Infectious Diseases Covid-19
Question 1. Discuss the epidemiology, clinical features, complications, and management of COVID-19.
Answer:
At the end of 2019, a novel coronavirus was identified as the cause of a cluster of pneumonia cases in Wuhan, China. The WHO designated the disease COVID-19 and the virus is designated severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
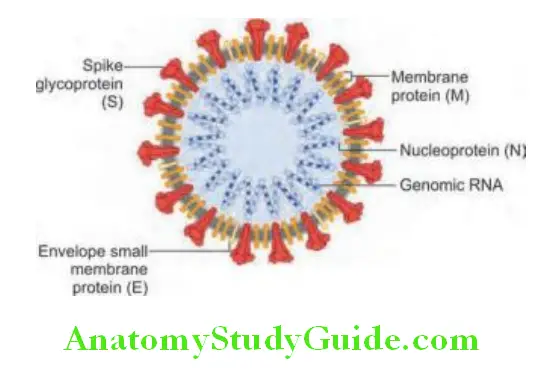
Read And Learn More: General Medicine Question And Answers
COVID-19 Transmission:
Person-to-person:
- Mainly via respiratory droplets
- Direct contact with mucous membrane (eyes/nose/mouth)
Viral shedding and period of infectivity:
- It can be transmitted prior to the onset of symptoms and throughout the course of illness, particularly early in the course.
- Transmission can occur during incubation period (from asymptomatic individuals)
- Duration of viral shedding is variable
Risk of transmission:
Depends on:
- Type and duration of exposure
- Use of preventive measures
- Amount of virus in respiratory secretions
- Environmental contamination—it is a potential source, especially in hospitals. Period of infectivity is ~6 days
- Risk of reinfection: Some antibodies are protective; not definitely established yet.
COVID-19 Clinical Features:
- Incubation period: <14 days
- Majority are asymptomatic
- Spectrum of disease:
- Mild—no/mild pneumonia
- Severe—>50% lung involvement, dyspnea, and hypoxia seen
- Critical—respiratory failure, shock, and multiple organ dysfunction syndrome (MODS)
- Death
- Most common manifestation/pneumonia—fever, cough, dyspnea, and bilateral lung infiltrates on imaging.

- Fever
- Dry/wet cough
- Dyspnea (new or worsening)
- Anosmia or other smell abnormalities
- Ageusia or other taste abnormalities
- Sore throat
- Myalgia
- Conjunctivitis
- Fatigue
- Confusion
- Chest pain
- Nausea/vomiting
- Diarrhea
- Chills/rigor
- Headache
- Rhinorrhea
- Tachypnea
- Hypoxia
COVID-19 Complications:
- Acute respiratory distress syndrome (ARDS)
- Cardiac arrhythmias
- Acute cardiac injury
- Shock
- Guillain–Barré syndrome
- Deep venous thrombosis (DVT) and pulmonary embolism
- Acute stroke
- Children—kawasaki disease and toxic shock syndrome. Multisystem inflammatory syndrome in children (MIS-C)
- Cytokine release syndrome
- Secondary infections—bacteremia, aspergillus, mucormycosis
COVID-19 Risk Factors for Severe Illness:
Laboratories:
- Lymphopenia with N/L >5
- CRP >100
- Ferritin >300
- LDH >245
- D-dimer >1,000 ng/mL
- High AST/ALT
- High PT/INR
- High trop-T
- AKI
- Increased CPK
Comorbidities:
- Age >50 years
- DM
- HTN
- Lung disease (COPD, BA, post-TB, and sequelae)
- CKD
- CLD
- HIV
- Malignancy (hematologic and solid organ tumors)
- Immunosuppression
- Obesity
Clinically:
- Hypoxia—SpO2 <94%
- HR >100 bpm
- RR >30 cpm
- SBP <90 mm Hg
- Altered mental status
Imaging:
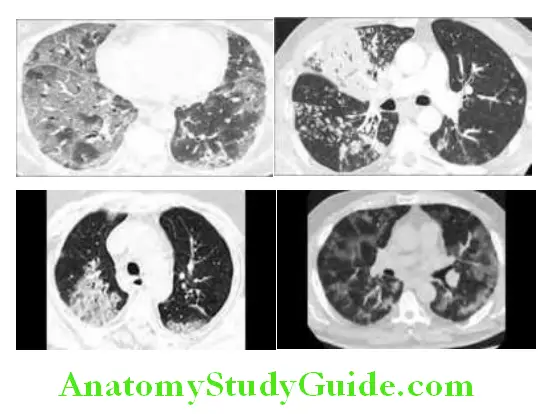
Chest X-ray: Normal in early/mild disease. Consolidation and ground glass opacities (GGO) in bilateral peripheral and lower lung zone distributions.
CT chest:
- GGO: 83% cases
- GGO with mixed consolidation
- Pleural thickening
- Interlobular septal thickening
- Air bronchograms
Less common findings:
- Bronchiectasis
- Pleural effusions
- Pericardial effusion
- Lymphadenopathy
- Crazy pavement pattern (GGO with superimposed septal thickening)
Chest CT Severity Score:
20 lung regions evaluated on chest CT using a system attributing scores of 0, 1, and 2 if parenchymal opacification involved 0%, less than 50%, or equal or more than 50% of each region. The CT-SS is defined as the sum of the individual scored in the 20 lung segment regions, which may range from 0 to 40 points.
Chest CT Severity Score Diagnosis:
- Clinical suspicion:
- New-onset fever/cough/dyspnea
- History of contact with a COVID positive case/suspect within last 14 days
- History of travel within last 14 days
- History of work in healthcare settings
- History of comorbidities/risk for severe illness
- RT-PCR:
- Preferred diagnostic test for COVID-19
- Specimens—nasopharyngeal swab/nasal swab from both anterior nares/nasal or nasopharyngeal wash or aspirate/oropharyngeal swab
- RT-PCR positive for SARS-CoV-2 indicates COVID-19 disease present
- Detectable SARS-CoV in upper respiratory tract specimens may persist for weeks after symptom onset
- Prolonged viral detection does not indicate ongoing infectiousness
- Single negative RT-PCR is sufficient to exclude COVID-19 in many individuals.
- False-negative RT-PCR has been well-documented. If clinical suspicion remains, repeat test is performed 24–48 hours after initial testing.
- Lower respiratory tract specimens (expectorated sputum, tracheal aspirate, and BAL fluid) for RT-PCR may be reserved for hospitalized patients. Induced sputum is not recommended for testing.
- Serology—Detects SARS-CoV-2 antigens nucleocapsid or spike protein. Uncertain sensitivity and specificity.
- Imaging—CXR/CT thorax, bilateral peripheral, and lower zone involvement
- ECG
Blood tests:
Basic Investigations:
- CBC
- ESR and CRP
- LFT
- RFT
- RBS
Others:
- D-dimer
- Ferritin
- LDH
- PT/INR
- Trop-T
- Procalcitonin
- ABG
Chest CT Severity Score Treatment:
- Influenza-like illness (ILI)—fever (>38°C) + cough with onset <10 days not requiring admission.
- Severe acute respiratory infection (SARI)—fever (>38°C) with cough with onset <10 days requiring admission.
General:
- No dietary restriction
- Good hydration
- RBS <180
- Continue ACE/ARBs, if present
- Avoid NASIDs other than PCT
- Avoid nebulization (aerosolization of the virus)
- Oxygen supplementation Hudson’s/venturi/HFNC/NIV/mechanical ventilation
Category A (ICMR):
Asymptomatic/mild symptoms:
- Tablet oseltamivir 75 mg 1-0-1 × 5 days
- Tablet azithromycin 500 mg 0-1-0 × 5 days
- Tablet HCQ 400 mg stat → 100 mg 1-0-1 × 4 days
- Tablet zinc 50 mg 0-1-0 × 7 days
- Tablet vitamin C 500 mg 1-1-1 × 7 days
- Injection enoxaparin 40 mg SC OD × 7 days (if D-dimer increased or CXR/CT suggestive of GGO)
Category B (ICMR):
Symptomatic with mild-moderate pneumonia with no signs of severe disease, RR 15–30 cpm, SpO2 90–94% at room air:
- Category A treatment PLU
- IV antibiotics if indicated
- Tablet N-acetylcysteine 600 mg 1-1-1 if cough+
- Injection dexamethasone 6 mg 1-0-0 × 10 days or till discharge, whichever is shorter
- Continuous SpO2 monitoring
- Oxygen via nasal prongs or Hudson’s mask
Category C (ICMR):
- Symptomatic with severe pneumonia, RR > 30 CPM, SpO2 <90% at room air or 90–94% with O2, ARDS, and septic shock.
- Category A and B treatment PLUS
- If septic shock: Injection Sepsivac 0.3 mL I/D OD × days
- NIV/MV
- Inotropes, if required
- Novel therapy as per clinical discretion: Discussed below
Drugs Approved for the Treatment of COVID-19:
Remdesivir:
- Acts against RNA-dependent RNA polymerase of SARS-CoV-2
- Indicated in those on supplemental oxygen/mechanical
ventilation/ECMO - Contraindicated in deranged LFT, GFR <30 mL/min
- Should not be used concurrently with HCQ
- Pregnancy is not a contraindication
IL-6 pathway inhibitors—tocilizumab, sarilumab, and siltuximab:
- Observational data revealed decreased risks of intubation and/or death
- Associated with an increased risk of secondary infections
Hydroxychloroquine/chloroquine:
- Was recommended initially, but FDA revoked emergency use authorization in June 2020, due to increased arrhythmias
- Contraindications for HCQ:
- QTc >500
- Porphyria
- Myasthenia gravis
- Retinal pathology
- Epilepsy
- Hypokalemia <3
- Pregnancy is not a contraindication.
Convalescent plasma:
- High neutralizing antibody titers is hypothesized to have clinical benefit when given early in the course of disease, however recent studies have not shown significant benefit
Lopinavir/ritonavir:
- Though recommended initially, clinical trials have failed to demonstrate efficacy
Favipiravir:
- An RNA polymerase inhibitor
- Used in mild disease
- Dosage of favipiravir for adults is 1800 mg orally twice daily on 1st day followed by 800 mg orally twice daily, up to maximum of 14 days.
Ivermectin:
- In vitro activity against SARS-CoV-2, various clinical trials of ivermectin are underway
Bamlanivimab:
- Recombinant neutralizing human IgG1-kappa monoclonal antibody directed against the spike (S) surface protein of SARS-CoV-2, derived from antigen specific B cells of a convalescent COVID-19 patient
- Binds to the receptor binding domain (RBD) of the S protein at a position overlapping the ACE-2 binding site
- Emergency use authorization (EUA) on 9th November 2020 for mild/moderate COVID-19 disease
Casirivimab and imdevimab:
- Casirivimab IgG1-kappa and imdevimab IgG1-lambda, recombinant human IgG1 monoclonal antibodies that bind simultaneously to the receptor binding domain of SARS CoV-2’s spike protein, and thereby preventing the entry of virus into the host cells.
- EUA by FDA on November 21st 2020. Approved for treatment of mild-to-moderate COVID-19 in adults
Baricitinib:
- Janus kinase inhibitor used as a treatment for adults with moderate to severe rheumatoid arthritis
Foralumab:
- Anti CD3 monoclonal antibody which induces T regulatory cells, resulting in IL-10 mediated antiinflammatory effect
AZD7442:
- It is a combination of two long-acting antibodies (LAABs) derived from convalescent patients after SARS-CoV-2 infection? The LAABs were optimized with half-life extension and reduced Fc receptor binding
- Other agents that have been proposed for COVID-19 therapy include the HCV
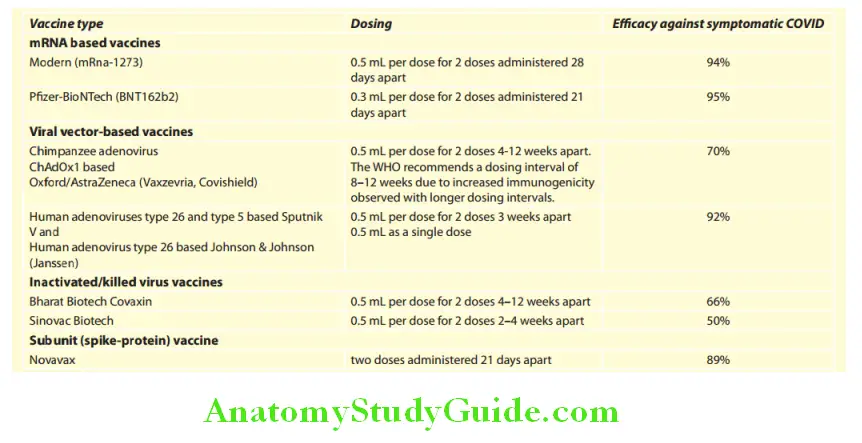
COVID-19 vaccines:
- The aim of the COVID-19 vaccine is to induce a protective immunity to, prevent symptomatic infection and further to prevent severe illness, hospitalization, and death.
Pyrexia (Fever) Of Unknown Origin:
Question 2. Define pyrexia of unknown origin (PUO). Discuss briefly your approach to a case of PUO or fever of unknown origin
(FUO).
(Or)
Describe types, investigation, and differential diagnosis of a case of pyrexia of unknown origin.
(Or)
Mention the etiology of pyrexia of unknown origin and discuss the investigation in a case of pyrexia of unknown origin.
Answer:
Pyrexia (Fever) Of Unknown Origin Defiition:
The definition of FUO (classical) was given by Petersdorf and Beeson in 196According to new classification, PUO is divided into four types:
Classic pyrexia of unknown origin (PUO) is classically defined as:
- Duration: Fever of at least 3 weeks duration.
- Temperature: Daily temperature persistently elevated above 101°F (38.3°C).
- Remains undiagnosed despite after a thorough history-taking, physical examination, and the following obligatory
(intelligent and intensive) investigations (or at least three outpatient visits or 3 days in hospital). - No known immunocompromised state (e.g., HIV or other immunosuppressing conditions).
Nosocomial PUO:
- Temperature: Daily temperature persistently elevated above 101°F (38.3°C) developing on several occasions.
- In a hospitalized patient (>24 hours) who is receiving acute care but no fever or incubating on admission.
- It is mandatory that the cause of fever is not found at least on 3 days of intelligent investigations, including at least 2 days incubation of cultures.
Neutropenic PUO:
- A temperature of >38.3°C (101°F) developing on several occasions
- Neutrophil count is below 500/mL or is expected to fall to that level in 1 or 2 days
- It is mandatory that the cause of fever is not found at least on 3 days of intelligent investigations, including at least 2 days incubation of cultures.
HIV-associated PUO:
- A temperature of more than 38.3°C (101°F) developing on several occasions
- Duration of fever is more than 4 weeks for outpatients or more than 3 days for hospitalized patients
- HIV infection confirmed
- It is mandatory that the cause of fever is not found at least on 3 days of intelligent investigations, including at least 2 days incubation of cultures.
Origin (Puo) Etiology:
Classic PUO is usually not due to a rare disease, but due to atypical presentation of common diseases.
Common causes of prolonged fever are:
Infections (40% Cases):
Most common infections causing PUO are tuberculosis, malaria, typhoid, and HIV. Other causes are:
1. Bacterial:
- Abscesses: Most commonly in the subphrenic space, liver, right lower quadrant, retroperitoneal space or the pelvis in women.
- Tuberculosis (TB): Disseminated tuberculosis occurring in immunocompromised patients, presents with more constitutional symptoms than localizing signs. Chest X-ray may be normal.
- Urinary tract infections (UTIs) are rare causes: Perinephric abscesses occasionally fail to communicate with the urinary system resulting in a normal urinalysis.
- Infective endocarditis: Culture-negative endocarditis occurs in 5–10% of endocarditis. The HACEK (H. parainfluenzae, H. aphrophilus, and H. paraphrophilus), Actinobacillus actinomycetemcomitans, Cardiobacterium hominis, Eikenella corrodens, and Kingella spp.) group are responsible for 5–10% cases of infective endocarditis and are the most common causes of gram-negative endocarditis in individuals without any abuse of intravenous drugs.
- Hepatobiliary infections: Cholangitis may develop without local signs and with only mildly elevated or normal liver function tests especially in the elderly patients.
Osteomyelitis:
- Brucellosis: Should be considered in patients with persistent fever and a history of contact with cattle, swine, goats or sheep, or patients who consume raw milk products.
- Borrelia recurrentis is responsible for tick borne relapsing fever.
- Other spirochetal diseases that can cause PUO include Spirillum minor (rat-bite fever), Borrelia burgdorferi (Lyme disease), and Treponema pallidum (syphilis).
2. Viral:
- Herpes viruses, such as Cytomegalovirus and Epstein–Barr virus (EBV) can cause prolonged febrile illnesses with constitutional symptoms without any significant organ manifestations, particularly in the elderly.
- HIV: Prolonged fever may be the only manifestation in patients with advanced HIV infection.
3. Fungi: Immunosuppression, the use of broad-spectrum antibiotics, the presence of intravascular devices and total parenteral nutrition all predispose individual to disseminated fungal infections.
4. Parasites:
- Malaria
- Toxoplasmosis: It should be considered in febrile patients with lymph node enlargement.
- Trypanosoma, leishmania, and amoeba species may rarely cause PUO.
5. Rickettsial organisms: Coxiella burnetii may cause chronic infections; chronic Q fever or Q fever endocarditis may be identified in patients with a PUO.
6. Psittacosis: Infection by the causative organism, Chlamydophila should be considered in a patient with PUO who has a history of contact with birds. Lepidopterism (fever with rash due to exposure to scales and toxic fluids of adult moths, butterflies or its caterpillars).
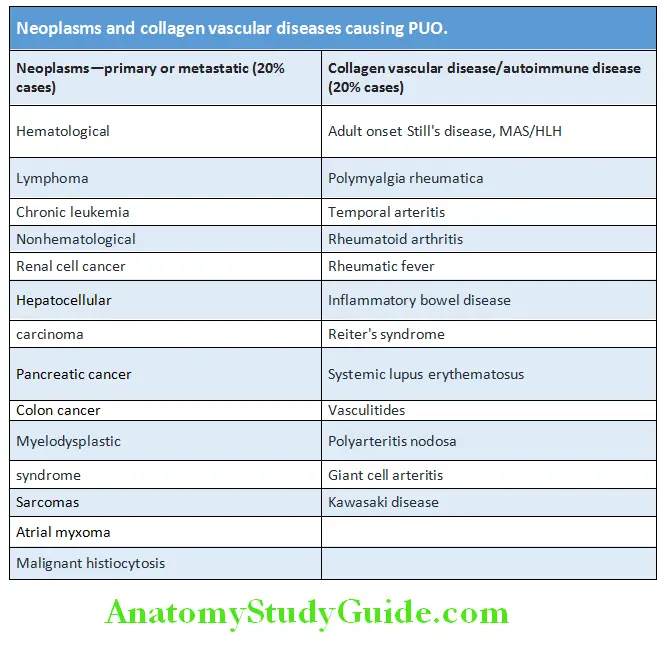
Neoplasms:
Primary or metastatic neoplasms constitute 20% of cases of PUO.
Collagen Vascular Disease/Autoimmune Disease:
Constitute 20% of cases of PUO.
Psychogenic Fevers:
- Habitual hyperthermia: It is seen in young females, characterized by temperatures of 99–100.5°F that occurs regularly or intermittently for years. No organic cause can be found.
- Afebrile PUO: In this, patient always complaints of feverishness but the temperature recorded is always <38.3oC.
- Exaggerated circadian rhythm: Normal person usually have an evening rise of temperature which is not normally apparent. In this condition, it becomes evident.
- Hysterical fever: In this, the patient is thinks and believes that he is always having fever.
- Malignant hyperthermia: It is a rare life-threatening condition triggered by exposure to certain drugs used for general anesthesia (especially all volatile anesthetics), nearly all gaseous anesthetics, and neuromuscular blocking agent like succinylcholine.
- Neuroleptic malignant syndrome (NMS): It is a rare, life-threatening, neurological disorder, most often caused by an adverse reaction to neuroleptic or antipsychotic drugs. It presents with muscle rigidity, fever, and autonomic instability.
Periodic Fevers:
For example, familial Mediterranean fever.
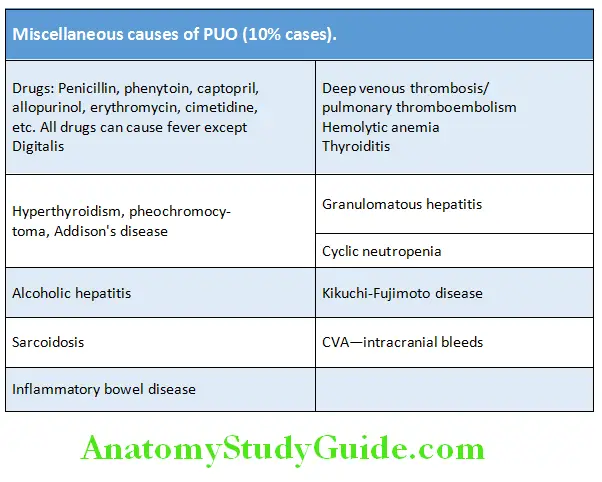
Miscellaneous:
Constitute 10% cases.
Undiagnosed (10% Cases):
About 10–15% of patients remain undiagnosed despite extensive investigations and in 75% of these the fever resolves spontaneously. In the remainder, other signs and symptoms make the diagnosis clear.
Pyrexia (Fever) Of Unknown Origin Clinical Approach to PUO:
History Taking:
1. Onset:
- Acute: Malaria and pyogenic infection
- Gradual: TB and typhoid fever
2. Character: High-grade fever is seen in urinary tract infections (UTI), malaria, thrombosis, and drug fever.
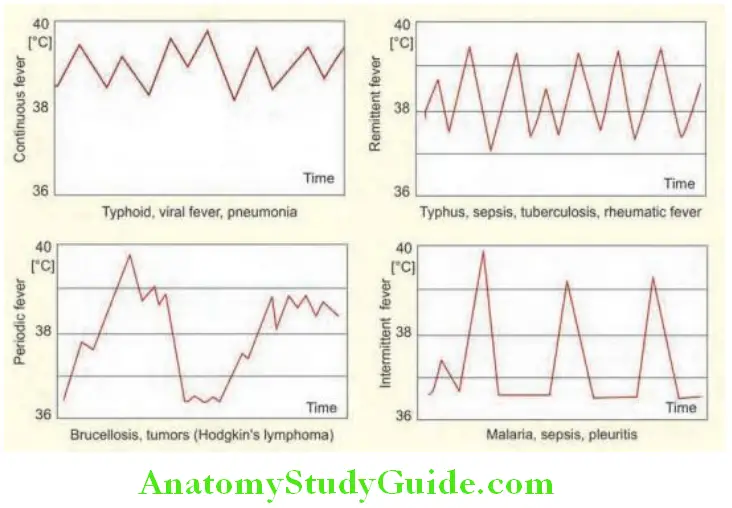
3. Pattern:
- Sustained/persistent: Typhoid fever and drugs
- Intermittent fever:
- Daily spikes: Malaria, abscess, tuberculosis (TB), and schistosomiasis
- Double quotidian, twice-daily spikes: Leishmaniasis, gonococcal endocarditis, and adult-onset Still’s disease
- Saddle back fever: Leptospirosis, dengue, and Borrelia
- Relapsing/recurrent fever: Nonfalciparum malaria, brucellosis, and Hodgkin’s lymphoma
4. Antecedents:
- Prior to onset of fever:
- Dental extraction: Infective endocarditis
- Urinary catheterization: UTI and bacteremia
5. Associated symptoms:
- Chills and rigors: Bacterial, rickettsial, and protozoal disease (malaria), influenza, lymphoma, leukemia, and druginduced
- Night sweats: TB and Hodgkin’s lymphoma
- Loss of weight: Malignancy and TB
- Cough and dyspnea: Miliary TB, multiple pulmonary emboli, AIDS patient with Pneumocystis pneumonia (PCP), and Cytomegalovirus (CMV)
- Headache: Giant cell arteritis, typhoid fever, sinusitis, meningitis, and drug fever
- Joint pain: Rheumatoid arthritis, systemic lupus erythematosus (SLE), vasculitis, adult-onset Still’s disease
- Abdominal pain: Cholangitis, biliary obstruction, perinephric abscess, Crohn’s disease, dissecting aneurysm, and gynecological infection
- Bone pain: Osteomyelitis and lymphoma
- Sore throat: Infectious mononucleosis, retropharyngeal abscess, and streptococcal infection
- Dysuria and rectal pain: Prostatic abscess and UTI
- Altered bowel habit: Inflammatory bowel disease (IBD), typhoid fever, schistosomiasis, and amebiasis
- Skin rash: Gonococcal infection, polyarteritis nodosa (PAN), non-Hodgkin’s lymphoma (NHL), dengue fever, and connective tissue disorders.
6. Review past medical history: Malignancy (e.g., leukemia, lymphoma, and hepatocellular carcinoma), HIV infection, diabetes mellitus, inflammatory bowel disease, collagen vascular disease [e.g., SLE and rheumatoid arthritis (RA)], giant cell arteritis, tuberculosis, and heart disease (e.g., valvular heart disease)
7.Past surgical history: Postsplenectomy/post-transplantation, prosthetic heart valve, catheter, AV fistula, and recent surgery/operation
8. Drug history:
- Immunosuppressive drug/corticosteroid
- Anticoagulants
- Before the fever: Drug fever occur within 3 months after starting, taking drugs may cause hypersensitivity and low grade fever, usually associated with rash.
- Due to the allergic reaction, direct effect of drug which impairs temperature regulation (e.g., phenothiazine), antiarrhythmic drug (e.g., procainamide and quinidine); antimicrobacterial agent (e.g., penicillin, cephalosporin, and hydralazine), and phenytoin.
- After the fever: May modify clinical pictures and mask certain infection, e.g., subacute bacterial endocarditis (SBE) and antibiotic allergy.
9. Family history: Whether anyone in family has similar problem (e.g., tuberculosis and familial Mediterranean fever)
10. Social history:
- Travel: Amebiasis, typhoid fever, malaria, and schistosomiasis
- Residential area: Malaria, leptospirosis, and brucellosis
- Occupation:
- Farmers, veterinarian, and slaughter-house workers: Brucellosis
- Workers in the plastic industries: Polymer-fume fever
- Contact with domestic/wild animal/birds: Brucellosis, psittacosis (pigeons), leptospirosis, Q fever, and toxoplasmosis
- Diet history:
- Unpasteurized milk/cheese: Brucellosis
- Poorly cooked pork: Trichinosis
- Intravenous drug user (IVDU): HIV-AIDS-related condition and endocarditis
- Sexual orientation: HIV, sexually transmitted disease (STD), and pelvic inflammatory disease (PID)
- Close contact with TB patients
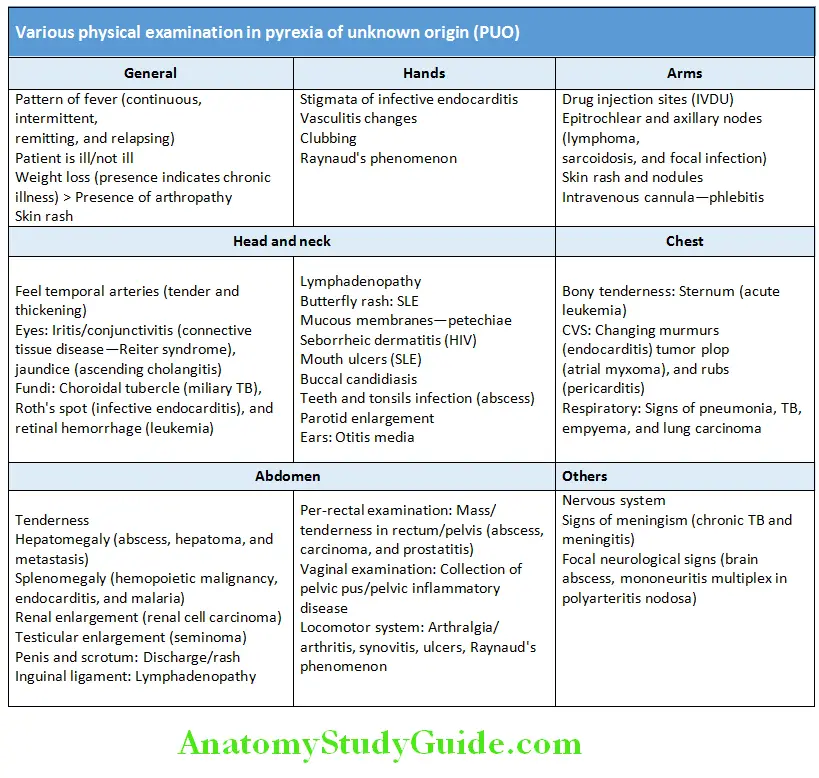
Physical Examination:
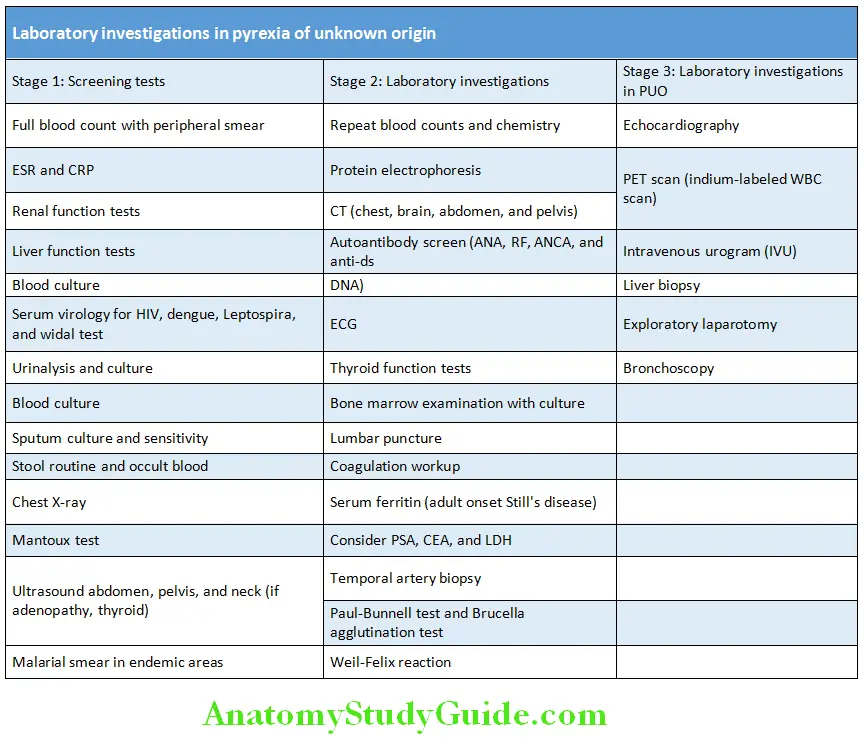
Pyrexia (Fever) Of Unknown Origin Investigations:
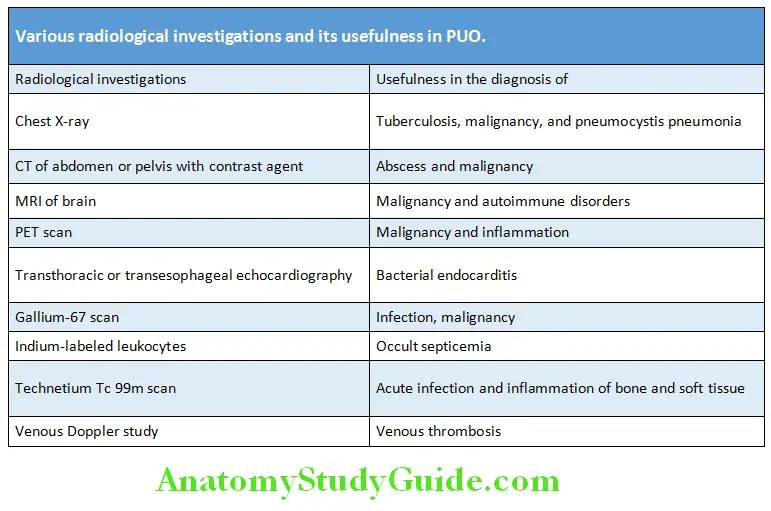
Treatment:
- Treat the underlying cause identified after investigations
- Use empirical broad-spectrum antibiotics
- Therapeutic trial of aspirin/steroids
- Naprosyn challenge for malignancy/lymphoma
- Empirical antitubercular treatment
Bacterial Infections:
Bacterial Infections Streptococcal Infections:
Streptococci are gram-positive cocci, 1 μm in diameter, nonmotile, and nonsporing. Many strains are capsulated.
Various products secreted by streptococci, which aid in their pathogenicity include streptolysin O and S, deoxyribonucleases, hyaluronidase, and erythrogenic toxins.
Streptococcal Pharyngitis:
- Incubation period is 2–4 days.
- Clinical features: Presents with abrupt onset of sore throat, dysphagia, headache, malaise, anorexia, and fever. The posterior pharyngeal wall is red and edematous. The tonsils are enlarged, red, and covered with yellowish exudate, which can be easily removed with a swab. Anterior cervical lymph nodes are enlarged and tender.
- Complications: Rheumatic fever and poststreptococcal glomerulonephritis are two major immunologically triggered complications of streptococcal infection of the upper oropharyngeal regions.
Treatment:
Pharyngitis responds readily to penicillin, erythromycin, cephalosporins (first generation given orally) or clindamycin 600–900 mg given 8th hourly.
Scarlet Fever:
Characterized by the development of an erythematous rash on the second day of illness. The primary lesion is in the throat. The rash is seen over the neck and trunk, the palms and soles are generally spared. The rash blanches on pressure. The rash subsides with extensive desquamation after 4–5 days.
Treatment:
Streptococcal lesions respond promptly to penicillin. A single intramuscular injection of benzathine penicillin G 600,000 units for children <25 kg and 1.2 million units for all others. Phenoxymethylpenicillin (penicillin V) 250 mg orally four times daily for 7–10 days is also equally effective. Erythromycin 250 mg 6 hourly for 7–10 days is given to patients allergic to penicillin. If suppuration develops, surgical drainage of the pus may be required.
Erysipelas:
Acute spreading infection of the skin and the subcutaneous tissue by streptococci. Face is commonly affected. The disease sets in abruptly with malaise, chills, headache, and vomiting. The skin lesions are erythematous with clear advancing margins which may show vesicles. The part is tender and local lymph node enlargement may occur.
Streptococcal Impetigo:
- It is inflammation of the skin characterized by isolated pustules which become crusted. Sites of predilection are around the mouth and nostrils.
- If left untreated they may ulcerate to produce shallow ulcers with crusts or scabs which may lead to pigmentation and
scarring. This stage is called ecthyma.
Cellulitis:
- This is spreading inflammation of the subcutaneous tissue due to entry of the organism through the abrasions of the skin.
- There is pain, tenderness, erythema, fever, and often regional lymphadenopathy.
Lymphangitis:
Acute lymphangitis may follow local trauma. This condition presents in the form of linear red streaks radiating from the site of entry to the draining lymph nodes.
Streptococcal Bacteremia:
Irrespective of the focus of entry and primary lesion, streptococcal bacteremia gives rise to metastatic foci of infection, such as suppurative arthritis, osteomyelitis, peritonitis, endocarditis, meningitis, or visceral abscesses.
Necrotizing Fasciitis (Synonym: Streptococcal Gangrene):
- This is a progressing destructive lesion of the subcutaneous tissue leading to necrosis of fascia and adipose tissue, but often sparing the skin. The organisms enter through trivial wounds, but within 24 hours the part is hot, swollen, tender, and edematous. The edema and violaceous hue spreads in all directions.
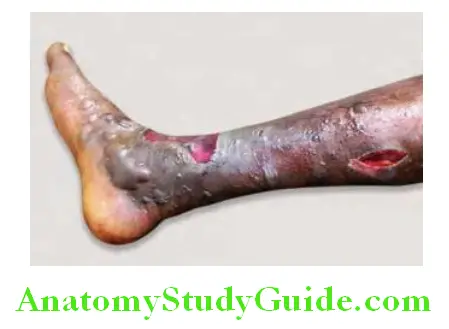
- It is more common in diabetes, immunocompromised individuals and those with local conditions impairing the vitality of the part, e.g., vascular occlusions, chronic edema, and infective lesions. Increase in the fascial compartmental pressure further jeopardizes the vascularity. Within 48 hours bullae develop, which progresses to gangrene within 4–5 days. The gangrenous area gets demarcated. General symptoms include severe prostration, toxemia, mental clouding, and delirium. If the diagnosis is missed, mortality is high.
Streptococcal Myositis:
- This is an uncommon lesion. Infection reaches the muscles by the bloodstream. Onset is with severe pain and swelling of muscles. Muscle compartment syndromes may develop. If unrecognized, mortality is over 80%.
- It should be differentiated from spontaneous gas gangrene. Presence of superficial crepitus favors the diagnosis of gas gangrene (clostridial myonecrosis).
Treatment: Includes the administration of broad-spectrum antibiotics and early surgical debridement.
Pneumonia and Empyema:
Streptococcal pneumonia usually follows a viral infection, and it manifests as bronchopneumonia. In many cases empyema develops as a complication.
Streptococcal Toxic Shock Syndrome:
Infection by group A streptococcus may lead to vascular collapse and organ failure. M-protein which is a constituent of the cell wall is the virulence factor, which plays the major role in the pathogenesis of toxic shock syndrome. It forms large aggregates with fibrinogen, in blood and tissues. These activate polymorphonuclear leukocytes intravascularly and this leads to the production of toxic shock syndrome.
Other Pathogenic Strains of Streptococci:
Group B streptococcus (Synonym: Streptococcus agalactiae):
This is a major pathogen found in the female genital tract, rectum, and also throat. Chorioamnionitis, septic abortion, and puerperal sepsis may occur during pregnancy. Urinary tract infection may occur in both sexes. Hematogenous spread may result in endocarditis, pneumonia, empyema, meningitis, and peritonitis. Immunocompromised hosts and elderly subjects are more susceptible.
Streptococcus viridans:
Viridans streptococci account for more than 40% cases of infective endocarditis. Streptococcus mutans which colonizes dental plaques is an important cause of dental caries.
Bacterial Infections Enterococci:
Causes urinary tract infections, biliary tract infections, septicemia, peritonitis, infective endocarditis, and abdominal suppuration.
Staphylococci:
Question 3. Write short note on:
- Diseases caused by staphylococci.
- Drugs used for treating staphylococcal septicemia.
Answer:
- Staphylococcus aureus: Causes pyogenic lesions, such as boils, carbuncles, wound infection, abscesses, impetigo, mastitis, osteomyelitis, pneumonia, septicemia, and pyemia.
- Coagulase negative staphylococci: It causes infection of cardiac and vascular prostheses, endocarditis, ventriculitis (cerebral), peritonitis in continuous ambulatory peritoneal dialysis, septicemia, and cystitis. Various diseases caused by staphylococci are listed in Table
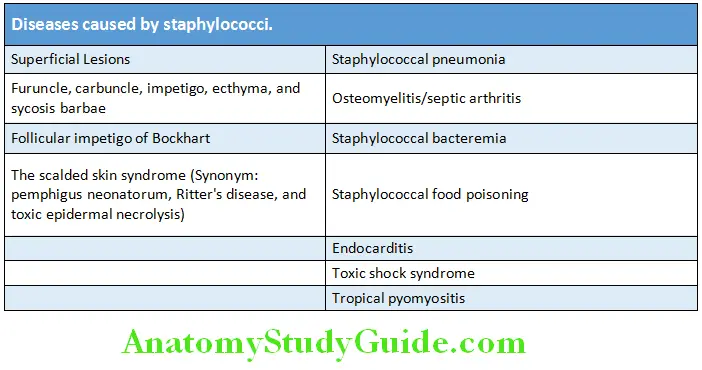
- Staphylococcus epidermidis is a common infective agent in implanted prosthetic devices. Most of the infections are acquired from hospital.
Treatment of staphylococcal infections:
- Antistaphylococcal antibiotics of the first choice:
- Oxacillin (methicillin)
- Cephalosporins of first generation (cefazolin and cephalothin)
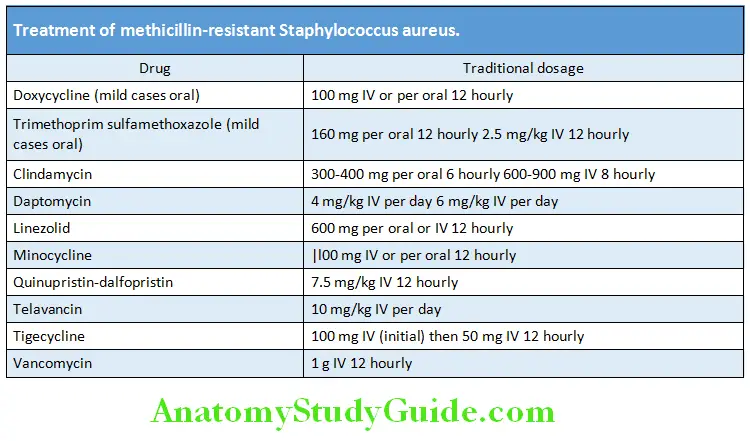
- Antistaphylococcal antibiotics of the second choice: For MRSA/VRSA
- Lincosamides (e.g., clindamycin), glycopeptides (vancomycin and teicoplanin), quinupristin/dalfopristin, chloramphenicol, minocycline, rifampin, trimethoprim sulfamethoxazole, fosfomycin, linezolid, daptomycin, tigecycline, and dalbavancin.
- Oritavancin, telithromycin, ceftobiprole medocaril anti-MRSA and cephalosporin antibiotics
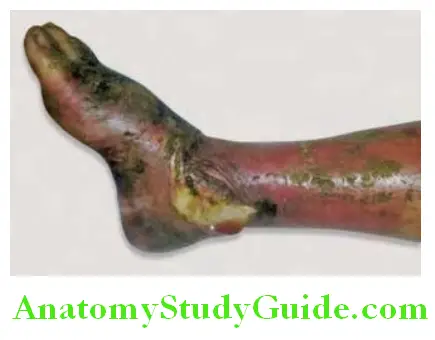
Community-acquired Methicillin-resistant Staphylococcus aureus (CA-MRSA)
Question 4. Write short note on community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA).
Answer:
- It is a type of staphylococcal bacteria that is resistant to certain antibiotics, such as methicillin, oxacillin, penicillin, and amoxicillin. It was first identified in 1968.
- The infection does not occur in individuals who are hospitalized or had any medical procedure. On the other hand, HA-MRSA occurs in the hospital setting.
- Outbreaks of CA-MRSA have occurred among categories such as: athletic teams (e.g., football, wrestling, rugby, and fencing), military barracks, correctional facilities, dormitories, daycares, and schools.
Factors that favor for MRSA transmission (5 Cs):
- Crowding
- Compromised skin (abrasions and cuts)
- Contaminated surfaces
- Frequent and skin-to-skin contact
- Lack of cleanliness
Feature: It tends to cause more aggressive skin and soft-tissue infections, necrotizing pneumonia, septic shock and bacteremia.
Treatment of methicillin-resistant Staphylococcus aureus (MRSA).
Methicillin-resistant Staphylococcus aureus Pneumococcal Infections:
Diseases caused by pneumococci are listed.
Treatment of pneumococcal infections:
- Pneumococci are generally susceptible to penicillin, but resistant strains are frequent. It is advisable to send the sputum for sensitivity tests before starting specific therapy. Benzyl penicillin is the drug of choice for uncomplicated pneumococcal infections.
- Multiple drug-resistant pneumococci respond to vancomycin 2 g/day given IV in divided doses or cefotaxime or ceftazidime. Levofloxacin is also effective.
Diseases caused by pneumococci:
- Pneumococcal pneumonia
- Extrapulmonary pneumococcal lesions
- Pneumococcal meningitis
- Pneumococcal peritonitis
Methicillin-resistant Staphylococcus aureus Meningococcal Infections:
Meningococcal Meningitis (Synonym: Cerebrospinal Fever):
Meningococcemia may be fulminant or chronic.
Fulminant meningococcemia:
This is characterized by abrupt onset, severe constitutional disturbances, peripheral vascular collapse, shock, and sometimes myocarditis. In some cases the illness may progress rapidly so that toxemia and shock may occur within hours.
These features are collectively called Waterhouse–Friderichsen syndrome. This syndrome is caused by hemorrhage into the adrenal glands resulting in acute adrenal failure. Toxic vasculitis aggravates the hypotension. Complications include endocarditis, allergic polyarthritis, pneumonia, and osteomyelitis.
Treatment:
Penicillin G is the antibiotic of choice and should be administered intravenously in a dose of 24 million units daily in divided doses.
Addition of glucocorticoids early in treatment favors prompt recovery and prevents complications considerably.
Ampicillin in a dose of 200–400 mg/kg body weight daily is also equally effective. The third-generation cephalosporins, especially
cefotaxime and ceftriaxone are equally effective.
Diphtheria:
Question 5. Discuss the clinical manifestations, complications, diagnosis, and management of diphtheria.
Answer:
- Diphtheria is a nasopharyngeal (respiratory diphtheria) and/or skin infection (cutaneous diphtheria) caused by Corynebacterium diphtheriae.
- Corynebacterium diphtheriae is a gram-positive bacillus. Corynebacterium diphtheriae remains localized at the site of infection but releases a powerful soluble exotoxin that damages the heart muscle and the nervous system.
Diphtheria Mode of Transmission:
- It is through airborne/droplet infection from active cases or carriers. Infection may also transmit through skin lesions.
- Incubation period: 2–7 days.
Pseudomembrane:
The diagnostic pathologic feature is mucosal ulcers coated by pseudomembrane. The pseudomembrane has a well-defined edge which appears as wash-leather, elevated, firm, and grayish-green (black in advanced stages) adherent membrane. It is surrounded by a zone of inflammation.
Diphtheria Clinical Features:
- Its manifestations may be local (due to the membrane) or systemic (due to exotoxin).
- Insidious in onset with a sore throat and fever being the usual manifestation. The fever is moderate but there is usually marked tachycardia.
Respiratory diphtheria:
Pharyngeal diphtheria: It is characterized by marked tonsillar and pharyngeal inflammation and the presence of a pseudomembrane. There may be regional often tender lymphadenopathy (cervical lymph nodes), and along with marked edema of submandibular areas produces the so-called “bull-neck” appearance (swelling of the neck). Pharyngeal diphtheria is associated with the greatest toxicity.
Laryngeal diphtheria: It is usually represents extension of the membrane from the pharynx. Extension and sloughing of membranes may produce fatal airway obstruction. It usually presents with a husky voice, a brassy cough and later dyspnea and cyanosis due to respiratory obstruction.
Nasal diphtheria: It is restricted to the nasal mucosa and is characterized by the presence of a unilateral, serosanguineous (frequently blood-stained) nasal discharge.
Cutaneous diphtheria:
It is uncommon but occurs in individuals with poor personal hygiene and with burns. It produces round, deep, “punchedout” skin ulcers with undermined edges and is covered by a gray-yellow or gray-brown adherent membrane. The ulcers occur more commonly on the lower and upper extremities, head, and trunk. Constitutional symptoms are not common.
Diphtheria Complications:
- Airway (laryngeal) obstruction: It may occur with advanced diphtheria. Airway obstruction may be either due to the sloughed pseudomembrane or extension of the pseudomembrane to the larynx or into the tracheobronchial tree (bronchopulmonary diphtheria). It is mainly observed in children because of their small airways.
- Cardiac complications: These include myocarditis with arrhythmias, cardiac failure, and ECG changes. They often develop weeks after initial episode of diphtheria. These are usually reversible.
- Neurological complications occur in 75% of cases.
- Palatal palsy may develop after 10 days.
- Polyneuropathy with weakness and paresthesia may develop 3–5 weeks after the onset of diphtheria.
- Paralysis of accommodation may manifest as difficulty in reading small print.
- Encephalitis can occur rarely.
- Other complications include pneumonia, renal failure, encephalitis, cerebral infarction, and pulmonary embolism.
Diphtheria Diagnosis:
- Clinical diagnosis
- Confirmation of diagnosis:
- Demonstration of Corynebacterium diphtheriae on methylene blue-stained preparations.
- Bacterial culture of Corynebacterium diphtheriae on Loeffler’s medium and toxin studies (Elek test).
Management:
- Patients should be hospitalized with close monitoring of cardiac and respiratory function. Patient should be isolated and strict bed rest.
- Diphtheria antitoxin is the only specific treatment. It is produced from hyperimmune horse serum and it neutralizes circulating toxin.
- It must be given as early in the course of diphtheria as possible without awaiting the result of a throat swab. Because any delay in administration can be dangerous because toxin once fixed to the tissues can no longer be neutralized by antitoxin.
- Dose: It is administered intravenously over 60 minutes after an initial test dose to exclude any allergic reaction. 20,000 to 40,000 units for pharyngeal/laryngeal disease of <48 hours duration, 40,000 to 60,000 units for nasopharyngeal disease, and 80,000 to 120,000 units for >3 days of illness or diffuse neck swelling (“bull neck”).
- Adverse reactions: It can cause two types of reactions, an immediate anaphylactic reaction and delayed serum sickness. The immediate anaphylactic reaction is treated with adrenaline and an antihistamine.
- Antibiotics should be given concurrently to eliminate C. diphtheriae and thereby remove the source of toxin production. Benzylpenicillin (1,200 mg four times daily IV) or amoxicillin (500 mg three times daily) is given for 2 weeks. Patients allergic to penicillin are given erythromycin (500 mg four times daily for 14 days).
- Tracheostomy or intubation may be needed for respiratory distress.
- Immunization: Primary diphtheria does not produce immunity against infection. Hence, following recovery all sufferers should be immunized with diphtheria toxoid. Close contacts should be protected by erythromycin prophylaxis and also by immunization. Vaccines include DPT (diphtheria, pertussis, tetanus) and DT (diphtheria, tetanus).
- Contact prophylaxis: Single dose of penicillin G benzathine [600,000 units intramuscularly (IM)] or oral erythromycin (500 mg four times daily for 7–10 days).
Tetanus:
Question 6. Write a short essay/note on tetanus.
Answer:
Tetanus is due to infection by toxin secreting clostridium namely Clostridium tetani. The organism is found in soil derived from animal and human excreta.
Tetanus Mode of infection: Infection enters the body through a contaminated wound (injury may be trivial). It can also develop as complication in intravenous drug misusers. Neonatal tetanus may develop following contamination of the umbilical stump, often after dressing the area (unhygienic practices) with dung (e.g., in many developing countries) or site of circumcision, causing tetanus neonatorum.
Pathogenesis: During circumstances unfavorable to the growth of Clostridium tetani, it forms spores and remain dormant for years in the soil. Spores germinate and organism multiplies only in the anaerobic conditions. Thus, it may multiply in areas of tissue necrosis or wherever the oxygen tension is reduced by the presence of other organisms (e.g., aerobic organism).
Clostridium tetani is not invasive and remain localized. Its clinical manifestations are due to the potent neurotoxin (exotoxin) called tetanospasmin.
Incubation period: Varies from 2 days to several weeks after injury. Shorter the incubation period, the more severe the attack and the worse the prognosis.
Clinical Features:
Question 7. Write a short essay/note on the clinical features of tetanus.
Answer:
Generalized tetanus is the most common form of tetanus.
- Lockjaw: General malaise is rapidly followed by the most important symptom namely trismus. It is due to spasm of the masseter muscles, which causes difficulty in opening the mouth and in masticating.
- Risus sardonicus: When the tonic rigidity involve the muscles of the face, neck, and trunk, contraction of the frontalis and the muscles at the angles of the mouth produces characteristic grinning expression known as “risus sardonicus”.
- Opisthotonus: Varying degree of rigidity develops in the muscles at the neck and trunk. The back is usually slightly arched (“opisthotonus”) and the abdominal wall appears board-like.
Severe disease:
- If the disease is severe, painful, violent, exhausting, and reflex spasms (convulsions) develop, usually within 24–72
hours of the initial symptoms and lasts for a few seconds to 3–4 minutes. The interval between the first symptom and
the first spasm is known as the “onset time”. - The spasms can be spontaneous or may be induced by stimuli such as movement or noise or by light. Laryngeal spasm can impair respiration; esophageal and urethral spasm can produce dysphagia and urinary retention, respectively.
Patients are mentally alert. - Autonomic involvement may produce cardiovascular complications (e.g., tachycardia, a labile blood pressure,
sweating, and cardiac arrhythmias).
Death: Spasms gradually increase in frequency and severity and death may occur from exhaustion, hypoxia, cardiac arrest, asphyxia, respiratory failure or aspiration pneumonia or exhaustion. Mild cases with rigidity usually recover.
Local tetanus is a milder form of the disease in which the pain, stiffness, increased tone or spasms of the muscles develop only near the infected wound. Prognosis is good and recovery usually occurs if treatment is commenced at this stage.
Cephalic tetanus: Uncommon but fatal. It usually develops due to entry of C. tetani through the middle ear. Cranial nerve
abnormalities (e.g., seventh nerve) are usual.
Neonatal tetanus is usually develops due to infection of the umbilical stump. Characterized by failure to thrive, poor sucking, grimacing, and irritability followed by intense rigidity and spasms. Mortality is almost 100%. It can be prevented by immunizing all women of childbearing age and providing clean delivery facilities.
Investigations/Diagnosis:
- Diagnosis is usually made on clinical grounds.
- C. tetani: Rarely possible to isolate from wounds (original locus of entry).
Differential diagnosis: Phenothiazine over dosage, strychnine poisoning, meningitis, and tetany.
Management/Treatment of Tetanus:
Question 8. Write a short essay/note on the treatment of tetanus.
Answer:
Suspected tetanus:
Care of the wound: Clean the wound and debrided if necessary, to remove the source of toxin.
Human tetanus immunoglobulin: In the dose of 250 units should be given along with an intramuscular injection of tetanus toxoid. If the patient is already immunized a single booster dose of the tetanus toxoid is given; otherwise the full three dose course of adsorbed vaccine is given.
Established tetanus:
Management of established disease should be started as soon as possible.
Prevent further toxin production: Debridement of wound and antibiotics (see below).
General supportive medical and nursing care. Patient is isolated in a quiet, well ventilated and darkened room. Maintain hydration
and nutrition and treat secondary infections.
Control of spasm: Nurse in a quiet room, avoid unnecessary stimuli. Benzodiazepines/IV diazepam is used to control spasms and sedate the patient. If spasms continue, paralyze patient and ventilate. Baclofen may be useful.
Intubation and mechanical ventilation: If the airway is compromised.
Magnesium sulfate infusion: Reduces the need for antispasmodics.
Antibiotics and antitoxin: Given even in the absence of an obvious wound. Drug of choice is intravenous metronidazole. Other antibiotic include penicillin (Benzylpenicillin 600 mg IV four times daily) and cephalosporins.
Neutralization of absorbed toxin: Human tetanus immunoglobulin (HTIG) 3,000–6,000 IU should is given by intramuscular injection to neutralize any circulating toxin. If HTIG is not available, immune equine tetanus immunoglobulin 10,000 IU should be given intramuscularly but there is a high incidence of severe allergic reactions.
If the patient recovers active immunization should be instituted, as immunity following tetanus is incomplete.
Anthrax:
Question 9. Write short essay on anthrax.
- Anthrax is a zoonotic disease caused by Bacillus anthracis.
- Bacillus anthracis is a gram-positive bacillus with a central spore. It produces toxins and is responsible for the clinical features of disease that most closely correlated with its virulence.
Anthrax Mode of Transmission:
It is through direct contact (inoculation of the spores) with an infected animal particularly herbivores. Infection is most frequent as an occupational disease in farmers, butchers, and dealers in wool and animal hides. Spores of Bacillus anthracis can also be ingested or inhaled. Deliberate release of anthrax spores is an important bioterrorist weapon.
Incubation period: 1–10 days.
Anthrax Clinical Manifestations:
It depends on the route of entry of the anthrax spores.
Cutaneous anthrax:
Question 10. Write short essay on cutaneous anthrax and malignant pustule.
Answer:
- It is the most common type of anthrax. It follows inoculation of spores into the subcutaneous of the exposed skin. Occupational exposure to anthrax spores during processing of hides and bone products results in cutaneous anthrax.
- Skin lesions (Hide-porter’s disease) begin as a small, itching, erythematous, maculopapule on an edematous hemorrhagic base. The lesion is initially painless despite edema. It enlarges to form a vesicle filled with serosanguinous fluid and is surrounded by gross edema (“malignant pustule”). The vesicle ulcerates and dries to form a central depressed thick black “eschar” surrounded by blebs. Despite marked edema, pain is infrequent.
- It is self-limiting illness in the majority of patients, but occasionally perivascular edema and regional lymphadenopathy may be associated with marked toxemia.
Inhalational anthrax (Woolsorter’s disease):
Question 11. Write short essay on inhalation anthrax and Woolsorter’s disease.
Answer:
- It is extremely rare and follows inhalation of spores into the lung producing “Woolsorter’s disease”
- Bioterrorism-related anthrax also due to inhalation of spores.
- It begins with fever, nonproductive cough, dyspnea, headache, and retrosternal discomfort. Patient develops bronchopneumonia. Symptoms of septicemia may develop 3–14 days following exposure.
- Pleural effusions (hemorrhagic) are common and meningitis may occur.
- Chest X-ray shows bronchopneumonia, widening of the mediastinum, and pleural effusions.
- Without rapid and aggressive treatment at the onset of symptoms, the mortality ranges from 50 to 90%.
Gastrointestinal anthrax:
- It is associated with ingestion of undercooked and contaminated meat products.
- The cecum is involved and it manifests as a severe gastroenteritis. The symptoms include nausea, vomiting, anorexia, and fever followed in 2–3 days by severe abdominal pain and bloody diarrhea.
- Toxemia, shock, and death may develop rapidly.
Inhalational anthrax Diagnosis:
- Demonstrating the organism: A stained smear of fluid taken from the edge of a skin lesion may demonstrate the organism.
- The organism may also be demonstrated in stools, laryngeal secretions, sputum, and CSF.
- Culture of blood and other body fluids: Bacillus anthracis can be cultured in mice, rabbits or guinea pigs.
- Serological examination: ELISAs for detecting antibodies to both the organism and a toxin.
- Chest X-ray may show mediastinal widening, bronchopneumonia, and pleural effusion.
Treatment:
Systemic anthrax with meningitis: Ciprofloxacin (400 mg TDS) plus meropenem plus linezolid/clindamycin.
Anthrax related to bioterrorism: Ciprofloxacin is the drug of choice. Dose is 400 mg IV ciprofloxacin BID. Once the patient stabilizes, ciprofloxacin is given orally in a dose of 500 mg BID. Duration of treatment is 60 days.
Raxibacumab and obiltoxaximab: 40–80 mg/kg human monoclonal antibody directed against the protective antigen has been shown in animal studies to improve survival in inhalation anthrax. Anthrax immunoglobulin is available to treat inhalational anthrax.
Prevention and Control:
Prophylaxis:
- Ciprofloxacin (500 mg twice daily) is recommended for individuals with high risk of exposure to anthrax spores.
- Doxycycline (100 mg BID) for 60 days.
- Vaccination of animals and persons at risk.
- When an infected animal dies, it should be burned and the area in which it was housed must be disinfected.
Plague:
Question 12. Write short essay/note on plague.
Answer:
- Plague is caused by a small gram-negative, nonmotile bacillus namely Yersinia pestis.
- 11 Yersinia species—three human pathogens namely Y. pestis, Y. pseudotuberculosis, Y. enterocolitica.
- One of three WHO quarantinable diseases.
- Plague is also called as “black death”. Historically, three plague pandemics have caused more than 200 million deaths, including the Black Death epidemic in 14th century Europe (Justinian 541 AD, Black Death 1,346, China 1,855).
- Used for biological warfare.
Plague Source of Infection:
The main reservoirs are woodland rodents (sylvatic rats) that spread infection to the domestic rat species (Rattus rattus) and finally infected rat fleas (Xenopsylla cheopis). These fleas bite humans when there is a sudden reduction in the rat population.
Plague Route of Infection:
- Rat flea bite: Most common route in humans is after bite of a plague-infected rat flea.
- Direct contact with infected tissues or fluids from sick or dead plague-infected animals. Hunters and trappers can develop plague from handling rodents.
- Droplet infection: Plague pneumonia or by laboratory exposure.
- Incubation period: 3–6 days (shorter in pneumonic plague).
Plague Clinical Features:
Type of plague: Four clinical forms are recognized: bubonic, septicemic, pneumonic, and cutaneous.
Bubonic plague:
- Most common form of the disease and occurs in about 90% of infected individuals.
- Onset is usually acute/sudden, with a high fever, rigor, chills, severe headache, dry skin, myalgia, nausea, vomiting and when severe, prostration. This is rapidly followed painful lymphadenopathy. Characteristically these lymph nodes are tender and suppurate in 1–2 weeks. Most common site for lymphadenopathy is the inguinal region or axilla. The swollen lymph nodes and surrounding tissue constitute the characteristic “bubo” called so because they are rarely fluctuant.
- Other manifestations include apathy, confusion, fright, anxiety, oliguria or anuria, tachycardia, toxemia, and hypotension. The spleen is usually palpable.
- Without treatment, complications, such as secondary septicemia, secondary pneumonia, and meningitis may occur. Mortality rate for untreated cases is 60%.
Septicemic plague:
- It may be primary without signs of primary disease (primary septicemic plague) or secondary as a complication of untreated bubonic plague or pneumonic plague (secondary septicemic plague).
- Primary septicemic plague presents as an acute fulminant infection characterized by high fever, chills, and malaise, but without any lymph node enlargement. Elderly individuals are more prone. The patient is toxic and may develop gastrointestinal symptoms, such as nausea, vomiting, abdominal pain, and diarrhea. Patients may develop hypotension, septic shock, renal failure, ARDS, and disseminated intravascular coagulation (DIC).
- Gangrene of acral regions (tip of the nose or the fingers and toes) may develop due to thrombosis of small artery in advanced stages (hence named Black Death).
- Left untreated, it deteriorates rapidly and the mortality approaches 100%.
Pneumonic plague:
- It may occur as a primary infection in the lung or as secondary infection (as a complication of the bubonic and septicemic plague—secondary pneumonia).
- Primary form: Develops within 1–6 days of exposure. It begins suddenly with features of a fulminant pneumonia. Patient develops malaise, high fever, vomiting, abdominal pain, diarrhea, and marked prostration. Soon followed by cough, dyspnea, copious blood-stained, frothy, highly infective sputum marked respiratory distress/failure, cyanosis, and septic shock. Without antibiotics, death occurs in almost all patients within 2 or 3 days.
- Chest X-ray: Shows bilateral infiltrates that may be nodular and progress to an ARDS-like picture.
Cutaneous plague:
It presents either as a pustule, eschar or papule or an extensive purpura. It can develop necrosis and gangrene.
Investigations and Diagnoses:
Diagnosis is based on clinical, epidemiological, and laboratory findings.
- Demonstration of organism: For rapid diagnosis, smears are prepared from blood, sputum, bubo aspirate (lymph node aspirate), and cerebrospinal fluid. They are stained with Gram, Giemsa or Wayson’s stains (contains methylene blue), and examined under microscopy. Y. pestis is seen as bipolar staining coccobacilli, giving a “safety pin” appearance.
- Culture of organism: From blood, sputum, and bubo aspirates.
- Serological diagnosis: A presumptive diagnosis in an appropriate clinical setting is possible by a rapid antigen detection test (by immunofluorescence, using Y. pestis F1 antigen-specific antibodies).
- Septicemic plague: Often associated with laboratory findings of DIC.
- Chest X-ray: In pneumonic plague, it shows evidence of multilobar consolidation, cavities or bronchopneumonia.
- Blood: WBC count 20,000/mm2 and/or thrombocytopenia in about 50% of patients.
Treatment/Management:
If the diagnosis is suspected on clinical and epidemiological grounds, urgent treatment is required even before the results of culture studies are available.
First choices:
- Streptomycin: 30 mg/kg per day IM (up to a total dose of 2 g) in two divided doses for 10 days.
- Gentamicin as effective OD dosing and less toxic.
Second choices:
- Tetracyclines: Doxycycline
- Fluoroquinolones: Ciprofloxacin, levofloxacin, ofloxacin, and moxifloxacin
- Chloramphenicol: 1st choice for meningitis ± aminoglycoside supportive treatment of ARDS, DIC, and shock
Prophylaxis:
- Control of rats and flea
- Avoid handling and skinning of wild animal in endemic areas.
- Prevention of human-to-human transmission: Patients with plague pneumonia should be isolated until at least 4 days of antibiotic treatment have been administered. For the other types of the plague, patients should be isolated for the first 48 hours or until clinical improvement begins. Attendants must wear gowns, masks, and gloves and healthcare workers should use highefficiency respirators.
- Chemoprophylaxis: Close contact with a patient with pneumonic plague should receive postexposure antibiotic prophylaxis (doxycycline 100 mg or ciprofloxacin 500 mg twice daily) for 7 days.
- Vaccine: A partially effective formalin-killed vaccine is available for those who travel to plague-endemic areas and individuals at occupational risk.
Botulism:
Question 13. Write a short essay/note on botulism.
Answer:
- Botulism is caused by neurotoxin (botulinum) produced by Clostridium botulinum.
- It produces a neurotoxin which is the most potent poison known and cause disease after ingestion of even picogram (as low as 0.05 g) amounts. There are seven types of toxins which are labeled as A to G.
Botulism Mode of Infection:
- Ingestion: It can contaminate many foodstuffs, such as canned or bottled foodstuff, in which it can multiply. Most cases of botulism are due to consumption of contaminated food being served undercooked. Contaminated honey causes in infant botulism, in which the organism colonizes the gastrointestinal tract.
- Inoculation: Wound botulism can develop in injection drug-users.
- Incubation period: 2 hours to 8 days.
Botulism Classifiation:
- Food-borne botulism: It occurs due to ingestion of toxin present in the contaminated food (fish and canned food) and it is the most common type of botulism.
- Infantile botulism: It develops due to ingestion of spores, which germinate in the gut and produce toxin.
- Wound botulism: It follows the contamination of wounds, street heroin injection contaminated with C. botulinum. Other types are inhalational and iatrogenic botulism.
Botulism Clinical Features:
Food-borne botulism:
- Initial symptoms are related to gastrointestinal symptoms, such as nausea and diarrhea. These are rapidly followed neurotoxic effects of toxin. The toxin causes mainly bulbar and ocular palsies (difficulty in swallowing, blurred or double vision, and ptosis), progressing to symmetrical descending limb weakness, diaphragmatic paralysis, respiratory paralysis, and death. No sensory deficits seen except blurring of vision.
- Absence of fever and patients are alert, remains responsive, though mild drowsiness may be present.
- Heart rate is normal or slow and blood pressure is normal.
- Parasympathetic dysfunction: Rare and may produce dry mouth, paralytic ileus, and dilated nonreactive pupils.
Infantile botulism:
- Characterized by onset of constipation, followed by weakness in sucking, crying or swallowing. Later there is progressive bulbar and muscle weakness of the extremities.
Wound botulism:
- It is similar to food-borne botulism except that GI upset does not occur.
Botulism Diagnosis:
- Diagnosis of botulism is usually based on clinical features.
- Detection of toxin: In blood (foodborne) or stools (infant botulism) or in the contaminated food.
- Culture of organism from wound.
Diffrential Diagnosis:
Guillain–Barré (GB) syndrome, myasthenia gravis, tick paralysis, diphtheria, and hypermagnesemia.
Treatment:
- It is mainly general supportive care with mechanical ventilation, prevention of secondary infection, and administration of antitoxin (not available in India). Equine serum heptavalent botulism antitoxin and human-derived botulism immune globulin are available in United States.
- Antibiotics are of no much use. However, penicillin G and metronidazole are used. The overall mortality is high, but those who survive the acute paralysis fully recover.
Whooping Cough:
Question 14. Describe the etiology, clinical features, complications, diagnosis, and management of whooping cough.
Answer:
- Whooping cough (pertussis) is an acute infection of the respiratory tract caused by Bordetella pertussis. Bordetella pertussis is a gram-negative coccobacillus.
- The term pertussis means “violent cough” which is the most prominent feature of the illness.
Whooping Cough Mode of spread: Pertussis is highly contagious and spreads by droplet infection.
Incubation period: 7–10 days.
Age group: Classic case is seen in childhood, with 90% occurring below 5 years of age.
Whooping Cough Clinical Features:
Catarrhal phase:
This first stage is highly infectious characterized by upper respiratory catarrh with rhinitis, lacrimation (conjunctivitis), lowgrade fever, and an unproductive cough. This stage lasts about 1–2 weeks.
Paroxysmal phase:
It is called so because of the characteristic paroxysms of coughing.
- Whoop: During this stage, the cough becomes spasmodic, more frequent and severe with repetitive bouts of 5–10 coughs.
- The coughing paroxysms episode may be terminated by a classic inspiratory audible whoop. The whoop is due to rapid inspiration against a closed glottis at the end of a paroxysm. It is observed only in younger patients in whom the lumen of the respiratory tract is narrowed due to mucus secretion and mucosal edema. These paroxysms usually terminate in vomiting. Early paroxysmal stage is also infectious.
- Other features include conjunctival suffusion and petechiae and ulceration of the frenulum of the tongue. Lymphocytosis is observed.
- This stage lasts for about 2 weeks and may be associated with many complications.
Convalescence phase:
It follows the paroxysmal phase during which slow resolution of whoop occurs. This phase can last 1–3 months, and cough may persist for several weeks to months.
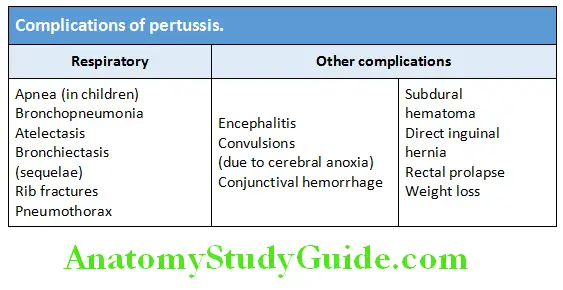
Complications:
Complications Diagnosis:
- Clinical diagnosis is not difficult if the classic symptoms, characteristic whoop and a history of contact with an infected individual are present.
- Blood: Peripheral blood may show lymphocytosis.
- Culture of nasopharyngeal secretions is the gold standard for diagnosis.
- B. pertussis DNA detection by PCR assay.
- Direct fluorescent antibody test.
Complications Management:
Antibiotics:
- Erythromycin for 7–14 days is the recommended treatment.
CDC recommends azithromycin for 5 days (500 mg day 1, followed by 250 mg day 2 through 5) or clarithromycin (500 mg twice daily for 7 days). - Trimethoprim/sulfamethoxazole reduces pertussis transmission and is an alternative treatment for patients who are allergic to macrolides.
- Cough suppressants are not effective.
- Others: Steroids, antihistamines, b-agonists, and immunoglobulins are not beneficial.
Complications Prevention:
Isolation of patient: Patients should be isolated to prevent contact with others, e.g., in hostels and boarding schools.
Catarrhal phase: If antibiotics have been given during catarrhal phase, patient is infectious until 5 days after starting antibiotics.
Paroxysmal phase: Patient is contagious till 3 weeks after the paroxysmal stage ends if not treated during catarrhal stage.
Chemoprophylaxis: Risk of transmission of B. pertussis within households is high. Hence, close contacts of patients should receive macrolides, especially if they are not vaccinated.
Active immunization: Pertussis is an easily preventable disease with active immunization by DPT vaccine. Immunity begins to decline 4–12 years after vaccination, and may make adolescent and adult susceptible to infection. Rarely, the vaccine can produce convulsions and neurological damage. Currently acellular effective vaccines with few adverse reactions are available. A triple vaccine containing tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) is available for use in adolescents and adults.
Enteric Fever:
Question 14. Describe the etiology, clinical features, investigations, diagnosis, complications, and treatment of typhoid fever. (or) Write short note on enteric fever (typhoid and paratyphoid fever).
Answer:
Enteric fever is the general term, which includes both typhoid and paratyphoid fever. It is an acute systemic illness characterized by fever, headache and abdominal discomfort.
Enteric Fever Causative Agent:
- Enteric fevers are caused by Salmonella typhi and Salmonella paratyphi. Salmonella are gram-negative, flagellate, motile, nonsporulating, and facultative anaerobic bacilli (rods). Boiling or chlorination of water and pasteurization of milk destroy the bacilli.
- Typhoid fever (enteric) is an acute systemic disease caused by infection with Salmonella typhi (also known as Salmonella enterica serovar Typhi). Paratyphoid fever is a clinically similar but milder disease caused by Salmonella paratyphi (Salmonella enterica serovar Paratyphi A, B or C).
Enteric Fever Source of Infection:
Humans are the only natural reservoir and include:
- Patient suffering from disease: Infected urine, feces, or other secretions from patients.
- Chronic carriers of typhoid fever: S. typhi or S. paratyphi colonizes in the gallbladder, urinary bladder, or biliary tree.
Enteric Fever Mode of transmission: From person-to-person contact.
- Ingestion of contaminated food (especially dairy products) and shellfish or water. Chronic carriers, often food handlers transmit the disease.
- Direct spread: Rare by finger-to-mouth contact with feces (fecal-oral route), urine, or other secretions is rare.
Incubation period: Usually 10–14 days and for paratyphoid it is shorter.
Pathogenesis:
- The typhoid bacilli (Salmonella) are ingested through contaminated food or water are able to survive in gastric acid of the stomach and reach mucosa of small intestine.
- In the small intestine, they penetrate the ileal mucosa, reach the submucosa and are phagocytosed by the macrophages in the Peyer’s patches.
- They are carried to the mesenteric lymph node via lymphatics and enter the bloodstream via the thoracic duct causing bacteremia.
- They colonize reticuloendothelial tissues (liver, gallbladder, spleen, and bone marrow), and multiply further and reenter bloodstream causing massive bacteremia (occurs towards the end of incubation period) and disease clinically manifests.
- In the intestine, the bacilli are localized to the Peyer’s patches and lymphoid follicles of the terminal ileum. They cause inflammation, plateau-like elevations of Peyer’s patches and necrosis, which results in characteristic oval typhoid ulcers.
Pathogenesis Clinical Features:
- Onset is gradual and nonspecific. Patients usually present with fever, anorexia, headache, abdominal pain, bloating, nausea, and vomiting.
- Febver: The temperature rises in a step-ladder fashion (step-ladder fever) to 40–41°C for 4 or 5 days in some cases. The hallmark of typhoid fever is continuous, persistent fever, often lasting 4–8 weeks in untreated patients.
- Early intestinal manifestations include constipation (especially in adults) or mild diarrhea (in children).
Pathogenesis Physical fidings:
- In the early stages abdominal tenderness, hepatosplenomegaly, lymphadenopathy, and a scanty maculopapular rash (“rose spots”) are found.
- Rose spots or “rose-red spots”: These are small 2–4 mm, pale-red maculopapular lesions on the skin that fade/ blanch on pressure appear on the chest and abdomen, which occur during first week and usually last only 2–3 days. They result from bacterial embolism and Salmonella can be cultured from the biopsy of these lesions.
- Mild hepatosplenomegaly: Spleen is soft and palpable (around the seventh to tenth day) may be accompanied by tender hepatomegaly.
- Relative bradycardia: The pulse is often slower than would be expected from the height of the fever.
- Intestinal manifestations: By the end of first week, constipation is succeeded by diarrhea and abdominal distension, with tenderness in the right iliac fossa. The stools are loose and greenish in color and characteristically described as “pea-soup” intestinal complications often develop in the third of fourth week of illness.
- If untreated by the end of second week: Patient may be profoundly ill
- By third week: Toxemia increases and patient may develop coma and die
- The fouth week of the illness is characterized by gradual improvement.
Complications of Typhoid:
Laboratory Diagnosis:
Question 16. Write short note on complications of typhoid fever (OR) Write short note on:
- Laboratory diagnosis of typhoid fever.
- Widal test.
Complications of typhoid:
- General complications: Toxemia, dehydration, peripheral circulatory failure, and DIC.
- Intestinal complications: The most common intestinal complication is ileus. Perforation of typhoid ulcer and hemorrhage from the ulcer may occur at the end of the second week or during the third week of the illness.
- Extraintestinal complications:
- Neurological: Delirium (muttering delirium), psychosis, seizures, coma vigil, catatonia, meningitis, encephalopathy, GB syndrome peripheral neuritis, and deafness.
- Miscellaneous: Myocarditis, endocarditis, pericarditis, pneumonia, cholecystitis, pyelonephritis, glomerulonephritis, osteomyelitis, arthritis, periostitis, hepatitis, and thrombophlebitis. Patients with sickle cell disease are susceptible to Salmonella osteomyelitis.
- Carrier state: Persistence of bacilli in the gallbladder or urinary tract may result in passage of bacilli in the feces or urine and causes a “carrier state” which is the source of infection to others. After clinical recovery, about 5–10% will continue to excrete S. typhi for several months and they are termed convalescent carriers.
Laboratory diagnosis of typhoid fever:
- Total leukocyte count: It shows leukopenia with relative lymphocytosis. Eosinophils are usually absent.
- Isolation of Bacilli:
- Blood culture: This is the “Gold Standard” investigation for diagnosis of typhoid. The maximum positivity of blood culture is in first week of fever in 90% of patients and remains positive in second week till the fever subsides. Blood culture rapidly becomes negative on treatment with antibiotics. During early phase, bone marrow culture aspirate is more sensitive than blood culture, even after a brief prior antibiotic treatment.
- Stool cultures: It is almost as valuable as blood culture and become positive in the third week.
- Urine culture: It reveals the organism in approximately 25% of patients by third week.
- Widal test/reaction: Classic Widal test measures agglutinating antibodies against O, H, and Vi antigens of S. typhi and H antigens of S. paratyphi A and B, but lacks sensitivity and specificity.
- Widal test (immunological reactions) becomes positive from end of the first week till fourth week. There are many false-positive (anamnestic reaction) and occasional falsenegative Widal reactions. Vi antigen is alone detected in the carrier state.
- The mean sensitivity, specificity, NPV, and PPV of Widal test remains below 80%. Therefore, Widal test should not be used as a diagnostic tool to rule out typhoid fever unless supported by invasive clinical pictures and other confirmatory tests.
- Other serologic tests: They are available for the rapid diagnosis of typhoid fever with a higher sensitivity.
- Molecular methods: PCR detects flagellin, somatic gene, and Vi gene.
Question 17. Write short note on
Answer:
- Treatment and prevention of typhoid fever.
- Chronic carrier state in typhoid.
- New drugs with dosage and duration used to treat enteric fever.
Treatment:
- General management: These include bed rest, isolation, and maintenance of nutrition and fluid intake.
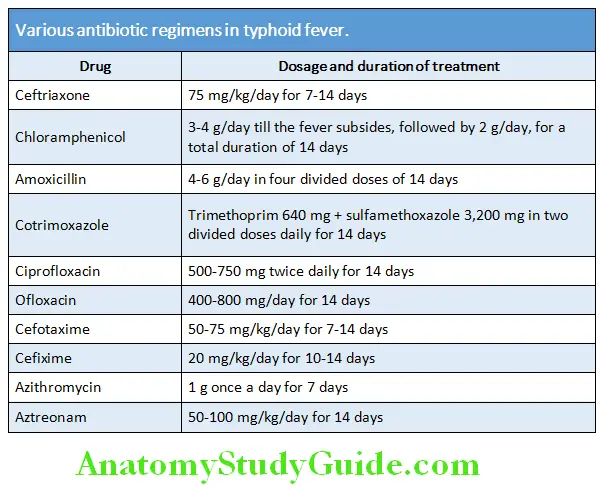
- Antibiotic therapy: Several antibiotics are effective in enteric fever and various drug regimens are presented in Table 4.9. It must be guided by culture and sensitivity report.
- Multidrug resistant strains: Certain strains of S. typhi (especially in India) are resistant to chloramphenicol, amoxicillin, and cotrimoxazole are called as multidrug resistant strains. These should be treated with ciprofloxacin.
- NARST-nalidixic acid resistant Salmonella typhi: Sometimes, strains that are sensitive to ciprofloxacin in vitro may not respond to ciprofloxacin. They are usually resistant to nalidixic acid when tested in vitro (NARST-nalidixic acid resistant Salmonella typhi). These patients need treatment for longer duration with ciprofloxacin or with ceftriaxone.
- Corticosteroids: It is indicated in patients with severe
toxemia, central nervous system manifestations, and DIC. Intravenous dexamethasone is given in the dose of 3 mg/kg as a loading dose, followed by 1 mg/kg every 6 hourly for 24 hours. - Treatment of complications: Intestinal perforation and hemorrhage occur in the third or fourth week of illness are managed accordingly.
Carrier state in typhoid:
- Asymptomatic carrier state: About 3–5% of patients develop long-term asymptomatic carrier state. Many carriers does not give history of typhoid fever and are probably had an undiagnosed mild infection.
- Chronic carriers: These carriers are usually older than 50 years and females with gallstones. S. typhi resides in the gallbladder, urinary bladder and even within the gallstones. They are intermittently excreted into the stool, thereby contaminating water or food. Vi antigen is positive in carriers.
- Chronic carriers should be given ciprofloxacin/ampicillin for 4 weeks. Cholecystectomy may be needed in some patients.
Prevention:
- Improved sanitation and living conditions: It is most important method to prevent typhoid fever. These measures include good hygiene, clean water, proper sewage disposal and proper water treatment. Travelers are advised to avoid drinking untreated water, ice in drinks, and eating ice creams.
- Vaccination: Three available typhoid vaccines are:
- Inactivated injectable: Two in number
- Heat-killed, phenol-extracted, whole-cell vaccine—because of several adverse reactions, this is not used at present.
- Vi-polysaccharide-parenteral administration in individuals >2 years; single dose.
- Oral live attenuated vaccines: Ty21a, a live, attenuated vaccine containing the S. typhi strain Ty21a is oral administered in individuals >6 years; one capsule every other day for three doses.
Food Poisoning:
Question 18. List the causes of food poisoning. Discuss briefly the manifestations, diagnosis, and management of food poisoning.
Answer:
- Food poisoning is an illness contracted by eating contaminated food.
- In most cases, food that causes food poisoning is contaminated by bacteria, such as salmonella or Escherichia coli (E. coli), or a virus, such as the norovirus. Some toxins can cause food poisoning within a short time. Vomiting is the main symptom of food poisoning.
- Each year about 5–33 million foodborne illness occurs.
- Campylobacter cause 1–6 million cases/year. Salmonella causes 2–4 million illnesses/year. E. coli causes about 21,000 cases each year.
Food infection versus intoxication:
Food infection:
- Bacteria are consumed
- Body reacts by raising temperature—fever
- Longer incubation
Food intoxication:
- Toxin contaminated food is eaten
- Shorter incubation
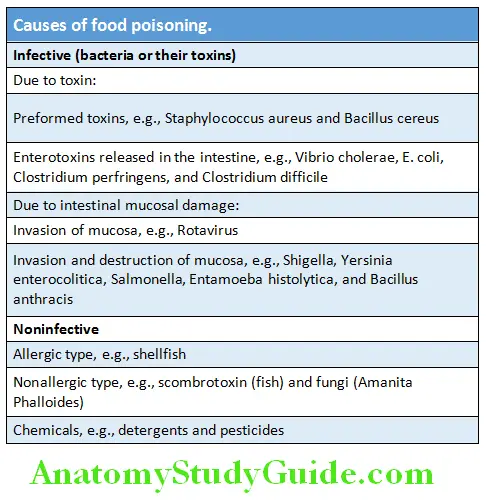
Causes of Food Poisoning:
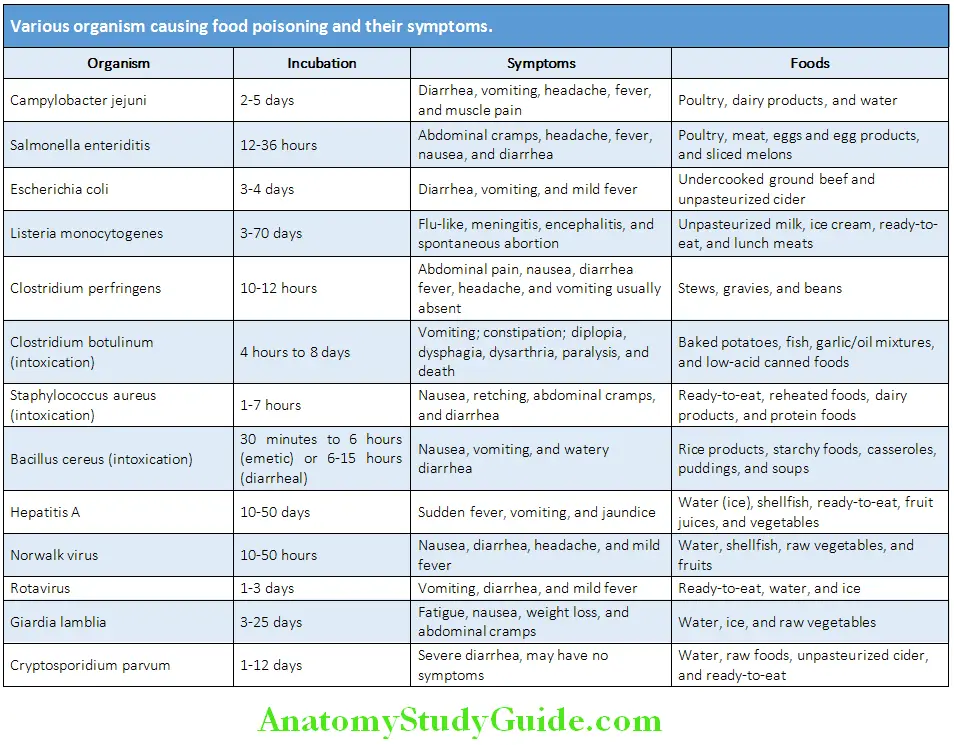
Various organism causing food poisoning and their symptoms are summarized in Table:
Food Poisoning Clinical Feature and Diagnosis:
The symptoms will be different depending on what type of contamination is responsible. Common symptoms of food poisoning are listed in Box 4.
Management:
- The symptoms of food poisoning subside in 2–3 days.
- The goal of treatment is to replace fluids and electrolytes lost through vomiting and diarrhea. If dehydration is severe and cannot be managed at home, patient needs to be admitted, intravenous saline and supportive treatment (antiemetics, antipyretics, probiotics, and antimotility agents) advised.
- Antibiotics to be used sparingly only in infective cases.
Dysentery:
Dysentery is defined as an acute inflammation of the large intestine (colitis) characterized by diarrhea with blood and mucus in the stools. Two causes are bacillary and amebic infections.
Common symptoms of food poisoning:
- Severe vomiting
- Diarrhea
- Headache
- Fever
- Abdominal pain
- Tiredness
Bacillary Dysentery (Shigellosis):
Question 19. Write short essay on the etiology, clinical presentation, diagnosis, and management of acute bacillary dysentery. (Or) Write short essay on clinical features of Shigellosis.
- Bacillary dysentery is an acute necrotizing infection of the distal small bowel and colon caused mostly by one of Shigella species. Shigella species that cause colitis are classified into four major subgroups namely Dysenteriae (most virulent), flexneri, boydii, and sonnei. Bacillary dysentery is one of the most common causes of bloody diarrhea. Other organisms causing bacillary dysentery include E. coli O157:H7, Salmonella, Campylobacter, etc.
- Shigella produces toxin (endotoxin as well as an exotoxin) that has cytotoxic, neurotoxic, and enterotoxic effects. When inflammation is severe, ileus, toxic megacolon, gross hemorrhage, and perforation may develop.
- Source of infection: Humans are the only natural reservoir.
- Mode of transmission: By ingestion through fecal-oral route or via fecally contaminated water and food. It can be acquired by oral contact with any contaminated surface (e.g., clothing, towels, and unwashed hands after defecation or skin surfaces) or flies.
- Incubation period: It ranges from 1 to 3 days.
Bacillary Dysentery (Shigellosis) Clinical features:
- Severity of infection: Disease severity varies from mild to severe. S. sonnei produces mild infection, S. flexneri infection is usually more severe, and S. dysenteriae may produce fulminating infection resulting in death within 48 hours.
- Symptoms start 24–48 hours after ingestion and usually presents as frequent small quantity of stools containing blood, mucus, and purulent exudate with little fecal material (dysentery). This is accompanied by fever, colicky abdominal pain, and tenesmus. Severe cases may show signs of systemic toxicity, dehydration and electrolyte disturbances.
- Physical examination may show tenderness over the colon in the left iliac fossa and hyperactive bowel sounds.
Bacillary Dysentery (Shigellosis) Complications:
Bacillary Dysentery (Shigellosis) Diagnosis:
- Stool culture is required for confirmation of Shigella infection.
- Sigmoidoscopy shows red and swollen mucosa covered by mucopus on the surface. The submucous veins are obscured.
- Enzyme immunoassay used for detecting Shiga toxins in stools.
- PCR for Shigella DNA in stools.
Management:
- Fluid and electrolyte deficits should be corrected by oral rehydration therapy or, if diarrhea is severe, by intravenous replacement of water and electrolyte loss.
- Antibiotic therapy: Infections caused by S. dysenteriae and S. flexneri should be given ciprofloxacin (500 mg twice daily for 3 days).
- Second-line agents include azithromycin and ceftriaxone.
- Antidiarrheal medication should be avoided. Codeine or loperamide may be given to control diarrhea in adults without dysentery.
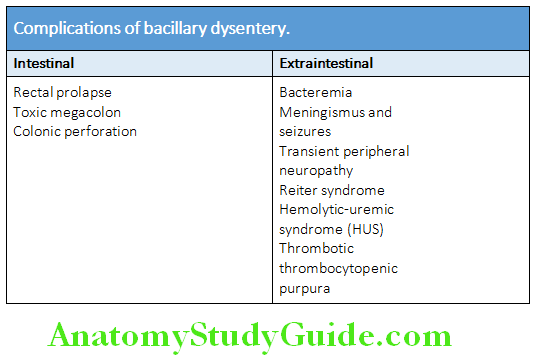
Amebiasis:
Question 20. Discuss the etiology, pathogenesis, clinical features, diagnosis, investigations, complications, and treatment of amebic dysentery/intestinal amebiasis.
Answer:
Amebiasis Etiology:
- Amebiasis is an infection caused by protozoan Entamoeba histolytica
- E. histolytica has three distinct stages:
- Trophozoite stage: Amebic trophozoites are seen in the stools of patients with acute symptoms.
- Precyst stage: In the colon, the trophozoite develops into a cyst through an intermediate form termed the precyst.
- Cyst stage: Amebic cysts are the infecting stage and are found only in stools. They are spherical and have thick chitin walls, and usually four nuclei.
- Source of infection: Humans are the only known reservoir for E. histolytica. It is reproduced in the colon of infected individual and passes in the feces.
- Mode of infection: It is acquired by fecal-oral route through ingestion of materials contaminated with human feces containing E. histolytica. Pathogenesis of amebiasis is depicted.
- Incubation period: About 2–6 weeks.
Amebic Colitis:
Question 21. Write a short note on amebic colitis/intestinal amebiasis. Amebic colitis can present in two forms: Amebic dysentery and nondysenteric amebic colitis.
Answer:
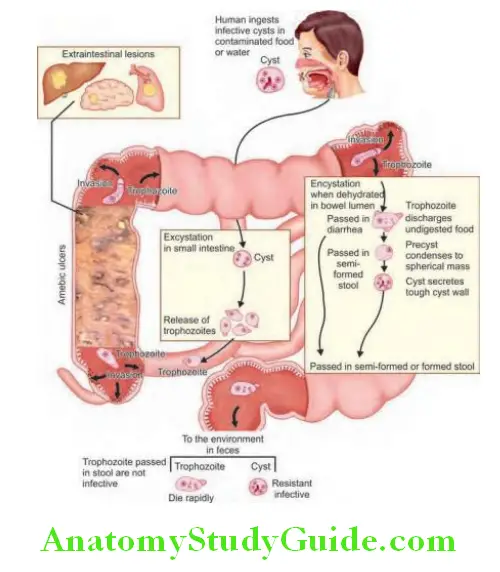
Amebic Dysentery:
Pathogenesis:
- The amebic cysts are passed in the stool of infected individuals and the cysts can contaminate water, food, or fingers. These cysts are tetra-nucleated and can remain viable for weeks to months. However, these are destroyed in temperature below 5°C or above 40°C.
- Amebic dysentery results from ingestion (fecal-oral transmission) of E. histolytica cysts. Amebic cysts colonize the epithelial surface of the terminal ileum and undergo further nuclear division, and release trophozoites. The trophozoites are carried to the large intestine and may colonize any part of the large intestine, but most frequently in the cecum and ascending colon causing amebic colitis. They produce the characteristic “flask-shaped” amebic ulcers with a narrow neck and broad base. These patients pass both cysts and trophozoites in the stool.
- Most often, the amebic infection remains subclinical. However, antibody response usually occurs even without local invasion. These asymptomatic patients should be treated to prevent transmission of infection to others and development of amebic colitis at a later period.
- Ameboma is a common complication of amebic dysentery. It is a localized granuloma which presents as a palpable mass in the rectum or causes a filling defect in the colon on radiography.
- Trophozoite may penetrate blood vessels and reach the liver through portal vein. In the liver they multiply and produce amebic liver abscesses in about 40% of patients with amebic dysentery. The liquid contents of this abscess have a characteristic pinkish color, which may later change to chocolate-brown (like anchovy sauce).
- They can also travel to lungs and brain.
Clinical features of intestinal amebiasis (amebic dysentery):
Question 22. Write short essay on the clinical features and treatment of intestinal amebiasis.
Answer:
- It may produce dysentery of varying severity. It presents with intermittent diarrhea consisting offoul smelling (offensive), loose, and watery stools that may contain mucus and blood. Sometimes diarrhea alternate with constipation.
- Other symptoms include abdominal pain/cramp (especially right lower quadrant which may simulate acute appendicitis), flatulence, and weight loss. Sometimes it is accompanied by systemic symptoms, such as headache, fever, nausea, and anorexia. Less commonly, it may present as acute amebic dysentery, resembling bacillary dysentery or acute ulcerative colitis.
- Physical examination: There may be tenderness over the cecum (amebic typhlitis), ascending colon and over the left iliac fossa (amebic point or Manson–Barr point) and tender hepatomegaly.
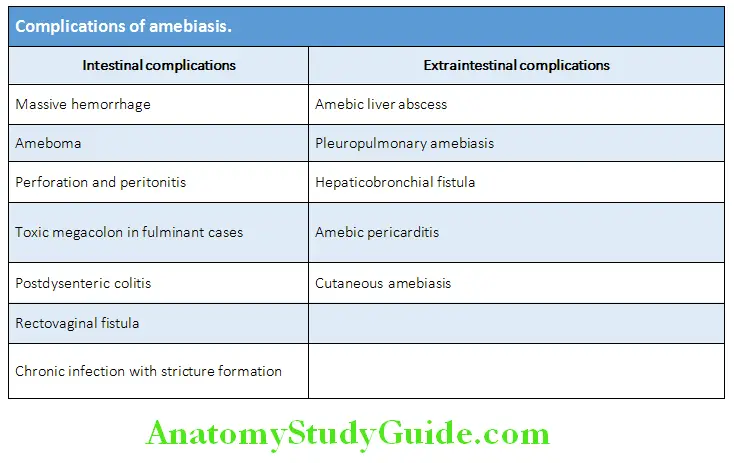
amebic dysentery Complications:
Diagnosis:
- Microscopic examination of stool: Presence of motile trophozoites containing red blood cells (hematophagous trophozoites) in fresh sample of stool will confirm the diagnosis. If a fresh stool sample cannot be examined immediately, it should be preserved with a fixative such as polyvinyl alcohol or kept cool (4°C). Presence of amebic cysts alone does not imply disease.
- Sigmoidoscopy and barium enema may show the characteristic “flask-shaped” ulcers with normal surrounding mucosa. The aspirated material or scrapings from the ulcer or biopsy of the ulcer may show the trophozoites. Colonic exudate obtained by sigmoidoscopy may microscopically show trophozoites.
- Serologic tests: Indirect hemagglutination test, ELISA or counter immunoelectrophoresis can detect antibodies in the blood. They are more useful in extraintestinal amebiasis.
- Amebic fluorescent antibody test is positive in about 90% of patients with liver abscess and in 60–70% with active colitis.
- Detection of E. histolytica antigen or DNA in a stool sample.
- Diagnosis of amebic liver abscess: It is often suspected on clinical grounds.
amebic dysentery Nondysenteric Amebic Colitis:
- Usually presents as recurrent bouts of diarrhea with or without mucus, but without any visible blood.
- Stools examination shows E. histolytica cysts or nonhematophagous trophozoite (trophozoites with no ingested RBCs).
Question 23. Write short note on uses of metronidazole. (or) Enumerate the differences between amebic dysentery and bacillary dysentery.
Answer:
Differences between amebic dysentery and bacillary dysentery are given in Table.
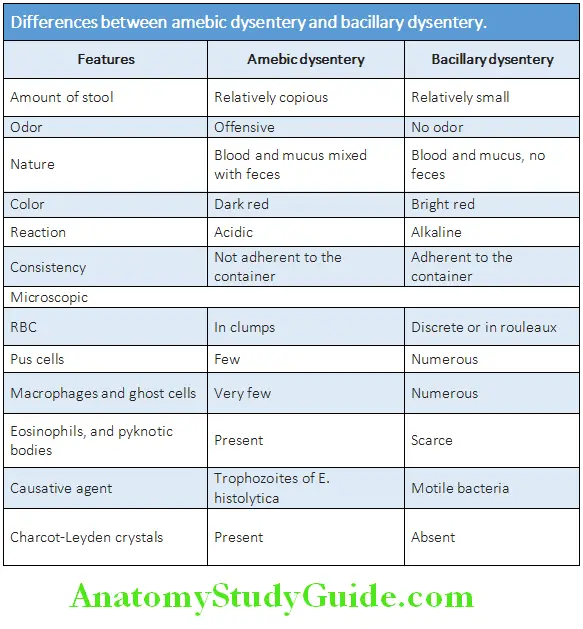
Uses of metronidazole:
- Amebiasis
- Giardiasis
- Trichomonas vaginalis
- Anerobic bacterial infections
- Pseudomembranous enterocolitis
- Ulcerative gingivitis
- Helicobacter pylori gastritis/peptic ulcer
Brucellosis:
Question 24. Write short essay/note on brucellosis (abortus fever; Malta fever; undulant fever; and Mediterranean fever). Name species that cause brucellosis.
Answer:
- Brucellosis (Malta fever, undulant fever, Mediterranean fever, and Rock fever of Gibraltar) is a zoonotic disease.
- Brucellosis is an infection caused by one of the four species of Brucella: Brucella melitensis (goats, sheep, and camels), Brucella abortus (cattle), Brucella suis (pigs), and Brucella canis (dogs).
- Brucella is a gramnegative organism.
- Natural reservoir of brucellosis is animals.
Brucellosis Mode of Infection:
- Ingestion: Infected animals may excrete Brucella in their milk, and human infection is acquired by ingesting contaminated dairy products (especially unpasteurized raw milk) and uncooked meat.
- Direct infection: Animal urine, feces, vaginal discharge, and uterine products may act as sources of infection and they may enter humans through abraded skin.
- Inhalation of infectious aerosols
- Pens, tables, and slaughter houses
- Conjunctival splashes and injection
- Person-to-person transmission is very rare, particularly mother-to-child.
Clinical Features:
Question 25. Write short essay on clinical features and treatment of brucellosis.
Answer:
Brucellosis involves many organ systems and has a very insidious onset with varying clinical signs. The most common sign in all patients is an intermittent/irregular fever with variable duration, which is termed as undulant fever.
Acute brucellosis (<2 months):
- Incubation period of acute brucellosis is 1–3 weeks.
- Acute onset in nearly 50% cases and presents with fever, malaise, headache, chills, fatigue, weakness, backache, generalized myalgia, and night sweats. Patients also have anorexia, lose weight, cough, and arthralgias.
- Fever: It is classically high swinging undulant pattern with rigors, although continuous and intermittent patterns may be
seen. - Physical findings are nonspecific and include splenomegaly (20% of cases may lead to hypersplenism and thrombocytopenia), lymphadenopathy (15% cases) and hepatomegaly (10% cases), sacroiliitis, and arthritis.
- A few may develop localized brucellosis in the form of osteomyelitis, splenic abscess, epididymo-orchitis, pneumonia, meningoencephalitis pleural effusion, and endocarditis.
Subacute brucellosis (2–12 months):
Features are similar to acute form, but less severe.
Chronic brucellosis (>12 months):
- This is characterized by low-grade fever easy fatigability, myalgia, arthralgia, occasional bouts of fever and neuropsychiatric manifestations (e.g., depression).
- Splenomegaly is usually observed.
- Bone and joint complications are the most common complications and include uveitis, sacroiliitis, spondylitis, peripheral arthritis, osteomyelitis, and bursitis.
Brucellosis Diagnosis:
- Culture: Definitive diagnosis depends on the isolation of the Brucella.
- Serological tests may also aid diagnosis and are of greater value in chronic disease. In Brucella agglutination test, a fourfold or greater rise in titer of agglutination antibody (IgM) is highly suggestive of brucellosis. Raised serum IgG level indicates current or recent infection; a negative test excludes chronic brucellosis.
- Other tests include ultrasound and CT, bone radiographs, radionuclide scans, and species-specific PCR tests.
Treatment:
- Regimen A: Doxycycline 100 mg PO BID for 6 weeks + streptomycin 1 g IM qd (means one a day) for the first 14–21 days
- Regimen B: Doxycycline 100 mg PO BID + rifampin 600–900 mg (15 mg/kg) PO qd for 6 weeks
Brucellosis Prevention and Control:
- Careful attention to hygiene when handling infected animals, eradication of infection in animals, and pasteurization of milk.
- No vaccine is available for humans.
Cholera:
Question 26. Write a short essay/note on cholera and cholera sicca.
Answer:
- Cholera is an acute illness caused by the curved, flagellated, halophilic gram-negative bacillus, Vibrio cholerae.
- Vibrio cholerae colonizes in the small intestine. It causes explosive, severe watery diarrhea with rapid depletion of extracellular fluid and electrolytes.
- Vibrio cholerae: It is killed by temperatures of 100°C in a few seconds but can survive in ice for up to 6 weeks. The major pathogenic strain has a somatic antigen (O1) and has two biotypes: classical and El Tor. New classical toxigenic strain, serotype O139 (Bengal serogroup) is still major cause of cholera in our country.
Cholera Mode of Transmission:
By the feco-oral route: Infection spreads via the stools or vomitus of symptomatic patients. Contaminated drinking water is the major source of the dissemination, although contaminated foodstuffs (shellfish and food contaminated by flies), and hands of contact carriers may contribute in epidemics. Incubation period is about 12–48 hours.
Pathogenesis:
- In the small intestine Vibrio cholerae proliferates and produces a powerful exotoxin. The exotoxin causes massive secretion of isotonic fluid into the intestinal lumen.
- Cholera toxin also releases serotonin (5HT, 5 hydroxy tryptamine) from enterochromaffin cells in the gut. This activates a neural secretory reflex in the enteric nervous system.
- V. cholerae also produces other toxins (zona occludens toxin, ZOT, and accessory cholera toxin, ACT) and are responsible for pathogenesis of cholera.
- Severe depletion of extracellular fluid produces hypotension, metabolic acidosis, and hypokalemia.
- In severe cases, massive fluid loss may lead to hypovolemic shock and acute tubular necrosis resulting in renal failure.
Cholera Clinical Features:
- Majority with cholera may have a mild illness that cannot be distinguished clinically from diarrhea due to other infective causes. Severe cases, present abruptly with severe, painless, watery diarrhea without pain or colic, followed by vomiting (vomiting may be absent).
- Rice-water stool: Following the evacuation of normal gut fecal contents, characteristic “rice water” stool is passed, consisting of clear fluid with flecks of mucus.
- Loss of fluid and electrolytes: Leading to intense dehydration with muscular cramps. The features of hypovolemic shock include cold, clammy and wrinkled (“washerwomen” skin) skin with loss of skin turgor, tachycardia, hypotension, and peripheral cyanosis. Features of dehydration include sunken eyes, hollow cheeks, and a diminished urine output. The blood pressure drops and the pulse becomes rapid and thready. Death from acute circulatory failure may occur unless fluid and electrolytes are promptly replaced. Rapid improvement occurs with proper treatment.
- Cholera sicca: Occasionally, a very severe form of the cholera occurs with accumulation of fluid into the dilated bowel. This kills the patient before typical gastrointestinal symptoms appear. It is called as cholera sicca!
Cholera Diagnosis:
- Clinical diagnosis can be easily made during an epidemic. Otherwise, the diagnosis requires bacteriological confirmation.
- Stool examination: “Hanging drop” preparation of the freshly passed stool shows the characteristic rapidly motile (shooting star motility) V. cholerae (this is not diagnostic, as Campylobacter jejuni may give a similar appearance).
- Culture of the stool or a rectal swab can isolate and identify the V. cholerae and also establish antibiotic sensitivity.
Treatment/Management:
Question 27. Write a short essay/note on treatment of cholera.
Answer:
1.Appropriate and effective rehydration therapy: Maintenance of circulation by replacement of water and electrolytes is the mainstay of treatment.
- Intravenous therapy is required if there is severe dehydration. Ringer’s lactate is the best fluid for intravenous replacement and should contain sodium chloride 5 g/L, potassium chloride 1 g/L and sodium bicarbonate (4 g/L) (“Dhaka solution”). Initial fluid deficit should be replaced within 3–4 hours. Patients with severe cholera may need 200–300 mL/kg of isotonic fluids in the initial 24 hours. It should be continued till the patient is hemodynamically stable and vomiting subsides.
- Oral rehydration: Once the vomiting stops and the patient is hemodynamically stable, fluid should be given orally up to 500 mL hourly. Oral rehydration solutions (ORS) are based on the observation that glucose (and other carbohydrates) increases absorption of sodium and water in the small intestine, even in the presence of secretory loss due to toxins.
- Oral rehydration solutions with resistant starch, based on either rice or cereal, shorten the duration of diarrhea and improves prognosis.
- Total fluid needed may exceed 50 L over a period of 2–5 days. Accurate assessment can be made by the use of a “cholera cot”, which has a reinforced hole under the patient’s buttocks, beneath which a graded bucket is placed.
2. Antibiotics: 3 days treatment with tetracycline 500 mg 6 hourly, a single dose of doxycycline 300 mg or ciprofloxacin 1 g in adults or cotrimoxazole one tablet daily reduces the duration or excretion of V. cholerae. It also reduces the total volume of fluid required for replacement. Other alternative drugs include erythromycin and furazolidone.
3. In children, zinc supplementation may reduce the severity of diarrhea. Prognosis is good with adequate treatment and there is gradual return to normal of clinical and biochemical parameters within 1–3 day
Cholera Prophylaxis:
- Vaccination: It is recommended by the WHO during potential or actual outbreak situations.
- Parenteral vaccination with a killed suspension (WC-rBS) of V. cholerae has some protective role.
- Both live attenuated and whole cell killed vaccine (both oral) are available. Oral vaccines containing killed V. cholerae and the B subunit of cholera toxin are of limited efficacy.
- Chemoprophylaxis by tetracycline 500 mg twice a day for 3 days. Mass single-dose vaccination along with tetracycline is also advised.
Leptospirosis:
Question 28. Write short essay on the etiology, clinical features, complications, investigations/laboratory diagnosis, complications, and treatment of leptospirosis. (or) Write a short note on leptospirosis.
Answer:
Leptospirosis is a globally important zoonotic disease caused by the spirochaete Leptospira interrogans.
Synonyms: Autumn fever, seven-day fever, canefield fever, swamp fever, Weil’s disease, rice-field fever, and swineherd’s disease.
Leptospirosis Etiology:
Leptospira consists of two species:
-
- Pathogenic Leptospira interrogans (now designated L. interrogans sensu lato)
- Saprophytic/free-living Leptospira biflexia (now designated L. biflexia sensu lato).
- Leptospirosis is the term used for an infection with any of the subtypes or subgroups of Leptospira interrogans.
Epidemiology:
Leptospirosis is the most widespread important zoonosis in the world.
Leptospirosis Source of Infection:
Leptospirosis is ubiquitous in wildlife and in many domestic animals. Reservoirs of organisms include rodents (most frequent), foxes, skunks, dogs, and domestic livestock. Many animals shed the organism into the urine in massive numbers for long period but infection is asymptomatic in these animals. Outbreaks of leptospirosis may occur with flooding.
Leptospirosis Mode of Transmission:
- Direct contact: Human infection occur either by direct contact with urine or tissue of an infected animal. Transmission may occur through cuts, abraded skin, and mucous membranes (nasopharynx, oral mucosa, conjunctiva, and vagina).
- Prolonged immersion in contaminated water favors invasion, as the spirochete can survive in fresh water for months and for up to 24 hours in sea water.
- Ingestion of contaminated water, soil or vegetation. Incubation period is usually 1–2 weeks (7–14 days) but ranges from 1 to 30 days.
Leptospirosis Pathogenesis:
Typically illness has two phases (biphasic illness)
1. Leptospiremic (initial/first) phase is named so because leptospirae are present in the blood and CSF during this phase. After entry of the organisms into the human, they proliferate and disseminate through blood into all organs (leptospiremic phase). The organisms can survive in the nonimmune host. They evade complement-mediated killing, resist ingestion and killing by neutrophils, monocytes and macrophages.
2. Immune (second) phase: During the immune phase, the antibodies appear and leptospirae disappear from the blood. However, the organism persist in various organs including liver, lung, kidney, heart, and brain.

Clinical Features:
Question 29. Write a short note on the signs and symptoms of Weil’s disease.
Answer:
Most patients with mild leptospirosis are asymptomatic and do not seek medical attention.
1. Leptospiremic phase: It lasts for up to a week. It produces a nonspecific illness and commonly encountered symptoms include:
- Fever: Usually high-grade with chills and rigors. May be
accompanied by malaise and weakness. - Severe headache: Retro-orbital or occipital.
- Myalgia: Muscle pain occurs in most patients and is severe. The muscles of the thighs and lumbar areas (calf and back) show severe tenderness and may show cutaneous hyperesthesia (causalgia).
- Conjunctival suffusion/congestion is very helpful and notable sign for detecting the disease. It is characterized by pericorneal reddening or hyperemia (redness without exudate) without conjunctivitis.
- Other less common features include anorexia, nausea, vomiting, abdominal pain, cough, pharyngitis, uveitis lymphadenopathy, hepatosplenomegaly, and skin rashes.
2. Immune (second) phase:
- In many patients, the first (leptospiremic) phase is followed by a period of apparent recovery, after which the symptoms worsen again for another 2–5 days (immune phase).
- During the second phase, meningitis, and iridocyclitis are more common.
- Meningitis: Presence of headache, fever, and neck stiffness may suggest meningitis. The CSF is usually show normal number of cells in the first 7 days of illness, although the organisms may be seen. With the onset of antibodies (during immune phase) in the serum aseptic meningitis develops in about 90% of patients, with abnormalities in CSF (refer investigations below).
- Other manifestations which are less frequent include change in the level of consciousness, encephalitis, cranial nerve palsies, and acute dilatation of the gallbladder with cholecystitis.
Weil’s Syndrome:
Question 30. Write a short note on Weil’s syndrome.
Answer:
Weil’s syndrome is not a specific subgroup of leptospirosis, but indicates severe leptospirosis. It can develop during the second (immune) phase of leptospirosis or as a progressive illness.
Components of Weil’s Syndrome:
It is dramatic life-threatening event, characterized by:
- Jaundice: The first manifestation of severe leptospirosis is usually jaundice. It develops between fifth and ninth days. Jaundice is deep and is not due to hepatocellular damage, but probably due to cholestasis or sepsis. Hepatic dysfunction such as acute hepatitis and sometimes fulminant may develop.
- Renal failure: Primarily due to impaired renal perfusion and acute tubular necrosis. It presents as oliguria or anuria (acute renal failure) with the presence of albumin, blood, and casts in the urine.
- Hemorrhagic manifestations are common, and include purpura, petechiae appearing on oral, vaginal or conjunctival mucosa, and large areas of bruising. In severe cases, there may be epistaxis, hemoptysis, hematemesis and melena (due to gastrointestinal bleeding), hemorrhage into adrenal glands, pleural, pericardial or subarachnoid spaces (subarachnoid hemorrhage). Thrombocytopenia is common cause of hemorrhagic manifestation.
- Pulmonary syndrome: Adult respiratory distress syndrome (ARDS), pulmonary hemorrhage, pneumonia, and effusion.
- CNS: Meningoencephalitis, subarachnoid hemorrhage, and seizures.
- Other features: DIC, hemolytic anemia, myocarditis and arrhythmias, and multiple organ dysfunction syndrome (MODS).
Weil’s Syndrome Investigations:
- Urine examination:
- During early part of the illness shows microscopic hematuria, pyuria, and proteinuria.
- Demonstration of organism: The tightly coiled spirochete may be visualized in the urine by phase contrast or darkfield microscopy.
- Blood:
- Total leukocyte count may vary, but polymorphonuclear leukocytosis (neutrophilia of more than 70%) with shift to left is very frequent.
- Anemia may develop due to intravascular hemolysis, azotemia, and blood loss caused by hemorrhage.
- Thrombocytopenia in severe infection.
- Elevated markers of inflammation: Raised erythrocyte sedimentation rate and C-reactive protein level.
- Raised blood urea nitrogen (BUN) and hyperkalemia occur with renal failure.
- Coagulation studies may show a prolonged prothrombin time which is reversible with vitamin K administration.
- Creatinine phosphokinase (CPK) are elevated in 50% of patients during the first week of illness. This helps in differentiating leptospirosis from viral hepatitis.
- Liver function tests show raised AST and ALT (up to five times normal), conjugated hyperbilirubinemia and raised alkaline phosphatase. Marked elevations of bilirubin and mild elevated transaminases level are characteristically found in Weil’s syndrome.
- Chest radiograph may reveal patchy bronchopneumonia and a small pleural effusion.
- Electrocardiographic abnormalities include bradycardia, low voltage, and nonspecific ST-T wave changes.
- CSF examination: It may be abnormal in up to 90% of patients.
- Cell counts are increased (but <500/mm 3) with predominance of neutrophils. Protein levels may be normal or raised and glucose level is normal. In severe jaundice, xanthochromia can be seen.
- Serological tests:
- IgM antibodies may be detected in blood by microscopic agglutination test (MAT), during the immune (second) phase of illness. IgM ELISA and immunofluorescent techniques are easy to perform and rapid immunochromatographic tests are specific but are of moderate sensitivity in the first week of disease.
- Demonstration of leptospiral antigen by radioimmunoassay or ELISA.
- Culture: Diagnosis can be confirmed by culture (on Fletcher’s medium) of the blood or CSF during the first week (leptospiremic phase) of illness or of the urine from the second week onwards. It may take several weeks.
- Detection of leptospiral DNA by PCR in blood during early symptomatic disease and in urine from the eighth day of illness. Modified Faine’s Criteria (Table 4.15) includes clinical, epidemiological, and laboratory criteria. A score of more than 26 indicates current leptospirosis.
Weil’s Syndrome Treatment:
Antibiotic therapy: Leptospirosis is highly susceptible to a broad range of antibiotics. Early antibiotic therapy may prevent the development of major organ system failure or lessen its severity, but it should be given irrespective of the stage of the infection.
- Mild disease: Oral treatment with following for 7 days effective when started within 4 days of onset of symptoms.
- Doxycycline (100 mg orally twice daily) or
- Ampicillin (750 mg four times daily)
- Amoxicillin (500 mg four times daily)
- Azithromycin (500 mg once daily)
- Severe leptospirosis should be treated with intravenous penicillin as soon as the diagnosis is considered.
- Intravenous penicillin (1.5 million units intravenous 6 hourly) or
- Ceftriaxone 1 g twice a day IV
- Cefotaxime 1 g IV four times a day
- Treatment of complications/general care of the patient
- Blood product transfusion for hemorrhage, anemia and thrombocytopenia
- ARDS may require mechanical ventilation. Renal failure may require dialysis. Intravenous corticosteroid is useful for the vasculitic nature of severe leptospirosis, particularly in the setting of pulmonary hemorrhage.
Melioidosis:
Question 31. Write short note on melioidosis.
Answer:
Melioidosis (Whitmore disease) is caused by Burkholderia pseudomallei; which is an aerobic, gram-negative motile bacillus, with “safety pin” appearance. It is an opportunistic pathogen found in water and moist soil and produces exotoxin.
Melioidosis Mode of Transmission:
- Contact with contaminated soil or water: It is the most common route of acquiring the disease.
- Other routes include aspiration or ingestion of contaminated water and inhalation of dust from soil. Transmission between infected animals and/or infected people is very rare. Person-to-person transmission may rarely occur through sexual contact with an individual with prostatic infection.
- Incubation period varies from 2 days to months to many years. B. pseudomallei can survive in phagocytic cells. Hence, melioidosis can result after a latent period.
- Risk factors: Immunosuppressive events or chronic diseases, such as diabetes, chronic alcohol use, chronic renal disease, and chronic lung disease.
Melioidosis Clinical Features:
- Most infections are asymptomatic.
- Acute pulmonary infection: It is the most common form which resembles tuberculosis with presentations of lobar or multilobar pneumonia, necrotizing lesions, and pleural effusions.
- Focal infection: Melioidosis can be limited to a focal infection. Localized lesions may occur in the skin (as a result of infected wounds) or various internal organs (as a result of septicemic spread). Many times focal infections become chronic conditions, such as lymphadenopathy, parotitis, cystitis, osteomyelitis, prostatitis, spondylitis, and septic arthritis.
- Severe systemic form: It manifests as severe septicemic form. This can be nondisseminated (only involving one organ) or disseminated. Rarely melioidosis can produce encephalomyelitis.
Melioidosis Diagnosis:
- It is difficult and has been called the great imitator (especially tuberculosis) because there are no pathognomonic lesions. Any organ can be affected and the lesions have no distinguishing characteristics.
- Isolation of the organism from blood, sputum, tissues or wound exudates by culture can help to diagnose the disease.
- Serological tests for titers may also be used for diagnosis. Serological tests available include agglutination tests, indirect hemagglutination, complement fixation, immunofluorescence, and enzyme assays. Improved methods for rapid diagnosis are being evaluated.
Melioidosis Rickettsial Diseases:
- Rickettsiae are obligate intracellular gram negative parasites.
- Most are zoonoses spread to humans by arthropods (except Q fever).
- Rickettsiae replicate within the cytoplasm of endothelial cells and smooth muscle cells of capillaries, arterioles, and small arteries causing necrotizing vasculitis.
- Most are febrile infections with a characteristic rash.
- An eschar is the characteristic lesion that develops at the site of inoculation.
- Among the major group of rickettsioses, the commonly reported diseases in India are:
- Scrub typhus,
- Murine (endemic) typhus,
- Indian tick typhus, and
- Q fever
Melioidosis Treatment of melioidosis:
- Systemic antibiotics: Initial intensive therapy with one of the following regimens:
- Ceftazidime (2 g IV every 6 hours)
- Meropenem (1 g IV every 8 hours)
- Imipenem (1 g IV every 6 hours)
- Addition of TMP-SMX (trimethoprim sulfamethoxazole) twice daily during the intensive phase and to be continued for 2–3 months.
- Surgical drainage of skin wounds can be effective for localized infection.
- Long-term treatment and multiple drugs will be necessary for chronic and severe cases. Relapses, as soon as 6 months after treatment, are common.
Treatment of rickettsial diseases:
- Tetracycline is the drug of choice
- Doxycycline 100 mg BID PO × 7–15 days
- Chloramphenicol 500 mg qid PO × 7–15 days
- IV Chloramphenicol 150 mg/kg per day for 5 days
- Coxiella endocarditis: Combination therapy
- Tetracycline + Cotrimoxazole
- Tetracycline + Rifampicin
Investigations in rickettsial disease:
- Polymerase chain reaction (PCR)
- Serological tests
- Indirect fluorescent antibody (IFA) test (Titer ≥1: 200)
- Complement fixation test
- Weil Felix test
- IgM ELISA test: Highly specific test
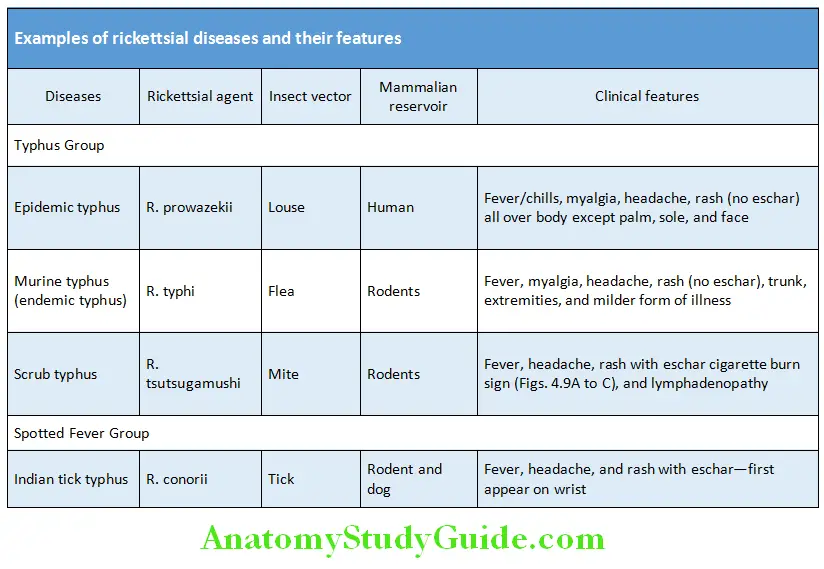
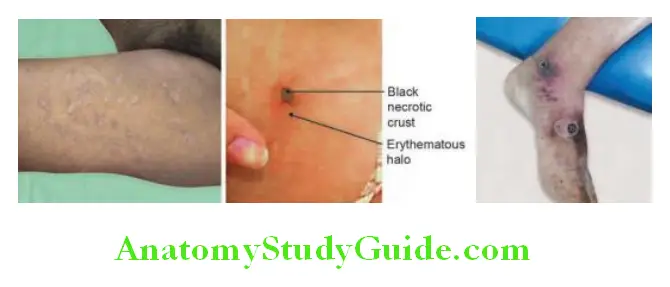
Viral Infections:
Measles (Rubella):
Question 32. Discuss the etiology, clinical features, complications, and prevention of measles (rubella).
Answer:
- Measles virus, the cause of measles is an RNA virus of the genus Morbillivirus in the family Paramyxoviridae.
- Measles is highly contagious; approximately 90% of susceptible household contacts acquire the disease.
- Maximal dissemination of virus occurs by droplet spray during the prodromal period (catarrhal stage).
Viral Infections Clinical Features:
Measles has three clinical stages:
1. Incubation stage
- This stage lasts for about 6–21 days to the first prodromal symptoms and another 2–4 days to the appearance of the rash.
- Period of infectivity is from 4 days before and 2 days after the onset of rash. Patients with compromised immunity can shed virus for the entire duration of illness.
2. Prodromal stage with an enanthem (Koplik spots) and mild symptoms
- Usually lasts 3–5 days and is characterized by low-grade to moderate fever, dry cough, coryza, and conjunctivitis.
Question 33. Write short note on Koplik’s spot.
Answer:
Koplik spots: Pathognomonic sign of measles are grayish white dots, usually as small as grains of sand, that have slight, reddish areolae; occasionally they are hemorrhagic.
- Usually tends to occur opposite the lower molars but can spread irregularly over the rest of the buccal mucosa.
- Rarely, they are found within the midportion of the lower lip, on the palate, described as “grains of salt on a red background” and on the lacrimal caruncle.
- They appear usually within 12–18 hours and disappear rapidly.
- Koplik’s spots begin to slough when the exanthem appears.
- The conjunctival inflammation and photophobia may suggest measles before Koplik spots appear. In particular, a transverse line of conjunctival inflammation, sharply demarcated along the eyelid margin, may be of diagnostic assistance in the prodromal stage.
1. Final stage with a maculopapular rash (exanthem) accompanied by high fever.
- Usually starts as faint macules on the upper lateral parts of the neck, behind the ears along the hairline, posterior parts of the cheek.
- Individual lesions become increasingly maculopapular as the rash spreads rapidly over the entire face, neck, upper arms, and upper part of the chest within approximately the first 24 hours.
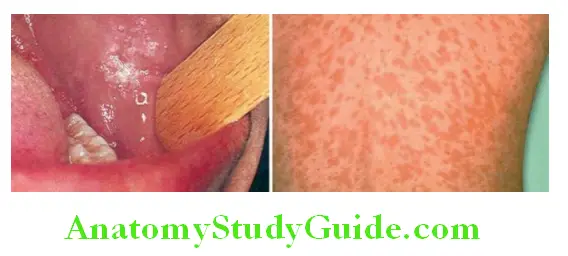
- During the succeeding 24 hours, the rash spreads over the back, abdomen, entire arm, and thighs.
- As it finally reaches the feet on the second to third day, it begins to fade on the face. Associated lymphadenopathy, conjunctivitis, and pharyngitis.
- In hemorrhagic measles (black measles), bleeding may occur from the mouth, nose or bowel.
Complete absence of rash (modified measles) is rare except:
- In patients who have received immunoglobulin (Ig) during the incubation period.
- In some patients with HIV infection.
- Occasionally in infants younger than 9 months of age who have appreciable levels of maternal antibody.
Koplik’s spot Diagnosis:
- Diagnosis is usually apparent from the characteristic clinical picture; laboratory confirmation is rarely needed.
- Testing for measles IgM antibodies is recommended in some situations. Measles IgM is detectable for 1 month alter illness, but sensitivity of IgM assays may be limited in the first 72 hours of the rash illness.
Koplik’s spot Complication:
They are more common in older children and adults.
Treatment:
There is no specific antiviral therapy and it is entirely supportive.
- Antipyretics (acetaminophen or ibuprofen) for fever
- Bed rest
- Maintenance of an adequate fluid intake
- Vitamin A supplementation: Recommended by the American Academy of Pediatrics for children:
- From 6 months to 2 years of age who are hospitalized for measles and its complications.
- Older than 6 months of age with measles and immunodeficiency.
- Recommended regimen as a single dose of:
- 100,000 IU orally for children 6 months to 1 year
- 200,000 IU for children 1 year and above
- Children with ophthalmologic evidence of vitamin A deficiency should be given additional doses the next day and 4 weeks later.
- Treatment of secondary bacterial infections: Prompt identification and treatment of secondary bacterial infections (with appropriate antibiotics).
- Measles encephalitis: Aerosolized and IV ribavirin may be useful.
Koplik’s spot Prevention:
- Passive immunization with intramuscular injection of human normal immune globulin (0.25 mL/kg; maximum: 15 mL) is effective for prevention and attenuation of measles within 6 days of exposure. Indications for passive immunization are mentioned in Table
- Active immunization: It is achieved by giving subcutaneous injection live attenuated measles virus. All children aged 12–15 months should be given measles vaccination. It can be given simultaneously in combination with rubella and mumps vaccines (MMR vaccine). Further MMR dose is given at the age of 4 years. This vaccination offer protection for at least 15 years.
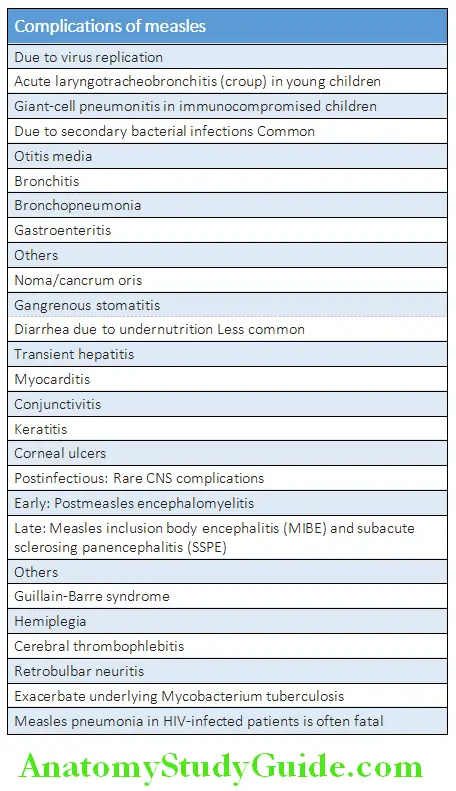

Contraindications for measles vaccine:
- Pregnant women
- Children with primary immunodeficiency
- Untreated tuberculosis, cancer, or organ transplantation
- Those receiving long-term immunosuppressive
therapy - Severely immunocompromised HIV-infected children
Rubella (German Measles):
Question 34. Write short note on
Answer:
- Rubella (German measles, 3 day measles)
- Rubella syndrome and congenital rubella syndrome
- Rubella is infection caused by a Rubella virus which is an enveloped RNA virus and member of the Togaviridae family. The name is derived from the Latin, meaning little red.
- Mode of spread via respiratory droplet infection: Maximum
infectivity from up to 10 days before the onset to 2 weeks after the onset of the rash. - After an incubation period of 14–21 days, the primary symptom of rubella virus infection is the appearance of a rash (exanthema) on the face which spreads to the trunk and limbs and usually fades after three days.
- The skin manifestations are called “blueberry muffin lesions”.
Rubella Clinical Features:
Depends on age of the patient and divided into acquired and congenital rubella. Symptoms are mild or absent in children under 5 years.
1. Acquired rubella
- Prodromal or catarrhal phase: Characterized by malaise, fever, headache, and mild conjunctivitis. Lymphadenopathy (particularly suboccipital and postauricular and posterior cervical lymph nodes) may be observed during the second week after exposure. Forchheimer spots (small petechial lesions on the soft palate) are suggestive but not diagnostic. Splenomegaly may be found.
- Eruptive or exanthematous phase: Occurs within 7 days of the initial symptoms. Characterized by pinkish red, macular rashes first appear behind the ears and on the forehead and then spread downwards to the trunk and limbs.
- These rashes usually last for 2–3 days. May be associated with polyarthritis and generalized lymphadenopathy which
may persist for 2 weeks. - Complications are rare. These include secondary bacterial infection of lung, arthralgia (commoner in females), hemorrhagic manifestations due to thrombocytopenia and postinfectious encephalitis.
2. Congenital rubella syndrome: It is the most serious consequence of rubella virus infection of mother during first trimester pregnancy.
- Mode of spread: From the transplacental transmission of the virus from an infected mother to the fetus.
- Consequences: The complications of rubella infection of the mother include miscarriage, fetal death, premature delivery or live birth with congenital defects. Infants infected with rubella virus in utero may have many defects. Most common are related to the eyes, ears, and heart. This combination of severe birth defects is known as congenital rubella syndrome.
- First trimester infections lead to abnormalities in 85% of cases and greater damage to organs.
- Second trimester infections lead to defects in 16%. > 20 weeks of pregnancy fetal defects are uncommon.
3. Expanded rubella syndrome additionally includes the following manifestations:
- Hepatosplenomegaly
- Thrombocytopenic purpura
- Intrauterine growth retardation
- Myocarditis
- Interstitial pneumonia
- Humoral and cellular immunodeficiency
Manifestations Classical triad of congenital rubella syndrome:
- Cataract
- Cardiac abnormalities
- Deafness

Manifestations Diagnosis:
Diagnosis is clinically made. However, laboratory diagnosis is needed especially in pregnancy.
- Detection of rubella specific IgM by ELISA in serum, confirmed by demonstration of IgG seroconversion (or a rising titer of IgG) in a serum taken 14 days later.
- Detection of viral genome in throat swabs (or oral fluid), urine and the products of conception (in the case of intrauterine infection).
Manifestations Treatment:
- No specific therapy is available.
- Symptom based treatment is given for various manifestations, such as fever and arthralgia.
Manifestations Prevention:
- Passive immunization: Human immunoglobulin can reduce the symptoms but does not prevent the teratogenic effects.
- Active immunization: Active immunization is by giving live attenuated rubella vaccine.
- Aim of vaccination: To reduce the frequency of infection and thereby decrease the chance of exposure to infection by susceptible pregnant females.
- Indications: Should be given to all children at the age of 15 months along with mumps and measles vaccine (MMR vaccine). A second dose is given to young females between the age of 11–13 years and all seronegative females of child-bearing age.
- Contraindications:
- Contraindicated during pregnancy or if there is a likelihood of pregnancy within 3 months of immunization. It is because of likely risk of vaccine-virus induced fetal damage.
- Patients with immune deficiency diseases or those on immunosuppressive drugs.
Mumps:
Question 35. Write short essay/note on mumps and its prevention.
Answer:
- Mumps is an acute systemic viral infection caused by paramyxovirus (RNA virus) and is characterized by swelling of the parotid glands.
- Mode of spread: The virus is transmitted by the respiratory route by droplet infection, by direct contact or through saliva and fomites. Humans are the only natural hosts of the mumps virus. The peak infectivity is 2–3 days before the onset of the parotitis and for 3 days afterwards.
- Incubation period is about 15–20 days (average 18 days).
Clinical Features:
Question 36. Write short essay/note on clinical manifestations of mumps. Primarily affects school-aged children and young adults (peaks at 5–9 years of age) and is uncommon before 2 years of age.
Answer:
- Prodromal symptoms: Nonspecific and includes malaise, low-grade fever, myalgia, anorexia, headache, anorexia, and tenderness at the angle of the jaw.
- Parotid gland enlargement: Prodromal symptoms are followed by severe pain over the parotid glands due to parotitis. Parotitis produces either unilateral or bilateral parotid swelling (75% of cases). It is accompanied by obliteration of the space between the earlobe and the angle of the mandible. The chief complaints at this stage are difficulty in eating, swallowing and talking. Submandibular gland involvement is less frequent.
Mumps Diagnosis:
- On the basis of the clinical features. In atypical cases diagnosis is confirmed by following investigations:
- Serological test: Demonstration of a mumps-specific IgM response in a blood or oral fluid during infection is diagnostic.
- Demonstration virus: Culture of virus or identification by genome (PCR) or antigen detection assays, from saliva, throat swab, urine and CSF.
Complications of Mumps:
Question 37. Write short essay/note on complications of mumps.
Answer:
Treatment:
- Mumps is generally a benign and self-resolving illness.
- Treatment is symptom-based and supportive. Adequate nutrition and mouth care. Analgesics to relieve pain.
Complications of Mumps Prevention:
MMR vaccine: To be given at the age of 15 months

Herpes Viruses Infecting Humans:
Question 38. Write short note on common herpes viruses infecting humans. Enumerate the diseases caused by them.
Answer:
Herpes viruses are double-stranded DNA viruses. The various diseases caused by them are listed in Table
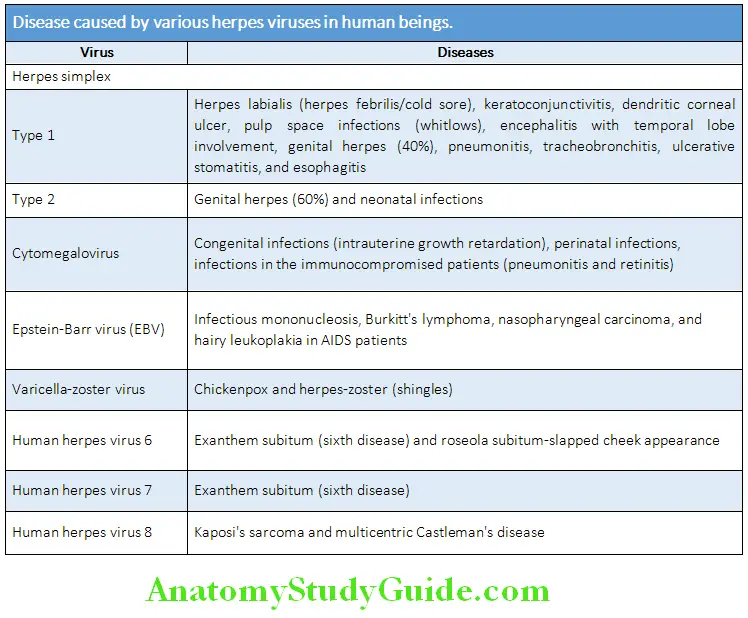
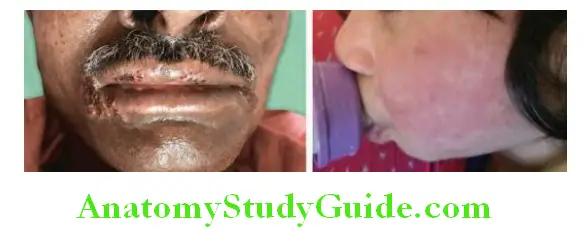
Infectious Mononucleosis (Glandular Fever):
Question 39. Write short essay/note on etiology, clinical features, investigations, complications, and treatment of infectious mononucleosis (glandular fever).
Answer:
Infectious Mononucleosis Etiology:
- Caused by Epstein–Barr virus (EBV). It is a herpes virus that infects and replicates in B lymphocytes in the submucosal lymphoid tissue of nasopharynx and oropharynx.
- Age: Peak incidence in 14–16 years for females and 16–18 years for males. Subclinical infection is very common.
- Mode of transmission is largely through saliva (e.g., kissing hence the nickname kissing disease).
- Incubation period: 7–10 days.
Infectious Mononucleosis Clinical Features:
- Usually presents with nonspecific prodromal symptoms followed by the classical triad of fever, severe pharyngitis, and tonsillitis lymphadenopathy (particularly posterior cervical lymph node enlargement but sometimes generalized) and hepatosplenomegaly.
- Other features: Petechial rashes on palate and maculopapular skin rash, the latter develops in 90% of patients who have received ampicillin or amoxicillin (inappropriately) for the sore throat.
- Gianotti–Crosti syndrome: A symmetrical rash on the cheeks with, multiple erythematous papules, which may coalesce into plaques, and persists for 15–50 days.
Infectious Mononucleosis Investigations:
- Peripheral blood smear: In >90% of, two-thirds are lymphocytes, 20–40% atypical lymphocytes. The atypical lymphocytes are large with larger eccentric and folded nuclei with a lower nuclear-to-cytoplasm ratio.
- Liver function tests: Raised liver enzymes
- Serological tests:
- Demonstration of heterophile antibodies: The following tests will be useful for demonstration of heterophile antibodies.
- Paul Bunnell test is characteristically positive: Sheep red blood cells agglutinate in the presence of heterophile antibodies.
- Monospot test is a sensitive slide test: Horse red cells agglutinate on exposure to heterophile antibodies.
- Demonstration specific antibodies against EBV antigens: These are demonstrated by ELISA.
- Antibody against viral capsid antigens (anti-VCA): These antibodies are initially of IgM type and later of IgG type (which persists for life).
- Antibodies to Epstein–Barr nuclear antigen (EBNA): This can be demonstrated by polymerase chain reaction (PCR).
Infectious Mononucleosis Complications of EBV Infection:
Infectious Mononucleosis EBV-associated Oncogenesis:

Treatment:
- Infectious mononucleosis is usually a self-limiting disease and majority requires no specific treatment and recovery rapidly.
- Symptomatic treatment includes rest, acetaminophen, etc.
- Corticosteroids are indicated when there are neurological complications (e.g., meningitis, encephalitis, severe hemolysis, marked thrombocytopenia, and marked tonsillar enlargement causing respiratory obstruction.
Chickenpox (Varicella):
Question 40. Discuss the clinical features, complications, and management of chickenpox.
Answer:
- Chickenpox is caused by ubiquitous and extremely contagious primary infection with varicella-zoster virus (also called as human herpes virus 3). It is usually a benign illness of childhood (5–9 years of age).
- Varicella zoster virus (VZV) is a dermotropic and neurotropic virus and is a member of the family Herpesviridae.
- It causes two distinct clinical entities: Varicella (chickenpox) and herpes zoster (shingles). Primary infection is chickenpox and usually occurs in childhood. The virus remains latent; which reactivate later in life giving rise to herpes zoster.
Chickenpox Mode of Transmission:
- By droplet infection from the upper respiratory tract.
- Direct contact with discharge from ruptured lesions on the skin. Humans are the only reservoirs of VZV. Chickenpox is contagious till pustules disappear.
- Incubation period is about 10–21 days.
Chickenpox Clinical Features:
- Distribution of skin lesions: Chickenpox presents with characteristic rash on the second day of illness that first appears on the trunk followed by the face, and finally on the limbs. Maximum lesions are found on the trunk and minimum on the periphery of the limbs (centripetal distribution).
- Appearance of skin lesions:
- Rash appear first as small pink macules and progress to papules, vesicles (Fig. 4.12B), and pustules within 24 hours. Finally these lesions dry up and form scabs.
- New lesions develop in crops every 2–4 days and each crop is associated with fever. Thus, lesions at all stages of development are seen in any area at the same time.
- Infectivity lasts from up to 4 days before the lesions appear till the last vesicles crust over.
- In immunocompromised patients the skin lesions are hemorrhagic and are numerous. Dissemination to other organs is quite common.
- Enanthem is rash/small spots on the mucous membranes may also be seen.
Complications of Chickenpox:

Question 41. Write short note on complications of chickenpox/varicella.
Answer:
Chickenpox/varicella Diagnosis:
- Mainly clinical, by recognition of the skin rash.
- If needed, it may be confirmed by:
- Detection of antigen (direct immunofluorescence) or DNA (PCR) of fluid aspirated from vesicles.
- Serology is used to identify seronegative individuals at risk of infection.
- Isolation of virus by culture of vesicular fluid.
- Tzanck smear: Prepared by scraping of the base of the vesicular lesion. It shows multinucleated giant cells and epithelial cells with eosinophilic intranuclear inclusion bodies. Its sensitivity is low (60%).
Question 42. Write short note on treatment of chickenpox/varicella.
Answer:
Chickenpox/varicella Management:
- No treatment is needed in majority of patients.
- Medical management of immunologically normal patient is by prevention of avoidable complications. These include good hygiene (daily bathing), meticulous skin care (to avoid secondary bacterial infection).
- Avoid aspirin use in children to prevent Reye’s syndrome.
- Symptomatic treatment for itching/pruritus includes antihistamines and local calamine lotion.
- Secondary bacterial infection is managed with local antiseptic or systemic antibiotics (e.g., cloxacillin).
Chickenpox/varicella Antiviral Therapy:
- Not required for uncomplicated primary VZV infection in children.
- Drugs used: Antiviral drugs that are used include acyclovir (15 mg/kg by mouth five times daily), valacyclovir (1 g three times daily) or famciclovir (250 mg three times daily) and famciclovir (500 mg TID) for 5–7 days.
- Indications: Though in healthy children, antiviral drugs may reduce the duration of disease when administered within 24 hours of symptoms. However, they are usually not recommended but are indicated in the following cases
- For uncomplicated chickenpox when the patient presents within 24–48 hours of onset of vesicles.
- All patients with complications.
- Immunocompromised patients and pregnant women, regardless of duration of vesicles. More severe disease in immunocompromised patients requires initial parenteral therapy. Immunocompromised patients may shed virus for a prolonged period and may require prolonged treatment.
Chickenpox/varicella Prevention:
Three methods for the prevention of VZV infections are:
1. Live attenuated vaccine: It is given to prevent chickenpox in immunocompetent children and adults who are at a high risk of infection. It is contraindicated in pregnant or immunocompromised patients.
2. Passive prophylaxis using zoster immune globulin (ZIG) or varicella-zoster immune globulin (VZIG):
- It may be given to individuals who are susceptible, those patients having high risk of severe disease or complication of chickenpox (e.g., immunocompromised, steroid treated or pregnant women with history of significant exposure). It should be given within 96 hours of exposure.
- It is given prophylactically to newborn infants, infant whose mother develops chickenpox within 5 days before or within 48 hours after delivery.
3. Antiviral therapy: As prophylaxis to individuals at high risk and not eligible for vaccine or after 96 hours of exposure. They significantly reduce the severity of disease if not completely preventive.
Shingles (Herpes Zoster):
Question 43. Write short essay on clinical features, diagnosis, and management of herpes zoster.
Answer:
Initial infection with VZV produces chickenpox. After this initial infection, VZV persists in latent form in the dorsal root ganglion of sensory nerves or cranial nerve ganglia. Shingles arises from the reactivation of his latent virus later in life.
Shingles Clinical Features:
- Age: It may occur at any age but most common in the elderly.
- Skin lesions: The onset of the skin rash is usually preceded (3–4 days before discrete vesicles occur in the skin) by severe pain (burning discomfort) in the affected dermatome. Though rash is similar to chickenpox, classically they are unilateral and restricted to a sensory nerve (i.e., dermatomal) distribution. This is associated with a brief dissemination of virus causing viremia and can produce distant satellite “chickenpox” lesions. Virus is from freshly formed vesicles may also cause chickenpox in susceptible contacts. Chickenpox may be contracted from a patient suffering from shingles but not vice versa. Occasionally, paresthesia may develop without rash (“zoster sine herpete”).
- Dermatome involved: Most commonly involves thoracic dermatomes or ophthalmic division of the trigeminal nerve (vesicles may develop on the cornea and can produce corneal ulceration leading to blindness). Bowel and bladder dysfunction may develop with sacral nerve root involvement. Occasionally it may cause cranial nerve palsy, myelitis or encephalitis.
Complications of Shingles (Herpes Zoster):
Question 44. Write short note on complications of herpes zoster.
Answer:
- Herpes zoster ophthalmicus, leading to loss of vision
- Ramsay Hunt syndrome: It develops due to geniculate ganglion involvement and characterized by involvement of cranial nerves (V and VII). A triad of
- ipsilateral facial paralysis/palsy (ipsilateral loss of taste and buccal ulceration), ear pain vesicles/rashes in the external auditory canal and auricle is seen. It may be mistaken for Bell’s palsy.
- Postherpetic neuralgia is seen in 10–15% of people with shingles. It results in troublesome persistent pain for 1–6 months or longer, following healing of the skin rash. It is more common in elderly patients.
- Granulomatous cerebral angiitis is a cerebrovascular complication. It leads to a stroke-like syndrome in patients with shingles, especially in an ophthalmic distribution.
Question 45. Write short note on drugs used for the treatment of herpes zoster.
Answer:
Herpes zoster Management:
- Early therapy with acyclovir or related agents reduce both earlyand late-onset pain, especially in patients above 65 years.
- Postherpetic neuralgia: Analgesics along with amitriptyline 25–100 mg daily or gabapentin (start with 300 mg daily and slowly increasing to 300 mg twice daily or more). Capsaicin cream (0.075%) may be of some help.
Human Papillomaviruses:
Question 46. Write short note on human papillomavirus.
Answer:
Human papillomavirus (HPV) is a nonenveloped, double-stranded DNA virus with strong tropism for stratified squamous
epithelial cells of skin and mucous membranes.
Human Papillomaviruses Types of HPV:
- High-risk types cause dysplastic lesions and invasive carcinoma in the cervix (e.g., HPV-16 and HPV-18).
- Low-risk types are responsible for condylomata acuminata (genital wart) and low-grade cervical lesions (e.g., HPV-6 and
HPV-11).
Human Papillomaviruses Mode of Infection:
- Direct contact: Usually mode of infection via direct contact with skin or mucous membrane lesion.
- Sexual contact: Anogenital HPV are usually transmitted sexually. Most important risk factor is number of sexual partners.
- Autoinoculation
- Incubation period for genital HPV varies from weeks to months.
Lesions/Diseases Caused by HPV:
Majority of HPV infections are subclinical and about 80% resolves spontaneously within 12 months.
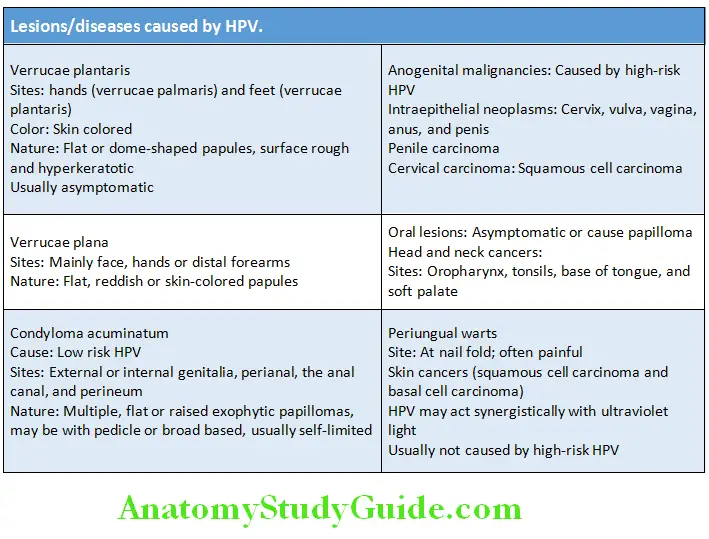
Human Papillomaviruses Diagnosis:
- On clinical presentation and appearance.
- Biopsy: Necessary to confirm dysplasia or carcinoma.
- Acetic acid test: HPV infections can be visualized after application of 3–5% acetic acid with a cotton swab. HPV-positive lesions appear white within 5 minutes.
- Endoscopic examination (colposcopy and proctoscopy): To visualize condyloma, dysplasia, and carcinomas.
- Molecular methods: To detect HPV viral DNA (e.g., hybrid capture two DNA test).
- PCR to detect HPV viral DNA.
Treatment/Therapy
No specific antiviral treatment available
- Verrucae
- Keratolysis: Mechanical removal with a sharp knife and application of salicylic acid: lactic acid: collodion (1:1:4). Vaseline is applied to the surrounding normal skin to prevent its corrosion by chemicals.
- Cryotherapy: Liquid nitrogen is used to freeze warts.
- Anogenital warts:
- Podophyllotoxin: Application causes death of infected cells. It is applied by patient himself at home twice/day for 3 consecutive days in cycles of several weeks. Serious systemic side effects can develop if it is applied to large areas and absorbed systemically. It is contraindicated during pregnancy.
- I miquimod: Induces the production of antiviral cytokines (e.g., interferon and tumor necrosis factor-α). A 5% cream to be applied thrice weekly for up to 4 months.
- Ablation: Either by surgery, laser, and electrocauterization.
- Cryotherapy by using liquid nitrogen.
- Trichloroacetic acid: Topical application of 50–90% trichloroacetic acid produces coagulation of cell protein and cell death.
- 5-Fluorouracil can be applied as cream.
HPV Vaccines:
- HPV vaccines induce high titers of antibodies which can neutralize infectious viruses. They can prevent about 70% of cervical dysplasias (which can progress to cancer if not treated).
- Types of HPV vaccines: Quadrivalent vaccine and bivalent vaccine
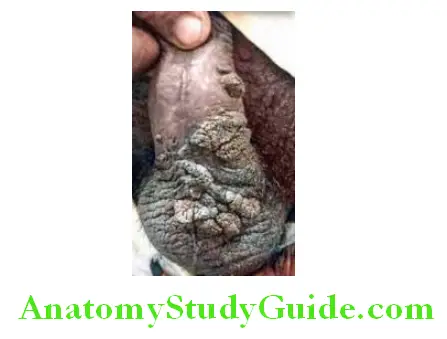
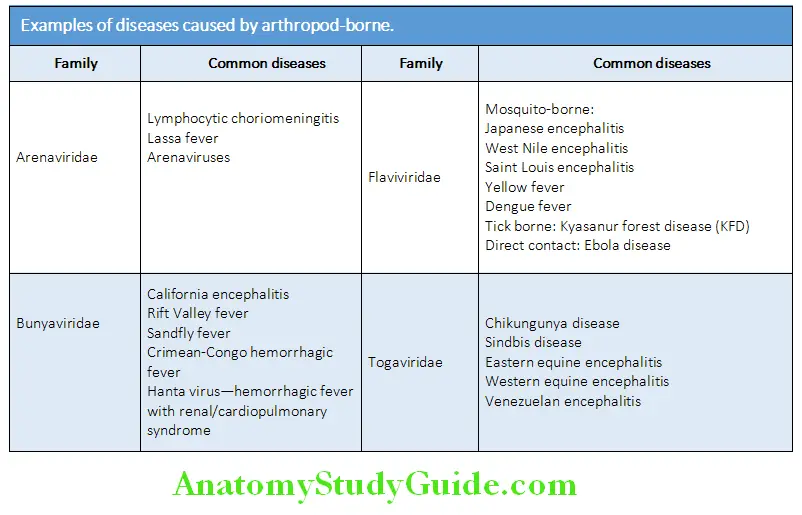
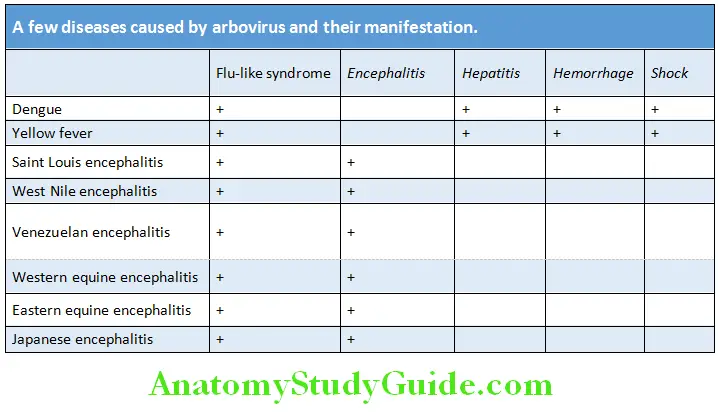
Arthropod-Borne Viruses (Arboviruses):
Question 47. Write short essay/note on
Answer:
- Classifiation of arboviruses
- Major clinical syndromes caused by arboviruses
Arthropod-borne viruses are called as arboviruses. Diseases caused by arboviruses are listed in Table 4.26 and manifestations of a few diseases are listed in Table.
Dengue:
Question 48. Describe classic dengue (break bone fever), dengue hemorrhagic fever (DHF), and dengue shock syndrome (DSS). (or) Write short essay on the clinical features, diagnosis, and management of dengue hemorrhagic fever.
Answer:
Dengue is the most rapidly spreading febrile illness caused by flavivirus and transmitted by mosquitoes. It is the most common arthropod-borne viral infection in humans in the world.
Source of Infection:
Humans suffering from dengue are main amplifying host of the virus and infective during the first 3 days of the illness (the viremic stage). About 2 weeks after feeding (day time) on an infected individual, mosquitoes become infective and remain so for life. Dengue is usually endemic.
Mode of Transmission:
All are transmitted by the daytime Aedes mosquito biting. Aedes aegypti is the most common mosquito involved in transmission which breeds in stagnant water (e.g., collections of water in containers, water-based air coolers, and tire dumps).
Incubation period: 4–10 days following the mosquito bite.
Dengue Classifiation of Dengue:
WHO (2008) classifiation of dengue:
WHO (2008) classifies dengue into nonsevere dengue (with and without warning signs) and severe dengue, the criteria for the same is presented in Figure 4.1Even nonsevere dengue patients without warning signs may develop severe dengue.
WHO (2016) has revised the classification and is presented in Table. Consequences of dengue virus infection are presented. Presently, expanded dengue has been defined and its features are presented.
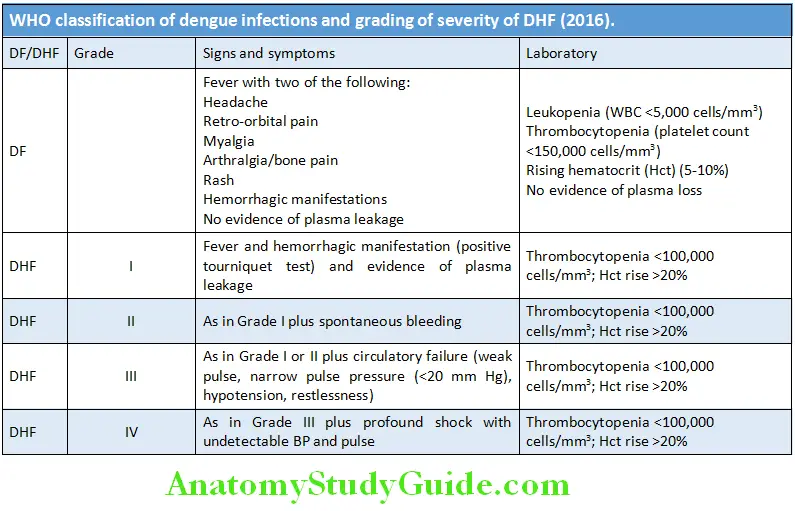
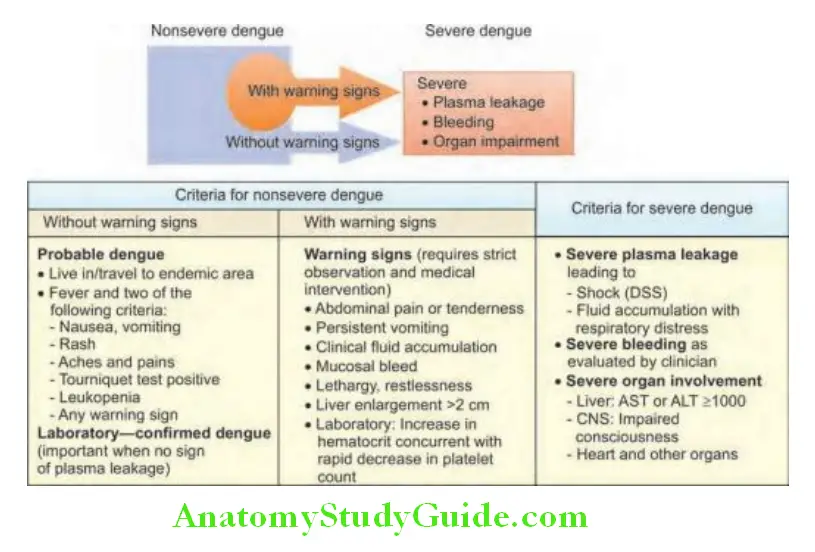

Pathogenesis of Severe Dengue:
- Severe dengue is characterized by plasma leakage, hemoconcentration, bleeding, and organ impairment.
- Plasma leakage: The cause of plasma leakage probably due to functional endothelial cell activation/dysfunction. The endothelial cell dysfunction in turn may be mediated by activation of infected monocytes and T cells, the complement system and mediators, monokines, cytokines, and soluble receptors.
- Bleeding: It may due to thrombocytopenia and associated platelet dysfunction or disseminated intravascular coagulation. Thrombocytopenia may be due to increased destruction or consumption (peripheral sequestration and consumption).
Pathogenesis of Severe Dengue Clinical Features:
Dengue is a systemic and dynamic disease. It has a wide clinical spectrum and includes both severe and nonsevere clinical manifestations. Asymptomatic infections are common (especially in children), but it is more severe in infants and the elderly.
Phases of Dengue:
After the incubation period, dengue begins abruptly and is followed by the three phases:
- Febrile
- Critical
- Recovery.
1. Febrile phase
- Characterized by the abrupt onset of high-grade fever and this phase last for 2–7 days. Fever may be continuous or saddle-back (see below).
- It often accompanied by facial flushing, skin erythema, generalized body ache (“break-bone fever”), myalgia, arthralgia, malaise, headache retrobulbar pain (worsens on eye movements), severe backache (which is a prominent symptom), anorexia, nausea, and vomiting. Some may have sore throat (pharyngitis), upper respiratory tract symptoms, and conjunctival suffusion. In children, high fever may produce febrile convulsions/seizures.
- Saddle-back fever: In few cases fever subsides and other symptoms disappear after 3–4 days. This remission may last for 2 days and then the fever returns together with others symptoms but milder. This biphasic or “saddle-back” pattern is considered characteristic of dengue.
- Mild hemorrhagic manifestations, such as petechiae and mucosal membrane bleeding (e.g., nose and gums) may be found.
- Liver is often enlarged and tender.
Phases of Dengue Laboratory findings:
- The earliest laboratory abnormality which has a high probability of dengue is a progressive decrease in total white cell count.
- Tourniquet test (Hess test):
- Sphygmomanometer (blood pressure) cuff is tied to the upper arm above the elbow and mark a circle of 5 cm diameter on the flexor aspect of forearm.
- The cuff is inflated to 80 mm for 5 minutes.
- Measure the number of petechiae present in the circle (already marked). A test is considered positive when 20 or more petechiae appear in the circle.
2. Critical phase:
- Around the time of falling (defervescence = the period of abatement of fever) of fever (usually on days 3–7 of illness), an increase in capillary permeability along with increase of hematocrit levels may occur. This marks beginning of critical phase.
- Consequences of plasma leakage are:
- Pleural effusion and ascites may develop.
- Shock occurs when there is a critical volume of plasma leakage and is often preceded by warning signs.
- Body temperature may be subnormal when shock occurs.
- Organ impairment: If shock is prolonged, it causes hypoperfusion of organs and leads to progressive organ impairment, metabolic acidosis, and disseminated intravascular coagulation (DIC). This in turn leads to severe hemorrhage and decrease of hematocrit in severe shock. Severe organ impairment (i.e., severe hepatitis, encephalitis or myocarditis) and/or severe bleeding may also develop without obvious plasma leakage or shock.
- Nonsevere dengue: Patients who improve after defervescence are said to have nonsevere dengue.
- Dengue with warning signs: Patients who deteriorate will manifest with warning signs and are said to have dengue with warning signs. Cases of dengue with warning signs usually recover with early intravenous rehydration. However, some cases will progress to severe dengue.
3. Recovery phase:
- If the patient survives the 24–48 hour critical phase, a gradual reabsorption of extravascular compartment fluid takes place during the subsequent 48–72 hours.
- It is characterized by improvement in the general well-being, return of appetite, disappearance of gastrointestinal symptoms, stabilization of hemodynamic status, and diuresis.
- Other features: Some patients may develop a rash of “isles of white in the sea of red”, and some may complain of generalized pruritus (particularly on hands and feet).
- Findings:
- Bradycardia and electrocardiographic changes are common.
- Hematocrit stabilizes or may be low due to the dilutional effect of reabsorbed extracellular fluid (which leaked due to increased vascular permeability).
- White blood cell count: Usually begins to rise soon after defervescence.
- Platelet count recovery is later than that of white blood cell count.
- Consequences of excessive intravenous fluids: If excessive intravenous fluids are administered, massive pleural effusion and ascites may develop leading to respiratory distress. During any phase (critical and/or recovery phases), excessive fluid therapy may cause pulmonary edema or congestive heart failure.
- The various clinical problems during the different phases of dengue are presented in Table

Severe Dengue:
Question 49. Write short note/essay on dengue shock syndrome.
Answer:
According to WHO, severe dengue (Fig. 4.15) is defined by one or more of the following:
- Plasma leakage that may lead to shock (dengue shock) and/or fluid accumulation, with or without respiratory distress
- Severe bleeding
- Severe organ impairment
Severe Dengue Mechanism of severe dengue:
Dengue shock:
- Time of occurrence: Usually develops around defervescence on day 4 or 5 (range 3–7 days) of illness. It is preceded by the warning signs.
- Mechanism of shock: In dengue, progression of increased vascular permeability (due to endothelial dysfunction) leads to worsening of hypovolemia and results in shock.
- Consequences: Initially compensatory mechanisms try to correct the process, but once they fail it goes into the decompensatory phase and organ dysfunction sets in.
- Severe dengue may be associated with coagulation abnormalities, but they are not sufficient to cause major bleeding. When major bleeding develops, it is always associated with profound shock, hypotension,thrombocytopenia, hypoxia, and acidosis. They can lead to multiple organ failure and advanced disseminated intravascular coagulation.
- Most deaths occur in patients with profound shock complicated by fluid overload. Complications of dengue are listed in Box
Complications of dengue:
- Dengue hemorrhagic fever, disseminated intravascular coagulation (DIC), hemophagocytic lymphohistiocytosis (HLH)
- Dengue shock syndrome, polyserositis acalculous cholecystitis, and acute pancreatitis
- Cerebral hemorrhage or edema, cranial nerve palsies, rhabdomyolysis, and hemolytic uremic syndrome
- Hepatitis, encephalitis, myocarditis, aseptic meningitis, GB syndrome, cortical venous thrombosis, pericarditis, atrial fibrillation, heart blocks, and cardiomyopathy
- Vertical transmission (if infection occurs within 5 weeks of delivery).
Diagnosis:
Question 50. Write short note on diagnosis of dengue fever.
Answer:
- Blood: Increasing hemoglobin and hematocrit, leukopenia, thrombocytopenia, and raised live enzymes (AST or ALT).
- Isolation of dengue virus from the blood (within first 5 days of onset of symptoms) by tissue culture.
- Detection of dengue virus RNA by molecular methods like RT-PCR or nucleic acid sequence base amplification (NASBA) during the first few days of illness is diagnostic.
- Detection of serum NS1 antigen by enzyme-linked immunosorbent assay (ELISA) is highly specific but less sensitive than PCR. It is positive early in the course of illness.
- Detection of virus specific IgM antibodies or of rising IgG titers in sequential serum samples (start after 5 days of onset).
- In severe dengue: Chest radiograph to look for pleural effusion, and ultrasound abdomen for ascites.
Question 51. Write short essay on management of dengue fever.
Answer:
Stepwise Approach to the Management of Dengue
At the first levels of care a stepwise approach should be applied as mentioned in Table.

Step I: Overall assessment:
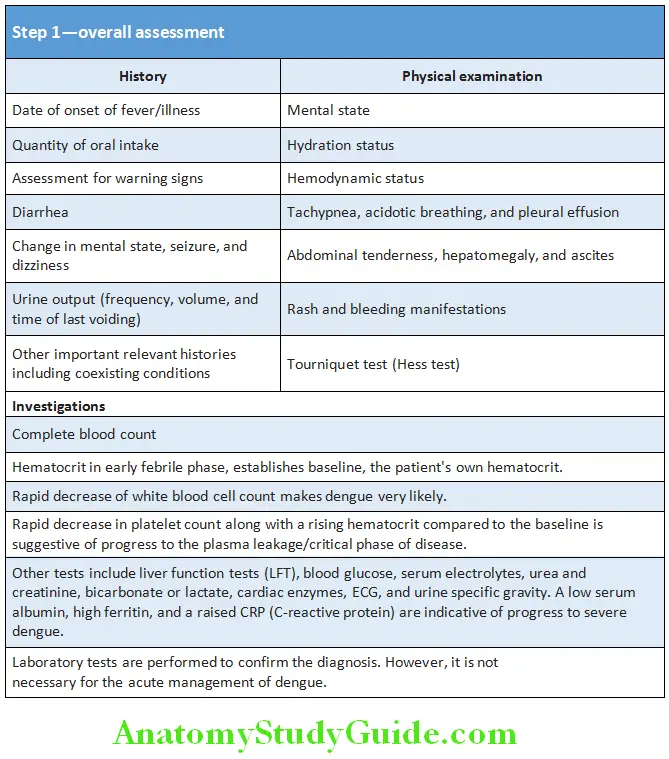
Step II: Diagnosis, assessment of disease phase and severity:
On the basis of step I determine
- Whether the disease is likely to be dengue
- If dengue, which phase (febrile, critical or recovery)
- Whether there are warning signs
- Hydration and hemodynamic status of the patient
- Whether the patient requires admission.
Step III: Management:
Treatment of group A patients (Can be managed at home):
Criteria for group A
- Patients who are able to tolerate adequate volumes of oral fluids and pass urine at least once every 6 hours.
- Do not have any of the warning signs, particularly when fever subsides.
Strategies to be followed:
- Daily review of patients for disease progression (decreasing white blood cell count, defervescence, and warning signs).
Treatment plan:
- Fluid intake: Encourage intake oral rehydration solution (ORS), fruit juice, and other fluids containing electrolytes and sugar to replace losses from fever and vomiting.
- Control of fever: Paracetamol for high fever and if patient is uncomfortable. Tepid sponge if fever is still high. Avoid use of NSAIDs (may aggravate gastritis or bleeding) and aspirin (may develop Reye’s syndrome).
- To be brought to hospital immediately if no clinical improvement or bleeding manifestations develop.
Treatment of group B patients
Patients may require hospital management for close observation, particularly as they approach the critical phase.
Criteria for group B patients:
These include patients with:
- Warning signs
- Coexisting conditions (e.g., pregnancy, infancy, old age, obesity, diabetes mellitus, renal failure, and chronic hemolytic diseases) that may make dengue or its management more complicated.
- Certain social circumstances (such as living alone or living far from a health facility without reliable means of transport).
Treatment plan: Obtain the reference hematocrit before the start of fluid therapy.
Intravenous flid:
- Give only isotonic solutions such as normal (0.9%) saline or Ringer’s lactate.
- Start with 5–7 mL/kg/hour for 1–2 hours, then reduce to 3–5 mL/kg/hour for 2–4 hours, and then reduce to 2–3 mL/kg/hour or less according to the clinical response.
Reassess clinical status and repeat hematocrit:
- If hematocrit remains the same or rises only minimally, continue with the same rate (2–3 mL/kg/hour) for another 2–4 hours.
- If vital signs are worsening and hematocrit is rising rapidly, increase rate to 5–10 mL/kg/hour for 1–2 hours.
- Reassess clinical status, repeat hematocrit and review fluid infusion rates accordingly.
- Monitor hematocrit, platelet count, creatinine, electrolytes, and liver function tests.
Treatment of group C patients:
Criteria for group C patients: These patients require emergency treatment and urgent referral when they are in the critical phase of disease or have severe dengue.
The criteria include patients with severe:
- Plasma leakage leading to dengue shock and/or fluid accumulation with respiratory distress.
- Hemorrhages
- Organ impairment (hepatic damage, renal impairment, cardiomyopathy, encephalopathy or encephalitis)
Treatment plan: All patients with severe dengue should be admitted to a hospital with intensive care facilities and blood transfusion.
Intravenous fluid: Judicious intravenous fluid resuscitation is the essential and usually sole intervention required.
- Replacement of plasma losses should be immediate and rapid with isotonic crystalloid solution or, in the case of hypotensive shock by colloid solutions. If possible, obtain hematocrit levels before and after fluid resuscitation.
Treatment of shock:
- Start intravenous fluid resuscitation with isotonic crystalloid solutions at 5–10 mL/kg/hour over 1 hour. Then reassess the patient’s condition (vital signs, capillary refill time, hematocrit, and urine output).
- If no improvement or shock worsens, fluid infusion rate can be increased. Vasopressors and inotropes need to be added.
Treatment of hemorrhagic complications:
- Avoid intramuscular injections.
- NSAIDs, antiplatelet agents, and anticoagulants to be withheld temporarily.
- Platelets transfusion if platelet count drops below 10,000/mm 3 or patient develops bleeding manifestations.
- If coagulopathy present, transfuse plasma. Pack red cell transfusion if hemoglobin drops below 8 g%. Steroids indicated if hemophagocytosis is diagnosed.
Management of complications appropriately prevention of dengue infections:
Prevention or reduction of dengue virus transmission depends on control of the mosquito vectors or interruption of human-vector contact.
- Control of the mosquito vectors
- Interruption of human-vector contact
- Currently, no vaccine or antiviral drug against dengue viral infections is available.
Chikungunya:
Question 52. Write short essay/note on chikungunya.
Answer:
- Chikungunya is a viral fever caused by an Alphavirus.
- It is usually not fatal. However during 2005–2006 outbreak occurred in India, where more than 200 deaths were reported mainly related to CNS involvement and fulminant hepatitis.
- Chikungunya (in Swahili of African dialect) means “that bends up”. It was used in reference to the stooped posture developed due to the arthritic symptoms of the disease.
Mode of spread: By bites from Aedes aegypti mosquitoes.
Source of infection: Humans are the major reservoir of chikungunya virus.
Incubation period: 2–12 days.
Epidemiology: Observed in Tamil Nadu, Karnataka, Kerala, Andhra Pradesh, Rajasthan, Gujarat, and Madhya Pradesh.
Significant numbers of cases have been reported from northern India including Delhi.
Chikungunya Clinical Features:
Symptoms:
- Fever (often severe): The fever usually lasts for 2–5 days and may be followed by an afebrile phase and then reappearance of fever (saddleback pattern).
- Maculopapular rash: Skin involvement is more common in children and occurs about 40–50% of cases. It may appear at the beginning or several days (day 2 or 3 of the disease) of the illness. It is usually a pruritic maculopapular rash predominating on the trunk and limbs.
- Arthralgia or arthritis: Adults are more prone to migratory polyarthritis, which produces early morning pain and swelling, most often in the small joints of the ankles, feet, hands, and wrists. However, larger joints may also be affected. Arthritis may persist for months and may become chronic (6 months to several years) in patients who are positive for human leukocyte antigen (HLA)-B27.
- Other symptoms: Constitutional symptoms and signs, such as abdominal pain, headache (last for a variable period), anorexia, nausea, conjunctival injection, and slight photophobia. Occasionally, bleeding from skin (petechiae), and epistaxis may occur. Arthritis and skin pigmentation may persist for months to years after convalescence.
Chikungunya Diagnosis:
- Blood: Leukopenia and thrombocytopenia may occur but is uncommon.
- Raised aspartate aminotransferases and C-reactive protein may be found.
- Demonstrating antibodies using ELISA method for diagnosis.
- Molecular methods include RT-PCR to detect structural genes of virus in the blood sample.
Chikungunya Treatment:
- Symptomatic: No specific treatment available. For arthralgias and arthritis NSAIDs (e.g., ibuprofen, diclofenac or naproxen) may be used. Rest is important to reduce morbidity.
- Hydroxychloroquine (200 mg once or twice a day) may be given to patients with prolonged joint pains.
Japanese Encephalitis:
Question 53. Write short note on Japanese encephalitis.
Answer:
- Japanese encephalitis is a mosquito borne encephalitis caused by a Flavivirus.
- Mode of transmission: It is a viral infection transmitted by infected Culex tritaeniorhynchus mosquitoes.
- Source of infection: It is a zoonotic disease maintaining Japanese encephalitis virus in nature through pig-mosquitopig and bird-mosquito-bird cycles. C. tritaeniorhynchus feeds mainly on the infected pigs and birds (e.g., herons and sparrows) and transmits the disease. Humans are accidental hosts.
- Incubation period: 5–15 days after the mosquito bite.
- Epidemiology: It is endemic in Bihar, Uttar Pradesh, Assam, Andhra Pradesh, Karnataka, Tamil Nadu, West Bengal, and Odisha states of India. It is most frequent in the rice growing countries (where irrigated rice fields are present).
Japanese Encephalitis Clinical Features:
Age group: In endemic regions, the infection occurs in children between 3 and 15 years of age because of high background immunity in older individuals.
Presenting Features:
- Prodromal period: Following incubation period, the illness starts with fever, severe rigors, headache, malaise nausea, diarrhea, vomiting, cough, and myalgia that last for 1–6 days. Weight loss is prominent.
- Acute encephalitic stage: During this stage, fever is high, there is neck rigidity, and neurological signs such as irritability, altered consciousness/behavior, hemiparesis, and convulsions develop. Mental deterioration occurs over a period of 3–4 days and may terminate in coma.
- Residual neurological defects features, such as cognitive and speech impairment, emotional liability, cranial nerve palsies (e.g., ocular palsies and deafness), hemiplegia, quadriplegia, and Parkinsonian presentation (extrapyramidal signs in the form of rigidity, dystonia, choreoathetosis, and coarse tremors) occur in about 70% of patients who have had central nervous system (CNS) involvement. Mortality ranges from 7% to 40% and is higher in children.
Japanese Encephalitis Diagnosis:
- Blood: Leukocytosis with neutrophilia
- Cerebrospinal fluid (CSF) examination:
- It shows raised pressure and cell count. During early stages, neutrophils may predominate but a lymphocytic pleocytosis is typical. CSF protein is moderately raised in about 50% of case.
- Virus isolation from CSF
- Detection of viral antigens in CSF by indirect immunofluorescence assay (IFA).
- Serological tests: Detect antibodies to viral antigens. The various tests include virus neutralization test, hemagglutination inhibition, and complement fixation. Antibody detection in serum and CSF by immunoglobulin M (IgM) capture ELISA is a rapid diagnostic test.
- EEG: Persistent abnormalities are common (particularly in children).
- Imaging studies by CT scan may show low-density areas in the temporal lobes. MRI scan is more sensitive in detecting early abnormalities.
Japanese Encephalitis Treatment:
- Treatment is supportive/symptomatic and includes management of fever, raised intracranial tension and convulsions.
Japanese Encephalitis Prevention:
- Control of the mosquito vectors
- Interruption of human-vector contact
- Vaccines: These include vaccines containing purified formalin-inactivated virus derived from mouse brain (requires three doses on 0, 7, and 14 days) and cell-culture derived live attenuated.
Smallpox (Variola):
Question 54. Write short essay on the clinical features, treatment, and prophylaxis of smallpox.
Answer:
- It is a severe disease with high mortality caused by variola virus that belongs to the Orthopoxvirus genus.
- Smallpox was eradicated worldwide by a global vaccination program and the last case was reported in 197In 1980, World Health Organization (WHO) declared world free of smallpox.
- Interest in smallpox has re-emerged because of its potential as a bioweapon. Deliberate introduction of the smallpox virus through aerosolization can produce an epidemic in a few days.
Mode of transmission: Virus spreads through the respiratory or oropharyngeal mucosa. The virus is present in contaminated aerosols, lesional tissue, body fluids, or fomites. It may also spread through contact with lesions (which contains numerous viable virions).
Incubation period: 12–17 days. During incubation period, the virus proliferate within the lymph nodes and subsequently disseminate and seed the other lymphoid tissues throughout the body.
Smallpox Clinical Features:
Prodromal period: It develops abruptly with high fever, headache, backache, prostration, and malaise.
Smallpox Skin Lesions:
- Contagious period: The disease is most contagious during the period from the development of the rash up to the 10th day. However, the patient remains contagious till scabs fall off.
- Within 2–3 weeks, the skin lesions develop into scabs that detach. Sometimes, it may lead to scar formation or become hypopigmented.
- Uncommon presentations are flat and hemorrhagic types of lesions.
- Smallpox: Characterized by soft, flat, confluent, or semiconfluent lesions.
- Hemorrhagic smallpox: Characterized by widespread hemorrhages into skin and mucous membranes with a very high case-fatality rate.
- Vaccination can modify course of disease with milder rash and lower mortality.
- Cause of death: May be due to disseminated intravascular coagulation (DIC), hypotension, and multiorgan failure.
Treatment:
Symptomatic and there is no effective postexposure therapy.
Smallpox Prophylaxis by Vaccine:
- It is a live attenuated vaccine that employs another virus of Orthopoxvirus genus called vaccinia.
- Vaccination can prevent or reduce the severity of infection if given within 4 days of exposure. However, most countries do not have adequate stock of vaccine.
Differences between smallpox and chickenpox are presented in Table
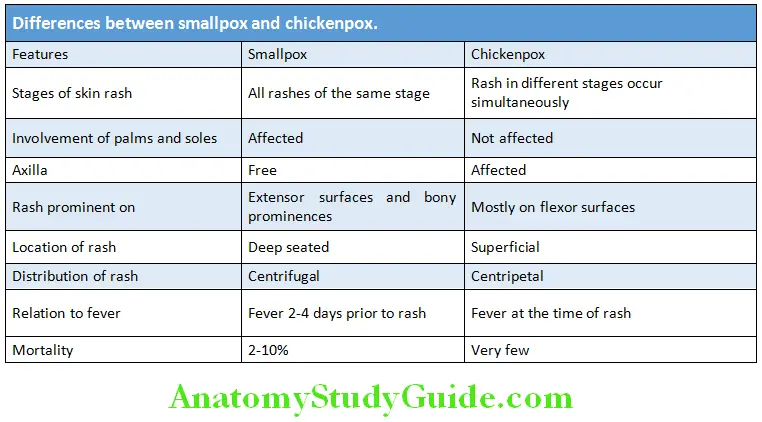
Rabies:
Question 55. Describe etiology, clinical features, and treatment of rabies. Add a note on postexposure prophylaxis.
Answer:
Rabies is caused by a rhabdovirus (genotype 1, single stranded RNA virus of the Lyssavirus genus). The virus has a marked affinity for central nervous tissue and the salivary glands of a wide range of mammals. Established infection is invariably fatal.
Mode of infection: By saliva usually through the bites or licks of an infected animal on abrasions or on intact mucous membranes. Other forms of transmission (aerosolized exposure in bat infested caves, postorgan transplant) are rare.
Source of infection: Humans are usually infected from dogs/fox (rarely cats) and bats.
Incubation period in humans: Varies from few weeks to several months. Unusually average is 1–3 months. In general, severe bites (especially on the head or neck) are associated with shorter incubation periods than those elsewhere.
Rabies Clinical Features:
- Clinical varieties: There are two distinct clinical varieties of rabies in humans:
- Furious rabies (the classic variety)
- Dumb rabies (the paralytic variety).
- The only characteristic feature in the initial prodromal period is the pain and tingling (paresthesia) at the site of the bite.
- There may be fever, malaise, and headache.
- After a prodromal period of 1–10 days, marked anxiety, agitation or depressive features, hallucinations, and paralysis may develop. It may be accompanied by spitting, biting and mania, with lucid intervals in which the patient is markedly anxious.
- Hyperexcitability is the hallmark and is precipitated by auditory or visual stimuli. The characteristic “hydrophobia” (fear of water) develops in 50% of patients. In hydrophobia, though the patient is thirsty, attempts at drinking (or eating) provoke violent/severe contractions of the diaphragm and other inspiratory (pharyngeal) muscles. Aerophobia (fear of air) is pathognomonic of rabies.
- Cranial nerve lesions and autonomic instability are common.
- Examination: It shows hyperreflexia, spasticity, and features of sympathetic overactivity (pupillary dilatation and diaphoresis).
- Patient develops convulsions, respiratory paralysis, and cardiac arrhythmias. Death usually occurs within 10–14 days of the onset of symptoms.
- Dumb rabies presents with a symmetrical ascending paralysis similar to Guillain–Barré syndrome. It commonly develops after bites from rabid bats.
Rabies Investigations/Diagnosis:
- Diagnosis is usually made on clinical grounds.
- Skin punch biopsy: To detect antigen with an immunofluorescent antibody test on frozen section.
- Reverse transcription polymerase chain reaction (RTPCR): Isolation of viral RNA
- Isolation of viruses: From saliva or the presence of antibodies in blood or CSF
- Corneal smear test: It is unreliable.
- Classic Negri bodies: They can be demonstrated at postmortem in 90% of patients with rabies. These are eosinophilic, cytoplasmic, ovoid bodies, 2–10 nm in diameter, found in large numbers in the neurons of the hippocampus and the cerebellum.
- Diagnosis on the biting animal: By using RTPCR, immunofluorescence assay (IFA) or tissue culture of the brain
Question 56. Write a short essay/note on treatment of rabies.
Answer:
Treatment/Management:
Rabies Established Disease:
- Once the CNS disease is established, treatment is symptomatic, as death is virtually inevitable. Only a few patients with established rabies survive.
- Intensive care: The patient should be isolated in a quiet, darkened room. Patients who received some postexposure prophylaxis should be given intensive care facilities to control cardiac and respiratory failure and nutritional support. Only palliative treatment can be given once symptoms have appeared.
- Heavy sedation: The patient should be heavily sedated with diazepam/morphine, supplemented by chlorpromazine if needed. Sedation should be done liberally in patients who are excitable. Nutrition and fluids should be given intravenously or through a gastrostomy.
- Milwaukee protocol using antivirals (ribavirin and amantadine) along with ketamine and midazolam infusion has been tried.
Rabies Prevention:
Pre-exposure prophylaxis:
High-risk individuals: Pre-exposure prophylaxis is indicated to individuals with a high-risk of contracting rabies. These include laboratory workers (who work with rabies virus), animal handlers (who handle potentially infected animals professionally), veterinarians, and those who live at special risk in rabies-endemic areas.
Method:
- Three doses on days 0, 7, and 28 (1.0 mL) of human diploid (HDCV) or chick embryo cell vaccine given by deep subcutaneous or intramuscular route.
- A reinforcing dose after 12 months and additional reinforcing doses are given every 3–5 years (depending on the risk of exposure).
Question 57. Write a short note on postexposure prophylaxis of rabies.
Answer:
Postexposure prophylaxi:

- Treatment of the wound: The wounds should be thoroughly and carefully cleaned with a quaternary ammonium detergent or soap
and water. Excise the damaged tissues, the wound left unsutured and open. - Rabies can be prevented, if treatment is started within a day or two of bite. For maximum protection, hyperimmune serum and vaccine are needed.
- Human rabies immunoglobulin should be given immediately at dose is 20 IU/kg body weight; half is injected/infiltrated around the bite wound and other half is given intramuscularly at a different site from the vaccine. Hyperimmune animal serum can be given but hypersensitivity reactions (including anaphylaxis) are common.
Postexposure prophylaxi Vaccine:
Essen schedule:
- Five dose intramuscular regimen—the course for postexposure prophylaxis should consist of intramuscular administration of five injections on days 0, 3, 7, 14, and 28.
- The sixth injection (D90) should be considered as optional and should be given to those individuals who are immunologically deficient, or at the extremes of age and on steroid therapy.
- Day 0 indicates date of first injection.
Updated Thai Red cross Schedule:
- This involves injection of 0.1 mL of reconstituted vaccine per ID site and on two such ID sites per visit (one on each deltoid area, an inch above the insertion of deltoid muscle) on days 0, 3, 7, and 28
- Human diploid cell strain vaccine (HDCV) is the safest vaccine, free of complications.
- Intradermal (ID) regimen—administration of a fraction of intramuscular dose of certain rabies vaccine on multiple sites in the layers of dermis of skin thereby reducing the cost of active immunization.
The treatment may be modified if animal involved (dog or cat) remains healthy throughout the observation period of 10 days by converting postexposure prophylaxis to pre-exposure vaccination by skipping the vaccine dose on day 14 and administering it on day 28
Ebola Virus Disease:
Question 58. Write short note on the clinical features and management of Ebola virus.
Answer:
Ebola virus disease (EVD), earlier known as Ebola hemorrhagic fever, is a severe, often fatal illness in humans. EVD outbreaks have a very high fatality rate of around 90%. EVD outbreaks occurred primarily in remote villages in Central and West Africa, near tropical rainforests.
Source of infection: Ebola virus is introduced into the human population through close contact with the blood, secretions, organs, or other bodily fluids of infected animals. Humans recovered from the disease can transmit the virus through semen for up to 7 weeks after recovery from illness.
Ebola Virus Disease Mode of Transmission:
The natural host of the Ebola virus is the fruit bats of the Pteropodidae family.
Spread of infection: The virus spreads in the human population through human-to-human transmission, with infection resulting from direct contact (through broken skin or mucous membranes) with the blood, secretions, organs or other bodily fluids of infected people, and indirect contact with environments contaminated with such fluids.
Ebola Virus Disease Clinical Features:
Acute EVD is characterized by sudden onset of high grade fever, associated with severe weakness, muscle pain, headache, vomiting, diarrhea, and skin rash.
Complications include acute renal failure, hemorrhagic manifestations, DIC, hepatitis, and multiple organ dysfunctio syndrome (MODS).
Diagnosis is clinical and serological tests available.
Ebola Virus Disease Treatment:
- Symptomatic and supportive.
- Isolation is of paramount importance.
- Handling of dead bodies, animal carcases needs to be emphasized.
- No vaccine available
Swine Flu:
Question 59. Write short note on Swine Flu.
Answer:
- Swine influenza virus (SIV) refers to influenza cases that are caused by orthomyxovirus endemic to pig populations. SIV strains have been classified either as influenza virus C or one of the various subtypes of the genus influenza virus A.
- In late March and early April 2009, cases of human infection with swine influenza A (H1N1) viruses were first reported in Southern California and near San Antonio, Texas.
- Swine influenza is a respiratory disease of pigs caused by type A influenza virus that regularly causes outbreaks of influenza in pigs that cause high levels of illness but low death rates in pigs. Swine influenza viruses may circulate among swine throughout the year, but most outbreaks occur during the late fall and winter months similar to outbreaks in humans. The classical swine flu virus (an influenza type A H1N1 virus) was first isolated from a pig in 1930.
- The disease originally was nicknamed swine flu because the virus that causes the disease originally jumped to humans from the live pigs in which it evolved. The virus is a “reassortant”—a mix of genes from swine, bird, and human flu viruses.
Swine Flu Source of Infection:
- Droplets from cough or sneeze of an infected individual
- Object contaminated by the cough or touch of an infected person
- Infected individual shed the virus from the day prior to illness onset until resolution of fever. Hence infected individuals should be considered to be contagious up to 7 days from the onset of illness.
Signs and Symptoms:
Patients aged above 65, children below 5 years, pregnant women (especially during the third trimester) and those with underlying medical conditions (e.g., asthma, diabetes, obesity, and heart disease), or those with weakened immune system (e.g., on immunosuppressive medications or infected with HIV) are at high risk of serious complications.
The Center for Disease Control and Prevention (CDC) criteria for suspected H1N1 influenza are listed in Table.
Complications: Adult respiratory distress syndrome (ARDS) and multiple organ dysfunction syndrome (MODS)

Swine Flu Diagnosis:
A confirmed case of novel influenza A (H1N1) virus infection is defined as a person with an influenza-like illness with laboratory confirmed novel influenza A (H1N1) virus infection by one or more of the following tests.
- Real-time RT-PCR
- Viral culture from swabs (nasal, oral, and secretions of respiratory tract) can be used for virus isolation.
Treatment:
Mainly supportive and consists of bed rest, increased fluid consumption, cough suppressants, and antipyretics and analgesics (e.g., acetaminophen and nonsteroidal anti-inflammatory drugs) for fever and myalgia.
- Severe cases may need intravenous hydration and other supportive measures.
Swine Flu Antiviral Agents:
- They may also be considered for treatment or prophylaxis.
- Neuraminidase inhibitors: They inhibit neuraminidase which is a glycoprotein on the surface of influenza virus that destroys an infected cell’s receptor for viral hemagglutinin. By inhibiting viral neuraminidase, neuraminidase inhibitor agents decrease the release of viruses from infected cells and, thus, decrease the spread of virus. Drugs include oseltamivir and zanamivir. Both are effective against both influenza A or B.
1. Oseltamivir (Tamiflu)
- Must be administered within 48 hours of symptom onset to provide optimal treatment.
- Adult dose:
- Treatment for acute illness: 75 mg PO BID for 5 days
- Prophylaxis: 75 mg PO qd (please refer to duration of prophylaxis specific for postexposure)
2. Zanamivir (Relenza)
- It is in powder form for inhalation via the Diskhaler oral inhalation device.
- Adult dose:
- Treatment for acute illness: 10 mg inhaled orally BID for 5 days
- Prophylaxis of household contact: 10 mg inhaled orally qd for 10 days (initiate within 36 hours)
- Prophylaxis for community outbreak: 10 mg inhaled orally qd for 28 days (initiate within 5 days of outbreak)
Other antiviral agents (e.g., amantadine and rimantadine) are not recommended because of recent resistance to other influenza strains.
Swine Flu Vaccination: Yearly vaccination with season specific strain vaccine before the annual flu season is recommended. Two types of vaccination are available namely intranasal and intramuscular.
Monovalent/trivalent vaccines are available.
Zika Virus:
Question 60. Write short note on Zika virus.
Answer:
- Zika virus (ZIKV) is an arbovirus, belongs to the Flavivirus genus.
- It is enveloped and icosahedral with a nonsegmented, single-stranded, positive-sense RNA genome. It was first identified in Zika forest of Uganda in 1947.
Zika Virus Mode of Spread:
- Zika virus is transmitted by Aedes mosquito species, such as A. aegypti, the extrinsic incubation period in mosquitoes is about 10 days. Rarely, mother to child transmission and one case of sexual transmission have been reported.
- The vertebrate hosts of the disease are monkeys and humans. The virus infects dendritic cells near the site of inoculation, and later spreads to lymph nodes and produces viremia.
Incubation period: 10 days.
Zika Virus Clinical Features:
It produces symptoms similar to a mild form of dengue fever with mild headaches, maculopapular rash, fever, malaise, conjunctivitis, and arthralgia. Within 2–4 days, the rash started fading, and fever resolved within 3 days.
Neurological complications: Guillain–Barré syndrome and other neurologic complications
Congenital infection leads to microcephaly, chorioretinal atrophy, hydranencephaly, intrauterine growth restriction (IUGR), and hydrops fetalis. In January 2016, the US Centers for Disease Control and Prevention (CDC) issued travel guidance on affected countries, including the use of enhanced precautions, and guidelines for pregnant women including considering postponing travel to affected areas. Most dangerous time is during first trimester of pregnancy and damage to the brain of fetus.
Zika Virus Diagnosis:
- Sample to be tested: Blood saliva and urine
- PCR: Useful in the first 3–5 days after the onset of symptoms. It directly detects the virus or specific viral antigens in the clinical specimen.
- Serology test: Detects the presence of antibodies and useful only after 5 days.
Treatment:
Only symptomatic. No specific antiviral drugs or vaccines are available.
Prevention and Control:
- Avoid travel to areas with an active infection
- Control of mosquito
Molluscum Contagiosum:
Question 61. Write short note on molluscum contagiosum.
Answer:
It is caused by a DNA poxvirus called the molluscum contagiosum virus (MCV).
MCV has no nonhuman-animal reservoir (infecting only humans).
Types: Four types of MCV named MCV-1 to 4; MCV-1 is the most prevalent and MCV-2 is seen usually in adults.
Mode of transmission: The virus spreads from person to person by touching of the affected skin. The virus also spreads by touching a surface with the virus on it, such as a towel, clothing, or toys.
Age group: Children and young adults are infected and also commonly seen in HIV patients.
Appearance of lesions: Lesions begin as small (3–6 mm) papules that are smooth, flesh-colored domes with a central dimple (umbilication). Inside the papule is a white, curd-like core that can be easily expressed. Occasionally, they may be complicated by secondary bacterial infections.
Site of lesions:
- It can occur anywhere on the skin and mucous membranes, but are usually grouped in one or two areas. Occasionally, they may be widely disseminated.
- Common sites: Head, eyelids, trunk, and genitalia (predominant site in adults).
Treatment: Imiquimod and retinoids have been used. Cryosurgery, curettage have also been used in the treatment
Nipah Virus Encephalitis:
Question 62. Write a short note on Nipah virus encephalitis.
Answer:
Nipah Virus Encephalitis Etiology:
It is caused by a RNA virus of the family Paramyxoviridae, genus Henipavirus first identified in 1999 outbreak, has caused annual outbreaks in Bangladesh, Malaysia, and India. Recent Indian outbreak has occurred in north Kerala in May 2018 has claimed lives.
Nipah Virus Encephalitis Transmission:
The Nipah virus is a zoonotic virus, which means it spreads to humans from either air or through saliva. The fruit bats are the primary carriers of Nipah virus. The virus is usually transferred through fluids from the bat. Close contacts with infected pigs and cattle can transmit the disease.
Nipah Virus Encephalitis Clinical Features:
- The incubation period ranges from 5 to 14 days. The illness presents with high grade fever and headache, followed by drowsiness, disorientation, and mental confusion. This can progress to encephalitis with brainstem dysfunction and coma within 24–48 hours.
- Other neurological signs include seizures, myoclonus, meningismus, autonomic dysfunction, and cerebellar involvement. One-third of patients develop pulmonary signs (ARDS), gastrointestinal, and renal involvement is seen in 5% of cases.
Nipah Virus Encephalitis Investigations:
- Thrombocytopenia, leukopenia, and elevated transaminases are seen in majority of patients.
- Diffuse bilateral lung infiltrates with basal atelectasis are seen in many cases.
- CSF shows lymphocytic pleocytosis.
- Serum/CSF immunoglobulin (Ig) M capture enzyme immunoassay (EIA) for detection of Nipah IgM antibodies and an indirect EIA for Nipah IgG antibodies are done for the specific diagnosis.
Treatment and Prognosis:
- Treatment is supportive only.
- The disease has a fatality rate of around 40%.
Nipah Virus Encephalitis Prevention:
- Stay away from pigs, bats, or domestic animals in endemic areas.
- Do not drink raw date, palm sap.
- Avoid fruits that have fallen from trees on the ground.
Fungal Infections:
Candidiasis (Moniliasis):
Question 63. Write short note on candidiasis (moniliasis).
Answer:
Infection of skin or mucous membranes (e.g., oral cavity and vagina) by Candida is known moniliasis.
- Most Candida infections occur when the normal commensal flora breach the skin or mucosal barriers. Candida resides normally in the skin, mouth, gastrointestinal tract, and vagina. Candida species usually live as benign commensals and do not cause disease in healthy individuals. Candida species (usually C. albicans) are the most common cause of human fungal infections. Candidas are small asexual fungi. Candida species are the most common nosocomial pathogens. Candida albicans is the most common cause of candidiasis.
- Candidiasis may be superficial (noninvasive) or invasive (disseminate). Invasive candidiasis can also be due to nonalbicans candida species (e.g., C. glabrata) and some of these are resistant to fluconazole.
Lesions/Conditions Caused Due to Candida:
- Oral thrush: It is characterized by white adherent, painless, discrete, or confluent patches in the mouth. Conditions that predispose to oral candida infection include use of broad-spectrum antibiotics, xerostomia, immune dysfunction (e.g., diabetes, immunosuppressive therapy, HIV infection, etc.) or the presence of removable prostheses and lichen planus.
- Vulvovaginal candidiasis: More common in diabetics. It presents with pruritus, pain, and vaginal discharge which is usually thin but may appear as whitish “curds” in severe cases.
- Cutaneous candidiasis: It may present as intertriginous infection (in the skin folds), paronychia (painful swelling at the nail-skin interface) balanitis (infection of the glans penis), pruritus ani (infection surrounding the anus), vulval candidiasis (in diabetic females), and scrotal candidiasis.
- Chronic mucocutaneous candidiasis: It is a heterogeneous infection of the hair, nails, skin, and mucous membranes which persists in spite of intermittent therapy. The onset is usually in infancy or within the first two decades of life. It may be mild and limited to a specific area of the skin or nails or may be severe form (Candida granuloma). It occurs in immunocompromised patients with leukemias, lymphomas and AIDS.
- Deeply invasive candida infections: It may or may not be due to hematogenous spread.
- Esophageal candidiasis: Deep esophageal infection may result from penetration by organisms from superficial esophageal erosions. It present as a dysphagia and retrosternal pain.
- Candidiasis of the urinary tract: It presents with hematogenous renal abscess and bladder thrush. Kidney infection may develop from catheter.
- Hematogenous dissemination of candida (invasive candidiasis or candidemia): Candida may enter into the intravascular compartment either from the gastrointestinal tract or, less often, from the skin through the site of an indwelling intravascular catheter. Then it may spread hematogenously to deep organs, such as brain, chorioretina, heart, and kidneys. It presents with retinal abscesses (extending into vitreous), pulmonary candidiasis, endocarditis, chronic meningitis, and arthritis.
Fungal Infections Diagnosis:
- Visualization of hyphae or pseudohyphae on wet mount (saline and 10% KOH) preparation or in scrapings from infected lesions under microscope.
- Tissue secretions stained with Gram’s stain, periodic acid-Schiff stain, or methenamine silver stain may show candida in association with inflammation.
- In invasive disease from blood cultures.
Treatment: Depends on the site and severity of infection.
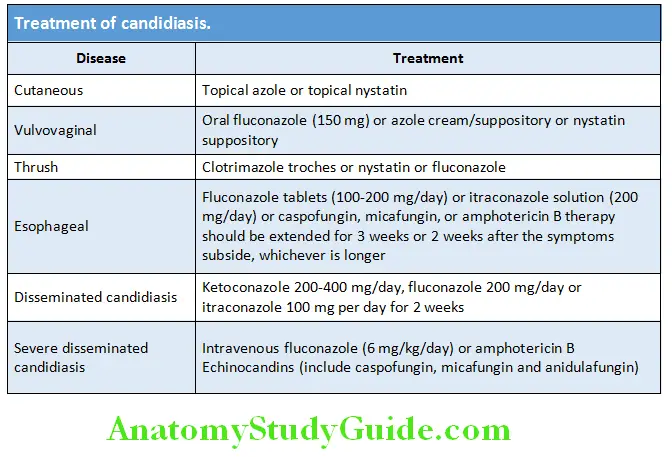
Superficial mycoses along with their etiologic agent are as follows:
- Pityriasis versicolor: Malassezia furfur
- Black piedra: Piedraia hortae
- White piedra: Trichosporon beigelii
- Tinea nigra: Exophiala werneckii
Superfiial Mycoses:
Question 64. Write short note on superficial mycoses and dermatophytes.
Answer:
Superficial mycoses are fungal infections of the outermost keratinized (cornfield) layers of the skin or hair shaft resulting in essentially no pathological changes.
Pityriasis Versicolor:
Superfiial Mycoses Etiology: Malassezia furfur (Pityrosporum orbiculare) which is a lipophilic yeast.
- Normal flora of the superficial epidermis and clusters around the openings of hair follicles.
- Saprophytic on normal skin of trunk, head, neck and appears in highest numbers in areas with increased sebaceous activity. Causes superficial chronic infection of stratum corneum.
Predisposing factors: Malnutrition, burns, corticosteroid therapy, immunosuppression, depressed cellular immunity, excess heat, and humidity. It is associated with increased sweating.
Superfiial Mycoses Clinical presentation:
- Most common in adolescent and young adult males.
- Multiple small, asymptomatic, red to fawn-colored, circular, macules, patches, or follicular papules. It is hypopigmented. There may be tan to dark brown macules and patches.
- Most common site: Back, underarm, upper arm, chest, neck, and occasionally on face.
- Microscopy of skin scrapings shows “spaghetti and meatballs” hyphae. Lesions fluoresce greenish yellow in Wood’s light.
Treatment: Topical selenium sulfide or 2% ketoconazole shampoo (apply and remove after 30–60 minutes and repeat daily for 7 days) or a topical imidazole cream (twice daily for 10 days). Oral ketoconazole or oral itraconazole (100 mg twice daily for 1 week) in resistant cases.
Altered pigmentation persists for months even after successful treatment.
Superfiial Mycoses Dermatophytes:
- Cutaneous fungi are called dermatophytes which are keratinophilic fungi—they possess keratinase allowing them to utilize keratin as a nutrient and energy source.
- They infect the keratinized (horny) outer layer of the scalp, glabrous skin, and nails causing tinea or ringworm by secreting keratinase—which degrades keratin with varied clinical manifestations and are caused by species of the fungal genera Trichophyton, Epidermophyton, and Microsporum (in order of commonality).
- Lesions on skin and sometimes nails have a characteristic circular pattern that was mistaken by ancient physicians as being a worm down in the tissue.
- These lesions are still today called ringworm infections even though the etiology is known to be a fungus rather than a worm.
Clinical manifestations of ringworm infections are called different names on basis of location of infection sites.
Superfiial Mycoses Kerion:
- Inflammatory reaction of tinea capitis caused by Microsporum canis or Trichophyton mentagrophytes.
- Presents as boggy indurated swellings with crusting and loose hairs.

- Hair follicles may discharge pus.
- In extensive lesions, fever, pain, and regional lymphadenopathy are present.
- Kerion may be followed by scarring and alopecia in areas of inflammation and suppuration.
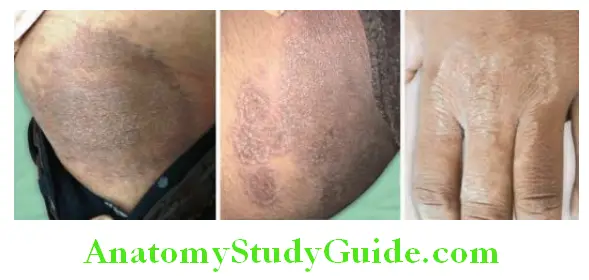
Favus:
- Caused by Trichophyton schoenleinii
- Characterized by the presence of yellowish, cup-shaped crusts known as scutula. Each scutulum develops round a hair, which pierces it centrally. The scutula have a distinctive mousy odor.
- Cicatricial alopecia is usually found in long-standing cases.
Favus Treatment of Mycoses:
- Topical: Miconazole, clotrimazole, econazole, and terbinafine
- Oral: Griseofulvin, ketoconazole, itraconazole, and terbinafine
Opportunistic Mycoses:
Question 65. Write short essay/note on opportunistic mycoses.
Answer:
- Occurs in humans with a compromised immune system.
- Causative agents are normal resident flora that becomes pathogenic only when the host’s immune defenses are altered, as in immunosuppressive therapy, in a chronic disease (e.g., diabetes mellitus), or during steroid or antibacterial therapy that upsets the balance of bacterial flora in the body.
Opportunistic Mycoses Etiology:

Opportunistic Mycoses Aspergillosis:
- Aspergillus species are ubiquitous saprophytes in nature.
- In nature >300 species of Aspergillus exist, few are important as human pathogens. These include
- A. fumigatus
- A. niger
- A. flavus
- A. terreus
- A. nidulans.
Opportunistic Mycoses Clinical Syndromes:
The Aspergillus species can cause a variety of clinical syndromes:
- Pulmonary aspergillosis: These include allergic asthma, bronchopulmonary aspergillosis, and aspergilloma.
- Invasive aspergillosis
- Superficial infections: Sinusitis, mycotic keratitis, and otomycosis
Treatment: Invasive aspergillosis is treated with intravenous amphotericin B, voriconazole.
Opportunistic Mycoses Mucormycosis:
It represents a group of life-threatening infections caused by fungi of the order Mucorales.
Rhizopus oryzae (in the family Mucoraceae) is by far the most common cause of infection.
Risk Factors:
- Impaired immune system: Mucormycosis typically causes infection primarily in patients with diabetes or defects in phagocytic function (e.g., associated with prolonged neutropenia or glucocorticoid treatment), solid organ or hematopoietic stem cell transplantation (HSCT), or malignancy. Post COVID-19, lots of cases have been reported.
- Elevated levels of free iron: Supports fungal growth in serum and tissues. Hence patients with raised levels of free iron also have increased risk. In iron-overloaded patients with end-stage renal failure, treatment with deferoxamine (iron chelator for the human host) predisposes to the development of rapidly fatal disseminated mucormycosis. Deferoxamine serves as a fungal siderophore, directly delivering iron to the mucorales.
Risk Factors Clinical Categories:
Mucormycosis can be divided into at least six clinical categories based on clinical presentation and the involvement of a particular anatomic site.
These are:
- Rhinocerebral (Most Common In Diabetics),
- Pulmonary (In Patients Undergoing Hematopoietic Stem Cell Transplant)
- Cutaneous
- Gastrointestinal
- Disseminated
- Miscellaneous.
Rhinocerebral Mucormycosis:
- The initial symptoms of rhinocerebral mucormycosis are nonspecific and include eye or facial pain and facial numbness followed by the onset of conjunctival suffusion, blurring of vision, and soft tissue swelling.
- Fever may be absent in up to half of cases, while white blood cell counts are typically elevated as long as the patient has functioning bone marrow.
- If untreated, infection usually spreads from the ethmoid sinus to the orbit, resulting in compromise of extraocular muscle function and proptosis, typically with chemosis.
- Onset of signs and symptoms in the contralateral eye, with resulting bilateral proptosis, chemosis, vision loss, and ophthalmoplegia, is ominous and suggests the development of cavernous sinus thrombosis.
- Upon visual inspection, infected tissue may appear to be normal during the earliest stages of fungal spread and then progresses through an erythematous phase, with or without edema, before the onset of a violaceous appearance and finally the development of a black necrotic eschar
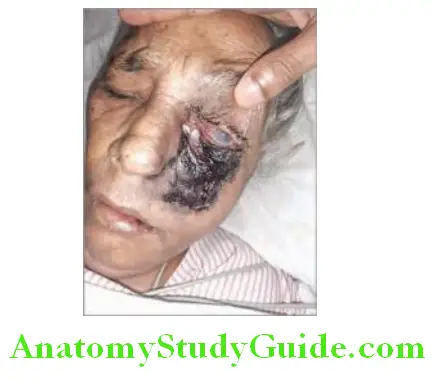
Defiitive Diagnosis:
- Diagnosis requires a positive culture from a sterile site (e.g., a needle aspirate, a tissue biopsy specimen, or pleural fluid). A probable diagnosis of mucormycosis can be done by culture from a nonsterile site (e.g., sputum or bronchoalveolar lavage).
- Biopsy with histopathologic evidence of invasive mucormycosis is the most sensitive and specific modality for definitive diagnosis. Biopsy reveals characteristic wide (6–30 mm), thick-walled, ribbon-like, aseptate hyphal elements that branch at right angles.
Treatment Surgical debridement plus:
- Amphotericin B deoxycholate: 1 mg/kg per day
- Liposomal AmB (LAmB): 5–10 mg/kg per day. However, dose escalation of LAmB to 10 mg/kg per day for CNS mucormycosis may be considered in light of the limited penetration of polyenes into the brain.
- Amphotericin B lipid complex (ABLC): 5–7.5 mg/kg
- Posaconazole or isavuconazole is used as step-down therapy for patients who have responded to amphotericin B
Mycetoma:
Question 66. Write short note on mycetoma. Write short note on actinomycosis.
Answer:
- Mycetoma is a chronic suppurative infection of the deep soft tissues and bones.
- Most common site is the limbs but can also occur in the abdominal or chest wall or head.
Mycetoma Etiology:
Caused by either:
- Aerobic or anaerobic branching gram-positive bacilli, Actinomycetales (actinomycetoma—60%)
- By true fungi, Eumycetes (eumycetoma—40%).
1. Actinomycetomas are caused by bacteria such as Actinomadura, Nocardia, and Streptomyces species.
2. Eumycetes: Many fungi cause eumycetomas. Most common are Madurella mycetomatis, M. grisea, Leptosphaeria
senegalensis and Scedosporium apiospermum.
Colored grains: Both the above groups characteristically produce colored grains; the color depends on the causative organism. Examples include black grains by eumycetoma, red and yellow grains by actinomycetoma, and white grains by either of them.
The disease develops mostly in the tropics and subtropics.
Mode of infection: It is acquired by inoculation (e.g., from a thorn) and most commonly affects the foot (Madura foot).
Mycetoma Clinical Features:
- It begins as a painless swelling at the implantation site. It becomes chronic and progressively, grows and spreads within the soft tissues, and extends into the underlying bone. There is usually little pain without fever or lymphadenopathy.
- Discharging sinuses: Nodules develop under the epidermis and rupture, producing sinuses through which grains (fungal colonies) may be discharged. Sinuses heal with scarring, while fresh sinuses appear at other places. It can cause progressive disability.
- Deeper soft tissue and bone involvement are less rapid and extensive in eumycetoma than actinomycetoma.
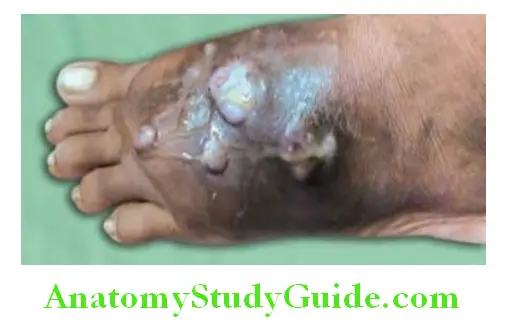
Diagnoses/Investigations:
- Demonstration of fungal grains in pus, and/or causative agent by histopathological examination of tissue
- Culture and sensitivity are needed for identification species and susceptibility testing.
- Serological tests: Not available.
Mycetoma Management:
- Actinomycetoma: Treated with prolonged antibiotic combinations. Usual combination is streptomycin and dapsone, with dapsone replaced by cotrimoxazole in cases with intolerance or refractory cases. Other combination includes cotrimoxazole plus amikacin, with rifampicin added in refractory cases and to prevent recurrence.
- Eumycetoma: Usually treated with a combination of surgery and antifungal therapy. Most commonly used antifungal drugs are itraconazole and ketoconazole (both 200–400 mg/day). Other drugs include terbinafine monotherapy, voriconazole, and posaconazole. Amphotericin B is usually not effective. Therapy is continued for 6–12 months or longer. In extensive and severe cases, amputation may be needed.
Mycetoma Nocardiosis:
- Nocardiosis is a gram-positive bacterial infection caused by aerobic actinomycetes of the genus Nocardia found in the soil.
- Mode of infection: It is an uncommon infection which occurs most frequently by direct traumatic inoculation or occasionally via inhalation or ingestion.
Mycetoma Clinical Features:
- Localized cutaneous infection: It causes localized cutaneous ulcers or nodules, most frequently in the lower limbs. In tropical countries, chronic destructive infection can produce actinomycetoma, involving soft tissues and bone.
- Systemic Nocardia infection: Occurs mostly in immunocompromised individuals and results in suppurative disease with lung and brain abscesses.
Microscopic appearance: Nocardia species appear as long, filamentous, and branching gram-positive rods. They are weakly acid-fast. They can be cultured but need prolonged incubation.
Treatment:
It depends on the result of culture and sensitivity.
- Systemic infection: Needs combinations of ceftriaxone, meropenem, amikacin, and cotrimoxazole. They are to be given for 6–12 months or longer. Abscesses are drained surgically wherever possible.
- Localized cutaneous infection: Usually treated with a single drug for 1–3 months.
Actinomyces israelii:
Actinomyces israelii can produce deep infection in the head and neck, and suppurating disease in the pelvis associated with intrauterine contraceptive devices (IUCDs).
Treatment: Penicillin or doxycycline.
Protozoal Infections:
Malaria:
Question 67. Draw a neat labeled diagram of life cycle of the malarial parasite. Discuss the etiology, epidemiology, clinical features, complications, investigations, and management of malaria. (or) Discuss the clinical features, diagnosis, and management of P. falciparum malaria (both uncomplicated and complicated P. falciform malaria).
Answer:
Malaria is a protozoan disease caused by Plasmodium. Human malaria is usually caused by one of four species of the genus Plasmodium namely
- P. falciparum
- P. vivax
- P. ovale
- P. malariae.
Occasionally, a species of malaria usually found in primates namely P. knowlesi (simian parasite) can affect man.
Plasmodium falciparum causes the most severe form of malaria than the other Plasmodium species. P. vivax is the most common cause of malaria in India.
Mode of transmission: By the bite of female anopheles mosquitoes. Malaria can also be transmitted through contaminated blood transfusions.
A comparison of the developmental characteristics of various species of Plasmodium is presented in Table
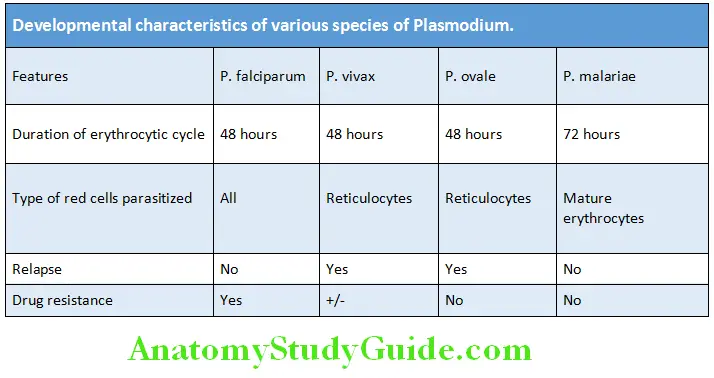
Life Cycle of the Malarial Parasite and Pathogenesis:
Question 68. Write short essay on life cycle of malarial parasite.
Answer:
The life cycle of Plasmodium species is simple because it involves only humans and mosquitoes. However, the development of the parasite is complex, because it passes through several morphologically distinct forms. Malarial parasites pass its life cycle in two different hosts namely:
- Human (intermediate host)
- Female anopheles mosquito (definitive host).
Malarial Parasite Human Cycle:
- Human cycle (infection) starts with the introduction of infectious sporozoite by the bite of infected female anopheles mosquito.
- The sporozoite is found in the salivary glands of mosquitoes. During mosquito bite, the mosquito takes a blood meal and sporozoites are released into the human’s blood. The different stages of human cycle are:
Pre-erythrocytic (Primary exoerythrocytic) stage:
Sporozoites cannot directly enter erythrocyte to start its erythrocyte stage, but undergoes development inside liver cells. The infection of the liver and development of sporozoites into merozoites are referred to as the pre-erythrocytic (exoerythrocytic) stage.
Within minutes of entry of sporozoites into the human blood (those which are not destroyed by the immune response), they are carried via the bloodstream and are rapidly (within 30 minutes) taken up by the liver.
They enter liver cells and these malaria parasites multiply by asexual reproduction (intrahepatic schizogony), releasing about 30,000 merozoites (asexual, haploid forms) when each infected liver cells ruptures.
This stage is asymptomatic and during this phase, the parasites are not found in the peripheral blood. The infected hepatocytes rupture and release merozoites into the bloodstream.
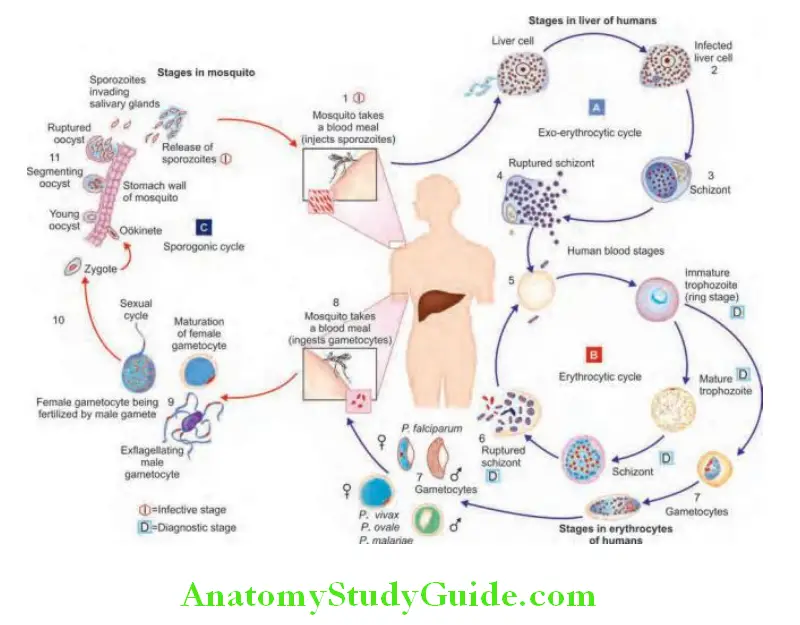
- During P. falciparum infection, rupture of swollen liver cells usually occurs within 8–12 weeks. P. falciparum and P. malariae have no persistent exoerythrocytic phase but a new outbreak of fever may result from multiplication of parasites in red cells which have not been eliminated by treatment and immune processes.
- In contrast, P. vivax and P. ovale releases merozoites into the bloodstream weeks to months after initial infection.
Malarial Parasite Hypnozoite (latent) stage:
The pre-erythrocytic phase disappears completely in P. falciparum, whereas a few parasites persist in the liver cells as dormant forms in P. vivaxand P. ovale.
The resting phase of parasite (latent phase) is called as hypnozoite. These hypnozoites are capable of developing into merozoites months or years later, causing relapsing malarial infection. Thus, the first attack of clinical malaria may develop long after the individual has left the endemic area.
Malarial Parasite Erythrocytic stage:
Once merozoites are released from the liver into the blood stream, they rapidly invade erythrocyte by penetration of the membrane. Within the red cells (erythrocytic stage), asexual division occurs.
- Asexual forms: In the red cells parasite develop through the stages of asexual forms changing from merozoite to trophozoite, to schizont and finally appearing as 8–24 new merozoites. These asexual forms of parasite can be demonstrated in the thick blood smears.
- Ring form: It is the first stage of the parasite in the red cell and is characterized by the presence of a single chromatin mass (ring form).
- Trophozoite: During this, the parasite assumes an irregular or amoeboid shape.
- Schizont: It is the next stage the parasite has consumed two-thirds of the RBC’s hemoglobin and has grown to occupy most of the cell. It shows multiple chromatin masses, each of which develops into a merozoite. Rupture of the schizont releases merozoites into the blood. This causes fever and the periodicity of fever depends on the species of parasite.
- Merozoites: Rupture of the red cell containing merozoites releases the merozoites into the bloodstream. These merozoites are capable of invading additional new erythrocyte and repeating the cycle. The characteristic clinical features of malaria such as paroxysmal fever, chills, and rigors develop during the release of these merozoites into the blood.
- Each cycle of the above process is called erythrocytic schizogony. The periodicity of such cycle takes about 48 h in P. falciparum, P. vivax, and P. ovale and about 72 hours in P. malariae. P. vivax, and P. ovale mainly attack reticulocytes and young erythrocytes, whereas P. malariae tends to attack old erythrocytes; P. falciparum will parasitize any stage of erythrocyte.
- Sexual forms: Most malaria parasites within the red cells develop into daughter merozoites. A few merozoites within erythrocytes develop not into trophozoites but undergo a different pathway of development into sexual forms called gametocytes (male and female gametocytes). These gametocytes are not released from the red cells until taken up by a feeding mosquito to complete the life cycle. These gametocytes are ingested by the mosquito during a blood meal when mosquito bites the infected human.
Malarial Parasite Mosquito cycle:
A female anopheles mosquito during its blood meal from an infected patient ingests both sexual and asexual forms of parasite, but it is only the mature sexual forms (gametocytes) capable of development.
The male and female gametocytes of malarial parasite fuse inside the mid-gut (stomach) of the mosquito to form a zygote.
- The zygote matures to form an ookinete, which penetrates the gut wall and form an oocyst.
- The oocyst matures and forms numerous sporozoites. These sporozoites have special predilection toward the salivary glands and reach maximum concentration in the salivary ducts of mosquito. The mosquito at this stage is capable of transmitting malarial infection.
- During the blood meal these sporozoites are inoculated into the new human host, thus completing the life cycle of Plasmodium.
Protection Against Malaria:
- Red blood cells containing hemoglobin F, C, or S impair growth of P. falciparum parasite. Patients with hemoglobin S especially heterozygotes (AS) are protected against the lethal complications of malaria.
- Attachment of merozoites to red blood cells is mediated via a specific erythrocyte surface receptor. In P. vivax, this receptor is related to the Duffy blood group antigen (Fya or Fyb). Thus, an individual who is Duffy-negative (as most of the West African population) will be resistant to infection by P. vivax.
Malarial Parasite Clinical Features:
- Malaria is a very common cause of fever in tropical countries. The first symptoms of malaria are nonspecific. P. vivax and P. ovale malaria
- Incubation period for P. vivax is 12–17 days, and for P. ovale is 15–18 days.
- Prodromal symptoms include headache, fatigue, abdominal discomfort, and muscle aches. These are more severe with P. malariae infections.
- Fever: The most common symptom of these “benign” malarias is paroxysms of fever. Fever starts with a rigor. The febrile paroxysm synchronizes with erythrocytic stage of the parasite.
- Tertian fever: In P. vivax and P. ovale malaria infections, the fever recurs every third day (tertian) interval (with 48 hours cycle between spikes).
- P. vivax: Patients suffering from P. vivax malaria may develop anemia, thrombocytopenia and mild jaundice with tender hepatosplenomegaly. Splenic rupture is more common. Occasionally, P. vivax malaria presently develops complications similar to those of P. falciparum malaria.
- P. ovale: The acute symptoms of P. ovale may be as severe as those of P. vivax infection, but anemia is less severe and the risk of splenic rupture is less common.
- P. vivax and P. ovale: They have a persistent hepatic cycle and a few parasites persist in the liver cells as dormant forms. This may give rise to relapses.
P. malariae malaria:
- Incubation period: 18–40 days
- Prodromal symptoms include headache, fatigue, abdominal discomfort, and muscle aches and may be more severe than P. vivax.
- Fever: The febrile paroxysm synchronizes with erythrocytic stage of the parasite.
- Quartan fever: In P. malariae infections, the fever recurs every fourth day (quartan) interval (72 hours interval between spikes).
- It is associated with gross splenomegaly, but splenic rupture is less common and anemia is less severe. Chronic P.malariae infections cause glomerulonephritis and nephrotic syndrome. P. malariae does not relapse, but a persisting undetectable parasitemia may produce repeated exacerbations and the risk of splenic rupture is less common.
P. falciparum malaria (malignant tertian or subtertian malaria):
Question 69. Write short essay on falciparum malaria.
Answer:
- It is the most dangerous type of malarias and patients may be either “killed or cured.”
- Incubation period: 7–14 days (mean 12 days)
- Prodromal symptoms: Onset is often insidious, with malaise, headache, myalgia, anorexia, and vomiting.
- Fever: Patients develop mild fever having no particular pattern and may last for several days before the onset of the classical “malarial paroxysms.”
- A high irregularly spiking unremitting fever or daily (quotidian) paroxysm is more commonly seen in falciparum malaria but usually due to mixed infection (falciparum + vivax).
- Physical findings: Patient is often anemic and jaundiced with moderate tender hepatosplenomegaly.
- Neurological complications can manifest as acute headache,
irritability, agitation, seizures, psychosis, and impaired consciousness.
P. falciparum malaria Jaundice in malaria:
Causes: Jaundice may be due to severe hemolysis and hepatic involvement (malarial hepatopathy) by malaria.
Hemolysis: The malarial parasite, especially P. falciparum infects a large number of RBCs. These are destroyed in the spleen and result in hemolytic anemia. It produces elevation of serum bilirubin (dominant unconjugated fraction) level without any significant elevation of the liver enzymes.
Malarial hepatitis (malarial hepatopathy): This term is used to describe dysfunction of liver cells in severe and complicated malaria. However inflammation of the liver parenchyma is never observed.
Severe Manifestations and Complications of Falciparum Malaria:
Question 70. Write short essay on
Answer:
- Complications of malaria
- Complications of falciparum malaria.
- WHO defines complicated falciparum malaria as one of the features shown in the Table 4.39 along with presence of asexual forms of P. falciparum in the peripheral smear. Acute renal failure, acute respiratory distress syndrome, and jaundice are uncommon in children.
- At highest risk of complications from malaria are nonimmune people, and children and pregnant females who live in endemic regions.
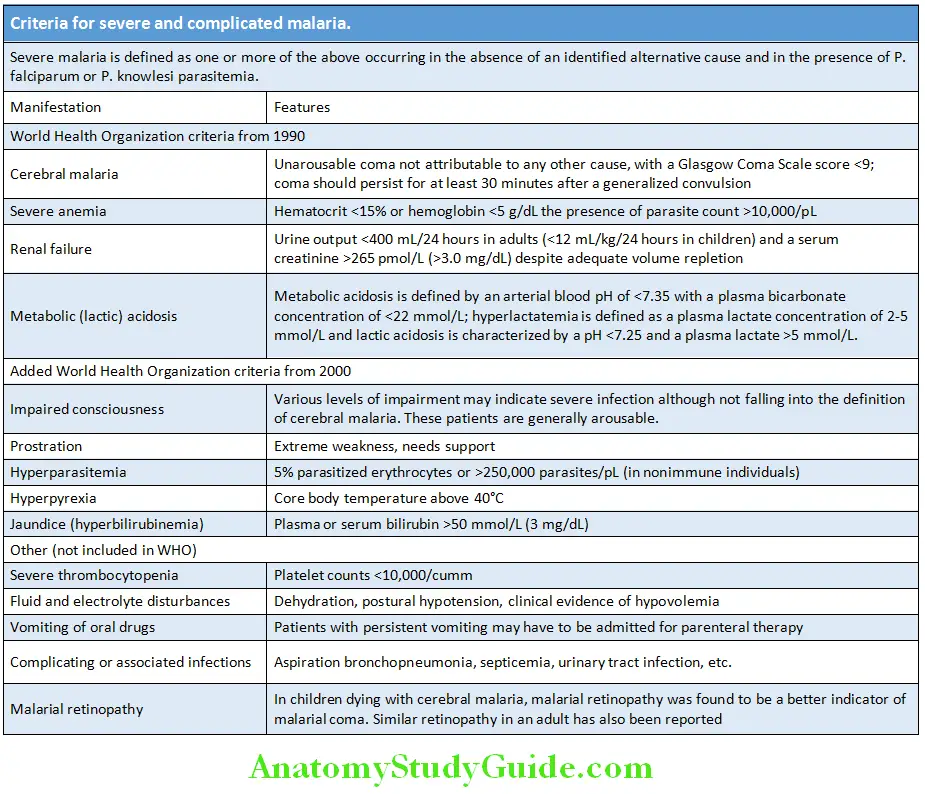
Cerebral malaria:
Question 71. Write short essay on clinical features and treatment of cerebral malaria. (or) Write long essay on cerebral malaria-etiopathogenesis, clinical features, and management.
Answer:
Cerebral malaria Definition: Cerebral malaria is defined as diffuse encephalopathy in a patient with falciparum malaria which is not attributable to any other cause.
Cerebral malaria Features: Cerebral malaria manifests as diffuse symmetric encephalopathy.
Neurological:
- Convulsions: Usually, generalized and often repeated. They occur in both children (50%) and adults (10%) but are more common in children.
- Most common neurological signs in adults are those of a symmetrical upper motor neuron lesion. The abdominal reflexes and cremasteric reflexes are absent.
- Mild neck stiffness may be seen, however, neck rigidity and photophobia and signs of raised intracranial tension are absent. Retinal hemorrhages occur in about 15% of cases, exudates are rare.
- Motor abnormalities like decerebrate rigidity, decorticate rigidity, and opisthotonus occur.
- Fixed jaw closure and tooth grinding (bruxism) are common.
- Neuropsychiatric manifestations, cerebellar signs, extrapyramidal syndromes, and multiple cranial nerve involvement are common in Indian patients.
- Residual neurological sequelae: They develop in about 5% of adults and 10% of children. These include hemiplegia, cerebral palsy, cortical blindness, deafness, aphasia, and ataxia. They are more frequent in patients with prolonged deep coma, repeated convulsions, those with hypoglycemia and severe anemia.
Ophthalmological:
- Eyes may be divergent and a pout reflex may be elicited by stroking the sides of the mouth. The corneal reflexes are preserved (except in deep coma). Transient abnormalities of eye movement (especially dysconjugate gaze) may be found.
- Ophthalmoscopy: Retinal hemorrhages may occur and indicate a poor prognosis. Papilledema is rare.
Bad prognostic signs:
- Include prolonged impaired consciousness, respiratory failure, renal failure, jaundice, and hypoglycemia.
- The CSF pressure is usually normal. The fluid is clear with <10 cells/µL, CSF lactic acid and protein levels are elevated.
- Cerebral malaria carries a mortality of around 20% in adults and 15% in children. Residual deficits are unusual in adults (<3%).
Anemia:
Anemia Renal failure:
- Acute kidney injury is common in severe falciparum malaria and usually occurs in adults. It causes progressive oliguria eventually resulting in anuria. It is accompanied by progressive increase serum creatinine and urea levels. It is usually reversible.
- Caused by renal cortical vasoconstriction and resultant hypoperfusion, sequestration, and resultant acute tubular necrosis due to microvascular obstruction and due to massive intravascular hemolysis in black water fever.
- Dehydration and hypovolemia can lead to renal hypoperfusion, but this is reversible with adequate rehydration.
Anemia Hypoglycemia:
Hypoglycemia is an important and common complication of severe falciparum malaria. It is particularly problematic occurs in three groups of patients:
- Children with severe disease
- Patients treated with quinine or quinidine
- Pregnant women (either on admission or following quinine
treatment)
Mechanism: In malaria, hypoglycemia develops due to various mechanisms.
Fluid, electrolyte, and acid-base disturbances:
- Hypovolemia: Characterized by low jugular venous pressure, postural hypotension, oliguria, and high urine specific gravity. This may be associated with signs of dehydration.
- Acidotic breathing: It may develop in severely ill patients in shock, hypoglycemia, hyperparasitemia, and renal failure.
- Lactic acidosis: It is a common complication and associated with raised lactic acid levels both in blood and CSF.
Noncardiogenic pulmonary edema:
- It is a serious complication of severe falciparum malaria in adults. It carries a high mortality rate (>80%). It can also develop in otherwise uncomplicated vivax malaria, where recovery is usual.
- It may develop several days after antimalarial therapy and at a time when the patient’s general condition is improving.
- Noncardiogenic pulmonary edema is associated with hyperparasitemia, renal failure, pregnancy, hypoglycemia, and lactic acidosis. It is aggravated by vigorous administration of IV fluid.
Circulatory collapse (“Algid malaria”):
- Algid malaria or hypotension due to peripheral circulatory failure may develop suddenly in severe malaria or it may be the presenting feature in some cases of malaria, with a systolic blood pressure <80 mm Hg in the supine position [<50 mm Hg in children], a cold, clammy, cyanotic skin, constricted peripheral veins, and rapid feeble pulse.
- This clinical picture is often associated with a complicating gram-negative septicemia and possible sites of associated infection should be sought in such patients, e.g., lung, urinary tract (especially if there is an indwelling catheter), meninges (meningitis), intravenous injection sites, intravenous lines, etc.
- Circulatory collapse is also observed in patients with pulmonary edema, metabolic acidosis, massive gastrointestinal hemorrhage, dehydration, and hypovolemia.
- Blood and urine culture should be done. Soon after collection of the specimen for culture, broad-spectrum antibiotics should be started.
Causes of neurological manifestations in malaria other than hyperparasitemia:
- High-grade fever
- Antimalarial drugs like chloroquine, quinine, mefloquine,
and halofantrine. - Hypoglycemia, either due to severe malaria or due to drugs
like quinine. - Hyponatremia, most often in the elderly.
- Severe anemia and hypoxemia can also cause cerebral
dysfunction, particularly in children.
Causes of anemia in malaria:
- Hemolysis of:
- Infected red cells
- Noninfected red cells (black water fever)
- Dyserythropoiesis
- Splenomegaly causing
- Erythrocyte sequestration
- Hemodilution
- Depletion of folate stores
- Hemorrhage secondary to thrombocytopenia/DIC
- Iron deficiency secondary to RBC breakdown
Causes of hypoglycemia in malaria:
- Failure to hepatic gluconeogenesis
- I ncreased consumption of glucose by host and malaria parasite.
- Treatment with quinine or quinidine. Both these drugs strongly stimulate pancreas to secrete insulin. This produces hyperinsulinemia resulting in hypogly
Malarial hemoglobinuria (Black water fever):
- Malaria hemoglobinuria is uncommon, and is usually associated with hyperparasitemia and/or severe disease. It may or may not be accompanied by renal failure.
- Patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency and other erythrocyte enzyme deficiencies may develop vascular hemolysis and hemoglobinuria when treated with oxidant drugs such as primaquine.
- Black water fever typically occurs in nonimmune patients with chronic falciparum malaria, taking antimalarials (especially quinine and primaquine) irregularly. It occurs more commonly in patients with G6PD deficiency.
- The hemolysis can occur so rapidly that the hemoglobin may drop significantly within a few hours and it may recur periodically at intervals of hours or days. Patient presents with headache, nausea, vomiting, and severe pain in the loins and prostration. Fever up to 39.4°C with a rigor is also seen. Urine is dark red to almost black.
- Bacterial coinfection—gram negative septicemia (Salmonella) is common in endemic areas.
Chronic complications of malaria:
- Tropical splenomegaly syndrome
- Quartan malarial nephropathy
- Burkitt’s lymphoma
Investigations and Diagnosis:
Question 72. Write short essay on laboratory tests in the diagnosis of malaria.
Answer:
Malaria should be considered in the differential diagnosis of febrile illness.
Microscopic demonstration of parasite:
- Peripheral smear examination: The diagnosis of malaria depends on the demonstration of asexual forms of the parasite in stained peripheral-blood smears. Both thin and thick smears should be examined whenever malaria is suspected. Smears are stained by one of the Romanowsky stains (e.g., Wright’s, Field’s, Leishman’s, or Giemsa).
- Bone marrow aspirate: Sometimes parasites cannot be detected in peripheral blood smears, even in patient with severe malarial infections. This may be due to partial antimalarials treatment or by sequestration of parasitized cells in deep vascular beds. In these, circumstances examination of smears of bone marrow aspirate reveals parasites or malaria pigment.
- Alternative microscopic methods have been tried, including faster methods of preparation, dark-field microscopy, and stains like benzothiocarboxypurine, acridine orange, and Rhodamine-12
- Quantitative buffy coat analysis (QBC): It is an alternative and probably more sensitive test than the peripheral smear for detecting the malarial parasite. In this test, the centrifuged buffy coat is stained with a fluorochrome (e.g., acridine orange) that “lights up” (microtube concentration methods with acridine orange staining) malarial parasites. The buffy coat smear is viewed under fluorescence microscopy
Immunodiagnosis:
Serological techniques: They can detect malarial antibodies, but these tests are not specific and are not done routinely. The tests include immunofluorescent assay (IFA) and enzyme-linked immunosorbent assay (ELISA).
PCR testing:
Molecular diagnosis by polymerase chain reaction (PCR) amplification of parasite nucleic acid (for parasite messenger RNA or DNA) is more sensitive than microscopy or rapid diagnostic tests.
Other laboratory fidings:
- Blood:
- Normochromic normocytic anemia, thrombocytopenia.
- Total leukocyte count is low to normal, but neutrophil leukocytosis may be observed in severe infections.
- Acute-phase protein: Erythrocyte sedimentation rate (ESR), plasma viscosity, and levels of C-reactive protein are high.
- Coagulation study: In severe infections, prothrombin time and partial thromboplastin time may be prolonged and antithrombin III levels are decreased.
- Other findings in severe malaria: In complicated malaria there may be metabolic acidosis and
- Low plasma concentrations of glucose, sodium, bicarbonate, calcium, magnesium, and albumin
- Elevated levels of lactate, BUN, creatinine, muscle and liver enzymes, bilirubin, and gamma globulin
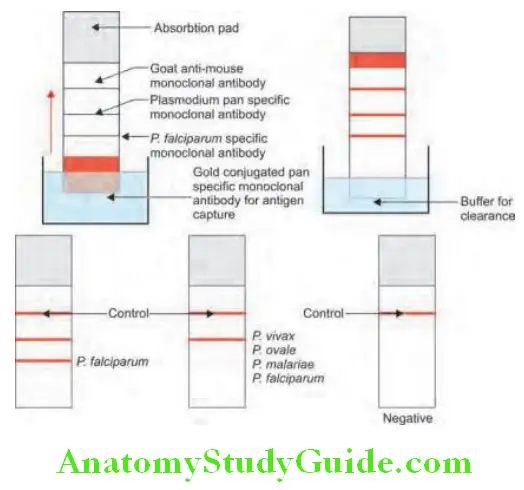
- Neuroimaging in cerebral malaria may show brain swelling, cortical infarcts, and hyperintense areas in white matter.

- Treatment of uncomplicated falciparum malaria
- Drugs used in resistant malaria/drug resistant malaria.
- Drugs for resistant falciparum malaria.
- Radical treatment of malaria
- Name antimalarial drugs
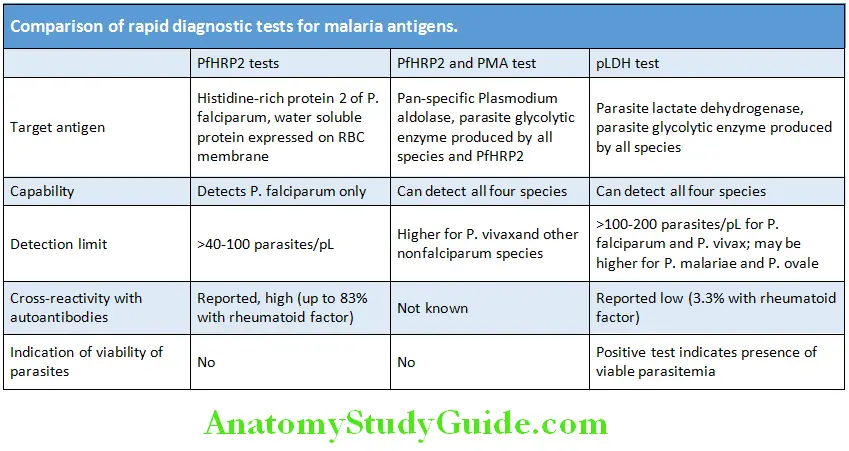
Investigations and Diagnosis Management/Treatment:
Aims of treatment are presented in Table:
To counter the threat of resistance of P. falciparum to monotherapies, and to improve treatment outcome, combinations of antimalarials are now recommended by WHO for the treatment of falciparum malaria. Two or more blood schizontocidal drugs with independent modes of action and thus unrelated biochemical targets in the parasite are used and at present artemisinin-based combination therapy
(ACT) is the recommended treatments for uncomplicated falciparum malaria.
Artemisinin-based combination therapy (ACT):
- Artemisinin and its derivatives (artesunate, artemether, artemotil, and dihydroartemisinin) produce rapid clearance of parasitemia and rapid resolution of symptoms. They reduce parasite numbers by a factor of approximately 10,000 in each asexual cycle, which is more than other current antimalarials (which reduce parasite numbers 100to 1000-fold per cycle). Artemisinin and its derivatives are eliminated rapidly.
- When given in combination with rapidly eliminated compounds (tetracyclines and clindamycin), a 7-day course of treatment with an artemisinin compound is required; but when given in combination with slowly eliminated antimalarials, shorter courses of treatment (3 days) are effective.
Nonartemisinin-based combinations (non-ACTs) include sulfadoxine—pyrimethamine with chloroquine (SP + CQ) or amodiaquine (SP + AQ). However, the prevailing high levels of resistance have compromised the efficacy of these combinations. There is no convincing evidence that SP + CQ provides any additional benefit over SP, so this combination is not recommended.
- The ACT used in the national program in India is artesunate + sulfadoxine + pyrimethamine. It is given a:
- 200 mg artesunate along with sulfadoxine 1,500 mg and pyrimethamine 75 mg on day 1.
- 200 mg artesunate on days 2 and
- Another option is to use quinine 600 mg salt three times daily for 5 days orally, followed by a single dose of sulfadoxine 1.5 g combined with pyrimethamine 75 mg.
- Artemisinin based combinations can be given in second and third trimester of pregnancy. Recommended treatment in first trimester
of pregnancy is quinine. - If sulfonamide sensitivity is suspected, quinine may be followed by tetracycline 250 m 6 hourly for 7 days.
Other drugs for treating chloroquine resistant P. falciparum are mefloquine (15 mg/kg followed by 10 mg/kg after 8 hours; generally given as ACT) and halofantrine. Recently, dihydroartemisinin (4 mg/kg/day) + piperaquine (18 mg/kg/day) combination for 3 days has been available.
Summary of treatment of malaria is presented in Table
- Radical cure of malaria due to P. vivax and P. ovale.
- Primaquine is given at a dose of 15 mg daily for 14 days. It destroys the hypnozoite phase in the liver.
Outlines the treatment of severe malaria:

Management of severe manifestations and complications of falciparum malaria are presented in Table. Recommendations for treatment of uncomplicated P. falciparum malaria in pregnancy are mentioned in Table.
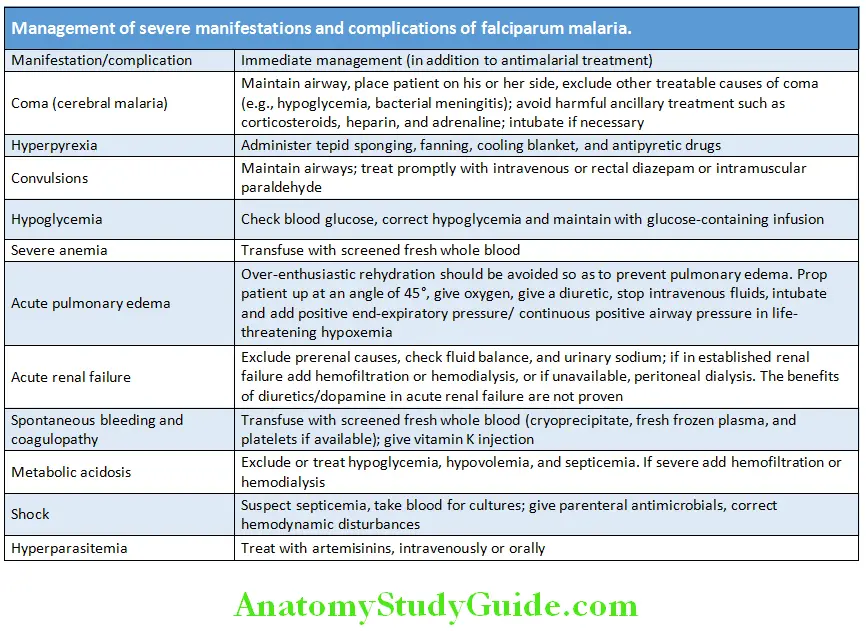

Chemoprophylaxis of malaria:
- For short-term chemoprophylaxis (<6 weeks), doxycycline in a dose of 100 mg daily. It should be started 2 days before travel and continued for 4 weeks after leaving the malarious area.
- For long-term chemoprophylaxis (>6 weeks), mefloquine in a dose of 5 mg/kg (up to 250 mg) weekly. It should be administered 2 weeks
before, during and 4 weeks after leaving the area.
Question 73. List the uses of chloroquine.
Answer:
Uses of chloroquine
- Drug of choice for clinical cure and suppressive prophylaxis of all types of malaria, except that caused by resistant P. falciparum
- Extraintestinal amebiasis
- Rheumatoid arthritis, SLE, and chikungunya
- Discoid lupus erythematosus
- Lepra reactions
- Photogenic reactions
- Infectious mononucleosis (symptomatic relief)
Tropical Splenomegaly Syndrome (TSS):
Question 74. Write short essay/note on tropical splenomegaly syndrome (TSS).
Answer:
Tropical splenomegaly syndrome or big spleen disease, also known as hyper-reactive malarial splenomegaly is massive enlargement of the spleen resulting from abnormal immune response to repeated attacks of malaria. It is seen among residents of endemic areas of malaria. It is one of the chronic complications of malaria.
Tropical Splenomegaly Syndrome Etiopathogenesis:
- Chronic or repeated malarial infections produce hypergammaglobulinemia; normocytic normochromic anemia; and massive splenomegaly.
- Tropical splenomegaly syndrome is characterized by the production of cytotoxic IgM antibodies to CD8+ T lymphocytes, antibodies to CD5+ T lymphocytes, and an increase in the ratio of CD4+ to CD8+ T cells. These result in continuous IgM production by B cell and the formation of cryoglobulins (IgM aggregates and immune complexes). This stimulates reticuloendothelial hyperplasia and leads to splenomegaly.
Tropical Splenomegaly Syndrome Clinical Features:
- Anemia and some degree of pancytopenia are usually found.
- There is increased vulnerability to respiratory and skin infections and many may die due to overwhelming sepsis.
- Physical examination shows massive splenomegaly, hepatomegaly and occasionally low-grade fever.
- The disease generally runs a benign course. Portal hypertension does not develop and the condition is reversible with antimalarial treatment. Some may evolve into malignant lymphoproliferative disorder (e.g., CLL).
Tropical Splenomegaly Syndrome Diagnosis:
- Antimalarial antibodies: High levels of antimalarial antibodies are found in blood. IgM levels are markedly elevated (up to 20 times).
- There is increase in the serum levels of polyclonal IgM with cryoglobulinemia, reduced C3 and the rheumatoid factor may be positive.
- Liver biopsy:
- Light microscopy of liver shows sinusoidal lymphocytosis and Kupffer cell hyperplasia.
- Immunofluorescence microscopy shows IgM aggregates phagocytosed by RE cells.
- Peripheral blood: The peripheral smear shows normocytic normochromic anemia with increased reticulocyte count. Leukopenia and thrombocytopenia may also be seen due to hypersplenism. Malarial parasites are not found in the peripheral blood.
Treatment of Tropical Splenomegaly Syndrome:
- Chloroquine weekly or proguanil daily has been found to be useful. These drugs may have to be continued for long periods, possibly for life.
- Severe anemia may require blood transfusion.
- Splenectomy may do more harm than good and it may be beneficial in only patients with splenic lymphoma.
- Splenic irradiation or antimitotic therapy is not beneficial and may be even dangerous.
Malaria Vaccines:
Question 75. Write short note on malarial vaccines.
Answer:
Malaria Vaccines Vaccine Types:
- The candidate malaria vaccines target the different phases of the parasite’s life cycle.
- The pre-erythrocytic vaccines target sporozoites or schizont infected liver cells, and are aimed at preventing infection by stopping the progression of hepatic stage. Even a single sporozoite escaping vaccine-induced immunity may cause a fully pathogenic blood stage infection, as was found during the clinical trials of the latest antisporozoite RTS, S vaccine.
- The erythrocytic stage vaccines are aimed at reducing parasite multiplication and growth in order to protect against clinical disease, particularly severe disease. These vaccines are designed to induce antibody responses against the targets on the asexual blood stage of the parasite such as merozoite surface proteins (such as MSP-1) or those contained in specialized organelles associated with invasion (such as AMA-1). But, the very short duration during which merozoites stay outside the red cells (about 2 minutes), the high degree of polymorphic variability, and the use of alternative invasion pathways by P. falciparum.
- Transmission-blocking vaccines are aimed at reducing malaria transmission by interrupting the parasite life-cycle in the mosquito by inducing antibodies that prevent either fertilization of the gametes in the mosquito gut or the further development of the zygote into sporozoites. These vaccines do not protect the immunized individual but rather provide herd benefit. Antidisease vaccination, by preventing the parasite surface protein parasite-derived Plasmodium falciparum erythrocyte membrane protein 1 (PfEMP1) from interacting with various vascular endothelial cell-surface receptors, may block the sequestration of parasite-infected erythrocytes and prevents the serious complications such as cerebral malaria or placental malaria. All these vaccines are in clinical trial stages. RTS, S is the most recently developed recombinant vaccine. It consists of the P. falciparum circumsporozoite protein from the pre-erythrocytic stage has been approved to vaccinate children aged 6 weeks to 17 months outside the European Union.
Recrudescence of malaria:
- Exacerbation of persistent undetectable parasitemia, due to survival of erythrocytic forms, no exoerythrocytic cycle (P.f. and P.m.)
Relapse:
- Reactivation of hypnozoites forms of parasite in liver, separate from previous infection with same species (P.v. and P.o.)
Recurrence or reinfection:
- Exoerythrocytic forms infect erythrocytes, separate from previous infection (all species)
- Cannot always differentiate recrudescence from reinfection
Leishmaniasis:
Question 76. Write short note on leishmaniasis.
Answer:
Leishmaniasis is a group of diseases caused by unicellular, flagellate, and intracellular protozoa of the genus Leishmania, which are transmitted by the bite of the female phlebotomine sandfly.
Clinical Syndromes:
Visceral Leishmaniasis (Kala-azar):
Question 77. Write short note on kala-azar or visceral leishmaniasis.
Answer:
Visceral leishmaniasis (kala-azar) is a generalized visceral infection caused by the protozoon Leishmania donovani complex (comprising L. donovani, L. infantum, and L. chagasi).
Visceral Leishmaniasis Epidemiology:
Mode of transmission: Sandfly of the Genera Phlebotomus.
- Endemic:
- Kala-azar is endemic in several countries. More than 90% of visceral leishmaniases are found in India, Bangladesh, Southern Sudan, Nepal, and Brazil.
- In India, it is endemic in the states of Bihar, West Bengal, Eastern parts of Uttar Pradesh, and parts of Odisha.
- Sporadic cases have been reported from several other parts of India.
Life cycle of Leishmania:
Leishmania occurs in two forms:
- Extracellular, flagellate promastigote form in the sandfly vector Intracellular, nonflagellate amastigote form in humans.
- Incubation period: Usually 1–6 months, but may be several years.
Clinical features of kala-azar:
- Age group: In India, adults and children are equally affected whereas elsewhere it is mainly seen in small children andinfants (except in adults with HIV coinfection).
- Organs involved: It primarily affects the host’s reticuloendothelial system. In visceral leishmaniasis, monocytes and macrophages of the spleen, liver, bone marrow, and lymph nodes are primarily affected.
- Onset: Great majority of individuals infected remain asymptomatic and onset in others may be abrupt.
- Fever: It is the first sign of infection. Few patients present with a low-grade fever, whereas others present with a high-grade,
usually accompanied by rigor and chills. The fever is intermittent showing a double rise of temperature in 24 hours (“camel hump fever”). The intensity of fever decreases over time, and patients may be afebrile for intervening periods ranging from
weeks to months. This is followed by a relapse of fever which is often less intense.
Physical fidings:
- Massive splenomegaly and hepatomegaly: Splenomegaly develops in the first few weeks and becomes massive as the disease progresses. Moderate hepatomegaly develops later.
- Lymphadenopathy: It is common in patients in Africa, the Mediterranean, and South America. In India, it is more common in patients from West Bengal.
- Blackish discoloration of the skin: Disease derived its name; kala-azar is derived from the Hindi word for “black fever.” Generalized blackish pigmentation with rough skin is prominent over face is an observed in advanced stage of disease and now it is rare.
- Anemia: Moderate to severe anemia develops rapidly, and can produce congestive cardiac failure and associated clinical
features. - Bleeding manifestations: Thrombocytopenia, often associated with hepatic dysfunction, may cause bleeding from the retina, gastrointestinal tract, and nose.
Cause of death: Death usually occurs within a year and is either due to bacterial infection or uncontrolled bleeding.
Complications:
- Hypoalbuminemia: May develop in advanced stage and may manifest as pedal edema, ascites, and anasarca (gross generalized edema and swelling).
- Immunosuppression: As the disease advances, severe immunosuppression may lead to secondary infections. These include tuberculosis, pneumonia, malaria, severe amebic or bacillary dysentery, gastroenteritis, herpes zoster, and chickenpox. Skin infections (e.g., boils, cellulitis, scabies, and cancrum oris) are common.
Physical fidings Investigations:
Direct evidences:
- Demonstration of amastigotes (Leishman–Donovan bodies): It can be demonstrated in stained smears of aspirates of bone marrow (50–70% cases), spleen (70–90% cases), liver, lymph nodes, or buffy coat of peripheral blood. Demonstration of LD bodies in splenic smears is the most useful for the diagnosis (98% sensitivity); however, there is a risk of serious hemorrhage in inexperienced hands. It is the gold standard for the diagnosis of visceral leishmaniasis (VL).
- Culture: The aspirated material can be cultured in the Novy-MacNeal-Nicolle (NNN) medium for the organism.
Indirect evidences:
- Blood: Pancytopenia is a common feature (i.e., anemia, granulocytopenia, and thrombocytopenia).
- Biochemical findings:
- Low serum albumin (hypoalbuminemia) and polyclonal hypergammaglobulinemia (high serum globulin), chiefly IgG followed by IgM in advanced cases.
- Liver function test: Mildly elevated bilirubin, AST/ALT and alkaline phosphatase may be seen.
- Serological/immunodiagnostic tests:
- These are less invasive and are useful in community surveillance studies.
- Leishmanin (Montenegro) skin test: Intradermal injection of killed culture of promastigotes produces a delayed-type
hypersensitivity and is positive only in patients with cured kala-azar. - Napier’s aldehyde (formal gel) test and Chopra’s antimony test: These are nonspecific test and should not be
employed for the diagnosis of VL. They are used for demonstration of increased immunoglobulins. - Detection of specific antibody: Various tests such as complement fiation test, indirect hemagglutination test, indirect
florescent antibody test, and countercurrent immunoglobulins have poor sensitivity and specifiity.
- Antigen detection by reverse western blotting, dot-enzyme immunoassay (EIA) and latex agglutination test.
- PCR: To detect DNA and identify species.
Post-Kala-azar Dermal Leishmaniasis (PKDL)
Time of occurrence: In India, Sudan, and other East African countries, 2–50% of patients develop skin lesions due to local parasitic infection concurrent with or after apparent recovery or cure of visceral leishmaniasis.
Post-Kala-azar Dermal Leishmaniasis Clinical features:
Skin lesions may be of three types namely: Hypopigmented macules, papules, or nodules and can occur in varying combinations.
- Hypopigmented macules: Earliest lesions and usually found on the trunk and extremities.
- Erythematous patches: These have butterfly distribution on the face.
- Yellowish pink nodules: They are generally found on the skin and rarely on mucus membrane of tongue and eye.
Diagnosis: Demonstration of parasite in the slit smear or by culture of the dermal tissue.
Treatment:
Question 78. Drugs used to treat kala-azar.
Answer:
General considerations: Blood transfusions to correct severe anemia and treatment of intercurrent bacterial infections with antibiotics.
Pentavalent Antimonial Compounds:
- Pentavalent antimonial compounds (sodium stibogluconate and meglumine antimoniate) were the first drugs to be used for the treatment of leishmaniasis and still remain the mainstay of treatment. The dose is 20 mg/kg either intravenously or intramuscularly for 40 days. However, many patients may fail to respond due to parasitic resistance.
- Side effects: Include arthralgia, myalgia, abdominal pain, raised liver enzymes, pancreatitis (in immunosuppressed patients), ECG
changes (T wave inversion and reduced amplitude), and rarely death due to cardiotoxicity. - For PKDL treatment for 3–4 months is necessary.
Amphotericin B:
Question 79. Write short note on amphotericin B and its side effects.
Answer:
Amphotericin B deoxycholate is currently used as a primary drug of choice in Bihar, India. In other parts of the world, it is used as a secondline drug when patient is unresponsive to initial antimonial treatment.
- Dose: 0.75–1.00 mg/kg given once daily or on alternate days for a total of 15–20 infusion. It has a cure rate of almost 100%.
- Side effects: Infusion-related side effects include high fever with chills, thrombophlebitis, diarrhea, and vomiting are common. Other side effects include hypokalemia and renal or hepatic toxicity and thrombocytopenia.
- Lipid formulations of amphotericin B: Some of above side effects of amphotericin B deoxycholate can be reduced by using lipidpreparations B lipid complex and amphotericin B colloidal dispersion. In India, liposomal amphotericin B is administered at a total dose of 10 or 15 mg/kg, administered in a single dose or as multiple doses over several days. It may be combined with 7–14 days of oral miltefosine.
Amphotericin B Miltefosine:
- Miltefosine is an alkyl phospholipid administered orally has shown excellent results. It is the first highly effective, oral drug for treating visceral leishmaniasis.
- Dose: 50 mg once a day if weight is <25 kg, and 50 mg twice a day if weight is >25 kg. Duration of treatment is 28 days.
- Side effects include vomiting and diarrhea, and occasionally, reversible hepatotoxicity and nephrotoxicity.
Amphotericin B Pentamidine:
Pentamidine isethionate was used. Dose of 3–4 mg/kg on alternate days for 5–25 weeks was recommended as a second-line drug to treat antimony resistant cases. However, it is abandoned because of its reduced effectiveness and serious side effects (e.g., hypoglycemia, hypotension and induction of diabetes mellitus).
Amphotericin B Other Drugs:
- Paromomycin (aminosidine): It is antibiotic with antileishmanial action and is highly effective and safe drug. It is given intramuscularly at dose of 11 mg of base/kg daily for 21 days.
- Azoles: ketoconazole, fluconazole, and itraconazole
- These oral antifungal agents have variable efficacy in leishmaniasis treatment.
Toxoplasmosis:
Question 80. Discuss the etiology, clinical manifestations, diagnosis, and management of toxoplasmosis.
Answer:
Toxoplasmosis is caused by the intracellular protozoan parasite Toxoplasma gondii.
Life Cycle of Toxoplasma gondii:
Man is the intermediate host and cat is the definitive host.
Mode of Transmission:
- Humans infection occurs via ingestion of:
- Oocyst discharged in the feces of infected reservoir animal (cat), contaminated salads, vegetables, and water.
- Raw or undercooked meats containing tissue cysts (bradyzoites). Sheep, pigs, and rabbits are the most common meat sources.
- Transplacental infection from mother to fetus may also occur.
Clinical Manifestations:
Question 81. Write short note on clinical features of toxoplasmosis.
Infection in immunocompetent host:
- Asymptomatic: Most acute toxoplasmosis is subclinical and goes unnoticed.
- Painless lymphadenopathy: In symptomatic patients, it is the most common presenting feature. It may be local or generalized and cervical lymphadenopathy is most common.
- Other features:
- Systemic symptoms are not observed in most patients. If present, these include fever, malaise, fatigue, myalgia, sore throat, and headache.
- Uncommon features are skin rash and confusion.
- Rarely: Myocarditis, encephalitis, pneumonia, hepatitis, and polymyositis.
- Completely resolution occurs within a few weeks to months.
- Ocular infection:
- Most often ocular involvement (chorioretinitis) is observed in congenital infection
- Examination shows gray white foci of retinal necrosis with adjacent choroiditis, vasculitis, hemorrhage, and vitreitis. Anterior uveitis may also be seen.
- Infection in immunocompromised host:
- In immunocompromised patients, toxoplasmosis is usually due to reactivation of latent infection.
- Central nervous system:
- It is the most common system affected.
- Manifestations include encephalopathy, meningoencephalitis, seizures, headache, and focal neurological deficits.
- Pneumonia and ocular involvement may also occur.
Congenital toxoplasmosis:
Question 82. Write short note on congenital toxoplasmosis.
Answer:
Congenital toxoplasmosis Transmission:
Acute toxoplasmosis is mostly subclinical and develops in 0.3–1% of pregnant women.
Clinical manifestations:
- Congenital toxoplasmosis may be asymptomatic, but can produce serious disease.
- CNS: Manifestations include hydrocephalus, microcephaly, encephalitis, convulsions, tremors, paralysis, and mental retardation. Cerebrospinal fluid may be xanthochromic with raised protein and mononuclear cells. Skull radiograph may show patches of calcification in the brain.
- Ophthalmic manifestations: Microphthalmus, nystagmus, chorioretinitis (common), and blindness.
- Other features: Hepatomegaly, jaundice, thrombocytopenia, and purpura may be seen.
Congenital toxoplasmosis Investigations:
- Blood: Mild lymphocytosis, raised ESR, and mild increase in liver enzymes.
- Serodiagnosis: Presence of significant levels ofToxoplasma specific IgM antibody indicates acute infection and absence of IgM antibodies virtually rules out acute infection. However, in immunocompromised patients most of the time toxoplasmosis is due to reactivation and there is no rise in IgM antibody. Hence, IgG antibodies are used for presumptive diagnosis.
- Sabin–Feldman methylene blue dye test: A dye test positive with a titer of 1:128 is diagnostic of active toxoplasmosis.
- Indirect fluorescent antibody test
- Fluorescent stain for toxoplasma (Goldman’s test): It is useful for detection of parasites in smears and biopsy specimens.
- ELISA test.
- PCR for toxoplasma DNA in ocular secretions and amniotic fluid
- Microscopic examination of smear or biopsy material (from a lymph node or muscle) shows the parasite.
- Inoculation of suspected material into laboratory-bred mice, guinea pigs may show characteristic histological changes.
- These include any of the three infectious stages: tachyzoites in groups, bradyzoites (tissue cysts), or sporozoites within oocyst.
- Investigations in toxoplasma encephalitis:
- CSF may be normal or show mild increase in cells and protein
- Serum: IgG antibodies to toxoplasma
- Radiological: CT scan of head may show multiple contrast-enhancing lesions.
However, MRI is more sensitive than CT scan in identifying the lesions. Single-photon emission computed tomography (SPECT) helps in
differentiating CNS lymphoma from toxoplasmic encephalitis.
Congenital toxoplasmosis Management:
- Acquired uncomplicated toxoplasmosis in an immunocompetent individual is self-limiting and does not require treatment.
- Treatment for severe (especially eye involvement) or progressive disease and for infection in immunocompromised patients
- Combination of sulfadiazine 2–4 g daily (1 g 6 hourly) and pyrimethamine in a single loading dose 25 mg daily both for 4–6 weeks, along with folinic acid/leucovorin. Steroids are added to the above regime for ocular toxoplasmosis.
- If the patient is sensitive to sulfonamides, clindamycin, atovaquone, or azithromycin can be used.
Toxoplasmosis in pregnant women:
- If a seronegative women acquire toxoplasmosis during pregnancy (especially during first trimester), or seroconverts during pregnancy, there is a greater risk of transmitting the infection to fetus giving rise to an infant with congenital toxoplasmosis. Such pregnancies should be terminated.
- The aim of management of pregnant women with toxoplasmosis is to reduce the risk of fetal complications.
- The recommended treatment for a pregnant woman with an established recent infection is spiramycin (3 g daily in divided doses) until term. However, whether it has any significant effect on the frequency or severity of fetal damage is not well established. Infected infants should be treated from birth.
Infections Caused By Helminths:
Cestodes (Tapeworms):
Cestodes (tapeworms) are ribbon-shaped worms which vary from a few millimeters to several meters in length.
Life Cycle:
The tapeworm passes its life cycle in two hosts:
- Definitive host: Human is the definitive host and harbors the adult worm.
- Intermediate host: It may be pig or cattle depending on type of tapeworm and they harbor larval forms.
Taenia solium:
Question 83. Write a short note on Taenia solium and cysticercosis.
Answer:
Taenia solium, the pork tapeworm, inhabits the intestinal lumen of humans (only definitive host). Adult worms are found only in human. Pigs are the usual intermediate hosts.
Taenia solium Disease in Humans:
Taenia solium causes:
- Intestinal infection: If cysticerci present in the undercooked pork is ingested.
- Cysticercosis (systemic infection from larval migration): If ova present in contaminated food and water are ingested. Feco-oral
autoinfection can occur but is rare. Patients with tapeworms usually do not develop cysticercosis and patients with cysticercosis do not usually harbor tapeworms.
Taenia solium Cysticercosis:
It is the condition in which human tissue is invaded by the larval form of Taenia solium.
Mode of infection:
- Human cysticercosis results from ingestion of ova/eggs ofTaenia solium which have contaminated water or vegetable.
- Feco-oral autoinfection: Human harboring adult worm may autoinfect himself either due to unhygienic personal habits.
- Reversal of peristaltic movements of the intestine whereby gravid segments are thrown back to the stomach which releases thousands of eggs into the stomach.
- The common sites of cysticerci are subcutaneous tissue, skeletal muscle, brain, and eyes.
Clinical features:
Question 84. Write short note on neurocysticercosis.
Answer:
Intestinal infection: Presence of adult tapeworm in the intestine usually do not produce symptoms. It is usually discovered when proglottids are found in feces or on underclothing.
Cysticercosis: They are usually range from 10 to 1,000.
- Superficially placed cysts: Those in subcutaneous tissues and muscles cause few or no symptoms. They produce palpable or visible nodule (pea-like ovoid bodies) under the skin or mucosa. Eventually, they die and undergo dystrophic calcification. The dead larvae may cause marked tissue response resulting in muscular pain, weakness, fever, and eosinophilia.
- Neurocysticercosis results from cysts in the brain. Heavy brain infections may present with meningoencephalitis, epilepsy, personality changes, staggering gait, space-occupying lesion, stroke (due to inflammatory changes in the wall of intracranial arteries located in the vicinity of cysticerci), or hydrocephalus. The cerebral signs usually occur after 5–20 years when the larvae die.
Neurocysticercosis Diagnosis:
- Stool examination: Eggs and proglottids in stool
- Radiological investigations:
- Radiographs of involved soft tissue (buttocks and thigh) may show calcified cysts (cigar-shaped/rice grain calcification) in muscles.
- CT scan of the brain may show calcified cysticerci, small hypodense lesions (ring or disk-like enhancing lesion) or small hypodense lesions with central bright spot. CT scan is more sensitive than MRI for identifying calcified cysts.
- MRI scan: It is better for identifying cystic lesions, enhancement, and cysticercosis in the ventricles.
- Serological test: Specific enzyme-linked immunoelectrodiffusion transfer blot (EITB) using lentil-lectin purified glycoproteins is highly sensitive and specific. IFA and ELISA test are also useful.
- Histological examination of the excised subcutaneous nodule or the lesion will show cysticerci.
Question 85. Write short note on.
Answer:
- Drug treatment of tapeworm.
- Treatment of neurocysticercosis.
Treatment:
Intestinal Infection:
For the adult worm in the intestine: Niclosamide (2 g as a single dose), praziquantel (5 mg/kg), or albendazole.
Neurocysticercosis:
- Albendazole: 15 mg/kg/day for 2–4 weeks is the drug of choice for parenchymal neurocysticercosis.
- An alternative drug is praziquantel (PZQ) in the dose of 50 mg/kg in three divided doses daily for 10 days. Single day course is effective in patients with a single cyst or low cyst burdens, but is less efficacious in those with heavier cyst burdens.
- Corticosteroids: Successful treatment is accompanied by increased local inflammation. Hence, prednisolone is given in the dose of 10 mg 8 hourly for 14 days, starting 1 day before, during and after the course of anthelminthic (albendazole or PZQ).
- Antiepileptic drugs should be administered for epilepsy until the reaction in the brain has subsided.
- Operative intervention may be necessary for internal hydrocephalus.
Echinococcus granulosus (Taenia Echinococcus) and Hydatid Disease:
Question 86. Write short note on hydatid disease.
Answer:
Life cycle of Echinococcus granulosus:
- Definitive host: Dog, wolf, fox, and jackal are the definitive host.
- Intermediate host: Human, sheep, pig, cattle, goat, etc., are intermediate host.
Hydatid Disease:
Hydatid cyst is the larval stage of E. granulosus producing tissue infection of humans or other intermediate hosts (sheep, cattle, camels, and other animals from contaminated water).
Mode of infection:
Human is infected accidentally by ingestion of eggs while handling a dog or drinking contaminated water. Canines (especially dogs) are the definitive hosts for E. granulosus.
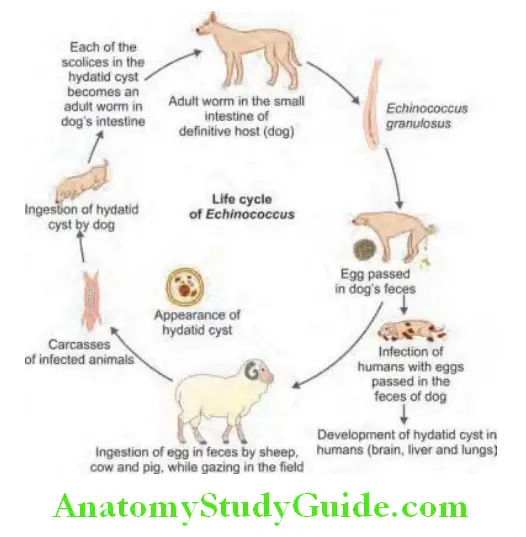
Echinococcus granulosus Clinical features:
- Humans may ingest the eggs while handling a dog or contaminated water. Usually, hydatid cyst is acquired in childhood and is diagnosed after a latent period of 5–20 years. It may cause pressure
symptoms and vary, depending on the site of the cyst (organ or tissue involved). - Liver: In about 75% of the patients, hydatid disease involves the right lobe of the liver and it contains a single cyst. The lesions in liver often present as palpable masses or abdominal pain.
- Obstruction of bile duct may produce obstructive jaundice.
- Intrabiliary extrusion of calcified hepatic cysts can cause recurrent cholecystitis.
- Rupture of a hydatid cysts: It may occur into the bile duct, peritoneal cavity, lung, pleura, or bronchus. It may cause fever, pruritus, urticarial rash, or an anaphylactoid reaction which may be fatal.
- Pulmonary hydatid cyst may rupture producing cough, chest pain, or hemoptysis.
- Hydatid cyst in central nervous system may produce epilepsy or blindness.
- Other organs involved: Rarely bone, heart, kidneys, spleen, ovary and thyroid.
Gross appearance of hydatid cyst.
Echinococcus granulosus Diagnosis:
- Radiological:
- X-ray: Routine chest radiographs is helpful in the diagnosis of hydatid cysts of lung. Lung lesions appear as round, irregular masses of uniform density. The “meniscus sign” or “crescent sign” is due to the presence of air between the pericyst and the laminated membrane. The “water lily sign” is due to an endocyst floating in a partially fluid-filled cyst. Occasionally, a chest X-ray may show smooth rim of calcified hepatic cyst.
- CT scan, ultrasonography and MRI can identify the hydatid cysts by showing scolices and daughter cysts.
- Casoni’s skin test is performed by intradermal injection of 0.2 mL of fresh sterile hydatid fluid. Positive test produces immediate hypersensitivity reaction (not routinely available).
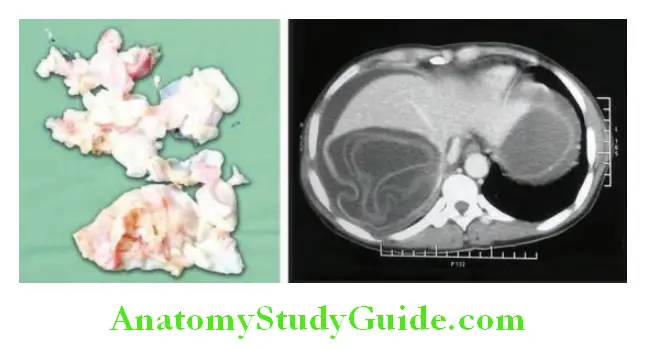
Serological test:
- Precipitin reaction, complement fixation, immunofluorescent tests, and ELISA are positive in 70–90% of patients.
- IHAT, ELISA using specific echinococcal antigens
Question 87. Write short note on medical management of hydatid disease.
Answer:
Hydatid disease Management/Treatment:
- Surgical excision of the cysts wherever possible is the treatment of choice. Great care is needed to avoid spillage of fluid and cavities are sterilized with 0.5% silver nitrate or 7% sodium chloride.
- In inoperable cases, “high-dose” albendazole (7.5 mg/kg/twice daily or 400 mg twice daily) is given for 1–3 months. In selected cases, the drug can combined with percutaneous puncture, aspiration, injection of scolicidal agent, and re-aspiration (PAIR)
- Praziquantel (20 mg/kg twice daily for 14 days) kills protoscolices and is useful perioperatively.
Enterobius vermicularis (Threadworm/Pinworm):
Question 88. Write a short note on enterobiasis, pinworm infection, and threadworm infection.
Answer:
Enterobiasis is an intestinal infection of humans caused by Enterobius vermicularis (threadworm).
Enterobius vermicularis Mode of Infection:
Humans are usually infected by the ingestion of eggs by direct transfer of eggs from the anus to the mouth by way of contaminated fingers or through contaminated food or water. Retroinfection occurs when the eggs hatch in the perianal region and the larvae migrate back into the bowel lumen.
Clinical Features:
Question 89. Write a short note on clinical features of Enterobius vermicularis.
Answer:
- Perianal itching (pruritus ani): It is the most common symptom, especially at night. This is due to the gravid female worm which migrates to anal region at night and lays ova around perianal region causing intense itching at night. The ova are carried to the mouth on the fingers and so reinfection or human-to-human infection is common (autoinfection). The adult worms may be seen moving on the buttocks or in the stool.
- In females: Migration of adult worm into the female genitalia. It may result in vaginal discharge, salpingitis, and endometritis.
- Other symptoms include irritability, insomnia, and enuresis.
Investigations and Diagnoses:
Question 90. Write a short note on laboratory features of pinworm infestations.
Answer:
- Direct visualizing the worms in the perineal and perianal region
- Stool examination for eggs may be positive in only 5–10% cases.
- Cellophane tape test: This test is performed by touching the adhesive surface of cellophane tape to the perianal area of skin several times in the morning and removing. It is then examined for eggs on a glass slide under the microscope. Sensitivity of test is about 90%.
Treatment/Management:
- Pyrantel pamoate given in a single oral dose of 11 mg/kg is the treatment of choice.
- Other drugs: A single oral dose of mebendazole (100 mg), albendazole (400 mg), or piperazine (4 g).
- Recurring infections: If infection constantly recurs in a family, each family member should be given mebendazole 100 mg twice daily for 3 days. This should be repeated after 2 weeks (for also to control autoreinfection). During this period all night clothes and bed linens are laundered. Finger nails must be trimmed and hands are washed before meals.
Ascaris lumbricoides (Roundworm):
Question 91. Write short note on ascariasis or roundworm infection.
Answer:
Ascariasis is an infection of humans caused by nematode Ascaris lumbricoides.
Clinical Features:
Question 92. Write short note on complications of roundworm infestations.
Answer:
- Infection is usually asymptomatic.
- Heavy infections are associated with nausea, vomiting, abdominal discomfort, anorexia, and malnutrition (fat, protein, carbohydrate, and vitamins) especially in children. However, serious morbidity and mortality are rare in ascariasis.
- The adult worm may be vomited or passed in the stool.
- Mechanical effects: The large size of the adult worm and its tendency to aggregate can form bolus of adult worms. It may produce volvulus, intussusception or obstruction of the small intestine (most common in the terminal ileum), hemorrhagic infarction, and perforation.
- Ectopic ascariasis:
- They may occasionally migrate and invade the appendix (causing acute appendicitis), or the bile duct (causing biliary obstruction, suppurative cholangitis, and liver abscess) and pancreatic ducts (pancreatitis).
- Pulmonary: Larval migration through the lungs may produce pulmonary eosinophilia, ascaris bronchopneumonia (characterized by fever, cough, dyspnea, wheeze, eosinophilic leukocytosis, and migratory pulmonary infiltrates), eosinophilic granulomas, bronchial asthma, and urticaria.
Roundworm infestations Investigations and Diagnoses:
- Stool examination: Microscopically shows typical ova in the feces and occasionally adult worms are expelled from the mouth or the anus.
- Radiographic study: Barium examination may demonstrate the worms.
- Blood shows eosinophilia.
Management/Treatment:
- Intestinal ascariasis: A single dose of albendazole (400 mg), pyrantel pamoate (11 mg/kg; maximum 1 g), ivermectin (150–200 μg/ kg) or mebendazole (100 mg twice daily for 3 days). The older drug, piperazine citrate is highly effective and less expensive, but slightly more toxic.
- Obstruction due to ascariasis adult worms should be treated with nasogastric suction, piperazine citrate, and intravenous fluids. Very rarely, surgical or endoscopic intervention may be needed for intestinal or biliary obstruction.
- Ascaris bronchopneumonia is treated symptomatically.
Trichuris trichiura (Whipworm):
Question 93. Write a short note on trichuriasis/whipworm infection.
Answer:
Trichuriasis is an intestinal infection of humans caused by Trichuris trichiura (whipworm).
Trichuris trichiura Clinical Features:
- Infection is usually asymptomatic, but mucosal damage at the attachment of worm in the intestine can produce petechiae. This may cause loss of 0.005 mL blood/worm/day by the patient and cause anemia.
- Occasionally intense infections in children may cause severe mucosal damage leading to colonic ulceration, persistent diarrhea/dysentery, growth retardation, or rectal prolapse.
- Treatment: Mebendazole in the dose of 100 mg twice daily for 3 days or albendazole 400 mg once daily for 3 days in the treatment of choice.
- Heavy infections may need repetitions of dose.
Ancylostomiasis (Hookworm):
Question 94. Discuss the etiopathogenesis, clinical features, and management and prevention of hookworm disease.
Answer:
- Ancylostomiasis is a symptomatic infection caused by parasitization with human hookworms Ancylostoma duodenale or Necator americanus.
- Hookworm infection is one of the major causes of anemia in the tropics and subtropics.
- Ancylostoma duodenale or old-world hookworm is found in East Asia, Africa, China, Japan, India, and the Pacific Islands. Necator americanus or new-world hookworm is found in South and Central America and the Caribbean.
Ancylostomiasis Mode of Infection:
It occurs through skin by filariform larvae when human walks bare-foot on the focally contaminated soil.
Ancylostomiasis Clinical Features:
- Lesion in skin: Allergic dermatitis/itching dermatitis (“ground itch dermatitis”) develops at the site of entry of the infective filariform larvae. The lesions are usually on the feet, particularly between the toes.
- Pulmonary manifestations: The passage of larvae through the lungs in a heavy infection produces cough with bloodstained sputum, fever. There may be bronchitis, patchy pulmonary consolidation/bronchopneumonia, and eosinophilia.
- Gastrointestinal manifestations: Light infections (especially in well-nourished individuals) are often asymptomatic.
- When the worms have reached the small intestine, heavier worm loads may produce epigastric pain, nausea, and vomiting resembling peptic ulcer disease. Other symptoms include abdominal distension, pica, and small frequent loose stools.
- Pathological effects: Chronic heavy infection, particularly on a background of malnourishment, may cause:
- Iron deficiency anemia due to chronic intestinal blood loss.
- Hypoproteinemia/hypoalbuminemia
- Retarded physical, mental, and sexual development may occur in older children with heavy infection in children.
Investigations and Diagnoses:
- Stool examination:
- Microscopic examination ofstool shows the characteristic eggs/ovum. The eggs of both Ancylostoma duodenale and Necator americanus are 60–70 µm in length and bounded by an ovoid transparent hyaline membrane.
- Occult blood: The stools rarely show gross blood, but tests for occult blood are usually positive.
- Blood:
- Peripheral blood smear: Characteristically shows microcytic hypochromic anemia. There may be eosinophilia (as high as 70–80%) in some cases.
- Hemoglobin level is usually low.
- Hypoalbuminemia is common in severe disease.
Question 95. Write short essay/note on
Answer:
- Treatment of ancylostomiasis
- Mention the drugs used in ancylostoma infestation.
Treatment/Management:
- Drugs: A single dose of albendazole in the dose of 400 mg is the treatment of choice. Other drugs include mebendazole (100 mg twice daily for 3 days) and pyrantel pamoate (single oral dose of 11 mg/kg). Eradication of infestation should be confirmed by follow-up stool examination 2 weeks after discontinuation of treatment.
- Improvement of nutrition with a high-protein diet.
- Anemia: Mild anemia usually responds to oral iron therapy. Severe anemia is often associated with anasarca, and heart failure. This should be treated carefully, if required with blood transfusions.
Cutaneous Larva Migrans (Creeping Eruption):
Question 96. Write short note on cutaneous larva migrans (creeping eruption).
Answer:
- Definition: Cutaneous larva migrans (CLM) is a creeping eruptions in the skin caused by the filariform larvae which aimlessly wander through the skin for weeks or months producing a reddish itch papule along the path of travel of the larvae (termed “larva migrans”).
- Cause: Cutaneous larva migrans is particularly observed with nonhuman hookworms namely Ancylostoma braziliense and A. caninum.
- Strongyloides stercoralis may produce creeping eruptions and its filariform larvae moves rapidly in short line; hence it is termed larva currens (reflecting fast movement of strongyloides larva).
- Sites affected: The main sites affected in cutaneous larva migrans are the dorsum and sole of the feet, buttocks, pelvic waist, legs, and shoulders. Infestation is usually self-limited. It can persist for a few days to months, but rarely for years.
- Loeffler’s syndrome (transient migratory pulmonary infiltrates, peripheral blood eosinophilia, and sputum eosinophilia) is found in many cases of cutaneous larva migrans.
Cutaneous Larva Migrans Clinical Features:
There are usually no systemic symptoms and diagnosis is purely made on clinical grounds. Skin shows multiple, clearly defined, intensely pruritic, linear, and serpiginous tracts.
Strongyloidiasis:
Question 97. Discuss clinical features, diagnosis, and treatment of strongyloidiasis.
Answer:
- Strongyloidiasis is an intestinal infection of humans caused by Strongyloides stercoralis.
- The adult worm of Strongyloides stercoralis is small and measures 2 mm in length. It is found in many parts of the tropics and subtropics. It lives in the small intestine namely upper part of jejunum.
Strongyloidiasis Treatment:
- Ivermectin: It is the drug of choice and given as a single dose in the dose of 150–200 µg/kg.
- Alternative drug is albendazole (200 mg twice a day for 5–7 days).
- Antihistamines may be given for intense itching.
Strongyloidiasis Clinical Features:
- Skin lesions: Larval migration through skin may produce itchy rashes at the site of entry. Autoinfection may produce migratory linear weal around the buttocks and lower abdomen (cutaneous larva currens).
- The classic triad of symptoms is:
- Abdominal Pain
- Diarrhea
- Urticaria.
- Pulmonary lesions: Larval migration through lungs may produce cough, dyspnea, hemoptysis, and bronchospasm.
- Intestinal lesions: Adult worms in the intestine may cause abdominal pain, bloating diarrhea, steatorrhea (malabsorption),
and weight loss. In heavy infections, damage to the small intestinal mucosa can cause even perforation. - Systemic infection: Systemic strongyloidiasis (the Strongyloides hyperinfection syndrome), with dissemination of larvae throughout the body. It may occur in immunocompromised states (e.g., HIV infection and corticosteroid therapy). This may produce diarrhea, severe, generalized abdominal pain, abdominal distension, pneumonia, meningoencephalitis, shock, and death. Gram-negative sepsis may develop.
Diagnosis:
Stool examination: Motile larvae (at least three samples) may be observed on microscopic examination of stool, especially after a period of incubation. Larvae may also be cultured from stool.
Blood: Eosinophilia may be found in some patients.
Duodenal/jejunal aspirate may show larvae.
Serological tests: ELISA for antibody to S. stercoralis.
In patients with disseminated infection: Stool examination, ELISA, and sputum and blood examination for larvae.
Question 98. Write short note on management of strongyloidiasis.
Answer:
Treatment:
- Ivermectin (200 µg/kg/day) for 2 days is the drug of choice.
- Albendazole: It is an alternative drug but is less effective drug. It is given orally at a dose of 400 mg twice daily for 7 days.
- Disseminated systemic infections: Both ivermectin and albendazole should be given and may be continued for 7 days or till the parasites are cleared.
Filariasis:
Question 99. Discuss the clinical features, diagnosis, and treatment of filariasis.
Answer:
Conditions included under filariasis:
- Lymphatic filariasis: Caused by Wuchereria bancrofti and Brugia malayi.
- Onchocerciasis (river blindness): Caused by Onchocerca volvulus
- Loiasis: Caused by Loa loa
Lymphatic Filariasis:
Question 100. Write short essay on lymphatic filariasis and bancroftian filariasis and their clinical manifestations and management.
Answer:
Clinical outcome of lymphatic filariasis may range from subclinical infection to hydrocele and elephantiasis. Life cycle of filariasis is presented.
Clinical Features:
Question 101. Write short essay on clinical features of filariasis.
Answer:
Acute filarial lymphangitis: It presents with fever, with chills and sweats, headache, and muscle pain. On examination there will be pain, tenderness, and erythema along the course of inflamed lymphatic vessels.
The whole episode may last for few days, but it may recur several times in a year. Temporary edema becomes more persistent and regional lymph nodes (e.g., inguinal) are enlarged painful and tender. Inflammation of the spermatic cord (funiculitis), epididymis (epididymitis), and testis (orchitis) is common.
- Chronic stage of the disease: It is characterized by development of permanent lymphedema (“elephantiasis”) of legs, scrotal edema, chylous ascites, chylous pleural effusion, and chyluria.
- Progressive enlargement of lymphedema may produce coarsening, corrugation, fissuring and bacterial infection of the skin, and subcutaneous tissue. This results in irreversible “elephantiasis.” The scrotum may become very large in size because of hydrocele. Chyluria and chylous effusions appear as milky and opalescent which on standing shows fat globules on the top of chylous fluid.
- Tropical pulmonary eosinophilia.
Question 102. Write short essay on laboratory findings in filariasis.
Answer:
- In the early stages of lymphangitis diagnosis can be made on clinical grounds, and peripheral blood eosinophilia.
- Microfilariae are not seen in the peripheral blood during early infestation.
- Peripheral blood:
- Eosinophilia: Host immune response to the parasite produces massive eosinophilia.
- Demonstration of microfilariae: In the peripheral blood at night.
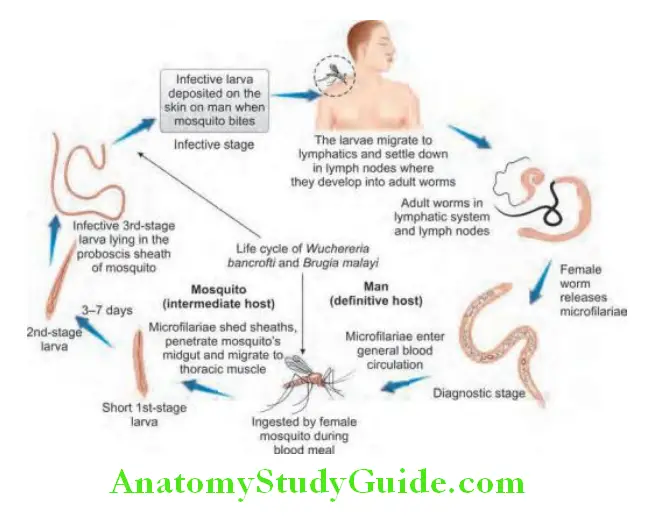
- Demonstration of microfilariae in other fluids: Microfilariae can be demonstrated in the hydrocele fluid and may occasionally show an adult worm.
- Diethylcarbamazine (DEC) provocation test: In this test, administration of 100 mg DEC usually produces positive blood specimens within 30–60 minutes.
- Radiology: Calcified filariae may be demonstrable by radiography.
- Serological tests:
- Antigen detection and antibody detection in the blood: By ELISA or immunochromatic method.
- PCR for DNA detection.
- Radionuclide lymphoscintigraphy: It is used to detect widespread lymphatic abnormalities.
- Tropical pulmonary eosinophilia: Serology is strongly positive and IgE levels are markedly raised but circulating microfilariae are not found. The chest X-ray shows miliary changes or mottled opacities. Lung function tests show a restrictive picture.
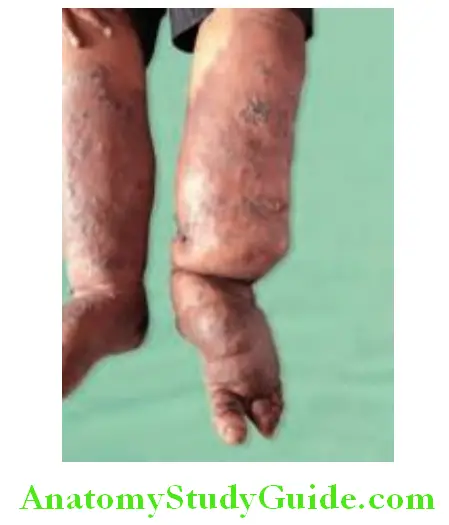
Filariasis Treatment:
- Aim of treatment: Reversing and halting the disease progression.
- Diethylcarbamazine (DEC) in the dose of 6 mg/kg in three divided doses for 12–21 days kills both microfilariae and adult worms.
- Adverse effects: Due to the host response to dying microfilariae. The intensity of reaction directly proportional to the microfilarial load. The main symptoms include fever, headache, nausea, vomiting, arthralgia, and prostration. They usually develop within 24–36 hours of the first dose of DEC. Antihistamines or corticosteroids may be needed to control these allergic reactions.
- Ivermectin kills only microfilariae, but not the adult worms.
- Combined albendazole (400 mg) and ivermectin (200 μg/kg) in a single dose, with or without DEC (300 mg), are also highly effective.
- Doxycycline 200 mg per day for 4 weeks plus ivermectin as a single dose (200 µg/kg provides additional benefit by eliminating the bacteria. This interrupts the parasite embryogenesis.
- Surgery may be of useful in the treatment established elephantiasis.
Filariasis Chronic lymphatic pathology:
- Prevention of secondary infection: Advised to take meticulous care of skin in lymphedematous limbs to prevent secondary bacterial and fungal infections.
- Control of lymphedema: Tight bandaging, massage and bed rest with elevation of the affected limb to control the lymphedema.
- Prevention of further damage: Prompt diagnosis and antibiotic therapy of bacterial cellulitis to prevent further lymphatic damage and worsening of existing elephantiasis.
- Surgery: Plastic surgery may be required in established elephantiasis. Hydroceles and chyluria can be repaired by surgery.
Nosocomial Infections (Hospital Infections):
Nosocomial Infections Introduction:
Question 103. Write short essay/answer on nosocomial infections.
Answer:
Nosocomial infection is the term derived from two Greek words “nosos” (disease) and “komeion” (to take care of).
Nosocomial Infections Definition: In its broad meaning, nosocomial infection is defined as any infection or disease that any individual suffers from the invasion of pathogens in hospital.
Requisites for using the term nosocomial infections are:
- Manifestation of the symptoms, first appear 48 hours or more after hospital admission or within 30 days after discharge from hospital.
- Any infection or disease which does not exist or is not in incubation period when the clients are hospitalized. They are the result of treatment in a hospital or a healthcare service unit, but secondary to the patient‘s original condition.
- Infection is caused by the invasion of disease-producing microorganisms acquired in hospital.
Health Care-associated Infection (HAI):
An infection that develops in a patient who is cared for in any setting where healthcare is delivered (e.g., acute care hospital, chronic care facility, ambulatory clinic, dialysis center, and surgicenter, home).
Classifiation of Nosocomial Infection:
According to the site of infections, nosocomial infection has the possibility to occur in every system and every site.
According to the source of the pathogens, nosocomial infections can be classified into:
- Reactivation of latent infection: TB, herpes viruses.
- Endogenous infections (autogenous infections): From normal commensals of the skin, respiratory, GI, and GU tract are common. It can occur when part of the individual’s flora becomes altered and results in an overgrowth of microorganism.
- When sufficient numbers of microorganisms normally found in one body cavity or lining are transferred to another body site, an endogenous infection develops. For example, transmission of enterococci, normally found in fecal material, from hands to the skin is a cause of wound infections.
- Exogenous infections (cross infections): They are due to the invasion of causative microorganisms from the source other than the individual’s themselves, such as from hospital personnel, the other individuals, various objects, and hospital environment.
- Inanimate environment: Aspergillus from hospital construction, Legionella from contaminated water.
- Animate environment: Hospital staff, visitors, other patients, and cross transmission are common.
Factors Predisposing to Hospital Infection
- Pre-existing condition
- Need for invasive devices
- Effect of surgery [(skin wound, tissue trauma, opening colonized viscus, immobilization, and implants of foreign material (joint prostheses, arterial graft)]
- Effect of antibiotic treatment (colonization by resistant bacteria and fungi)
- Effect of immunosuppressive treatment (corticosteroids, cancer chemotherapy, radiotherapy, and transplant immunosuppression)
- Exposure to healthcare workers and other patients, who may transmit pathogens.
- Exposure to pathogens in the hospital environment
Epidemiology: It develops in ~10% of patients admitted to hospital.
Nosocomial Infections Etiology: Staphylococcus aureus, Enterobacteria, Pseudomonas aeruginosa, Acinetobacter species, coagulase-negative Staphylococci, Enterococci, and Candida are the most frequent pathogens.
Mechanisms of Transmission:
- Contact: Direct (person-person), indirect (transmission through an intermediate object-contaminated instruments)
- Airborne: Organisms that have a true airborne phase as pattern of dissemination (TB, Varicella)
- Common-vehicle: Common animate vehicle as agent of transmission (ingested food or water, blood products, and IV
fluids) - Droplet: Brief passage through the air when the source and patient are in close proximity
- Arthropod borne
Main Groups of Nosocomial Infection:
Question 104. Write short essay/answer on common types of nosocomial infections.
Answer:
Intravascular Catheter-related Infections
- Localized: Peripheral thrombophlebitis, abscess
- Systemic: Bacteremia, sepsis, endocarditis, and metastatic infection.
Microbiology: Coagulase-negative staphylococci (S. epidermidis), S. aureus, Candida species, Enterococcus species, and gram-negative rods.
Main Groups of Nosocomial Infection Clinical features:
- Local signs of inflammation at the site of entry: Redness, tenderness, swelling, slight purulent exudate—prominent in peripheral lines, less in central lines.
- Systemic signs: Fever, rigors, and no other source of fever
- Clinical improvement after catheter removal
- Elevated inflammatory markers, typical pathogens in blood culture.
Main groups of nosocomial infections:
- Catheter-related infections
- Pneumonia
- Respiratory tract infections other than pneumonia
- Surgical site infections
- Urinary tract infections
- Gastrointestinal infections
- Central nervous system infections
Management of Catheter/Line-related Sepsis:
- Two blood cultures, one from catheter, one from peripheral to differentiate colonization versus catheter-related bacteremia
- Catheter removal, culture of the tip—semiquantitative >15 CFU = infection (insertion of a new catheter not immediately)
- Antibiotics, if symptoms did not resolve after removal, guided by culture results.
- Long-time catheters (Broviac, Hickman)—catheter treatment, antibiotic locks
Prevention and Control:
- Choice of catheter and insertion site
- Aseptic and atraumatic insertion (skin disinfection and barrier precautions), fixation to the skin
- TPN—total parenteral nutrition (all-in-one bags) prepared in the pharmacy under sterile precautions
- Minimize opening of the IV set for additive drugs, every opening strictly aseptic
- Maintaining adequate hygiene and dressing of the insertion site and regular review of insertion site
- Replacing giving set and catheter at appropriate intervals
- Giving set every 72 hours, if lipid emulsions or blood products—24 hours
- Catheter—peripheral 48 hour, central 7 days, tunneled line much longer
- Removal of catheter if no longer necessary
Hospital-acquired, Nosocomial Pneumonia:
Prevention:
- Pulmonary toilet
- Change position second hourly: Elevate head to 30–45°
- Deep breathing, incentive spirometry
- Frequent suctioning
- Bronchoscopy to remove mucous plugging
Other Respiratory Tract Infections:
Tracheobronchitis, sinusitis—frequent in mechanically ventilated patients’ risk factor for development of nosocomial pneumonia and sepsis.
Urinary Tract Infections (UTI)—Urinary Catheter-related Infections:
Source of uropathogens:
- Endogenous: Most common
- Catheter insertion
- Retrograde movement up the urethra (70–80%)
- Patient’s own enteric flora (E. coli)
- Exogenous:
- Cross contamination of drainage systems
- May cause clusters of UTI’s.
Main Groups of Nosocomial Infection Etiology: Gram-negatives (E. coli, Klebsiella pneumoniae, Pseudomonas, and Proteus species), enterococci, and Candida
species.
Risk factors: Length of catheterization (5%/day), opened drainage system, diabetes, female sex, and age above 50 years.
Colonization is often asymptomatic, but there is a risk of ascending infection—pyelonephritis, bacteremia, and sepsis.
Main Groups of Nosocomial Infection Diagnosis:
Pyuria: >5 leukocytes per high-power field
Urine culture: Significant bacteriuria ≥105 CFU/mL.
Treatment:
- Catheter removal or change
- Antibiotics in symptomatic UTI
Prevention:
- Avoid catheter when possible and discontinue ASAP—MOST IMPORTANT
- Aseptic insertion by trained healthcare workers
- Maintain closed system of drainage
- Ensure dependent drainage
- Minimize manipulation of the system
- Silver coated catheters
- Maintenance of good patient hygiene
- Replacing catheter at appropriate intervals
- ICU-bacteriological screening—urine culture at appropriate intervals
Surgical Site Infections (SSI):
Infections in the site of surgery and related bacteremias
CDC classifiation
- Superficial SSI: Skin and subcutaneous tissue involved
- Deep SSI: Deep soft tissue, fascia, and muscles
- Organ SSI: Organs and cavities.
Microbiology: S. aureus, coagulase-negative staphylococci, enterococci, E. coli, Pseudomonas, and Enterobacter.
Gastrointestinal Infections:
Antibiotic-associated colitis:
Precipitated by previous antibiotic treatment, mainly lincosamides, ampicillin, and cephalosporins.
Clostridium difficile toxin A (enterotoxin) and toxin B
(cytotoxin)
Transmission via spores may cause hospital epidemics.
- Postantibiotic diarrhea, simple colitis
- Pseudomembranous colitis
- Toxic megacolon
Main Groups of Nosocomial Infection Diagnosis:
Clinical: Diarrhea, fever, abdominal pains, vomiting, and related to antibiotic treatment.
Laboratory: C. difficile toxin detection—ELISA, antigen detection—latex agglutination (stool culture on selective agar, cytotoxicity test on tissue culture = gold standard, rarely available).
Colonoscopy: Typical morphology with pseudomembranes,
typical histology in biopsy specimens.
Central Nervous System Infections:
CSF shunt-related, rare occurrence, mostly Staphylococcus aureus and Pseudomonas, often serious course (nosocomial ventriculitis) and high mortality.
Strategies to Reduce Nosocomial Infection:
Question 105. Write short essay/answer on measures to minimize nosocomial infection.
Answer:
- Modify host: Risk factors such as age, underlying disease are difficult to change.
- Reduce patient exposure to pathogens.
- Reduce the number and virulence of nosocomial pathogens.
Exposure Reduction:
- Use of aseptic technique during patient care
- Regular handwashing
- Proper isolation of patients known or suspected of harboring infectious diseases
Handwashing frequently is the single most important measure to prevent nosocomial infections.
Antibiotic Prophylaxis
Antibiotics: Bactericidal, effective against considered pathogens, with respect to local resistance, not betalactamases inducers. Short course of prophylaxis—first dose IV at induction of anesthesia, second dose if surgery >3 hours.
Prevention of SSI
- Preoperative:
- Minimizing the preoperative hospital stay
- Cutting better than shaving of surgical site
- Antibiotic prophylaxis, if indicated
- Surgical team clothing and washing
- Skin decolonization
- Chlorhexidine
- Intranasal mupirocin for S. aureus carriers
- Intraoperative:
- Regardful (tissue-saving) operation technique
- Bleeding control
- Excision of foreign bodies and devitalized tissue
- Isolation of incised bowel
- Minimizing of surgical staff and their movement
- Postoperative: Aseptic care of the site, sterile covering
Strategies to Reduce Nosocomial Infection Treatment:
- Stop precipitating antibiotic
- Rehydration, diet
- Antibiotic: Per oral. Metronidazole 3–4 × 500 mg p.o. or vancomycin 4 × 125–250 mg treat at least 7 days
- Probiotic drugs, colestipol, and rifaximin have been tried.
- Toxic megacolon—colectomy
Sepsis:
Question 106. Discuss management of a patient with septic shock.
Answer:
Systemic Inflammatory Response Syndrome (SIRS):
- SIRS is the body’s response to a clinical insult (e.g., infection, inflammation, stress, trauma, and burns)
- Must have at least two of the following:
- Temperature >38.5°C or <36°C
- Heart rate >90 beats/min
- Respiratory rate >20 breaths/min or PaCO2 <32 mm Hg WBC >12,000 cells/mm 3, <4,000 cells/mm 3, or >10% immature (band) forms
Question 107. Write short note on sepsis/SIRS/ MODS/Septic shock.
Answer:
Sepsis: SIRS + suspected or confirmed infection (documented
via cultures or visualized via physical exam/imaging). Latest definition of sepsis—Sepsis is defined as lifethreatening organ dysfunction caused by the dysregulated host response to infection. (Sepsis = suspected infection PLUS q SOFA = 2) q SOFA3 criteria: Hypotension-systolic BP <100 mm Hg, altered mental statusGCS <13, respiratory rate >2A continuum of severity describing the host systemic inflammatory response
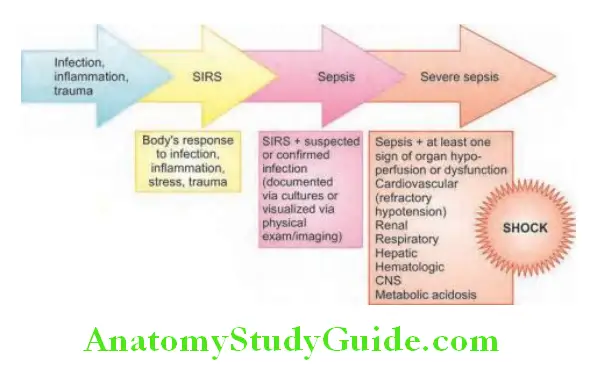
Severe sepsis Septic shock:
All three must be met within 6 hours:
Documentation of a suspected source of infection
Two or more manifestations of SIRS criteria:
- Temperature >38.3°C/101 F or <36°C/96.8°F
- Heart rate >90
- Respiratory rate >20
- WBC >12 or <4 or >10% bands
Organ dysfunction, evidenced by any one of the following:
- SBP <90 or MAP <65, or a SBP decrease of more than 40 patients
- Cr >2.0 or urine output <0.5 cc/kg/ hour for 2 hours
- Bilirubin >2 mg/dL (32.4 mol/L)
Or if a provider documents severe sepsis, r/o sepsis, possible sepsis, or septic shock
There must be documentation of septic shock present and
Tissue hypoperfusion persisting in the hour after crystalloid fluid administration, evidenced by:
- SBP <90
- MAP <65
- Decrease in SBP by >40 points from the patients baseline
- Lactate ≥4
Or if the criteria are not met, but there is provider documentation of septic shock or suspected septic shock
Multiple organ dysfunction syndrome (MODS)
- Altered organ function in an acutely ill patient
- Homeostasis cannot be maintained without intervention
Etiology of Sepsis:
Septic shock may be caused by gram-positive (most common) or gram-negative bacteria, fungi, and, very rarely, protozoa or Rickettsiae. The common gram-positive bacteria include Staphylococcus aureus, enterococci, Streptococcus pneumoniae, and gram-negative bacilli which are resistant to usual antibiotics.
Management of Sepsis:
Surviving Sepsis Campaign:
- Step 1: Identify sepsis
- Step 2: Categorize sepsis
- Step 3: Initiate treatment
Risk factors for sepsis are listed in Table 4.46 and pathogenesis of septic shock is illustrated
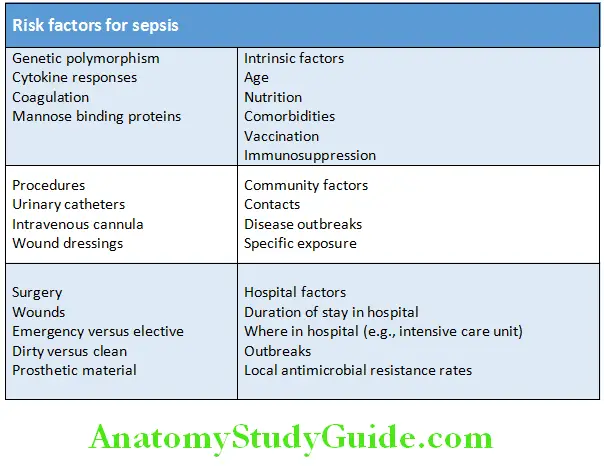
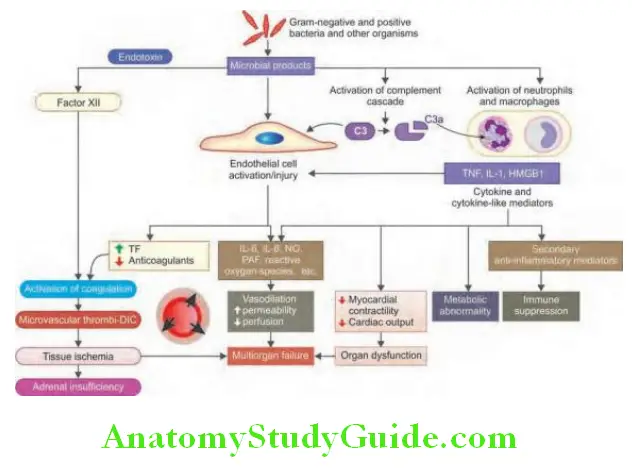
Identifying acute organ dysfunction as a marker of severe sepsis is presented in Table
- Initial resuscitation: Volume resuscitation with normal saline 30 mL/kg over first 30 minutes → if mean arterial pressure <70 mm Hg start inotropes (discussed later).
- Diagnosis:
- Blood culture: Before the initiation of antimicrobial therapy, at least two blood cultures should be obtained at least one drawn percutaneously at least one drawn through each vascular access device if inserted longer than 48 hours.
- Other cultures such as urine, cerebrospinal fluid, wounds, respiratory secretions, or other body fluids should be obtained as per the clinical situation.
- Other diagnostic studies such as imaging and sampling should be performed promptly to determine the source and causative organism of the infection.
- Antibiotic therapy:
- Start intravenous antibiotic therapy within the first hour of recognition of severe sepsis after obtaining appropriate cultures.
- Empirical choice of antimicrobials should include one or more drugs with activity against likely pathogens, both bacterial or fungal.
- Penetrate presumed source of infection
- Guided by susceptibility patterns in the community and hospital
- Continue broad spectrum therapy until the causative organism and its susceptibilities are defined.
- Source control: Evaluate patients for focus of infection amenable to source control measures
- Fluid therapy
- Crystalloids: Always use first!
- Colloids: FFP if coagulopathy, RBCs if HCT <30, especially, if risk of CAD. Avoid albumin (increase ECF and is costly).
- Vasopressors
- Initiate vasopressor therapy if appropriate fluid challenge fails to restore adequate blood pressure and organ perfusion.
- Either norepinephrine or dopamine is first-line agents to correct hypotension in septic shock.
- Norepinephrine is more potent and may be more effective than dopamine at reversing hypotension in septic shock patients.
- Dopamine may be particularly useful in patients with compromised
systolic function but causes more tachycardia and may be more arrhythmogenic.
- Inotropic therapy
- In patients with low cardiac output despite of adequate fluid resuscitation, dobutamine can be used to increase cardiac output.
- Should be combined with vasopressor therapy in the presence of hypotension.
- Steroids:
- In patients with septic shock who require vasopressor therapy to maintain blood pressure intravenous corticosteroids are recommended.
- Administer intravenous hydrocortisone 200–300 mg/day for 7 days in three or four divided doses or by continuous infusion.
- Recombinant human activated protein C (rhaPC) [drotrecogin alfa
(activated)]: It is recommended in patients at a high risk of death- APACHE II (Acute Physiology and Chronic Health Evaluation II) score >25, or
- Sepsis-induced multiple organ failure, or
- Septic shock, or
- Sepsis induced acute respiratory distress syndrome
- Patients should have no absolute or relative contraindication related to bleeding risk that outweighs the potential benefit of rhAPC.
- Blood product administration: Red blood transfusion should be given only when hemoglobin decreases to <7 g/dL. Target hemoglobin of 7–9 g/dL.
- Mechanical ventilation
- Sepsis-induced acute lung injury (ALI)/adult respiratory distress syndrome (ARDS)
- High tidal volumes, >6 mL/kg, coupled with high plateau pressures, >30 cm H 2O, should be avoided. A minimum amount of positive end expiratory pressure should be set to prevent lung collapse at end-expiration.
- Sedation, analgesia, and neuromuscular blockade in sepsis
- The use of protocols and daily interruption/lightening of continuous sedative infusion have shown to reduce the duration of mechanical ventilation and length of stay.
- Glucose control: Following initial stabilization of patients with severe sepsis, blood glucose to <150 mg/dL. Glycemic control strategy must include a nutrition protocol with the preferential use of the enteral route.
- Renal replacement: Continuous veno-venous hemofiltration and intermittent hemodialysis are considered equivalent in acute renal failure. Continuous hemofiltration offers easier management of fluid balance in hemodynamically unstable septic patients.
- Bicarbonate therapy: Bicarbonate is not recommended for improving hemodynamics or reducing vasopressor requirements or for the treatment of hypoperfusion induced lactic academia with pH >7.1
- Deep vein thrombosis prophylaxis: DVT prophylaxis with either low-dose unfractionated heparin or low molecular weight heparin
should be used in severe sepsis patients. - Stress ulcer prophylaxis: Stress ulcer prophylaxis should begiven to all patients with severe sepsis. H 2 receptor blockers are more efficacious than sucralfate and are the preferred agents. Proton pump inhibitors compared to H 2 blockers have not been assessed.
Principles of sepsis management:
- Recognize sepsis early and determine severity.
- Organ dysfunction is determined by Sequential Organ Failure Assessment Score (SOFA) which include paO2, bilirubin, creatinine, and platelet count.
- Early antibiotics are critical to resolution of shock.
- Resuscitate severe sepsis and septic shock as soon as possible.
- Early goal directed therapy
“Surviving sepsis campaign (SSC)”: International Guidelines for management of sepsis and septic shock.
Question 108. Discuss key elements in first hour sepsis bundles.
Answer:
The fie key elements of hours-1 bundle:
- Measure lactate level. Remeasure if initial lactate is >2 mmol/L.
- Obtain blood culture prior to administration of antibiotics.
- Administer broad-spectrum antibiotics.
- Begin rapid administration of 30 mL/kg crystalloid for hypotension or lactate ≥4 mmol/L
- Apply vasopressors if patient is hypotensive during or after fluid resuscitation to maintain MAP >65 mm Hg.
Sepsis bundles Anti-Infective Therapy:
Antimicrobial Combination Therapy:
Limit combination therapy to special situations:
- Synergistic killing, e.g., β-lactam plus aminoglycoside for endocarditis
- Mixed infections or severe infections of unknown cause, e.g., limb infections in diabetic patients
- To prevent the emergence of resistance, e.g., M. tuberculosis
- Initial empiric therapy
Multidrug Therapy:
Tuberculosis, infective endocarditis, malaria, HIV, and leprosy
Antibiotic Chemoprophylaxis:
- Infective endocarditis: Amoxicillin + gentamicin
- Splenectomy: Penicillin
- Rheumatic fever: Penicillin
- Tuberculosis: INH
- Meningitis: Meningococcal and H. influenzae: Rifampicin Beta-lactam antibiotics
Question 109. Write a short essay/note on β-lactam antibiotics. (Or) Mention four β-lactam antibiotics with dosage. (Or) Mention cephalosporins and name drugs belonging to third generations.
Answer:
These antibiotics have a β-lactam ring structure and their bactericidal (lysis of bacteria) action is due to their inhibition enzymes involved in cell wall formation in the bacteria [penicillin binding proteins (PBP)].
β-lactam antibiotics Types of beta-lactam antibiotics:
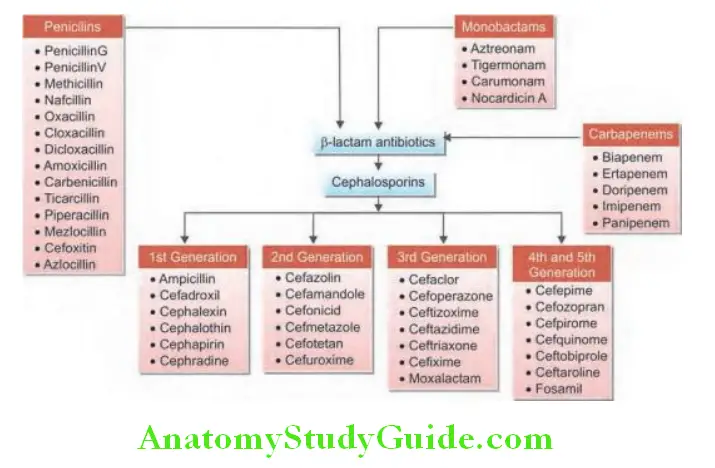
Adverse effects of beta-lactam antibiotics:
Drug Resistance:
Antibiotic resistance is a challenging and growing problem. The most important mechanism of drug resistance is destruction of drugs by beta-lactamase enzymes.
β-lactamases:
Question 110. Write a short essay/note on β-lactamases.
Answer:
- β-lactam antibiotics have a β-lactam ring structure. They exert a bactericidal action by inhibiting enzymes involved in cell wall synthesis [penicillin binding proteins (PBP)].
- β-lactamases are bacterial enzymes produced by many gram-positive and gram-negative bacteria. Theses enzymes can inactivate β-lactam antibacterials by hydrolysis of β-lactam ring structure and results in infective compounds.
- Production of β-lactamases by these bacteria is the most important factor that contributes to β-lactam antibiotic resistance.
- Many serine-active β-lactamases inhibitors (e.g., clavulanic acid, sulbactam, and tazobactam) in combination with β-lactam antibiotics are used to reduce drug resistance by bacteria containing β-lactamases.
β-lactamases Adverse effects of beta-lactam antibiotics:
- Generalized allergy to penicillin
- Gastrointestinal upset and diarrhea
- Mild reversible hepatitis
- Leukopenia, thrombocytopenia and coagulation deficiencies, and interstitial nephritis and potentiation of aminoglycoside-mediated renal damage
- Thrombophlebitis with parenteral β-lactams
β-lactamases Extended-Spectrum β-Lactamases (ESBL):
Several isolates of Enterobacteriaceae and Pseudomonas have been identified which produce newer β-lactamases. These enzymes include
Plasmid-Mediated Cephamycinases,
Extended-Spectrumβ-Lactamases (Esbls)
Carbapenemhydrolyzing Enzymes Or Carbapenemases.
Major Sources of Lactamases:
- Most commonly β-lactamases are produced by Klebsiella species, Escherichia coli but can occur in other GNB (gram-negative bacilli), including Enterobacter, Proteus, Salmonella, and Citrobacter species, Morganella morganii, Shigella dysenteriae, Serratia marcescens, Pseudomonas aeruginosa, and Burkholderia cepacia.
- Mutant, plasmid-mediated β-lactamases enzymes are derived from amino acid substitutions in native β-lactamases (particularly TEM-1, TEM-2, and SHV-1). Typically associated with multidrug resistance (fluoroquinolones, cotrimoxazole, and aminoglycosides).
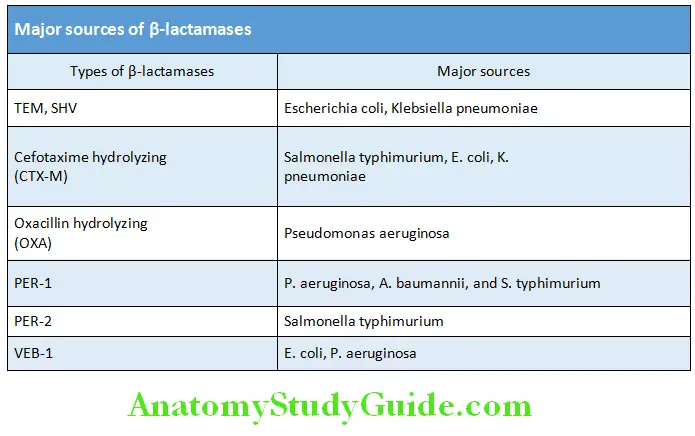
β-lactamases Actions:
- β-lactamases are able to hydrolyze all penicillins, cephalosporins (except cephamycins), and monobactams. These are associated with multidrug resistance (fluoroquinolones, cotrimoxazole, and aminoglycosides).
- The ESBLs are a group of enzymes capable of hydrolyzing and cause resistance to the oxyiminocephalosporins (cefotaxime, ceftazidime, ceftriaxone, cefuroxime, and cefepime) and monobactams (aztreonam), but not the cephamycins (cefoxitin and cefotetan) or carbapenems (imipenem, meropenem, doripenem, and ertapenem).
Risk factors: Predisposing to infection or colonization with ESBLproducing pathogens is presented in Table.
Treatment recommendations for infections with ESBL producers:
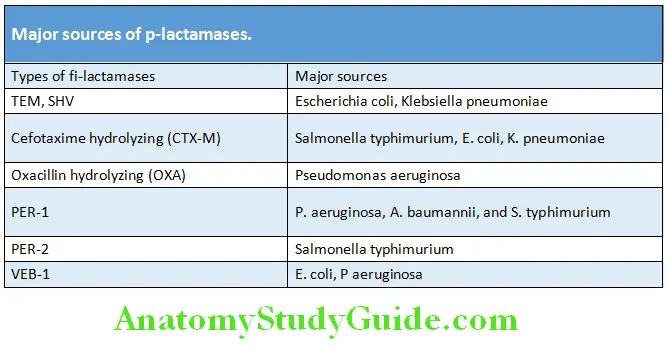
Carbapenem-Resistant Enterobacteriaceae
Klebsiella pneumoniae Carbapenemase (KPC)
Clonal outbreaks in New York, Israel, Greece, Colombia, Brazil, China, and Canada (Montreal)
Question 111. Write a short essay/note on New Delhi Metallo-β-lactamase (NDM-1).
Answer:
NDM-1 (New Delhi Metallo-β-lactamase)
- It is a novel metallo-β-lactamase (MBL) coded by a novel gene bla
(NDM-1) in Enterobacteriaceae. - First, it was reported in a Swedish male diabetic, who returned to
- Sweden after having treatment for gluteal abscess in December 2007 at a hospital in New Delhi. Since the immediate previous treatment was in New Delhi it was named as NDM-1.
- Subsequently, Enterobacteriaceae family containing this genetic element was detected in various regions of India, Pakistan, Bangladesh, Britain, and many other countries.
- Metallo-β-lactamase (MBL) enzymes mediate resistance to almost β-lactam agents (except monobactams, e.g., aztreonam) including carbapenems.
- Organism carrying this gene has multidrug resistance and is susceptible only to tigecycline and colistin

Miscellaneous:
Amikacin:
Question 112. Write short note on amikacin.
Answer:
- It is a semisynthetic derivative of kanamycin and has pharmacokinetics, dose, and toxicity.
- Outstanding feature: Amikacin has the wide spectrum of activity and effective against many organisms that are resistant to other aminoglycosides. However, it is effective in relatively higher dose against Pseudomonas, Proteus, and Staphylococci.
- Indications: The range of conditions in which amikacin can be used is as same as for gentamicin. It is reserved for hospital acquired gram-negative bacterial infections where gentamicin/tobramycin resistance is high. It is effective against tuberculosis, but rarely used for its treatment.
- Toxicity: It causes more hearing loss than vestibular disturbance, nephrotoxicity, and neurotoxicity.
- Dose: 15 mg/kg/day in 1–3 doses; urinary tract infections 7.5 mg/kg/day. Best given as single daily infusion (minimizes the toxicity).
Indications of Doxycycline:
Question 113. Write short note on indications of doxycycline.
Answer:
Doxycycline is a tetracycline antibiotic. It is indicated for many different bacterial infections, such as acne, urinary tract infections, intestinal infections, eye infections, gonorrhea, chlamydia, etc.
Cephalosporins:
Question 114. Write short note on cephalosporins.
Answer:
Cephalosporins are group of semisynthetic antibiotics derived from cephalosporin-C obtained from a fungus Cephalosporium.
They are chemically related to penicillins.
- First generation cephalosporins: Cephalexin, cephradine, and cefadroxil
- Second generation cephalosporins: Cefuroxime, cefuroxime axetil, and cefaclor
- Third generation cephalosporins: Cefotaxime, ceftizoxime, ceftriaxone, ceftazidime, cefoperazone, cefixime, cefpodoxime, cefdinir, ceftibuten, and cefetamet pivoxil
- Fourth generation cephalosporins: Cefepime and cefpirome
Side effects of sulfonamides:
- Nausea, vomiting, and epigastric pain
- Crystalluria
- Hypersensitivity reactions
- Hepatitis
- Contact sensitization with topical use
- Hemolysis in persons with G6PD deficiency
- Precipitation of kernicterus in premature newborn
Side Effcts of Tetracycline, Chloramphenicol, and Quinolones
Question 115. Write short note on side effects of tetracycline, chloramphenicol, and quinolones.
Answer:
Side Effcts of Sulfonamides
Question 116. Write short note on side effects of sulfonamides.
Classifiation of Antifungal Drugs:
(or) Write short note on antifungal drugs.
Ketoconazole:
(or) Write short note on ketoconazole.
Answer:
Ketoconazole is antifungal drug belonging to the azole group. It inhibits enzymes involved in steroid hormone biosynthesis and prevents the growth of the fungus. Ketoconazole is available for oral administration (causes severe hepatitis and it has been discontinued in many countries) and for topical application (e.g., for the treatment of tinea, cutaneous candidiasis).
Antiviral Agents:
Ivermectin:
Question 117. Write short note on ivermectin.
Answer:
It is an extremely potent semisynthetic derivative of the antinematodal principle obtained from Streptomyces avermitilis.
Indications:
- Drug of choice for single dose treatment of onchocerciasis and strongyloidiasis
- Bancroftian and brugian filariasis
- Cutaneous larva migrans and ascariasis
- Scabies and head lice
Indications for Intravenous Gamma Globulin:
Question 118. Write short note on indications for intravenous gamma globulin.
Answer:
Indications for intravenous gamma globulin:
Transplantation:
- Allogeneic bone marrow transplantation
- Kidney transplantation with a high antibody recipient or with an ABO incompatible donor
- Hematopoietic stem cell transplantation in patients older than 20 years
Immunodefiiency:
- Common variable immunodeficiency (CVID)
- Primary immunodeficiency disorders associated with defects in humoral immunity
Others:
- Chronic inflammatory demyelinating polyneuropathy (CIDP)
- Chronic lymphocytic leukemia
- Immune-mediated thrombocytopenia
- Kawasaki disease
- Pediatric HIV type 1 infection:
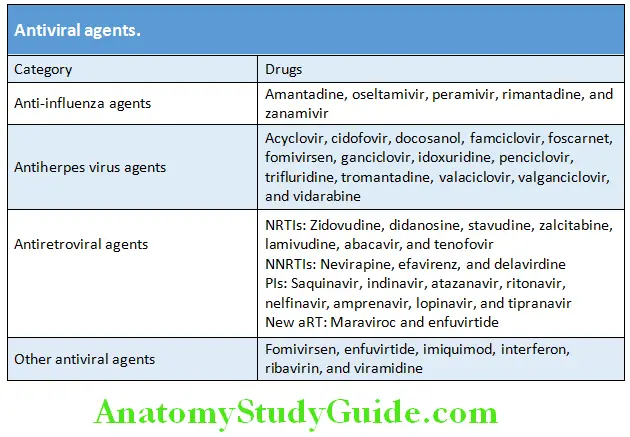
Commonly used Immunoglobulins:
- Human immunoglobulins:
- Human normal immunoglobulins: Hepatitis A, measles, rabies, tetanus, and mumps
- Human specific immunoglobulins: Hepatitis B, varicella, and diphtheria
- Non-human (antisera): Bacterial (e.g., diphtheria, tetanus, gas gangrene, and botulism) and viral (e.g., rabies)
Infections Associated with Skin Rash:
Question 119. Write short note on infections associated with skin rash.
Answer:

Infections associated with skin rash
Mnemonic: Very Sick Person Must Take Double Eggs.
- Varicella (Chicken pox): 1st day (rash is often 1st sign in children)
- Scarlet fever: 2nd day
- Pox (Small pox): 3rd day
- Measles (Rubella or 14 day measles): 4th day (Remember: Koplik spots appear in pre-eruptive phase on 2nd day of fever)
- Typhus: 5th day
- Dengue: 6th day
- Enteric fever: 7th day
Leave a Reply