The Liver, Biliary Tract and Exocrine Pancreas
Liver
Liver Normal Structure:
Table of Contents
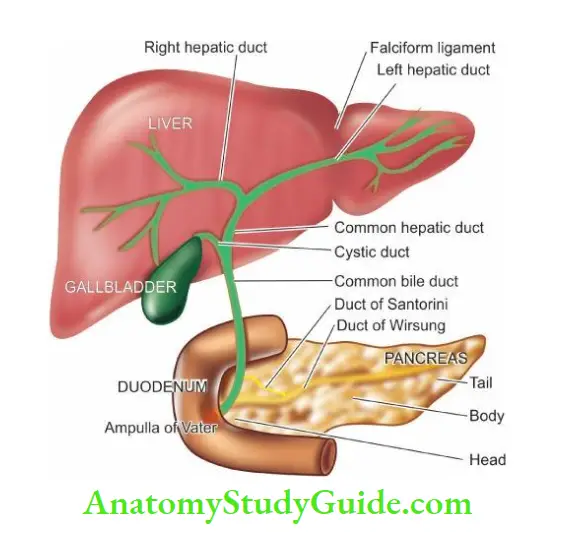
- Anatomy The liver is the largest organ in the body weighing 1400-1600 gm in the males and 1200-1400 gm in the females.
- There are 2 main anatomical lobes right and left, the right being about six times the size of the left lobe.
- The right lobe has a quadrate lobe on its inferior surface and a caudate lobe on the posterior surface.
- The right and left lobes are separated anteriorly by a fold of peritoneum called the falciform ligament, inferiorly by the fissure for the ligaments teres, and posteriorly by the fissure for the ligaments venous.
Read And Learn More: Systemic Pathology Notes
- The porta hepatis is the region on the inferior surface of the right lobe where blood vessels, lymphatics and common hepatic duct form the hilum of the liver.
- A firm smooth layer of connective tissue called Glisson’s capsule encloses the liver and is continuous with the connective tissue of the porta hepatis forming a sheath around the structures in the porta hepatis.
- The liver has a double blood supply the portal vein brings the venous blood from the intestines and spleen, and the hepatic artery coming from the coeliac axis supplies arterial blood to the liver.
- This dual blood supply provides sufficient protection against infarction in the liver.
- The portal vein and hepatic artery divide into branches to the right and left lobes in the porta.
- The right and left hepatic ducts also join in the porta to form the common hepatic duct.
- The venous drainage from the liver is into the right and left hepatic veins which enter the inferior vena cava.
- Lymphatics and the nerve fibres accompany the hepatic artery into their branchings and terminate around the porta hepatis.
Liver History:
- The hepatic parenchyma is composed of numerous hexagonal or pyramidal classical lobules, each with a diameter of 0.5 to 2 mm.
- Each classical lobule has a central tributary from the hepatic vein and at the periphery are 4 to 5 portal tracts or triads containing branches of the bile duct, portal vein and hepatic artery.
- Cords of hepatocytes and blood-containing sinusoids radiate from the central vein to the peripheral portal triads.
- The functioning lobule or liver acinus as described by Rappaport has a portal triad in the centre and is surrounded at the periphery by portions of several classical lobules.
- However, in most descriptions of the pathology of the liver, the term lobule is used in its classical form.
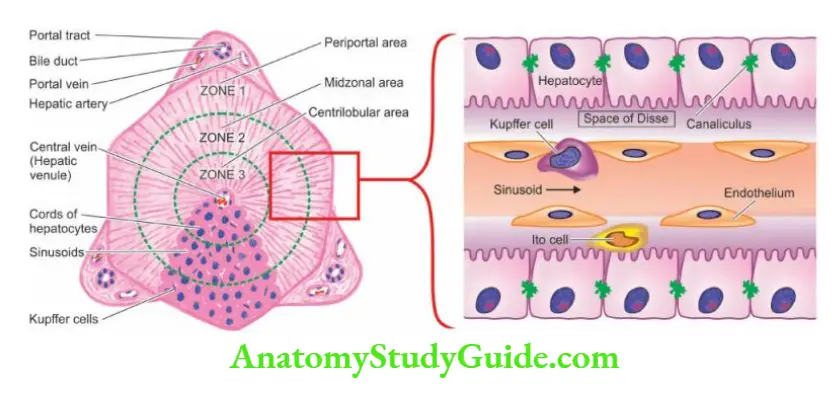
The blood supply to the liver parenchyma flows from the portal triads to the central veins. Accordingly, the hepatic parenchyma of the liver lobule is divided into 3 zones.
- Zone 1 or the periportal (peripheral) area is closest to the arterial and portal blood supply and hence bears the brunt of all forms of toxic injury.
- Zone 3 or the centrilobular area surrounds the central vein and is most remote from the blood supply and thus suffers from the effects of hypoxic injury.
- Zone 2 is the intermediate mid-zonal area.
- The hepatocytes are polygonal cells with a round single nucleus and a prominent nucleolus. The liver cells have a remarkable capability to undergo mitosis and regeneration.
- Thus it is not uncommon to find liver cells containing more than one nucleus and having polyploidy up to octoploid.
- A hepatocyte has 3 surfaces: one facing the sinusoid and the space of Disse, the second facing the canaliculus, and the third facing neighbouring hepatocytes.
- The blood-containing sinusoids between cords of hepatocytes are lined by discontinuous endothelial cells and scattered flat Kupffer cells belonging to the reticuloendothelial system.
- The space of Disse is the space between hepatocytes and sinusoidal lining endothelial cells. A few scattered fat-storing Ito cells lie within the space of Disse.
- The portal triad or tract besides containing the portal vein radicle, the hepatic arteriole and bile duct, has a few mononuclear cells and scanty connective tissue considered to be an extension of Glisson’s capsule.
- The portal triads are surrounded by a limiting plate of hepatocytes. The intrahepatic biliary system begins with the bile canaliculi interposed between the adjacent hepatocytes.
- The bile canaliculi are simply grooves between the contact surfaces of the liver cells and are covered by microvilli.
- These canaliculi join at the periphery of the lobule to drain eventually into terminal bile ducts or ductules (canal of Hering) which are lined by cuboidal epithelium.
Functions The liver performs multifold functions listed below:
- Manufacture and excretion of bile.
- Manufacture of several major plasma proteins such as albumin, fibrinogen and prothrombin.
- Metabolism of proteins, carbohydrates and lipids.
- Storage of vitamins (A, D and B12) and iron.
- Detoxification of toxic substances such as alcohol and drugs.
Liver Cell Necrosis
Closely linked to microanatomy of the liver are different types of liver cell necrosis. All forms of injury to the liver such as microbiologic, toxic, circulatory or traumatic, result in necrosis of liver cells. The extent of involvement of hepatic lobule in necrosis varies. Accordingly, liver cell necrosis is divided into 3 types: diffuse (submassive to massive), zonal and focal.
1. Diffuse (Submassive To Massive) Necrosis:
- When there is extensive and diffuse necrosis of the liver involving all the cells in groups of lobules, it is termed diffuse, or submassive to massive necrosis. It is most commonly caused by viral hepatitis or drug toxicity.
2. Zonal Necrosis:
- Zonal necrosis is necrosis of hepatocytes in 3 different zones of the hepatic lobule. Accordingly, it is of 3 types; each type affecting the respective zone is caused by different etiologic factors
- Centrilobular necrosis: is the commonest type involving hepatocytes in zone 3 (located around the central vein).
- Centrilobular necrosis is a characteristic feature of ischaemic injuries such as in shock and CHF since zone 3 is farthest from the blood supply.
- Besides, it also occurs in poisoning with chloroform, carbon tetrachloride and certain drugs.
- Midzonal necrosis: this is uncommon and involves zone 2 of the hepatic lobule.
- This pattern of necrosis is seen in yellow fever and viral hepatitis.
- In viral hepatitis, some of the necrosed hepatocytes of the mid-zone are transformed into acidophilic, rounded Councilman bodies.
- Periportal (peripheral) necrosis: is seen in zone 1 involving the parenchyma closest to the arterial and portal blood supply.
- Since zone 1 is the most well-perfused, it is most vulnerable to the effects of circulating hepatotoxins example phosphorus poisoning and eclampsia.
3. Focal Necrosis
- This form of necrosis involves small groups of hepatocytes irregularly distributed in the hepatic lobule.
- Focal necrosis is most often caused by microbiologic infections. These include viral hepatitis, miliary tuberculosis, typhoid fever and various other forms of bacterial, viral and fungal infections.
- Focal necrosis may also occur in drug-induced hepatitis.
Liver Function Tests
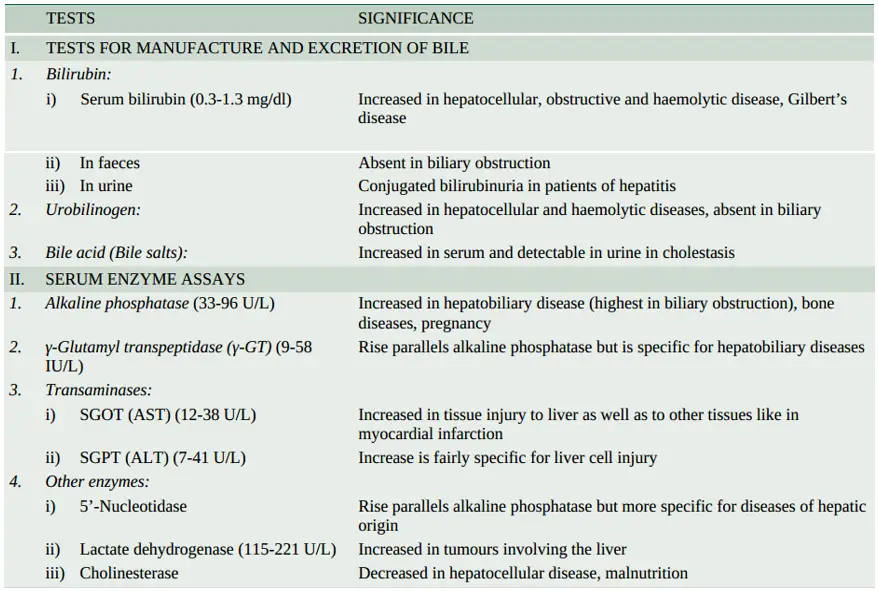
In view of the multiplicity and complexity of the liver functions, it is obvious that no single test can establish the disturbance in liver function. Thus a battery of liver function tests is employed for accurate diagnosis, to assess the severity of the damage, to judge prognosis and to evaluate therapy. These tests are described below in relation to major liver functions. A summary of various liver function tests is given in.

Tests For Manufacture And Excretion Of Bile
- Bile is produced by the liver, stored in the gallbladder and secreted via biliary ducts into the duodenum. Bile consists of biliary phospholipids and primary and secondary bile acids.
- To understand the mechanisms underlying biliary pathology, it is important to understand normal bilirubin metabolism.
- In brief, jaundice will develop if bilirubin is excessively produced, there is impaired hepatic uptake and conjugation of bilirubin, or it is insufficiently excreted into the duodenum.
- Tests employed to assess the synthesis and elimination of bilirubin pigment, urobilinogen and bile acids are as follows
1. Bilirubin Bilirubin pigment can be detected in serum, faeces and urine.
- Serum bilirubin estimation: is based on the van den Bergh diazo reaction by spectrophotometric method. Diazo reagent consists of diazotised sulfanilic acid.
- Water-soluble conjugated bilirubin gives a direct van den Bergh reaction with a diazo reagent within one minute, whereas alcohol-soluble unconjugated bilirubin is determined by an indirect van den Bergh reaction.
- The addition of alcohol to the reaction mixture gives a positive test for both conjugated and unconjugated bilirubin pigment.
- The unconjugated bilirubin level is then estimated by subtracting the direct bilirubin value from this total value.
- The serum of normal adults contains less than 1 mg/dl of total bilirubin, out of which less than 0.25 mg/dl is conjugated bilirubin.
- Bilirubin level rises in diseases of hepatocytes, obstruction to biliary excretion into the duodenum, haemolysis, and defects of hepatic uptake and conjugation of bilirubin pigment such as in Gilbert’s disease.
- In faeces, excretion of bilirubin is assessed by inspection of stools. Clay-coloured stool due to the absence of faecal excretion of the pigment indicates obstructive jaundice.
- In urine, the presence of bilirubin imparts deep yellow colour. Conjugated bilirubinuria can be detected by commercially available ‘dipsticks’, Fouchet’s test, foam test or dictates tablet method.
- Bilirubinuria does not occur in normal subjects nor is unconjugated bilirubin excreted in the urine. Bilirubinuria occurs only when there is raised level of conjugated bilirubin (filterable).
- Its excretion depends upon the level of conjugated bilirubin in plasma that is not protein-bound and is therefore available for glomerular filtration.
- Bilirubinuria appears in patients with hepatitis before the patient becomes jaundiced.
2. Urobilinogen
- Urobilinogen is normally excreted in the urine. Its semiquantitative estimation in the urine can be done by preparing dilutions with Ehrlich’s aldehyde reagent or by the ‘dipstick’ method.
- An increase in urobilinogen in the urine is found in hepatocellular dysfunctions such as alcoholic liver disease, cirrhosis and malignancy of the liver.
- It is also raised in haemolytic disease and in pyrexia. In cholestatic jaundice due to complete biliary obstruction, urobilinogen disappears from the urine.
3. Bile Acids (Bile Salts)
- The primary bile acids (cholic acid and chenodeoxycholic acid) are formed from cholesterol in the hepatocytes.
- These bile acids on secretion into the gut come in contact with colonic bacteria and undergo deconjugation with the production of secondary bile acids (deoxycholic acid and lithocholic acid).
- Most of these bile acids are reabsorbed through enterohepatic circulation and reach the liver.
- Only about 10% of the total bile acids are excreted in the faeces normally as unabsorbable toxic lithocholic acid.
- Hepatobiliary diseases with cholestasis are associated with raised levels of serum bile acids which are responsible for producing itching (pruritus).
- These acids are excreted in the urine by active transport and passive diffusion and can be detected by simple methods such as Hay’s test and ‘dipsticks’.
4. Bromsulphalein Excretion
- Bromsulphalein (BSP) is a dye that is removed from circulation by the same mechanisms of binding, conjugation and excretion as bilirubin.
- BSP is injected intravenously and a sample of venous blood 45 minutes later is tested for the percentage of injected dye remaining in the blood; normally there is <5% dye retention in blood after 45 minutes.
- The test is rarely performed nowadays because of the availability of enzyme estimations which are better indicators of hepatic dysfunction.
- Presently, the only value of the BSP excretion test is in the diagnosis of Dubin-Johnson syndrome.
2. Serum Enzyme Assays
- Determination of certain serum enzymes is considered useful in various types of liver injury, whether hepatocellular or cholestatic, as well as in quantifying liver damage.
- A combination of serum transaminases and alkaline phosphatase estimation is adequate to diagnose liver injury.
1. Alkaline Phosphatase: Serum alkaline phosphatase is produced by many tissues, especially bone, liver, intestine and placenta and is excreted in the bile.
- Most of the normal serum alkaline phosphatase (range 33-96 U/L) is derived from bone. Elevation in the activity of the enzyme can thus be found in diseases of the bone, liver and in pregnancy.
- In the absence of bone disease and pregnancy, elevated serum alkaline phosphatase levels generally reflect hepatobiliary disease.
- The greatest elevation (3 to 10 times normal) occurs in biliary tract obstruction. Slight to moderate increase is seen in parenchymal liver diseases.
- such as hepatitis and cirrhosis and in metastatic liver disease.
- It is possible to distinguish serum hepatic alkaline phosphatase from bony alkaline phosphatase by fractionation into isoenzymes but this is not routinely done.
2. γ-Glutamyl Transpeptidase (γ-Gt)
- The primary source of the enzyme, γ-GT, in serum, is the liver. Its serum level parallels serum alkaline phosphatase and is used to confirm that the elevated serum alkaline phosphatase is of hepatobiliary origin.
- Besides its elevation in cholestasis and hepatocellular disease, the levels are high in patients with alcohol abuse even without liver disease.
3. Transaminases (Aminotransferases)
Assessment of liver cell necrosis is most frequently done by estimation of the following 2 serum enzymes:
- Serum aspartate transaminase or AST (formerly glutamic oxaloacetic transaminase or SGOT) AST or SGOT is a mitochondrial enzyme released from the heart, liver, skeletal muscle and kidney. Its normal serum level is 0.20-0.65 mkat/L (12-38 U/L).
- Serum alanine transaminase or ALT (formerly glutamic pyruvic transaminase or SGPT) ALT or SGPT is a cytosolic enzyme primarily present in the liver.
-
- Its normal serum level is 0.12-0.70 mkat/L (7-41 U/L). Serum levels of SGOT and SGPT are increased on damage to the tissues producing them.
- Thus serum estimation of SGPT (ALT) which is fairly specific for liver tissue is of greater value in liver cell injury, whereas SGOT (AST) level may rise in acute necrosis or ischaemia of other organs such as the myocardium, besides liver cell injury.
- Transaminase estimations are useful in the early diagnosis of viral hepatitis. Very high levels are seen in extensive acute hepatic necrosis such as in severe viral hepatitis and acute cholestasis.
- Alcoholic liver disease and cirrhosis are associated with mild to moderate elevation of transaminases.
4. Other Serum Enzymes: Determination of a few other serum enzymes is done sometimes but without any extra diagnostic advantage over the above-mentioned enzyme assays.
These are as under:
- 5’-Nucleotidase is another phosphatase derived from the liver. Its determination is useful to distinguish alkaline phosphatase of hepatic origin from that of bony tissue.
- Lactate dehydrogenase (LDH) is found to be elevated in the serum of patients with metastatic liver involvement.
- Choline esterase synthesised by the liver is diminished in hepatocellular disease and malnutrition due to impaired synthesis.
3. Tests For Metabolic Functions
The liver is the principal site of metabolism and synthesis of plasma proteins and amino acids, lipids and lipoproteins, carbohydrates and vitamins, besides detoxification of drugs and alcohol.
- Amino Acid And Plasma Protein Metabolism: Amino acids derived from the diet and from tissue breakdown are metabolised in the liver to ammonia and urea.
-
- A number of plasma proteins and immunoglobulins are synthesised on polyribosomes bound to the rough endoplasmic reticulum within the hepatocytes and discharged into plasma.
- Based on these metabolic functions of the liver, serum estimation of proteins, immunoglobulins and ammonia and aminoaciduria are employed to assess the liver cell damage.
1. Serum proteins: Liver cells synthesise albumin, fibrinogen, prothrombin, alpha-1- antitrypsin, haptoglobin, ceruloplasmin, transferrin, alpha-fetoproteins and acute phase reactant proteins.
- The blood levels of these plasma proteins are decreased in extensive liver damage.
- Routinely estimated are the total concentration of serum proteins (normal 6.7 to 8.6 gm/dl), serum albumin (normal 3.5 to 5.5 gm/dl), serum globulin (normal 2 to 3.5 gm/dl) and albumin/globulin (A/G) ratio (normal 1.5-3:1).
- Electrophoresis is used to determine the proportions of α1, α2, β and γ globulins.
- Due to the availability of protein electrophoresis, thymol turbidity and flocculation tests based on altered plasma protein components have been discontinued.
- Hypoalbuminaemia may occur in liver diseases having significant destruction of hepatocytes. Hyperglobulinaemia may be present in chronic inflammatory disorders such as cirrhosis and chronic hepatitis.
2. Immunoglobulins: The levels of serum immunoglobulins produced by lymphocytes and plasma cells (IgG, IgM and IgA) show nonspecific abnormalities in liver diseases and represent inflammatory or immune response rather than liver cell dysfunction.
- IgA is the predominant immunoglobulin in bile and its level is raised in cirrhosis, IgG is markedly raised in chronic active hepatitis and IgM is markedly increased in primary biliary cirrhosis.
3. Clotting factors: The hepatic synthetic function of several clotting factors can be assessed by a few simple coagulation tests.
- Prothrombin time and partial thromboplastin time, both of which reflect the activities of various clotting factors, are prolonged in patients with hepatocellular disease.
- Prothrombin time is dependent upon the hepatic synthesis of clotting factors and intestinal uptake of vitamin K, a fat-soluble vitamin.
- Thus, obstruction of the bile duct and intrahepatic cholestasis which result in vitamin K deficiency due to impaired lipid absorption, are associated with prolonged prothrombin time.
- However, parenteral injection of vitamin K will normalise prothrombin time if the prolongation was due to obstruction, but there will be no improvement in prothrombin time if there is extensive hepatocellular disease.
4. Serum ammonia: High blood levels of ammonia are found in acute fulminant hepatitis, cirrhosis and hepatic encephalopathy.
- The rise in serum ammonia is due to the inability of the severely damaged liver to convert ammonia to urea. Thus, urea synthesis is reduced in chronic liver disease.
2. Lipid And Lipoprotein Metabolism: Lipids synthesised in the liver include cholesterol and cholesterol esters, phospholipids and triglycerides.
- These lipids are insoluble in water and are carried in circulation with three major types of lipoproteins which contain apoproteins.
- These are high-density lipoproteins (HDL), low-density lipoproteins (LDL) and very low-density lipoproteins (VLDL).
Blood lipids Estimations of total serum cholesterol, triglycerides and lipoprotein fractions are frequently done in patients with liver disease.
- There is a rise in total serum cholesterol in cholestasis, probably due to the retention of cholesterol which is normally excreted in the bile (normal < 200 mg/dl). Serum triglyceride is also elevated in cholestasis.
- Values are lowered in acute and chronic diffuse liver diseases and in malnutrition.
3. Carbohydrate Metabolism: The liver plays a central role in carbohydrate metabolism. Blood glucose level is lowered in fulminant acute hepatic necrosis. In chronic liver disease, there is impaired glucose tolerance and relative insulin resistance.
4. Immunologic Tests
Liver diseases are associated with various immunologic abnormalities which may be nonspecific immunologic reactions or maybe antibodies against specific etiologic agents.
- Nonspecific Immunologic Reactions: These include the following:
- Smooth muscle antibody to the actin component of muscle is formed in certain hepatic disorders with hepatic necrosis. It appears that hepatocytes have a protein which is immunologically similar to actin.
- Mitochondrial antibody develops in patients with primary biliary cirrhosis.
- The antinuclear antibody is present in some patients with chronic hepatitis. The LE cell test may be positive in these cases.
2. Antibodies To Specific Etiologic Agents: These vary according to the etiologic agent causing the liver cell injury.
- Hepatitis B surface antigen (HBsAg) can be demonstrated in cases of serum hepatitis. A confirmed positive test for HBsAg is definite proof of hepatitis B infection.
- Hepatitis B core antibody (HBc) can be detected in all patients with hepatitis B.
- Hepatitis B antigen (HBeAg) can be found in chronic varieties of hepatitis B.
- Amoeba antibodies to Entamoeba histolytic develop in patients with amoebic liver abscess.
5. Ancillary Diagnostic Tests
In addition to the laboratory tests described above, two ancillary tests which are invariably done by the physician are ultrasonography and percutaneous liver biopsy and/or FNAC.
1. Ultrasonography: Ultrasound (US) examination of the liver is indicated in the following situations:
- Cholestasis of various etiologies to see the dilated intra- and extrahepatic canalicular tree.
- Space-occupying lesions (SOLs) within the liver to determine whether they are neoplasms or non-neoplastic cysts.
- To provide US guidance for FNAC or liver biopsy.
2. Fnac And/Or Percutaneous Liver Biopsy
- Lastly, FNAC and percutaneous liver biopsy are employed to examine the microscopic changes of hepatic morphology in various diseases.
- Both these tests are done after evaluation of signs of obstruction since these tests are contraindicated in cholestasis.
- FNAC and liver biopsy are otherwise easily performed bedside tests of value. Their main indications are as follows
- the hepatocellular disease of unknown cause,
- suspected cases of chronic hepatitis,
- hepatomegaly of various etiologies,
- splenomegaly of unknown cause,
- fever of unknown cause, and
- SOLs visualised in radiologic examination
Liver: Normal Structure, Necrosis and Function Tests
- The liver is the largest organ in the body with an average weight of 1500 gm in males and 1300 gm in females.
- It has microscopic lobules composed of cords of hepatocytes and 3-5 portal tracts in each lobule.
- All forms of liver cell injury result in necrosis of liver cells in the hepatic lobule which may be diffuse (submassive to massive), zonal and focal.
- Zonal necrosis may pertain to the respective zone: centrilobular (zone 1), mid-zonal (zone 2) and periportal (zone 3).
- Major functions performed by the liver are metabolism, synthesis, storage, detoxification and excretion.
- Manufacture and excretion of bile can be tested in blood, urine and stool.
- Serum enzymes can be estimated to assess the hepatocellular injury.
- Blood tests for the manufacture of proteins, lipids and carbohydrates are routinely done.
- Specific immunologic tests, ultrasound and FNA or biopsy are done in some clinical situations.
Gilbert’S Syndrome
- This is the commonest of the familial, genetically-determined diseases of the liver affecting 2-5% of the population.
- Gilbert’s syndrome is characterised by mild, benign, unconjugated hyperbilirubinaemia (serum bilirubin 1-5 mg dl) which is not due to haemolysis. The condition is inherited as an autosomal dominant character.
- The defect in bilirubin metabolism is complex and appears to be reduced activity of UDP-glucuronosyl transferase with decreased conjugation, or an impaired hepatic uptake of bilirubin. The jaundice is usually mild and intermittent.
Morphologic Features: There are no morphologic abnormalities in the liver except for some increased lipofuscin pigment in centrilobular hepatocytes.
- The prognosis of patients with Gilbert’s syndrome is excellent, though chronic jaundice persists throughout life.
Crigler-Najjar Syndrome: Crigler-Najjar syndrome is a rare form of familial nonhaemolytic jaundice with very high unconjugated hyperbilirubinaemia. There are 2 forms of this condition: type 1 and type 2.
Type 1 Crigler-Najjar syndrome: This is inherited as an autosomal recessive disorder.
- There is a complete absence of conjugating enzyme UDP-glucuronosyl transferase in the hepatocytes and hence no conjugated bilirubin is formed.
- There is extreme elevation of unconjugated bilirubin (usually more than 20 mg/dl) with a high risk of developing permanent CNS damage from kernicterus.
- The prognosis is generally fatal, with death coming from kernicterus usually in the first year of life.
Morphologic Features: There are no significant morphologic changes except for some canalicular stasis.
Type 2 Crigler-Najjar syndrome: This is inherited as an autosomal dominant disease. There is a deficiency of the enzyme UDP-glucuronosyl transferase but not a complete absence.
- Thus, unconjugated hyperbilirubinaemia is generally mild to moderate (usually less than 20 mg/dl). The occurrence of kernicterus is exceptional and patients respond well to phenobarbital therapy.
- Morphologic Features: There are no morphologic changes in the liver
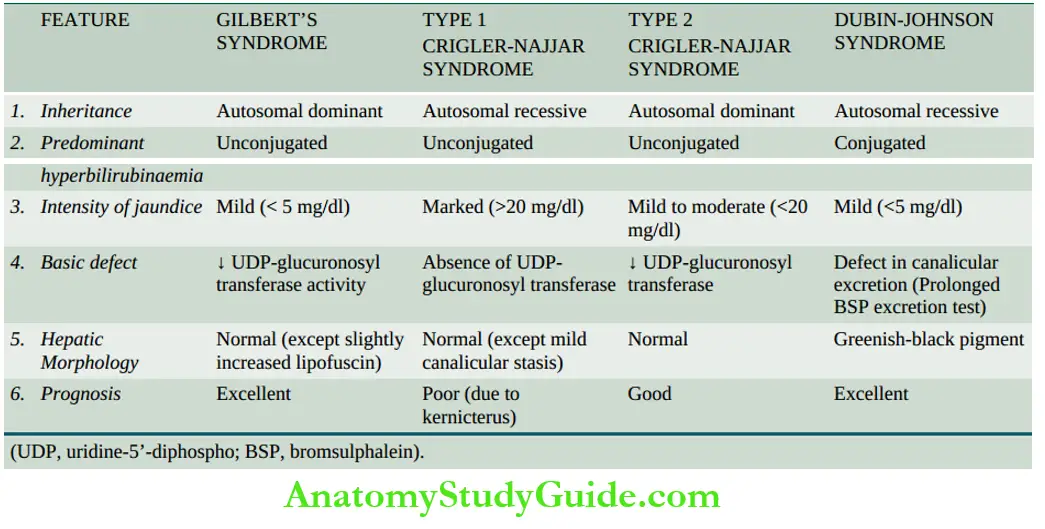
- Dubin-Johnson Syndrome: Dubin-Johnson syndrome is an autosomal recessive disorder characterised by predominantly conjugated hyperbilirubinaemia (usually less than 5 mg/dl) with a genetic defect in canalicular excretion of conjugated bilirubin.
-
- A prolonged BSP dye excretion test is diagnostic of Dubin-Johnson syndrome.
- Morphologic Features Grossly, the condition differs from other forms of hereditary hyperbilirubinaemias in producing greenish-black pigmented liver.
- Microscopically, The hepatocytes show dark-brown, melanin-like pigment in the cytoplasm, the exact nature of which is obscure but it is neither iron nor bile.
- Unrelated viral hepatitis mobilises the hepatic pigment of Dubin-Johnson syndrome leading to its excretion in urine but the pigment reappears after recovery from viral hepatitis.
- The disease runs a benign course and does not interfere with life.
Other Rare Intrahepatic Cholestatic Disorders: Besides Dubin-Johnson syndrome, there are a few rarer familial disorders having intrahepatic (conjugated) hyperbilirubinaemias (ranging from mild to recurrent to severe jaundice)
Rotor syndrome: There is mild chronic jaundice similar to Dubin-Johnson syndrome but differs from it in having no brown pigment in the liver cells.
- The disease has autosomal recessive inheritance. The defect probably lies in intrahepatic storage of bilirubin and no cholestasis but the exact molecular basis is not known.
Benign recurrent intrahepatic cholestasis (BRIC):
- In this, there are recurrent attacks of pruritus and jaundice in early childhood followed by resolution and long gaps for recurrences.
- The genetic defect has been identified and two types of BRIC (type 1 and type 2) have been categorised.
Progressive familial intrahepatic cholestasis (FIC): Based on a mutational defect, three genotypes of FIC have been identified but all three types have similar phenotypes. progressive severe cholestasis beginning in childhood.
Neonatal Hepatitis
- Neonatal hepatitis, also termed giant cell hepatitis or neonatal hepatocellular cholestasis, is a general term used for the constant morphologic change seen in conjugated hyperbilirubinaemia as a result of known infectious and metabolic causes listed in or may have an idiopathic aetiology.
- ‘Idiopathic’ neonatal hepatitis is more common and accounts for 75% of cases.
- Though all the cases with either known etiologies or idiopathic types are grouped together under neonatal hepatitis, all of them are not necessarily inflammatory conditions, thus belying their nomenclature as ‘hepatitis’.
- The condition usually presents in the first week of birth with jaundice, bilirubinuria, pale stools and high serum alkaline phosphatase.
Morphologic Features: Irrespective of the aetiology, there is a morphologic similarity in all these cases. The histologic features are as under:
- Loss of normal lobular architecture of the liver.
- Presence of prominent multinucleate giant cells derived from hepatocytes.
- Mononuclear inflammatory cell infiltrates the portal tracts with some periportal fibrosis.
- Haemosiderosis.
- Cholestasis in small proliferated ductules in the portal tract and between necrotic liver cells.
Biliary Atresias
- Biliary atresias, also called infantile cholangiopathies, are a group of intrauterine developmental abnormalities of the biliary system.
- Though they are often classified as congenital, the abnormality of development in most instances is extraneous infection during intrauterine development or shortly after birth that brings about inflammatory destruction of the bile ducts.
- The condition may, therefore, have various grades of destruction ranging from the complete absence of bile ducts termed atresia, to a reduction in their number called paucity of bile ducts.
- Depending upon the portion of the biliary system involved, biliary atresias may be extrahepatic or intrahepatic.
Extrahepatic Biliary Atresia: The extrahepatic bile ducts fail to develop normally so that in some cases the bile ducts are absent at birth, while in others the ducts may have been formed but start undergoing sclerosis in the perinatal period.
- It is common to have multiple defects and other congenital lesions. Extrahepatic biliary atresia is found in 1 per 10,000 live births. Cholestatic jaundice appears by the first week after birth.
- The baby has severe pruritus, pale stools, dark urine and elevated serum transaminases.
- In some cases, the condition is correctable by surgery, while in the vast majority, the atresia is not correctable and in such cases, hepatic portent (Kasai procedure) or hepatic transplantation must be considered.
- Death is usually due to intercurrent infection, liver failure, and bleeding due to vitamin K deficiency or oesophageal varices. Cirrhosis and ascites are late complications appearing within 2 years of age.
- Morphologic Features Grossly, the liver is enlarged and dark green. The atretic segments of the biliary system are reduced to cord-like structures.
- Histologically, the condition must be distinguished from idiopathic neonatal hepatitis as surgical treatment is possible in extrahepatic biliary atresia but not in the latter.
- Besides, α-1- antitrypsin deficiency also produces a similar appearance in liver biopsy. The main histologic features are as under:
- Inflammation and fibrous obliteration of the extrahepatic ducts with the absence of bile in them.
- Ductular proliferation and periductular inflammation.
- Cholestasis and bile thrombi in the portal area.
- Periportal fibrosis and later secondary biliary cirrhosis.
- Transformation of hepatic parenchyma to neonatal (giant cell) hepatitis in 15% of cases.
Intrahepatic Biliary Atresia: Intrahepatic biliary atresia is characterised by biliary hypoplasia so that there is a paucity of bile ducts rather than their complete absence.
- The condition probably has its origin in viral infection acquired during the intrauterine period or in the neonatal period.
- Cholestatic jaundice usually appears within the first few days of birth and is characterised by high serum bile acids with associated pruritus, and hypercholesterolaemia with the appearance of xanthomas by the first year of life.
- Hepatic as well as urinary copper concentrations are elevated. In some cases, intrahepatic biliary atresia is related to α-1-antitrypsin deficiency.
Morphologic Features: The microscopic features are as follows:
- A paucity of intrahepatic bile ducts.
- Cholestasis.
- Increased hepatic copper.
- Inflammation and fibrosis in the portal area, eventually lead to cirrhosis.
Reye’s Syndrome
- Reye’s syndrome is defined as an acute postviral syndrome of encephalopathy and fatty change in the viscera. The syndrome may follow almost any known viral disease but is most common after influenza A or B and varicella.
- Viral infection may act singly, but more often its effect is modified by certain exogenous factors such as by administration of salicylates, aflatoxins and insecticides.
- These effects cause mitochondrial injury and decreased activity of mitochondrial enzymes in the liver. This eventually leads to a rise in blood ammonia and accumulation of triglycerides within hepatocytes.
- The patients are generally children between 6 months and 15 years of age.
- Within a week after a viral illness, the child develops intractable vomiting and progressive neurological deterioration due to encephalopathy, eventually leading to stupor, coma and death.
- Characteristic laboratory findings are elevated blood ammonia, serum transaminases, bilirubin and prolonged prothrombin time.
- Morphologic Features Grossly, the liver is enlarged and yellowish-orange. Microscopically, hepatocytes show small droplets of neutral fat in their cytoplasm (microvesicular fat).
- Similar fatty change is seen in the renal tubular epithelium and in the cells of skeletal muscles and heart. The brain shows oedema and sometimes focal necrosis of neurons.
Jaundice
- Jaundice is yellow pigmentation of the skin or sclerae by bilirubin.
- It is due to a rise in bilirubin levels in the blood (hyperbilirubinaemia) above normal (0.3-1.3 mg/dl).
- Normally, bilirubin formed in the body is transported and metabolised through the liver and excreted through the intestines and kidneys.
- Predominantly unconjugated hyperbilirubinaemia is due to increased production and decreased hepatic uptake and conjugation, while reduced excretion causes mainly conjugated hyperbilirubinaemia.
- Neonatal jaundice appears at serum bilirubin level of more than 3 mg/dl and is more often unconjugated hyperbilirubinaemia.
- Hereditary non-haemolytic hyperbilirubinaemias are familial disorders of bilirubin metabolism; the most common is Gilbert’s syndrome; others are Crigler-Najjar syndrome (types 1 and 2), and Dubin-Johnson syndrome.
- Gilbert’s and Dubin-Johnson’s syndrome have excellent prognoses.
- Neonatal hepatitis or giant cell hepatitis is a morphologic change seen in conjugated hyperbilirubinaemia as a result of known infectious and metabolic causes or may be idiopathic.
- Biliary atresias (intrahepatic and extrahepatic) are intrauterine developmental abnormalities of the biliary system.
- Reye’s syndrome is an acute postviral syndrome of encephalopathy and fatty change in the viscera, most importantly in the liver.
Hepatic Failure
- Though the liver has a marked regenerative capacity and a large functional reserve, hepatic failure may develop from severe acute and fulminant liver injury with massive necrosis of liver cells (acute hepatic failure), or from advanced chronic liver disease (chronic hepatic failure).
- Acute hepatic failure develops suddenly with severe impairment of liver functions whereas chronic liver failure comes insidiously.
- The prognosis is much worse in acute hepatic failure than that in chronic liver failure.
Etiology Acute and chronic hepatic failure result from different causes:
- Acute (fulminant) hepatic failure occurs most frequently in acute viral hepatitis.
Other causes are hepatotoxic drug reactions (for example anaesthetic agents, nonsteroidal anti-inflammatory drugs, and anti-depressants), carbon tetrachloride poisoning, acute alcoholic hepatitis, mushroom poisoning and pregnancy complicated with eclampsia. - Chronic hepatic failure is most often due to cirrhosis. Other causes include chronic active hepatitis, chronic cholestasis (cholestatic jaundice) and Wilson’s disease.
Manifestations: In view of the diverse functions performed by the liver, the syndrome of acute or chronic hepatic failure produces complex manifestations.
The major manifestations are briefly discussed below and diagrammatically illustrated.
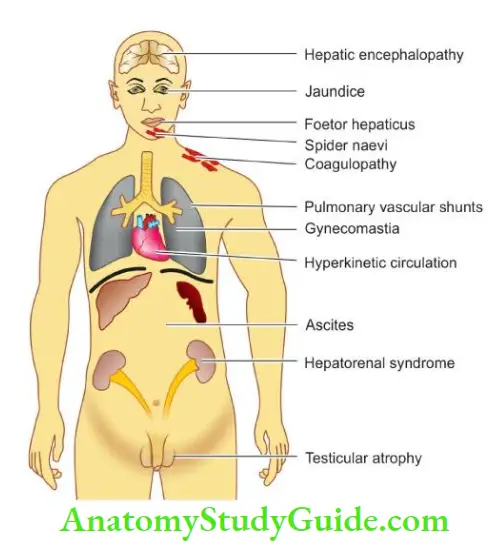
1. Jaundice: Jaundice usually reflects the severity of liver cell damage since it occurs due to the failure of liver cells to metabolise bilirubin.
- In acute failure such as in viral hepatitis, jaundice nearly parallels the extent of liver cell damage, while in chronic failure such as in cirrhosis, jaundice appears late and is usually of mild degree.
2. Hepatic encephalopathy (Hepatic coma): Neuropsychiatric syndrome may complicate liver disease of both acute and chronic types.
- The features include disturbed consciousness, personality changes, intellectual deterioration, low-slurred speech, flapping tremors, and finally, coma and death.
- The genesis of CNS manifestations in liver disease is toxic products not metabolised by the diseased liver.
- The toxic products may be ammonia and other nitrogenous substances from intestinal bacteria which reach the systemic circulation without detoxification in the damaged liver and thus damage the brain.
- Advanced cases of hepatic coma have poor prognoses but may respond favourably to hepatic transplantation.
3. Hyperkinetic circulation: All forms of hepatic failure are associated with hyperkinetic circulation characterised by peripheral vasodilatation, increased splanchnic blood flow and increased cardiac output.
- There is increased splenic flow but reduced renal blood flow resulting in impaired renal cortical perfusion. These changes result in tachycardia, low blood pressure and reduced renal function.
4. Hepatorenal syndrome: The term hepatorenal syndrome is applied to patients of both acute and chronic hepatic failure who develop renal failure as well, in the absence of clinical, laboratory or morphologic evidence of other causes of renal dysfunction.
- Hepatorenal syndrome develops in about 10% of cases of acute and chronic liver diseases. Acute renal failure is usually associated with oliguria and uraemia but with good tubular function.
- The histology of the kidney is virtually normal, suggesting a functional defect for renal failure.
- The pathogenesis of the syndrome is poorly understood but appears to be initiated by an effective reduction of the renal blood flow (effective hypovolaemia) as a consequence of systemic vasodilatation and pooling of blood in portal circulation.
- The renal failure in the hepatorenal syndrome is reversible with improvement in hepatic function.
- Diagnosis of the hepatorenal syndrome should be made only after excluding other causes producing concomitant damage to both organs, circulatory failure leading to acute tubular necrosis and other forms of reversible tubular damage.
5. Hepatopulmonary syndrome: The pulmonary changes in chronic hepatic failure such as in cirrhosis consist of pulmonary vasodilatation with intra-pulmonary arteriovenous shunting.
- This results in ventilation-perfusion inequality that may lead to impaired pulmonary function, clubbing of fingers and sometimes cyanosis.
6. Coagulation defects: Impaired synthesis of a number of coagulation factors by the diseased liver may result in coagulation disorders.
- These include disseminated intravascular coagulation (consumption coagulopathy), thrombocytopenia and the presence of fibrin degradation products in the blood.
7. Ascites and oedema: Chronic liver failure due to cirrhosis may result in portal hypertension and ascites.
- Decreased synthesis of albumin by the liver resulting in hypoproteinaemia and consequent fall in plasma oncotic pressure, increased hydrostatic pressure due to portal hypertension and secondary hyperaldosteronism, contribute to the development of ascites and oedema in these patients.
8. Endocrine changes: may be found in association with chronic hepatic failure. The changes are more common in alcoholic cirrhosis in active reproductive life.
- In the male, the changes are towards feminisation such as gynecomastia and hypogonadism. In the female, the changes are less towards masculinisation but atrophy of gonads and breasts occurs.
- The underlying mechanism appears to be changed end-organ sensitiveness to sex hormones in cirrhosis.
9. Skin changes: In alcoholic cirrhosis ‘arterial spiders’ having radiating small vessels from a central arteriole are frequent in the vascular region drained by the superior vena cava such as in the neck, face, forearms and dorsum of hands.
- Less frequently, palmar erythema, especially in the hypothenar and thenar eminences and on the pulps of the fingers, is observed in chronic liver disease.
10. Foetor hepatitis: A sweetish pungent smell of the breath is found in severe cases of acute and chronic hepatocellular diseases.
- It appears to be of intestinal origin, possibly due to failure of the liver to detoxify sulfur-containing substances absorbed from the gut
Hepatic Failure
- Hepatic failure may develop from severe acute and fulminant liver injury with massive necrosis of liver cells (acute hepatic failure), or from advanced chronic liver disease (chronic hepatic failure).
- The clinical manifestations are systemic and include jaundice, hepatic coma, hyperkinetic circulation, hepatorenal syndrome, hepatoma syndrome, coagulation defects, ascites, oedema, endocrine changes, skin changes and foetor hepatica.
Circulatory Disturbances
- Vascular disorders of general nature involving the liver such as chronic passive congestion and infarction have already been discussed. Hepatic and portal venous obstruction and hepatic arterial obstruction are considered here.
Hepatic Venous Obstruction
- The central veins of lobules of the liver are tributaries of the hepatic veins. In the normal liver, there are no anastomoses between the hepatic vein and portal vein but in the cirrhotic liver, there are such anastomoses.
- Normal pressure in the free hepatic vein is about 6 mmHg.
- Three uncommon diseases produced by obstruction of the hepatic veins are Budd-Chiari syndrome (hepatic vein thrombosis), hepatic veno-occlusive disease and bacillary angiomatosis peliosis hepatis.
- Budd-Chiari Syndrome (Hepatic Vein Thrombosis)
- Budd-Chiari syndrome in its pure form consists of slowly developing thrombosis of the hepatic veins and the adjacent inferior vena cava, while some workers include hepatic veno-occlusive disease (described below) in this syndrome.
Aetiology The aetiology of hepatic venous thrombosis in about a third of cases is unknown (idiopathic), while in the remaining cases various causes associated with increased thrombotic tendencies are attributed:
- Polycythaemia vera
- Paroxysmal nocturnal haemoglobinuria
- Use of oral contraceptives
- Pregnancy and postpartum state
- Intra-abdominal cancers (for example hepatocellular carcinoma)
- Chemotherapy and radiation
- Myeloproliferative diseases
- Formation of membranous webs in the suprahepatic portion of the inferior vena cava (either congenital or as a consequence of organised thrombosis).
Morphologic Features Grossly, the liver is enlarged, swollen, red-purple and has a tense capsule.
- Histologically, the changes in sudden hepatic vein occlusion are those of centrilobular congestion, necrosis and rupture of sinusoids into the space of Disse.
- In slowly developing thrombosis, the changes are more chronic and include fibrosing reaction in the centrilobular zone that may progress to cardiac cirrhosis.
Clinical Features: Budd-Chiari syndrome is clinically characterised by either an acute form or chronic form depending upon the speed of occlusion.
- In the acute form, the features are abdominal pain, vomiting, enlarged liver, ascites and mild icterus.
- In the more usual chronic form, the patients present with pain from the enlarged tender liver, ascites and other features of portal hypertension.
The acute form of illness leads to acute hepatic failure and death, whereas in the chronic form, the patient may live for months to a few years.
Hepatic Veno-occlusive Disease
- The hepatic veno-occlusive disease consists of intimal thickening, stenosis and obliteration of the terminal central veins and medium-sized hepatic veins.
- The venous occlusion results in pathologic changes similar to those of Budd-Chiari syndrome and can be distinguished from the latter by the demonstration of the absence of thrombosis in the major hepatic veins.
- The aetiology of hepatic veno-occlusive disease can be explained by the following associations:
- Since this condition is more widespread in countries such as Africa, India and certain other tropical countries where ‘bush tea’ (medicinal tea) is consumed that contains hepatotoxic alkaloids, it is implicated in the aetiology.
- The disease has also been found after high-dose chemotherapy administered before bone
marrow transplantation. - It is also seen as part of a rare hereditary veno-occlusive disease with immunodeficiency which results from a gene mutation.
Bacillary Angiomatosis and Peliosis Hepatis
- Although sinusoidal dilatation can occur secondary to many liver diseases, peliosis hepatis is an uncommon condition of primary sinusoidal dilatation that results in blockage of blood outflow and may result in massive intraperitoneal haemorrhage.
- Although the exact aetiology is not known, peliosis hepatis and another related condition, bacillary angiomatosis, have been found to occur in HIV-infected patients whose CD4+ T cell counts fall below 100/µl.
- Opportunistic infection with Bartonella henselae in poor hygienic conditions in these cases results in blood-filled cysts in the liver partly lined by endothelial cells and having mixed inflammatory cells in a fibromyxoid background.
- Etiologic association of peliosis hepatis with consumption of anabolic steroids and oral contraceptives has also been suggested and is self-limiting with the withdrawal of the offending agent.
Portal Venous Obstruction
Obstruction of the portal vein may occur within the intrahepatic course or in the extrahepatic site.
- Intrahepatic cause of portal venous occlusion is hepatic cirrhosis as the commonest and most important followed in decreasing frequency by tumour invasion, congenital hepatic fibrosis and schistosomiasis.
- Extrahepatic causes of portal vein obstruction are intra-abdominal cancers, intra-abdominal sepsis, direct invasion by tumour, myeloproliferative disorders and upper abdominal surgical procedure followed by thrombosis.
- The effects of portal venous obstruction depend upon the site of obstruction. The most important effect, irrespective of the site of occlusion or cause, is portal hypertension and its manifestations.
- If the obstruction is in the extrahepatic portal vein along with the extension of occlusion into the splenic vein, it may result in venous infarction of the bowel. Pylephlebitis may be followed by multiple pyaemic liver abscesses.
Hepatic Arterial Obstruction
- Diseases from obstruction of the hepatic artery are uncommon. Rarely, accidental ligation of the main hepatic artery or its branch to the right lobe may be followed by fatal infarction.
- Obstruction of the small intrahepatic arterial branches usually does not produce any effects because of good collateral circulation.
Hepatic Circulatory Disturbances
- Obstruction of the hepatic veins may occur and produce Budd-Chiari syndrome (hepatic vein thrombosis), hepatic veno-occlusive disease and peliosis hepatis.
- Budd-Chiari syndrome is idiopathic or due to increased thrombotic tendencies.
- The veno-occlusive disease is related to the consumption of alkaloids.
- In peliosis hepatis and bacillary angiomatosis that occurs in HIV-infected patients, there is sinusoidal dilatation.
- Obstruction of the portal vein may occur from intrahepatic (for example cirrhosis) or extrahepatic (for example abdominal cancer) causes.
- hepatic arterial obstruction is rare; besides, there is good collateral circulation to circumvent the ill effects of obstruction of smaller intrahepatic arterial branches.
Viral Hepatitis
The term viral hepatitis is used to describe infection of the liver caused by hepatotropic viruses. Currently, there are five main types of hepatotropic viruses causing distinct types of viral hepatitis:
- Hepatitis A virus (HAV), causes a faecally-spread self-limiting disease.
- Hepatitis B virus (HBV), causes a parenterally transmitted disease that may become chronic.
- Hepatitis C virus (HCV), previously termed non-A, non-B (NANB) hepatitis virus involved chiefly in transfusion-related hepatitis.
- Hepatitis delta virus (HDV) is sometimes associated as a superinfection with hepatitis B infection.
- Hepatitis E virus (HEV), causes water-borne infection.
- While HBV is a DNA virus, all other human hepatitis viruses are RNA viruses.
- Though a number of other viral diseases such as infection with Epstein-Barr virus (in infectious mononucleosis), arbovirus (in yellow fever), cytomegalovirus, herpes simplex and several others affect the liver but the changes produced by them are nonspecific; the term ‘viral hepatitis’ is strictly applied to infection of the liver by the hepatitis viruses.
Etiologic Classification
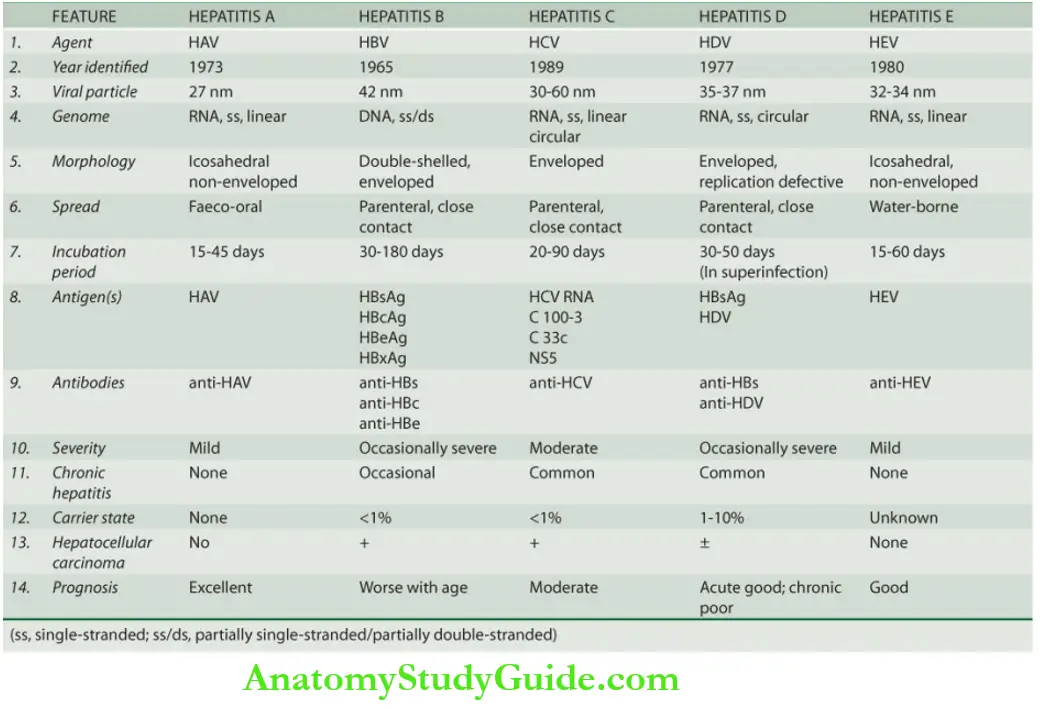
- Based on the etiologic agent, viral hepatitis is currently classified into five etiologic types hepatitis A, hepatitis B, hepatitis C, hepatitis D, and hepatitis E. The contrasting features of major types are presented.
Hepatitis A
- Infection with HAV causes hepatitis A (infectious hepatitis). Hepatitis A is responsible for 20- 25% of clinical hepatitis in the developing countries of the world but the incidence is much lower in the developed countries.
- Hepatitis A is usually a benign, self-limiting disease and has an incubation period of 15-45 days. The disease occurs in epidemic form as well as sporadically.
- It ]is almost exclusively spread by the faecal-oral route. The spread is related to close personal contact such as overcrowding, and poor hygienic and sanitary conditions.
- Frozen and stored contaminated foods and water have been blamed for many epidemics. The most frequently affected age group is 5-14 years; adults are often infected by the spread from children
Hepatitis A Virus (Hav): The etiologic agent for hepatitis A, HAV, is a small, 27 nm diameter, icosahedral non-enveloped, single-stranded RNA virus.
- The viral genome has been characterised but only a single serotype has been identified. HAV infection can be transmitted to primates and the virus can be cultivated in vitro.
- Inactivation of viral activity can be achieved by boiling for 1 minute, by ultraviolet radiation, or by contact with formaldehyde and chlorine.
- The virus is present in the liver cells, bile, stool and blood during the incubation period and in the prehistoric phase but viral shedding diminishes after the onset of jaundice.
- Chronic carriers have not been identified for HAV infection.
Pathogenesis: Evidence that hepatitis caused by HAV has an immunologic basis comes from a demonstration of the following antibodies acting as serum markers for hepatitis A infection:
- IgM anti-HAV antibody appears in the serum at the onset of symptoms of acute hepatitis A.
- IgG anti-HAV antibody is detected in the serum after acute illness and remains detectable indefinitely. It gives life-long protective immunity against reinfection with HAV.
Hepatitis D
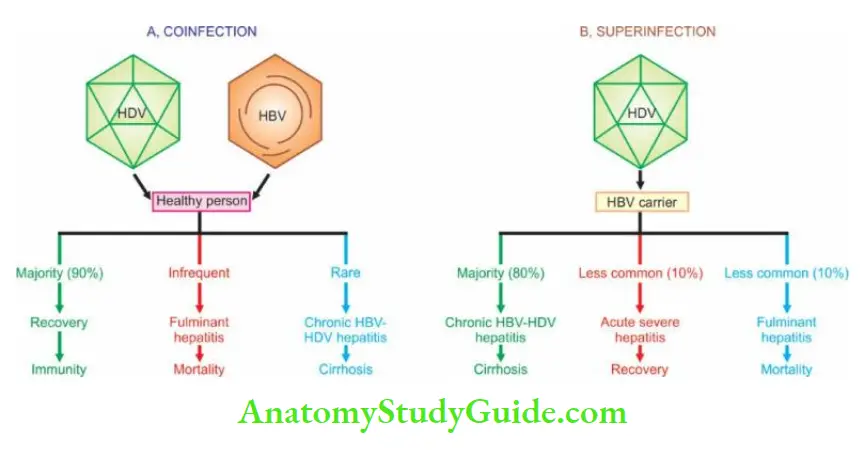
- Infection with delta virus (HDV) in the hepatocyte nuclei of HBsAg-positive patients is termed hepatitis D.
- HDV is a defective virus for which HBV is the helper. Thus, hepatitis D develops when there is a concomitant hepatitis B infection.
- HDV infection and hepatitis B may be simultaneous (co-infection), or HDV may infect a chronic HBsAg carrier (superinfection).
- With coinfection, acute hepatitis D may range from mild to fulminant hepatitis but fulminant hepatitis is more likely in such simultaneous delta infection. Chronicity rarely develops in coinfection.
- With superinfection (incubation period 30-35 days), chronic HBV infection gets worsened indicated by the appearance of severe and fulminant acute attacks, progression of carrier stage to chronic delta hepatitis or acceleration towards cirrhosis.
- The occurrence of hepatocellular carcinoma is, however, less common in HBsAg carriers with HDV infection. HDV infection is worldwide in distribution though the incidence may vary in different countries.
- Endemic regions are Southern Europe, Middle-East, South India and parts of Africa.
- The high-risk individuals for HDV infection are the same as those for HBV infection intravenous drug abusers, homosexuals, transfusion recipients, and healthcare workers.
Hepatitis Delta Virus(HDV): The etiologic agent, HDV, is a small single-stranded RNA particle with a diameter of 36 nm. It is double-shelled—the outer shell consists of HBsAg and the inner shell consists of delta antigen provided by a circular RNA strand.
- It is highly infectious and can induce hepatitis in any HBsAg-positive host. HDV replication and proliferation take place within the nuclei of liver cells. Markers for HDV infection include the following:
- HDV identification in the blood and in the liver cell nuclei.
- HDAg is detectable in the blood and on fixed liver tissue specimens.
- Anti-HD antibody in acute hepatitis which is initially IgM type and later replaced by IgG type anti-HD antibody which persists for life to confer immunity against reinfection.
Pathogenesis: HDV, unlike HBV, is thought to cause a direct cytopathic effect on hepatocytes.
- However, there are examples of transmission of HDV infection from individuals who themselves have not suffered from any attack of hepatitis, suggesting that it may not be always cytopathic.
Hepatitis C
- The diagnosis of this major category of hepatitis was earlier made after the exclusion of infection with other known hepatitis viruses in those times and was initially named non-A, non-B (NANB) hepatitis.
- However, after it was characterised, it was renamed hepatitis C. Hepatitis C infection is acquired by blood transfusions, blood products, haemodialysis, parenteral drug abuse and accidental cuts and needle-pricks in health workers. About 90% of post-transfusion hepatitis is of hepatitis C type.
- About 1-2% of volunteer blood donors and up to 5% of professional blood donors are carriers of HCV.
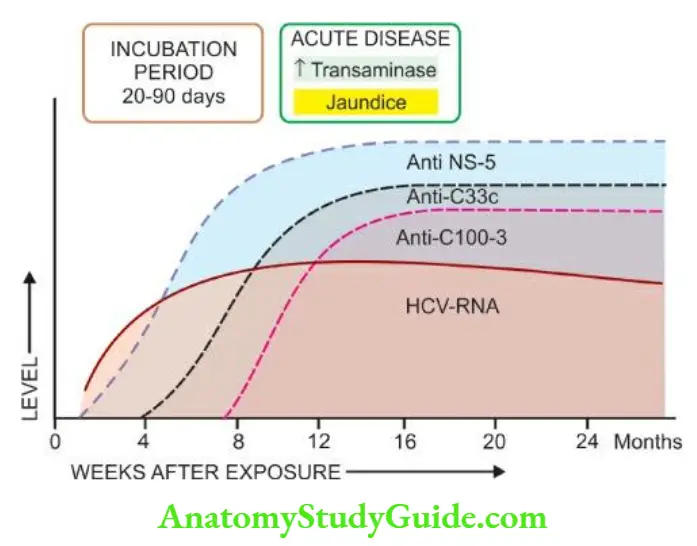
- Hepatitis C has an incubation period of 20- 90 days (mean 50 days). Clinically, acute HCV hepatitis is milder than HBV hepatitis but HCV has a higher rate of progression to chronic hepatitis than HBV.
- Persistence of infection and chronic hepatitis are the key features of HCV.
- Occurrence of cirrhosis after 5 to 10 years and progression to hepatocellular carcinoma are other late consequences of HCV infection.
- Currently, HCV is considered a more important cause of chronic liver disease worldwide than HBV.
- Hepatitis C Virus (HCV) HCV is a single-stranded, enveloped RNA virus, having a diameter of 30-60 nm. HCV genome has about 3000 amino acids.
- The genomic organisation of HCV shows a 5’ terminal end, C (capsid) region and the envelope regions E1 and E2 in the exons
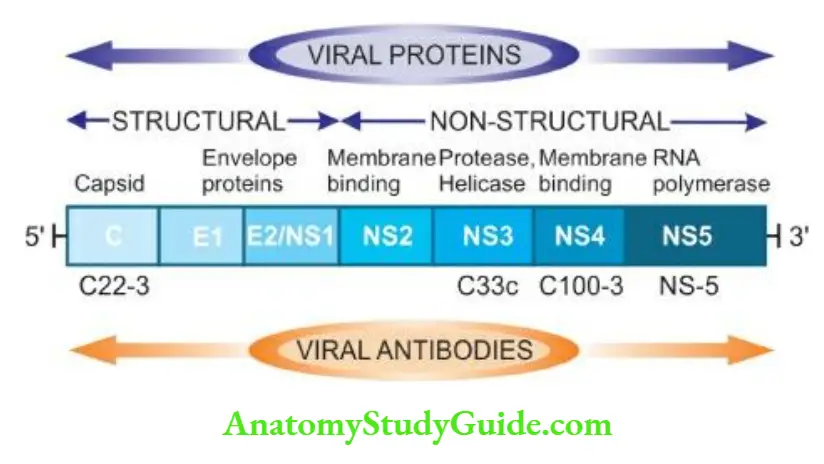
- The viral proteins result in corresponding serologic and virologic markers for HCV infection as under
1. Anti-HCV: antibodies. Three generations of anti-HCV IgG assays are available:
- First-generation antibodies are against C100-3 region proteins and appear 1 to 3 months after infection.
- Second-generation antibodies are against C200 and C33c proteins and appear about one month earlier than the first-generation.
- Third-generation antibodies are against C22-3 and NS-5 region proteins and are detected even earlier.
2. HCV-RNA: HCV infection is, however, confirmed by HCV-RNA employing PCR technique which can be detected within a few days after exposure to HCV infection, much before the appearance of anti-HCV and persists for the duration of HCV infection.
Pathogenesis: HCV induces hepatocellular injury by a cell-mediated immune mechanism supported by the following:
- It is possible that the host lymphoid cells are infected by HCV.
- HCV-activated CD4+ helper T lymphocytes stimulate CD8+ T lymphocytes via cytokines elaborated by CD4+ helper T cells.
- The stimulated CD8+T lymphocytes, in turn, elaborate antiviral cytokines against various HCV antigens.
- Further support to this T-cell mediated mechanism comes from the observation that immune response is stronger in those HCV-infected persons who recover than those who harbour chronic HCV infection.
- There is some role of certain HLA alleles and innate immunity in rendering variable responses by different hosts to HCV infection.
- Natural killer (NK) cells also seem to contribute to the containment of HCV infection.
- In a subset of patients, there is crossreactivity between viral antigens of HCV and host autoantibodies to liver-kidney microsomal antigen (anti-LKM) which explains the association of autoimmune hepatitis and HCV hepatitis.
Hepatitis E
- Hepatitis E is an enteric virus, previously labelled as an epidemic or enteric transmitted variant of non-A non-B hepatitis.
- The infection occurs in young or middle-aged individuals, primarily seen in India, other Asian countries, Africa and Central America.
- The infection is generally acquired by contamination of water supplies such as after monsoon flooding. However, compared with HAV, secondary person-to-person infection does not occur with HEV.
- Thus HEV has some common epidemiologic features with HAV. HEV infection has particularly high mortality in pregnant women but is otherwise a self-limited disease and has not been associated with chronic liver disease.
Hepatitis E Virus (HEV): HEV is a single-stranded 32-34 nm, icosahedral non-enveloped virus. The virus has been isolated from the stools, bile and liver of infected persons. Serologic markers for HEV include the following:
- Anti-HEV antibodies of both IgM and IgG classes. Both fall rapidly after acute illness but routine serologic testing for HEV antibodies is not available.
- HEV-RNA.
Clinicopathologic Spectrum
- Among the various etiologic types of hepatitis, evidence linking HBV and HCV infection with the spectrum of clinicopathologic changes is stronger than with other hepatotropic viruses.
- The typical pathologic changes of hepatitis by major hepatotropic viruses are virtually similar. HAV and HEV, however, do not have a carrier stage nor cause chronic hepatitis.
- The various clinical patterns and pathologic consequences of different hepatotropic viruses can be considered under the following headings:
- Carrier state
- Asymptomatic infection
- Acute hepatitis
- Chronic hepatitis
- Fulminant hepatitis (Submassive to massive necrosis)
In addition, progression to cirrhosis and association with hepatocellular carcinoma is known to occur in certain types of hepatitis which are discussed separately later.
1. Carrier State
An asymptomatic individual without manifest disease, harbouring infection with hepatotropic virus and capable of transmitting it is called a carrier state. There can be 2 types of carriers:
- An ‘asymptomatic healthy carrier’ who does not suffer from ill effects of the virus infection but is capable of transmitting.
- An ‘asymptomatic carrier with chronic disease’ is capable of transmitting the organisms. As stated before, hepatitis A and E do not produce the carrier state.
- Hepatitis B is responsible for the largest number of carriers in the world, while concomitant infection with HDV more often causes progressive disease rather than an asymptomatic carrier state.
- There is geographic variation in the incidence of HBV carrier state: while in the normal population in the US and Western Europe, it is less than 0.5%, its prevalence is much higher in Asian and tropical countries (5-20%).
- An estimated 2-3% of the general population are asymptomatic carriers of HCV.
- Data on HBV carrier state reveal the role of 2 important factors rendering the individual more vulnerable to harbour the organisms early age at infection and impaired immunity.
- Whereas approximately 10% of adults contracting hepatitis B infection develop a carrier state, 90% of infected neonates fail to clear HBsAg from the serum within 6 months and become HBV carriers.
- Clinical recognition of HBV carrier state is done by the persistence of HBsAg in the serum of an infected person who fails to clear HBsAg from the blood for more than 6 months. Concomitant infection of HDV with HBV depends upon the demonstration of anti-HD.
Morphologic Features: Carriers of HBV may or may not show changes on liver biopsy.
- Healthy HBV carriers may show no changes or minor hepatic changes such as the presence of finely granular, ground-glass, eosinophilic cytoplasm as evidence of HBsAg.
- Asymptomatic carriers with chronic disease may show changes in chronic hepatitis and even cirrhosis.
2. Asymptomatic Infection
- These are cases which are detected incidentally to have an infection with one of the hepatitis viruses as revealed by their raised serum transaminases or by detection of the presence of antibodies but are otherwise asymptomatic.
3. Acute Hepatitis
- The most common consequence of all hepatotropic viruses is acute inflammatory involvement of the entire liver. In general, type A, B, C, D and E run similar clinical course and show identical pathologic findings.
- Clinically, acute hepatitis is categorised into 4 phases: incubation period, pre-icteric phase, icteric phase and post-icteric phase.
1. Incubation period: It varies among different hepatotropic viruses: for hepatitis A it is about 4 weeks (15-45 days); for hepatitis B the average is 10 weeks (30-180 days); for hepatitis D about 6 weeks (30-50 days); for hepatitis C the mean incubation period is about 7 weeks (20-90 days), and for hepatitis E it is 2-8 weeks (15-60 days).
- The patient remains asymptomatic during the incubation period but the infectivity is highest during the last days of the incubation period.
2. Pre-icteric phase: This phase is marked by prodromal constitutional symptoms that include anorexia, nausea, vomiting, fatigue, malaise, distaste for smoking, arthralgia and headache.
- There may be low-grade fever preceding the onset of jaundice, especially in hepatitis A. The earliest laboratory evidence of hepatocellular injury in the pre-icteric phase is the elevation of transaminases.
3. Icteric phase: The prodromal period is heralded by the onset of clinical jaundice and the constitutional symptoms diminish.
- Other features include dark-coloured urine due to bilirubinuria, clay-coloured stools due to cholestasis, pruritus as a result of elevated serum bile acids, loss of weight and abdominal discomfort due to the enlarged, tender liver.
- The diagnosis is based on deranged liver function tests (for example elevated levels of serum bilirubin, transaminases and alkaline phosphatase, prolonged prothrombin time and hyperglobulinaemia) and serologic detection of hepatitis antigens and antibodies.
4. Post-icteric phase: The icteric phase lasting for about 1 to 4 weeks is usually followed by clinical and biochemical recovery in 2 to 12 weeks. The recovery phase is more prolonged in hepatitis B and hepatitis C.
- Up to 1% of cases of acute hepatitis may develop a severe form of the disease (fulminant hepatitis), and 5-10% of cases progress to chronic hepatitis.
- Evolution into the carrier state (except in HAV and HEV infection) has already been described above.
Morphologic Features: Grossly, the liver is slightly enlarged, soft and greenish. Histologically, the changes are as follows:
1. Hepatocellular injury: There may be variation in the degree of liver cell injury but it is most marked in zone 3 (centrilobular zone):
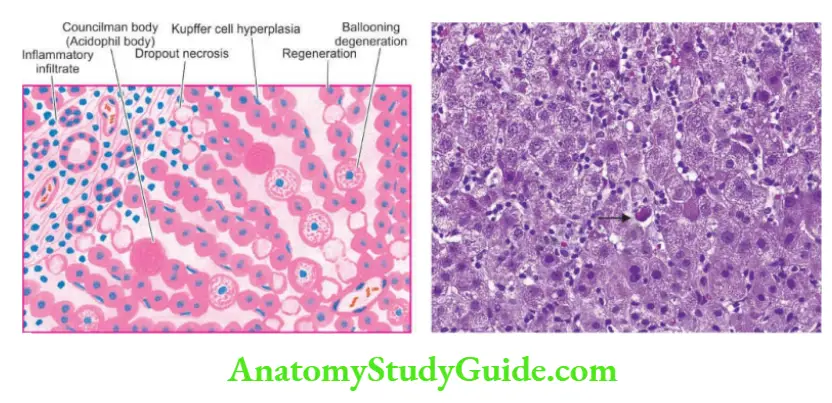
- Mildly injured hepatocytes appear swollen with granular cytoplasm which tends to condense around the nucleus (ballooning degeneration).
- Others show acidophilic degeneration in which the cytoplasm becomes intensely eosinophilic, the nucleus becomes small and pyknotic and is eventually extruded from the cell, leaving behind necrotic, acidophilic mass called Councilman body or acidophil body by the process of apoptosis.
- Another type of hepatocellular necrosis is dropout necrosis in which isolated or small clusters of hepatocytes undergo lysis.
- Bridging necrosis is a more severe form of hepatocellular injury in acute viral hepatitis and may progress to fulminant hepatitis or chronic hepatitis (discussed below).
Bridging necrosis is characterised by bands of necrosis linking portal tracts to central hepatic veins, one central hepatic vein to another, or a portal tract to another tract.
2. Inflammatory infiltrate: There is infiltration by mononuclear inflammatory cells, usually in the portal tracts, but may permeate into the lobules.
3. Kupffer cell hyperplasia: There is reactive hyperplasia of Kupffer cells many of which contain phagocytosed cellular debris, bile pigment and lipofuscin granules.
4. Cholestasis: Biliary stasis is usually not severe in viral hepatitis and may be present as intracytoplasmic bile pigment granules.
5. Regeneration: As a result of necrosis of hepatocytes, there is lobular disarray. Surviving adjacent hepatocytes undergo regeneration and hyperplasia.
- If the necrosis causes the collapse of the reticulin framework of the lobule, healing by fibrosis follows, distorting the lobular architecture.
- The above histologic changes apply to viral hepatitis by various types of hepatotropic viruses in general and by HBV in particular.
- It is usually not possible to distinguish histologically between viral hepatitis of various etiologies, but the following morphologic features may help in giving an etiologic clue:
- HAV hepatitis is a pan-lobular involvement by heavy inflammatory infiltrate compared to other types.
- HCV hepatitis causes milder necrosis, with fatty change in hepatocytes, showing the presence of lymphoid aggregates in the portal triads and degeneration of bile duct epithelium.
4. Chronic Hepatitis
- Chronic hepatitis is defined as continuing or relapsing hepatic disease for more than 6 months with symptoms along with biochemical, serologic and histopathologic evidence of inflammation and necrosis.
- The majority of cases of chronic hepatitis are the result of infection with hepatotropic viruses—hepatitis B, hepatitis C and combined hepatitis B and hepatitis D infection.
- However, some non-viral causes of chronic hepatitis include Wilson’s disease, α-1-antitrypsin deficiency, chronic alcoholism, drug-induced injury and autoimmune diseases.
- The last name gives rise to autoimmune or lupoid hepatitis which is characterised by positive serum autoantibodies (for example antinuclear, anti-smooth muscle and anti-mitochondrial) and a positive LE cell test but negative for serologic markers of viral hepatitis.
- Until recent years, prediction of the prognosis of chronic hepatitis used to be made on the basis of morphology which divided it into 2 main types chronic persistent and chronic active (aggressive) hepatitis.
- A third form, chronic lobular hepatitis is distinguished separately by some as a mild form of lobular inflammation without inflammation of portal tracts but these cases often recover completely.
- However, subsequent studies have revealed that morphologic subtypes do not necessarily correlate with prognosis since the disease is not essentially static but may vary from a mild form to severe and vice versa.
- Besides, two other factors which determine the vulnerability of a patient of viral hepatitis to develop chronic hepatitis are impaired immunity and the extreme age at which the infection is first contracted.
- Currently, therefore, chronic hepatitis is classified on the basis of aetiology and hepatitis activity score (described below).
The frequency and severity with which hepatotropic viruses cause chronic hepatitis vary with the organisms as under
- HCV infection accounts for 40-60% of cases of chronicity in adults. HCV infection is particularly associated with the progressive form of chronic hepatitis that may evolve into cirrhosis.
- HBV causes chronic hepatitis in 90% of infected infants and in about 5% of adult cases of hepatitis B.
- HDV superinfection on HBV carrier state may be responsible for chronic hepatitis in 10-40% of cases.
- HAV and HEV do not produce chronic hepatitis.
Morphologic Features: The pathologic features are common to both HBV and HCV infection and include the following lesions.
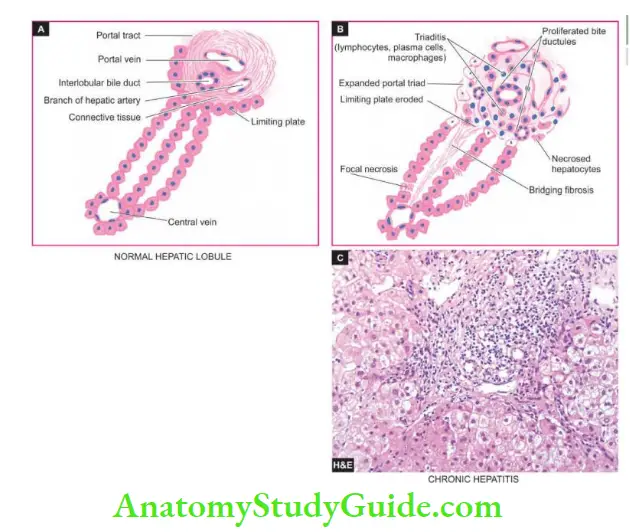
1. Piecemeal necrosis: is defined as periportal destruction of hepatocytes at the limiting plate (piecemeal = piece by piece). Its features in chronic hepatitis are as under:
- Necrosed hepatocytes at the limiting plate in a periportal zone. Interface hepatitis due to expanded portal tract by infiltration of lymphocytes, plasma cells and macrophages.
- Expanded portal tracts are often associated with proliferating bile ductules as a response to liver cell injury.
2. Portal tract lesions: All forms of chronic hepatitis are characterised by variable degrees of changes in the portal tract.
- Inflammatory cell infiltration by lymphocytes, plasma cells and macrophages (triaditis).
- Proliferated bile ductules in the expanded portal tracts.
- Additionally, chronic hepatitis C may show lymphoid aggregates or follicles with reactive germinal centres and infiltration of inflammatory cells in the damaged bile duct epithelial cells.
3. Intralobular lesions: Generally, the architecture of the lobule is retained in mild to moderate chronic hepatitis.
- There are focal areas of necrosis and inflammation within the hepatic parenchyma.
- Scattered acidophilic bodies in the lobule.
- Kupffer cell hyperplasia.
- The more severe form of injury shows bridging necrosis (i.e. bands of necrosed hepatocytes that may bridge portal tract-to-central vein, central vein-to-central vein, and portal tract-Autoportal tract).
- Regenerative changes in hepatocytes in cases of persistent hepatocellular necrosis.
- Cases of chronic hepatitis C show moderate fatty change.
- Cases of chronic hepatitis B show scattered ground-glass hepatocytes indicative of an abundance of HBsAg in the cytoplasm.
- Immunohistochemical stains can be employed for the diagnosis of specific hepatitis viruses such as anti-HBs immunostain, anti-HBc, anti-HD immunostains etc.
4. Bridging fibrosis: The onset of fibrosis in chronic hepatitis from the area of interface hepatitis and bridging necrosis is a feature of irreversible damage.
- At first, there is periportal fibrosis at the sites of interface hepatitis giving the portal tract a stellate-shaped appearance.
- Progressive cases show bridging fibrosis connecting portal tract-to-portal tract or portal tract-to-central vein traversing the lobule.
- End-stage of chronic hepatitis is characterised by dense collagenous septa destroying lobular architecture and forming nodules resulting in postnecrotic cirrhosis.
-
- As a prognostic indicator of chronic hepatitis, a histologic grading of chronic hepatitis (ranging from none to minimal/mild to moderate and severe) was originally described by Knodell and Ishak.
- A combined histologic grade leads to hepatitis activity index (HAI) and takes the following features into consideration:
A. Necroinflammatory activity:
- Periportal necrosis i.e. piecemeal necrosis and/or bridging necrosis (ranging from a score of 0 as ‘no necrosis’ to a score of 4 as ‘multilobular necrosis’).
- Intralobular necrosis, focal or confluent (ranging from score 0 as ‘none’ to score 4 for ‘>10 foci’ for focal necrosis, and score 6 as ‘panacinar/multiacinar’ for confluent necrosis).
- Extent and depth of portal inflammation (ranging from grade 0 as ‘no inflammation’ to grade 4 having ‘marked portal inflammation’).
B. Stage of fibrosis:
- Extent and density of fibrosis (ranging from score 0 as ‘no fibrosis’ to score 6 as ‘cirrhosis’).
Clinical Features: The clinical features of chronic hepatitis are quite variable ranging from mild disease to a full-blown picture of cirrhosis.
- Mild chronic hepatitis shows only slight but persistent elevation of transaminases (‘transaminitis’) with fatigue, malaise and loss of appetite.
- Other cases may show mild hepatomegaly, hepatic tenderness and mild splenomegaly.
- Laboratory findings may reveal prolonged prothrombin time, hyperbilirubinaemia, hyperglobulinaemia and markedly elevated alkaline phosphatase.
- Systemic features of circulating immune complexes due to HBV and HCV infection may produce features of immune complex vasculitis, glomerulonephritis and cryoglobulinaemia in a proportion of cases.
- However, clinical features do not correlate with the morphologic appearance of the liver biopsy.
- Some patients may have a mild form of disease without progressing for several years while others may show rapid evolution into cirrhosis with its complications over a period of a few years.
- Patients of long-standing HBV and HCV chronic infection are known to evolve into hepatocellular carcinoma.
5. Fulminant Hepatitis (Submassive to Massive Necrosis)
- Fulminant hepatitis is the most severe form of acute hepatitis in which there is rapidly progressive hepatocellular failure.
- Two patterns are recognised submassive necrosis having a less rapid course extending up to 3 months; and massive necrosis in which the liver failure is rapid and fulminant occurring in 2-3 weeks.
- Fulminant hepatitis of either of the two varieties can occur from viral and non-viral etiologies:
- Acute viral hepatitis accounts for about half the cases, most often from HBV and HCV; less frequently from combined HBV-HDV and rarely from HAV.
However, HEV infection is a serious complication in pregnant women. In addition, herpesvirus can also cause serious viral hepatitis. - Non-viral causes include acute hepatitis due to drug toxicity (for example acetaminophen, nonsteroidal anti-inflammatory drugs, isoniazid, halothane and anti-depressants), poisonings, hypoxic injury and massive infiltration of malignant tumours into the liver.
- The patients present with features of hepatic failure with hepatic encephalopathy. The mortality rate is high if hepatic transplantation is not undertaken.
- Acute viral hepatitis accounts for about half the cases, most often from HBV and HCV; less frequently from combined HBV-HDV and rarely from HAV.
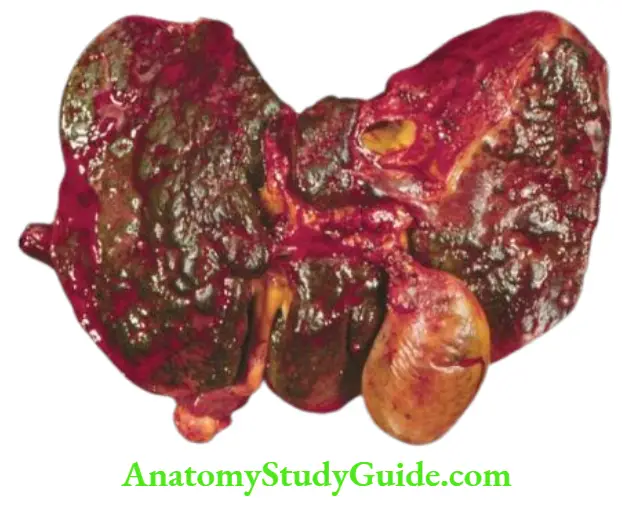
Morphologic Features: Grossly, the liver is small and shrunken, often weighing 500- 700 gm. The capsule is loose and wrinkled.
- The sectioned surface shows diffuse or random involvement of hepatic lobes.
- There are extensive areas of muddy-red and yellow necrosis (previously called acute yellow atrophy) and patches of green bile staining. Histologically, two forms of fulminant necrosis are distinguished—submassive and massive
necrosis.- In submassive necrosis, large groups of hepatocytes in zone 3 (centrilobular area) and zone 2 (mid zone) are wiped out leading to a collapsed reticulin framework.
Regeneration in submassive necrosis is more orderly and may result in the restoration of normal architecture - In massive necrosis, the entire liver lobules are necrotic.
As a result of the loss of hepatic parenchyma, all that is left is the collapsed and condensed reticulin framework and portal tracts with proliferated bile ductules plugged with bile. Inflammatory infiltration is scanty.
Regeneration, if it takes place, is disorderly forming irregular masses of hepatocytes. Fibrosis is generally not a feature of fulminant hepatitis.
- In submassive necrosis, large groups of hepatocytes in zone 3 (centrilobular area) and zone 2 (mid zone) are wiped out leading to a collapsed reticulin framework.
Immunoprophylaxis And Hepatitis Vaccines
- The best prophylaxis against viral hepatitis remains the prevention of its spread to the contacts after detection and identification of the route by which infection is acquired such as from food or water contamination, sexual spread or parenteral spread.
- Of late, however, immunoprophylaxis and a few hepatitis vaccines have been developed and some more are under development.
- The principle underlying either of these two forms of prophylaxis is that the persons who develop a good antibody response to the antigen of the hepatotropic virus following active infection are protected against the disease on reinfection.
- Thus, pre-testing of persons may be carried out so as to determine their antibody levels. Immunoprophylaxis and hepatitis vaccination are unnecessary if the pre-testing for antibodies is positive.
1. Hepatitis A Passive immunisation with immune globulin as well as active immunisation with a killed vaccine are available.
2. Hepatitis B Earlier, only passive immunoprophylaxis with standard immune globulin was used. Later, active immunisation against HBsAg was introduced. Current recommendations include pre-exposure and post-exposure prophylaxis with recombinant hepatitis B vaccine:
- Pre-exposure prophylaxis is done for individuals at high-risk e.g. healthcare workers, haemodialysis patients and staff, haemophiliacs, intravenous drug users etc.
Three intramuscular injections of hepatitis vaccine at 0, 1 and 6 months are recommended. - Post-exposure prophylaxis is carried out for unvaccinated persons exposed to HBV infection and includes prophylaxis with the combination of hepatitis B immune globulin and hepatitis B vaccine.
3. Hepatitis D infection can also be prevented by hepatitis B vaccine.
4. Hepatitis C Currently, the hepatitis C vaccine has yet not been feasible though antibodies to the HCV envelope have been developed.
5. Hepatitis E It is not certain whether immune globulin (like for HAV) prevents hepatitis E infection or not but a vaccine against HEV is yet to be developed.
Viral Hepatitis
- Viral hepatitis is an infection of the liver caused by hepatotropic viruses. A, B, C, D and E.
- Hepatitis A is a faecally-spread self-limiting disease. Early, IgM and later IgG, antibodies appear in the blood.
- Hepatitis B is transmitted parenterally through blood and blood products. There are several immunological markers in the serum: HbsAg, anti-HBs, HbeAg, anti-Hbe, HbcAg, and anti-HBc.
- Hepatitis D develops with hepatitis B either as a co-infection or may infect an HBsAg carrier (superinfection).
- Hepatitis C infection is acquired by blood transfusions.
- Hepatitis E is an enteric infection.
- The carrier state exists for hepatitis B and C only.
- The morphology of acute and fulminant hepatitis caused by different hepatotropic viruses is similar.
- Chronic hepatitis may occur in infection with hepatitis B, C and combined HBV-HDV.
- Immunoprophylaxis by a few hepatitis vaccines is available for high-risk populations.
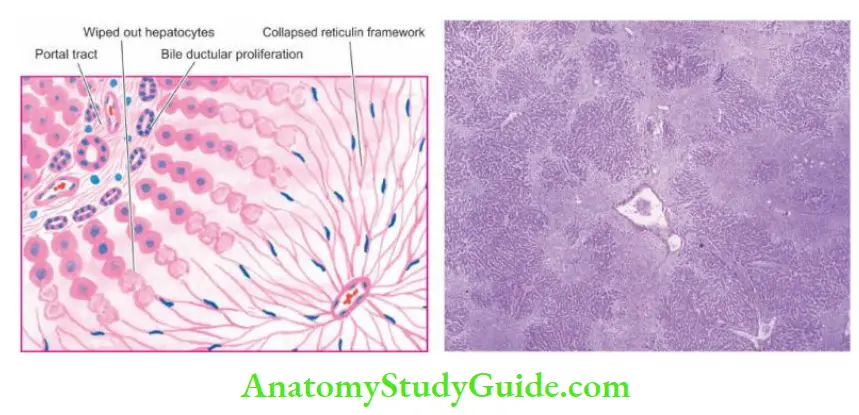
Other Infections
- Apart from viral hepatitis, the liver is affected by infections with bacteria, spirochaetes and fungi and is involved in some parasitic infestations. Some common examples of such conditions are described below.
Chemical And Drug Injury
Hepatic Drug Metabolism: The liver plays a central role in the metabolism of a large number of organic and inorganic chemicals and drugs which gain access to the body by inhalation, injection, or most commonly, via the intestinal tract.
- The main drug metabolising system resides in the microsomal fraction of the smooth endoplasmic reticulum of the liver cells via P-450 cytochrome and cytochrome reductase enzyme systems.
- Other steps involved in drug metabolism are its conjugation with an endogenous molecule, its active transport from the hepatocytes and ultimately its excretion in the bile or in urine depending upon the molecular weight of the substance.
- A number of risk factors predispose an individual to hepatic drug injury such as pre-existing liver disease, ageing, female sex and genetic inability to perform a particular biotransformation.
- Hepatotoxicity Toxic liver injury produced by drugs and chemicals may virtually mimic any form of naturally-occurring liver disease.
- In fact, any patient presenting with liver disease or unexplained jaundice is thoroughly questioned about a history of drug intake or exposure to chemicals.
Hepatotoxicity: from drugs and chemicals is the commonest form of iatrogenic disease. The severity of hepatotoxicity is greatly increased if the drug is continued after symptoms develop.
- Among the various inorganic compounds producing hepatotoxicity are arsenic, phosphorus, copper and iron.
- Organic agents include certain naturally-occurring plant toxins such as pyrrolizidine alkaloids, mycotoxins and bacterial toxins.
- The synthetic group of organic compounds are a large number of medicinal agents.
- In addition, exposure to hepatotoxic compounds may be occupational, environmental or domestic that could be accidental, homicidal or suicidal ingestion.
In general, drug reactions affecting the liver are divided into two main classes:
1. Direct or predictable, when the drug or one of its metabolites is either directly toxic to the liver or it lowers the host’s immune defence mechanism.
- The adverse effects occur in most individuals who consume them and their hepatotoxicity is dose-dependent example carbon tetrachloride.
2. Indirect or unpredictable or idiosyncratic, when the drug or one of its metabolites acts as a hapten and induces hypersensitivity in the host.
- In many instances, drug hepatotoxicity is associated with the appearance of autoantibodies to liver-kidney microsomes ( anti-LKM2) directed against cytochrome P450 enzyme.
- The hepatotoxicity by this group does not occur regularly in all individuals and the effects are usually not dose-related example acetaminophen.
- A simplified clinicopathologic classification of important hepatic drug reactions and the agents causing them is presented.
- The changes produced by hepatotoxic agents may vary from mild, which are diagnosed only by elevated serum transaminases, to instances of massive necrosis and death.
The pathologic changes by hepatotoxins include 2 large categories:
- Acute liver disease is characterised by cholestasis, hepatocellular necrosis, fatty change, granulomatous reaction or vascular disease.
- Chronic liver disease is characterised by variable degrees of fibrosis, cirrhosis or neoplasia
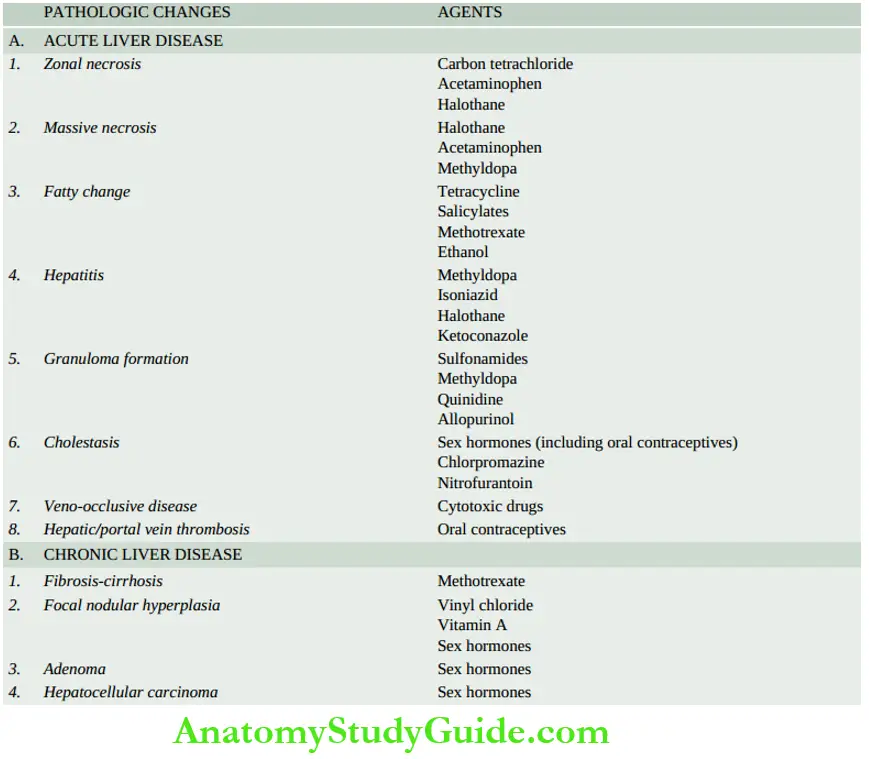
As such, the pathologic changes induced by hepatotoxins are indistinguishable from the respective disease states.
Chemical and Drug Injury
- The liver plays a central role in the metabolism of a large number of organic and inorganic chemicals and drugs which enter the body by inhalation, injection, or via the intestinal tract.
- A toxic liver injury produced by drugs and chemicals may mimic any form of naturally-occurring liver disease.
- Drug reactions affecting the liver may be direct or predictable, and indirect or unpredictable.
Cirrhosis
Cirrhosis of the liver is one of the ten leading causes of death in the Western world. It represents the irreversible end-stage of several diffuse diseases causing hepatocellular injury and is characterised by the following 4 features:
- It involves the entire liver.
- The normal lobular architecture of the hepatic parenchyma is disorganised.
- There is a formation of nodules separated from one another by irregular bands of fibrosis.
- It occurs following hepatocellular necrosis of varying aetiology so that there are alternate areas of necrosis and regenerative nodules.
However, regenerative nodules are not essential for the diagnosis of cirrhosis since biliary cirrhosis and cirrhosis in haemochromatosis have little regeneration.
Pathogenesis
- Irrespective of the aetiology, cirrhosis involves a combination of a few processes: hepatocellular necrosis, healing by fibrosis, formation of compensatory regenerative nodules and changes in the vascular pattern of the hepatic parenchyma.
Fibrogenesis: Continued destruction of hepatocytes causes collapse of normal lobular hepatic parenchyma followed by fibrosis around necrotic liver cells.
- Fibrosis in the liver lobules may be portal-central, portal-portal, or both. The mechanism of fibrosis is by increased synthesis of type I and III collagen in the space of Disse.
- There is a proliferation of fat-storing Ito cells underlying the sinusoidal epithelium which are transformed into myofibroblasts and fibrocytes.
- Besides collagen, two glycoproteins, fibronectin and laminin, are deposited in excessive amounts in areas of liver cell damage.
- Stimulants for fibrosis are several growth factors (for example platelet-derived growth factor receptor-β, transforming growth factor-β, metalloproteinase), vasoactive factors, cytokines, lymphokines and chemokines released from lymphocytes, Kupffer cells, endothelial cells and hepatocytes.
Regenerative Nodules: The surviving hepatocytes act as stimulants for the growth and proliferation of more hepatocytes under the influence of growth factors.
- This compensatory proliferation of hepatocytes is restricted within fibrous nodules forming regenerative nodules.
Vascular Reorganisation: Due to damaged hepatic parenchyma and the formation of fibrous nodules, the new vessels formed in the fibrous septa are connected to the vessels in the portal triad ( branches of hepatic artery and portal vein) and then the blood is drained into the hepatic vein.
- This way, the blood bypasses the hepatic parenchyma.
- Moreover, due to fibrous proliferation in the space of Disse, gaps in the hepatic sinusoids are closed which results in the development of capillaries in the sinusoids (capillarization of sinusoids).
Vascular Reorganisation Classification
- Cirrhosis can be classified on the basis of morphology and aetiology.
A. Morphologic Classification: There are 3 morphologic types of cirrhosis micronodular, macronodular and mixed. Each of these forms may have an active and inactive form.
- An active form is characterised by continuing hepatocellular necrosis and inflammatory reaction, a process that closely resembles chronic hepatitis.
- An inactive form, on the other hand, has no evidence of continuing hepatocellular necrosis and has sharply defined nodules of surviving hepatic parenchyma without any significant inflammation.
1. Micronodular cirrhosis: In micronodular cirrhosis, the nodules are usually regular and small, less than 3 mm in diameter.
- There is diffuse involvement of all the hepatic lobules forming nodules by thick fibrous septa which may be portal-portal, portal-central, or both.
- Micronodular cirrhosis includes the etiologic type of alcoholic cirrhosis (or nutritional cirrhosis or Laennec’s cirrhosis) and represents the impaired capacity for regrowth as seen in chronic alcoholism.
2. Macronodular cirrhosis: In this type, the nodules are of variable size and are generally larger than 3 mm in diameter.
- The pattern of involvement is more irregular than in micronodular cirrhosis, sparing some portal tracts and central veins, and more marked evidence of regeneration.
- Macronodular cirrhosis corresponds to post-necrotic (or post-viral hepatitis) cirrhosis of the etiologic classification.
3. Mixed cirrhosis: In mixed type, some parts of the liver show a micronodular appearance while other parts show a macronodular pattern.
- All the portal tracts and central veins are not involved in fibrosis but instead, some of them are spared. A mixed pattern is a kind of incomplete expression of micronodular cirrhosis.

2. Etiologic Classification: Based on the etiologic agent for cirrhosis, various categories of cirrhosis are described as given in.
Specific Types Of Cirrhosis
Alcoholic Liver Disease and Cirrhosis
- Alcoholic liver disease is the term used to describe the spectrum of liver injury associated with acute and chronic alcoholism.
- There are three sequential stages in alcoholic liver disease: alcoholic steatosis (fatty liver), alcoholic hepatitis and alcoholic cirrhosis.
- Before discussing the features of alcoholic liver disease and cirrhosis, a brief outline of ethanol metabolism is outlined below and is discussed earlier in Chapter 9.
Ethanol Metabolism: One gram of alcohol gives 7 calories but alcohol cannot be stored in the body and must undergo obligatory oxidation, chiefly in the liver.
- Thus, these empty calories make no contribution to nutrition other than to give energy.
- Ethanol after ingestion and absorption from the small bowel circulates through the liver where about 90% of it is oxidised to acetate by a two-step enzymatic process involving two enzymes.
- Alcohol dehydrogenase (ADH) is present in the cytosol, and acetaldehyde dehydrogenase (ALDH) in the mitochondria of hepatocytes. The remaining 10% of ethanol is oxidised elsewhere in the body.
First step: Ethanol is catabolised to acetaldehyde in the liver by the following three pathways, one major and two minor
- In the cytosol, by the major rate-limiting pathway of alcohol dehydrogenase (ADH).
- In the smooth endoplasmic reticulum, via microsomal P-450 oxidases (also called microsomal ethanol oxidising system, MEOS), where only part of ethanol is metabolised.
- In the peroxisomes, minor pathways via catalase such as H2O2.
Acetaldehyde is toxic and may cause membrane damage and cell necrosis. Simultaneously, the cofactor nicotinamide-adenine dinucleotide (NAD) which is a hydrogen acceptor, is reduced to NADH
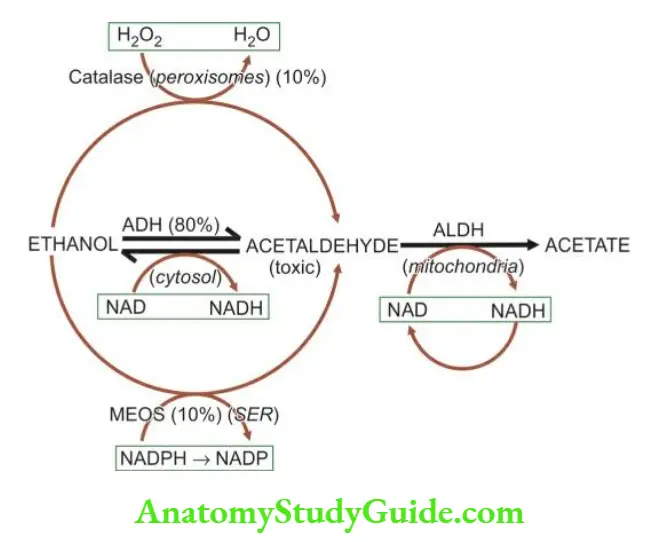
Second step: In the mitochondria, ALDH acts as a co-enzyme and converts acetaldehyde to acetate.
- Most of the acetate on leaving the liver is finally oxidised to carbon dioxide and water, or converted by the citric acid cycle to other compounds including fatty acids.
- Simultaneously, the same cofactor, NAD, is reduced to NADH resulting in increased NADH: NAD redox ratio which is the basic biochemical alteration occurring during ethanol metabolism.
- A close estimate of NADH NAD ratio is measured by the ratio of its oxidised and reduced metabolites in the form of lactate-pyruvate ratio and β-hydroxy butyrate-acetoacetate ratio.
Risk Factors For Alcoholic Liver Disease: All those who indulge in alcohol abuse do not develop liver damage.
- The incidence of cirrhosis among alcoholics at autopsy is about 10-15%. Why some individuals are predisposed to alcoholic cirrhosis is not clearly known, but a few risk factors have been implicated. These are as under
1. Drinking patterns: Most epidemiologic studies have attributed alcoholic cirrhosis to chronic alcoholism.
- Available evidence suggests that chronic and excessive consumption of alcohol invariably leads to fatty liver in >90% of chronic alcoholics, progression to alcoholic hepatitis in 10-20% of cases, and eventually to alcoholic cirrhosis in more than 10 years.
- It is generally agreed that continued daily imbibing of 60-80 gm of ethanol in any type of alcoholic beverage for at least 10 years is likely to result in alcoholic cirrhosis.
- Liver injury is related to the quantity of ethanol contained in alcoholic beverages consumed and its duration, but not related to the type of alcoholic beverage consumed.
- The ethanol content in an alcoholic beverage is given on the label of the container, but in general, it is about 4-6% in beer, 10-12% in wine, and about 40-50% in brandy, whisky and scotch.
- Intermittent drinking for a long duration is less harmful since the liver is given a chance to recover.
2. Gender: Women have increased susceptibility to developing advanced alcoholic liver disease with much lesser alcohol intake (20-40 g/day).
- This gender difference in disease progression is unclear but is probably linked to the effects of oestrogen.
3. Malnutrition: Absolute or relative malnutrition of proteins and vitamins is regarded as a contributory factor in the evolution of cirrhosis.
- The combination of chronic alcohol ingestion and impaired nutrition leads to alcoholic liver disease and not malnutrition per se.
- It appears that calories derived from alcohol displace other nutrients leading to malnutrition and deficiency of vitamins in alcoholics.
- Additional factors contributing to malnutrition in alcoholics are chronic gastritis and pancreatitis.
- The evidence in favour of the synergistic effect of malnutrition in chronic alcoholism comes from clinical and morphologic improvement in cases of alcoholic cirrhosis on treatment with protein-rich diets.
4. Infections: Intercurrent bacterial infections are common in cirrhotic patients and may accelerate the course of the disease.
- Lesions similar to alcoholic cirrhosis may develop in nonalcoholic patients who have had viral infections in the past.
5. Genetic factors: The rate of ethanol metabolism is under genetic control.
- It is chiefly related to altered rates of elimination of ethanol due to genetic polymorphism for the two main enzyme systems, MEOS (microsomal P-450 oxidases) and alcohol dehydrogenase (ADH).
- Various HLA histocompatibility types have been associated with the susceptibility of individuals to alcoholic liver damage but no single genotype has been identified yet.
6. Hepatitis B and C infection: Concurrent infection with either HBV or HCV is an important risk factor for the progression of alcoholic liver disease.
- HBV or HCV infection in chronic alcoholics leads to the development of alcoholic liver disease with much less alcohol consumption (20-50 g/day), disease progression at a younger age, having greater severity, and increased risk to develop cirrhosis and hepatocellular carcinoma, and overall poorer survival.
Pathogenesis: Exact pathogenesis of alcoholic liver injury is yet unclear as to why only some chronic alcoholics develop the complete sequence of changes in the liver while others do not.
- However, knowledge and understanding of ethanol metabolism have resulted in discarding the old concept of liver injury due to malnutrition.
- Instead, it is now known that ethanol and its metabolites are responsible for ill effects on the liver in a susceptible chronic alcoholic having the above-mentioned risk factors.
- Briefly, the biomedical and cellular pathogenesis due to chronic alcohol consumption culminating in morphologic lesions of alcoholic steatosis (fatty liver), alcoholic hepatitis and alcoholic cirrhosis can be explained under and is schematically illustrated in.
1. Direct hepatotoxicity by ethanol: There is evidence to suggest that ethanol ingestion for a period of 8-10 days regularly may cause a direct hepatotoxic effect on the liver and produce fatty change.
- Ethanol is directly toxic to microtubules, mitochondria and the membrane of hepatocytes.
2. Hepatotoxicity by ethanol metabolites: Major hepatotoxic effects of ethanol are exerted by its metabolites, chiefly acetaldehyde.
- Acetaldehyde levels in the blood are elevated in chronic alcoholics. Acetaldehyde produces hepatotoxicity by the formation of two adducts
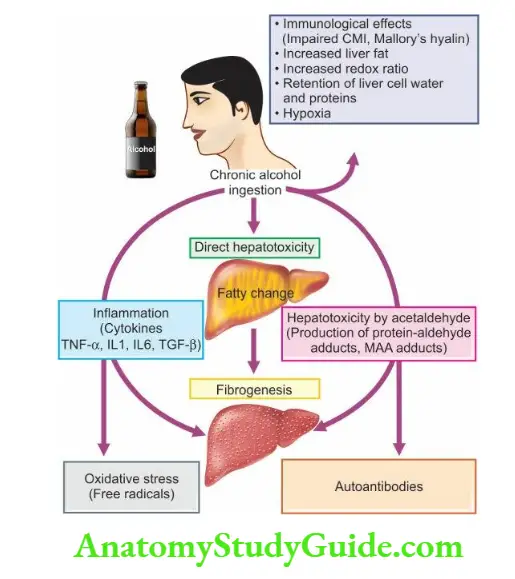
- Production of protein-aldehyde adducts which are extremely toxic and can cause cytoskeletal and membrane damage and bring about hepatocellular necrosis.
- Formation of malon-di-aldehyde-acetaldehyde (MAA) adducts which produce autoantibodies and initiate an autoimmune response.
- These adducts have also a role in hepatic fibrogenesis due to peroxisome proliferator-activated receptor (PPAR)-γ on hepatocytes.
3. Oxidative stress: Oxidation of ethanol by the cytochrome-450 oxidases (MEOS) leads to the generation of free radicals which causes oxidative damage to the membranes and proteins.
4. Immunological mechanism: Cell-mediated immunity is impaired in alcoholic liver disease. Ethanol causes direct immunologic attacks on hepatocytes.
- In a proportion of cases, alcohol-related liver cell injury continues unabated despite cessation of alcohol consumption which is attributed to immunologic mechanisms.
- An immunological mechanism may also explain the genesis of Mallory’s alcoholic hyalin though a more favoured hypothesis for its origin is the aggregation of intermediate filaments of keratin type due to alcohol-induced disorganisation of the cytoskeleton.
5. Inflammation: Chronic ethanol ingestion is not only injurious to hepatocytes but also damages the intestinal cells.
- The injured intestinal cells elaborate endotoxins which release proinflammatory cytokines, chiefly tumour necrosis factor-α, IL-1, IL-6 and TGF-β.
- These cytokines and endotoxemia produce apoptosis and necrosis of hepatocytes and initiate inflammatory reactions in the alcohol-damaged liver.
6. Fibrogenesis: Main event facilitating hepatic fibrogenesis is the activation of stellate cells by various stimuli:
- by damaged hepatocytes,
- by Malon-di-aldehyde-acetaldehyde adducts,
- by activated Kupffer cells, and
- direct stimulation by acetaldehyde.
All forms of collagen are increased and there is increased transformation of fat-storing Ito cells into myofibroblasts and fibrocytes.
7. Increased redox ratio: Marked increase in the NADH: NAD redox ratio in the hepatocytes results in an increased redox ratio of lactate-pyruvate, leading to lactic acidosis.
- This altered redox potential has been implicated in a number of metabolic consequences such as in fatty liver, collagen formation, occurrence of gout, impaired gluconeogenesis and altered steroid metabolism.
8. Retention of liver cell water and proteins: Alcohol is inhibitory to the secretion of newly synthesized proteins by the liver leading to their retention in the hepatocytes.
- Water is simultaneously retained in the cell in proportion to the protein and causes swelling of hepatocytes resulting in hepatomegaly in alcoholics.
9. Hypoxia: Chronic ingestion of alcohol results in increased oxygen demand by the liver resulting in a hypoxic state which causes hepatocellular necrosis in the centrilobular zone (zone 3).
Redox changes are also more marked in zone 3.
10. Increased liver fat: The origin of fat in the body was discussed in Chapter 3.
- In chronic alcoholism, there is a rise in the amount of fat available to the liver which could be from exogenous (dietary) sources, excess mobilisation from adipose tissue or increased lipid synthesis by the liver itself.
This may account for lipid accumulation in the hepatocytes
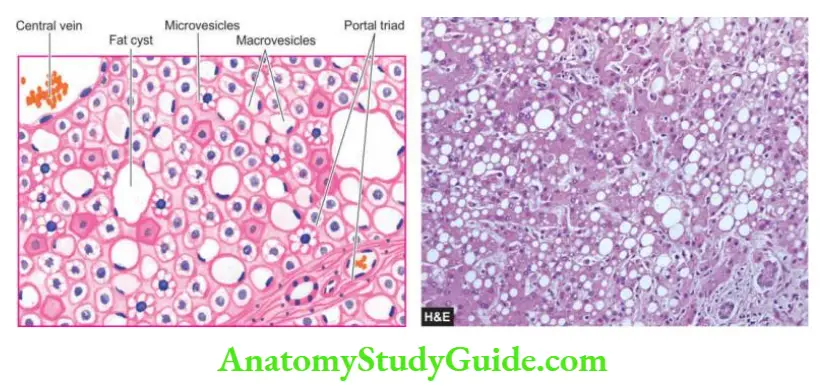
- Morphologic Features Three types of morphologic lesions are described in alcoholic liver disease—alcoholic steatosis (fatty liver), alcoholic hepatitis and alcoholic cirrhosis.
1. Alcoholic Steatosis (Fatty Liver): The morphologic changes in a fatty change in the liver have already been described on and are briefly considered here.
Grossly, the liver is enlarged, yellow, greasy and firm with a smooth and glistening capsule.
Microscopically, the features consist of initial microvesicular droplets of fat in the hepatocyte cytoplasm followed by more common and pronounced features of macrovesicular large droplets of fat displacing the nucleus to the periphery.
- Fat cysts may develop due to the coalescence and rupture of fat-containing hepatocytes. Less often, lipo granulomas consisting of a collection of lymphocytes, macrophages and some multinucleate giant cells may be found.
2. Alcoholic hepatitis: develops acutely, usually following a bout of heavy drinking. Repeated episodes of alcoholic hepatitis superimposed on the pre-existing fatty liver are almost certainly a forerunner of alcoholic cirrhosis.
- Histologically, the features of alcoholic hepatitis are as follows
- Hepatocellular necrosis Single or small clusters of hepatocytes, especially in the centrilobular area (zone 3), undergo ballooning degeneration and necrosis.
- Mallory bodies or alcoholic hyalin These are eosinophilic, intracytoplasmic inclusions seen in perinuclear locations within swollen and ballooned hepatocytes.
- They represent aggregates of cytoskeletal intermediate filaments (keratin). They can be best visualised with connective tissue stains like Masson’s trichrome and chromophobe aniline blue, or by the use of immunoperoxidase methods.
- Mallory bodies are highly suggestive of, but not specific for, alcoholic hepatitis since Mallory bodies are also found in certain other conditions such as.
- primary biliary cirrhosis, Indian childhood cirrhosis, cholestatic syndromes, Wilson’s disease, intestinal bypass surgery, focal nodular hyperplasia and hepatocellular carcinoma.
3. Inflammatory response The areas of hepatocellular necrosis and regions of Mallory bodies are associated with an inflammatory infiltrate, chiefly consisting of polymorphs and some scattered mononuclear cells.
- In more extensive necrosis, the inflammatory infiltrate is more widespread and may involve the entire lobule.
4. Fibrosis Most cases of alcoholic hepatitis are accompanied by pericellular and perivenular fibrosis, producing a web-like or chickenwire-like appearance. This is also termed creeping collagenous.
3. Alcoholic Cirrhosis: is the most common form of a lesion, constituting 60-70% of all cases of cirrhosis.
- Several terms have been used for this type of cirrhosis such as Laennec’s cirrhosis, portal cirrhosis, hobnail cirrhosis, nutritional cirrhosis, diffuse cirrhosis and micronodular cirrhosis.
Grossly, alcoholic cirrhosis classically begins as micronodular cirrhosis (nodules less than 3
mm diameter), the liver is large, fatty and weighing usually above 2 kg.
- Eventually over a span of years, the liver shrinks to less than 1 kg in weight, and becomes nonfatty, having macronodular cirrhosis (nodules larger than 3 mm in diameter), resembling postnecrotic cirrhosis.
- The nodules of the liver due to their fat content are tawny-yellow, on the basis of which Laennec in 1818 introduced the term cirrhosis first of all (from Greek cirrhosis = tawny).
- The surface of the liver in alcoholic cirrhosis is studded with diffuse nodules which vary little in size, producing a hobnail liver (because of the resemblance of the surface with the sole of an old-fashioned shoe having short nails with heavy heads).
- On the cut section, spheroidal or angular nodules of fibrous septa are seen.
Microscopically, alcoholic cirrhosis is a progressive alcoholic liver disease. Its features include the following:
1. Nodular pattern Normal lobular architecture is effaced in which central veins are hard to
find and are replaced with nodule formation.
2. Fibrous septa The fibrous septa that divide the hepatic parenchyma into nodules are initially delicate and extend from the central vein to portal regions, or portal tract to portal tract, or both.
- As the fibrous scarring increases with time, the fibrous septa become dense and more confluent.
3. Hepatic parenchyma The hepatocytes in the islands of surviving parenchyma undergo slow proliferation forming regenerative nodules having disorganised masses of hepatocytes.
- The hepatic parenchyma within the nodules shows extensive fatty change early in the disease. But as the fibrous septa become thicker, the amount of fat in hepatocytes is reduced.
- Thus, there is an inverse relationship between the amount of fat and the amount of fibrous scarring in the nodules.
4. Necrosis, inflammation and bile duct proliferation: The etiologic clue to the diagnosis in the form of Mallory bodies is hard to find in fully-developed alcoholic cirrhosis.
- The fibrous septa usually contain sparse infiltration of mononuclear cells with some bile duct proliferation.
- Bile stasis and increased cytoplasmic haemosiderin deposits due to enhanced iron absorption in alcoholic cirrhosis are some other noticeable findings
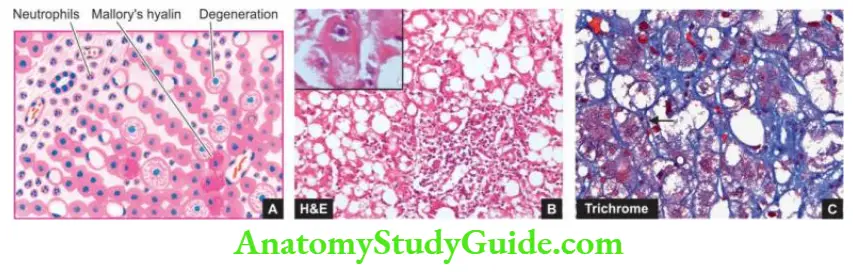
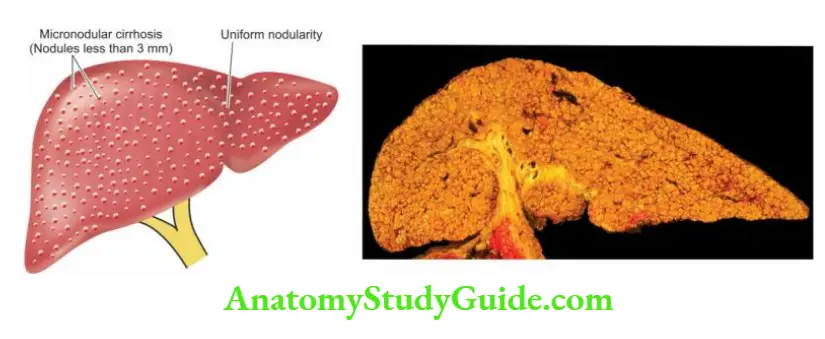
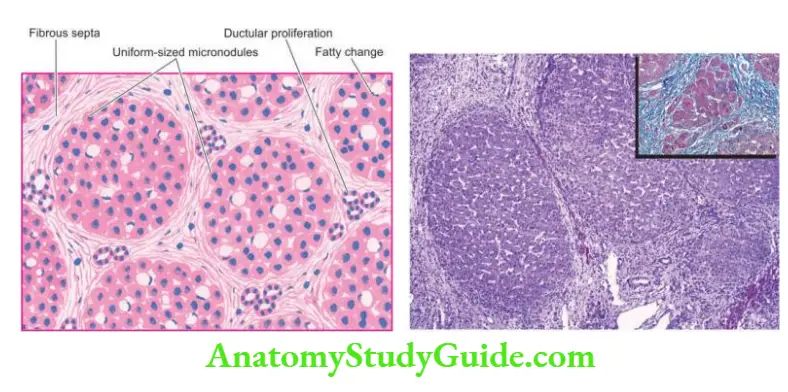
Laboratory Diagnosis: The clinical manifestations and complications of cirrhosis, in general, are described.
- The laboratory findings in the course of alcoholic liver disease may be quite variable and liver biopsy is necessary in doubtful cases.
The progressive form of the disease, however, generally presents the following biochemical and haematological alterations
- Elevated transaminases; increase in SGOT (AST) is more than that of SGPT (ALT).
- Rise in serum γ-glutamyl transpeptidase (γ-GT).
- Elevation in serum alkaline phosphatase.
- Hyperbilirubinaemia.
- Hypoproteinaemia with reversal of albumin-globulin ratio.
- Prolonged prothrombin time and partial thromboplastin time.
- Anaemia.
- Neutrophilic leucocytosis in alcoholic hepatitis and in secondary infections.
Post-necrotic Cirrhosis
Post-necrotic cirrhosis, also termed post-hepatitic cirrhosis, macronodular cirrhosis and coarsely nodular cirrhosis, is characterised by large and irregular nodules with broad bands of connective tissue and occurs most commonly after previous viral hepatitis.
Aetiology: Based on epidemiologic and serologic studies, the following factors have been implicated in the aetiology of post-necrotic cirrhosis.
1. Viral hepatitis About 25% of patients give a history of recent or remote attacks of acute viral hepatitis followed by chronic viral hepatitis.
- The most common association is with hepatitis B and C; hepatitis A is not known to evolve into cirrhosis.
- It is estimated that about 20% of cases of HBV chronic hepatitis and about 20-30% of cases of HCV chronic hepatitis progress to cirrhosis over 20-30 years.
2. Drugs and chemical hepatotoxins: A small percentage of cases may have originated from toxicity due to chemicals and drugs such as phosphorus, carbon tetrachloride, mushroom poisoning, acetaminophen and α-methyldopa.
3. Others: Certain infections ( for example brucellosis), parasitic infestations (for example clonorchiasis), metabolic diseases (for example Wilson’s disease or hepatolenticular degeneration) and advanced alcoholic liver disease may produce a picture of post-necrotic cirrhosis.
4. Idiopathic After all these causes have been excluded, a group of cases remain in which the aetiology is unknown.
Morphologic Features: Typically, post-necrotic cirrhosis is macronodular type. Grossly, the liver is usually small, weighing less than 1 kg, and having a distorted shape with irregular and coarse scars and nodules of varying size.
The sectioned surface shows scars and nodules varying in diameter from 3 mm to a few centimetres.
Microscopically, the features are as follows:
1. Nodular pattern: The normal lobular architecture of hepatic parenchyma is mostly lost and is replaced by nodules larger than those in alcoholic cirrhosis.
- However, uninvolved portal tracts and central veins in the hepatic lobules can still be seen in some parts of surviving parenchyma.
2. Fibrous septa: The fibrous septa dividing the variable-sized nodules are generally thick.
3. Necrosis, inflammation and bile duct proliferation: Active liver cell necrosis is usually inconspicuous.
- Fibrous septa contain prominent mononuclear inflammatory cell infiltrate which may even form follicles, especially in cases following HCV chronic hepatitis.
- Often there is an extensive proliferation of bile ductules derived from collapsed liver lobules.
4. Hepatic parenchyma: Liver cells vary considerably in size and multiple large nuclei are common in regenerative nodules. Fatty change may or may not be present in the hepatocytes.
- Clinical Features: Besides the general clinical features described on, postnecrotic cirrhosis is seen as frequent in women as in men, especially in the younger age group.
- Like in alcoholic cirrhosis, the patients may remain asymptomatic or may present with prominent signs and symptoms of chronic hepatitis.
- Splenomegaly and hypersplenism are other prominent features. The results of haematologic and liver function tests are similar to those of alcoholic cirrhosis.
- Out of the various types of cirrhosis, post-necrotic cirrhosis, especially when related to hepatitis B and C virus infection in early life, is more frequently associated with the development of hepatocellular carcinoma later.
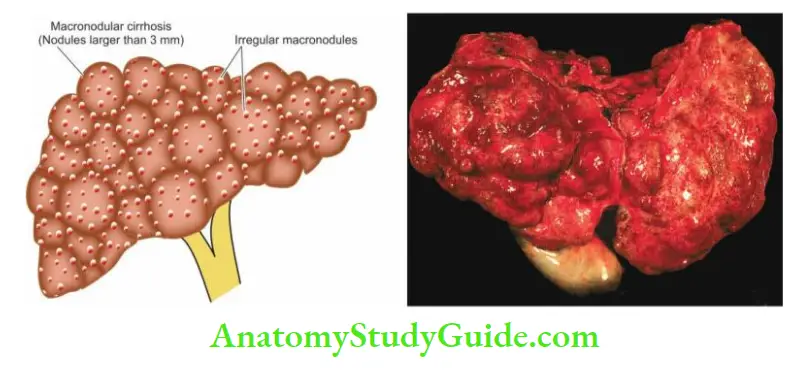
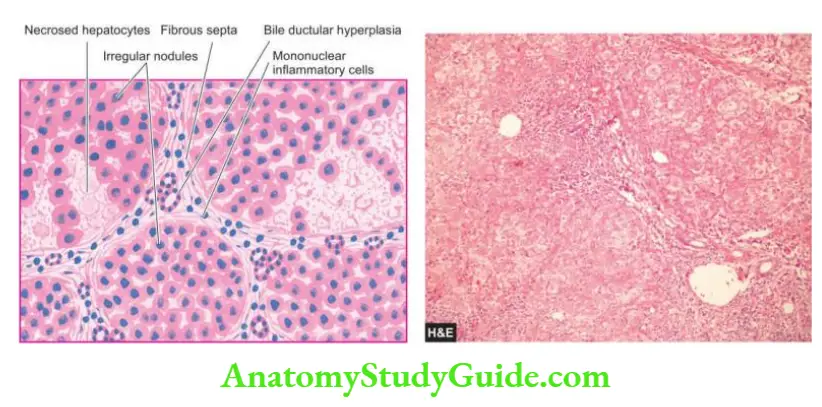
- Although the etiologic and morphologic diagnosis of cirrhosis is attempted by pathologists, invariably it is difficult to reconstruct the course of events which led to end-stage cirrhosis, especially in a liver biopsy.
- However, the main contrasting features of alcoholic and post-necrotic cirrhosis on liver specimens are given in.
Non-Cirrhotic Portal Fibrosis
- Non-cirrhotic portal fibrosis (NCPF) is a group of congenital and acquired diseases in which there is localised or generalised hepatic fibrosis without nodular regenerative activity and there is the absence of clinical and functional evidence of cirrhosis.
- Besides, the patients of NCPF are relatively young as compared to those of cirrhosis and develop repeated bouts of haematemesis in the course of the disease.
- A similar condition described in Japan has been named idiopathic (primary) portal hypertension with splenomegaly.
The type common in India, particularly in young males, is related to the following etiologic factors:
- Exposure to trace elements, particularly chronic arsenic ingestion in drinking water.
- Intake of orthodox medicines.
- Infections, particularly of the umbilical cord, infective diarrhoea and sepsis, cause infection in portal circulation and lead to thrombophlebitis.
- Familial aggregation of disease and its relationship to HLA-DR3.
Morphologic Features: Grossly, the liver is small, and fibrous and shows prominent fibrous septa on both external as well as on cut surf
- Standing out of portal tracts due to their increased amount of fibrous tissue in triad without significant inflammation.
- Obliterative sclerosis of portal vein branches in the portal tracts (obliterative portoven).
Hepatic Tumours And Tumour-Like Lesions
- The liver is the site for benign tumours, tumour-like lesions, and both primary and metastatic malignant tumours. However, metastatic tumours are much more common than primary tumours.
- Tumour-like lesions are masses (space-occupying lesions or SOLs) which may clinically and by imaging studies mimic tumours and include cysts in the liver which may be single or multiple.
- These cysts are mainly of three types congenital, simple (nonparasitic) and hydatid (Echinococcus) cysts. Primary hepatic tumours may arise from hepatic cells, intrahepatic bile duct epithelium, or non-epithelial structures.
- The current WHO classification of tumours of the liver and intrahepatic bile duct epithelium is listed in. Common and important examples are discussed below.
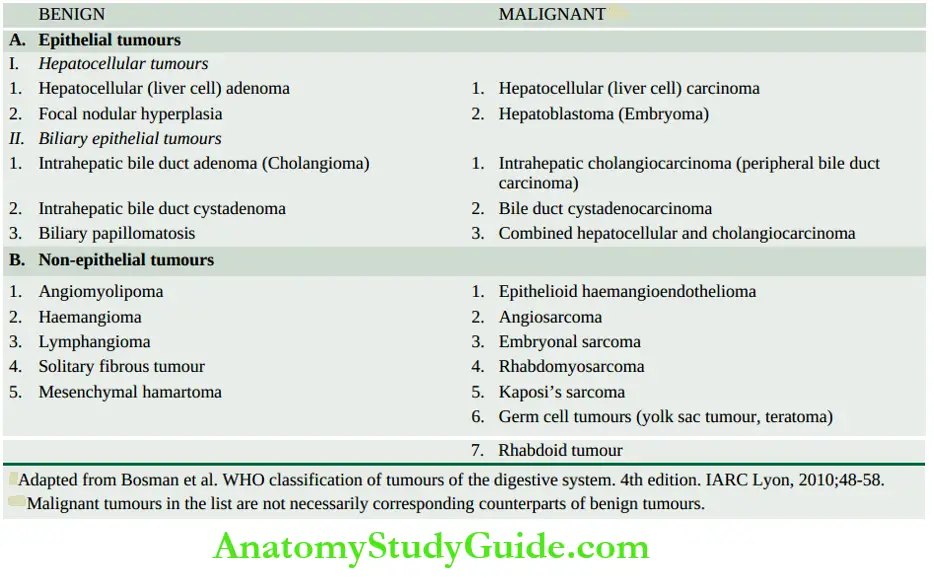
Benign Hepatic Tumours
- These are uncommon and some of them are incidental autopsy findings.
Focal Nodular Hyperplasia
- These lesions are more common in young women taking oral contraceptives.
Grossly, Focal nodular hyperplasia is a well-demarcated tumour-like nodule occurring underneath the Glisson’s capsule.
- The nodules may be single or multiple, measuring about 5 cm in diameter. It may be tan-yellow or bile-stained. The sectioned surface shows a central fibrous scar.
- Histologically, it is composed of a central stellate fibrous scar having a proliferation of bile ductal epithelium at the margin but without portal triads or central hepatic veins in the scar. Hepatocytes are seen surrounding the scar.
Hepatocellular (Liver Cell) Adenoma
- Adenomas arising from hepatocytes are rare and are reported in women in the reproductive age group in association with the use of oral contraceptives, sex hormone therapy and with pregnancy.
- The tumour presents as an intrahepatic mass that may be mistaken for hepatocellular carcinoma and may rupture causing severe intraperitoneal haemorrhage.
Morphologic Features: Grossly, the tumour usually occurs singly but about 10% are multiple. It is partly or completely encapsulated and slightly lighter in colour than the adjacent liver or maybe bile-stained.
- The tumours vary from a few centimetres up to 30 cm in diameter. On the cut section, many of the tumours have varying degrees of infarction and haemorrhage.
Histologically, liver cell adenomas are composed of sheets and cords of bland-looking hepatocytes without mitotic activity.
- The hepatocytes in adenomas contain a greater amount of glycogen than the surrounding liver cells and may sometimes show fatty change.
- Hepatocellular adenomas lack portal tracts and bile ducts but bile canaliculi containing bile plugs may be present. Numerous blood vessels are generally present in the tumour which may be thrombosed.
- Thrombosis leads to infarction and may result in rupture with intraperitoneal haemorrhage.
Bile Duct Adenoma (Cholangioma)
- Intrahepatic or extrahepatic bile duct adenoma is a rare benign tumour. The tumour may be small, composed of acini lined by biliary epithelium and separated by a variable amount of connective tissue, or larger cystadenomas having loculi lined by biliary epithelium.
Haemangioma
Haemangioma is the commonest benign tumour of the liver. The majority of them are asymptomatic and discovered incidentally. Rarely, a haemangioma may rupture into the peritoneal cavity.
Morphologic Features Grossly, haemangiomas appear as solitary or multiple, circumscribed, red-purple lesions, commonly subcapsular and varying from a few millimetres to a few centimetres in diameter.
They are commonly cavernous type giving the sectioned surface a spongy appearance.
Histologically, haemangioma of the liver shows characteristic large, cavernous, blood-filled spaces, lined by a single layer of endothelium and separated by connective tissue.
- Some haemangiomas may undergo progressive fibrosis and may later get calcified.
Malignant Hepatic Tumours
- Among the primary malignant tumours of the liver, hepatocellular (liver cell) carcinoma accounts for approximately 85% of all primary malignant tumours, cholangiocarcinoma for about 5-10%, and infrequently mixed pattern is seen.
- Other tumours listed are less common. Hepatic haemangiosarcoma and embryonal sarcoma resemble in morphology their counterparts elsewhere in the body.
Hepatocellular Carcinoma
- Hepatocellular carcinoma (HCC) or liver cell carcinoma, also termed hepatoma, is the most common primary malignant tumour of the liver.
- The tumour shows marked geographic variations in incidence which is closely related to HBV and HCV infection in the region.
- Whereas the prevalence of HCC is less than 1% of all autopsies in the United States and Europe, the incidence in sub-Saharan Africa and South-East Asia (particularly China) is high (2-8%).
- Liver cell cancer is more common in males than in females in the ratio of 4:1.
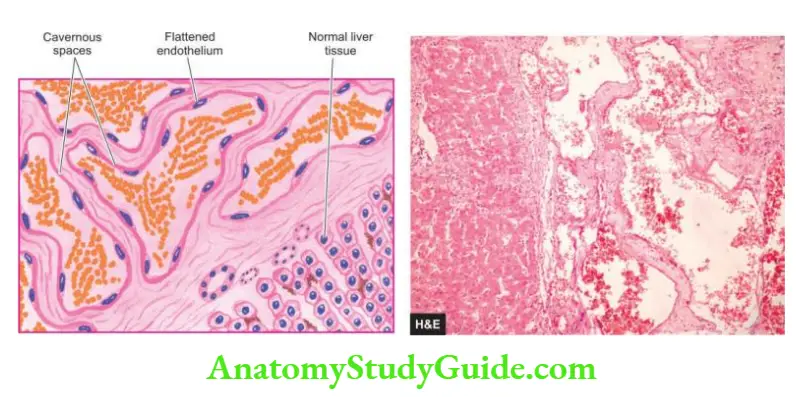
- The peak incidence occurs in 5th to 6th decades of life but in high-incidence areas where HBV and HCV infection is prevalent, it occurs a decade or two earlier. The tumour supervenes cirrhosis in 70-80% of cases.
Etiopathogenesis A number of etiologic factors are implicated in the aetiology of HCC, the most important being HBV and HCV infection, and association with cirrhosis.
1. Relation to chronic HBV infection: Genesis of HCC is linked to prolonged infection with HBV. The evidence in support is both epidemiologic and direct.
- The incidence of HBsAg positivity is higher in HCC patients. For example, in Taiwan, HBsAg-positive carriers have more than 200 times greater risk of developing HCC than HBsAg-negative patients, particularly when the infection is acquired in early life.
- In African and Asian patients, 95% of cases of HCC have anti-HBc.
- There is more direct evidence of the integration of the HBV-DNA genome in the genome of tumour cells of HCC.
2. Relation to chronic HCV infection: Long-standing HCV infection has emerged as a major factor in the aetiology of HCC, generally after 20-30 years of infection. The shreds of evidence in support are as under
- Developed countries where a higher incidence of HCC was earlier attributed to endemic HBV infection (for example in Japan), it has shown a remarkable shift to HCV infection.
- However, in countries of Africa and Asia, chronic HBV infection is still the predominant etiologic factor in the pathogenesis of HCC.
- The patients having anti-HCV and anti-HBc antibodies together have a three times higher risk of developing HCC than those with either antibody alone.
- Chronic HCV infection for a long duration more often produces cirrhosis prior to the development of HCC.
- On the other hand, in HCC developing following chronic HBV infection, half the cases are preceded by cirrhosis and the remainder have chronic HBV hepatitis.
- It is also possible that HBV and HCV infection may act synergistically to predispose to HCC.
3. Cirrhosis and precursor lesions: Cirrhosis of all etiologic types is more commonly associated with HCC but the most frequent association is with macronodular post-necrotic cirrhosis.
- The mechanism of progression to HCC appears to be chronic regenerative activity in cirrhosis, or that the damaged liver in cirrhosis is rendered vulnerable to carcinogenic influences.
- Dysplastic nodules or adenomatous hyperplasia of hepatocytes are precursor lesions of HCC which are identified by cellular enlargement, nuclear hyperchromatism and multinucleate cells, are found in 60% of cirrhotic livers with HCC and in only 10% of non-cirrhotic livers.
4. Relation to alcohol abuse: It has been observed that chronic alcoholism is a major risk factor, particularly seen in Western countries.
- There is about a four-fold increased risk of developing HCC in alcohol abusers. It is possible that alcohol may act as a co-carcinogen with HBV or HCV infection, but alcohol does not appear to be a hepatic carcinogen per se.
5. Mycotoxins: An important mycotoxin, aflatoxin B1, produced by a mould Aspergillus flavus, can contaminate poorly stored wheat grains or groundnuts, in hot and humid places.
- This is responsible for the higher incidence of HCC in some developing countries of Asia and Africa. Aflatoxin B1 is carcinogenic; it may act as a co-carcinogen with hepatitis B or may suppress the cellular immune response.
6. Chemical carcinogens: A number of chemical carcinogens can induce liver cancer in experimental animals. These include:
- Butter-yellow, safrole and nitrosamines are used as common food additives.
- Bush trees containing pyrrolizidine, and tannin acid.
- Pollutants such as pesticides and insecticides.
7. Miscellaneous factors Limited role of various other factors in HCC has been observed.
- These include the following:
- Haemochromatosis
- α-1-antitrypsin deficiency
- Prolonged immunosuppressive therapy in renal transplant patients
- Other types of viral hepatitis
- Non-alcoholic steatohepatitis (NASH)
- Tobacco smoking
- Parasitic infestations such as clonorchiasis and schistosomiasis
- Glycogen storage diseases.
The molecular pathogenesis of HCC can be explained on the basis of genetic mutations induced by one or more of the above major etiologic factors which damage the DNA of hepatocytes and result in neoplastic transformation.
These mutated genes are as under:
- Inactivation of tumour suppressor oncogene p53 by HBV.
- Binding of X-protein (HBxAg) generated from X-gene of HBV to p53.
- Mutations of oncogenes such as KRAS
- Mutations in receptors for hepatocyte growth factors example c-MYC, and c-MET.
- Activation of WNT and AKT pathways.
Morphologic Features Grossly, HCC may form one of the following 3 patterns of growth, in decreasing order of frequency:
- Expanding type Most frequently, it forms a single, yellow-brown, large mass, most often in the right lobe of the liver with central necrosis, haemorrhage and occasional bile-staining. It may be deceptively encapsulated.
- Multifocal type Less often, multifocal, multiple masses, 3-5 cm in diameter, scattered throughout the liver are seen.
- Infiltrating (Spreading) type Rarely, the HCC forms diffusely infiltrating tumour mass.
-
- Microscopically, the tumour cells in the typical HCC resemble hepatocytes but vary with the degree of differentiation, ranging from well-differentiated to highly anaplastic lesions.
- Most of the HCCs have trabecular growth patterns. The tumour cells have a tendency to invade and grow along blood vessels. Thus important diagnostic features are the patterns of tumour cells and their cytologic features:
1. Histologic patterns These include the following:
- The trabecular or sinusoidal pattern is the most common. The trabeculae are made up of 2-8 cell-wide layers of tumour cells separated by vascular spaces or sinusoids which are endothelium-lined.
- Pseudoglandular or acinar pattern is seen sometimes. The tumour cells are disposed around the central cystic space formed by degeneration and breakdown in solid trabeculae.
- The compact pattern resembles the trabecular pattern but the tumour cells form large solid masses with inconspicuous sinusoids.
- The scirrhous pattern is characterised by more abundant fibrous stroma.
2. Cytologic features The typical cytologic features in the HCC consist of cells resembling hepatocytes having vesicular nuclei with prominent nucleoli.
- The cytoplasm is granular and eosinophilic but becomes increasingly basophilic with increasing malignancy.
- Aside from these features, a few other cellular features are pleomorphism, bizarre giant cell formation, spindle-shaped cells, tumour cells with clear cytoplasm, the presence of bile within dilated canaliculi, and intracytoplasmic Mallory’s hyalin.
- Immunohistochemically, hepatocellular carcinoma cells stain positively with AFP, EMA, keratin etc.
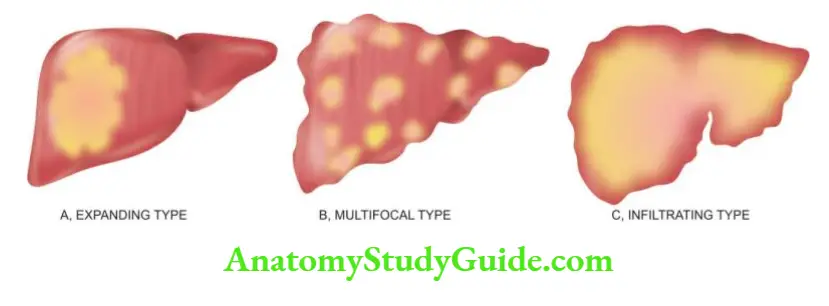
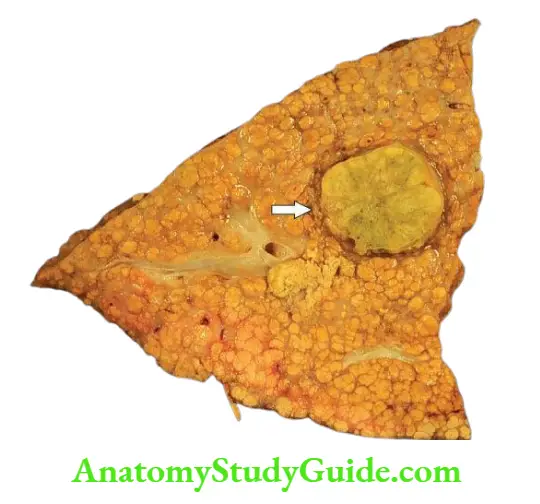
Fibrolamellar Carcinoma: A clinicopathologic variant of the HCC is fibrolamellar carcinoma of the liver found in young people of both sexes.
- The tumour forms a single large mass which may be encapsulated and occurs without background changes of cirrhosis or any other chronic liver disease.
Histologically, the tumour is composed of eosinophilic polygonal cells (oncocytes) forming cords and nests which are separated by parallel bands of fibrous stroma.
The prognosis of fibrolamellar carcinoma is better than other forms of HCC.
Clinical Features: Hepatic cancer may remain undetected initially because it often occurs in patients with underlying cirrhosis.
- The usual features consist of hepatomegaly with a palpable mass in the liver, right upper quadrant pain or tenderness, and less often, jaundice, fever and haemorrhage from oesophageal varices.
- Ascites with RBCs and malignant cells are found in about half the patients. Rarely, systemic endocrine manifestations due to paraneoplastic syndrome are observed such as hypercalcaemia, hypoglycaemia, gynaecomastia and acquired porphyria.
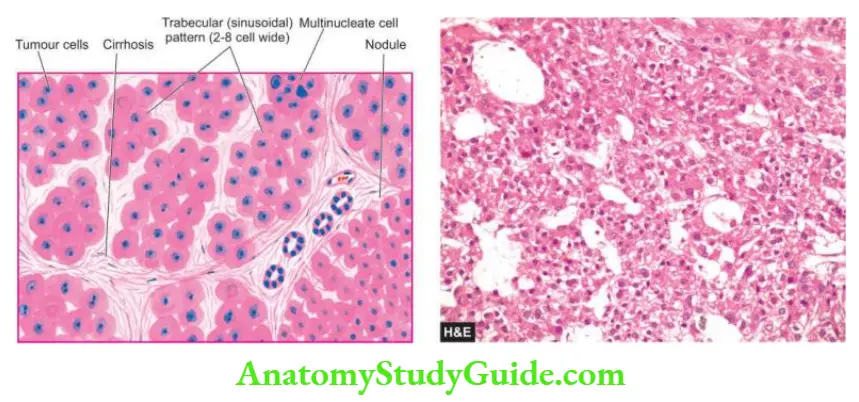
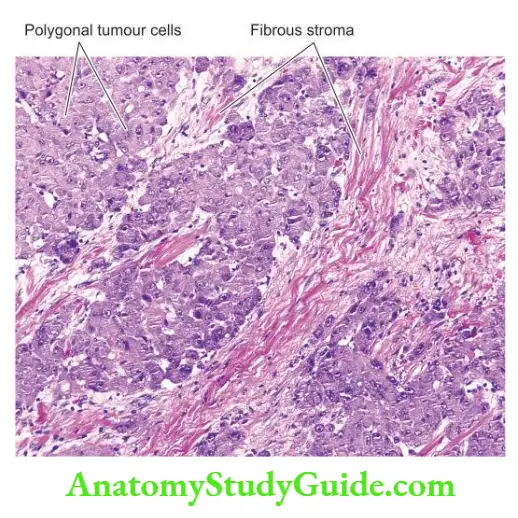
- Laboratory findings yield nonspecific results like anaemia, and markedly elevated serum alkaline phosphatase as found in cirrhosis.
- But high serum alpha-foetoprotein (AFP) level is quite specific; very high levels of AFP (above 500 ng/ml) are observed in 70-80% of cases of HCC.
- However, it lacks sensitivity since AFP is also found elevated in yolk sac tumours, cirrhosis, chronic hepatitis, massive liver necrosis and normal pregnancy.
- Ultrasound of the liver has been reported to be more sensitive than elevated AFP levels. An abnormal type of prothrombin, des-γ- carboxy prothrombin, is also elevated and correlates well with AFP levels.
Spread The HCC can have both intrahepatic and extrahepatic spread which faithfully reproduces the structure of the primary tumour:
- Intrahepatic spread occurs by a haematogenous route and forms multiple metastases in the liver.
- Extrahepatic spread occurs via hepatic or portal veins to different sites, chiefly to lungs and bones, and by lymphatic route to regional lymph nodes at the porta hepatis and to mediastinal and cervical lymph nodes.
- The causes of death from HCC are cachexia, massive bleeding from oesophageal varices, and liver failure with hepatic coma. Usually, survival after diagnosis of HCC is less than 2 years.
Cholangiocarcinoma
- Cholangiocarcinoma is the designation used for carcinoma arising from bile duct epithelium within the liver (intrahepatic or peripheral cholangiocarcinoma).
- Carcinomas arising from the large hilar ducts (hilar cholangiocarcinoma) and from extrahepatic ducts are termed bile duct carcinomas.
- None of the etiologic factors related to HCC have any role in the genesis of cholangiocarcinoma.
- However, the etiological factors involved in it are exposure to radioopaque dye thorotrast, anabolic steroids, clonorchiasis and fibrocystic disease.
- The tumour affects older people and the clinical features are those of HCC but with prominence of jaundice.
Morphologic Features Grossly, the tumour is firm to hard and whitish.
- Microscopically, the tumour has a glandular structure. The tumour cells resemble biliary epithelium but without bile secretion.
- They form various patterns such as tubular, ductular or papillary. The stroma consists of fibrous tissue with little or no capillary formation.
- Occasionally, mucinous, signet-ring and adenosquamous types patterns are found. An uncommon variant is combined hepatocellular-cholangiocarcinoma.
Hepatoblastoma (Embryoma)
- Hepatoblastoma is a rare malignant tumour arising from primitive hepatic parenchymal cells. It presents before the age of 2 years as progressive abdominal distension with anorexia, failure to thrive, fever and jaundice.
- It is more common in boys. The concentration of serum AFP is high. The tumour grows rapidly and causes death by haemorrhage, hepatic failure or widespread metastases.
Morphologic Features Grossly, the tumour is circumscribed and lobulated mass measuring 5-25 cm in size, having areas of cystic degeneration, haemorrhage and necrosis. Microscopically, hepatoblastoma consists of two components.
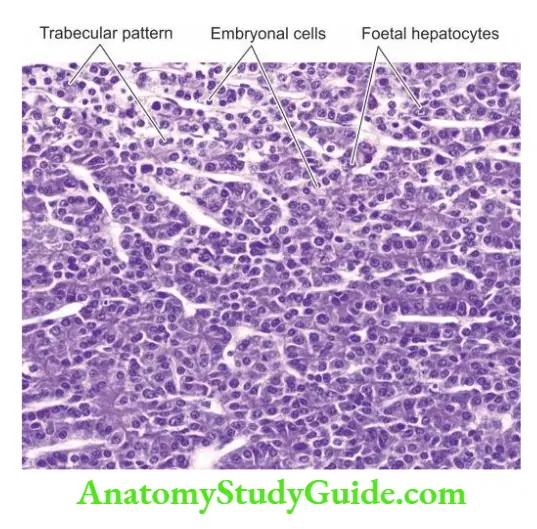
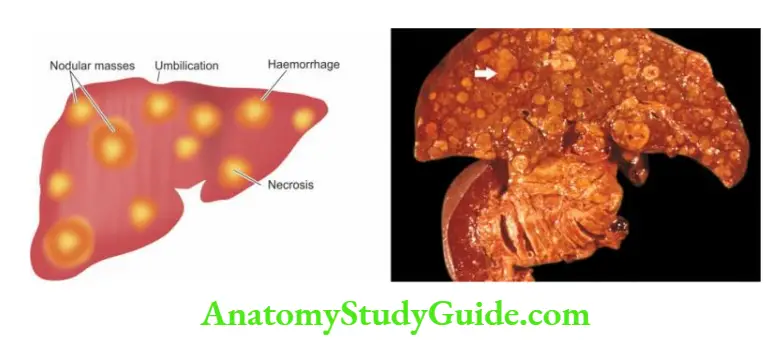
1. Epithelial component: contains two types of immature liver cells embryonal hepatocytes are small with dark-staining, hyperchromatic nuclei and scanty cytoplasm, while foetal hepatocytes are larger with more cytoplasm that may be granular or clear.
- The epithelial cells are organised in trabeculae, ribbons or rosettes.
2. Mesenchymal component: includes fibrous connective tissue, cartilage and osteoid of variable degree of maturation. Extramedullary haematopoiesis is a frequent accompaniment.
Secondary Hepatic Tumours
- Metastatic tumours in the liver are more common than the primary hepatic tumours. Most frequently, they are blood-borne metastases, irrespective of whether the primary tumour is drained by portal vein or systemic veins.
- The most frequent primary tumours metastasising to the liver, in descending order of frequency, are those of the stomach, breast, lungs, colon, oesophagus, pancreas, malignant melanoma and haematopoietic malignancies.
- Sarcomas rarely metastasise to the liver. Occasionally, metastatic involvement may be present in the absence of obvious evidence of a primary tumour.
- Aside from general features of disseminated malignancy such as anorexia, cachexia and anaemia, the patients have hepatomegaly with nodular free margin.
- There is little hepatic dysfunction until late in the course of hepatic metastatic disease.
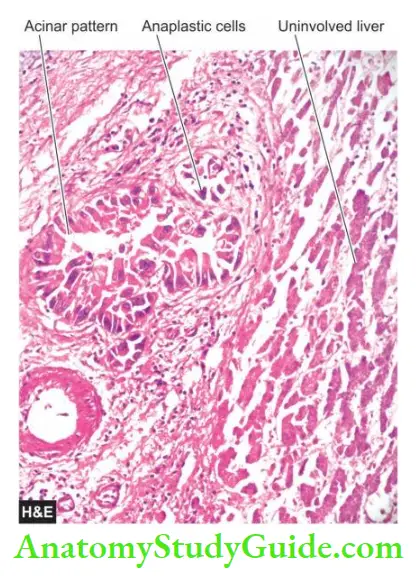
Morphologic Features: Grossly, most metastatic carcinomas form multiple, spherical, nodular masses which are of variable size. The liver is enlarged and heavy, weighing 5 kg or more.
- The tumour deposits are white, well-demarcated, soft or haemorrhagic. The surface of the liver shows characteristic umbilication due to central necrosis of nodular masses.
- Histologically, metastatic tumours generally reproduce the structure of the primary lesions.
Hepatic Tumours and Tumour-like Lesions
- Tumour-like lesions are solitary or multiple cysts in the liver example congenital, simple and parasitic.
- Benign hepatic tumours are uncommon and include focal nodular hyperplasia, hepatocellular (liver cell) adenoma, bile duct adenoma (cholangioma) and haemangioma.
- Hepatocellular carcinoma or hepatoma is the most common primary malignant tumour of the liver. The tumour supervenes cirrhosis in 70-80% of cases, most often on postnecrotic and alcoholic cirrhosis.
- In general, HCC has a poor prognosis but a variant form, fibrolamellar carcinoma, that occurs in younger age groups has a better prognosis.
- Hepatoblastoma is a rare malignant tumour presenting before the age of 2 years.
- Metastatic tumours in the liver are more common than the primary hepatic tumours. Most often, they are blood-borne metastases from various primary sites.
Biliary Tract
Normal Structure
- Anatomy The Gallbladder is a pear-shaped organ, 9 cm in length and has a capacity of approximately 50 ml. It consists of the fundus, body and neck that tapers into the cystic duct.
- The two hepatic ducts from the right and left lobes of the liver unite at the porta hepatis to form the common hepatic duct which is joined by the cystic duct from the gallbladder to form the common bile duct.
- The common bile duct enters the second part of the duodenum posteriorly. In about 70% of cases, it is joined by the main pancreatic duct to form the combined opening in the duodenum (ampulla of Vater).
- In 30% of cases, the common bile duct and the pancreatic duct open separately into the duodenumed from the duodenum forming the sphincter of Oddi.
Histology Histologically, the gallbladder, unlike the rest of the gastrointestinal tract, lacks the muscularis mucosae and submucosa. The wall of the gallbladder is composed of the following 4 layers.
1. Mucosal layer: It has a single layer of tall columnar epithelium which is thrown into permanent folds that are larger and more numerous in the neck of the gallbladder.
- Beneath the epithelium is delicate lamina propria that contains capillaries, and in the region of the neck, a few acinar glands are present.
2. Smooth muscle layer: External to the lamina propria are smooth muscle bundles in layers— inner longitudinal, middle oblique, and outer circular.
3. Perimuscular layer: Outer to the muscle layer is a zone of fibrous connective tissue with some interspersed fat cells.
4. Serosal layer: The perimuscular layer is covered by serosa on the peritoneal surface of the gallbladder. The peritoneum covers the gallbladder except in the region of the gallbladder fossa where it is embedded in the liver.
- The extrahepatic bile ducts are also lined by tall columnar epithelium that overlies the lamina propria. It is surrounded by a dense layer of fibromuscular tissue.
- The ducts which lie between the lobules of the liver and receive bile from the canaliculi are lined by cuboidal or flattened cells.
Functions The main function of the gallbladder is to store and concentrate the bile secreted by the liver and then deliver it into the intestine for digestion and absorption of fat.
- The concentrating ability of the gallbladder is due to its absorptive mucosal surface that has numerous folds. Normally, the liver secretes approximately 500 ml of bile per day and the gallbladder concentrates it 5-10 times.
- The motility, concentration and relaxation of the gallbladder are under the influence of a peptide hormone, cholecystokinin, released from neuroendocrine cells of the duodenum and jejunum.
- Three groups of diseases of the biliary tract are discussed below: gallstones, cholecystitis, and
tumours.
Cholecystitis
- Cholecystitis or inflammation of the gallbladder may be acute, chronic, or acute superimposed on chronic. Though chronic cholecystitis is more common, acute cholecystitis is a surgical emergency.
Acute Cholecystitis
- In many ways, acute cholecystitis is similar to acute appendicitis. The condition usually begins with obstruction, followed by infection later.
Etiopathogenesis Based on the initiating mechanisms, acute cholecystitis occurs in two types of situations—acute calculous and acute acalculous cholecystitis.
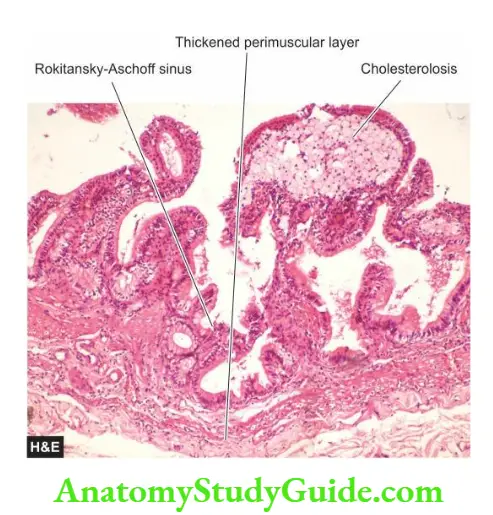
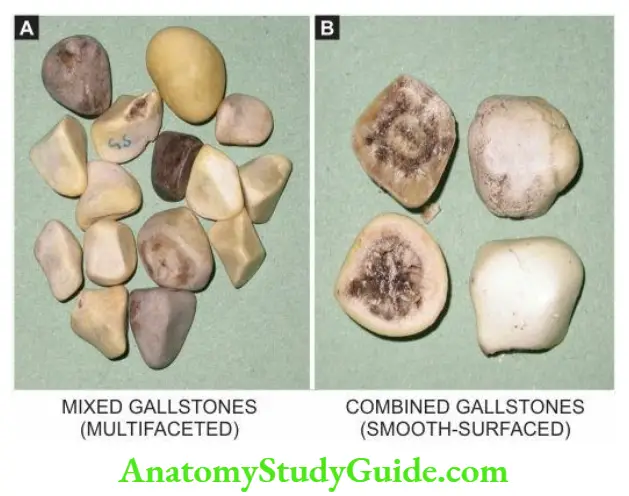
- Acute calculous cholecystitis In 90% of cases, acute cholecystitis is caused by an obstruction in the neck of the gallbladder or in the cystic duct by a gallstone.
- The commonest location of impaction of a gallstone is in Hartmann’s pouch. The obstruction results in distension of the gallbladder followed by acute inflammation which is initially due to chemical irritation.
- Later, however, secondary bacterial infection, chiefly by E. coli and Streptococcus faecalis, supervenes.
- Acute acalculous cholecystitis The remaining 10% of cases of acute cholecystitis do not contain gallstones.
- In such cases, a variety of causes have been assigned such as previous nonbiliary surgery, multiple injuries, burns, recent childbirth, severe sepsis, dehydration, torsion of the gallbladder and diabetes mellitus.
- Rare causes include primary bacterial infections like salmonellosis and cholera and parasitic infestations.
Morphologic Features: Except for the presence or absence of calculi, the two forms of acute cholecystitis are morphologically similar. Grossly, the gallbladder is distended and tense.
- The serosal surface is coated with fibrinous exudate with congestion and haemorrhages. The mucosa is bright red. The lumen is filled with pus mixed with green bile.
- In calculous cholecystitis, a stone may get impacted in the neck or in the cystic duct.
- When obstruction of the cystic duct is complete, the lumen is filled with purulent exudate and the condition is known as empyema of the gallbladder.
Microscopically, the wall of the gallbladder shows marked inflammatory oedema, congestion and neutrophilic exudate.
- There may be frank abscesses in the wall and gangrenous necrosis with rupture into the peritoneal cavity (gangrenous cholecystitis).
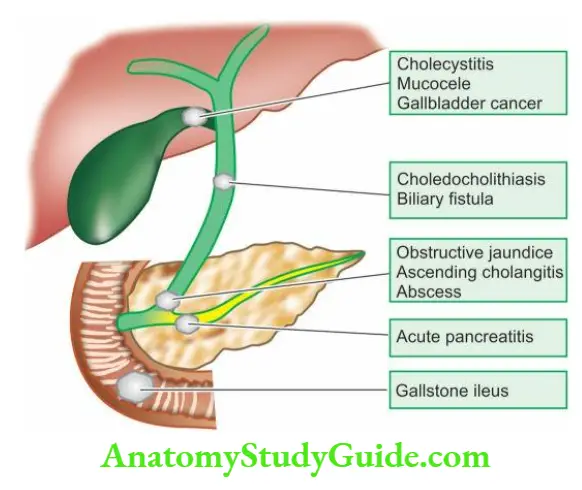
Clinical Features: The patients of acute cholecystitis of either type have similar clinical features. They present with severe pain in the upper abdomen with features of peritoneal irritation such as guarding and hyperaesthesia.
- The gallbladder is tender and may be palpable. Fever, leucocytosis with neutrophilia and slight jaundice are generally present.
- Early cholecystectomy within the first three days has a mortality of less than 0.5% and the risk of complications such as perforation, biliary fistula, recurrent attacks and adhesions is avoided.
- However, medical treatment brings about resolution in a fairly large proportion of cases though chances of recurrence of attack persist.
Chronic Cholecystitis
- Chronic cholecystitis is the commonest type of clinical gallbladder disease. There is an almost constant association of chronic cholecystitis with cholelithiasis.
Etiopathogenesis The association of chronic cholecystitis with mixed and combined gallstones is virtually always present. However, it is not known what initiates the inflammatory response in the gallbladder wall.
- Possibly, supersaturation of the bile with cholesterol predisposes to both gallstone formation and inflammation. In some patients, repeated attacks of mild acute cholecystitis result in chronic cholecystitis.
Morphologic Features Grossly, the gallbladder is generally contracted but may be normal or enlarged.
- The wall of the gallbladder is thickened which on the cut section is grey-white due to dense fibrosis or may be even calcified.
- The mucosal folds may be intact, thickened, or flattened and atrophied. The lumen commonly contains multiple mixed stones or a combined stone.
Histologically, the features are as under:
- Thickened and congested mucosa but occasionally mucosa may be totally destroyed.
- Penetration of the mucosa deep into the wall of the gallbladder up to the muscular layer to form Rokitansky-Aschoff’sinuses.
- Variable degree of chronic inflammatory reaction, consisting of lymphocytes, plasma cells and macrophages, present in the lamina propria and subserosal layer.
- Variable degree of fibrosis in the subserosal and subepithelial layers.
A few morphologic variants of chronic cholecystitis are considered below:
- Cholecystitis glandular, when the mucosal folds fuse together due to inflammation and result in the formation of crypts of epithelium buried in the gallbladder wall.
- Porcelain gallbladder is the pattern when the gallbladder wall is calcified and cracks like an eggshell.
- Acute on chronic cholecystitis is the term used for the morphologic changes of acute cholecystitis superimposed on changes of chronic cholecystitis.
Clinical Features Chronic cholecystitis has ill-defined and vague symptoms. Generally, the patient—a fat, fertile, female of forty or fifty, presents with abdominal distension or epigastric discomfort, especially after a fatty meal.
- There is a constant dull ache in the right hypochondrium and epigastrium and tenderness over the right upper abdomen.
- Nausea and flatulence are common. Biliary colic may occasionally occur due to the passage of stone into the bile ducts. Cholecystography usually allows radiologic visualisation of the gallstones.
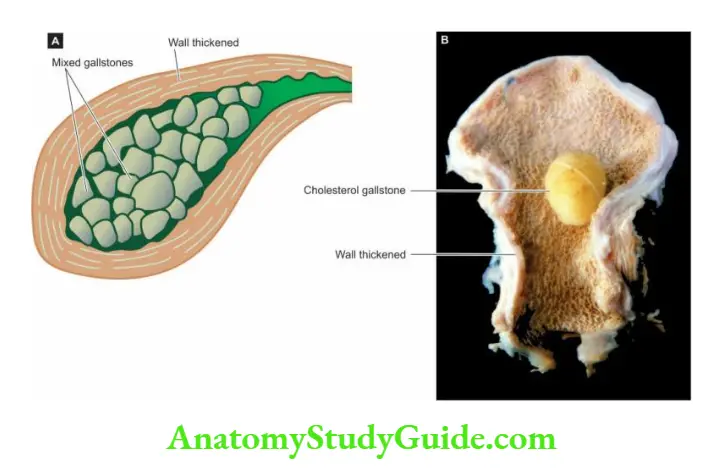
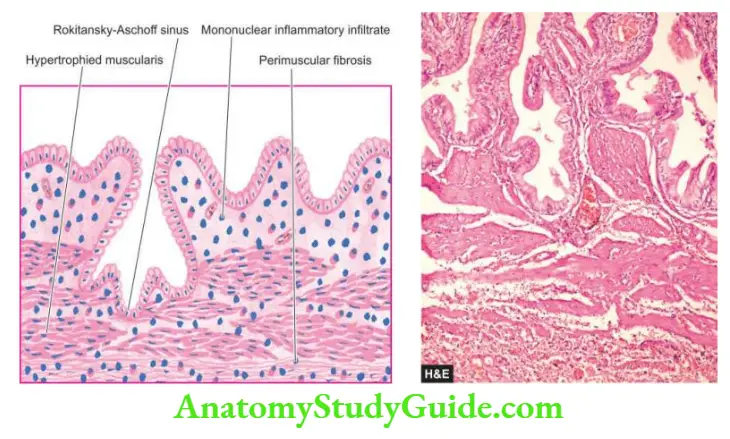
Cholecystitis
- Acute cholecystitis is clinically similar to acute appendicitis. It usually begins with obstruction, followed by infection later.
- Chronic cholecystitis is invariably associated with mixed and combined gallstones.
- Generally, the patients of chronic cholecystitis are fat, fertile, female of forty or fifty, presenting with abdominal distension or epigastric discomfort.
Developmental Anomalies
- The significant developmental anomalies of the pancreas are ectopic or aberrant pancreatic tissue in Meckel’s diverticulum, anomalies of the ducts, and cystic fibrosis. Only the last name is discussed here.
Cystic Fibrosis
- Cystic fibrosis of the pancreas or fibrocystic disease is a hereditary disorder of autosomal recessive inheritance characterised by viscid mucous secretions in all the exocrine glands of the body (mucoviscidosis) and is associated with increased concentrations of electrolytes in the eccrine glands.
- It is quite common in whites (1 per 2000 live births) The defect is a genetic mutation in the CFTR gene, an acronym for coding protein for cystic fibrosis transmembrane conductance regulator located on the long arm of chromosome 7 (7q).
- CFTR is a cAMP-regulated anion channel expressed on the apical surface of epithelial cells that line the airways, pancreatic ducts, and other tissues.
- When it is absent (for example in F508 mutation) or its activity is reduced, chloride and bicarbonate transport are both reduced.
- The clinical manifestations may appear at birth or later in adolescence and pertain to multiple organs and
systems such as pancreatic insufficiency, intestinal obstruction, steatorrhoea, malnutrition, hepatic cirrhosis and respiratory complications. - The terms ‘cystic fibrosis’ and ‘fibrocystic disease’ are preferable over ‘mucoviscidosis’ in view of the main pathologic change of fibrosis produced as a result of obstruction of the passages by viscid mucous secretions.
Morphologic Features Depending upon the severity of involvement and the organs affected, the pathologic changes are variable. Most of the changes are produced as a result of obstruction by viscid mucous.
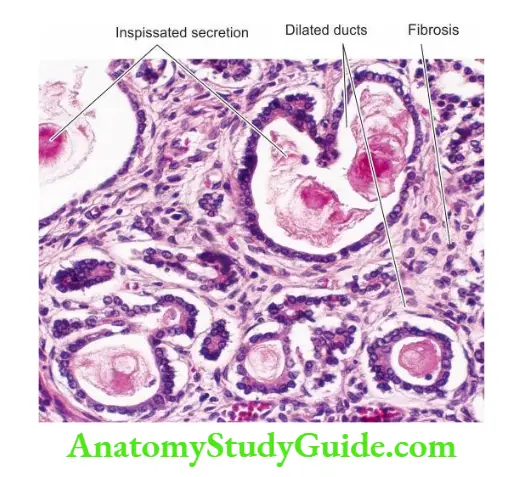
1. Pancreas: The pancreas is almost invariably involved in cystic fibrosis. Grossly, pancreatic lobules are ovoid rather than rhomboid. Fatty replacement of the pancreas and grossly visible cysts may be seen.
Microscopically, the lobular architecture of pancreatic parenchyma is maintained. There is increased interlobular fibrosis.
- The acini are atrophic and many of the acinar ducts contain inspissated secretions or laminated, eosinophilic concretions.
- Rarely, inflammation, fat necrosis and cyst formation may be seen. The islet tissue (endocrine pancreas) generally remains intact.
- Atrophy of the exocrine pancreas may cause impaired fat absorption, steatorrhoea, intestinal obstruction, avitaminosis A and cystic fibrosis-related diabetes mellitus.
2. Liver: The bile canaliculi are plugged by viscid mucous which may cause diffuse fatty change, portal fibrosis and ductular proliferation. More severe involvement may cause biliary cirrhosis.
3. Respiratory tract Changes in the respiratory passages are seen in almost all typical cases of cystic fibrosis.
- The viscid mucous secretions of the submucosal glands of the respiratory tract cause obstruction, dilatation and infection of the airways.
- The changes include chronic bronchitis, bronchiectasis, bronchiolitis, bronchiectasis, peribronchiolar pneumonia and inflammatory nasal polyps.
- These superimposed microbial infections include Haemophilus influenza, Staphylococcus aureus, Pseudomonas aeruginosa, Aspergillus etc.
4. Salivary glands Pathologic changes in the salivary glands are similar to those in the pancreas and include obstruction of the ducts, dilatation, fibrosis and glandular atrophy.
5. Sweat glands Hypersecretion of sodium and chloride in the sweat observed in these patients may be reflected pathologically by diminished vacuolation of the cells of eccrine glands.
- A sweat chloride level of more than 70 mEq/ml is diagnostic of cystic fibrosis.
Cystic Fibrosis of the Pancreas
- Cystic fibrosis of the pancreas is a hereditary condition having viscid mucous secretions in all the exocrine glands of the body (mucoviscidosis).
- The disease has an autosomal recessive inheritance due to a genetic mutation in the CFTR gene.
- Clinical features are apparent in homozygotes only due to obstruction by viscid secretions. These include features pertaining to affected organs such as the pancreas (cystic fibrosis), lungs (infections), liver (biliary cirrhosis), salivary glands (fibrosis and atrophy) and sweat glands (atrophy).
Pancreatitis
- Pancreatitis is an inflammation of the pancreas with acinic cell injury. It is classified into acute and chronic forms both of which are distinct entities.
Acute Pancreatitis
- Acute pancreatitis is an acute inflammation of the pancreas presenting clinically with an ‘acute abdomen’.
- The severe form of the disease associated with macroscopic haemorrhages and fat necrosis in and around the pancreas is termed acute haemorrhagic pancreatitis or acute pancreatic necrosis.
- The condition occurs in adults between the age of 40 and 70 years and is commoner in females than in males.
- The onset of acute pancreatitis is sudden, occurring after a bout of alcohol or a heavy meal.
- The patient presents with abdominal pain, vomiting and collapse and the condition must be differentiated from other diseases producing acute abdomens such as acute appendicitis, perforated peptic ulcer, acute cholecystitis, and infarction of the intestine following sudden occlusion of the mesenteric vessels.
- Characteristically, there is the elevation of serum amylase level within the first 24 hours and elevated serum lipase level after 3 to 4 days, the latter being more specific for pancreatic disease. Glucosuria occurs in 10% of cases.
Etiology Two leading causes associated with acute pancreatitis are alcoholism and cholelithiasis, both of which are implicated in more than 80% of cases.
- Less common causes of acute pancreatitis include trauma, ischaemia, shock, extension of inflammation from the adjacent tissues, blood-borne bacterial infection, viral infections, certain drugs (for example thiazides, sulfonamides, oral contraceptives), hypothermia, hyperlipoproteinaemia and hypercalcaemia from hyperparathyroidism.
- Rarely, familial pancreatitis is encountered. In a proportion of cases of acute pancreatitis, the aetiology remains unknown (idiopathic pancreatitis).
Pathogenesis Destructive changes in the pancreas are attributed to the liberation and activation of pancreatic enzymes.
- Though more than 20 enzymes are secreted by the exocrine pancreas, 3 main groups of enzymes which bring about destructive effects on the pancreas are as under
- Proteases such as trypsin and chymotrypsin play the most important role in causing proteolysis.
- Trypsin also activates the kinin system by converting prekallikrein to kallikrein, and thereby the clotting and complement systems are activated.
- This results in inflammation, thrombosis, tissue damage and haemorrhages found in acute haemorrhagic pancreatitis.
- Lipases and phospholipases degrade lipids and membrane phospholipids.
- Elastases cause the destruction of the elastic tissue of the blood vessels. The activation and release of these enzymes are brought about by one of the following mechanisms
- Acinic cell damage caused by etiologic factors such as alcohol, viruses, drugs, ischaemia and trauma result in the release of intracellular enzymes.
- Duct obstruction caused by cholelithiasis, chronic alcoholism and other obstructing lesions is followed by leakage of pancreatic enzymes from the ductules into the interstitial tissue.
- Block in exocytosis of pancreatic enzymes occurring from nutritional causes results in the activation of these intracellular enzymes by pancreatic lysosomal hydrolases.
Morphologic Features Grossly, in the early stage, the pancreas is swollen and oedematous.
- Subsequently, in a day or two, the characteristic variegated appearance of grey-white pancreatic necrosis, chalky-white fat necrosis and blue-black haemorrhages are seen.
- In the typical case, the peritoneal cavity contains blood-stained ascitic fluid and white flecks of fat necrosis in the omentum, mesentery and peripancreatic tissue. The resolved lesions show areas of fibrosis, calcification and ductal dilatation.
Microscopically, the following features in varying grades are noticeable:
- Necrosis of pancreatic lobules and ducts.
- Necrosis of the arteries and arterioles with areas of haemorrhages.
- Fat necrosis.
- Inflammatory reaction, chiefly by polymorphs, around the areas of necrosis and haemorrhages.
Complications A patient with acute pancreatitis who survives may develop a variety of systemic and local complications
Systemic complications
- Chemical and bacterial peritonitis
- Endotoxic shock
- Acute renal failure
Local sequelae
- Pancreatic abscess
- Pancreatic pseudocyst
- Duodenal obstruction
- Mortality in acute pancreatitis is high (20-30%). Patients succumb to hypotensive shock, infection, acute renal failure, and DIC.
Chronic Pancreatitis
- Chronic pancreatitis or chronic relapsing pancreatitis is the progressive destruction of the pancreas due to repeated mild and subclinical attacks of acute pancreatitis.
- Most patients present with recurrent attacks of severe abdominal pain at intervals of months to years. Weight loss and jaundice are often associated.
- Later manifestations include associated diabetes mellitus and steatorrhoea. Abdominal radiographs show calcification in the region of the pancreas and the presence of pancreatic calculi in the ducts.
Etiology Most cases of chronic pancreatitis are caused by the same factors as acute pancreatitis.
- Thus, most commonly, chronic pancreatitis is related to chronic alcoholism with a protein-rich diet, and less often to biliary tract disease.
- Familial hereditary pancreatitis, though uncommon, is more frequently chronic than the acute form.
- Other rare causes of chronic pancreatitis are hypercalcaemia, hyperlipidaemia and developmental failure of fusion of dorsal and ventral pancreatic ducts.
Pathogenesis Acute haemorrhagic pancreatitis seldom develops into chronic pancreatitis, but instead develops pancreatic pseudocysts following recovery.
- Pathogenesis of alcoholic and non-alcoholic chronic pancreatitis is explained by different mechanisms
- Chronic pancreatitis due to chronic alcoholism accompanied by a high-protein diet results in an increase in protein concentration in the pancreatic juice which obstructs the ducts and causes damage.
- Non-alcoholic cases of chronic pancreatitis seen in tropical countries (tropical chronic pancreatitis) result from protein-calorie malnutrition. Genetic factors play a role in some cases of chronic pancreatitis.
Morphologic Features Grossly, the pancreas is enlarged, firm and nodular. The cut surface shows a smooth grey appearance with a loss of normal lobulation.
- Foci of calcification and tiny pancreatic concretions to larger visible stones are frequently found. Pseudocysts may be present.
Microscopically, depending upon the stage of development, the following changes are seen:
- Obstruction of the ducts by fibrosis in the wall and protein plugs or stones in the lumina.
- Squamous metaplasia and dilatation of some inter- and intralobular ducts.
- Chronic inflammatory infiltrate around the lobules as well as the ducts.
- Atrophy of the acinar tissue with a marked increase in interlobular fibrous tissue.
- Islet tissue is involved in the late stage only.
Complications Late-stage of chronic pancreatitis may be complicated by diabetes mellitus, pancreatic insufficiency with steatorrhoea and malabsorption and formation of pancreatic pseudocysts.
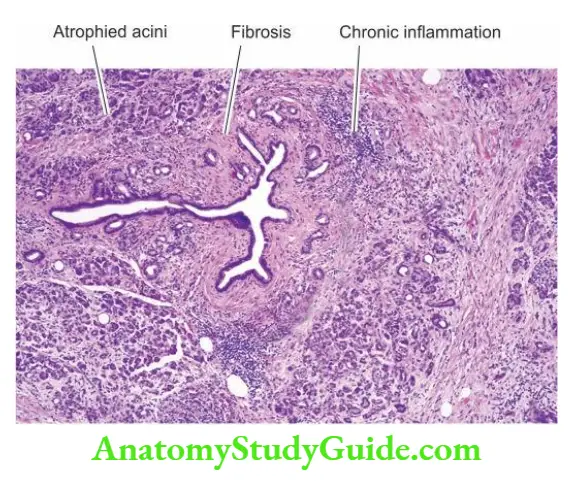
Pancreatitis
- Acute pancreatitis presents clinically with ‘acute abdomen’.
- The two leading causes associated with acute pancreatitis are alcoholism and cholelithiasis.
- Chronic pancreatitis is the progressive destruction of the pancreas due to repeated mild and subclinical attacks of acute pancreatitis.
- Chronic pancreatitis may be complicated by diabetes mellitus, steatorrhoea and pancreatic pseudocysts.
Tumours And Tumour-Like Lesions
- Tumour-like masses of the exocrine pancreas include congenital cystic disease (involving the pancreas, liver and kidney) and pancreatic pseudocysts.
- True pancreatic tumours are classified into benign (for example serous cystadenoma, fibroma, lipoma and adenoma) and malignant (carcinoma of the pancreas).
- Out of all these, only two pancreatic lesions—pseudocyst and carcinoma of the pancreas, are common and discussed here.
Pancreatic Pseudocyst
- The pancreatic pseudocyst is a localised collection of pancreatic juice, necrotic debris and haemorrhage. It develops following either acute pancreatitis or trauma.
- The patients generally present with abdominal mass that produces pain, intraperitoneal haemorrhage and generalised peritonitis.
Morphologic FeaturesGrossly, the pseudocyst may be present within or adjacent to the pancreas. Usually, it is solitary, unilocular, measuring up to 10 cm in diameter with thin or thick walls.
- Microscopically, the cyst wall is composed of dense fibrous tissue with marked inflammatory reactions.
- There is evidence of preceding haemorrhage and necrosis in the form of deposits of haemosiderin pigment, calcium and cholesterol crystals.
- The lumen of the cyst contains serous or turbid fluid. The cyst does not show any epithelial lining.
Carcinoma Of Pancreas
- Pancreatic cancer is the term used for cancer of the exocrine pancreas. Besides pancreatic cancer, a well-differentiated neuroendocrine tumour called panNET (earlier called islet cell tumour) is also described in the pancreas.
- It is one of the most common cancers, particularly in Western countries and Japan. In the United States, cancer of the pancreas is the second most common cancer of the alimentary tract after colorectal cancer, is more common in African Americans and accounts for 5% of all cancer deaths in that country.
- It is commoner in males than in females and the incidence increases progressively after the age of 50 years.
- Etiology A significant increase in the incidence of pancreatic cancer has been observed in the UK and the US during the last 50 years.
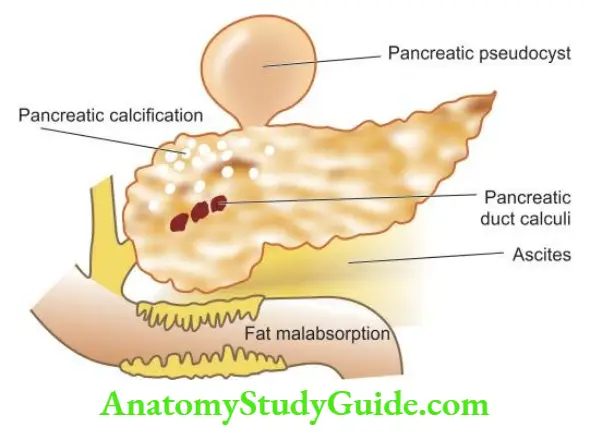
Little is known about the aetiology of pancreatic cancer. However, the following factors have been implicated in its aetiology:
- Smoking: Heavy cigarette smokers have a higher incidence than non-smokers. However, it is not known whether tobacco metabolites have a direct carcinogenic effect on the pancreas or by some other unknown mechanism.
- Diet and obesity: A diet with high total caloric value and high consumption of animal proteins and fats is related to a higher incidence of pancreatic cancer. Obesity is a risk factor for pancreatic cancer.
- Chemical carcinogens: Individuals exposed to β-naphthylamine, benzidine and nitrosamines have a higher incidence of cancer of the pancreas.
- Diabetes mellitus: Patients with long-standing diabetes mellitus have a higher incidence.
- Chronic pancreatitis patients are at increased risk.
- H. pylori infection has been reported to have an association with pancreatic cancer.
- Genetic factors have been found to have an association with pancreatic cancer example its occurrence in first-degree relatives in 10% of cases, and occurrence in certain hereditary syndromes (Lynch, FPC, HNPCC).
- However, excessive consumption of alcohol or coffee, and cholelithiasis are not risk factors for pancreatic cancer.
Pathogenesis: Pancreatic cancer evolves through precursor lesions of pancreatic intraepithelial neoplasia (PanIN).
- At the genetic level, a combination of mutations in the K-RAS gene, Cdkn2a, TP53 and SMAD4 genes have been found in almost all cases, both precursor and invasive cancer of the pancreas
- The earliest alterations found in low-grade pancreatic intraepithelial neoplasia (PanIN) lesions are the shortening of telomeres and activating point mutations in K-RAS.
- Subsequent to mutated KRAS, mutation in the tumour suppressor gene CDKN2A occurs. The latter mutations are more common in high-grade than in low-grade lesions such as PanIN.
- Advanced cases of PanINs and invasive pancreatic carcinoma develop mutations in the tumour suppressor genes TP53 and SMAD4.
Morphologic Features The most common location of pancreatic cancer is the head of the pancreas (70%), followed in decreasing frequency, by the body and the tail of the pancreas.
- Grossly, carcinoma of the head of the pancreas is generally small, homogenous, poorly defined, grey-white mass without any sharp demarcation between the tumour and the surrounding pancreatic parenchyma.
- The tumour of the head extends into the ampulla of Vater, common bile duct and duodenum, producing obstructive biliary symptoms and jaundice early in the course of the illness.
- Carcinomas of the body and tail of the pancreas, on the other hand, are fairly large and irregular masses and frequently infiltrate the transverse colon, stomach, liver, spleen and regional lymph nodes.
Microscopically, most pancreatic carcinomas arise from the ductal epithelium which normally comprises less than 4% of total pancreatic cells, whereas carcinoma of the acini constitutes less than 1% of pancreatic cancers.
- The following histologic patterns of pancreatic carcinoma are seen:
- Pancreatic intraepithelial neoplasia (PanIN) are lesions surrounding ductal adenocarcinoma and are classified into low grade and high grade.
- These are in situ carcinomas that may be intraductal papillary mucinous neoplasm (IPMN), intraductal tubulopapillary neoplasm (ITPN), mucinous cystic neoplasm (MCN), and intra-ampullary papillary-tubular neoplasm.
- High-grade PanIN is often seen in the vicinity of ductal carcinomas. These may remain confined as in situ, but as they advance they may be associated with invasive components.
- Ductal adenocarcinoma is the most common malignant tumour of the pancreas.
- It is generally a well-differentiated carcinoma having glandular architecture, lined by mucin-secreting malignant columnar cells. Perineural invasion is frequent and has prognostic significance.
- Several other variants have been described but they are less common. A few examples are colloid carcinoma (or mucinous noncystic carcinoma), signet-ring cell carcinoma, acinar carcinoma and adenosquamous carcinoma.
Clinical Features Clinical symptoms depend upon the site of origin of the tumour. Generally, the following features are present:
- Obstructive (cholestatic) jaundice More often and early in the course of disease in cases with carcinoma head of the pancreas (80%), and less often in cancer of the body and tail of the pancreas.
- It is characterised by: dark urine, clay-like stools, pruritus, and very high serum alkaline phosphatase.
- Other features include abdominal pain, anorexia, weight loss, cachexia, weakness and malaise, nausea and vomiting, migratory thrombophlebitis (Trousseau’s syndrome), GI bleeding and splenomegaly.
- Tumour markers CA19.9 and CEA can be performed on blood as well as fluid or cytology specimens. Their levels are found to be elevated in pancreatic cancer.
- The prognosis of pancreatic cancer is dismal median survival is 6 months from the time of diagnosis. Approximately 10% of patients survive 1-year and the 5-year survival is poor 1 to 2%.
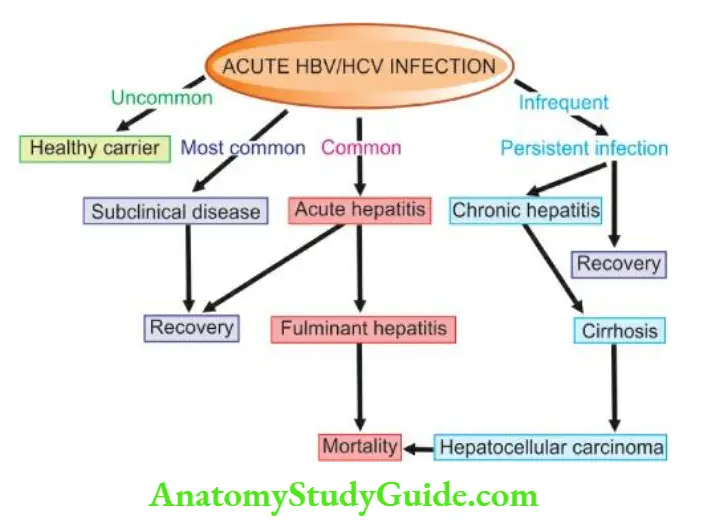
Leave a Reply