Physical Pharmaceutics 1 Long Question And Answers
Question 1. Define solubility. Explain the various factors affecting the solubility of drugs.
Answer:
Solubility:
The term solubility can be defined as a property of a substance (solute) to dissolve in a given solvent. A solute is any substance that can be either solid or liquid or gas dissolved in a solvent.
Factors affecting solubility:
Solubility of a compound depends upon the physical and chemical properties of the solute and the solvent, as well as upon factors such as,
- Temperature
- pH
- Particle size
- Crystal structure
- Molecular structure
- Nature of solvent
1. Temperature
- Generally, as the T increases the solubility increases.
- For the effect of T on solubility we have to consider two criteria
- Solubility increases with temperature.
- This is the case for most of the solvents.
- The situation is different for gases. With the increase of the temperature they became less soluble in each other and water, but more soluble inorganic solvents.
Endothermic Reactions:
- During the dissolution process, the energy (heat) is absorbed.
- Thus rise in T will lead to an increase in the solubility of a solid in the solution with a positive heat of the solution
Exothermic Reactions:
- During the dissolution process, the energy (heat) is evolved.
- Thus rise in T will lead to a decreased solubility of a solid in the solution with a negative heat of the solution.
Non-Polar Compounds:
- The forces holding the particles together are small, and any interaction between solute and solvent is small.
- No detectable heat effect on the POLAR substance.
Polar Compounds:
- Either decrease OR increase in solubility. In polar substances, it takes energy to separate the molecule from surrounding molecules & if energy is supplied in the form of heat, producing a cooling effect.
- Also, there is the possibility of interaction between the solute and solvent with the formation of a dipole-dipole type bond, and this interaction will tend to give off heat.
- Depending on which of the two interactions is greatest you can get an increase or decrease in temperature
2. pH
We have to consider for
- Nonionizable Substances:
- Little effect of pH on nonionizable substances.
- Solubility can be increased by a change of dipole moment.
- Ionizable Substances:
- Depends On Hederson-Hesselbalch equation.
- For Acidic drug pH= pKa+log [(S-S0)/S0]
- S = Overall solubility of a substance
- For basic drug
- So = Solubility of unionized species
- pH= pKa+log [S0/(S-S0)] S-So=solubility of ionized species pH of a substance is related to its pKa and the concentration of the ionized and unionized forms of the substance.
- The effect of pH on a weak acid (A) and a weak base (B)
- Depends On Hederson-Hesselbalch equation.
3. Particle Size
As the P.S decreases solubility increases due to an increase in the sin-face area. But after a very small P.S decrease in P.S will decrease solubility due to the formation of agglomerates.
log S/S0=[2yM/2.303RTpr]:
S = Solubility ofsmall particle ofradius r
M = Molecular weight
S0 = Normal solubility
R = Gas constant
y = Surface tension
ρ = Density
4. Crystal Structure
- The amorphous form of drugs is more soluble than the Crystalline form.
- Solubility: Solvates >Anhydrous > Hydrates
- The effect of crystal structure on solubility is explained in the chapter on preformulation in the subtopic crystallinity
5. Molecular Structure
Change in the molecular structure highly affects the solubility of the compound. Example: Introduction of the hydrophilic group in hydrophobic substance may improve solubility.
Examples:
- Introduction of hydrophilic group Benzene into phenol with increased solubility.
- Conversion into salt Generally all salt forms are soluble.
- Esterification of Chloramphenicol into palmitate form for taste masking
6. Pressure
Solid and liquid solutes: For the majority of solid and liquid solutes, pressure does not affect solubility.
- Gas Solute:
- As for gases Henry’s law states that the solubility of a gas is directly proportional to the pressure of this gas.
- This is mathematically presented as p = kc, where k is a temperature-dependent constant for a gas.
- A good proof of Henry’s law can be observed when opening a bottle of carbonated drink. When we decrease the pressure in a bottle, the gas that was dissolved in the drink bubbles out of it.
Question 2. Describe the principle, construction, and working of the Abbes refractometer. Write a note on its applications.
Or
Define refractive index. Discuss in detail the working of Abbe’s refractometer.
Answer:
Abbes refractometer:
A refractometer is an instrument that measures the refractive index by making use of total internal reflection.
- Pulfrich designed a refractometer enabling the refractive index of a liquid to be easily obtained, which consists of a block of glass G with a polished and vertical face.
- On top of G is cemented a circular glass tube V.
- The liquid L is placed in V, and a convergent beam of monochromatic light is directed so that the liquid-glass interface is illuminated.
- On observing the light refracted through G by a telescope T, a dark and light field of view is seen.
- Abbe designed a refractometer for measuring the refractive index of liquids whose principle is illustrated as follows:
- Two similar prisms X and, Y are placed on table A, the prism X being hinged at H so that it could be swung away from Y, A drop of the liquid is placed on the surface a, which is matt, and the prisms are placed together so that the liquid is squeezed into a thin film between them.
- Light from a suitable source is directed towards the prisms using a mirror m, where it strikes the surface a, and is scattered by the matt surface into the liquid film.
- The emergent rays are collected in a telescope T directed toward the prisms, and the field of view is divided into a dark and bright portion.
- Table A is then turned until the dividing line between the dark and bright fields is on the crosswires of the telescope, which is fixed.
- The reading on a scale S, which is attached to, and moves with, the table, gives the refractive index of the liquid directly.
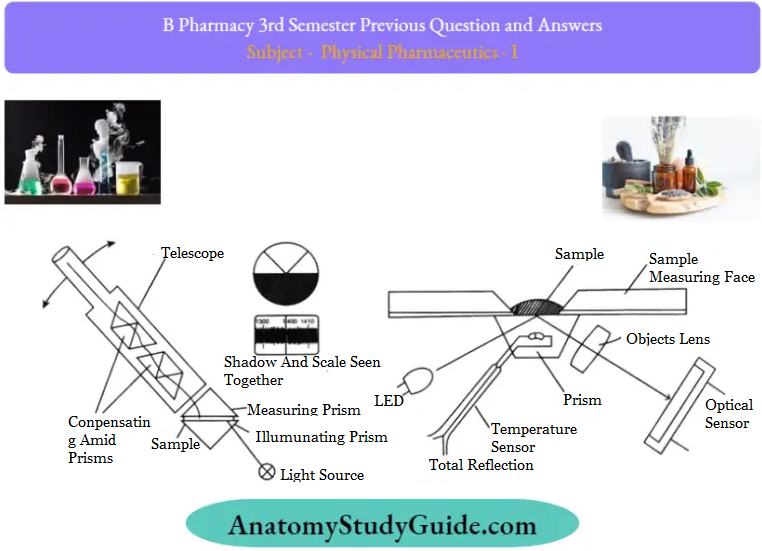
Abbe’s refractometer Applications:
- Abbe’s refractometer is used to measure the refractive index of the given organic liquid.
- Using a particular monochromatic light source, the apparatus is calibrated with water as the liquid.
- Adjust the micrometer screw to focus the boundary between the bright and dark regions.
Question 3. Define adsorption isotherm. Explain Freundlich and Langmuir’s adsorption isotherm.
Answer:
Adsorption isotherms have been of immense importance to researchers dealing with environmental protection and adsorption techniques.
- The two primary methods used for predicting the adsorption capacity of a given material are known as the Freundlich and Langmuir isotherms.
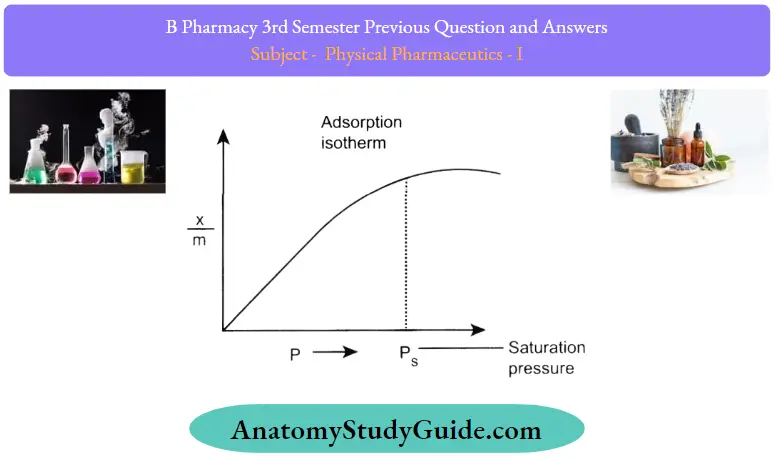
- An adsorption isotherm is a graph that represents the variation in the amount of adsorbate (x) adsorbed on the surface of the adsorbent with the change in pressure at a constant temperature.
- As we know from Le Chatelier’s principle, the direction of equilibrium in a reaction shifts in the direction in which stress is relieved.
- So, here we can see that upon application of excess pressure on the system, the equilibrium shifts in the direction where the number of molecules decreases so that the pressure in the system decreases
- From the graph, we also observe that after attaining a pressure Ps, that is the saturation pressure, the variation in the amount of adsorbent adhering to the adsorbate becomes zero.
- This happens because the surface area available for adsorption is limited and as all the sites are occupied, a further increase in pressure does not cause any difference.
Question 4. Different adsorption isotherms have been proposed by different scientists namely:
Answer:
- Langmuir isotherm
- Freundlich isotherm
- BET theory
The graph shown above shows the isotherm proposed by Freundlich.
Freundlich Adsorption Isotherm:
Freundlich adsorption gives the variation in the quantity of gas adsorbed by a unit mass of solid adsorbent with the change in pressure of the system for a given temperature.
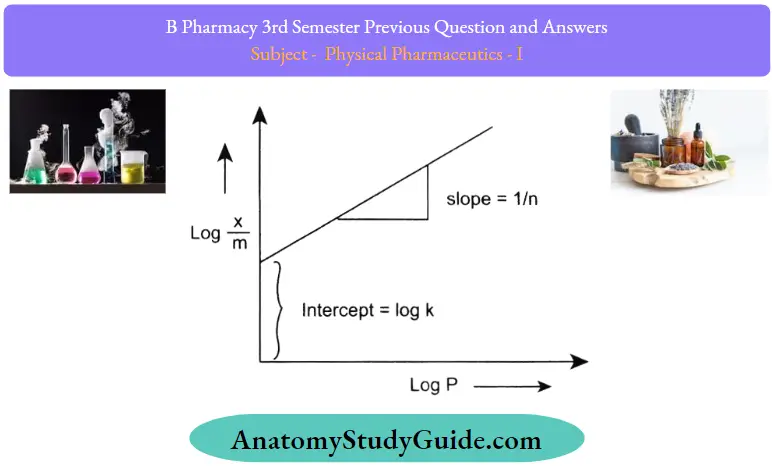
The expression for the Freundlich isotherm can be represented by the following equation:
x/m = kP1/n
Where,
n>l
Where x is the mass of the gas adsorbed, m is the mass of the adsorbent, P is the pressure and n is a constant that depends upon the nature of the adsorbent and the gas at a given temperature. Taking the logarithm on both sides of the equation, we get,
Log x/m = log k+ 1/n log P
The plot of this equation is a straight line as represented by the following curve
Langmuir Adsorption Isotherms:
The Freundlich adsorption isotherm is followed by another two isotherms, Langmuir adsorption isotherms, and BET theory. The Langmuir adsorption isotherms predict linear adsorption at low adsorption densities and a maximum surface coverage at higher solute metal concentrations.
The Langmuir adsorption isotherm has the form:
θ = Kp1 + Kpθ = Kp1 + Kp
Where
- θ is the fraction of the surface covered by the adsorbed molecule.
- K is an equilibrium constant known as the adsorption coefficient.
- { K= ka/kd = rate constant for adsorption/ rate constant for desorption}
- p is the pressure.
The Langmuir adsorption is applicable for monolayer adsorption onto a homogeneous surface when no interaction occurs between adsorbed species.
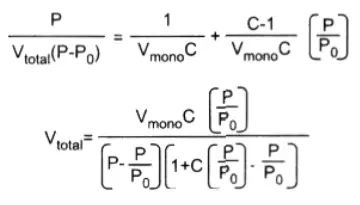
BET Adsorption Isotherm:
The theory of multilayer adsorption proposed by Brunauer, Emmett, and Teller in 1938 (BET Theory) assumes that physisorption results in the formation of multilayer adsorption. The theory also assumes that the solid surface has uniform sites of absorption and that adsorption at one site does not affect adsorption at neighboring sites.
After the formation of the monolayer, the adsorption process can continue with the formation of a multilayer involving the second layer, third layer, and so on. The equation for BET is
Adsorption Applications:
Following are the applications of adsorption
- Gas masks: Poisonous gases get adsorbed to the surface of the mask and prevent its encounter when used by coal miners.
- Production of vacuum: Traces of air are adsorbed on charcoal and removed from devices undergoing the process of evacuation.
- Removal of moisture: Silica gel pellets are used for the adsorption of moisture in medicines and new plastic bottles to control humidity.
- Removal of color: The juice extracted from the cane is treated with animal charcoal for the removal of the coloring agent to get a clear liquid solution.
- As Catalysts: Suitable materials are used as a catalyst such that reactants get adhered to its surface, thus enabling the reaction to proceed at a faster rate and increasing the rate of reaction
Question 5. Explain the various factors influencing the dissolution of poorly soluble drugs.
Answer:
1. Factors Relating to the Physicochemical Properties of The Drug
Solid-Phase Characteristics:
- The solid-phase characteristics of drugs, such as amorphicity and crystallinity.
- Have been shown to have a significant effect on the dissolution rate, studies have demonstrated that the amorphous form of a drug usually exhibits greater solubility and higher dissolution rate as compared to that exhibited by the crystalline form, Piccolo and Saks3.
- Showed that the dissolution rate of amorphous erythromycin estolate is markedly lower than the crystalline form of erythromycin estolate, as exemplified.
Polymorphism:
- The polymorphic forms of drugs have been shown to influence changes in the solubilizing characteristics and thus the dissolution rate of the drug substance.
- Numerous reports have shown that polymorphism and the states of hydration, salvation, and/or complexation markedly influence the dissolution characteristics of the drug.
Coprecipitation andlor Complexation
- Numerous reports have shown that polymorphism and the states of hydration, salvation, and/or complexation markedly influence the dissolution characteristics of the drug.
- Radical Characteristics According to Nemst Brunner’s theory, the dissolution rate is directly proportional to the surface area of the drug.
- Since the surface area increases with decreasing particle size, higher dissolution rates may be achieved through the reduction of particle size.
- This effect has been highlighted by the superior dissolution rate observed after the micronization of certain sparingly soluble drugs as opposed to the regularly milled form.
2. Factors related to drug product formulation A variety of factors concerning the formulation:
A variety of factors concerning the formulation of a drug product can directly influence the dissolution rate of the active ingredient contained within it. Once these factors are completely characterized, one can use this information to achieve custom-tailored drug dissolution profiles. This information is then employed in the development of optimally effective dosage forms.
Excipients and Additives:
- Most solid dosage forms incorporate more than one excipient for various purposes together with the active ingredient 21 in the formulation.
- It has been shown that the dissolution rate of a pure drug can be altered significantly when mixed with various adjuncts.
- These adjuncts include diluents, binders, lubricants, granulating agents, disintegrants, and so on.
- In the following discussion, we address the influence of excipients on the rate of dissolution of the active ingredient from a dosage form.
Particle Size:
- Several investigators have concluded that in most instances, a reduction in particle size of drugs contained in tablets or capsules will enhance dissolution and absorption.
- This can most likely be attributed to procedure 22 employed in tablet production: that is, mixing the drug with usually hydrophilic diluents and subsequent granulation will result in a more hydrophilic surface, even for originally hydrophobic drug particles.
- Finholt et al.25,26. have extensively evaluated the effect of particle size on the dissolution rate of drugs from granules and tablets.
- Figures illustrate this phenomenon with phenacetin and phenobarbital as the test compounds. Several other investigators have reported similar results26,20,27.28.
Granulating agent and binder:
- It has been reported by several investigators that binder and granulating agents incorporated in tablet formulation and other solid dosage forms can markedly influence the dissolution characteristics of the drug from the dosage form.
- Solvong and finholt19 have shown that Phenobarbital tablets granulated with gelatin solution provide a faster dissolution rate in human gastric juice than those prepared using sodium carboxymethylcellulose or polyethylene glycol 6000 as a binder.
Disintegrating Agents:
- Several reports have been published in the literature demonstrating the effect of various disintegrating agents on the dissolution rate of tablets 27-29.
- It must be noted that the type and amount of disintegrating agent employed in the formulation significantly controls the overall rate of dissolution of the dosage form.
Lubricants:
- Lubricants that are commonly incorporated in the formulation of solid dosage forms fall predominantly in the class of hydrophobic compounds. Consequently, the nature, quality, and quantity of the lubricant added can affect the dissolution rate.
- The effects of various lubricants on the dissolution rate of salicylic acid tablets were studied by Levy and Gumtow 30.
- They concluded that magnesium stearate, a hydrophobic lubricant, tends to retard the dissolution rate of salicylic acid tablets, whereas sodium lauryl sulfate enhances dissolution, due to its hydrophilic character combined with surface activity, which increases the microenvironment pH surrounding the weak acid and increases wetting and better solvent penetration into the tablets
The interfacial tension between drug and dissolution medium:
The properties of the interface between the drug and the dissolution medium can become a deciding factor as far as the dissolution rate is concerned. The characteristics can be modified by the addition of an agent that acts at the interface
Surfactant:
The drugs that are practically insoluble in aqueous medium (<0.01%) are of increasing therapeutic interest, particularly due to the problems associated with their bioavailability when administered orally. Drugs with low solubilities when incorporated with surfactants can enhance their dissolution rate
3. Factor related to the dissolution testing device Eccentricity of Agitating (Stirring) Element:
The current official compendium specifies that the stirring shaft must rotate smoothly without significant wobble. The lord significance gives the experimenter full right to ensure that such wobble does not significantly affect the dissolution rate.
- Additionally, USP XX/NF XV states that the axis of rotation of the stirring shaft must not deviate > 2 mm from the axis of the stirring vessel.
- This implies that this specification permits eccentricity up to ±2 nun but that such eccentricity must not significantly affect the dissolution rate. This is certainly an excessive amount.
Vibration:
- The speed of the rotational device selected by the official compendium is 100 rpm. Other speeds are specified for certain drugs.
- Precise speed control is best obtained with a synchronous motor that locks into line frequency 25.
- Such motors are not only more rugged but are far from reliable. Periodic variations in rpm might result in a possible disturbance in rotational acceleration.
This phenomenon, present in almost all rotational devices, is commonly referred to as torsional vibration.
- Such vibration indicates a variation in the velocity of rotation for short periods. Their average velocity was well within ±4% of the specified rate.)
- Vibration is a common variable introduced into a dissolution system due to various causes. It can affect changes in the flow patterns of the dissolution medium.
- Additionally, it can introduce unwanted energy to the dynamic system. Both effects may result in significant changes in the dissolution ratel-8
Agitation Intensity:
It can be stated with a significant amount of certainty that the degree of agitation, or the stirring conditions, is one of the most important variables to consider in dissolution. Given the background on the various theories of dissolution, it is apparent that agitation conditions can markedly affect diffusion-controlled dissolution because the thickness of the diffusion layer is inversely proportional to agitation speed. Wurster and Taylor 39 employed the empirical relationship.
K = a(N)b
Where N is the agitation rate, K is the reaction (dissolution) rate, and a and b are constants. For diffusion-controlled processes, b = I Dissolution that is interfacial-reaction rate-controlled will the independent of agitation intensity, and thus b = 0
Flow Pattern Disturbances;
For dissolution-rate data to be reproducible and reliable, the flow pattern should be consistent from test to test
- The geometry and alignment of the stir-ring device, external vibration, and rotational speed are some of the factors that can influence flow patterns.
- In 1978, DRTL conducted an extensive examination of these factors and their influence on dissolution testing.
- They concluded that the geometry of the rotating paddle and/or basket, the flask dimensions, and the sampling positions can all introduce various types of flow patterns that can alter the dissolution characteristics of the drug product.
- Coxat al.29 further suggested that variations in the smoothness of the round-bottomed flask can make significant differences in flow patterns as well.
- The influence on flow patterns of the vertical distance of the basket or paddle from the lowest point of the bottom of the round-bottomed flask should also be considered. The official compendium specifies this distance to be 2.5 cm (±2 mm).
Sampling Probes, Position, and Filters:
- The large probe can affect the hydrodynamics of the system and therefore the dissolution rate of some dosage forms, causing results that differ from those obtained by manual sampling 30-35.
- USP/NF states that samples should be removed at approximately half the distance from the bottom of the basket or paddle to the surface of the dissolution medium and not closer than 1 cm to the side of the flask.
- The choice of a filter should be preceded by an investigation of the adsorption characteristics of the drug and the particular filter material.
Question 6. Factors related to dissolution test parameters:
Answer:
Temperature:
USP/NF specifics that the dissolution medium must be held at 37°C (0.5°). Although most commercial water baths can meet this standard of performance, failure to meet this requirement is not uncommon.
- It is often assumed that the water bath temperature and the flask temperature are the same. Plastic flasks have a heat transfer coefficient of approximately 3.5 times less than that of glass
- M. As the temperature difference between the bath and the flask’s medium is lowered, the amount of heat transferred into the flasks is reduced.
- It is vital to cover the flasks at least during dissolution testing.
- Since the drug solubility is temperature dependent, its careful control during the dissolution process is crucial.
The effect of temperature variations of the dissolution medium depends mainly on the temperature-solubility curves of the drug and excipients in the formulation.
Stokes’ equation 36 explains the temperature dependency of a dissolved molecule and diffusion coefficient:
D = k T
6πηr
Where k is the Boltzmann constant and the denominator expresses the Stokes force for a spherical molecule, n is the viscosity, and r is the radius of the molecule.
Dissolution Medium:
The constituents, nature, and overall characteristics of the dissolution medium have a significant bearing on the dissolution performance of a drug substance. Also, the selection of the proper dissolution medium for dissolution testing depends on the solubility of the drug as well as on economics and practicality. Factors such as dissolved gases, media pH, and viscosity of the medium are significantly influential as far as the dissolution rate is concerned.
Viscosity:
The dissolution rate decreases with the increased viscosity of the dissolution medium; especially in the case of diffusion diffusion-controlled dissolution process, viscosity has very little effect on the interfacial-controlled dissolution process.
Sorption:
The relative density of the tablets was found to decrease, resulting in increased disintegration time with an increase in water sorption-rate constants.
Humidity:
About the dissolution rate of a drug substance, humidity is usually associated with storage effects.
- Moisture has been shown to influence the dissolution of many drugs from solid dosage forms and decreases the dissolution of prednisone tablets exposed to the environment with varying degrees of relative humidity.
- Hanson found that three of the four formulations of prednisone tablets showed drastic decreases in dissolution rate when the tablets were exposed to high humidity.
- However, did not find significant changes in the dissolution behavior of the two drugs, except for weight gain.
- The environmental conditions to which the dosage forms are exposed, moisture content in particular, should be rigorously assessed reproducible and reliable dissolution data are to be obtained.
- Additionally, humidity during the manufacture of the dosage form should be carefully controlled to guarantee the quality of the product from batch to batch
Detection Errors:
Analytical methods be checked carefully for each dissolution system. Extreme care must also be exercised when laboratory methods are introduced into quality control to ensure that no part of the equipment interferes with sensitive determinations.
Question 7. Discuss dielectric constant and dipole moment with their applications in pharmacy.
Answer:
The dielectric constant of any substance refers to the relative permittivity of the dielectric substance. It is the proportion of the permittivity of the material to the permittivity of the free space.
K = e/e0
e = Permittivity ofthe substance
e0 = Permittivity of the free space
The Theory Behind Dielectric Constant:
This is a prime parameter to characterize a capacitor. A capacitor is an electronic component designed to store electric charge. This is widely built by sandwiching a dielectric insulating plate in between the metal conducting plates. The dielectric property plays a major role in the functioning of the capacitor.
The layer made up of dielectric material decides, how effectively the capacitor can store the charge. Picking the right dielectric material is crucial. Thus, we can also define it as ‘the ratio of the electric field without a dielectric (E0) to the net field with a dielectric (E).’
K = E0 E
Here, the value of E0 is always greater than or equal to E. Thus, The value of the dielectric constant is always greater than 1.
The greater the value of K more charge can be stored in a capacitor.
In the capacitor, the capacitance is given by
C = kC0
Thus, filling the gap between the plates with dielectric material will increase its capacitance by the factor of the dielectric constant value.
In the parallel plate capacitor, the capacitance is given by:
C=K ε0 Ad
Where,
- C is the capacitance of the parallel plate capacitor.
- K is the dielectric constant.
- ε0 is the permittivity of the free space.
- A is the area of parallel conducting plates
- D is the separation between parallel conducting plates
The capacitance value can be maximized by increasing the value ofthe dielectric constant and by decreasing the separation between the parallel conducting plates.
Dielectric Constant Value:
Thus, the value of the dielectric constant is crucial in building various electronic components. The following table gives some typical values of dielectric constants:

Dielectric Applications:
- The dielectric has the property of making the space larger or smaller than the actual size.
- For example, when a dielectric material is placed between two charges, it reduces the electric field force acting between them, as if they were moved away.
- When an electromagnetic wave passes through a dielectric medium, the speed of the wave is reduced, and has a shorter wavelength
Dipole moment:
A bond dipole moment is a measure of the polarity of a chemical bond between two atoms in a molecule. It involves the concept of electric dipole moment, which is a measure of the separation of negative and positive charges in a system.
The bond dipole moment is a vector quantity since it has both magnitude and direction. An illustration describing the dipole moment that arises in an HCl (hydrochloric acid) molecule is provided below.

Dipole Moment has a Magnitude and a direction
It can be noted that the symbols δ+ and δ– represent the two electric charges that arise in a molecule which are equal in magnitude but are of opposite signs.
They are separated by a set distance, which is commonly denoted by ‘d’.
- The dipole moment of a single bond in a polyatomic molecule is known as the bond dipole moment and it is different from the dipole moment of the molecule as a whole.
- It is a vector quantity, i.e. it has magnitude as well as definite directions.
- Being a vector quantity, it can also be zero as the two oppositely acting bond dipoles can cancel each other.
- By convention, it is denoted by a small arrow with its tail on the negative center and its head on the positive center
- In chemistry, the dipole moment is represented by a slight variation of the arrow symbol. It is denoted by a cross on the positive center and an arrowhead on the negative center. This arrow symbolizes the shift of electron density in the molecule.
- In the case of a polyatomic molecule, the dipole moment of the molecule is the vector sum of all present bond dipoles in the molecule
Dipole Moment Formula:
A dipole moment is the product of the magnitude of the charge and the distance between the centers of the positive and negative charges. It is denoted by the Greek letter μ
Mathematically,
Dipole Moment (μ) = Charge (Q) distance of separation (r)
It is measured in Debye units denoted by ‘D’. 1 D = 3.33564 ×10-30 C.m, where C is Coulomb and m denotes a meter.
The bond dipole moment that arises in a chemical bond between two atoms of different electronegativities can be expressed as follows:
μ = δ.d
Where,
- μ is the bond dipole moment,
- δ is the magnitude of the partial charges δ+ and δ–,
- And d is the distance between δ+ and δ–
The bond dipole moment (μ) is also a vector quantity, whose direction is parallel to the bond axis. In chemistry, the arrows that are drawn to represent dipole moments begin at the positive charge and end at the negative charge.
When two atoms of varying electronegativities interact, the electrons tend to move from their initial positions to come closer to the more electronegative atom. This movement of electrons can be represented via the bond dipole moment.
Examples:
Dipole moment of BeF2
In a beryllium fluoride molecule, the bond angle between the two beryllium-fluorine bonds is 180°. Fluorine, being the more electronegative atom, shifts the electron density towards itself. The individual bond dipole moments in a
BeF2 molecules are illustrated below.
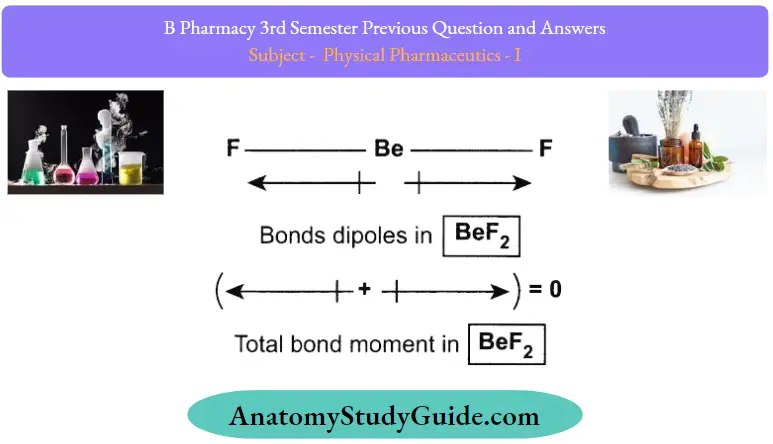
From the illustration provided above, it can be understood that the two individual bond dipole moments cancel each other out in a BeF2 molecule because they are equal in magnitude but are opposite in direction. Therefore, the net dipole moment of a BeF2 molecule is zero.
The dipole moment of H2O (Water):
In a water molecule, the electrons are localized around the oxygen atom since it is much more electronegative than the hydrogen atom. However, the presence of a lone pair of electrons in the oxygen atom causes the water molecule to have a bent shape (as per the VSEPR theory). Therefore, the individual bond dipole moments do not cancel each other out as is the case in the BeF2 molecule. An illustration describing the dipole moment in a water molecule is provided below.
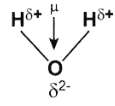
The bond angle in a water molecule is 104.5°. The individual bond moment of an oxygen-hydrogen bond is 1.5 D. The net dipole moment in a water molecule is found to be 1 .84D.
Dipole moment Applications:
1. Distinction Between Polar and Non-Polar Molecules:
The molecules having dipole moment are called polar molecules whereas molecules having zero dipole moment are said to be non-polar molecules.
For example:
- Molecules such as H2, N2, O2, Cl2, etc. have non-polar bonds and zero value of dipole moment.
- Molecules such as CO2, BF3, CH4, etc., have polar bonds but zero value of dipole moment.
- Molecules such as HF, HCl, NH3, and H2O, have polar bonds and their dipole moment is greater than zero. In other words, they are polar molecules.
2. Ionic Character in A Molecule:
The dipole moment gives an idea about the ionic character of a bond or a molecule. For example, let us calculate the percentage of ionic character in the HCl molecule.
Experiments have shown that the dipole moment of HCl is 1.03 D and its bond length is 1.275A°. Now for 100% ionic character the charge developed on H and Cl atoms would be 4.8 × 10 -10 e. s. u.
Therefore, the dipole moment in case of 100% ionic character is given as:
μ = 4.8 × 10-10 × 1.275 × 10-8 e. s. u.-cm
= 6.12 × 10-8 e. s. u.-cm = 6.12 D
The observed dipole moment
μ = 1.03 D
Therefore, Percentage Ionic character = 1.03/6.12 × 100 = 16.83%
In general, the larger the value of the dipole moment is, the more the ionic character.
3. Shapes of The Molecules:
- The value of dipole moment helps to predict the shape of the molecule. For example, experiments show that the dipole moment of BeF2 is zero.
- This is possible only if bond dipoles of two Be- F bonds cancel each other. In other words, the two bond dipoles must be oriented in opposite directions.
- This is possible if the molecule is linear.
- Similarly, water has a dipole moment of 1.83 D.
- Thus, its molecule cannot be linear because the bond dipoles do not cancel each other. Thus, the molecule must have an angular shape.
Question 8. Defines surface tension. Explain the principle involved in the determination of surface tension by the capillary rise method.
Answer:
“Surface tension is the tension of the surface film of a liquid caused by the attraction of the particles in the surface layer by the bulk of the liquid, which tends to minimize surface area”.
To find the surface tension of water by the capillary rise method
Apparatus/ Materials Required:
Three capillary tubes of different radii
- A tipped pointer clamped in a metallic plate with a handle
- Traveling microscope
- Adjustable height stand
- A flat bottom open dish
- Thermometer
- Clean water in a beaker
- Clamp and a stand
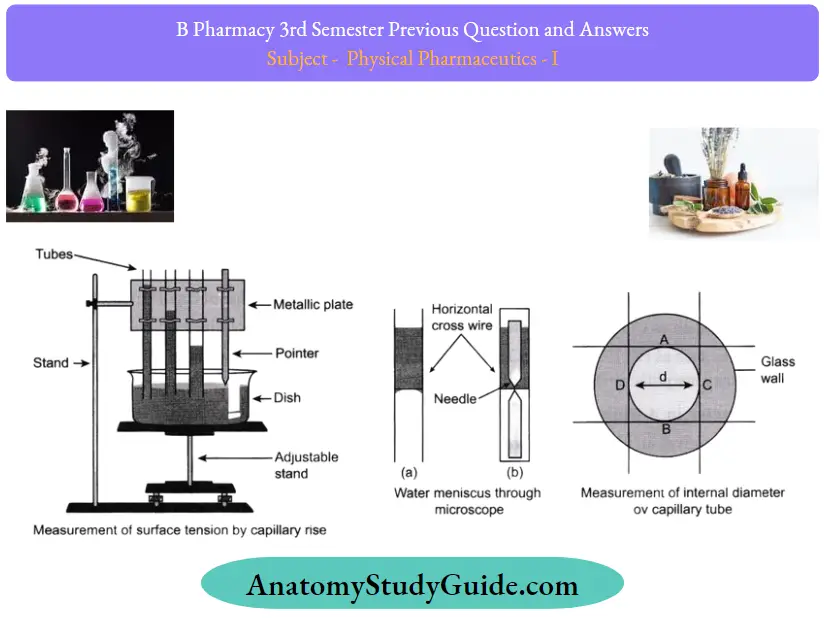
Tension Theory
The surface tension of water is given by the formula
T = r(r+h/3)ρg2cosθ
Tension Procedure
1. Arranging the apparatus
- Place the adjustable height stand on the table and make its base horizontal by leveling the screws.
- Take dirt and grease-free water in an open dish with a fat bottom and put it on top of the stand.
- Take three capillary tubes of different radii.
- Clean the tubes and dry them and then clamp them to a metallic plate to increase the radius. Clamp a pointer after the third capillary tube.
- Clamp the horizontal handle ofthe metallic plate in a vertical stand so that the capillary tube and the pointer become vertical.
- Adjust the height of the metallic plate that the capillary tubes dip in the water in an open dish.
- Adjust the position ofthe pointer such that the tip touches the water surface
2. Measurement of capillary rise
- Calculate the least count of the traveling microscope for vertical and horizontal scales.
- Raise the microscope to a suitable height pointed towards the capillary tube with a horizontal axis.
- Focus the microscope to the first capillary tube.
- Make the horizontal cross wire touch the central part of the concave meniscus seen convex through the microscope
- Note the reading of the microscope on the vertical scale.
- Move the microscope horizontally and bring it in front of the second capillary tube.
- Lower the microscope and repeat steps 11 and 12
- Likewise, repeat steps 11 and 12 for the third capillary
- Lower the stand for the pointer tip to be visible.
- Move the microscope horizontally and bring it in front of the pointer.
- Lower the microscope and make the horizontal cross-wire touch the tip of the pointer
3. Measurement of the internal diameter of the capillary tube:
- Place the first capillary tube horizontally on the adjustable stand.
- Focus the microscope on the end dipped in water. A white circle with a green strip will be visible.
- Make the horizontal cross-wire touch the inner circle at point A.
- Put the values h and r for each capillary tube separately and find the values of T using the following formula:
T = r(h + r/3)ρg2cosθ
Find the mean value of the obtained T values as follows:
T = T1+T2+T3
= ____________ dynes cm-1
Question 9. State and explain the distribution law. Mention its limitations and applications.
Answer:
Nemst gave a generalization governing the distribution of a solute between two non-miscible solvents. This is called as Nemst distribution law.
Distribution law Statement:
If a solute X distributes itself between the two immiscible solvents A and B at constant temperature and X is in the same molecular condition in both solvents,
Then
Concentration of X in A / Concentration of X in B = KD
IF Cl denotes the concentration of the solute in the solvent,
A and C2 denote the concentration in solvent B, then it is expressed as Cl / C2 = KD
KD = Distribution co-efficient or partition co-efficient or distribution ratio
Distribution law Limitations:
- Dilute solutions: The cone of the solute must be low in two solvents. This law does not hold good when the concentration is high.
- Constant temperature: Temperature should be kept constant throughout the experiment since solubility is dependent on temperature.
- Same molecular state: Solute must be in the same molecular state in both solvents.
- Equilibrium concentration: This is achieved by shaking the mixtures for a longer time.
- Non-miscibility of solvents: So, the solvents are to be allowed for separation for a sufficient time
Distribution law Applications:
1. Solvent extraction:
- This is the process used for the separation of organic substances from aqueous solution is shaken with an immiscible organic solvent such as ether in a separating funnel.
- The distribution ratio is in favor of ether, most of the organic substances pass into the ethereal layer.
- The ethereal layer is separated and ether is distilled off. Organic substance is left behind.
2. Partition chromatography:
- A paste of the mixture is applied at the top of a column of silica soaked in water. Another immiscible solvent is allowed to flow down the column.
- Each component of the mixture is portioned between the stationary liquid phase and the mobile liquid phase.
- The various components of the mixture are extracted by hexane in order of their distribution coefficients.
3. Determination of solubility:
Suppose the solubility of iodine in benzene is to be determined. Iodine is shaken with water and benzene. At equilibrium concentrations of iodine in benzene and water are found experimentally and the value of the distribution coefficient is calculated
Cb / Cw = Kd
Sb / Sw = Kd
Sb = Solubility in benzene
Sw = Solubility in water
4. Determination of dissociation:
Suppose a substance X is dissociated in an aqueous layer and exists as single molecules in ether. If x is the degree of dissociation the distribution law is modified as
Cl / C2 (1-x) = K
Cl = concentration of X in benzene
C2= concentration of X in the aqueous layer
The value of X can be determined from conductivity measurements, while C1 and C2 are found experimentally. Thus the value of K can be calculated. Using this value of K, the value of x for any other concentrations of X can be determined.
Question 10. Define interfacial tension. Explain in detail any one method for the determination of the interfacial tension.
Answer:
The interfacial tension (y) is the net force per unit length that is exerted on the contact surface between one phase (solid or liquid) and another (solid, liquid, or gas).
- The net force is vertical to the contact surface and is directed towards the interior of the phases.
- When one of the phases is a gas it is usually called surface tension.
- The phases in contact are immiscible, that is, they cannot dissolve together to form a solution.
- The region of contact between the phases is a geometric separation surface called an interface. Interfacial tension is due to intermolecular forces present at the interface.
- Interfacial tension plays an important role in many interfacial phenomena and processes, such as emulsion production and oil production.
DU NOUY method:
It utilizes the interaction of a platinum ring with the surface of the liquid. The ring is submerged below the interface by moving the stage where the liquid container is placed. After immersion, the stage is gradually lowered, and the ring pulls up the meniscus of the liquid. Eventually this meniscus tears from the ring. Before this event, the volume (and thus the force exerted) of the meniscus passes through the maximum value and begins to drop before the actual tearing event.
Steps:
It utilizes the interaction of a platinum ring with the surface of the liquid. The ring is submerged below the interface by moving the stage where the liquid container is placed. After immersion, the stage is gradually lowered, and the ring pulls up the meniscus of the liquid. Eventually this meniscus tears from the ring.
Before this event, the volume (and thus the force exerted) of the meniscus passes through the maximum value and begins to drop before the actual tearing event
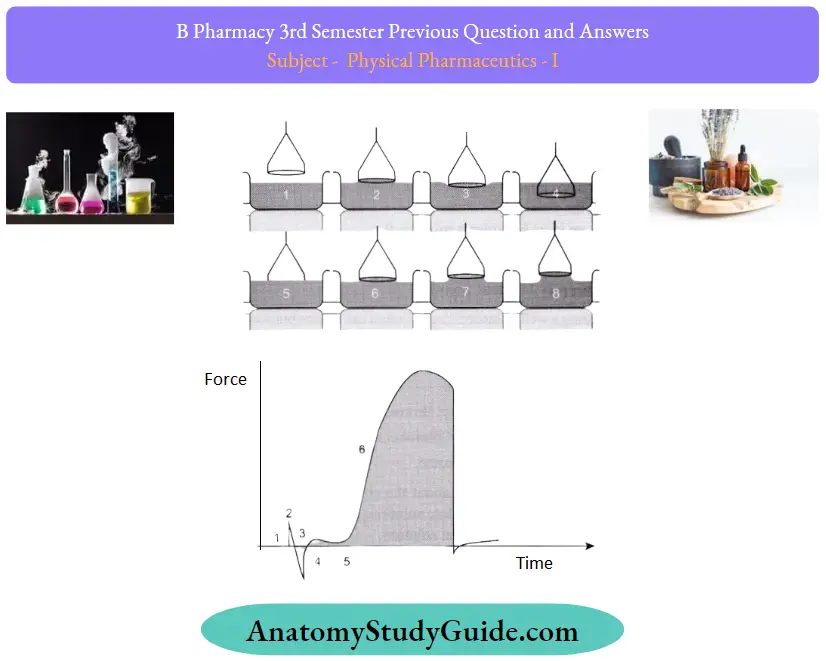
The different stages of the experiment can be identified
- The ring is above the surface and the force is zeroed.
- The ring hits the surface and there is a slight positive force due to the adhesive force between the ring and the surface.
- The ring must be pushed through the surface (due to surface tension) which causes a small negative force.
- The ring breaks the surface and a small positive force is measured due to the supporting wires of the ring.
- When lifted through the surface the measured force starts to increase.
- The force keeps increasing until
- The maximum force is reached.
- After the maximum, there is a small decrease in the force until the lamella breaks (or the ring is pushed back below the surface).
The calculation of the surface or interfacial tension by this technique is based on the measurement of this maximum force. The depth of immersion of the ring and the level to which the ring is raised when it experiences the maximum pull are irrelevant to this technique. The original calculations are based on the ring with the infinite diameter (or wire) and do not consider the excess liquid that is pulled up due to the proximity of one side of the ring to the other. Nowadays correction factors are routinely used. For the utilization of the correction factor, the density of the liquid must be known
Question 11. Define azeotropic mixtures. With the help of a neat diagram explain in detail the fractional distillation process.
Or
Write the principle and workings of the Dunouy tensiometer
Answer:
An azeotropic mixture is a mixture of substances that have the same concentration of vapor and fluid phases. It is a mixture that contains two or more liquids. A zeotropic mixture has constant or same boiling points and the mixture’s vapor will also have the same composition as the liquid.
Fractional Distillation Procedure:
Few fractional distillation apparatuses are required for the process. It includes a distilling flask, condenser, receiver, fractionating column, thermometer, and heat source.
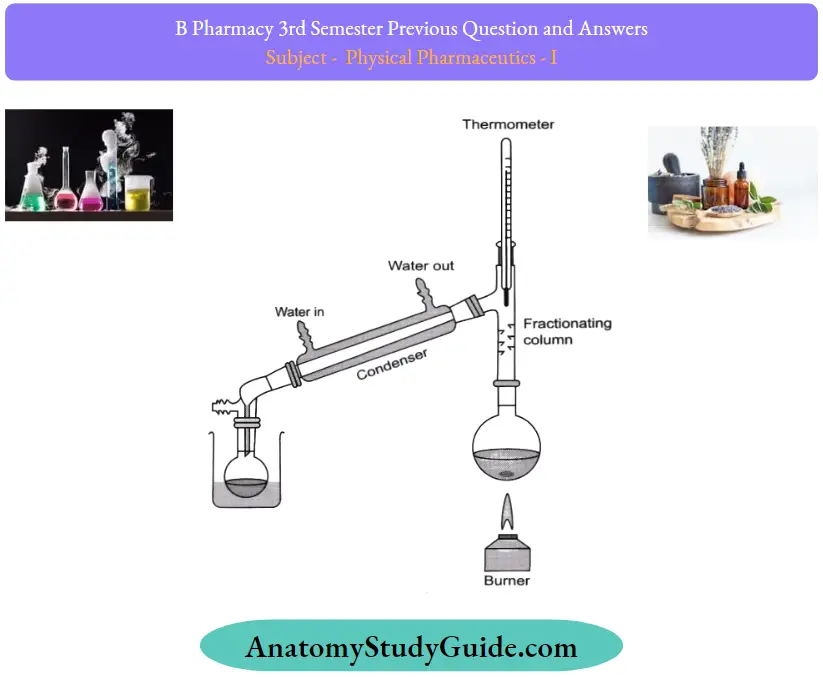
After setting up the apparatus, a mixture of two miscible liquids A and B is taken where A has more volatility than substance B. The solution is added to the distilling flask while the fractionating column is connected at the tip of the flask. Heat is applied which increases the temperature slowly. The mixture then starts to boil and vapors start rising in the flask. The vapors are from the volatile component A. The vapors then start moving through the fractionating column into the condenser where it is cooled down to form a liquid which is collected in the receiver. Throughout the process, vaporization and condensation take place repeatedly until the two mixtures are separated.
Fractional Distillation Applications:
- Fractional distillation is used for the purification of water as well as separating acetone and water.
- Fractional distillation is used in several industries like oil refineries and chemical plants mainly for purification and separation of many organic compounds.
- Fractional distillation is also used for the separation of(liquefied) air. Components like liquid nitrogen and oxygen as well as concentrated argon are obtained.
- Distillation is used in the production of high-purity silicon from chlorosilanes. The silicon is widely used in semiconductors.
Question 12. Define optical rotation. Discuss in detail the working of the polarimeter.
Answer:
Optical rotation is the angle through which the plane of polarization is rotated when polarized light passes through a layer of a liquid. Optical rotation is the effect that is determined by the concentration of chiral molecules and their molecular structure in a substance.
Every optical active substance has its specific rotation. A polarimeter consists of a light source, two polarizing prisms or filters, a liquid cell placed between the two polarizers, an eyepiece, and a protractor for measuring the angular rotation of light by the solution
Optical rotation Construction:
- The polarimeter is made up of two Nicol prisms (the polarizer and the analyzer). The polarizer is fixed and the analyzer can be rotated.
- The prisms may be thought of as slits SI and S2. The light waves may be considered to correspond to waves in the string. The polarizer SI allows only those light waves that move in a single plane.
- This causes the light to become plane-polarized. When the analyzer is also placed in a similar position it allows the light waves coming from the polarizer to pass through it.
- When it is rotated through the right angle no waves can pass through the right angle and the field appears to be dark.
- If a glass tube containing an optically active solution is placed between the polarizer and analyzer the light now rotates through the plane of polarization through a certain angle, the analyzer will have to be rotated in the same angle.
Optical rotation Working:
Polarimeters measure this by passing monochromatic light through the first of two polarising plates, creating a polarized beam. This first plate is known as the polarizer. This beam is then rotated as it passes through the sample.
After passing through the sample, a second polarizer, known as the analyzer, rotates either via manual rotation or automatic detection of the angle. When the analyzer is rotated such that all the light or no light can pass through, then one can find the angle of rotation which is equal to the angle by which the analyzer was rotated(θ) in the former case or (90-θ) in the latter case.

Operating principle of an optical polarimeter:
- Linear polarizer
- Linearly polarized light
- Sample tube containing molecules under study
- Optical rotation due to molecules
- Rotatable linear analyzer
- Detector
Question 13. Define interfacial tension. Explain in detail the working of the stalagmometer. Write its applications.
Answer:
Interfacial tension is the force of attraction between the molecules at the interface of two fluids. At the air/liquid interface, this force is often referred to as surface tension. The SI units for interfacial tension are milli-newtons per meter (mN/m). These are equivalent to the former units of dynes per centimeter (dyne/cm).
Stalagmometer
A stalagmometer is a device for investigating surface tension using the stalagmometric method. It is also called a stactometer or stalogometer. The device is a capillary glass tube whose middle section is widened. The volume of a drop can be predetermined by the design of the stalagmometer.
The lower end of the tube is narrowed to force the fluid to fall out of the tube as a drop. In an experiment, the drops of fluid flow slowly from the tube in a vertical direction. The drops hanging on the bottom of the tube start to fall when the volume of the drop reaches a maximum value that is dependent on the characteristics of the solution. At this moment, the weight of the drops is in an equilibrium state with the surface tension.
Based on Tate’s law:
The drop falls when the weight (mg) is equal to the circumference (2πr) multiplied by the surface tension (σ). The surface tension can be calculated provided the radius of the tube (r) and mass of the fluid droplet (m) are known. Alternatively, since the surface tension is proportional to the weight of the drop
The fluid of interest may be compared to a reference fluid of known surface tension (typically water):
In the equation, and a, represent the mass and surface tension of the reference fluid and m2 and <x, the mass and surface tension of the fluid of interest. If we take water as a reference fluid
- If the surface tension of water is known which is 72 dyne/ cm, we can calculate the surface tension of the specific fluid from the equation.
- The more drops we weigh, the more precisely we can calculate the surface tension from the equation. The stalagmometer must be kept clean for meaningful readings.
- There are commercial tubes for the stalagmometric method in three sizes: 2.5, 3.5, and 5.0 (ml).
- The 2.5-ml size is suitable for small volumes and low viscosity, 3.5 (ml) for relatively viscous fluids, and of 5.0 (ml) for large volumes and high viscosity.
- The 2.5-ml size is suitable for most fluids.
Stalagmometer Procedure:
1. Experimental procedure (drop-weighing method):
- Mount the clean and dry stalagmometer on the vertical stand.
- Weigh the mass of the weighing bottle mO.
- Fill the beaker with distilled water. Mount the tubing with the water pump on the top end of the gmometer. Immerse the bottom end of the stalagmometer in water and fill it up, such that the water level is above the wide part of the stalagmometer.
- Remove the water pump and collect 20 water drops into the weighing bottle.
- Weigh the mass of the weighing bottle with water and determine the mass of 20 drops.
- Empty the weighing bottle and stalagmometer, dry them, and prepare for the next measurement.
- Repeat steps 2-6 for liquids with unknown surface tension.
- Knowing the temperature in the laboratory, determine the water surface values, and calculate the surface tensions of studied liquids according to the equation
2. Experimental procedure (drop-counting method):
- Fill the stalagmometer up to the top mark with distilled water. Release water to the weighing bottle and count how many drops it takes to decrease the water level in the stalagmometer down to the bottom mark. Write down the number of drops nH20.
- Empty and dry the weighing bottle and stalagmometer, and prepare them for the next measurement.
- Repeat steps 1 and 2 for liquids with unknown surface tension.
- Write down the densities of studied liquids according to the notes on bottles, and the density of distilled water at the actual temperature in the laboratory. Using the equation, calculate the surface tension for all the studied liquids.
- Compare results obtained via the drop-weight method and drop-counting method
Stalagmometer Applications
Used to measure surface tension
Question 14. Write any two applications of buffers in the pharmacy.
Answer:
The buffer is a combination of an acid-base aqueous solution adjusted to an accurate pH value. The buffer is used to maintain a specific pH of the solution, it is used in the analysis and manufacture of various manufacturing processes such as food processing, electroplating, manufacture of medicines especially injection, ear drops, eye droplets, suspension, dissolution of tablets, etc.
- Improving Purity: Proteins are purified depending on the fact that amphoteric compounds are slightly soluble at their isoelectric point. For example, insulin precipitates from the aqueous solution in the pH range of 5 to 6. This technique is used for insulin purification.
- Increased Stability: Because of hydrolysis, many compounds are unstable in aqueous solutions. These solutions can be stabilized by regulating the pH. For example, the stability of vitamins is within a narrow range of pH only.
- Enhanced solubility: If the pH of the solution is not properly maintained, then the drug dissolution can precipitate. This principle applies in the dosage forms manufacturing, and some pharmaceutical ingredients and drugs dissolve only at a specific pH, hence, it is necessary to maintain the right pH of the solution.
- Optimizing Biological Activity: Enzymes contain most activity only on certain pH values. For example, at pH 1.5, there is a maximum activity of pepsin
Question 15. Define the term ligand with examples.
Answer:
A ligand is an ion or molecule, which donates a pair of electrons to the central metal atom or ion to form a coordination complex.
- Ligands can be anions, cations, and neutral molecules.
- Ligands act as Lewis bases (donate electron pairs) and central metal atoms are viewed as Lewis acid (electron pair acceptor).
- Occasionally ligands can be cations (NO+, N2H5+) and electron-pair acceptors.
Examples: Anionic ligands are F–, Cl–, Br–, I–, S–, CN–, NCS–, OH– , NH2– and neutral ligands are NH3, H2O, NO, CO
Question 16. Write the application of inclusion complexes with examples.
Answer:
An inclusion compound is a complex in which one chemical compound (the ‘host’) forms a cavity in which molecules of a second compound (‘guest’) are entrapped. These complexes generally do not have any adhesive forces working between their molecules and are therefore also known as no-bond complexes.
1. Channel lattice types:
The well-known starch-iodine solution is a channel type of complex, in which the iodine molecules are trapped within the spirals of the glucose molecules. The crystals of deoxycholic acid are arranged to form a channel into which the complexing molecule fits.
Inclusion complexes Applications
- Separation of optical isomers.
- In an analysis of dermatological creams.
2. Layer type:
Compounds such as clay, and montmorillonite can entrap hydrocarbons, alcohols, and glycols. They form alternate monomolecular layers of guest and host.
Layer type Applications:
- Very limited
- Useful for catalysis on account of a larger surface area
3. Clathrates:
Warfarin sodium is a clathrate of water and isopropyl alcohol. It is available as a white crystalline powder. During crystallization, certain substances form a cage-like lattice in which the coordinating compound is entrapped.
Clathrates Applications:
Stores gaseous volatile and toxic substances by the mechanism of clathrates.
4. Monomolecular inclusion complexes:
They involve the entrapment of a single guest molecule in the activity of one host molecule. Most of the host molecules are cyclodextrins.
Monomolecular Inclusion Applications:
- Enhanced solubility: The solubility of retinoic acid is increased to 160mg/liter by complexation with beta-cyclodextrin.
- Enhanced dissolution: The dissolution rate of drugs such as famotidine and Tolbutamide is increased by complexation with beta-cyclodextrin
- Enhanced stability: The stability of drugs such as aspirin, benzocaine, and ephedrine is improved when complexed with beta-cyclodextrin. The inclusion complex will also protect the rugs from preventing exposure of the functional groups to the exterior of the environment.
- Sustained release: Ethylate beta-cyclodextrin retard the release of drugs such as diltiazem and isosorbide dinitrate when complexed. The drug releases slowly for prolonged periods and provides a sustained effect
Question 17. Explain real solutions with examples.
Answer:
The solution which does not obey the Raoult law over the entire range of compositions is called as a nonideal or real solution. These solutions deviate from ideal behavior. Two main situations can cause non-ideal solutions to form:
Situation 1:
- Non-ideal solutions can form when forces of attraction between dissimilar molecules are weaker than between similar molecules. At this point, a heterogeneous (non-mixing) solution may still occur, but it is not always the case.
- The resulting solution has a larger enthalpy of solution than pure components of the solution, causing the process to be endothermic (heat is absorbed to move the reaction forward)
Example-1
A common example of a type of solution where this behavior is seen is in mixtures of carbon disulfide and acetone. Carbon disulfide is non-polar and acetone is polar.
- Since carbon disulfide is non-polar, the intermolecular attractions are
- London dispersion forces are known to be weak compared to other types of intermolecular forces.
- However, since acetone is polar, it has dipole-dipole forces, which are known to be very strong.
- Putting these two components together in a mixture results in dipole-induced dipole interactions.
- Since dipole-dipole-induced forces are not nearly as strong as the dipole-dipole interactions between acetone molecules in a pure substance, carbon disulfide-acetone mixtures are non-ideal solutions.
Situation 2:
- Non-ideal solutions can also form when intermolecular forces between dissimilar molecules are larger than those between similar molecules.
- In this case, interactions between these two types of molecules release more energy than is taken in to separate the two types of molecules.
- This energy is released in the form of heat, making the solution process exothermic.
Example-2
An example of this kind of non-ideal solution is a mixture of acetone and chloroform.
Activity Coefficients:
The chemical activity of a compound corresponds to the active concentration of that particular compound. However, due to intermolecular forces, we know this is not the case; therefore, we introduce an activity coefficient, labeled y, as a unit-less correctional factor. This coefficient takes into account the non-ideal characteristics of a mixture and it is between 0 and 1
For example:
1. The relationship between the activity of a component and its concentration for ideal mixtures is defined by
al = C pure …………. (1)
2. While the same relationship for real (non-ideal) mixtures is defined as follows
al = γ C pure …………… (2)
- If the interactions attract each other: γ < l
- If the interactions repel one another: γ > l
- If there are no interactions: γ = l
Question 18. Discuss in detail the electrometric determination of pH.
Answer:
One of the most widely accepted methods for hydrogen ion determination (pH) is the electrometric method. This method is highly accurate and used in laboratory work and by researchers. The accuracy of the pH value is 0.1 to 0.0001.
Apparatus for pH Determination:
- The apparatus required for the determination of water pH are:
- pH meter that is suitable for field as well as laboratory analysis. These can have either one or two electrodes.
- Distilled Water
- Standard Buffer solutions with pH of 4, 7, and 10.
- The solutions of known standards can be used.
- Thermometer that can read 77 ± 18°C to the nearest value of 0.1 degree Celsius
- Glass stirring rod
- Minimum capacity scale to read up to 1.1 lb
Procedure for pH Determination:
The procedure of determining of pH of water involves the following steps:
- The water sample is properly mixed and stirred using a glass rod
- By using a watch glass, a sample of water equal to 40ml (5ml more or less) is added to the beaker. The temperature of the water is allowed to stabilize by placing the sample stand for 1 hour.
- In between this time stirring can be done.
- After 1 hour, the temperature of the water is measured and this temperature is adjusted in the pH meter. Hence the pH meter shows a temperature similar to that of the sample.
- All these adjustments to the apparatus must be performed and fixed before the test is conducted.
- There are some pH meters with automatic temperature controls. In such cases, the instructions provided by the manufacturer have to be followed.
The standard solutions are used to standardize the pH meter. Here also the temperature is adjusted as mentioned above procedure.
- Next, into the water sample, the electrodes are inserted. The beaker is turned and adjusted so that there is good contact between the electrodes and the water.
- Before starting the reading, the electrodes have to be placed in the solution for more than 30 seconds.
- This period is required for the proper stabilizing of the meter to have a proper reading. In pH meters that have an automatic reading system, a signal will be provided to tell that the meter is stabilized. Once the reading is shown, it must be read to the nearest tenth ofthe whole number.
- If the value shows to 1 00th place then it has to be rounded off. The tenth place digit is left if the 100th place is less than 5.
- For values greater than 5 after decimal, it is rounded to 1 unit. If the 100th place is equal to 5, the nearest even number is taken as a rounded value. The apparatus must be maintained after each use.
- The electrodes used are washed thoroughly with distilled water. If there is any form of film around the electrodes, it has to be cleared.
- Wiping of the electrodes must be avoided as this will result in polarization which will result in a slow response to the experiment.
Precautions and Tips:
- The pH meter can be standardized by measuring the 7-pH buffer solution or any other solution of standard pH. Sometimes, the manufacturer of the pH meter may suggest other methods of standardizing, which too have to be followed.
- The electrodes have to be inserted into the water so that it does not touch the bottom of the beaker. Bottom contact with damage may cause damage to the electrodes.
- Any cause of slow response due to the polarization can be solved by washing the electrodes thoroughly.
- The periodic check has to be conducted to check the electrodes
- During the electrode storage, they have to be kept moist. And also follow the instructions of the manufacturer
Question 19. Write a note on the solubility of gases in liquids.
Answer:
- The solubility of a gas in a liquid depends on the following factors such as temperature, partial pressure of the gas on the liquid, the nature of the solvent, and the nature of the gas.
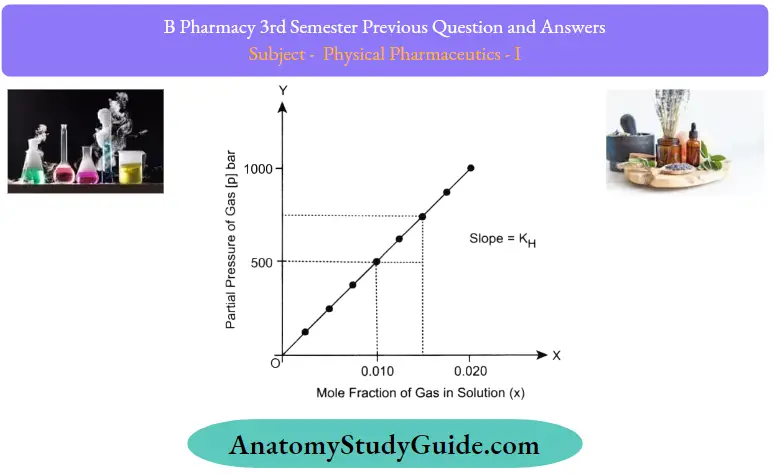
- Henry’s law states that the solubility of the gas in liquid is directly proportional to the partial pressure f that gas at a constant temperature. It was formulated by William Henry in 1803
- Where K = Henrys constant
- If the solubility is based on the molar fraction of a gas in the solution then Henry’s law states that the “Partial pressure of the vapour phase is proportional to the molar fraction of the gas in the solution and is expressed as
S ∝ P
S = KP
Where K= Henry constant
The concentration of dissolved gas depends on the partial pressure of the gas. If partial pressure is low then the concentration of gas will also decrease. If the partial pressure is doubled, the number of collisions with the surfaces will also be doubled and eventually, it will produce more dissolved gas. Henry’s law applied to increase the solubility of carbon dioxide in carbonated drinks and soda water, the bottle is sealed under high pressure. But there are still limitations of Henry’s law.
Because Henry’s law is applicable only under the following conditions when:
- There is no chemical reaction between gas and solvent.
- The gas should not undergo dissociation in the solvent.
- The pressure of the gas is not too high.
- The temperature is not too low.
Question 20. Write a note on surface-free energy.
Answer:
Surface tension maintains the surface area of a liquid to a minimum value. The surface area can be increased provided some work is being done against the force at the surface. Surface-free energy is defined as the work required to increase the area of a liquid by I sqm. The derivation to estimate the surface free energy is as follows
Measuring the surface energy with contact angle measurement:
The most common way to measure surface energy is through contact angle experiments. In this method, the contact angle of the surface is measured with several liquids, usually water and diiodomethane. Based on the contact angle results and knowing the surface tension of the liquids, the surface energy can be calculated. In practice, this analysis is done automatically by a contact angle meter.
There are several different models for calculating the surface energy based on the contact angle readings.
- The most commonly used method is OWRK which requires the use of two probe liquids and gives out as a result the total surface energy as well as divides it into polar and dispersive components.
- The contact angle method is the standard surface energy measurement method due to its simplicity, applicability to a wide range of surfaces and quickness. The measurement can be fully automated and is standardized.
- Measuring the surface energy of a solid- other methods The surface energy of a liquid may be measured by stretching a liquid membrane (which increases the surface area and hence the surface energy).
- In that case, to increase the surface area of a mass of liquid by an amount, δA, a quantity of work, γ δA, is needed (where y is the surface energy density of the liquid).
- However, such a method cannot be used to measure the surface energy of a solid because stretching of a solid membrane induces elastic energy in the bulk in addition to increasing the surface energy.
The surface energy of a solid is usually measured at high temperatures. At such temperatures the solid creeps and even though the surface area changes, the volume remains approximately constant.
If γ is the surface energy density of a cylindrical rod of radius r and length l at high temperature and a constant uniaxial tension P, then at equilibrium, the variation of the total Helmholtz free energy vanishes and we have
δF = -Pδl + γ δA = 0
y = Pδl /δ a where F is the Helmholtz free energy and the surface area of the rod:
Also, since the volume (V) of the rod remains constant, the variation (SV) of the volume is zero, that is,
V = πr²l is constant
δV = 2πrlδr + πr²δl
Therefore, the surface energy density can be expressed as
γ = PI /πr (l-2r)
The surface energy density of the solid can be computed by measuring P, r, and 1 at equilibrium.
This method is valid only if the solid is isotropic, meaning the surface energy is the same for all crystallographic orientations. While this is only strictly true for amorphous solids (glass) and liquids, isotropy is a good approximation for many other materials. In particular, if the sample is polygranular (most metals) or made by powder sintering (most ceramics) this is a good approximation.
In the case of single-crystal materials, such as natural gemstones, anisotropy in the surface energy leads to faceting. The shape of the crystal (assuming equilibrium
Leave a Reply