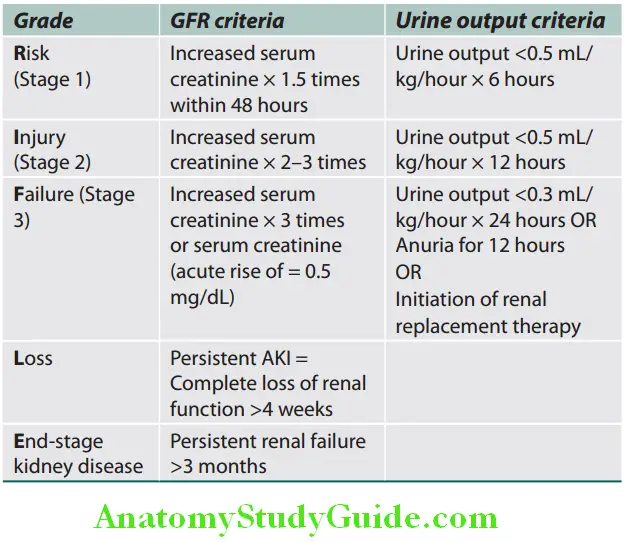
RIFLE Criteria:
- The distinction between acute and CKD or acute on chronic kidney disease, cannot easily be done in case of uremia. Acute
- The Dialysis Quality Initiative group proposed the RIFLE (Risk, Injury, Failure, Loss, End-stage renal disease) criteria to classify AKI.
- These criteria indicate an increasing degree of renal damage is of predictive value for mortality.
RIFLE criteria for the classification of acute kidney injury:
Acute Kidney Injury Network Classification:
Acute kidney injury network (AKIN) has proposed a modification of the RIFLE criteria. It includes less severe AKI, a time constraint of 48 hours, and gives a correction for volume status before classification. According to this, AKI is classified into three stages.
- Stage 1 is the same as the risk category of RIFLE with the addition of an increase in serum creatinine by 0.3 mg/dL within 48 hours.
- Stages 2 and 3 are the same as the injury and failure categories of RIFLE.

Etiopathogenesis:
Causes:
Question 10. Write a short essay/note on the causes of acute renal failure/acute kidney injury.
(or)
Define and enumerate the causes of acute kidney injury/azotemia.
Answer:
At rest, a normal kidney receives about 25% of the cardiac output. The etiology of AKI is diverse.
Etiopathogenesis Classification:
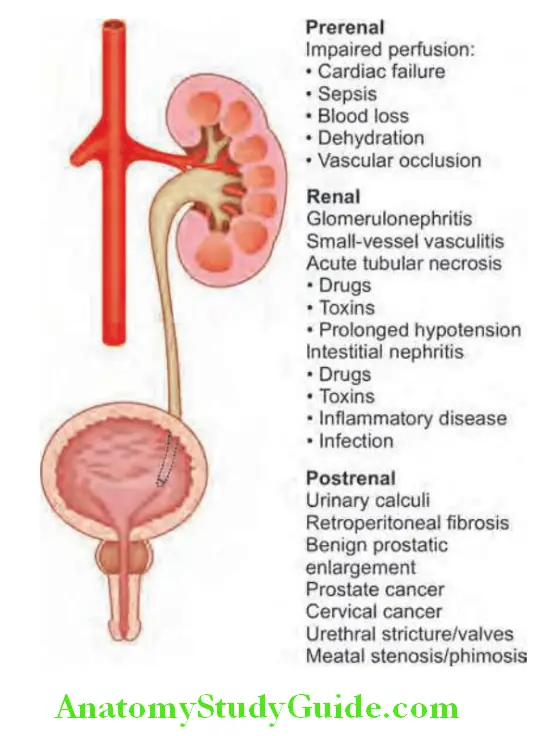
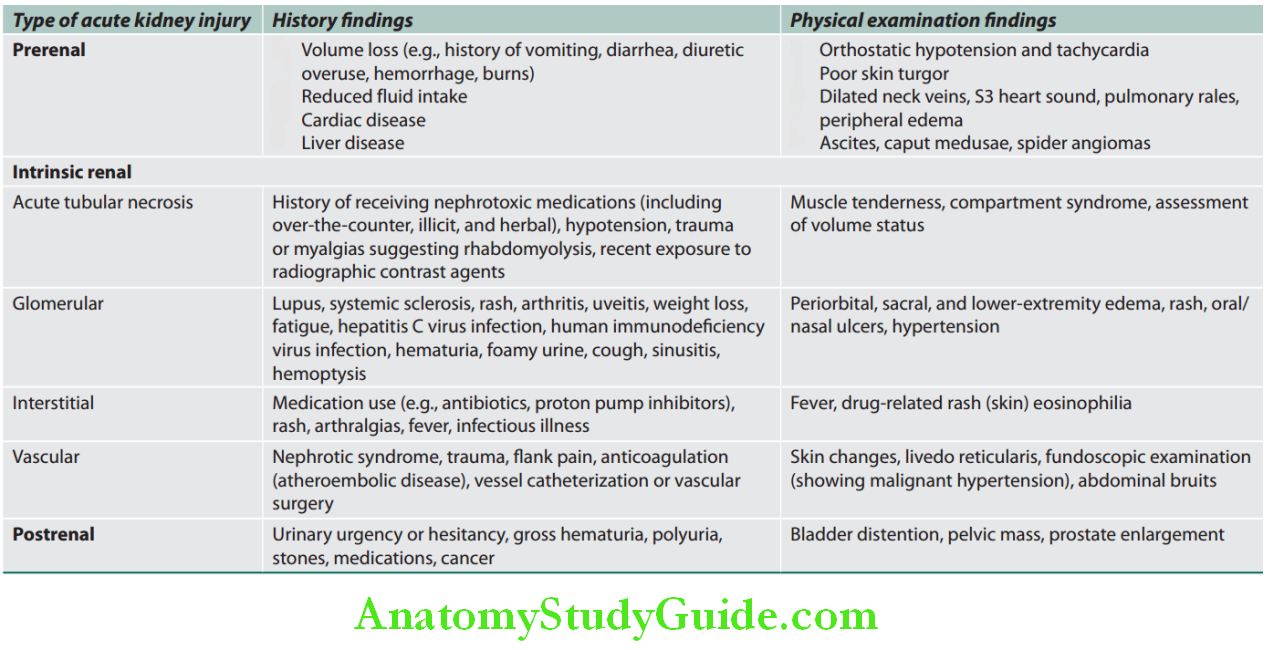
Causes of AKI are traditionally divided into three broad anatomical categories: Prerenal, intrarenal (intrinsic renal parenchymal disease), and postrenal causes. The most common cause is ATN followed by prerenal causes.
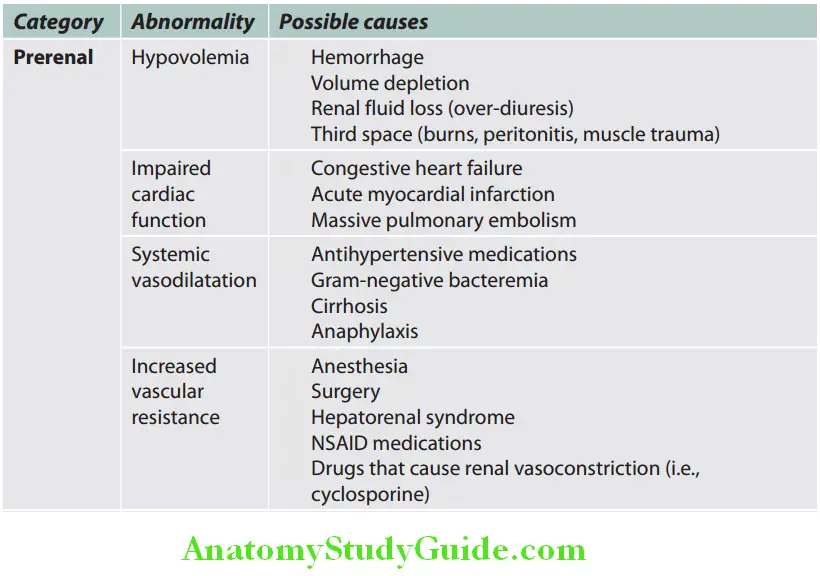
- Prerenal causes:
- The precipitating event is renal hypoperfusion which may be due to a reduction in the volume of extracellular fluid or disease states associated with decreased effective arterial volume or other causes.
- Kidneys are inadequately perfused and the GFR is markedly reduced and produces oliguria (urine output <400 mL)
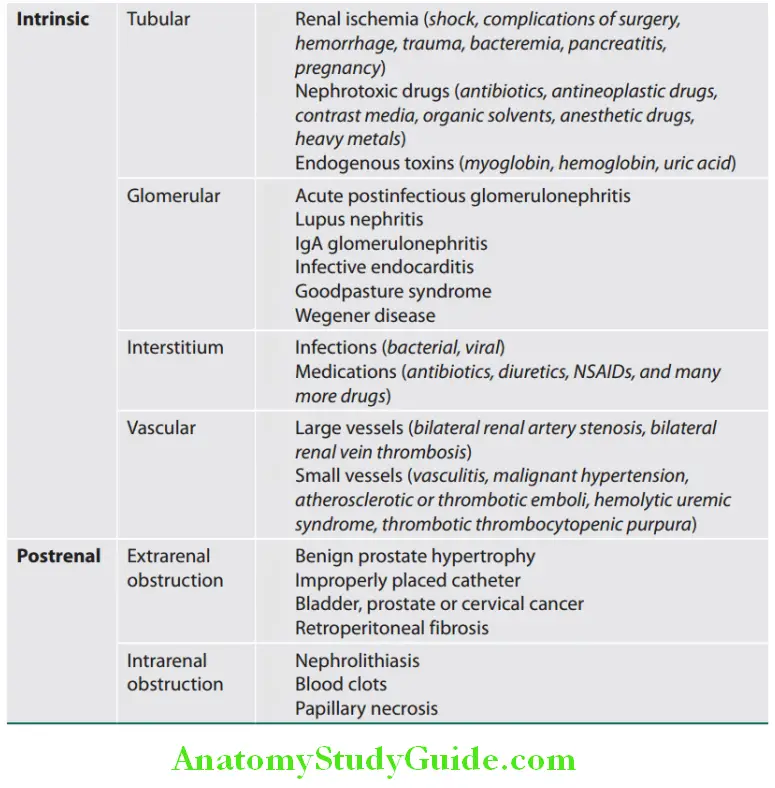
- Renal causes:
- Intrinsic diseases of the kidney causing
- AKI are classified according to the primary histologic site of injury: Tubules, Interstitium, Vasculature or Glomerulus.
- Renal tubular epithelial (RTE) cell injury, commonly known as acute tubular necrosis (ATN) occurs more commonly due to ischemia of any cause, but can also be damaged by specific renal toxins. ATN results in ischemia and necrosis of the tubular epithelial cells.
- However, when the causative factors are removed the tubular cells can regenerate. Postrenal causes: These include obstruction of the urinary tract at any point in its course from the tubule to the urethra.
Classification of causes of acute kidney injury:
Etiopathogenesis Clinical Features:
General Symptoms:
- Irrespective of the cause ARF present with symptoms related to uremia. These include anorexia, nausea, vomiting, intellectual clouding, drowsiness, fits, coma, pruritus, hemorrhagic episodes (e.g., epistaxis and gastrointestinal hemorrhage), and dyspnea due to fluid overload.
- Physical findings include asterixis, myoclonus, pericardial rub, and evidence of fluid overload in the form of edema, elevated jugular venous pressure (JVP), and crepitation.
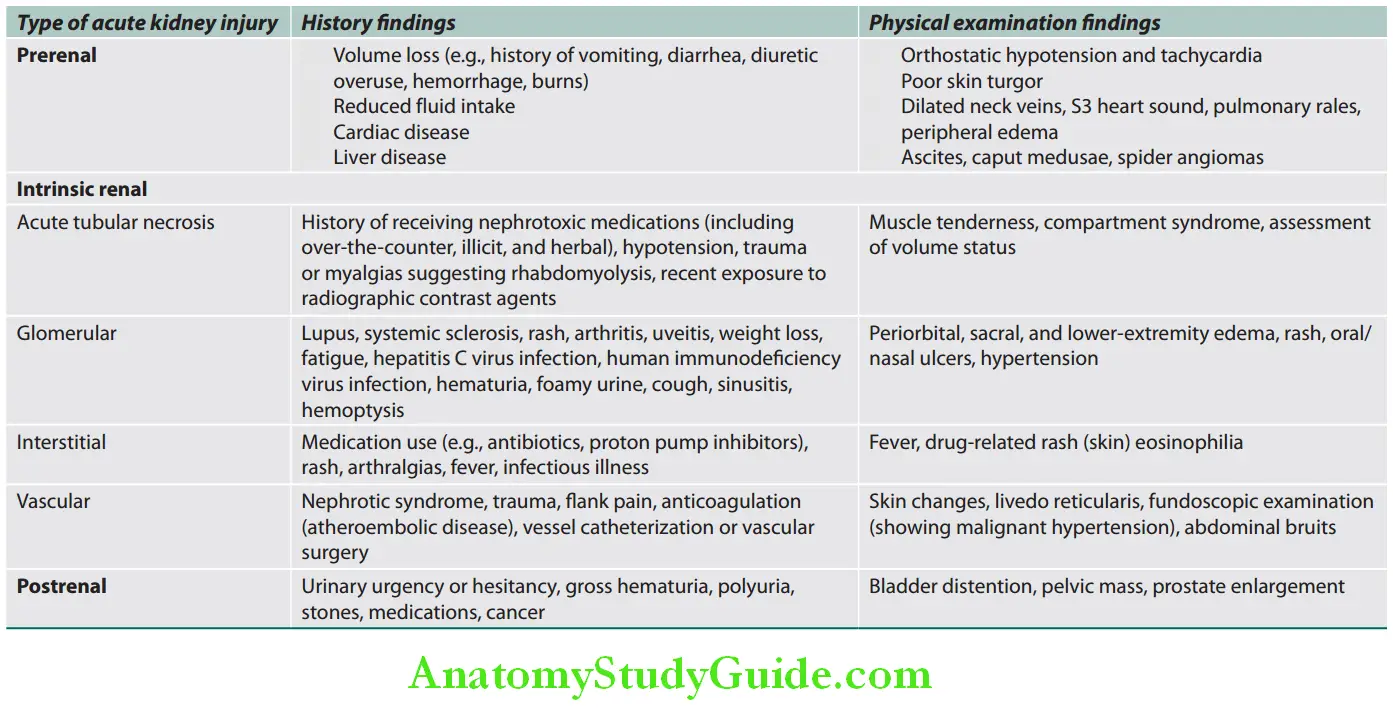
Question 11. How will you differentiate renal from prerenal failure?
(or)
Describe clinical and laboratory differences between prerenal and renal azotemia.
Answer:
AKI Due to a Prerenal Disorder:
- Most common form of AKI
- Rise in serum creatinine/BUN due to inadequate renal plasma flow and intraglomerular hydrostatic pressure to support normal glomerular filtration.
- Usually no or reversible parenchymal injury, but may be associated with ATN.
- Renin-angiotensin-aldosterone system responds to decreased effective circulating volume, but renal autoregulation fails once systolic blood pressure falls below 80 mm Hg.
- Nonsteroidal anti-inflammatory drugs—prevent renal afferent vasodilatation.
- Angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (ACEIs/ARBs)—prevent renal efferent vasoconstriction.
ARF Due to Renal Causes:
The most common causes—are sepsis, ischemia, and nephrotoxins. Prerenal AKI can lead to ATN in some cases. Acute tubular necrosis is the most important cause of ARF due to intrinsic renal diseases. The clinical course in ATN/ARF is divided into three phases:
- Oliguric (initiation) phase:
- Clinical features depend on the initiating event that caused the ischemic form of AKI. Patients develop symptoms due to fluid overload and azotemia. Fluid overload results in raised JVP, pedal edema, ascites, and pulmonary edema.
- Mild reduction of urine output and increase in BUN. Hyperkalemia occurs commonly during this phase. This phase usually lasts for about 10–14 days. About 40% of the patients may have normal urine output which is called nonoliguric renal failure. The electrolyte disturbances are less in these patients.
- Maintenance phase: During this phase, there is a sustained decrease in urine output in the range of 40–400 mL/day (oliguria), salt and water overload, rising BUN level, hyperkalemia, metabolic acidosis, and other features of uremia.
- This phase lasts for days to weeks.
- Diuretic (recovery) phase: During this phase, there is a steady increase in urine output, and in a few days the patient develops polyuria with a urine output that may reach up to 3 L/day. During this phase the tubular concentrating capacity is defective, and there is uncontrolled loss of large amounts of water, sodium, and potassium (leading to hypokalemia) in the urine. Once the renal tubular function returns to normal, BUN, creatinine levels, and urine volume also return to normal.
- ARF due to glomerulonephritis (GN) presents with hypertension, proteinuria, and hematuria.
- Drug-induced acute tubule-interstitial nephritis patients may present with fever, skin rash, and arthralgia.
Ischemia—associated AKI:
- Kidneys receive 20% of cardiac output, account for 10% of oxygen consumption although they constitute only 0.5% of human body mass.
- Renal outer medulla—one of the most hypoxic regions in the body – due to:
- Architecture of blood vessels that supply tubules
- Enhanced leukocyte-endothelial interactions in small vessels causing inflammation
- This causes:
- Reduced blood flow to metabolically active s3 segment of proximal tubule
- Mitochondrial dysfunction and release of reactive oxygen species (ROS) leading to tubular injury
Exogenous toxins—contrast agents:
- Iodinated contrast agents (CT):
- Risk of contrast nephropathy increased in presence of CKD, diabetic nephropathy, multiple myeloma, congestive cardiac failure (CCF).
- Diagnosis: Rise in serum creatinine within 24–48 hours following exposure, peak at 3–5 days, resolving within 1 week.
- Severe dialysis requiring AKI only occurs in the setting of other coexisting conditions mentioned above.
- Others: High-dose gadolinium (MRI), oral sodium phosphate solutions (bowel purgatives), drugs
Endogenous toxins:
- Rhabdomyolysis—traumatic crush injuries, muscle ischemia during vascular/orthopedic surgery, compression during come/immobilization, prolonged seizure activity, excessive exercise, heat stroke, malignant hyperthermia, infections, metabolic disorders such as hypophosphatemia and severe hypothyroidism, myopathies—release of myoglobin.
- Massive hemolysis—release of hemoglobin.
- Tumor lysis syndrome—after initiation of cytotoxic therapy (lymphoma, ALL, myeloma)
AKI Due to Postrenal Causes:
- Partial or total obstruction to unidirectional flow of urine—leading to increased hydrostatic pressure and interference with glomerular filtration
- Obstruction can be anywhere from renal pelvis to tip of urethra.
- B/L renal obstruction or U/L obstruction of a single functioning kidney, setting of CKD, reflex vasospasm of contralateral kidney.
- Causes:
- Bladder neck obstruction—prostate disease, neurogenic bladder, and therapy with anticholinergic drugs.
- Obstructed Foley’s catheter
- Ureteric obstruction—intraluminal obstruction—calculi, blood clots, sloughed renal papillae; infiltration of ureteric wall—neoplasia; external compression—retroperitoneal fibrosis, neoplasia, abscess, and surgical damage.
- If untreated, obstructive nephropathy leads to irreversible tubulointerstitial fibrosis (i.e., intrinsic disease).
Prerenal failure Investigations:
Serum Creatinine and Urea:
- The rate of rise in serum creatinine and urea is determined by the rate of protein catabolism (tissue breakdown).
- Raised serum creatinine and urea levels are the most consistent findings. In ARF due to prerenal causes, there is adisproportionate elevation of serum urea in relation to serum creatinine.
Question 12. List the causes of raised serum creatinine.
Answer:
Drawbacks:
Rise in creatinine is an unreliable indicator of early renal injury because:
- Normal serum creatinine level is an influenced by several nonrenal factors (age, gender, muscle mass, medications, hydration and nutrition status, and tubular secretion).
- More than 50% loss of renal function must be lost before serum creatinine rises.
- Serum creatinine does not reflect true GFR: This is because several hours to days must elapse before a new equilibrium between presumably steady state production and decreased excretion of creatinine is established.
Causes of raised serum creatinine:
- Azotemia: Prerenal, renal, and postrenal
- Large amount of consumption of meat
- Hypothyroidism, acromegaly, and gigantism
- Rhabdomyolysis
- Drugs: Statins, fibrates
Raised serum creatinine Other Investigations:
- Other biochemical findings: Include hyperkalemia, hypocalcemia, hyperphosphatemia, and hyperuricemia.
- Urine analysis:
- RBCs/RBC casts: Glomerulonephritis, vasculitis, malignant hypertension, and thrombotic microangiopathy
- WBCs/WBC casts: Interstitial nephritis, GN, pyelonephritis, allograft rejection, and malignant infiltration of kidney
- Renal tubular epithelial cells/RTE casts/pigmented casts: ATN, TIN, acute cellular allograft rejection, myoglobinuria, hemoglobinuria
- Granular casts: ATN, GN, TIN, and vasculitis
- Eosinophiluria: Allergic interstitial nephritis, atheroembolic disease, pyelonephritis, cystitis, GN
- Crystalluria: Acute uric acid nephropathy, calcium oxalate (ethylene glycol intoxication), drugs/toxins (acyclovir, indinavir, sulfadiazine, and amoxicillin)
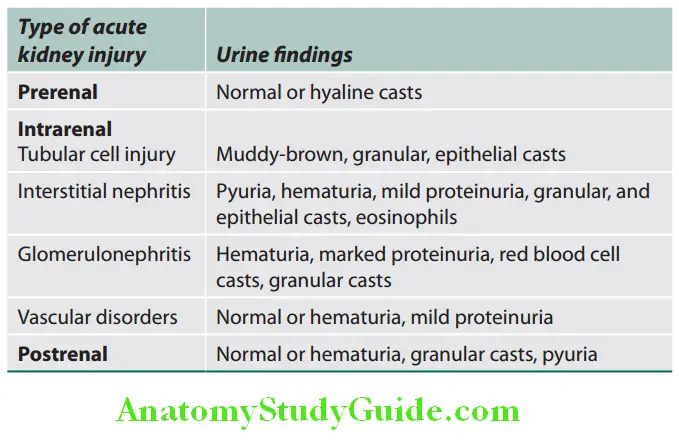
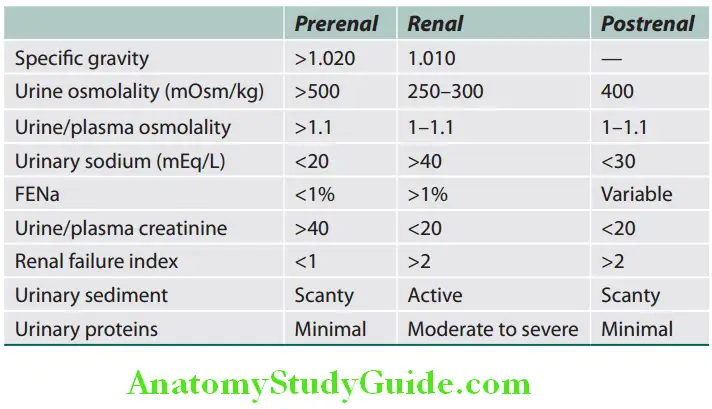
Proteinuria:
- Mild—<1 g/day—ischemia or nephrotoxin associated AKI
- Severe (nephrotic range) >3.5 g/day—GN, vasculitis, toxins that affect both glomerulus and tubulointerstitium such as NSAIDs, minimal change disease
- Electrocardiogram: May show features of hyperkalemia.
- Chest radiograph: May show pulmonary edema and pleural effusion.
- Others:
- In rapidly progressive glomerulonephritis (RPGN): Systemic causes (e.g., Wegener’s granulomatosis) must be excluded by appropriate investigations (e.g., cAN-CA). A kidney biopsy may be necessary.
- Kidney injury molecule 1 (KIM 1): Ischemia or nephrotoxin associated AKI
- Neutrophil gelatinase-associated lipocalin (NGAL): Cardiopulmonary bypass-associated AKI
- Insulin-like growth factor-binding protein 7 (IGFBP7): Marker of development of severe AKI
- Tissue inhibitor of metalloproteinase-2 (TIMP-2):
- Marker of development of severe AKI
- Fractional excretion of sodium: The ratio of sodium clearance to creatinine clearance, increases the reliability of this index. However, it may remain low in some renal diseases. Fraction of filtered sodium load that is reabsorbed by the tubules—measure of kidney’s ability to reabsorb sodium as well as endogenously and exogenously administered factors that affect tubular reabsorption. Helps in differentiating between prerenal azotemia and ATN, but has limited role.
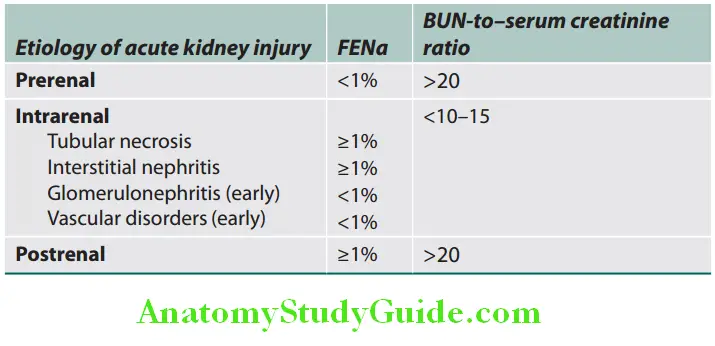
Complications of Acute Kidney Injury:
Question 13. Mention the complications of acute kidney injury (acute renal failure).
Answer:
- Metabolic: Hyperkalemia, hypocalcemia, hyperphosphatemia, hypermagnesemia, hyperuricemia, metabolic acidosis (increased anion gap)
- Cardiovascular: Cardiac arrhythmias, pulmonary edema, pericarditis/pericardial effusion
- Gastrointestinal: Gastrointestinal hemorrhage
- Neurologic: Encephalopathy, neuropathy, seizures
- Hematologic: Anemia, bleeding—platelet dysfunction
- Miscellaneous: Infections (pneumonia, urinary tract infection, septicemia)
Acute Kidney Injury Management:
General measures and management of complications:
- Fluid balance: Advisable to restrict fluid intake.
- Amount of fluid to be given depends upon the degree of edema, and fluid loss through urine, gastrointestinal tract and skin.
- Usually intake restricted to about 400 mL/day in addition to the abovementioned fluid losses.
- Sodium balance: Sodium is restricted to avoid volume expansion and overhydration
- Hyponatremia is common and is usually due to excessive fluid administration.
- Hypernatremia may occur occasionally, due to excessive administration of sodium bicarbonate for correction of acidosis.
- Potassium balance: Hyperkalemia is the leading cause of death in ARF.
- Acid-base balance: In most patients acidosis is of moderate degree and does not require treatment. However, in advanced cases intravenous sodium bicarbonate may be necessary. Acidosis if accompanied by severe hyperkalemia and fluid overload is best treated with dialysis.
- Calcium-phosphorus balance: Both hypocalcemia and hypercalcemia are observed in the maintenance phase of ARF and are not serious clinical problems. Phosphate retention occurs in patients can be controlled with aluminum hydroxide, lanthanum carbonate, sevelamer, calcium acetate or calcium carbonate [bind phosphate within the gastrointestinal tract (GIT) and eliminated in stool].
- Diet:
- Restrict dietary proteins to about 40 g/day. Suppress endogenous protein catabolism to a minimum level by giving as much energy as possible in the form of carbohydrates and fats. Patients treated by blood purification techniques are given 70 g or more protein/day.
- Hypercatabolic patients may need higher nitrogen intake to prevent negative nitrogen balance.
- Restrict the salt intake.
- Vitamin supplements are usually necessary.
- Systemic complications of ARF: Infections and gastrointestinal bleeding are two important complications.
- Infectious complications [very high (80%)] includes pulmonary, urinary, and wound infections (in post-traumatic and postoperative patients). Infections should be treated promptly by appropriate antibiotics.
- Gastrointestinal bleeding (in 40% of patients) may prove fatal and treated by proton pump inhibitors and gastroprotective agents. Qualitative platelet dysfunction, which results in a hemorrhagic diathesis.
- Use of drugs: Great care is necessary in the use of drugs and nephrotoxic drugs should be avoided.
Treatment of the underlying cause of the AKI:
- Identify the cause (by simple initial investigations such as ultrasound or may require additional investigations, including renal biopsy) and correct it, if possible.
Specific therapy:
- No specific treatment for ATN, other than restoring renal perfusion.
- Intrinsic kidney disease may require specific therapy (e.g., immunosuppressive drugs such as corticosteroids and cyclophosphamide in some causes of rapidly progressive glomerulonephritis).
- “Postrenal” obstruction requires urgent relief of obstruction. Once the blood chemistry returns to normal, the underlying cause should be treated whenever possible.
- Drug-induced acute tubule-interstitial nephritis usually recovers after stopping the offending drug, but sometimes, short course of steroids may be helpful.
- If conservative measures fail, dialysis and hemofiltration may be necessary. These techniques purify blood and/or remove excess fluid.
- Main indications of dialysis and hemofiltration in ARF are listed in lists the preventive strategies for conditions with high risk of acute kidney injury
Question 14. Write short note on indications for dialysis in acute renal failure.
Answer:



Cause of death: Most common causes of death in ARF (in the absence of dialysis) are hyperkalemia and pulmonary edema, followed by infection and uremia.
Glomerular Diseases:
The syndromes of glomerular disease are mentioned.
Syndromes of glomerular disease:
- Acute nephritic syndrome
- Rapidly progressive glomerulonephritis
- Nephrotic syndrome
- Chronic nephritis
- Asymptomatic urinary abnormalities (hematuria, proteinuria or both)
Glomerulonephritis:
- Glomerulonephritis: Inflammation of glomeruli and most are due to an immunologically mediated injury.
- Glomerulopathy: Glomerular diseases without apparent inflammation. There is an overlap between these terms.
Causes of Glomerulonephritis:
Pathogenesis:
Main mechanism is antibody-mediated glomerular injury.
1. Immune complex-mediated:
- Glomerular injury develops due to deposition of circulating antigen-antibody complexes (immune complexes) in the glomerulus. There is trapping of circulating antigen-antibody complexes within glomeruli which results in glomerular damage. The antibodies are not against any of glomerular constituents, and the immune complexes localize within the glomeruli.
- The antigen may be exogenous [e.g., bacteria as in poststreptococcal glomerulonephritis (PSGN) or endogenous (e.g., antibodies to host DNA in patients with systemic lupus erythematosus (SLE)].
2. Anti-GBM antibody-induced glomerulonephritis:
It develops due to injury by antibodies to the insoluble fixed (intrinsic) glomerular basement antigens. Anti-GBM antibody-induced GN is responsible for <5% of cases of GN. This type of injury is caused due to antibodies which are produced against intrinsic fixed antigens (that are normal components) of the GBM proper.
Causes of glomerular diseases:
Primary glomerulonephritis/glomerulopathy’s:
Acute proliferative glomerulonephritis: Postinfectious, others
- Rapidly progressive (crescentic) glomerulonephritis
- Minimal-change disease
- Membranous glomerulopathy
- Membranoproliferative glomerulonephritis
- Focal segmental glomerulosclerosis
- IgA nephropathy
- Chronic glomerulonephritis
Systemic diseases with glomerular involvement:
- Systemic immunological diseases: Systemic lupus erythematosus
- Metabolic diseases: Diabetes mellitus
- Vasculitis: Microscopic polyarteritis/polyangiitis, Wegener granulomatosis, Henoch–Schönlein purpura
- Amyloidosis
- Goodpasture syndrome
- Bacterial endocarditis
Hereditary disorders:
- Alport syndrome
- Thin basement membrane disease
- Fabry disease
Terms used in glomerular diseases:
Focal: Some glomeruli, but not all show the lesion.
- Diffuse (global): Most of the glomeruli (>75%) show the lesion.
- Segmental: Only a part of the glomerulus is affected (most focal lesions are also segmental, e.g., focal segmental glomerulosclerosis).
- Proliferative: Increase in cell numbers due to hyperplasia of one or more of the resident glomerular cells with or without inflammation.
- Membranous: Capillary wall thickening due to deposition of immune deposits or alterations in basement membrane.
- Crescent formation: Proliferation of parietal epithelial cell with mononuclear cell infiltration in Bowman’s space.
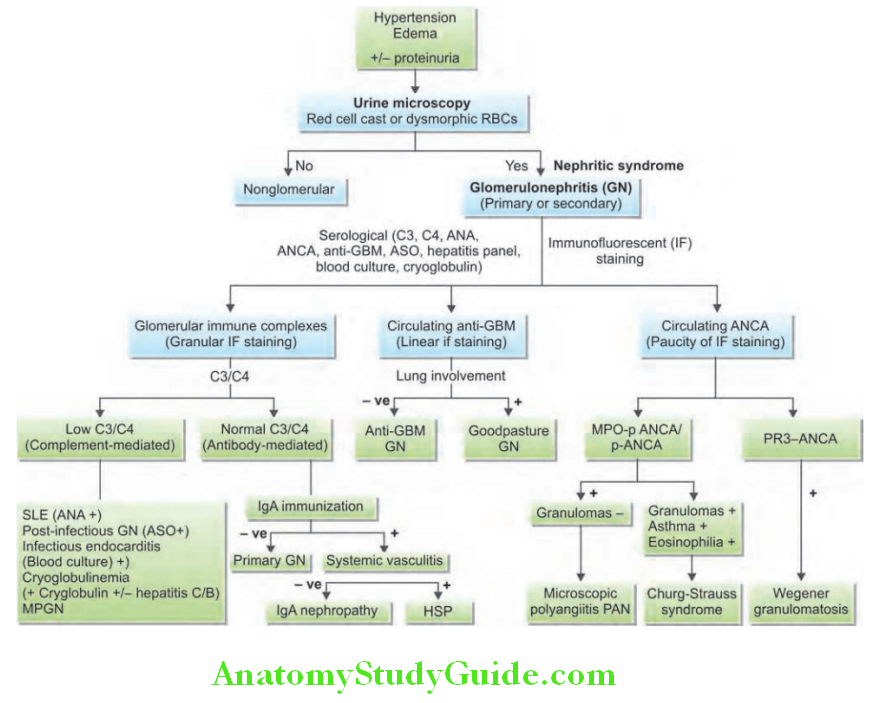
Characteristics of acute nephritic syndrome are presented.
Characteristics of acute nephritic syndrome:
- Hematuria (gross or microscopic)
- Red cell casts in the urine
- Azotemia (temporary)
- Temporary oliguria (due to decreased glomerular filtration rate)
- Hypertension
- Proteinuria*
- Edema* (periorbital, leg or sacral)
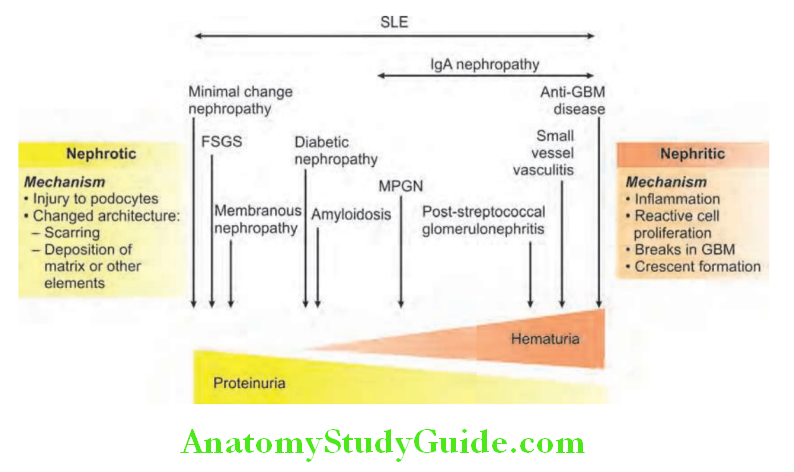
Spectrum of glomerular diseases are presented and various causes of proliferative and nonproliferative GN.
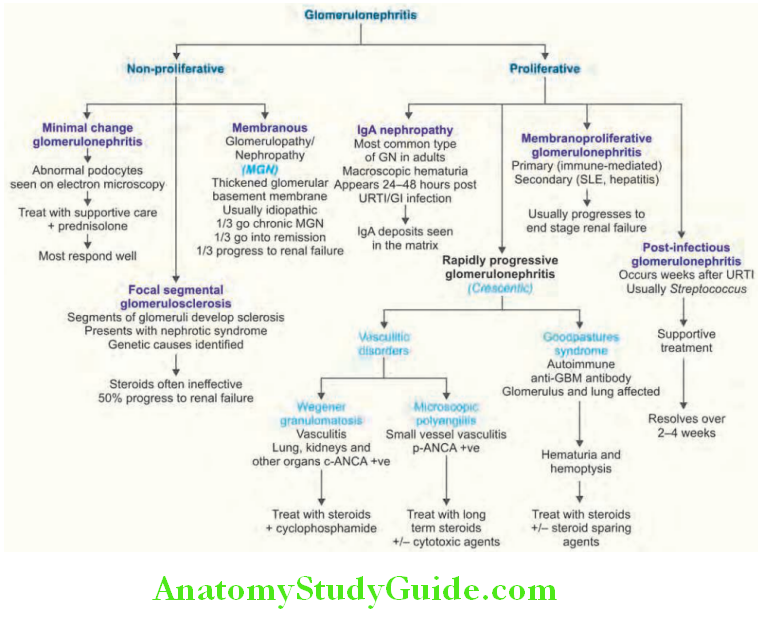
Acute Proliferative Glomerulonephritis:
These are immune complexes mediated diseases.
The inciting antigen may be:
- Exogenous, for example, Postinfectious GN which commonly follows streptococcal infection, but may also associated with other infections
- Endogenous,for example, Nephritis of SLE
- It is characterized histologically by cellular proliferation (mesangial and endothelial) associated with infiltration by leukocytes (neutrophils, macrophages).
Poststreptococcal (Postinfectious) Glomerulonephritis:
Question 15. Write short essay/note on acute glomerulonephritis or acute nephritic syndrome and its causes and clinical features.
(or)
Discuss the etiology, pathogenesis, clinical features, diagnosis, complications, and management of acute poststreptococcal glomerulonephritis [acute glomerulonephritis (AGN)].
Answer:
Poststreptococcal glomerulonephritis is specific subtype of postinfectious glomerulonephritis. It is common in developing countries and one of the common causes of acute nephritic syndrome.
Age group: Most frequently seen in children between 6 and 10 years of age, but may develop in adults.
Etiology and Pathogenesis:
- The primary streptococcal infection usually involves the pharynx (pharyngitis) or the skin (impetigo/pyoderma). Skin infections are usually associated with overcrowding and poor hygiene.
- Only certain strains of Group Ab-hemolytic streptococci are nephritogenic. More than 90% are due to types 12, 4, and 1.
- Commonly associated with poor personal hygiene, overcrowding and skin diseases such as scabies.
- It is an immunologically mediated disease and evidences to support this are:
- Latent period: It manifests usually after a latent period of 1–4 weeks following primary streptococcal infection.
- Cutaneous infections are associated with longer latent period. This latent period is compatible with the time required for the production of antibodies and the immune complex formation.
- Antibodies against streptococcal antigens: Majority of patients show increased titers of antibodies against one or more streptococcal antigens. These antibodies include: antistreptolysin O (ASO), antideoxyribonuclease B (antiDNase B), antistrepokinase, antihyaluronidase, and antinicotinyl adenine dinucleotidase.
- Hypocomplementemia: Immune complexes activate and utilize complement components and more than 90% of patients reveal decreased complement (C3 and C4) levels in the blood (hypocomplementemia).
- Immune complex deposits: Electron microscopy shows glomeruli with electron dense deposits of immune complexes. Immunofluorescence shows granular fluorescence to the immune deposits.
- Streptococcal antigens in the glomeruli: Many cationic antigens unique to nephritogenic strains of streptococci can be demonstrated in the glomeruli. Example, nephritis-associated streptococcal plasmin receptor (NAPlr), streptococcal pyrogenic exotoxin B (SpeB) and its zymogen precursor (zSpeB).
Poststreptococcal Clinical Features:
- Onset is often abrupt.
- Usually the affected child suddenly develops malaise, fever, nausea, oliguria, and hematuria (characteristically, urine appears smoky or red or cola-colored urine) 1–4 weeks after recovery from a sore throat.
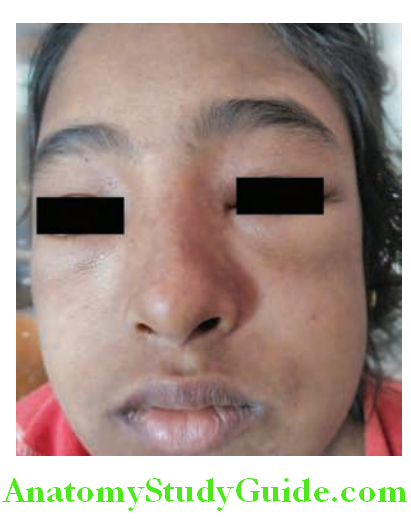
- Periorbital edema (causes puffiness of face), and mild to moderate hypertension is usually observed. Edema initially appears in areas of low tissue pressure (periorbital areas), followed by involvement of dependent portions of the body, and may be associated with ascites and/or pleural effusion.
- In adults clinical features are atypical. They may present with the sudden appearance of hypertension or edema, and elevation of BUN. poststreptococcal glomerulonephritis)
Poststreptococcal Investigations:
Various laboratory fidings in acute poststreptococcal glomerulonephritis (PSGN):

Question 16. Write short essay/note on diagnostic criteria and management of nephritic syndrome.
(or)
Write short essay/note on urinary findings in acute glomerulonephritis.
Answer:
Nephritic syndrome Management/Treatment:
- Supportive treatment during acute PSGN: These include rest, salt restriction, diuretics and antihypertensives.
- Dialysis is necessary when there is severe oliguria, fluid overload, and hyperkalemia.
- Steroids and cytotoxic drugs are of no value. However, if recovery is slow or if RPGN develops, corticosteroids (methylprednisolone) may be of some help.
Complications:
Question 17. Write short note on the complications of acute poststreptococcal glomerulonephritis/nephritic syndrome.
Answer:
- Rapidly progressive glomerulonephritis
- Pulmonary edema
- Hypertensive encephalopathy
- Renal failure
Nephritic syndrome Prognosis:
Majority of patients with the epidemic form of PSGN have an excellent prognosis.
- Children: Prognosis is good and more than 95% totally recover. Minority may develop a RPGN.
- Adults: Less benign. They may recover promptly or develop RPGN or progress to chronic glomerulonephritis (hypertension and/or renal impairment).
Prevention: Pharyngitis caused by streptococci should be treated promptly by antibiotics which protects against development of GN.
Rapidly Progressive Glomerulonephritis/Crescentic Glomerulonephritis:
Question 18. Write short essay/note on the causes, clinical features, investigations, and treatment of rapidly progressive glomerulonephritis.
(or)
Write short essay/note on crescentic glomerulonephritis.
Answer:
Crescentic Glomerulonephritis Definition: Rapidly progressive glomerulonephritis is a syndrome, characterized by rapid and progressive loss of renal function (usually a 50% reduction in the GFR within 3 months) associated with severe oliguria and signs of nephritic syndrome. If not treated death occurs due to renal failure within weeks to months. Histologically, it is characterized by extensive crescents (usually >50%).
Crescent:
Crescents are formed by the proliferation of the parietal epithelial cells lining Bowman capsule along with the infiltration of monocytes and macrophages.
Crescentic Glomerulonephritis Classification:
Rapidly progressive glomerulonephritis classified into three types based on immunological findings. Each type may be idiopathic or associated with a known disorder.
Crescentic Glomerulonephritis Clinical Features:
Crescentic Glomerulonephritis Investigations:
- Blood:
- Leukocytosis and anemia
- Blood urea and serum creatinine levels: Usually raised.


- Urinalysis:
- Moderate proteinuria (1–4 g/day)
- Microscopic hematuria
- RBC and WBC casts.
- Others:
- Complement levels (C3 and C4): May be decreased in immune-complex mediated RPGN.
- Circulating anti-GBM antibodies: In Goodpasture syndrome.
- Antineutrophil cytoplasmic antibody: In pauci-immune RPGN.
- Serum cryoglobulin levels: May be raised in cryoglobulinemia.
- Abdominal ultrasound: Normal sized kidneys.
- Chest X-ray: Patients with Goodpasture syndrome and vasculitides may show diffuse opacities if associated with pulmonary hemorrhage.
- Renal biopsy: Shows crescents.
Complement Levels in Nephritic Syndrome:

Algorithm of approach to the patient presenting with acute glomerulonephritis/nephritic syndrome is presented.
Nephrotic Syndrome:
Question 19. Discuss the etiology, pathogenesis, clinical features, diagnosis, and management of nephrotic syndrome.
(or)
Define nephrotic syndrome. Discuss the differential diagnosis in a 30-year-old male presenting with anasarca.
Answer:
Features of nephrotic syndrome are mentioned.
Characteristics of nephrotic syndrome:
- Massive/heavy proteinuria (>3.5 g of protein/24 hours)
- Hypoalbuminemia
- Generalized edema
- Hyperlipidemia and lipiduria
Nephrotic Syndrome Pathophysiology:
- Massive proteinuria is characterized by daily loss of 3.5 g or more of protein (less in children) in the urine.
- Normally, the glomerular capillary wall acts as a size and charge dependent barrier for the plasma filtrate.
- Proteinuria in nephrotic syndrome is due to increased permeability of glomerular capillary wall to plasma proteins. This increased permeability is due to glomerular inflammation, change in the surface electrical charge, and an alteration in the pore size.
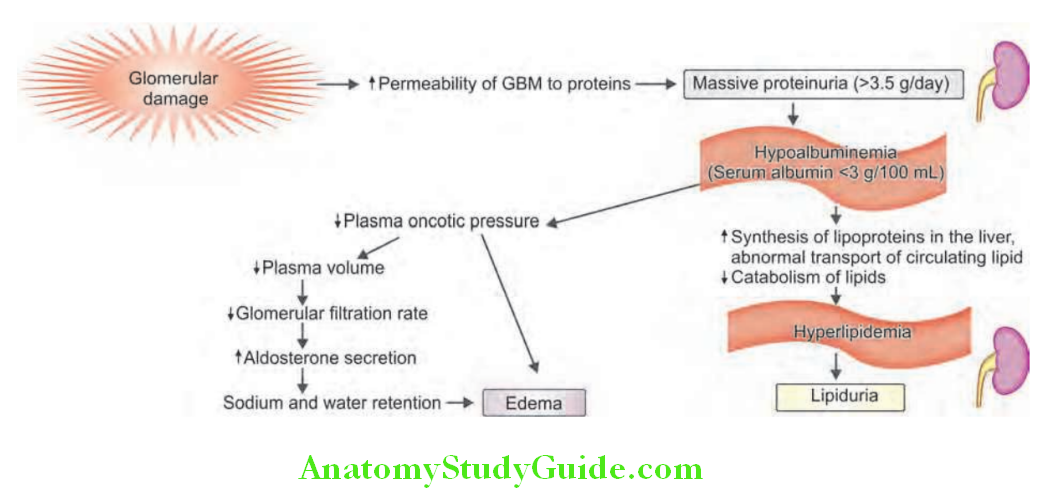
-
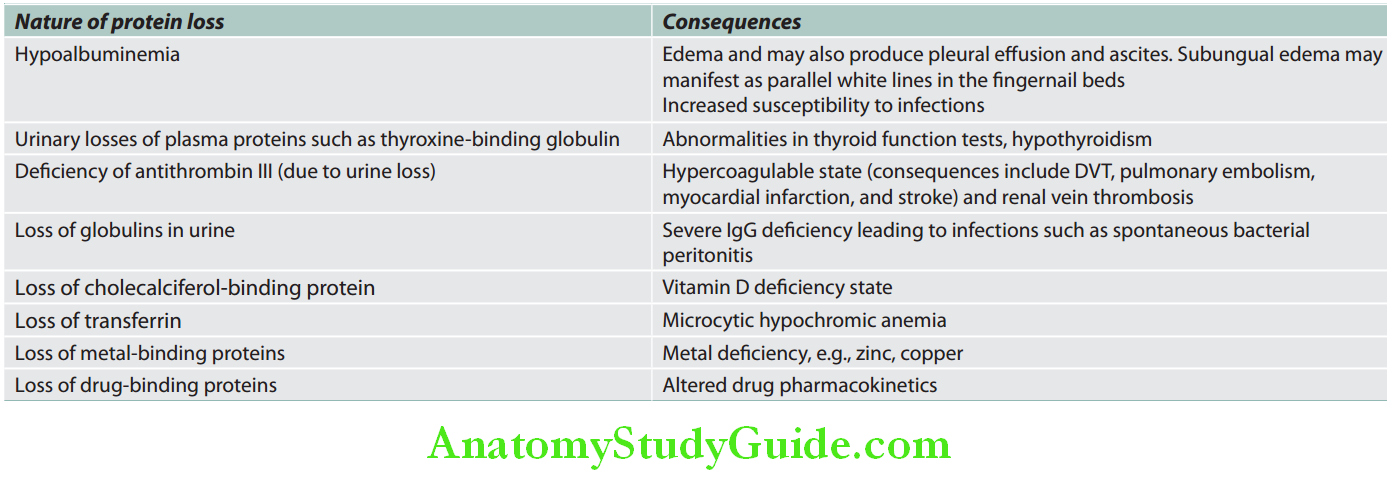
- The major proportion of protein lost in the urine is albumin, and rarely globulins. Consequences of protein loss.
- Hypoalbuminemia
- Massive proteinuria decreases the serum albumin levels (hypoalbuminemia).
- Hypoalbuminemia decreases the colloid osmotic pressure of the blood resulting in a disturbance in the Starling forces acting across peripheral capillaries.
- Massive proteinuria decreases the serum albumin levels (hypoalbuminemia).
Nephrotic Syndrome Treatment:
It depends on the factors involved in the pathogenesis.
- Supportive therapy:
- Control of infection
- Control of volume status (dialysis may be needed)
- Specific therapy:
- Plasma exchange to remove circulating antibodies and in patients presenting with life-threatening pulmonary hemorrhage.
- Steroids methylprednisolone—500–1,000 mg/day for 3 days to suppress inflammation from antibody already deposited in the tissue.
- Immunosuppressive therapy (e.g., cyclophosphamide, azathioprine, mycophenolate) such as cyclophosphamide to suppress further antibody synthesis.
- Infliximab and rituximab.
- The hypovolemia also triggers the RAA system. This causes increased reabsorption of sodium and water by the kidney, resulting in edema.
- Generalized edema
- Soft and pitting
- Most marked in the periorbital regions and dependent portions of the body.
- Associated with pleural effusions and ascites.
- Hyperlipidemia and lipiduria
- Hyperlipidemia: Most patients with nephrotic syndrome have raised blood levels of cholesterol, triglyceride, very-low-density lipoprotein, low-density lipoprotein, Lplipoprotein, and apoprotein. It increases risk of atherosclerosis and cardiovascular disease.
- Causes of hyperlipidemia: Increased synthesis of lipoproteins in the liver due to low plasma colloid oncotic pressure. Abnormal transport of circulating lipid. Decreased catabolism of lipids
- Lipiduria: Hyperlipidemia is followed by leakage of lipoproteins across the glomerular capillary wall → leaked lipoprotein is reabsorbed by tubular epithelial cells → then shed along with the degenerated cells → appears in urine either as free fat or as oval fat bodies.
Causes of Nephrotic Syndrome:
Question 20. Write short essay on causes/etiology, pathogenesis, clinical features, investigations, and treatment of minimal change disease.
Answer:
Causes of nephrotic syndrome:
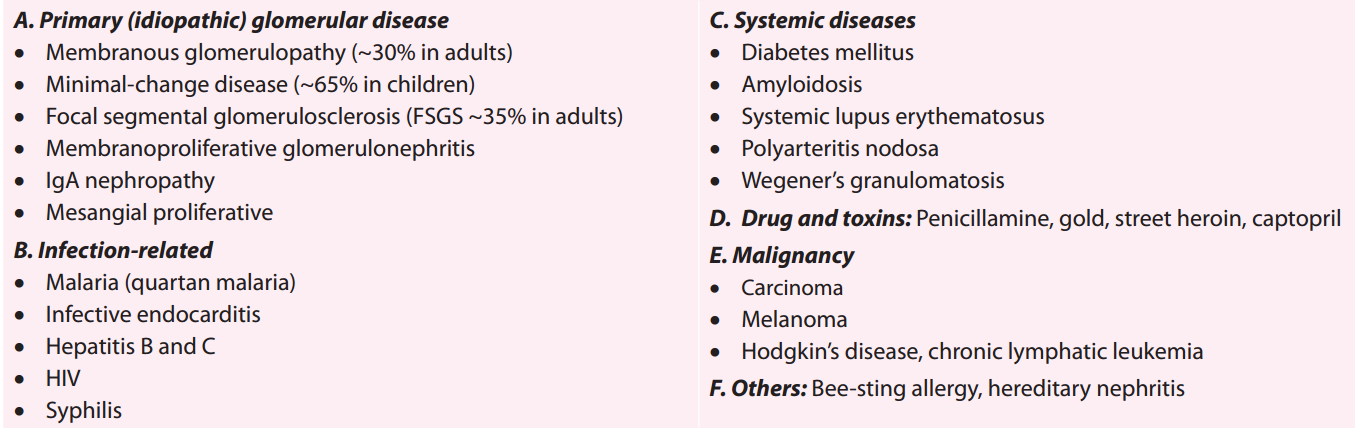

- Minimal change disease (MCD):
- Minimal change disease is named so because the glomerular changes are absent or minimal and glomeruli appear normal under light microscopy. But under electron microscopy, it shows diffuse effacement (loss) of foot processes of visceral epithelial cells (podocytes).
- Minimal change disease is the major cause of nephrotic syndrome in children (80%), but it is less common in adults (20%).
- Age: Peak incidence between 2 and 6 years of age.
- Focal and segmental glomerulosclerosis (FSGS)
- FSGS is characterized by the sclerosis that involves only part of the capillary tuft (i.e., segmental) of some glomeruli (i.e., focal).
- Accounts for about one-third of cases of nephrotic syndrome in adults.
- Usually manifest as nephrotic syndrome or heavy proteinuria, hypertension, renal insufficiency, and occasionally hematuria.
- Causes
- Membranous nephropathy:
- Characterized by uniform diffuse thickening of the glomerular capillary wall. This is due to the accumulation of electron-dense deposits along the subepithelial side of the glomerular basement membrane.
- Common cause (~30%) of the nephrotic syndrome in adults.
- Gender and age: Male predominance and high incidence between 30 to 50 years of age.
- Causes
- About 75% of patients present with nephrotic-range proteinuria and 50% present with microscopic hematuria.
Causes of focal and segmental glomerulosclerosis:
Primary (idiopathic):
Secondary:
- Viruses: HIV infection
- Drugs: Heroin addiction
- Sickle-cell disease
- Massive obesity
- Congenital (e.g., unilateral agenesis) and acquired (e.g., reflux nephropathy) reductions in renal mass
Hereditary forms: Inherited mutations in genes that encode podocyte proteins, e.g., podocin, α-actinin 4
Causes of membranous nephropathy:
Primary/idiopathic: No identifiable cause in about 85% of patients
Secondary (20–30% cases): In association with:
- Therapeutic drugs: Penicillamine, captopril, nonsteroidal anti-inflammatory drugs (NSAIDs), gold
- Malignant neoplasms, e.g., carcinomas of the lung and colon, lymphomas and melanoma
- Autoimmune disease, e.g., systemic lupus erythematosus (SLE), thyroiditis
- Infections: Chronic hepatitis B, hepatitis C, quartan malaria, syphilis, schistosomiasis
Investigations in Nephrotic Syndrome:
Question 21. Write a short note on urinary findings in nephrotic syndrome.
Answer:
- Urine examination
- Proteinuria: Twenty-four hour urinary protein estimation.
- Microscopy: Red cells and red cell casts and waxy casts may be present. However, in minimal change disease, RBCs and red cell casts are not seen. Shows lipiduria
- Serum albumin: Reduced
- Serum cholesterol: Raised
- Renal biopsy: May be necessary for histological diagnosis.
- Other investigations: Depending on the suspected secondary causes appropriate investigations are to be performed.
Nephrotic Syndrome Management:
1. General measures:
Measures to reduce proteinuria: These measures are necessary if immunosuppressive drugs and other specific measures against the underlying cause do not benefit.
- Angiotensin-converting enzyme inhibitors and/or angiotensin II receptor antagonists: They reduce proteinuria in all types of GN and also slow the rate of progression of renal failure by lowering glomerular capillary filtration pressure. Blood pressure and renal function should be monitored regularly during their administration.
Measures to control complications:
- Treatment of edema:
- Initially, it is treated by dietary salt (sodium) restriction, rest and a thiazide diuretic (e.g., chlorthalidone, bendroflumethiazide). The weight loss should not be more than 1 kg/day. Aggressive diuretic therapy may precipitate ARF due to reduction in intravascular volume.
- If not responsive, furosemide 40–120 mg daily with the addition of amiloride (5 mg daily), and serum potassium concentration should be monitored.
- Gut mucosal edema in nephrotic syndrome may cause malabsorption of diuretics (as well as other drugs). Thus, if there is resistance to oral diuretic treatment, parenteral administration is required.
- In diuretic-resistant patients and those with oliguria and uremia in the absence of severe glomerular damage (e.g., in minimal-change nephropathy) edema may be treated by infusion of salt-poor albumin as a temporary measure combined with diuretic therapy. However, most of infused albumin will be excreted by the kidneys within 1–2 days.
- Dietary proteins: It is advisable to take normal protein and should be about 0.8–1.0 g/kg. A high-protein diet (approximately 80–90 g protein daily) increases proteinuria and may be harmful in the long-term. However, malnutrition should be prevented.
- Hypercoagulable state: Develops due to loss of coagulation factors (e.g., antithrombin) in the urine and an increase in production of fibrinogen by liver. It predisposes to venous thrombosis and thromboembolism. Therefore, avoid prolonged bed rest. Long-term prophylactic anticoagulant therapy is desirable and it is indicated in patients who have already developed deep venous thrombosis or arterial thrombosis.
- Lipid abnormalities: They increase in the risk of myocardial infarction or peripheral vascular disease. Hypercholesterolemia is treated with an HMG-CoA reductase inhibitor and dietary restrictions of lipids.
- Vitamin D supplementation: To be given if there is biochemical evidence of vitamin D deficiency.
- Sepsis: It is a major cause of death in nephrotic syndrome. The increased susceptibility to infection is partly due to loss of immunoglobulin in the urine. They are particularly susceptible to pneumococcal infections and pneumococcal vaccine should be given to these patients. Early detection and aggressive treatment of infections should be done. Vaccinations prophylactically is advisable.
2. Treatment of underlying cause:
Minimal change disease:
- In children:
- Initial treatment by high-dose corticosteroid therapy with prednisolone 60 mg/m 2 daily (up to a maximum of 80 mg/day) for a maximum of 4–6 weeks.
- Followed by alternate day prednisolone at a dose of 40 mg/m 2 (1 mg/kg in adults) for further 4–6 weeks.
- More than 95% of children respond to the above therapy.
- Children who respond within the first 4 weeks of corticosteroid therapy are termed “steroid responsive.” Those who relapse on withdrawal of corticosteroid therapy are termed “steroid dependent.”
- Relapse:
- One-third of patients relapse on steroid withdrawal, and remission is once more induced with steroid therapy.
- In patients who have frequent relapses or develop unacceptable corticosteroid side effects, long-term remission can be achieved by a course of cyclophosphamide 1.5–2.0 mg/kg daily is given for 8–12 weeks with concomitant prednisolone 7.5–15 mg/day.
- Steroid unresponsive patients may also benefit by cyclophosphamide. Not more than two courses of cyclophosphamide should be given because of the risk of side-effects (e.g., azoospermia).
- An alternative to cyclophosphamide is ciclosporin 3–5 mg/kg/day (ciclosporin is potentially nephrotoxic).
- Adults: Response rates are significantly lower and response may occur late (12 weeks with daily steroid therapy and 12 weeks of maintenance with alternate-day therapy).
- Prognosis: Excellent, although it may show remission and relapses.
Focal and segmental glomerulosclerosis:
- Steroids: It is beneficial in only 20–30% patients and usually prednisolone is given in the dose of 0.5–2 mg/kg/day.
- Cyclosporine may be effective in reducing or stopping urinary protein excretion.
- Cyclophosphamide, chlorambucil or azathioprine may be used as second-line therapy in adults.
- About 50% progress to end-stage renal failure.
Membranous glomerulonephritis:
- Oral high-dose corticosteroids are not useful for producing either a sustained remission of nephrotic syndrome or preserving renal function.
- Alkylating agents: Cyclophosphamide and chlorambucil are effective. However, because of long-term toxicity, these drugs should be reserved for patients who have severe or prolonged nephrosis (i.e., proteinuria >6 g/day for >6 months), renal insufficiency, and hypertension. Cyclophosphamide, cyclosporine and chlorambucil in combination with steroids may be helpful.
- Anti-B lymphocyte therapy is more effective against T lymphocytes than broad-spectrum immunosuppressive agents. Anti-CD20 antibodies (rituximab, which ablates B lymphocytes) improve renal function, reduce proteinuria and increase the serum albumin.
- Prognosis: Spontaneous remission may occur in 40%, 3–40% may develop repeated remissions and relapses and 10–20% patients may develop progressive renal failure.
Immunoglobulin A Nephropathy (Berger’s Disease):
Question 22. Write short essay/note on IgA nephropathy.
Answer:
- Characterized by focal and segmental proliferative GN with predominant IgA deposition in the glomerular mesangium.
- Clinical features:
- Occurs in children and young males.
- Presents with asymptomatic/painless microscopic hematuria or recurrent macroscopic hematuria generally within 1–2 days after an upper respiratory or gastrointestinal viral infection.
- Proteinuria occurs and in 5% it can be in the nephrotic range.
- Occasionally, it may present as ARF or nephritic syndrome.
- Diagnosis by renal biopsy: Immunofluorescence microscopy shows prominent IgA deposits in the mesangial regions.
- Prognosis:
- Usually good, especially in patients with normal blood pressure, normal renal function, and absence of proteinuria at presentation.
- Complete remission uncommon.
- Risk of development of end-stage renal failure in about 25% of patients with proteinuria of more than 1 g/day, elevated serum creatinine, hypertension, ACE gene polymorphism and presence of tubulointerstitial fibrosis on renal biopsy.
Immunoglobulin A Nephropathy Management:
- Steroids: Used for patients with proteinuria of 1–3 g/day, mild glomerular changes only, and preserved renal function.
- They reduce proteinuria and stabilize renal function.
- Use of immunosuppressive therapy is controversial.
- Combination therapy: In patients with progressive disease (creatinine clearance <70 mL/min), prednisolone with cyclophosphamide can be used for 3 months followed by maintenance with prednisolone and azathioprine.
- Tonsillectomy: May reduce proteinuria and hematuria in patients with recurrent tonsillitis.
- Combination of ACE inhibitor and angiotensin II receptor antagonist: Can be given to all patients, with or without hypertension and proteinuria. This combination therapy reduces proteinuria and preserves renal function.
Hereditary Nephritis or Alport’s Syndrome:
- Most common familial nephropathies, characterized by familial occurrence of progressive hematuria, nephritis, and sensorineural loss of hearing.
- Common in females. Male patients develop severe renal disease with progressive renal failure occurring before the fourth decade. Most females have a normal life-span.
- Pathology: Electron microscopy—shows irregular thickening of GBM, splitting and splintering of the lamina densa and small, round, electron-dense granulations are present within the lucent zones. GBM lesions are the hallmark but not specific of Alport’s syndrome.
- Clinical presentation:
- Major presentation: Macroscopic or microscopic hematuria and may be observed at birth.
- Other presenting features: Proteinuria, edema, hypertension, renal failure, and deafness.
- Microscopic or recurrent episodes of macroscopic hematuria following upper respiratory infection (or physical exertion) may resemble postinfective glomerulonephritis.
- Incidental leukocyturia and pyuria may lead to erroneous diagnosis of UTI.
- Nephrotic syndrome can occur with increasing proteinuria. Hypertension develops with progressive renal insufficiency.
- Deafness is more frequent in males and progression of hearing loss usually indicates poor prognosis.
- Ocular changes include anterior lenticonus (most common) and associated posterior or anterior cataract.
Immunoglobulin A Nephropathy Treatment: No specific measure to prevent progression of renal disease. Treatment includes renal replacement therapy, by longterm hemodialysis or renal transplant. Hearing defect can be temporarily compensated by the use of hearing aid
Tubulointerstitial Diseases:
- Tubulointerstitial nephropathy is an inflammatory condition affecting primarily the renal tubules and interstitium.
- Structural changes in the glomeruli develop later and results in progressive decline in GFR, glomerular proteinuria and volume-dependent hypertension.
- It accounts for 20–40% of cases of CRF and 10–25% of cases of ARF.
Acute Tubulointerstitial Nephropathy:
- Tubulointerstitial nephritis is a frequent cause of AKI that can lead to CKD.
- Affects renal tubules and interstitial components of the renal parenchyma
- Characterized: Tubular dysfunction with electrolytes abnormalities (moderate proteinuria, varying degrees of renal impairment)
Two most common causes of acute TIN are drugs or toxins and infections.
Immunoglobulin A Nephropathy Etiology:
Drug-induced acute TIN:
- After exposure to a causative drug, renal dysfunction may occur within a few hours but can occur after weeks or months.
- Preceded or accompanied by the triad of fever (70–100%), skin rash (30–50%) and eosinophilia (transient). Skin rash and eosinophilia are not found in TIN caused by NSAIDs.
- Microscopic hematuria, pyuria, and proteinuria are present in almost all cases. Eosinophiluria (>50% of WBC) is a sensitive marker of drug-induced TIN.
- Patients may need steroids (methylprednisolone 500–1000 mg/day for 3 days).
Etiology of acute tubulointerstitial nephropathy:

Analgesic Nephropathy:
- Prolonged analgesic abuse leads to a nephropathic process characterized bycapillary sclerosis, chronic tubulointerstitial diseases and papillary necrosis.
- Imaging reveals shrunken kidneys with calcification of renal papillae.
- Risk of developing renal disease is dependent upon the frequency and duration of analgesic consumption, the cumulative amount of individual analgesic exceeding 3 kg (phenacetin, acetaminophen, or aspirin).
Tubulointerstitial Nephritis and Uveitis Syndrome:
- Combination of TIN and uveitis
- Abnormal renal function, abnormal urinalysis
- Anterior uveitis—photophobia, eye pain and redness, eyelid edema, rapidly progressive loss of vision, as well as symptoms of systemic illness, including weight loss, fever, and fatigue
- Seen in adolescent girls
- Has good prognosis
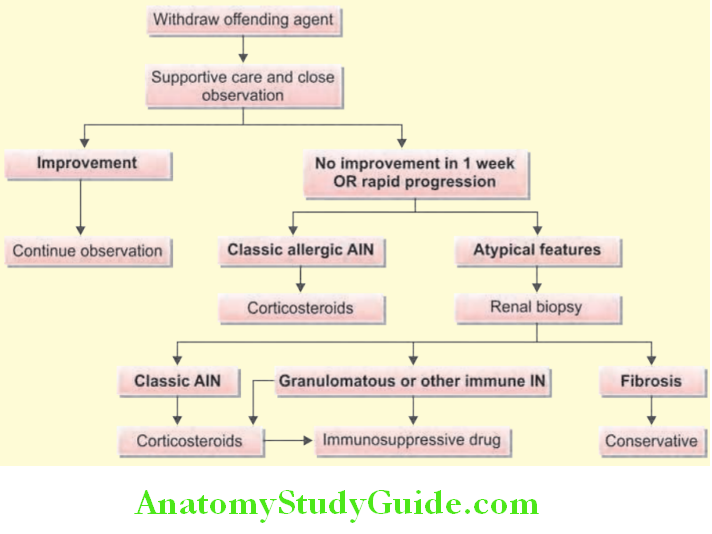
Treatment of Interstitial Nephritis:
- Stop the offending agent
- Prednisone at a dose of 1 mg/kg/day (to a maximum of 40–60 mg) for a minimum of 1–2 weeks, beginning a gradual taper after the serum creatinine has returned to or near baseline.
- Cyclophosphamide (2 mg/kg/d)
- Mycophenolate
- Plasmapheresis.
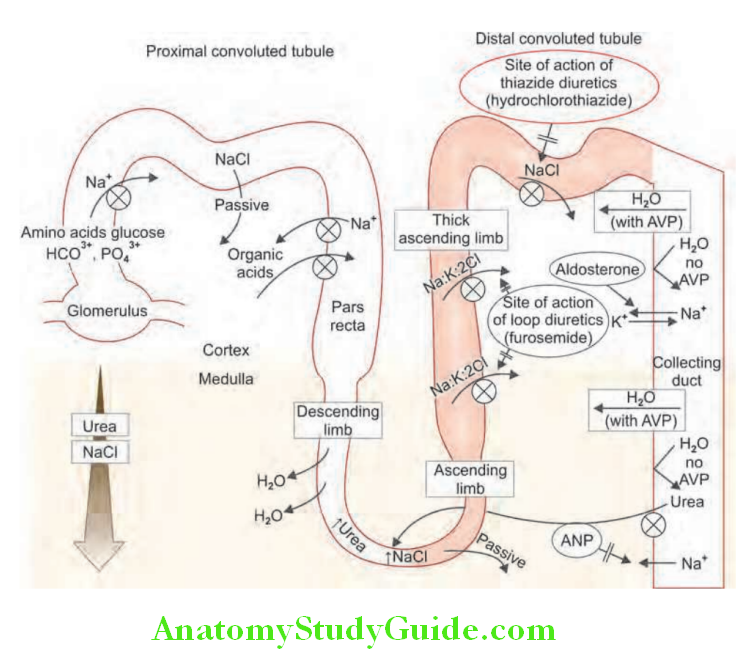
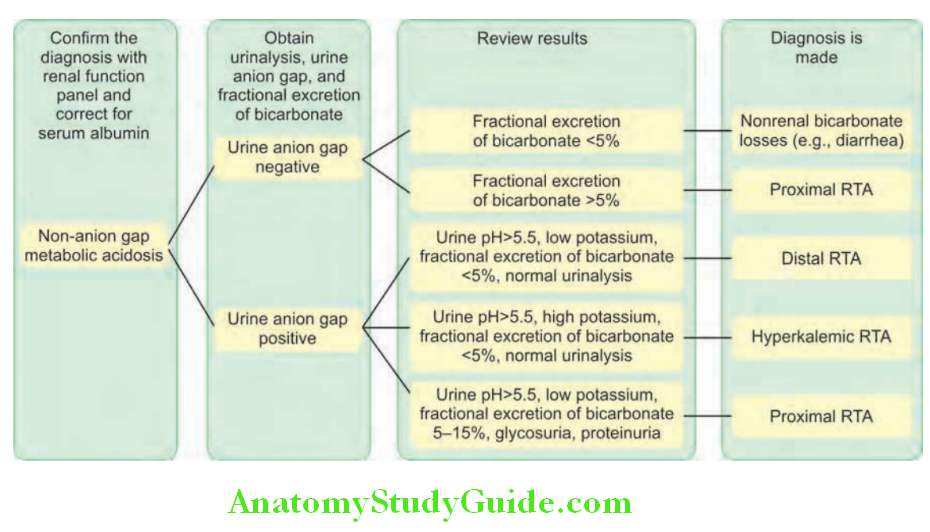
Renal Tubular Acidosis:
Question 23. Write short essay/note on renal tubular acidosis.
Answer:
- Renal tubular acidosis refers to hyperchloremic (normal anion gap) metabolic acidosis in the presence of normal or almost normal renal function.
- Renal tubular acidosis arises as a result of defects in the tubular transport of HCO3- and/or H+.
- Most forms of RTA are usually asymptomatic; rarely, life-threatening electrolyte imbalances may occur.
Mechanism:
Renal tubular acidosis can be due to a defect in one of three processes:
- Impaired acid secretion in the late distal tubule or cortical collecting duct intercalated cells (classical distal RTA).
- Impaired bicarbonate reabsorption in the proximal tubule (proximal RTA).
- Impaired sodium reabsorption in the late distal tubule or cortical collecting duct. It is associated with reduced secretion of both potassium and H+ ions (hyperkalemia distal RTA).
Lists types of renal tubular acidosis (RTA):
Types of renal tubular acidosis (RTA):
- Type 1 RTA or distal tubular RTA
- Type 2 RTA or proximal RTA
- Type 3 RTA or mixed RTA
- Type 4 RTA or hypoaldosteronism hyperkalemia RTA
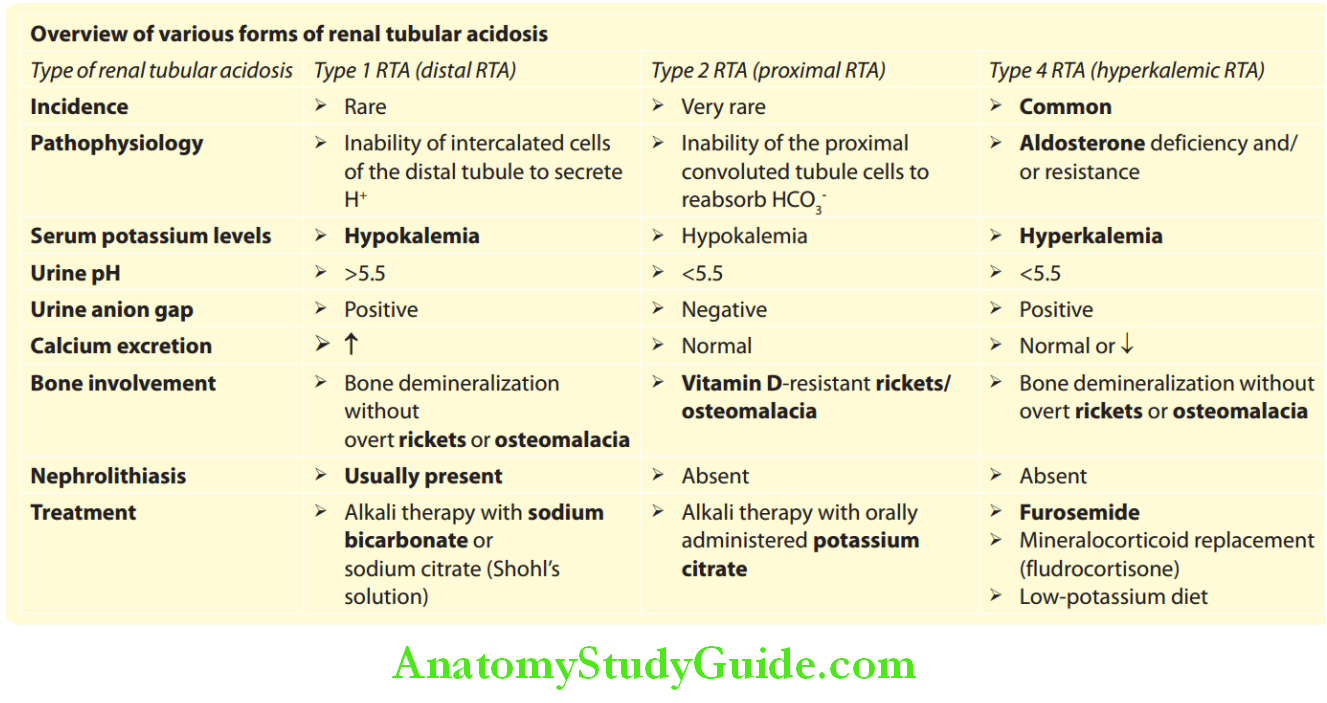
Renal Tubular Acidosis Types:
Type 1 (“Distal” Renal Tubular Acidosis):
- Most common type of RTA.
- Causes: It occurs due to a failure of H+ (hydrogen ion) excretion in the distal tubule.
Causes of type 1 renal tubular acidosis (RTA):
- Primary/hereditary
- Nephrocalcinosis (producing damage of cortical collecting duct)
- Chronic urinary tract obstruction
- Hypergammaglobulinemic states
- Drugs and toxins, e.g., ifosfamide, amphotericin B, lithium, and toluene
- Renal transplant rejection
- Autoimmune disease, e.g., Sjögren’s syndrome
- Cirrhosis of liver
- Sickle cell anemia
Consequences:
- Acidosis
- Hypokalemia (few exceptions)
- Failure to lower the urine pH below 5.3 despite systemic acidosis
- Low urinary ammonium production
- Renal calculus formation due to hypercalciuria, hypocitraturia (citrate inhibits calcium phosphate precipitation), and alkaline urine (which favors precipitation of calcium phosphate). Calculus produces hematuria, pain, and recurrent urinary infections.
- Nephrocalcinosis: Deposition of calcium in the kidney parenchyma.
- Depletion/mobilization of calcium (demineralization) from bones causing rickets in children and osteomalacia in adults.
Renal Tubular Acidosis Diagnosis:
- Urinary pH > 5.3 in presence of systemic acidosis
- Plasma bicarbonate (HCO–) <20 mEq/L
- Acid load test:
- Give ammonium chloride by mouth (100 mg/kg) and check pH of urine hourly and plasma HCO3at 3 hours.
- Plasma HCO3
- should drop below 21 mmol/L.
- Diagnosis is confirmed if urine pH remains >5.3 despite a plasma HCO3 of 21 mmol/L.
Renal Tubular Acidosis Treatment:
- Correction of the acidosis: Oral sodium bicarbonate or sodium citrate reverses bone demineralization.
- Potassium supplements: Potassium citrate in case of hypokalemia, stone formation, and nephrocalcinosis.
- Thiazide diuretics: They cause volume contraction and increased proximal sodium bicarbonate reabsorption.
Type 2 (“Proximal”) Renal Tubular Acidosis:
- Very rare and is due to failure of filtered sodium bicarbonate reabsorption in the proximal tubule. It leads to appearance of bicarbonate in urine and subsequent acidosis.
- Causes of type 2 RTA: Inherited forms of isolated type 2 RTA may show both autosomal dominant and recessive patterns of inheritance.
Consequences: Type 2 RTA usually occurs as part of a generalized tubular defect, together with urinary wasting of amino acids, phosphate, and glucose (Fanconi’s syndrome), as well as bicarbonate and potassium.
- Acidosis less severe than type 1 RTA
- Hypokalemia
- Inability to lower the urine pH below 5.5 despite systemic acidosis
- Appearance of bicarbonate in the urine despite a subnormal plasma bicarbonate
- Bone demineralization due to phosphate wasting.
Type 3 Renal Tubular Acidosis:
- It represents a combination of type 1 and type 2.
- Inherited type 3 RTA: It is caused by mutations resulting in carbonic anhydrase type II deficiency. It is characterized by osteopetrosis, cerebral calcification, and mental retardation.
Type 4 Renal Tubular Acidosis:
It is also known as “hyporeninemic hypoaldosteronism.” Its features are listed.
- Causes of type 4 RTA is presented.
Causes of type 2 renal tubular acidosis (RTA):
- Inherited
- Paraproteinemia
- Amyloidosis
- Hyperparathyroidism
- Heavy metal toxicity
- Drugs and toxins such as antiretroviral drugs, ifosfamide, lead, and cadmium
- Wilson’s disease
Renal Tubular Acidosis Treatment:
- Large/massive doses of sodium bicarbonate: May be required to overcome the renal “leak” of bicarbonate.
- Potassium supplementation: Often necessary because loss of bicarbonate in urine potentiates hypokalemia.
Features of type 4 renal tubular acidosis:
- Hyperkalemia
- Reduced plasma bicarbonate and hyperchloremia
- Normal ACTH stimulation test
- Low basal 24 hours urinary aldosterone
- Reduced response of plasma renin and plasma aldosterone to stimulation
- Correction of hyperkalemia by fludrocortisone
Treatment of type 4 RTA:
- Treatment of aldosterone deficiency: With a mineralocorticoid (For Example, fludrocortisone) and glucocorticoid for cortisol deficiency (if present).
- Hyporeninemic hypoaldosteronism by fludrocortisone and accompanying hypertension and edema are treated by thiazide or loop diuretic. Diuretics are also necessary for the control of hyperkalemia.

Chronic Kidney Disease:
Question 24. Write a short note on chronic kidney diseases.
Answer:
- Chronic kidney disease previously termed CRF or insufficiency.
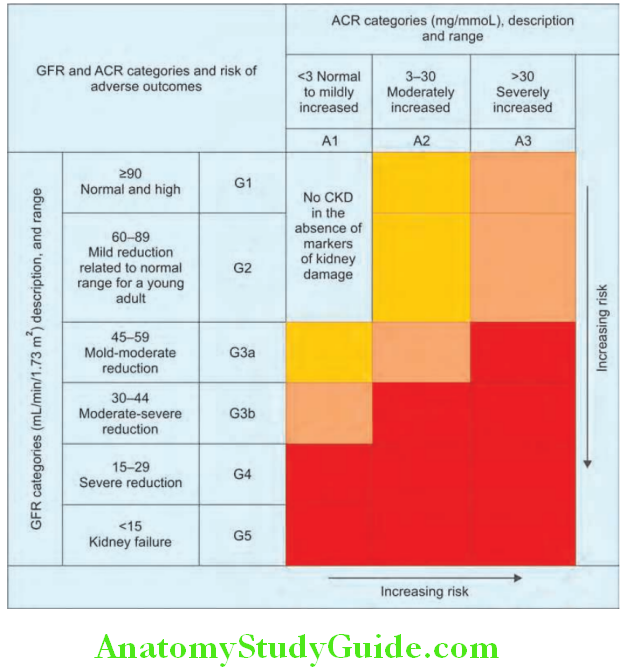
- Chronic kidney disease refers to a spectrum of long-standing (more than 3 months), usually progressive processes associated with irreversible worsening of renal function and decline in GFR. CKD spectrum ranges from abnormalities detectable only by laboratory testing to uremia.
- Revised CKD classification based upon GFR and albuminuria KDIGO 2013 is presented

Definition:
Question 25. Discuss the clinical and biochemical features of chronic kidney disease (CKD).
Answer:
Definition of chronic kidney disease (CKD):
- Glomerular filtration rate (GFR) of <60 mL/minute/1.73 m 2 for 3 months or more, with or without kidney damage or a urinary albumin to creatinine ratio >65 mg/mmol or protein creatinine ratio of 100 mg/mmol.
OR
Kidney damage for 3 or more months with or without decreased GFR, as evidenced by any of the following: - Microalbuminuria: Albumin excretion rate 30–300 mg/day in urine or urinary albumin >30 mg/day excretion of creatinine.
- Macroalbuminuria: Albumin excretion rate in urine 300 mg/day.
- Pathologic abnormalities such as abnormal findings on renal biopsy.
- Radiologic abnormalities such as scarring or polycystic kidneys on renal ultrasound scan.
Causes of Chronic Kidney Disease:
Important causes of chronic kidney disease:

Clinical Approach to Chronic Kidney Disease:
History:
- Duration of symptoms
- Drug intake: These include nonsteroidal anti-inflammatory agents, analgesic, and other medications (e.g., herbal medicines).
- Previous medical and surgical history, e.g., chemotherapy, SLE, malaria.
- Previous urinalysis or urea and creatinine values if performed.
- Family history of renal disease.
Symptoms:
- Unfortunately, early stages of CKD may be asymptomatic, despite the progressive loss of kidney function and accumulation of numerous metabolites.
- Usually there is a rough correlation between serum urea and creatinine levels and symptoms. Symptoms are common when the serum urea level exceeds 40 mmol/L.
Clinical Features:
Question 26. Write short essay/note on the clinical features of CKD.
Answer:
Clinical features of chronic kidney disease (CKD):
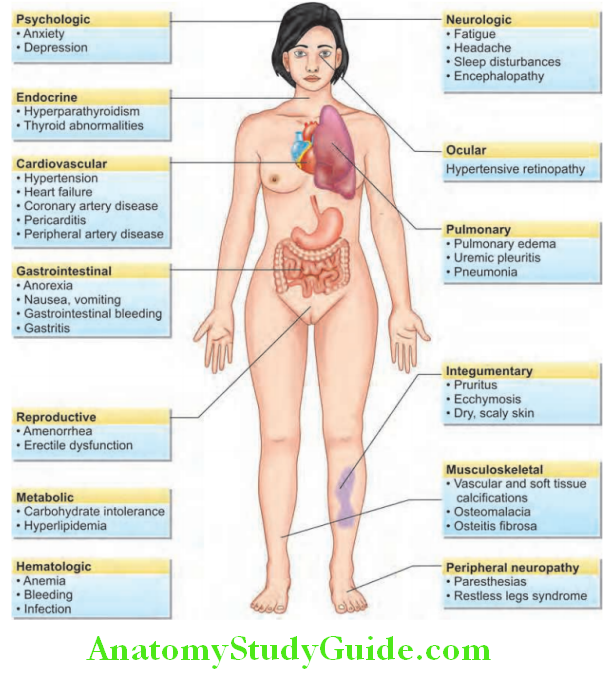
Complications of Chronic Renal Failure:
Question 27. Write short essay/note on the complications of chronic renal failure.
Answer:
Anemia: Various causes of anemia in CRF are listed.
Causes of anemia in chronic renal failure (CRF):
- Deficiency of erythropoietin (most important)
- Toxic effects of uremia on bone marrow precursor cells
- Bone marrow fibrosis secondary to hyperparathyroidism
- Deficiency of hematinic: Iron, vitamin B 12, folate because of reduced dietary intake due to anorexia.
- Intestinal absorption of iron is also impaired.
- Increased red cell destruction: Abnormal red cell membranes
- Increased blood loss
- Occult gastrointestinal bleeding
- Blood sampling
- Blood loss during hemodialysis
- Capillary fragility
- Due to platelet dysfunction and capillary fragility
Question 28. Write short essay/note on renal osteodystrophy.
Answer:
Metabolic Bone Disease: Renal Osteodystrophy:
The term “renal osteodystrophy” (bone mineral disorder), constitutes various forms of bone disease that may develop alone or in combination in CRF.
It includes:
- Hyperparathyroid bone disease (osteitis fibrosa cystica),
- Osteomalacia
- Osteoporosis
- Osteosclerosis
- Adynamic bone disease.
Pathogenesis of bone disease:
- Phosphate retention owing to reduced excretion by the kidneys release offibroblast growth factor 23 (FGF 23) and other phosphaturic agents by osteoblasts as a compensatory mechanism.
- Actions of FGF 23 are:
- Causes phosphaturia to normalize the plasma phosphate level.
- It downregulates 1a-hydroxylase to reduce intestinal absorption of phosphate.
- Decreased production of the la-hydroxylase enzyme by the kidney results in reduced conversion of 25-(OH) 2D 3 (25-hydroxyvitamin D) to the more metabolically active 1,25-(OH)2D3(1,25-dihydroxycholecalciferol).
- Its consequences are:
- Decreased activation of vitamin D receptors (VDRs) in the parathyroid glands leads to increased release of parathyroid hormone (PTH) causing secondary hyperparathyroidism.
- Decreased intestinal absorption of calcium causes hypocalcemia which leads in turn to increased PTH production by the parathyroid glands.
- Phosphate retention also indirectly lowers ionized calcium and these together results in an increase in PTH synthesis and release. The raised serum phosphate combine with calcium in the extracellular space, causing ectopic calcification in blood vessels and other tissues.
- Parathyroid hormone causes reabsorption of calcium from bone and increased reabsorption of calcium from proximal renal tubules.
This prevents hypocalcemia induced by 1,25-(OH)2D3 deficiency and phosphate retention.
- Secondary hyperparathyroidism causes increased osteoclastic activity, cyst formation and bone marrow fibrosis (osteitis fibrosa cystica).
- Long-standing secondary hyperparathyroidism finally causes hyperplasia of the glands with autonomous or “tertiary” hyperparathyroidism.
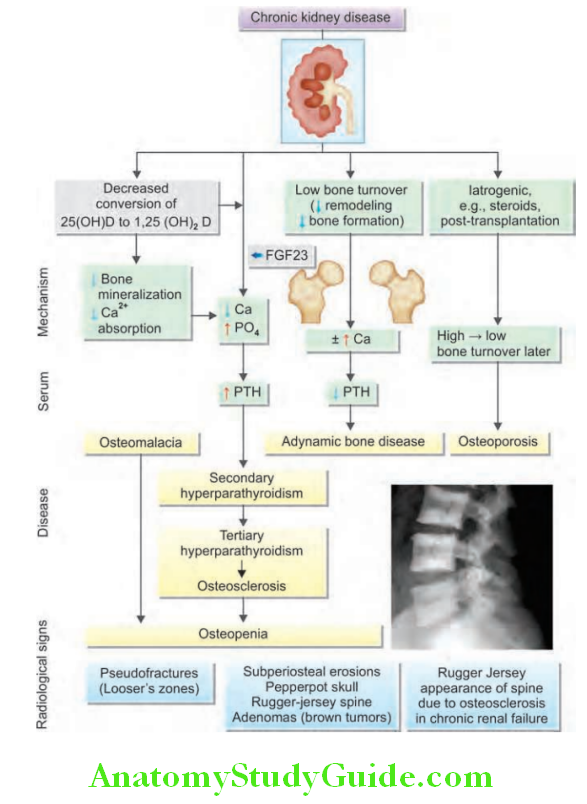
- Osteomalacia: It is due to impaired mineralization of osteoid caused by deficiency 1,25-(OH)2D3 and hypocalcemia.
- Osteosclerosis: Literally means “hardening of bone” characterized by increased bone density and is due to the direct result of long-standing PTH excess. Alternating bands of sclerotic and porotic bone in the spine give rise to a characteristic “rugger-jersey” appearance on X-ray.
- Osteoporosis is probably related to malnutrition and is commonly found in CRF, often after transplantation and the use of corticosteroids.
- Adynamic bone disease is the condition of in which both bone formation and resorption are depressed.
- Gastrointestinal complications:
- Reduced gastric emptying and increased risk of reflux esophagitis
- Increased risk of peptic ulceration and acute pancreatitis
- Constipation (especially in patients on continuous ambulatory peritoneal dialysis (CAPD)
- Gastrointestinal bleed.
- Metabolic abnormalities:
- Gout: Urate retention is a common in CRF.
- Insulin requirement and resistance: Insulin is catabolized by and to some extent excreted via the kidneys. Thus, insulin requirements in diabetic patients reduce as renal failure progresses. By contrast, end-organ resistance to insulin is observed in advanced renal impairment.
- Lipid metabolism abnormalities:Include
- Impaired clearance of triglyceride-rich particles and
- Hypercholesterolemia.
- Endocrine abnormalities: Include
- Hyperprolactinemia
- Increased luteinizing hormone (LH) levels in both sex
- Decreased serum testosterone levels
- Absence of normal cyclical changes in female sex hormones, resulting in oligomenorrhea or amenorrhea,
- Abnormalities of growth hormone secretion and action
- Abnormal thyroid hormone levels.
- Muscle dysfunction proximal myopathy.
- Nervous system
- Central nervous system:
- Unusual combination of depressed cerebral function and decreased seizure threshold.
- Dialysis disequilibrium develops if rapid correction of severe uremia is done by hemodialysis owing to osmotic cerebral swelling.
- Dialysis dementia is a syndrome characterized by progressive intellectual deterioration, speech disturbance, myoclonus, and fits.
- Autonomic nervous system:
- Increased circulating catecholamine level
- Impaired baroreceptor sensitivity
- Impaired efferent vagal function.
- Peripheral nervous system:
- Median nerve compression in the carpal tunnel due to b2-microglobulin-related
amyloidosis, - Restless legs syndrome,
- Polyneuropathy
- Psychiatric problems (anxiety, depression, phobias, and psychoses).
- Median nerve compression in the carpal tunnel due to b2-microglobulin-related
- Central nervous system:
- Cardiovascular disease: Increased (16-fold) incidence of cardiovascular disease, particularly myocardial infarction, cardiac failure, sudden cardiac death and stroke, uremic pericarditis or dialysis pericarditis. Hypertension develops in about 80% of patients with CRF.
- Malignancy: Raised incidence of malignancy. Malignant change can occur in polycystic kidney disease. Lymphomas, primary liver cancer, and thyroid cancers can also develop.
Pathogenesis of bone disease Investigations:
Lists the purpose of investigations in CKD:
Purpose of investigations in chronic renal failure:
- To identify the underlying cause wherever possible.
- To identify reversible factors (e.g., hypertension, urinary tract, obstruction, nephrotoxic drugs, and salt and water depletion).
- To screen for complications (e.g., anemia, renal osteodystrophy) and cardiovascular risk factors.
Urinalysis:
- Physical examination: Fixed specific gravity around 1.010 (isosthenuria) is seen in CRF.
- Chemical examination:
- Hematuria may indicate glomerulonephritis.
- Proteinuria, if heavy, is strongly suggestive of glomerular disease.
- Glycosuria with normal blood glucose level is common in CRF.
- Urine microscopy:
- White cells in the urine usually indicate bacterial urinary infection, sterile pyuria suggests papillary necrosis or renal tuberculosis.
- Eosinophils indicate allergic tubulointerstitial nephritis or cholesterol embolization.
- Red cells source may be from anywhere in the urinary tract between the glomerulus and the urethral meatus.
- Red-cell casts are suggestive of glomerulonephritis.
- Granular casts indicate active renal disease.
- Broad casts are seen in CRF.
- Urine culture should be performed. Early-morning urine samples should be cultured for tuberculosis.
Urine Biochemistry:
- 24-hour creatinine clearance is useful to know the severity of renal failure.
- Urine osmolality is a measure of concentrating ability.
- Urine electrophoresis and immunofixation for the detection of light chains in myeloma.
Serum Biochemistry:
- Serum urea and creatinine: The most consistent abnormalities in CRF are elevated levels of urea and creatinine. The level of serum creatinine correlates with the degree of renal damage.
- Electrophoresis and immunofixation for myeloma.
- Extreme elevations of creatine kinase and a disproportionate elevation in serum creatinine and potassium compared to urea suggestive of rhabdomyolysis.
- Other biochemical abnormalities include hypocalcemia, hyperphosphatemia, hyperuricemia and hyperkalemia.
Hematology:
- Anemia
- Eosinophilia suggestive of allergic TIN, vasculitis, or cholesterol embolism.
- Peripheral smear with fragmented red cells (Burr cells) and/or thrombocytopenia suggestive of intravascular hemolysis due to accelerated hypertension, hemolytic uremic syndrome or thrombotic thrombocytopenic purpura.
- Markedly raised erythrocyte sedimentation rate (ESR) is suggestive of myeloma.
Immunology:
- Low complement components may be seen in active glomerular disease (e.g., SLE, PSGN).
- Autoantibody screening helpful autoimmune diseases (e.g., SLE).
- Antibodies to streptococcal antigens (ASOT, anti-DNase B) if PSGN is suspected.
- Antibodies to hepatitis B and C
- Antibodies to HIV when HIV-associated renal disease is suspected.
- Malaria can cause glomerular disease in the tropics.
Radiological Investigation:
Ultrasound: Ultrasonography to assess the renal size and to exclude hydronephrosis. Renal ultrasound usually shows shrunken kidneys in CRF. In diabetic glomerulosclerosis, amyloidosis, polycystic kidney diseases, HIV nephropathy, bilateral hydronephrosis, and myeloma, the kidneys may be of normal size.
Plain abdominal radiography and CT (without contrast) to exclude low-density renal stones or nephrocalcinosis. CT may also useful for the diagnosis of retroperitoneal fibrosis and useful in some patients with suspected obstructive nephropathy.
MRI: Magnetic resonance angiography is useful in renovascular disease.
Renal Biopsy:
Aim: To establish the nature and extent of renal disease, which helps in treatment and predicting prognosis. It should be performed in patient with unexplained renal failure and normal-sized kidneys (exception diabetic glomerulosclerosis), unless there are strong contraindications.
Management of Chronic Renal Failure:
Management of CRF can be divided into three parts:
- Establishing the diagnosis and etiology of CKD and to detect any reversible factors
- Measures to prevent/slow down the further damage to the kidney (progression of CKD)
- Supportive measures (e.g., dialysis or transplantation) when necessary
Treatments aimed at specific causes of CKD: Optimization glucose control in diabetes mellitus, immunomodulatory agents for glomerulonephritis, and emerging specific therapies to retard cytogenesis in polycystic kidney disease. Any reversible factors should be detected and treated.
Reversible factors in chronic kidney disease:

Measures to Reduce the Symptoms and Progression of CRF:
- Following measures may stabilize or slow the decline of renal function.
- Control of hypertension and proteinuria: Goals of treatment include: Blood pressure should be reduced <130/80 mm Hg and proteinuria < 0.3 g/24 hours. If creatinine is below 3 mg/dL, ACE inhibitors and ARBs (inhibit the angiotensin-induced vasoconstriction of the efferent arterioles of the glomerular microcirculation) are the drugs of choice.
- Diet
- Protein restriction to 0.60 and 0.75 g/kg/day, i.e., about 40 g/day (with higher amount of essential amino acids). It reduces symptoms associated with uremia and may also slow the rate of renal decline at earlier stages of renal disease.
- Avoid foods with high potassium.
- Salt restriction is necessary in most of the patients. However, patients with salt-losing nephropathy (tubulointerstitial disease) require a high-salt intake.
- Slowing progression of diabetic renal disease by control of blood glucose and maintaining HbA1c in the range of 7.0–7.5.
- Use of lipid-lowering agents: Hypercholesterolemia is common in patients with significant proteinuria, and in patients with CKD. Lipid lowering reduces vascular events in nondialysis CKD.
- Cessation of smoking.
- Exercise and weight reduction.
- Avoid nephrotoxic medications.
Corrections of complications:
Calcium and phosphate control and suppression of PTH:
- Treatment of hypocalcemia and hyperphosphatemia: Both should be treated aggressively.
- Hypocalcemia:
- Treated with calcitriol or alphacalcidol (1-α-hydroxyvitamin D3) or paricalcitol (19-nor-1,25-dihydroxyvitamin D3) and calcium supplementation.
- Serum calcium level should be monitored to avoid hypercalcemia. Oral calcium carbonate also decreases the bioavailability of dietary phosphates.
- Hyperphosphatemia: Treated with phosphate binders. Previously, aluminum hydroxide was used to bind phosphate in the gut and was producing aluminum toxicity. Others include:
- Calcium carbonate and calcium acetate (serum calcium should be monitored).
- Polymer sevelamer carbonate (an anion-exchange resin).
- Lanthanum carbonate (a nonaluminum, noncalcium phosphate-binding agent) has higher incidence of side effects.
- Treatment of hyperparathyroidism: By the use of
- Calcium carbonate or acetate.
- Vitamin D analogs.
- Calcimimetics activate the calcium-sensing receptor in the parathyroid gland, thereby inhibiting PTH secretion. Cinacalcet is used in patients who are on dialysis.
Maintenance fluid and electrolyte balance:
- Fluid retention: If there is evidence of fluid retention intake of dietary sodium is restricted and loop diuretics may be necessary.
- Hyperkalemia: Usually responds to dietary restriction of potassium intake. Occasionally, ion-exchange resins may be necessary to remove potassium in the gastrointestinal tract. If hyperkalemia occurs during diuretic therapy, reduce or stop potassium-sparing diuretics, ACE inhibitors, and ARBs.
- Acidosis: Sodium bicarbonate may be effective, but can cause edema and hypertension owing to extracellular fluid expansion. Calcium carbonate, also used as a calcium supplement and phosphate binder is useful in treating acidosis.
Question 29. Write short essay/note on the management of anemia in chronic renal failure.
Answer:
Treatment of anemia in chronic renal failure The anemia of erythropoietin (EPO) deficiency is treated with recombinant (synthetic) human EPO (erythropoietin-alpha or -beta, or the longer-acting darbepoetin-alpha). Administration of erythropoietin-alpha through subcutaneous route is contraindicated in CRF and the intravenous route is used, initially 50 U/kg of epoetin-alpha over 1–5 minutes three times/week. EPO is less effective in the presence of iron deficiency, active inflammation or malignancy, and in patients with aluminum overload (found in dialysis).

Diffrences between acute kidney injury (AKI) and chronic kidney disease (CKD):
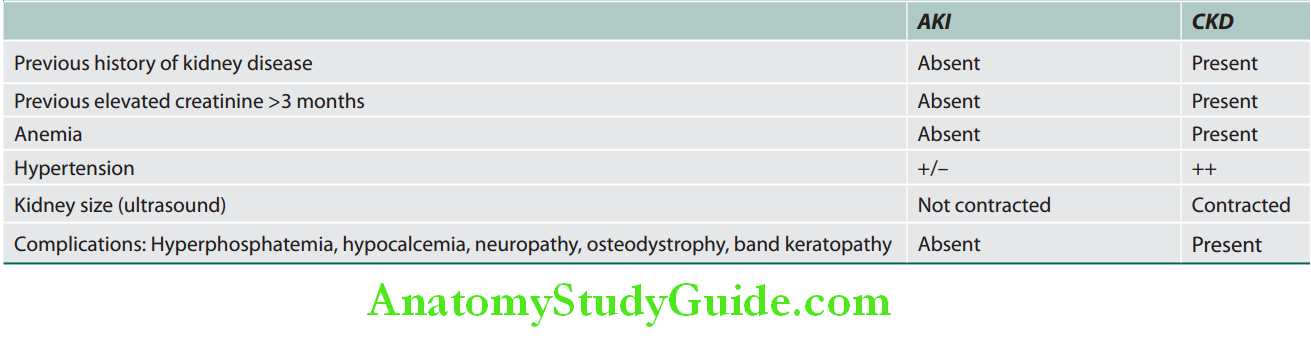
Cystic Diseases Of Kidney:
Polycystic Kidney Diseases:
Types: Two types
- Autosomal-recessive (childhood) polycystic kidney disease (ARPKD):
- Mutation in PKHD1 gene on chromosome 6, the gene that encodes for fibrocystin and biliary epithelial epithelial tissue
- A specialized layer of tissue formed by closely aggregated cells that line the outer surface of organs, blood vessels, the skin, and the inner surface of body cavities. Divided into squamous, cuboidal, and columnar types
- docIcon General histology → Epithelium → cells.
- All patients also have cysts in the liver and patients who survive infancy may develop congenital hepatic fibrosis and intrahepatic bile duct dilatation (Caroli disease).
- Autosomal-dominant (adult) polycystic kidney disease (ADPKD):
- ADPKD is the MC inherited renal disease
- ADPKD is caused by mutations in
-
- PKD1 on chromosome 16 codes for polycysti n-1 (PC1)
- PKD2 on chromosome 4 codes for polycysti n-2 (PC2)
- 10% patients may have no family history; due to spontaneous mutation
- PKD2: Slower disease progression
-
- It characterized by multiple thin-walled, spherical cysts in cortex and medulla of both kidneys. These cysts expand and cause destruction of kidney parenchyma and leads to renal failure.
- Associated extrarenal anomalies:
- Cysts in other organs such as liver (polycystic liver disease in more than 75% of cases) spleen, pancreas, thyroid, ovary, endometrium, seminal vesicles, and epididymis.
- Intracranial Berry aneurysms in the circle of Willis (in 10–30%) which may rupture and cause subarachnoid hemorrhages.
- Mitral valve prolapse and other cardiac valvular anomalies. Colonic diverticula, abdominal or inguinal hernias are associated.
- Clinical presentation
Question 30. Discuss the clinical features and complications of adult polycystic kidney disease.
Answer:
- May be at any age from the second decade and majority of patients remain asymptomatic until the fourth decade of life.
- Presenting manifestations
- Acute loin/flank pain (heaviness or dragging sensation), and/or hematuria (owing to hemorrhage into a cyst), UTI (pyelonephritis and renal cyst infection) or urinary tract stone formation and renal colic (due to passage of blood clots in the urine).
- Vague abdominal/loin or discomfort due to increased size of the kidneys
- Subarachnoid hemorrhage due to rupture of berry aneurysm
- Renal hemorrhage and hematuria
- Hypertension and its complications
- Complications of associated liver cysts
- When sufficient quantity of nephrons are destroyed, patient develops CKD and symptoms of uremia and/or anemia
- Polycythemia (due to increased erythropoietin production) is a rare complication and presentation of ADPKD.
- Rarely renal carcinoma
- Physical examination: Bilateral large abdominal masses of irregular kidneys.
- Investigation:
- Abdominal ultrasound is used for establishing the definitive diagnosis.
- MRI is more sensitive than ultrasound and can detect small cysts.
- IVU: Rarely performed nowadays and may demonstrate the characteristic “drooping water-lily sign.”
- Screening: The children of patients with established ADPKD should undergo screening.
Adult polycystic kidney disease Treatment:
- Control of hypertension
- Control of pain
- Treatment of infection
- Management of CKD—dialysis or transplantation (if required) and
- Specific treatment—vaptans, mammalian target of rapamycin (mTOR) inhibitors (sirolimus, everolimus), and somatostatin
analogs have been tried in treatment of ADPKD.
Medullary Sponge Kidney:
- Medullary sponge kidney (MSK) is characterized by nonprogressive dilatation of collecting ducts and tubules.
- Usually inherited as autosomal dominant
- May be associated with other developmental and genetic disorders
- Often asymptomatic, and may manifest with hematuria, nephrolithiasis, and infection.
Medullary Cystic Disease or Juvenile Nephronophthisis:
- Two distinct genetic and age-related forms with similar renal morphology. Autosomal recessive manifests during childhood and is frequently associated with extrarenal abnormalities such as ophthalmologic, mental retardation, cerebellar ataxia, skeletal anomalies, and hepatic fibrosis.
- The disease progresses to end-stage renal disease (ESRD) before the age of 25 years.
Obstructive Uropathy:
- Obstructive uropathy is a term used for obstruction in the urinary passage involving the urethra, bladder, ureters or pelvicalyceal system. It leads to impedance to urine flow and consequent damage to renal function.
- Nature of obstruction: It is acute or chronic.
- Acute obstruction: The changes in kidney function are completely reversible after the relief of obstruction.
- Chronic obstruction: It leads to irreversible structural and functional changes.
Site and Cause of Urinary Tract Obstruction:

Nephrolithiasis:
- Nephrolithiasis (also known as kidney stones or renal calculi) is a common disorder characterized by the formation of aggregates of microscopic crystals into solid objects (stones).
- Constituent: Usually contain calcium or phosphate along with small amounts of proteins and glycoproteins.
- Size: They vary greatly in size from millimeters to centimeters.
- Age and gender: It usually appears during middle age and is more common in men than women (M:F = 2:1).
- Risk of recurrence: Calculi recur in about 50% of cases within 3–5 years.
Etiology:
Question 31. Write short note on predisposing factors for renal stones.
Answer:
- Normal urine contains inhibitors of crystal formation (citrate, inorganic magnesium, pyrophosphate, glycosaminoglycans and nephrocalcin) that prevent the formation of calculi.
- When the chemical concentration of urine is altered, the calculus forming substances may exceed their maximum solubility in water and may favor crystal formation.
Predisposing factors for renal stones:
Environmental and dietary factors:
- Conditions that favor low volume of urine: High atmospheric temperatures, low intake of fluid
- Diet: High protein, high sodium, low calcium
- High excretion of sodium, oxalate, and urate excretion
- Low excretion of citrate
Acquired causes:
- Hypercalcemia
- Ileal disease or resection (increases oxalate absorption and urinary excretion)
- Renal tubular acidosis (RTA) type I (distal)
- Infection: Urinary infection by urea-splitting organisms such as Proteus, Pseudomonas, Klebsiella, Staphylococcus, and Mycoplasma
Congenital and inherited disorders:
- Medullary sponge kidney
- Familial hypercalciuria
- Cystinuria
- Renal tubular acidosis (RTA) type I (distal)
- Primary hyperoxaluria
Congenital and inherited disorders Types:
Calcium Stones (Oxalate Calculus/Calcium Oxalate):
- Most (80%) renal stones are composed of calcium complexed with oxalate (calcium oxalate) or phosphate (calcium phosphate) or a mixture of these (calcium oxalate + calcium phosphate). These stones are radiopaque.
- Causes.
Uric Acid Stones:
- Commonly found in patients with hyperuricemia (e.g., gout) and diseases involving rapid cell turnover (e.g., leukemias). However, more than 50% of patients have neither hyperuricemia nor increased urinary excretion of uric acid.
- Uric acid is insoluble in acidic urine and urine pH below 5.5 may predispose to uric acid stones.
- Uric acid stones are radiolucent stones.
- Causes.
Types and causes of renal stones:
Type:
- Calcium oxalate and/or calcium phosphate (~80%)
- Idiopathic hypercalciuria: Most common
- Hypercalciuria and hypercalcemia
- Hyperoxaluria: Enteric (oxalate-containing foods, salt, protein-meat) vitamin C abuse, primary
- Hyperuricosuria
- Idiopathic
- Uric acid (~7%)
- Associated with hyperuricemia
- Associated with hyperuricosuria
- Idiopathic
- Struvite (magnesium, ammonium, phosphate) ~10%
- Urinary tract infection
- Cystine (~2%)
- Others/unknown (~1%)
Struvite Stones or (Triple stones/Magnesium, Ammonium, Phosphate Stones):
- They are composed of calcium phosphate often with magnesium and ammonium phosphate and are known as struvite stones or triple phosphate stones.
- Causes: These stones are unique in that they develop after infections of the urinary tract by urea-splitting bacteria (e.g., Proteus), which convert urea to ammonia → produces alkaline pH + slowing of urine flow (people with abnormal urinary drainage, such as those with ureteral reflux, neurogenic bladder, other forms of bladder dysfunction, or ureteral diversions) → precipitation of magnesium, ammonium, phosphate (struvite), and calcium phosphate (apatite).
Cystine Stones:
- Cystine stones are uncommon and associated with cystinuria, which is due to genetic defects in the renal reabsorption of cystine or other amino acids.
- Stones form at low urinary pH (acidic urine)
- Cystine stones also are radiopaque
- Recur frequently in most affected individuals
- Diagnosis is suggested by a positive urine nitroprusside test and confirmed by analysis of the calculus.
- Urine sediment: It may show the characteristic hexagonal cystine crystals and crystal formation is favored in acidic urine (low pH).
Cystine Stones Clinical Manifestations:
- Most common presenting symptoms:
- Severe colicky flank and/or abdominal pain radiating to the anterior abdominal wall
- Hematuria (microscopic or gross). This symptom complex termed renal or ureteric colic. These symptoms are due to partial or full obstruction produced by stones while passing through the ureter.
- Asymptomatic: Stone is incidentally discovered in an abdominal computed tomography or ultrasound examination for other purposes.
- Symptoms depending on the site of stone:
- Stones in the renal pelvis usually are painless unless infection or obstruction is present.
- Ureteral stones may cause nausea, vomiting, and severe abdominal and/or flank pain radiating into the groin, urethra, or genitalia.
- Stones at the ureterovesical junction may produce dysuria, frequency, and urgency, even in the absence of infection.
- Once in the bladder, stones are normally passed out without difficulty, but they can remain there and grow to very large sizes in patients with bladder dysfunction.
- Fever and pyuria suggest associated UTI and must be confirmed by urine culture.
- Chronic and complete obstruction can result in hydronephrosis and loss of function in the affected kidney.
Cystine Stones Diagnosis/Investigations:
- CT-KUB (CT of kidney, ureter, and bladder) is the gold standard for diagnosis. It defines the size and location of any stones (including nonradiopaque stones, such as those containing uric acid and cysteine) in the urinary tract.
- Abdominal ultrasound or intravenous pyelograms (IVPs) are generally inferior in sensitivity and specificity compared to the computed tomography scan.
- A plain X-ray of the abdomen has limited diagnostic utility, but may be useful to track progression of previously diagnosed radiopaque stones.
- Urine analysis: To confirm the presence of blood and check for evidence of infection. Urinary sediment should be examined under the microscope for crystals. Any stones passed in urine should be saved for analysis. Metabolic evaluation of stones should always be performed before starting on a therapeutic regimen. Urine culture should be done when clinically indicated.
- Laboratory tests: These include serum studies for renal function, electrolytes, calcium, magnesium, phosphate, and uric acid. If there is hypercalcemia, a PTH level should be obtained.
Important investigations in patients with renal calculi:
- Routine urine examination (albumin, RBC, WBC, casts and crystals, and pH)
- Renal functions (blood urea, serum creatinine)
- Imaging (ultrasonography and IV urography)
- To find out underlying cause
- Chemical analysis of calculus passed in urine
- Plasma calcium, phosphate, and parathormone
- 24-hour urine for estimation of calcium, oxalate, urate, cysteine
Cystine Stones Treatment:
Cystine Stones Acute Management:
- Acute colic should be treated with an effective/powerful analgesic. Diclofenac orally or IV infusion or as a suppository is very effective. However, opiates may be needed.
- Increase the water intake to maintain a daily urine output of 2 L or more. If this is not possible, intravenous fluids should be given to increase urinary output.
- Stone removal strategies are based on the size of the stone.
- Most small calculi (<5 mm in diameter) usually pass spontaneously.
- Alpha blockers (e.g., tamsulosin) facilitate spontaneous expulsion of distal ureteral stones of <6 mm size.
- Stones >1 cm diameter usually need urological or radiological intervention. Extracorporeal shock wave lithotripsy (ESWL) causes fragmentation of most of the stones into pieces <5 mm, which then pass spontaneously. Ureteroscopy with a YAG laser (for larger stones) and percutaneous nephrolithotomy also can be used. Open surgery is rarely necessary.
- Surgical removal is attempted only if the stone passage becomes complicated. Complicating features include failure to pass stone in 3–7 days, acute renal failure, gross hematuria with clots, or infection. Open surgical procedures are very rarely performed.
- Endoscopic or surgical removal is recommended for:
- Persistent pain not relieved by analgesia
- Stone obstructing a single kidney, or bilateral ureteral obstruction and/ or pyonephrosis.
Cystine Stones Chronic Management:
- Fluid: Increase the urine volumes (dilution) so that it reduces supersaturation and prevents new stone formation. Increase the fluid intake until daily urine output exceeds 2–3 L.
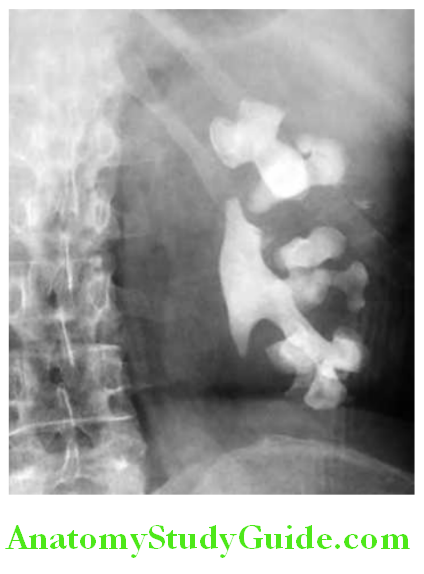
- Avoid: Vitamin D supplements (increase calcium absorption and excretion) and vitamin C supplementation (increases oxalate excretion).
- Calcium-containing stones
- Modification of diet to reduce risks of stone formation in recurrent stone formers (>5 years between episodes).
- Thiazide diuretics (e.g., 12.5–25 mg of hydrochlorothiazide) reduce urinary calcium excretion and are useful in hypercalciuric calcium stone formers.
- Potassium citrate (60 mEq/day) in divided doses is used in hypocitraturia and also helps to prevent recurrence in patients with normal urinary citrate levels.
- Oxalate: Avoid foods rich in oxalate (spinach, rhubarb).
- Uric acid stones:
- In patients with hyperuricosuria (>1,000 mg of uric acid per 24 hours) and normal serum calcium, allopurinol in the dose of 100–200 mg/day reduce uric acid synthesis.
- Alkalinization of the urine to a pH of 7 or above to increase uric acid solubility and to dissolve uric acid crystals. This is achieved by supplementation with potassium citrate, potassium bicarbonate, or sodium bicarbonate in divided doses, or acetazolamide to alkalinize the nocturnal urine.
- Cystine stones: Decrease the urinary concentration of cystine below the solubility limit of 200–300 mg/L. Apart from the methods already described, alkalinization of the urine to a pH above 7.5 is critical. This usually can be achieved with Shohl’s solution (contains sodium citrate).
Urinary Tract Infections:
Question 32. Discuss the etiopathogenesis, clinical features, investigations, and management of urinary tract infection.
Answer:
Definition:
A UTI is associated with multiplication of organisms in the urinary tract and is defined as the presence of more than 105 organism/mL in the midstream sample of urine (MSU).
Urinary Tract Infections Clinical Presentation:
Clinical spectrum of urinary tract infection (UTI):

Anatomic Classifiation of Urinary Tract Infections:
- Lower urinary tract infections: Include cystitis (bladder), prostatitis, and urethritis.
- Upper urinary tract infections: Include infection of kidneys and their collecting systems (pyelonephritis) and perinephric abscess.
Etiology:
Question 33. What are the predisposing causes of urinary tract infection?
(or)
Write short essay on.
Answer:
- Etiology and factors responsible for urinary tract infections.
- Risk factors for urinary tract infections.
Causative organisms: Majority (~85%) of UTI are the caused by gram-negative bacilli which are normal inhabitants of the intestinal tract (enteric origin).
- Most common pathogens: Escherichia coli (80% cases), Proteus, Klebsiella, Enterobacter, and Pseudomonas.
- Less common: Streptococcus faecalis, staphylococci, and fungi.
- In immunocompromised patients: Viruses (polyoma virus, cytomegalovirus, and adenovirus).
Pathogenesis:
Question 34. Differences between male and female urinary tract infection.
(or)
Write short note on causes of urinary tract infection in male.
Answer:
Route of infection: Bacteria can reach urinary tract via the bloodstream, the lymphatics or by direct extension (e.g., from a vesicocolic fitula), but in majority of cases via the ascending transurethral (via the urethra) route.
1. Ascending infection: It is the most common route of infection of the renal parenchyma, i.e., pyelonephritis. It is a form of endogenous infection, where the source of infecting organisms is the patient’s own fecal flora. The infection ascends from the lower urinary tract into the renal parenchyma. Different steps in the pathogenesis of pyelonephritis are:
- Colonization of the distal urethra and introitus (in the female): By enteric or coliform bacteria from the perineum due to poor hygiene and hormonal effects.
- Entry from the urethra to the bladder: Organisms may enter the bladder during urethral catheterization or other instrumentation. Urinary infections are more common in females, because of:
- Shorter urethra (4 cm)
- Absence of prostatic fluid which has antibacterial properties
- Hormonal changes in women which affect the adherence of bacteria to the mucosa.
- Trauma to the urethra during sexual intercourse facilitates entry of introital bacteria into the bladder.
- Gram-negative enteric organisms residing around the anal region also colonize the periurethral region.
- Urinary tract obstruction and stasis of urine
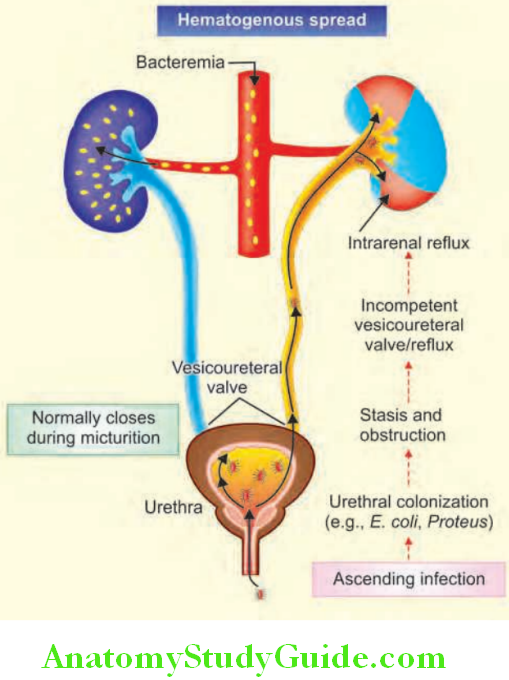
- Obstruction/bladder dysfunction: Causes incomplete emptying and increased residual volume of urine.
- Examples: benign prostatic hypertrophy, tumors, strictures, or calculi.
- Neurogenic bladder dysfunction: Diabetes, spinal cord injury, tabes dorsalis, and multiple sclerosis predisposes to UTI.
- Stasis: Organisms introduced into the bladder can multiply when there is stasis.
2. Hematogenous route: It is less common route of infection. Because of rich blood supply, bacteria can seed the kidneys during the course of septicemia or infective endocarditis through the bloodstream. It occurs with nonenteric organisms (e.g., staphylococci), fungi, and viruses. Hematogenous infections occur in:
- The Presence Of Ureteral Obstruction,
- Debilitated Patients
- Patients receiving immunosuppressive therapy.
Clinical Features:
Question 35. List the differences between complicated and uncomplicated UTI.
Answer:
Clinical features of UTI:

Uncomplicated versus Complicated UTI:
Differences between uncomplicated versus complicated urinary tract infection (UTI):
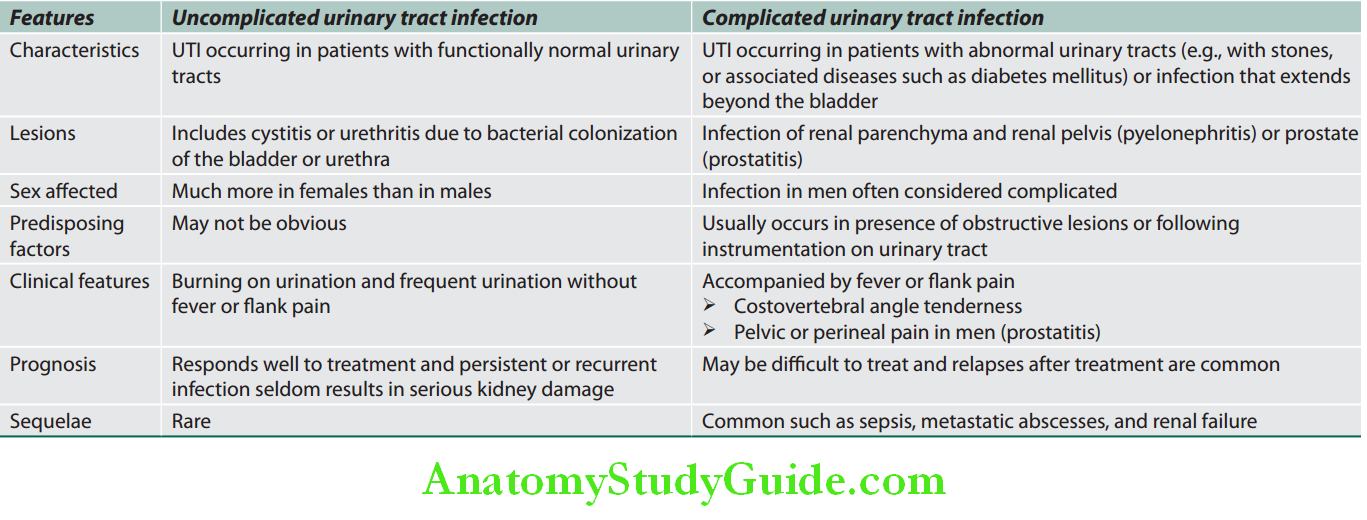
Acute urethral syndrome: About one-third of females with dysuria and pregnancy have either insignificant bacteria in midstream culture or completely sterile cultures and is known as acute urethral syndrome. It is often due to infection with usual organisms (culture shows only 102–104 bacteria) or due to unusual organism (Neisseria gonorrhoeae, Chlamydia trachomatis).
Causes of Dysuria:
- Urinary tract infections (e.g., urethritis, cystitis, vaginitis, pyelonephritis)
- Sexually transmitted infections
- Genital herpes
- Chlamydia
- Gonorrhea
- Inflammation and irritation of bladder and urethra, For Example, stones
Factors that indicate a complicated UTI:
- Anatomical abnormality of the urinary tract
- Bacterial persistence in spite of adequate therapy
- Childhood UTI
- Diabetes
- Elderly
- Functional abnormality of the urinary tract [vesicoureteric reflux (VUR); neurogenic dysfunction]
- Gestational (pregnancy); Gender-male
- Hospital acquired (catheter induced, instrumentation, stents)
- Immunosuppression
Factors Determining Symptomatic Infection:
- Virulence: Ability of the organism to adhere to epithelial cells determines the degree of virulence.
- Innate host defense: The following hosts defense mechanisms are required for the prevention of UTI:
- Neutrophils: Activation of neutrophils is necessary for bacterial killing and their impaired function predisposes an individual to severe UTI.
- Complement: Complement activation with IgA production by uroepithelium (acquired immunity) plays a major role in defense against UTI.
- Commensal organisms (e.g., lactobacilli): Form part of the normal host defense and their eradication by spermicidal jelly or disruption by certain antibiotics results in overgrowth of E. coli.
- Urine flow and normal micturition: Wash out bacteria and stasis of urine predisposes to UTI.
- Uroepithelium—mannosylated proteins: Such as Tamm–Horsfall proteins (THPs), have antibacterial properties and are present in the mucus and glycocalyx covering uroepithelium. Normally, they interfere with bacterial binding to uroepithelium. Thus, disruption of this uroepithelium by trauma (e.g., sexual intercourse or catheterization) predisposes to UTI.
- Pregnancy is associated with an increased incidence of UTI.
- Factors that favor UTI include:
- Decreased Ureteral Tone (Progestational Activity)
- Decreased Ureteral Peristalsis
- Transient incompetence of the vesicoureteral valves.
Honeymoon Cystitis (or “Honeymoon Disease”):
Question 36. Write short note on honeymoon cystitis.
Answer:
Honeymoon cystitis is caused as a result of frequent or prolonged sexual activity, as would typically be expected in the honeymoon period of a marriage. It can occur when a woman has sex for the first time, or when a woman has sex after a long period of time without any sexual activity.
Honeymoon Cystitis Symptoms: Same as with cystitis due to other cause.
During sex, E. coli on the skin around your anus can be transferred to urethra of women (for example, via partner’s figers or penis).
Honeymoon Cystitis Investigations:
Urine Examination:
- Dipstick tests: Most gram-negative organisms reduce nitrates to nitrites and dipstick tests (nitrite test) are used to detect nitrite in urine. Dipsticks that detect significant pyuria depend on the release of esterases from leukocyte (leukocyte esterase test).
- Positive dipstick tests for both nitrite and leukocyte esterase are highly predictive of acute infection.
- Microscopic examination: For leukocytes, leukocyte casts and red cells.
- Culture and sensitivity of a freshly voided clean-catch midstream specimen of urine.

Causes of sterile pyuria:
- Partially treated UTI
- Tuberculosis of urinary tract
- Calculi in urinary tract
- Infection with other organisms (e.g., Chlamydia, Corynebacterium, etc.)
- Tumors of bladder
- Chemical cystitis/drugs
- Prostatitis
- Interstitial nephritis
- Appendicitis
Special Investigations:
- Prostatitis
- Per rectal examination of the prostate
- Prostatic massage followed by urine culture
- Cystitis: Cystoscopy.
- Renal ultrasonography CT-KUB, MR urogram: To identify obstruction, cysts, and calculi.
- Intravenous urography (IVU), including a postvoid film of bladder: To identify physiological and/anatomical abnormalities of urinary tract.
- Dimercaptosuccinic acid (DMSA) renal scan: For pyelonephritis.
- Micturating cystourethrogram (MCU) to identify vesicoureteric reflux and disturbed bladder emptying.
- In females with recurrent UTI, pelvic examination to exclude cystocele, rectocele, and uterine prolapse.
Honeymoon Cystitis Treatment:
- Antibiotic therapy: The choice of antibiotic depends on the result of urine culture and sensitivity of urine.
- The commonly employed antibiotics include cotrimoxazole (trimethoprim and sulfamethoxazole 1 double strength tablet two times daily), ampicillin (250 mg three times daily), amoxicillin (250 mg 8-hourly three times daily), oral cephalosporin, nitrofurantoin (50 mg three times daily), and quinolones. Treatment is over 3–5 days in uncomplicated infections (nitrofurantoin for 7 days) and for 7–10 days in complicated infections.
- Hospitalized patients:
- No risk factors for infection with a multidrug-resistant organism → ceftriaxone (1 g IV once daily) or piperacillin-tazobactam (3.375 g IV every 6 hours)
- At least one risk factor for infection with a multidrug-resistant organism → antipseudomonal carbapenem (imipenem 500 mg IV every 6 hours, meropenem 1 g IV every 8 hours, or doripenem 500 mg IV every 8 hours).
- Enterococcus species or MRSA are suspected → add vancomycin, daptomycin, or linezolid.
- “Single-shot” treatment with 3 g of amoxicillin or 1.92 g of cotrimoxazole can be used in patients with bladder symptoms of less than 36 hours duration and without any previous history of UTI.
- If patients has a calculi, catheter or other obstructions, no antibiotic is necessary unless symptomatic.
- Fluid intake: A high (2 L daily) fluid intake during treatment and for some subsequent weeks to initiate water diuresis, so as to maintain a high rate of urine flow.
- Other measures:
- Regular complete emptying of urinary bladder at 2- to 3-hour intervals.
- Alkalinization of urine
- Urinary analgesics (e.g., phenazopyridine) and antispasmodics (e.g., hyoscyamine) to be given for detrusor spasm.
- If patients presents for the first time with high fever, loin pain and tenderness, urgent renal ultrasound examination is performed to exclude an obstructed pyonephrosis. If this is present percutaneous nephrostomy is performed to drain it.
- Cranberry juice inhibits adherence of uropathogens to uroepithelial cells, hence advisable to prevent recurrent UTI.
- In females: The incidence of UTIs can be reduced by
- Adequate Perineal Hygiene,
- Emptying the bladder before bedtime and before and after intercourse
- Application of 0.5% cetrimide cream to periurethral area before intercourse. Atrophic vaginitis should be identified and treated in postmenopausal women.
- Avoidance of constipation (may impair bladder emptying).
Asymptomatic bacteriuria: It requires treatment
- If a patient is pregnant
- In case of renal transplantation
- Before planning for a urologic surgery. Treatment of asymptomatic bacteriuria in pregnant women decreases the occurrence of pyelonephritis.
Acute Pyelonephritis:
Question 37. Discuss the etiology, clinical features, investigations, diagnosis, and management of acute pyelonephritis.
(or)
Write short essay/note on common causes, diagnostic features, and management of upper urinary tract infections.
Answer:
Acute Pyelonephritis Definition: Acute pyelonephritis is an acute suppurative inflammation of upper urinary tract affecting the tubules, interstitium, and renal pelvis. There is often coincident cystitis.
Acute Pyelonephritis Clinical Features:
Classic triad:
- Loin pain
- Fever
- Tenderness over the kidneys.
Presence of this triad and significant bacteriuria usually indicates acute pyelonephritis.
- Sudden onset of pain in one or both loins (costovertebral/renal angle), radiating to the iliac fossa and suprapubic area.
- Fever with chills and rigors, and malaise
- Tenderness and guarding in the renal angle
- Dysuria, frequency, and urgency. Frequent passage of small amount of urine accompanied by scalding, cloudy urine.
- Acute pyelonephritis caused by the presence of obstruction (e.g., stone, tumor, bladder neck obstruction, enlarged prostate), can be very severe and may progress to renal abscess.
- Rarely, acute pyelonephritis may be associated with necrotizing papillitis/papillary necrosis. Causes include diabetes mellitus, chronic urinary obstruction, analgesic nephropathy, and sickle-cell disease.
Investigations:
Question 38. Write short note on urinary abnormalities in acute pyelonephritis.
Answer:
- Peripheral blood: Characteristically shows leukocytosis.
- Urine examination
- Microscopy shows numerous pus cells and organisms, WBC casts, some red cells, and epithelial cells.
- Culture of midstream urine (MSU) shows growth the causative organism.
- Other investigations: When the diagnosis is uncertain, other investigations are required to exclude anatomical abnormalities of the urinary tract. When acute pyelonephritis is severe, the possibility of any obstruction or abscess formation must be excluded. It is also necessary, if there is inadequate response or rapid recurrence of pyelonephritis following appropriate therapy.
- Ultrasonography: Most frequently done, but is relatively insensitive.
- Computerized tomography (CT) is performed if ultrasound is normal. It can detect calculi, obstruction, hemorrhage, gas, enlargement of kidney, and inflammatory masses (damaging renal function).
- Intravenous urography is usually not done at present.
- Radionuclide scan: The radionuclide agent is injected into bladder through the suprapubic route may be useful to detect reflux.
- Dimercaptosuccinic acid renal scan: If performed within 2 days DMSA can help diagnose acute pyelonephritis. It is advisable to scan after 4–6 months of an acute episode to detect renal parenchymal damage.
Acute pyelonephritis Management:
Intravenous antibiotics: Intravenous ampicillin, amoxicillin plus aminoglycosides (e.g., tobramycin), cephalosporin (e.g., cefuroxime), quinolone (e.g., ciprofloxacin), or a combination beta-lactam/betalactamase inhibitor (piperacillin-tazobactam) or carbapenem (meropenem) (discussed above).
Persistent or Recurrent UTI:
Question 39. Write short essay/note on persistent or recurrent urinary tract infection and its management.
Answer:
- In some patients with UTI, the causative organism persists on repeat culture of urine in spite of treatment, or reinfection develops with any organism after an interval. Such cases, there is more probably an underlying cause is present and hence a more detailed investigation is necessary.
- Recurrent infections are common in females.
- Recurrent UTI, especially in the presence of an underlying cause may produce permanent renal damage. If an underlying cause cannot be treated, suppressive antibiotic therapy can prevent recurrence and reduce the risk of septicemia and renal damage.
- Culture and sensitivity of urine should be at regular intervals.
Persistent or Recurrent UTI Treatment: Two or three antibiotics in sequence, rotating every 6 months often reduces the emergence of resistant organisms. Other methods discussed above
Emphysematous Pyelonephritis:
Question 40. Write short note on emphysematous pyelonephritis.
Answer:
- It is a type of severe acute pyelonephritis most often caused by E. coli and Klebsiella pneumoniae.
- The major risk factors for emphysematous UTIs are diabetes and urinary tract obstruction.
- The infections primarily occur in women at a mean age of about 60 years.
- Abdominal pain is the major clinical manifestation with renal angle tenderness. Pneumaturia may be seen after bladder catheterization.
- The diagnosis of emphysematous UTIs is made by abdominal imaging; computed tomography is more sensitive than plain films and can detect obstructing lesions.
- Treatment: Parenteral antibiotics, percutaneous drainage/nephrectomy.
Chronic Pyelonephritis:
Question 41. Write short essay on the clinical features, investigations, and management of chronic pyelonephritis (reflux nephropathy).
Answer:
Chronic pyelonephritis is a chronic inflammation of tubulointerstitial tissue leading to scarring of calyces, pelvis, and renal parenchyma.
Chronic Pyelonephritis Types:
- Reflux nephropathy (chronic reflux-associated pyelonephritis/atrophic pyelonephritis): It results recurrent UTIs from a combination of factors such as:
- Congenital vesicoureteral reflux and intrarenal reflux: Chronic pyelonephritis is so often associated with vesicoureteric reflux and some feel that it is better named “reflux nephropathy.” Reflux may be unilateral or bilateral and accordingly causes scarring of one or both the kidney.
- Superimposition of a urinary infection acquired in infancy or early childhood.
- Chronic obstructive pyelonephritis: Develops due to recurrent infections superimposed on obstructive lesions, which lead to renal inflammation parenchymal atrophy, and scarring.
Chronic pyelonephritis is an important cause of renal damage and ESRD. Morphological changes are depicted.
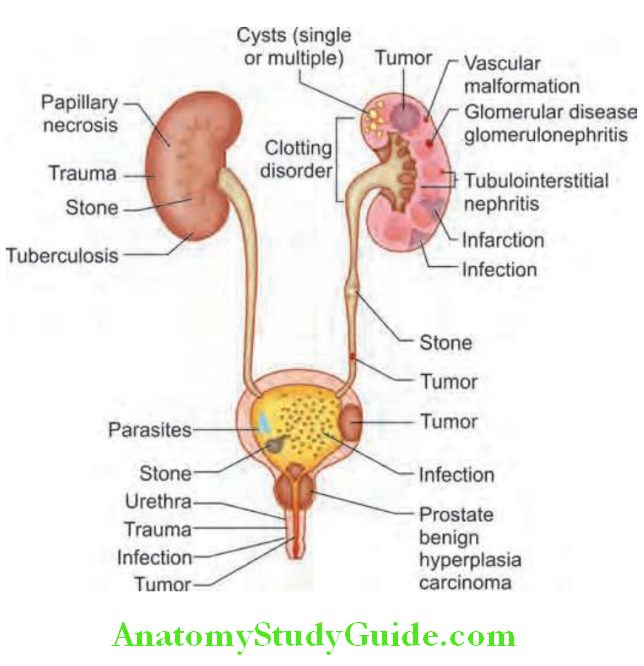
Chronic Pyelonephritis Clinical Features:
- Reflux pyelonephritis may be of silent onset. Clinical features includes back pain, lassitude, or symptoms of uremia or hypertension. Other symptoms include frequency of micturition, dysuria, pyuria, and bacteriuria.
- Xanthogranulomatous pyelonephritis (XGP): Predominantly affects elderly women. The condition is commonly associated with renal calculi or obstructive uropathy and is usually unilateral. The kidney is enlarged, and nonfunctioning and may even mimic a tumor.
Chronic Pyelonephritis Investigations:
- Culture of the urine: E. coli is the most common organism causing infection. Other organisms include Proteus, Pseudomonas aeruginosa, and staphylococci.
- Ultrasound of kidneys
- CT scan of the kidneys: Diagnostically, it reveals irregular renal outlines, clubbed calyces, and a variable decrease in the size of kidney. The condition may be unilateral or bilateral and may affect entire or part of the kidney.
- DMSA scan is more accurate than IVU.
- Intravenous urogram: Shows localized contraction of the renal substance associated with clubbing of the adjacent calyces.
- Micturating cystourethrogram (MCU): Reveals vesicoureteric reflux, but has been replaced by radionuclide cystography scan.
- Cystoscopy and urography: May detect any abnormality causing obstruction to the flow of urine.
Chronic Pyelonephritis Management:
- Eradicating predisposing factors:
- Examples include treatment of calculi and malformations.
- Surgery is performed if the vesicoureteric reflux persists and is indicated only when the disease is confined to one kidney. Most cases of childhood reflux likely to disappear spontaneously. Patients with end stage renal failure require renal replacement.
- Treatment of infection: Meticulous early detection and control of infection can prevent further scarring and allow normal growth of the kidneys. Unfortunately, once the parenchyma is scarred it becomes susceptible to blood–borne reinfection sometimes with a different and resistant organism. Thus, antibiotics may be of temporary benefit and progressive renal damage is common.
- Use of appropriate antibiotics for 7 days.
- If the infection cannot be eradicated suppressive therapy may be given several months with trimethoprim (100 mg at bed time) or nitrofurantoin (50 mg at bed time).
- Morphological changes in reflx nephropathy compared with normal.
- Other measures:
- Complete emptying of the bladder should be advised.
- Double micturition is advised if reflux. This consists of emptying of the bladder and then again second time attempt to empty the bladder, after about 10–15 minutes.
- Control of hypertension because severe hypertension is a relatively common cause of end-stage renal failure in childhood or adult life.
Tuberculosis Of The Urinary Tract:
Question 42. Describe renal tuberculosis.
(or)
Briefly outline the clinical features, investigations, and treatment of tuberculosis of the urinary tract.
Answer:
Tuberculosis of the urinary tract results from hematogenous spread from a distant primary focus of infection that is often impossible to identify.
Types of lesions in renal tuberculosis:
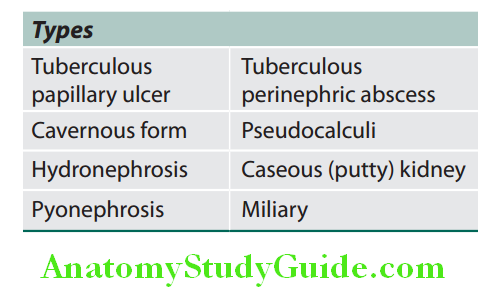
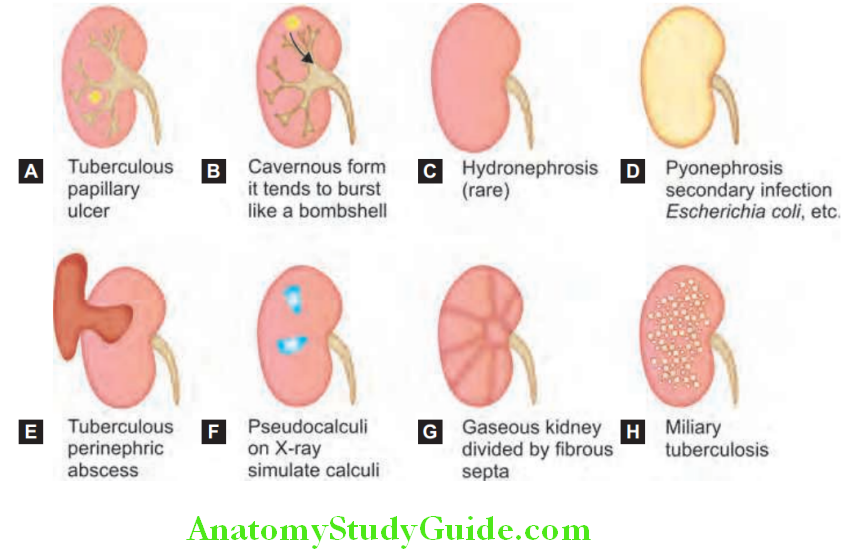
Tuberculosis Of The Urinary Tract Etiology and Pathology:
- Tuberculosis usually involves one kidney. Rarely, tuberculosis may be bilateral as part of the generalized process of miliary tuberculosis.
- Tuberculous granulomas in the region of renal pyramid may coalesce to form an ulcer and discharge mycobacteria and pus cells into the urine.
- Untreated lesions of renal tuberculosis may enlarge and form tuberculous abscess in the parenchyma.
- Fibrosis at the necks of the calyces and the renal pelvis may cause tuberculous pyonephrosis.
- Extension of tuberculous pyonephrosis or renal abscess may result in perinephric abscess and the kidney may be progressively replaced by caseous material (putty kidney). This may become calcified (cement kidney).
- Renal tuberculosis is often followed by infection of the ureters and bladder leading to ureteral stricture and contraction of bladder.
- Rarely, cold abscesses may form in the loin. In male, tuberculous epididymo-orchitis may develop even without any lesion in bladder.
Tuberculosis Of The Urinary Tract Clinical Features:
- Usually occurs between 20 and 40 years of age
- Affects males more commonly than females (2:1)
- Affects right kidney more commonly than the left
Tuberculosis Of The Urinary Tract Symptoms:
These include a dull ache in the renal angle, increased frequency of micturition, dysuria, and constitutional symptoms such as fever (evening rise of temperature) malaise and weight loss. Tuberculous cystitis present with hematuria and painful micturition.
- Physical examination: Shows an enlarged palpable kidney and tenderness in the renal angle.
Tuberculosis Of The Urinary Tract Investigation:
- Urine examination:
- Microscopic examination: Sterile pyuria is characterized by urine containing pus cells, but fails to grow any organism on routine culture. This is the characteristic feature.
- Urine examination for tubercle bacilli: Five early morning consecutive samples of urine should be examined (because there is usually intermittent excretion of tubercle bacilli). Alternatively, a 24-hour urine collection can be examined for acid-fast bacillus (AFB).
- Culture of the urine: Diagnosis of active tuberculous infection depends on culture of mycobacteria from earlymorning urine samples.
- Radiography:
- Plain KUB radiograph: It may show calcification (pseudocalculi) in the renal parenchyma and ureter.
- Chest X-ray: Should be done in all cases to exclude active or previous evidence of pulmonary tuberculosis.
- IVU: May reveal irregular contour of the kidney, hydrocalyx, cold abscess, displacement of adjacent calyces, and small contracted bladder (systolic or thimble bladder).
- Ultrasonography: May show dilated calyces, hydronephrosis, small abscesses, and areas of calcification.
- Excretion urography: May show cavitating lesions in the renal papillary areas with calcification. May also show ureteral obstruction with hydronephrosis.
- Cystoscopy: May show a characteristic “golf hole” appearance of the ureteric orifice due to sclerosing periureteritis.
Tuberculosis Of The Urinary Tract Treatment:
- Antituberculous chemotherapy: Similar to that for pulmonary tuberculosis. Renal ultrasonography or excretion urography should be done 2–3 months after initiation of treatment as ureteric strictures may first develop in the healing phase.
- Surgical treatment may be required in selected cases.
Renal Replacement Therapies:
Question 43. Write short essay/note on renal replacement therapy.
Answer:
Renal Replacement Therapies Requirement:
Renal replacement therapy (RRT) may be required on a temporary measure in patients with AKI or on a permanent measure for CKD.
Main options of renal replacement therapy:
- Peritoneal dialysis
- Intermittent hemodialysis (HD) combined with ultrafiltration, if necessary
- Intermittent hemofiltration
- Continuous arteriovenous or venovenous hemoiltration
- Hemodiafiltration
- Renal transplantation.
Aim of renal replacement techniques:
- To replace all the excretory functions of the normal kidney namely excretion of nitrogenous wastes
- Maintenance of:
- Plasma biochemistry
- Normal electrolyte concentrations
- Normal extracellular volume (fluid balance)
Options of renal replacement therapy:
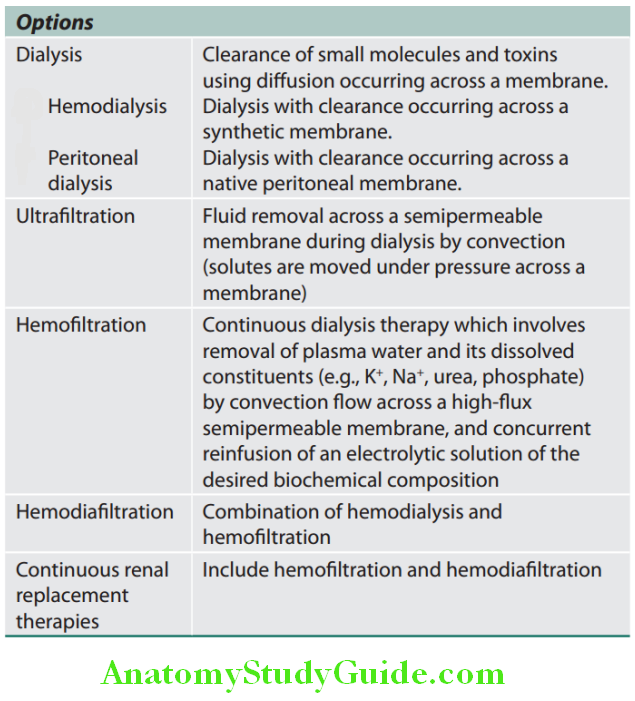
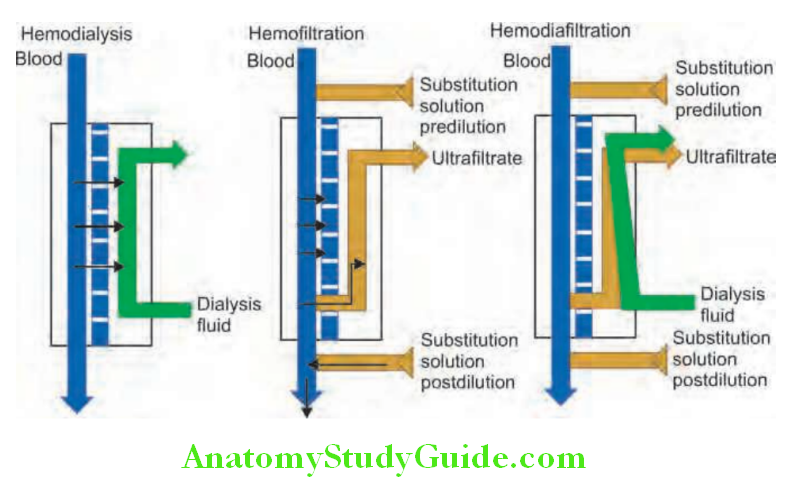
Hemodialysis:
Question 44. Write short note on hemodialysis.
(or)
Describe the types and indications for dialysis. Enumerate complications of hemodialysis.
Answer:
Hemodialysis is the most common form of RRT used in ESRD and also in AKI.
Hemodialysis Basic Principles:
In hemodialysis, blood from the patient is pumped through an array of semipermeable membranes (the dialyzer, often called an “artificial kidney”).
This brings the blood into close contact with dialysate (dialysis fluid) flowing countercurrent to the blood on the other side of membrane.
This allows accumulated uremic toxins (e.g., urea and creatinine) and electrolytes (e.g., potassium) to diffuse across a semipermeable membrane from the blood (where they are in high concentrations), to the dialysis fluid on the other side (where they are in low concentrations). This in turn results in changes in the plasma biochemistry toward that of the dialysate due to the diffusion of molecules down their concentration gradients.
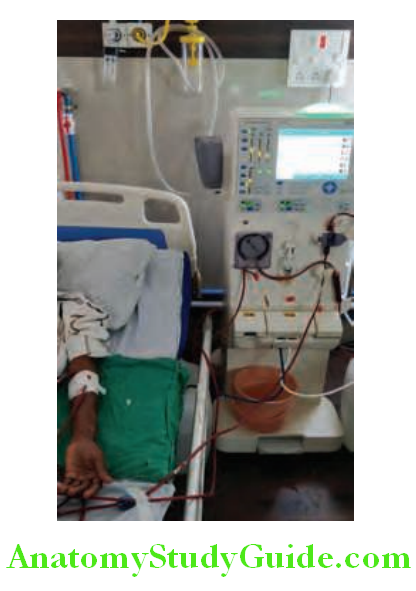
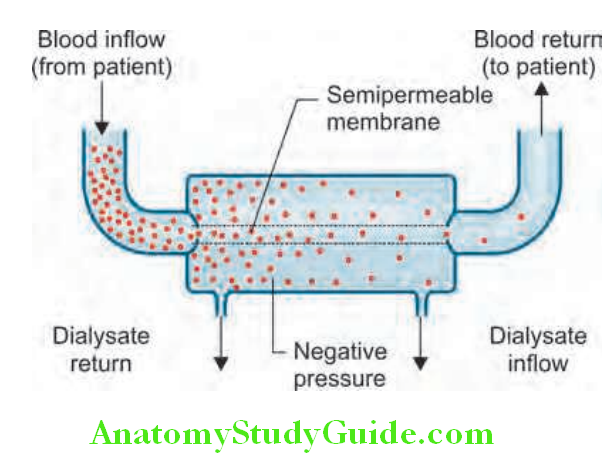
Hemodialysis Access for Hemodialysis:
Usually for adequate dialysis, required blood flow is 200–300 mL/minute and the dialysate flow is 500 mL/minute. Hemodialysis involves gaining access to the circulation through.
Arteriovenous fistula: For long-term dialysis access to the bloodstream is achieved by surgical construction of an arteriovenous fistula, usually in the forearm using the radial or brachial artery and the cephalic vein.
Arteriovenous shunt: In patients with poor-quality veins or arterial disease (e.g., diabetes mellitus) access is obtained by interposing a piece of synthetic material [polytetrafluoroethylene (PTFE) grafts] between native artery and native vein.
Central venous catheter: If immediate dialysis is required, a large bore double lumen cannula may be inserted into a central vein (subclavian, jugular or femoral). Each session of hemodialysis usually lasts for 4–5 hours. Two or three sittings/week may be required. Indications of RRT discussed under AKI.
Types of hemodialysis are depicted:

Complications:
Question 45. Write short essay/note on complications of hemodialysis.
Answer:
- Hypotension during dialysis:
- It is the major complication and its contributing factors include:
- Excessive removal of extracellular fluid and hypervolemia,
- Shift of fluid into intracellular compartment (due to rapid reduction in urea in the extracellular compartment) and inadequate “refilling” of the blood compartment from the interstitial compartment,
- Abnormalities of venous tone
- Autonomic neuropathy
- Acetate Toxicity That Causes Vasodilatation
- Left ventricular failure.
- Other complications:
- Cardiac arrhythmias due to potassium and acid-base shifts
- Hemorrhage due to anticoagulants and venous needle discontinuation
- Air embolism due to disconnected or defect lines and equipment malfunction
- Anaphylactic reactions to the dialyzer may rarely occur to ethylene oxide (used to sterilize most dialyzers) or in patients receiving ACE inhibitors (with polyacrylonitrile dialyzers).
- Hard-water syndrome due to failure to soften water resulting in a high calcium concentration prior to mixing with dialysate concentrate.
- Hemolytic reactions
- Infections usually occur with vascular access devices (catheter or fistula).
- Pulmonary edema due to fluid overload. Other features include hypothermia, platelet consumption, and electrolyte disturbances.
- Dialysis disequilibrium syndrome: May develop following a dialysis session. It is characterized by nausea, vomiting, restlessness, headache, hypertension, myoclonic jerks, and in severe case, seizures and coma. This is because of rapid changes in plasma osmolality leading to cerebral edema.
- Dialysis dementia: May develop in patients on long-term hemodialysis. It is characterized by speech dyspraxia, myoclonic jerks, dementia, seizures and later, death. It is mainly due to aluminum toxicity and some may be due to viral infection.
The rate of death from cardiac disease is higher in patients on hemodialysis when compared to patients on peritoneal dialysis or after renal transplantation.
Acute Dialysis in Critically Ill Patients:
Continuous Renal Replacement Therapy:
Hemodialysis Advantages:
- It is used in critically ill patients who cannot tolerate large fluid shifts and hypotension that frequently occurs during standard hemodialysis.
- It allows slow and isotonic fluid removal. Thus produces excellent hemodynamic tolerance, even in patients with shock or severe fluid overload.
- Since dialysis is continuous, volume removal and correction of metabolic abnormalities can be modified at any time. This allows rapid adjustment in critically ill patients.
Continuous renal replacement therapy modalities:
The most common is continuous venovenous hemodiafiltration. This combines convective and diffusive clearance through a dialyzer with reinfusion of electrolyte-rich solutions.
Hemodialysis Indications:
Continuous renal replacement therapy (CRRT) is of choice in patients with ARF combined with hemodynamic instability, cerebral edema, severe fluid overload, and encephalopathy.
Hemodialysis Complications:
Embolization, arteriovenous fitula formation, hemorrhage, and infection from catheter access.
Peritoneal Dialysis:
Question 46. Write a short note on peritoneal dialysis and its indications.
Answer:
In peritoneal dialysis, the peritoneal membrane of the patient acts as a semipermeable membrane. Through this diffusion of water and solutes takes place and this avoids the need of extracorporeal circulation of blood.
Very simple, low-technology treatment when compared to hemodialysis.
Peritoneal Dialysis Principle: Solutes diffuse from blood across the peritoneal membrane to peritoneal dialysis fluid down a concentration gradient and water diffuses through osmosis.
Indications for peritoneal dialysis:
- Preferred mode of dialysis for infants and young children
- Patients with severe hemodynamic instability on hemodialysis
- Patients with difficult vascular access
Peritoneal Dialysis Procedure:
- A plastic or silicone tube/catheter is placed into the peritoneal cavity through the anterior abdominal wall
- Dialysate is instilled into the peritoneal cavity, usually under gravity.
- Urea, creatinine, phosphate, and other uremic toxins pass across the peritoneal membrane into the dialysate because of their concentration gradients.
- Water (with solutes) is attracted into the peritoneal cavity by osmosis.
- The fluid is removed by gravity after 30–60 minutes, and the fluid is changed regularly to repeat the process several times. This is called as intermittent peritoneal dialysis and is mainly used in ARF.
When chronic peritoneal dialysis required, a soft catheter is inserted, with its tip in the pelvis with an exit site in the lateral abdominal wall.
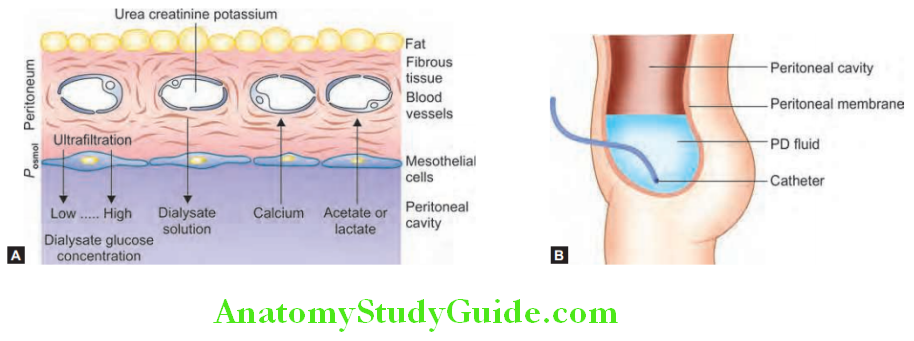
Peritoneal Dialysis Complications:
- Technical complications: Catheter-related exit site infection, tunnel infection, peritonitis, pericatheter leakage, poor drainage, catheter migration rarely bowel perforation.
- Raised intra-abdominal pressure: Hernias, fluid leaks, rectal prolapse, vaginal prolapse, low back pain, scrotal edema, and decrease in appetite, sclerosing encapsulating peritonitis.
Forms of Peritoneal Dialysis:
- Continuous ambulatory peritoneal dialysis:
- In this type of peritoneal dialysis, dialysate is present within the peritoneal cavity continuously, except when dialysate is being exchanged.
- Dialysate exchanges are performed 3–5 times a day.
- About 1.5–3 L bags of dialysate are infused into the peritoneal catheter using a sterile no-touch technique.
- Each exchange takes 20–40 minutes.
- This technique is useful for maintenance of peritoneal dialysis in patients with end-stage renal failure (CRF).
- Nightly intermittent peritoneal dialysis (NIPD):
- In this type of peritoneal dialysis, an automated device is used to perform exchanges each night (while the patient is asleep).
- Apart from that night, sometimes dialysate is left in the peritoneal cavity during the day, to increase the time during which biochemical exchange is occurring.
- Tidal dialysis: In this type of peritoneal dialysis, a residual volume of dialysate is left within the peritoneal cavity with continuous cycling of smaller volumes in and out.
Differences between hemodialysis and peritoneal dialysis are presented:
Differences between hemodialysis and peritoneal dialysis:
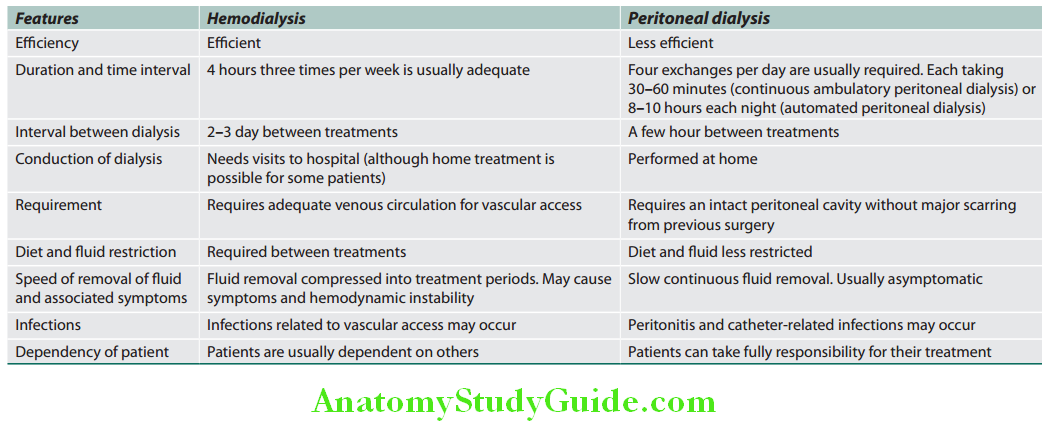
Renal Transplantation:
Question 47. Write a short essay/note on renal transplantation.
Answer:
Successful renal transplantation offers the best chance of long-term survival with almost complete rehabilitation in ESRD.
Renal Transplantation Advantages:
- Treatment of choice for most patients with advanced (end-stage) renal failure.
- Method of RRT having significant survival advantage when compared to dialysis patients.
- Freedom from dietary and fluid restriction
- Corrects anemia and infertility in CRF patients
- Reduces the need for parathyroidectomy Donor is usually a living close relative or a cadaveric donor.
Factors Determining the Success:
- Donor: Living donors should be healthy and should be free of hypertension, diabetes, or malignant disease. With the acceptance of the concept of brain death in India, cadaveric transplants are being also performed at present.
- ABO (blood group) compatibility between donor and recipient is required.
- Matching donor and recipient for HLA type:
- Among the 6 HLA types in each person (A/B/C/DP/DQ/DR), three HLA types are matched (HLA DR, HLA-B, HLA-A)
- It is preferable to have a donor with a HLA-identical that of the recipient. Matching for HLA-DR antigens (class II) appears to be most important for graft survival. Matching at the HLA-B locus (class I) has only a minor effect on graft outcomes.
- Complete compatibility at HLA-A, HLA-B, and HLA-DR offers the best chance of long survival of the graft, followed by a single HLA mismatch (i.e., antigen possessed by the donor and not possessed by the recipient).
- Class I antigens (HLA-A, B, and C) are detected by a lymphocyte assay and class II (HLA-DR) by mixed lymphocyte culture.
- Cross-matching: Presensitization (i.e., presence of antibodies against donor ABO blood group and HLA class I antigens) is detected by a cross-matching. Transplantation is contraindicated if it is positive.
Renal Transplantation Contraindications:
Renal Transplantation Technique:
- Before transplantation, the recipient receives a hemodialysis to ensure a relatively normal metabolic state.
- In transplantation, donor human kidney (either from a cadaveric donor or from a living close relative) is placed in an extraperitoneal pouch in the iliac fossa of the recipient.
- The renal artery and vein of donor kidney are anastomosed to the recipient’s iliac vessels. The donor ureter is placed into the recipient’s bladder. The recipient’s original kidneys are left undisturbed.
- Eighty percent of grafts now survive for 5–10 years in the best centers, and 50% for 10–30 years.
Absolute and relative contraindications for renal transplantation:
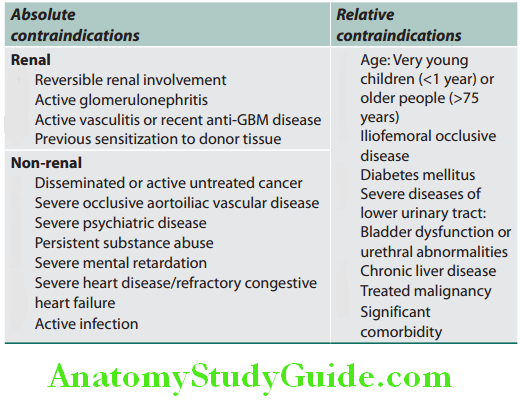
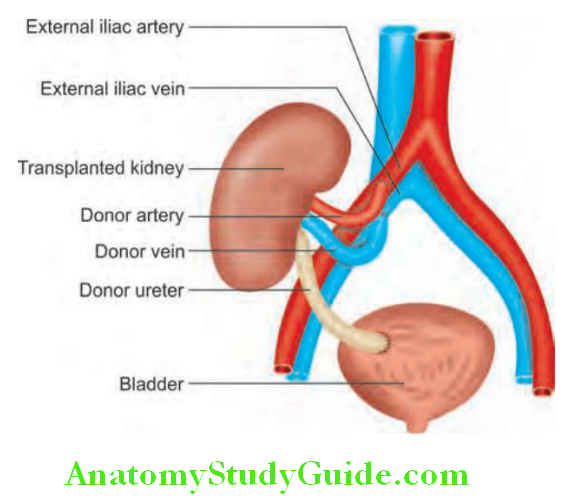
Immunosuppressive therapy:
- Unless the donor is genetically identical (i.e., an identical twin), immunosuppressive treatment is required to prevent rejection.
- Immunosuppressive therapy varies from center to center and combination of drugs is often used.
- Commonly used drugs include corticosteroids, antiproliferative agents (e.g., azathioprine, mycophenolate mofetil), calcineurin inhibitor (e.g., ciclosporin, tacrolimus, sirolimus), and antibodies [polyclonal antithymocytic globulin (ATG), antilymphocytic globulin (ALG), and monoclonal (OKT3) antibodies]. Newer drugs include basiliximab and daclizumab (antibodies against interleukin-2 receptor).
- Immunosuppressive agents can be classified as:
- Induction immunosuppression:
- Depleting agents:
- Anti Cd-52: Alemtuzumab
- OKT3: Anti-CD3 monoclonal antibodies
- Rabbit ATG
- Nondepleting agents:
- Anti-CD-25: Basiliximab, daclizumab
- Maintenance immunosuppression:
- Calcineurin inhibitors: Cyclosporine, tacrolimus
- Mycophenolate mofetil
- Steroids
- mTOR inhibitors: Sirolimus
- Depleting agents:
Renal Transplantation Complications:
- Acute tubular necrosis: It is the most common cause of cadaveric graft dysfunction (up to 40–50%).
- Technical failures:
- These include:
- Occlusion or stenosis of the arterial anastomosis
- Occlusion of the venous anastomosis
- Urinary leaks owing to damage to the lower ureter, or defects in the anastomosis between ureter and recipient bladder.
- Transplantation rejection: In spite of prophylactic use of immune suppressants before or at the time of transplantation, most recipients undergo one or more type of rejection.
- Types of graft rejection:
- Hyperacute rejection:
- A special type of compliment-mediated rejection occurs if the host has preformed anti-donor antibodies (for example, anti-ABO blood type antibodies) in the circulation before transplantation. These antibodies bind to endothelium of graft organ, activates complement followed by vascular thrombosis.
- Occurs within minutes. The transplanted kidney must be removed immediately to prevent a severe systemic inflammatory response and generalized clotting.
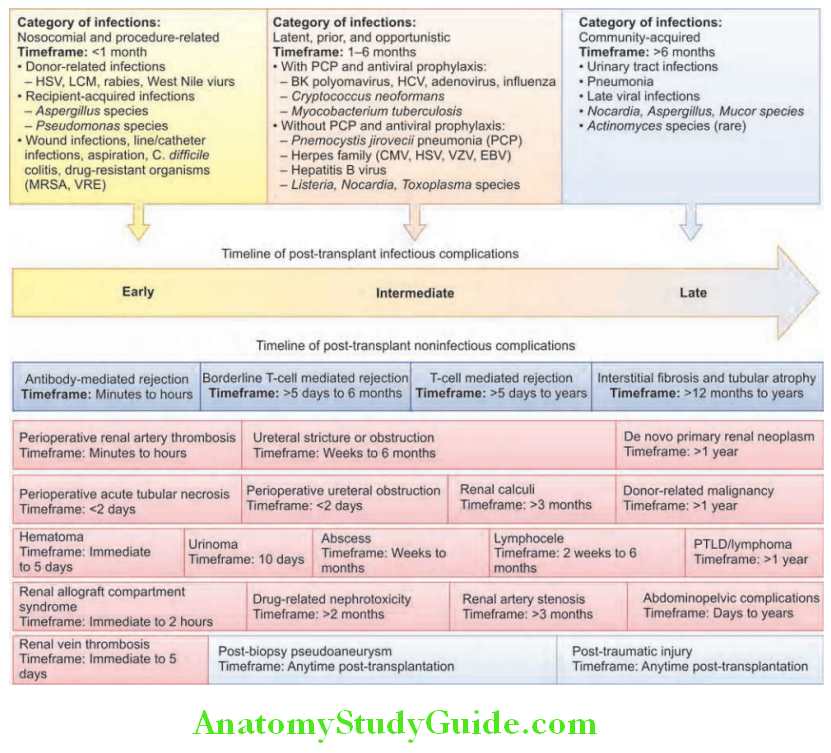
- Acute rejection:
- Time of occurrence: Within days to weeks after transplantation in the nonimmunosuppressed host.
- Type: Either cellular or humoral immune mechanisms may predominate.
- Acute cellular rejection: Occurs within few months after transplantation and develops renal failure.
- Acute humoral rejection (rejection vasculitis): Main target of the antibodies is the graft vasculature → manifest as vasculitis. Caused by anti-donor antibodies.
- Features: Acute rejection is characterized by deterioration of renal function, hypertension, weight gain, tenderness and swelling of the graft, fever and appearance of protein, lymphocytes, and renal tubular cells in the urine sediment.
- If the diagnosis is not clear, percutaneous needle biopsy is performed for histopathological examination.
- Chronic rejection: Few patients may develop irreversible chronic graft rejection.
- Other complications:
- Infection is the most important cause of morbidity and mortality. Risk of infections can be divided into three distinct time periods.
Renal Transplantation Treatment:
- Use additional immunosuppressant including intravenous pulses of methylprednisolone and antilymphocyte antibody.
- Antibody-mediated rejection is less likely to respond to corticosteroids and may be treated with plasma exchange (to remove the antibody) and intravenous immunoglobulin
1. First month post-transplant:
These include:
- Infections present in the recipient before transplant and aggravated by immunosuppressive therapy (e.g., tuberculosis, systemic fungal infections, hepatitis B or C, HIV or smouldering bacterial infections);
- Infections transmitted through contaminated allograft; and
- Routine postsurgical bacterial infections of the wound, IV lines, and urine catheters.
2. 1–6 months post-transplant: These include:
- Viral infections (e.g., cytomegalovirus, Epstein–Barr virus, hepatitis viruses, and HIV)
- Superinfection with opportunistic bacteria or fungi (e.g., Pneumocystis carinii, Listeria monocytogenes, and Aspergillus).
More than 6 months post-transplant:
These include:
- Chronic progressive disease due to viral infections acquired earlier [e.g., progressive cytomegalovirus (CMV) chorioretinitis, progressive liver disease due to hepatitis B or C virus, and EBV-associated lymphoproliferative disorders];
- Opportunistic infections with pathogens such as Pneumocystis carinii, Cryptococcus, Listeria monocytogenes, and Nocardia asteroides in patients with chronic graft dysfunction who have received multiple courses of antirejection therapy; and
- Infections similar to those in the community.
- Post-transplantation lymphoproliferative disorders: Epstein–Barr virus-associated malignancies are common in those who received biological agents and in children.
- Malignancy: Immunosuppressive therapy is associated with increases the risk of skin tumors (e.g., basal and squamous cell carcinoma). Incidence is 5–6% in Western countries and lower (2%) in India.
- Cardiovascular disease is responsible for deaths in 20–30% of patients, especially the elderly and diabetics.
- Post-transplant hypertension develops in up to 80% of patients early in the postoperative course and in 50% of stable recipients on maintenance immunosuppression.
- Post-transplant osteoporosis is common due to treatment with steroids.
- Recurrent and de novo renal disease is common (e.g., primary FSGS, type II membranoproliferative glomerulonephritis, diabetic nephropathy and IgA nephropathy, hyperoxaluria).
- Vascular: Renal artery thrombosis and renal artery stenosis.
- Urologic: Ischemic necrosis of the lower end of the ureter may produce urinary leak, calyceal-cutaneous fistula. Vesicoureteric reflux into the graft may lead to increased frequency of UTIs.
Renal Biopsy:
Question 48. Write short note on renal biopsy and its indications.
Answer:
It is used to establish the nature and extent of renal disease. Biopsy is performed transcutaneously with ultrasound guidance. Biopsy material is examined under light microscopy, electron microscopy and by immunofluorescence. Indications for renal biopsy are presented in Table 13.37 and contraindications and complications of renal biopsy are presented.
Renal Biopsy Treatment:
Use additional immunosuppressant including intravenous pulses of methylprednisolone and antilymphocyte antibody.
Antibody-mediated rejection is less likely to respond to corticosteroids and may be treated with plasma exchange (to remove the antibody) and intravenous immunoglobulin.
Indications for renal biopsy:

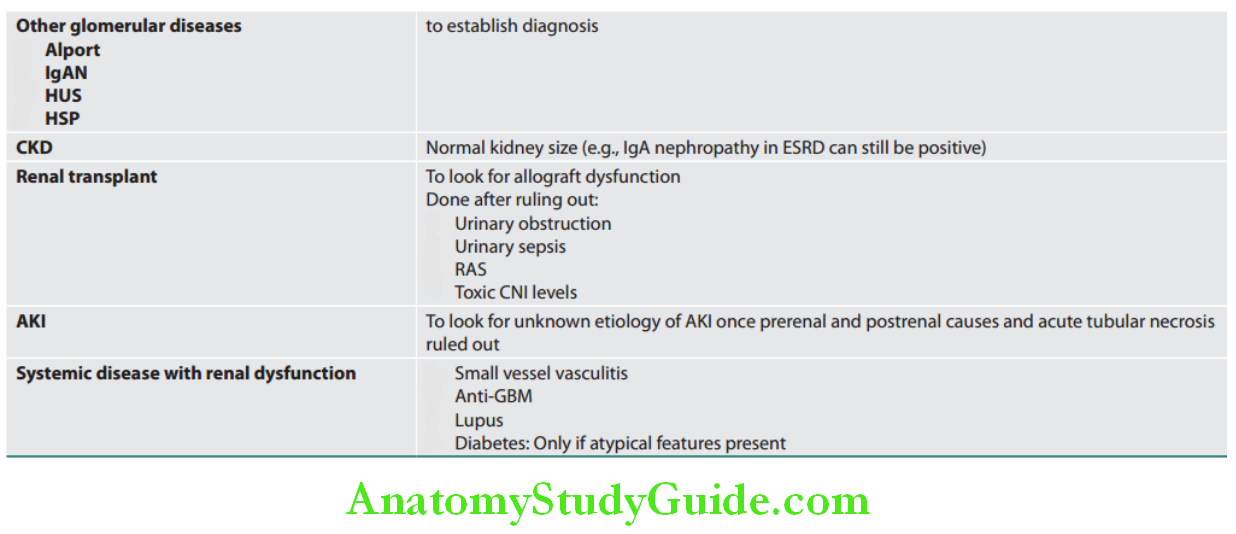
Contraindications and complications of renal biopsy:

Leave a Reply