Fluorides Introduction
Fluorine is an extremely reactive and the most electronegative element. It combines with almost every element/organic radical and is very rarely found in a free form. McKay and Churchill’s study identified fluoride as the causative factor of mottled enamel.
Table of Contents
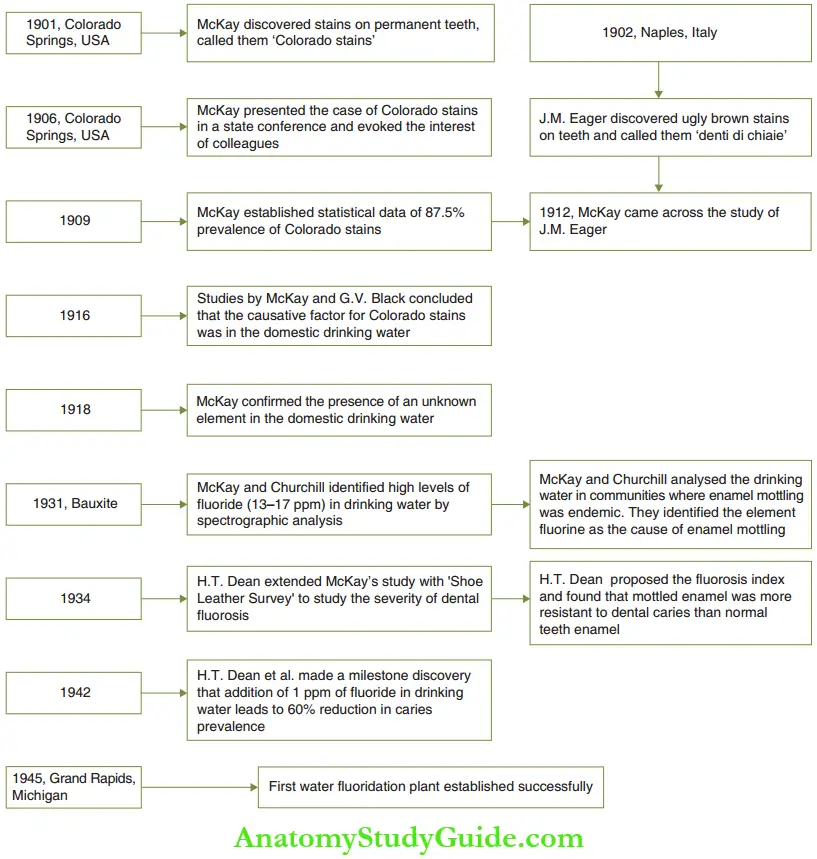
The anti-cariogenic effectiveness of fluoride was first identified by H. T. Dean after many years of research on McKay and Churchill’s study of mottled enamel. Further, H. T. Dean discovered that 1 ppm of fluoride in drinking water can decrease the prevalence of dental caries by 60%. A brief history of the initiation of water fluoridation is illustrated.
Read And Learn More: Paediatric Dentistry Notes
Cariostatic Mechanisms
The cariostatic mechanism of fluoride operates in two phases. They are the pre-eruptive and post-eruptive phases. Fluoride ingested during the development of the tooth or in the pre-eruptive phase has been reported to have an influence on the alteration of crown morphology.
The pits and fissures are reported to be shallower and cuspal inclines are less steep with optimal fluoride ingestion during tooth development. The effects of fluoride intake in the post-eruptive phase are discussed in the following text.
- Post-Eruptive Phase Fluorine acts on the enamel crystals, plaque, and plaque bacteria in the post-eruptive phase. It acts on enamel crystals to achieve the following three effects:
- Improve crystallinity
- Decrease solubility of crystals
- Catalyse stable apatite crystals
- Enamel crystals are hydroxyapatite crystals made of calcium, oxygen, and hydrogen ions. The chemical formula is Ca10 (PO4)2OH2. The crystalline arrangement of hydroxyl apatite brings together hydroxyl ions (OH–). Two hydroxyl ions react to form a water molecule, which is expressed out or lost with only one residual oxygen atom (–O—link). This is considered as a void or a lattice imperfection. Thsecrystallatticeimperfectionsoccurring due to missing hydroxyl groups (OH–) make the enamel crystals more reactive and soluble. Fluoride ions and hydroxyl ions have the same hydration number and similar ionic radii – 29 and 33 angstrom, respectively. Hence, a fluoride ion can take the void space created due to the loss of a hydroxyl ion easily. This eliminates the lattice imperfections and makes the apatite molecule less reactive, more stable, and relatively insoluble to acids. Such an insoluble molecule is called ‘for hydroxyapatite’ or ‘fluorapatite crystal’.
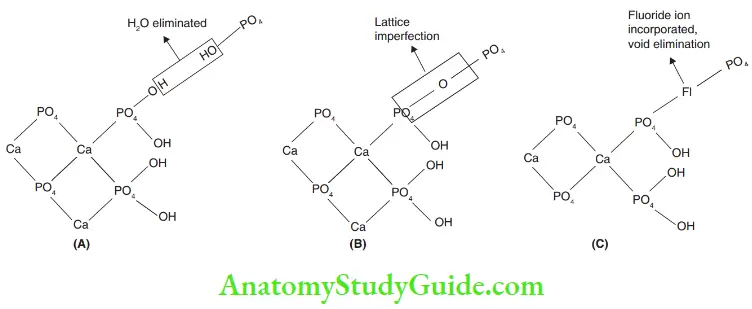 1. (A) Water molecule expressed out.
1. (A) Water molecule expressed out.
2. (B) Lattice imperfection due to loss of hydroxyl group.
3. (C) Fluoride ion takes up void space – fluorapatite crystal. - Ca10 (PO4)OH2 + 2F– = Ca16(PO4)F2 + 2OH–
- Gram-positive rods and cocci are the major organisms in the oral cavity and in the plaque biofilm. Of these, the streptococcus species are the most cariogenic, specifically Streptococcus mutans. Fluoride has a direct effect on cariogenic bacteria. It acts on plaque and plaque bacteria by the following three methods:
- At 2 ppm concentration, fluoride retards the uptake and transport of glucose or glucose analogs into the cells of oral streptococci.
- Fluoride retards the intracellular glycolysis of microorganisms by retarding the enzyme Enolase. Enolase mediates the conversion of phosphoenol pyruvate to pyruvic acid, which is later converted to lactic acid. Lactic acid demineralizes enamel leading to tooth decay. As the enzyme production is reduced, the acid production is decreased leading to decreased caries conduciveness. Hence, fluoride decreases the cariogenic potential of the micro-organism.
- Fluoride ions help in the mineralization of enamel crystals. Surface enamel and plaque biofilm are a reservoir of fluoride ions. Microbial activity produces lactic acid, which decreases the oral pH from 7 to 5.5. Enamel dissolution begins in this acidic environment. However, when the oral pH falls, fluoride is released from surface enamel and plaque. This leads to remineralization of the demineralised enamel.
- Enamel crystals are hydroxyapatite crystals made of calcium, oxygen, and hydrogen ions. The chemical formula is Ca10 (PO4)2OH2. The crystalline arrangement of hydroxyl apatite brings together hydroxyl ions (OH–). Two hydroxyl ions react to form a water molecule, which is expressed out or lost with only one residual oxygen atom (–O—link). This is considered as a void or a lattice imperfection. Thsecrystallatticeimperfectionsoccurring due to missing hydroxyl groups (OH–) make the enamel crystals more reactive and soluble. Fluoride ions and hydroxyl ions have the same hydration number and similar ionic radii – 29 and 33 angstrom, respectively. Hence, a fluoride ion can take the void space created due to the loss of a hydroxyl ion easily. This eliminates the lattice imperfections and makes the apatite molecule less reactive, more stable, and relatively insoluble to acids. Such an insoluble molecule is called ‘for hydroxyapatite’ or ‘fluorapatite crystal’.
Fluoridation
Modes of associating the fluoride ion with the individual can be referred to as ‘fluoridation’. Fluoridation may be achieved by ingestion of a substrate that is treated (added) with fluoride compounds. This is termed ‘systemic fluoridation as the fluoride ion gets to participate in the metabolism of the individual.
Fluoride is an important trace element to the body but its advantages are maximal to teeth in lieu of anti-cariogenicity. Hence, fluoride compounds may be directly applied on the tooth surface allowing fluoride to get incorporated into the enamel surface. There is no ingestion/systemic effect. This is termed ‘topical fluoridation.

The cariostatic mechanisms of fluoride are put to clinical use with systemic and topical fluoridation methods.
Systemic modes of fluoridation are water fluoridation, milk fluoridation, salt fluoridation, and fluoride supplements. Therapeutic formulations include fluoride tablets, multi-vitamin–fluoride combination tablets, and fluoride drops. Topical fluoride involves the application of fluoride solutions/gels that contain 9000–22,000 ppm of fluoride on the tooth surface.
- Systemic Fluorides Systemic fluoridation includes the addition of fluoride in community drinking water, school water supplies, salt, and milk. Dietary fluoride supplements also form an important part of systemic fluorides.
- Systemic Water Fluoridation McKay’s research revealed that enamel mottling was related to the element fluorine in drinking water. The survey conducted by H. T. Dean reaffirmed McKay’s study. He proposed an inverse relationship between the incidence of very mild mottling and dental caries. Continued systematic efforts of H. T. Dean established the optimal threshold of fluoride in drinking water as 1 ppm. At this level, fluoride led to a maximum reduction of caries with only sporadic instances of mild fluorosis of aesthetic disturbance. In 1978, the World Health Organization supported fluoridation and made it evident that the optimum level of fluoride, that is, 1 ppm in drinking water, is physiologically and medically safe in addition to being useful in dental caries prevention. The optimum recommended fluoride levels vary with climate as the average consumption of water increases in warmer climates. In cold climates, the recommended fluoride levels may be as high as 1.2 ppm. In extremely hot climates, a level of about 0.7 ppm and in moderate climates, 1 ppm is recommended. The optimum fluoride concentration of a particular community can be calculated by the equation given in Figure. Systemic water fluoridation can be implemented only in areas that have a central pipe water supply system. In India, only 30% of the population receives water through a centralized water supply. School water fluoridation is done by adding 4–5 ppm of fluoride to the centralized school drinking water supply. Fluoride levels are higher as the students spend less time at school. School water fluoridation is extremely advantageous to children from non-fluoridated/less-fluoridated endemic areas.

- Salt Fluoridation Fluoridation of salt is a viable and feasible method of fluoride ingestion systemically as its distribution can be easily monitored. On average, an individual on an Indian diet consumes 5–8 g of salt per day. An addition of 250 mg of fluoride per kilogram of salt has a similar cariostatic effect to systemic water fluoridation. Also, the addition of fluoride to salt does not alter its color or taste. 200–250 mg of fluoride added to a kilogram of salt does not lead to toxicity. This is because the renal clearance of fluoride is more rapid than that of sodium or chloride.
- Milk Fluoridation Milk is one of the most widely consumed food products throughout the world. Hence, it was thought that the addition of fluoride to milk could prevent tooth decay. In 1952, Ziegler proposed the possibility of milk fluoridation in Switzerland. Contemporarily, milk was reported as an effective vehicle for carrying fluorides by Imamura in Japan in 1953 and by Russof in the USA in 1955. In 1971, the ‘Burrow Foundation’ was founded by Edgar Wilfred Burrow in England. The objective of this charity foundation was to promote caries prevention primarily through milk fluoridation and aimed to make this method as common as water fluoridation. Compounds that have been used to fluoridate milk are calcium fluoride, sodium fluoride, disodium mono fluorophosphate, and disodium silicofluoride. Of these, sodium fluoride has been the most extensively used agent for large-scale milk fluoridation. 2–2.5 mg of sodium fluoride is added to 1 L of milk to attain a fluoride concentration of 2–2.5 ppm.
- The advantages of milk fluoridation are as follows:
- Wide consumption, especially in the younger age group
- Good absorption
- A little alteration in taste
- The cariostatic effect of fats and proteins could occur as an added benefit
- The advantages of milk fluoridation are as follows:
- Fluoride Supplements Dietary fluoride supplements include fluoride tablets and drops. Usually, they contain sodium fluoride. They are available with the multivitamin complex also. On ingestion of a 2.2-mg sodium fluoride tablet, the net absorbed fluorine into the systemic circulation is almost 1 mg. Fluorine tablets are available in the following formulations – 2.2, 1.1, and 0.55 mg. These doses result in net absorption of 1, 0.5, and 0.25 of fluorine ion, respectively. Fluoride drops deliver 0.1 mg of fluoride per drop.
- Dosage The dosage of these supplements has to be calculated according to the following:
- Age of the child
- Weight of the child
- Pattern
- The presence or absence of other preventive measures
- Dosage The dosage of these supplements has to be calculated according to the following:
- Fluoride supplements are indicated beyond the age of 2 years. They are usually taken up to the age of 13 years. The recommended dosages of fluoride by Nikiforuk and Fraser for children are given in Figure. Sodium fluoride tablets are commercially available under the trade names, Fluor-a-day, Tymaflor, and Luride (Hoy Ltd, London, UK). Sodium fluoride tablets are available in combination with vitamins also. Chewable sodium fluoride tablets act as a source of systemic and topical fluoride intake and prevent dental caries. The various techniques of systemic fluoridation discussed earlier are summarised in Figure.


- Systemic Water Fluoridation McKay’s research revealed that enamel mottling was related to the element fluorine in drinking water. The survey conducted by H. T. Dean reaffirmed McKay’s study. He proposed an inverse relationship between the incidence of very mild mottling and dental caries. Continued systematic efforts of H. T. Dean established the optimal threshold of fluoride in drinking water as 1 ppm. At this level, fluoride led to a maximum reduction of caries with only sporadic instances of mild fluorosis of aesthetic disturbance. In 1978, the World Health Organization supported fluoridation and made it evident that the optimum level of fluoride, that is, 1 ppm in drinking water, is physiologically and medically safe in addition to being useful in dental caries prevention. The optimum recommended fluoride levels vary with climate as the average consumption of water increases in warmer climates. In cold climates, the recommended fluoride levels may be as high as 1.2 ppm. In extremely hot climates, a level of about 0.7 ppm and in moderate climates, 1 ppm is recommended. The optimum fluoride concentration of a particular community can be calculated by the equation given in Figure. Systemic water fluoridation can be implemented only in areas that have a central pipe water supply system. In India, only 30% of the population receives water through a centralized water supply. School water fluoridation is done by adding 4–5 ppm of fluoride to the centralized school drinking water supply. Fluoride levels are higher as the students spend less time at school. School water fluoridation is extremely advantageous to children from non-fluoridated/less-fluoridated endemic areas.
- Topical Fluorides Topical fluoride acts by inhibiting demineralization and promoting remineralization at the interface of tooth and oral fluids. Saliva is normally supersaturated with tooth minerals. During an acid attack, calcium and phosphate from the tooth leach out leading to net mineral loss (due to acid dissolution). The presence of fluorides inhibits this process and promotes remineralization by causing fluorapatite crystal formation. They cause precipitation of hydroxyapatite crystals, larger in size than the original. Topical fluoride can be used at home or can be applied professionally. Self-applied fluorides include dentifrices and mouth rinses. Professionally applied fluorides include sols, gels, varnishes, and foams. shows various forms of topical fluorides.
- Forms of Topical Fluorides
- Self-applied – At-home use
- Denitrifies – Contain 1000–1500 ppm of fluoride
- Mouth rinse – Sodium fluoride in the concentration of 0.05 ppm for daily use and 0.2 ppm for weekly use
- Professionally applied – In-office use
- Sols – Sodium fluoride, stannous fluoride, and APF Sol
- Gel – APF gel
- Varnishes – Sodium fluoride and stannous fluoride
- Foams – APF and sodium fluoride
- Self-applied – At-home use
- Forms of Topical Fluorides
- Self-Applied Fluorides
- Fluoride dentifrices: The most commonly used form of topical fluoride is fluoride dentifrice. Fluoride dentifrices act by mechanical action (abrasive system) with a toothbrush. The history of tooth-cleaning dentifrices dates back to 5000 BC. The ancient Egyptians used toothpowder to clean teeth with their fingers. The Romans and Greeks too improvised on the technique. Brick powder and powdered china clay were the earlier used tooth powders. In 1850, glycerine was formulated to be added to the abrasive powders that evolved into ‘toothpaste’ out of these ‘tooth powders’. The earliest toothpaste was in jars. In 1873, Colgate started large-scale manufacture of toothpaste and supplied them in jars. In 1892, Dr. Washington Sheffid invented Dr. Sheffid Crème dentifrice and supplied it in collapsible tubes.
- Fluoride incorporation into toothpaste began in 1950 when Crest came out with its first fluoridated toothpaste carrying an ADA acceptance. Since then, a lot of research has been done on the quantity of fluoride to be added to toothpaste. Concentrations varying from 200 to 5000 ppm of fluoride have been studied. Parameters such as optimal efficacy at caries prevention and lower systemic chronic toxicity were studied.
- The current understanding of the research gives the following data:
- The fluoride concentration that exhibits optimal decay prevention is between 1000 and 1500 ppm
- The addition of more fluoride does not significantly enhance the efficacy of decay prevention
- However, concentrations higher than 1000– 1500 ppm can decrease the therapeutic index of the formulation making it unsafe, if accidentally ingested.
- In children, the toothpaste formulation can contain 350–450 ppm of fluoride as there is a greater risk of accidental ingestion.
- In contrast to toothpaste, topical fluorides contain 9000–22,500 ppm fluoride. They seem to be more effective, at the outset. However, it is also proven that frequent application of low concentrations of fluoride through dentifrices is more beneficial in reducing caries as compared to less frequent application of high fluoride compounds such as in topical fluoride application.
- The various fluoride compounds that have been added to toothpaste to achieve this fluoride concentration are sodium fluoride, stannous fluoride, amine fluoride, and sodium monofluorophosphate. Of these, the more commonly used compounds are sodium fluoride and sodium monoflorophosphate.

- Fluoride mouth rinses: Fluoride was first introduced in mouth rinses in 1946. Sodium fluoride APF and stannous fluoride can be formulated as mouth rinses. Sodium fluoride, acidulated phosphate fluoride (APF) and stannous fluoride mouth rinses are commercially available as Cavi Rinse, Phosflr, and S-FLO, respectively. The mouth rinses are prescribed either weekly or daily. The recommended concentration for daily and weekly use is 0.05 and 0.2, respectively. Mouth rinses have an edge over dentifrices in decay prevention. Dentifrices are ineffctive in interdental areas and less effient at proximal surfaces. Mouth rinses can fly inside interdental areas and protect proximal areas, thus delivering fluoride to contact areas. However, fluoride mouth rinses can only be used as an adjuvant to dentifrices and never solely.
- Fluoride dentifrices: The most commonly used form of topical fluoride is fluoride dentifrice. Fluoride dentifrices act by mechanical action (abrasive system) with a toothbrush. The history of tooth-cleaning dentifrices dates back to 5000 BC. The ancient Egyptians used toothpowder to clean teeth with their fingers. The Romans and Greeks too improvised on the technique. Brick powder and powdered china clay were the earlier used tooth powders. In 1850, glycerine was formulated to be added to the abrasive powders that evolved into ‘toothpaste’ out of these ‘tooth powders’. The earliest toothpaste was in jars. In 1873, Colgate started large-scale manufacture of toothpaste and supplied them in jars. In 1892, Dr. Washington Sheffid invented Dr. Sheffid Crème dentifrice and supplied it in collapsible tubes.
- Professionally Applied Topical Fluorides The various chemical agents that are used as professionally applied topical fluoride agents are sodium fluoride, stannous fluoride, and acidulated phosphate fluoride. The various physical forms in which these agents are applied are sols (solution), gels, varnishes, and foams.
- Indications Professionally applied topical fluoride application is rightly indicated in the following patients:

- Individuals with high caries activity: Fluoride incorporation into the enamel surface can decrease decay activity.
- Patients receiving radiation of the head and neck region: Salivary glands are directly affected by therapeutic radiation, which results in xerostomia. Xerostomia enhances decay susceptibility.
- Patients who are mentally and physically challenged: The special children have poor dexterity to perform oral hygiene measures effectively. This predisposes the oral environment to enhanced decay activity.
- Patients undergoing orthodontic, periodontal, or restorative treatment: During these phases, severe restrain is imposed on maintaining teeth surfaces plaque-free. This increases the plaque bacteria colonization enhancing decay susceptibility. Fluoride application can effectively counter this enhanced decay proneness.
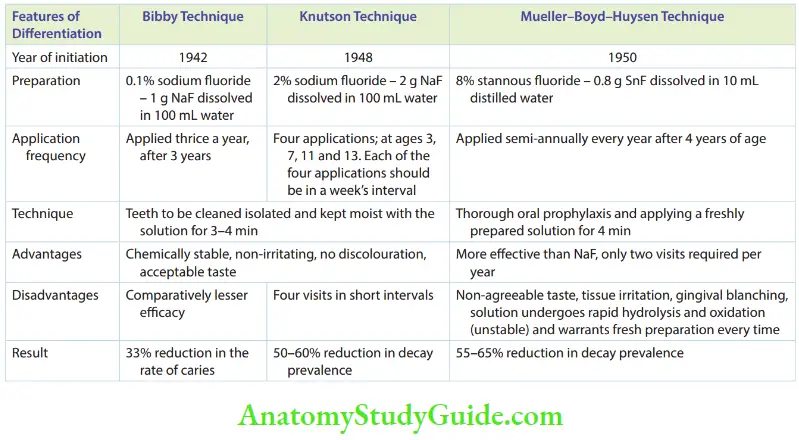
- Fluoride solutions: They can be applied by three common techniques. The application techniques and effects of fluoride solutions are listed in Table.
- APF solution and gel: The application of APF on enamel was introduced by Brudevold in 1960. This was based on the fact that slightly demineralized enamel acquired more fluoride than unaffected enamel. Brudevold developed a solution that was acidulated with orthophosphoric acid and buffered to a pH of 3. APF contains 1.23% fluoride ion concentration. APF is available in the form of a solution and gel. The solution was prepared as follows: 20 g of sodium fluoride + 1 L 0.1-M phosphoric acid + 50% hydrochloric acid (pH 3.0) For the APF gel, methylcellulose or hydroxyethyl cellulose was added (pH 4.5) to the above solution. The APF solution is applied once or twice a year after oral prophylaxis and isolation. The teeth have to be kept moist with the solution for 4 minutes. The patient is instructed not to eat, drink, or rinse the mouth for half an hour after the application of the solution. APF gel is applied by the tray method. Five milliliters of gel are placed on a disposable foam-lined tray. The patient is asked to exert slight pressure on the cheeks and tongue. Biting movements are simulated in this position. APF gel is a thixotropic gel. Although it is a gel at normal consistency, when it is physically stimulated the viscosity is brought down and it becomes a sol. Hence on biting movements, the thixotropic gel flows into the inter-proximal area as its flow increases on the application of pressure. The use of the saliva ejector is mandatory as the solution held in the tray may be ingested. The advantages are that the APF requires only two applications in a year and self-application of the gel is also possible.
- The disadvantages are as follows:
- Increased chair side time because of continued application of the solution
- Acidic, sour, and bitter taste as the fluoride preparation is acidulated
- Cannot be stored in glass containers as the acid of the APF reacts with the glass to form fluorosilicic acid. Fluoride ions are used in this process. As the number of fluoride ions becomes lesser, the potency of the solution decreases.
- The acid of the fluoride solution can cause surface roughening of porcelain for composite restoration.
- The disadvantages are as follows:
- Fluoride varnishes: They were developed with the idea of prolonging the contact of fluoride with enamel. The slow-release mechanism of the varnish could lease fluoride when most needed. The two widely used varnishes are Duraphat and Fluor Protector. The fluoride varnishes are recommended to be used biannually. There are no contraindications to the use of fluoride varnishes.
- Fluoride solutions: They can be applied by three common techniques. The application techniques and effects of fluoride solutions are listed in Table.
- Indications Professionally applied topical fluoride application is rightly indicated in the following patients:
The Fluor protector contains 7000 ppm fluoride. It is prepared by dissolving 0.7% polyurethane-based lacquer in chloroform. The active component in this varnish is dichlorosilane. The amount of fluoride deposited in the surface layer of enamel is very high. It forms tags that penetrate into the enamel and thus inhibit further fluoride penetration. Duraphat contains 22,600 ppm of fluoride, that is, 2.26%. It contains sodium fluoride in organic lacquer. Duraphat is more effective than floor protector.
Fluoride Toxicity
Fluoride is a ‘twin-edged sword’. It is highly efficient in the prevention of decay. At the same time, ingestion of a higher volume of fluoride can lead to toxic manifestations. The toxicity can surface only with the ingestion of fluoride-incorporated substrates such as water, dentifrice, and topical fluoride agents.
One bolus ingestion of a substantially large Florida entity is termed ‘acute fluoride toxicity’. This can happen when a child mishandles an adult toothpaste tube and swallows almost all of its contents.
Prolonged ingestion of fluoride compounds, the concentration of which is higher than the permissible dose, leads to the demonstration of signs/symptoms of chronic toxicity. A child who has been drinking groundwater containing 5 ppm fluoride for the first 10 years of life demonstrates chronic fluoride toxicity.
- Acute Fluoride Toxicity The probably toxic dose or, PTD or fluoride is the minimum dose that can cause serious or life-threatening systemic signs and symptoms. It warrants immediate therapeutic intervention and hospitalization. The exact toxic dose cannot be accurately stated. Children with oral fluoride ingestion of 5.0 mg/kg body weight have been reported to have complications. This can be considered as the PTD. This does not mean that doses below 5.0 mg/kg body weight are harmless. Sodium fluoride and stannous fluoride generally have more potential to cause acute toxicity than mono fluorophosphate, calcium fluoride, and amine fluoride. 10 mL of sodium fluoride mouth rinse is advocated for one-time use. 110 mL of 0.2%, that is, 10 times the prescribed volume, of sodium fluoride mouth rinse, produces acute toxicity. Similarly, 50 g of 1000 ppm of fluoride-containing toothpaste swallowed bya 1-year-old child is the probable toxic dose (5 mg/kg body weight). Fift fluoride supplement tablets consumed at a time can cause acute toxicity. Acute fluoride toxicity may also result from the ingestion of topical fluoride agents as they contain 9000–22,500 ppm of fluoride. Solutions, foams, and thixotropic gels pose a higher risk than gels and varnishes owing to the difference in their viscosities. To prevent ingestion of these agents, it is mandatory to follow certain basic tenets during topical fluoride application. These are illustrated in Figure. Precise efforts must be taken to ensure that small children do not have access to fluoride mouth rinses, tablets, and dentifrices. Personal supervision is a must during the use of dentifrices.

- Clinical Features The signs and symptoms of acute fluoride toxicity vary with the concentration of fluoride ingested. Ingestion of 2 mg/kg body weight of fluoride can cause nausea, vomiting, headache, sweating, epigastric distress, nasal/oral mucus discharge, excess salivation, and diarrhea. Ingestion of 5 mg/kg of fluoride has more serious symptoms such as hypotension, cardiac arrhythmias, disturbances in electrolyte balance (hypocalcemia and hyperkalemia), respiratory distress, metabolic acidosis, and coma.
- Treatment Attempts must be made to eliminate the toxic dose of fluoride from the body and minimize further absorption of fluoride. The following line of management is advocated in case of acute toxicity:
- The vital signs should be supported.
- Administration of an emetic if the patient possesses a gag reflex: This is done if the patient does not vomit, is not unconscious, and does not have convulsions.
- Investigation of blood: pH, plasma fluoride concentration and serum chemistry profiles have to be performed.
- Glucose must be administered through 4 lines to reverse hyperkalemia and calcium gluconate must be given to maintain sodium and calcium levels.
- Na2HCO3 or Ringer’s lactate must be given to attain the following:
- Increased urinary flow
- Decreased acidosis
- Increased pH
- Increased excretion of fluoride
- The patient should be continuously monitored till normalcy is attained.
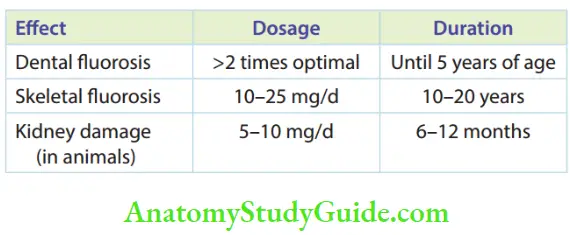
- Chronic Fluoride Toxicity The toxic effects of chronic excessive fluoride ingestion are listed in Table, with the dosage and duration of ingestion.
- Clinical Features Chronic ingestion of fluoride has been reported to cause allergy, carcinoma, birth abnormalities, mutation, and other genetic disorders. Costoflorosis has been reported in individuals with chronic fluoride intake. The following features are observed in these patients:
-
-
- Increased bone density
- Bones of extremities show thickening, exocytosis, and osteophytes
- Calcification of tendons and ligaments
-
Clinically, the pain has been reported in the joints of the hands, feet, and spine. As severity increases, stiffness of gait and limitation of movement occurs. When the toxicity is most severe, there is crippling of movement and extreme pain in the spine. Joints become rigid, and virtual immobilization of the individual occurs. The patient has to be treated by a physician under emergency care.
Fluoride for Teeth Summary
- Fluorine is the most electronegative and extremely reactive element with anti-cariogenic effectiveness well documented.
- A brief history of fluorides with reference to preventive dentistry is given in Figure.
- The cariostatic mechanisms of fluoride are operational in two phases:
- Post-eruptive phase
- Enamel crystals
- Improve crystallinity
- Decrease solubility of crystals
- Catalyse stable apatite crystals
- Plaque and plaque bacteria
- Promote remineralization of decalcified enamel
- Decrease acid formation
- Inhibit bacterial metabolism and growth
- Enamel crystals
- Pre-eruptive phase
- Alteration in crown morphology
- Fluoridation can be of two types: systemic and topical

- Fluoridation can be of two types: systemic and topical
- Alteration in crown morphology
- Post-eruptive phase
- Acute fluoride toxicity occurs from the ingestion of a large amount of fluoride. Probably toxic dose: The PTD of fluoride is the minimum dose that could cause serious or life-threatening systemic signs and symptoms that might trigger immediate therapeutic intervention and hospitalization.
- Chronic fluoride toxicity: Fluoride has been reported to cause allergy, carcinoma, birth abnormalities, mutation, and other genetic disorders.

Leave a Reply