The Gastrointestinal Tract Oesophagus
Normal Structure:
Table of Contents
- The oesophagus is a muscular tube extending from the pharynx to the stomach. In an adult, this distance measures 25 cm. However, from the clinical point of view, the distance from the incisor teeth to the gastro-oesophageal (GE) junction is about 40 cm.
- The region of the proximal oesophagus at the level of cricopharyngeus muscle is called the upper oesophageal sphincter, while the portion adjacent to the anatomic gastro-oesophageal junction is referred to as the lower oesophageal sphincter.
Read And Learn More: Systemic Pathology Notes
- Histologically, the wall of the oesophagus consists of mucosa, submucosa, muscularis propria and adventitia/serosa.
- The mucosa is composed of non-keratinising stratified squamous epithelium overlying lamina propria except at the lower end for a distance of 0.5 to 1.5 cm.
- The basal layer of the epithelium may contain some melanocytes, argyrophilic cells and Langerhans’ cells. At the lower end of the oesophagus, there is a sudden change from stratified squamous epithelium to mucin-secreting columnar epithelium; this is called the junctional mucosa.
- The submucosa consists of loose connective tissue with a sprinkling of lymphocytes, plasma cells, and occasional eosinophils and mast cells. Mucus-producing glands are scattered throughout the submucosa.
- The muscular propria is composed of 2 layers of smooth muscle an inner circular coat and an outer longitudinal coat.
- The proximal portion of the oesophagus contains skeletal muscle fibres from the cricopharyngeus muscle. The parasympathetic nerve supply by the vagus nerve is in the form of extrinsic and intrinsic plexuses.
- The adventitia/serosa is the outer covering of the oesophagus. Serosa is present in the intraabdominal part of the oesophagus only, while elsewhere the peri oesophageal adventitia covers it.
- The major functions of the oesophagus are swallowing by peristaltic activity and preventing the reflux of gastric contents into the oesophagus.
- Oesophageal diseases discussed here are congenital anomalies, muscular dysfunctions, inflammatory diseases, and tumours.
- Congenital Anomalies
- Congenital anomalies of the oesophagus are uncommon and are detected soon after birth.
- These include a few rare anomalies such as agenesis (congenital absence of oesophagus) which is incompatible with life, duplication of oesophagus (double oesophagus) and congenital stenosis (i.e. fibrous thickening of the oesophageal wall and atrophy of the muscularis propria). However, oesophageal atresia and trachea-oesophageal fistula are relatively more common.
Oesophageal Atresia And Tracheo-Oesophageal Fistula: In About 85% of cases, congenital atresia of the oesophagus is associated with tracheo-oesophageal fistula, usually at the level of the tracheal bifurcation. For survival, the condition must be recognised and corrected surgically within 48 hours of the birth of the newborn.
Clinically, the condition is characterised by regurgitation of every feed, hypersalivation, attacks of cough and cyanosis. Death usually results from asphyxia, aspiration pneumonia and fluid-electrolyte imbalance.
Morphologically, the condition is recognised by a cord-like non-canalised segment of the oesophagus having a blind pouch at both ends.
Muscular Dysfunctions:
These are disorders in which there is motor dysfunction of the oesophagus, manifested clinically by dysphagia. These include achalasia, hiatus hernia, oesophageal diverticula, and webs and rings.
Achalasia (Cardiospasm):
Achalasia of the oesophagus is a neuromuscular dysfunction due to which the cardiac sphincter fails to relax during swallowing and results in progressive dysphagia and dilatation of the oesophagus (mega-oesophagus).
Etiology: There is a loss of intramural neurons in the wall of the oesophagus.
Most cases are of primary idiopathic achalasia i.e. congenital. Secondary achalasia may occur from some other causes which include Chagas’ disease (an epidemic parasitosis with Trypanosoma cruzi), infiltration into the oesophagus by gastric carcinoma or lymphoma, certain viral infections, and neurodegenerative diseases.
Morphologic Features: There is dilatation above the short contracted terminal segment of the oesophagus.
Muscularis propria of the wall may be of normal thickness, hypertrophied as a result of obstruction, or thinned out due to dilatation. Secondary oesophagitis may supervene and cause oesophageal ulceration and haematemesis.
Hiatus Hernia:
Hiatus hernia is the herniation or protrusion of part of the stomach through the oesophageal hiatus of the diaphragm. Oesophageal hiatal hernia is the cause of diaphragmatic hernia in 98% of cases.
The condition is diagnosed radiologically in about 5% of apparently normal asymptomatic individuals.
In symptomatic cases, especially the elderly women, the clinical features are heartburn (retrosternal burning sensation) and regurgitation of gastric juice into the mouth, both of which are worsened due to heavy work, lifting weights and excessive bending.
Etiology: The basic defect is the failure of the muscle fibres of the diaphragm that form the margin of the oesophageal hiatus. This occurs due to shortening of the oesophagus which may be congenital or acquired. The congenitally short oesophagus may be the cause of hiatus hernia in a small proportion of cases.
More commonly, it is acquired due to secondary factors which cause fibrous scarring of the oesophagus as follows:
- Degeneration of muscle due to ageing.
- Increased intra-abdominal pressure such as in pregnancy, abdominal tumours etc.
- Recurrent oesophageal regurgitation and spasm causing inflammation and fibrosis.
- An increase in fatty tissue in obese people causes decreased muscular elasticity of the diaphragm.
Morphologic Features:
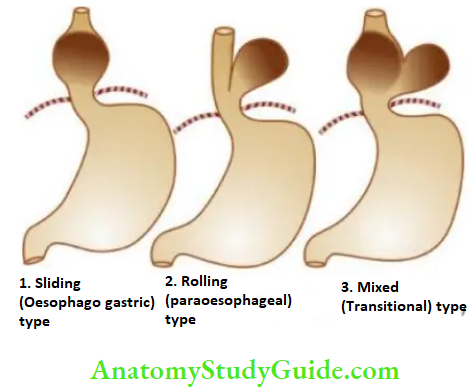
There are 3 patterns in hiatus hernia:
- Sliding or oesophagogastric hernia is the most common, occurring in 85% of cases.
- The herniated part of the stomach appears as a supradiaphragmatic bell due to sliding up on both sides of the oesophagus.
- A rolling or para-oesophageal hernia is seen in 10% of cases. This is a true hernia in which the cardiac end of the stomach rolls up para-oesophageal, producing an intrathoracic sac.
- Mixed or transitional hernia constitutes the remaining 5% of cases in which there is a combination of sliding and rolling hiatus hernia.
Oesophageal Diverticula:
Diverticula are the outpouchings of the oesophageal wall at the point of weakness. They may be congenital or acquired.
Congenital diverticula occur either at the upper end of the oesophagus or at the bifurcation of the trachea.
Acquired diverticula may be of 2 types:
Pulsion (Zenker’s) type It is seen in the region of the hypopharynx and occurs due to oesophageal obstruction such as due to chronic oesophagitis, carcinoma etc. The mucosa and submucosa herniate through the weakened area or through a defect in the muscular propria.
Traction type It occurs in the lower third of the oesophagus from contraction of fibrous tissue such as from pleural adhesions, scar tissue of healed tuberculous lesions in the hilum, silicosis etc.
Complications of diverticula include obstruction, infection, perforation, haemorrhage and carcinoma.
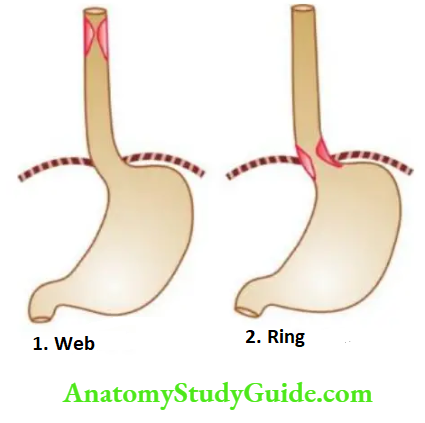
Oesophageal Webs And Rings:
Radiological shadows in the oesophagus resembling ‘webs’ and ‘rings’ are observed in some patients complaining of dysphagia.
WEBS These are located in the upper oesophagus, seen more commonly in adult women, and associated with dysphagia, iron deficiency anaemia and chronic atrophic glossitis (Plummer-Vinson syndrome).
Rings: Those located in the lower oesophagus, not associated with iron-deficiency anaemia, nor occurring in women alone, are referred to as ‘Schatzki’s rings’.
Morphologic Features: The rings and webs are transverse folds of mucosa and submucosa encircling the entire circumference, or are localised annular thickenings of the muscle. These give characteristic radiological shadows.
Inflammatory Diseases:
Inflammation of the oesophagus, or oesophagitis, occurs most commonly from reflux, although a number of other clinical conditions and infections may also cause oesophagitis.
Reflux (Peptic) Oesophagitis:
Reflux of gastric juice is the commonest cause of oesophagitis.
Pathogenesis: Gastro-oesophageal reflux, to an extent, may occur in normal healthy individuals after meals and in early pregnancy. However, in some clinical conditions, the gastroesophageal reflux is excessive, resulting in inflammation of the lower oesophagus.
These conditions are as under:
- Sliding hiatus hernia
- Chronic gastric and duodenal ulcers
- Nasogastric intubation
- Persistent vomiting
- Surgical vagotomy
- Neuropathy in alcoholics, diabetics
- Oesophagogastrostomy.
Morphologic Features: Endoscopically, the demarcation between normal squamous and columnar epithelium at the junctional mucosa is lost.
The affected distal oesophageal mucosa is red, erythematous, friable and bleeds on touch. In advanced cases, there are features of chronic disease such as nodularity, strictures, ulcerations and erosions.
Microscopically, the reflux changes in the distal oesophagus include basal cell hyperplasia and deep elongation of the papillae touching close to the surface epithelium. Inflammatory changes vary according to the stage of the disease.
In the early stage, mucosa and submucosa are infiltrated by some polymorphs and eosinophils; in the chronic stage, there is lymphocytic infiltration and fibrosis of all the layers of the oesophageal wall.
Barrett Oesophagus:
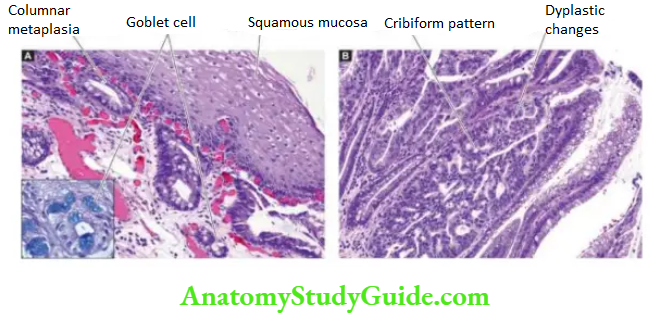
This is a condition in which, following reflux oesophagitis, stratified squamous epithelium of the lower oesophagus is replaced by columnar epithelium (columnar metaplasia).
The condition is seen more commonly at a later age and is caused by factors producing gastro-oesophageal reflux disease (described above).
Barrett’s oesophagus is a premalignant condition evolving sequentially from Barrett epithelium (columnar metaplasia with goblet cells) to dysplasia to carcinoma in situ and finally to oesophageal adenocarcinoma.
Morphologic Features: Endoscopically, the affected area is red and velvety. Hiatus hernia and peptic ulcer at the squamocolumnar junction (Barrett’s ulcer) are frequently associated.
Microscopically, the changes are as under:
- A most common finding is the replacement of squamous epithelium by metaplastic columnar cells, along with goblet cells and Paneth cells (intestinal metaplasia. Based on the length of the oesophagus involved, it is further divided into long segment (=3 cm) and short segment (<3 cm) involvement.
- Intestinal metaplasia may be accompanied by dysplastic changes of the columnar epithelium or glands ranging from low to high grade.
- There may be changes in a peptic ulcer due to the presence of fundic gastric glands, or cardiac mucous glands.
- Inflammatory changes, acute or chronic, are commonly accompanied.
- There may be the development of stricture in the long segment.
High-grade dysplasia may progress to invasive adenocarcinoma of the oesophagus in up to 20% of cases the reported risk of development of adenocarcinoma in Barrett’s oesophagus is at the rate of 0.5% per year after diagnosis. Therefore, surveillance endoscopic biopsies are advised.
Other Causes Of Oesophagitis
These include infective and non-infective causes:
Infective Causes:
A number of opportunistic infections in immunosuppressed individuals can cause oesophagitis.
Some of these agents are as follows:
- Candida (Monilial) oesophagitis
- Herpes simplex (Herpetioesophagitis
- Cytomegalovirus
- Tuberculosis.
Non-infective Causes:
- Eosinophilic oesophagitis caused by radiation, corrosives
- Intake of certain drugs (anticholinergic drugs, doxycycline, tetracycline)
- Ingestion of hot, irritating fluids
- Crohn’s disease
- Various vesiculobullous skin diseases.
Oesophageal Tumours:
Benign tumours of the oesophagus are uncommon and small in size (less than 3 cm).
The epithelial benign tumours project as intraluminal masses arising from squamous epithelium (squamous cell papilloma), or from columnar epithelium (adenoma).
The stromal or mesenchymal benign tumours are intramural masses such as leiomyoma and others like lipoma, fibroma, neurofibroma, rhabdomyoma, lymphangioma and haemangioma.
For all practical purposes, malignant tumours of the oesophagus are carcinomas because sarcomas such as leiomyosarcoma and fibrosarcoma occur with extreme rarity.
Oesophageal Cancer:
Carcinoma of the oesophagus is diagnosed late after symptomatic oesophageal obstruction (dysphagia developed and the tumour has transgressed the anatomical limits of the organ. The tumour occurs more commonly in men over 50 years of age.
Prognosis is dismal: With standard methods of therapy (surgical resection and/or irradiation), 70% of the patients die within one year of diagnosis. The five-year survival rate is 5-10%.
Etiology:
A number of conditions and factors predispose to the development of oesophageal cancer:
1. Diet and personal habits
- Heavy smoking
- Alcohol consumption
- Intake of foods contaminated with fungus
- Nutritional deficiency of vitamins and trace elements.
2. Oesophageal disorders
- Oesophagitis (especially Barrett oesophagus in adenocarcinoma)
- Achalasia
- Hiatus hernia
- Diverticula
- Plummer-Vinson syndrome.
3. Other factors
- Race more common in the Chinese and Japanese than in Western races; more frequent in blacks than whites.
- Family history association with tylosis (keratosis palmaris et plantaris).
- Genetic factors predisposition with coeliac disease, epidermolysis bullosa, tylosis.
- HPV infection is a recent addition to etiologic factors.
At the molecular level, abnormality of the p53 tumour suppressor gene has been found associated with a number of above risk factors, notably with consumption of tobacco and alcohol, and in cases having proven Barrett’s oesophagus.
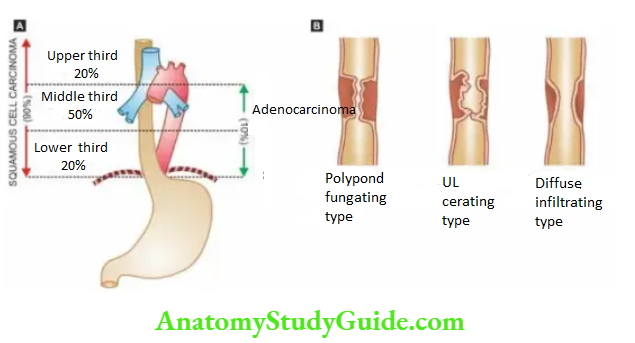
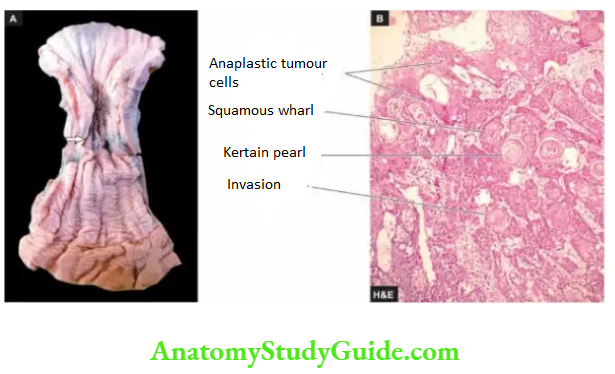
Morphologic Features: Carcinoma of the oesophagus is mainly of 2 types squamous cell and adenocarcinoma. The sites of predilection for each of these 2 forms.
Squamous Cell Carcinoma: Squamous cell or epidermoid carcinoma comprises 90% of primary oesophageal cancers.
It is exceeded in incidence by carcinoma colon, rectum and stomach among all the gastrointestinal cancers.
The disease occurs in the 6th to 7th decades of life and is more common in men than women. The sites of predilection are the three areas of oesophageal constrictions.
Half of the squamous cell carcinomas of the oesophagus occur in the middle third, followed by the lower third, and the upper third of the oesophagus in that order of frequency.
Grossly, 3 types of patterns are recognised:
Polypoid fungating type is the most common form. It appears as a cauliflower-like friable mass protruding into the lumen.
Ulcerating type is the next common form. It looks grossly like a necrotic ulcer with everted edges.
Diffuse infiltrating type appears as an annular, stenosis narrowing of the lumen due to infiltration into the wall of the oesophagus.
Microscopically, the majority of the squamous cell carcinomas of the oesophagus are well-differentiated or moderately differentiated. Prickle cells, keratin formation and epithelial pearls are commonly seen.
However, non-keratinising and anaplastic growth patterns can also occur. An exophytic, slow-growing, extremely well-differentiated variant, verrucous squamous cell carcinoma, has also been reported in the oesophagus.
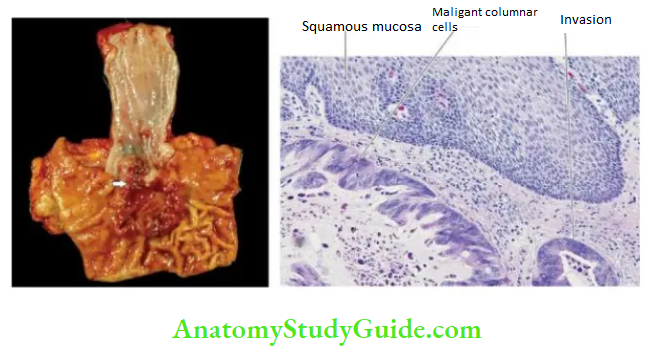
Adenocarcinoma: Adenocarcinoma of the oesophagus constitutes less than 10% of primary oesophageal cancer. It occurs predominantly in men in their 4th to 5th decades.
The common locations are lower and middle third of the oesophagus. These tumours have a strong and definite association with Barrett’s oesophagus in which there are foci of gastric or intestinal type of epithelium.
Grossly, oesophageal adenocarcinoma appears as a nodular, elevated mass in the lower oesophagus and often extends through gastric cardia.
Microscopically, adenocarcinoma of the oesophagus can have 3 patterns:
- The intestinal type is the adenocarcinoma with a pattern similar to that seen in the adenocarcinoma of the intestine or stomach.
- Adenosquamous type is the pattern in which there is an irregular admixture of adenocarcinoma and squamous cell carcinoma.
- Adenoid cystic type is an uncommon variety and is akin to similar growth in the salivary gland i.e. a cribriform appearance in an epithelial tumour.
Adenocarcinoma of the oesophagus must be distinguished from adenocarcinoma of the gastric cardia. This is done by identifying normal oesophageal mucosa on the distal as well as proximal margin of the tumour.
Spread: Oesophageal cancer spreads locally as well as to distant sites.
1. Local spread This is the most important mode of spread and is of great importance for surgical treatment. Local spread may occur in the transverse as well as longitudinal direction.
The tumour may invade below into the stomach, above into the hypopharynx, into the trachea resulting in tracheo-oesophageal fistula, and may involve the larynx causing hoarseness.
The tumour may invade the muscular wall of the oesophagus and involve the mediastinum, lungs, bronchi, pleura and aorta.
2. Lymphatic spread Submucosal lymphatic permeation may lead to multiple satellite nodules away from the main tumour. Besides, the lymphatic spread may result in metastases to the cervical, para-oesophageal, tracheobronchial and subdiaphragmatic lymph nodes.
Haematogenous spread Blood-borne metastases from oesophageal cancer are rare, probably because the death occurs early due to the invasion of important structures by other modes of spread. However, metastatic deposits by haematogenous route can occur in the lungs, liver and adrenals.
Diseases of the Oesophagus:
- Oesophageal atresia and trachea-oesophageal fistula are more common congenital anomalies of the oesophagus.
- Achalasia (or cardiospasm) and hiatus hernia are important muscular dysfunctions of the oesophagus.
- Barrett’s oesophagus is intestinal metaplasia along with the presence of goblet cells in the squamous-lined oesophagus; it may be associated with dysplastic changes which may progress to adenocarcinoma.
- Common cancer of the oesophagus is squamous cell type, most common in the middle third of the oesophagus.
Stomach
Normal Structure:
The stomach is a ‘gland with cavity’, extending from its junction with the lower end of the oesophagus (cardio junction with the duodenum (pylorus).
The lesser curvature is the inner concavity on the right, while the greater curvature is the outer convexity on the left side of the stomach.
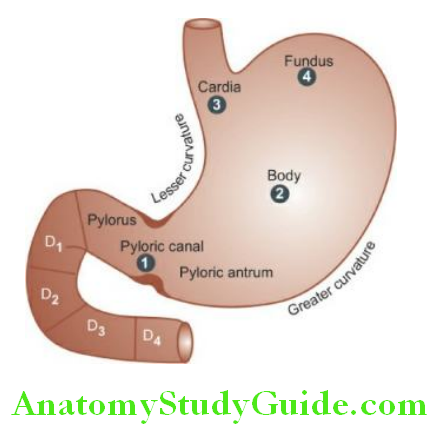
The stomach has 5 anatomical regions:
Cardia is the oesophagogastric junction and lacks the sphincter.
Fundus is the portion above the horizontal line drawn across the oesophagogastric junction.
The body is the middle portion of the stomach between the fundus and the pyloric antrum.
The pyloric antrum is the distal third of the stomach.
Pylorus is the junction of the distal end of the stomach with the duodenum. It has powerful sphincter muscle.
The mucosal folds in the region of the body and the fundus are loose (rugae), while the antral mucosa is somewhat flattened.
The gastric canal is the relatively fixed portion of the pyloric antrum and the adjoining lesser curvature; it is the site for numerous pathological changes such as gastritis, peptic ulcer and gastric carcinoma.
The stomach receives its blood supply from the left gastric artery and the branches of the hepatic and splenic arteries with widespread anastomoses.
Numerous gastric lymphatics which communicate freely with each other are also present. The innervation of the stomach is by the vagi and branches of the sympathetic which are connected with ganglia in the muscular and submucous layers.
Histologically, the wall of the stomach consists of 4 layers serosa, muscular, submucosa and mucosa.
1. Serosa is derived from the peritoneum which is deficient in the region of lesser and greater curvatures.
2. muscular consists of 3 layers of smooth muscle fibres the outer longitudinal, the middle circular and the inner oblique. Nerve plexuses and ganglion cells are present between the longitudinal and circular layers of muscle.
The pyloric sphincter is the thickened circular muscle layer at the gastroduodenal junction.
3. Submucosa is a layer of loose fibroconnective tissue binding the mucosa to the muscularis loosely and contains branches of blood vessels, lymphatics and nerve plexuses and ganglion cells.
4. Mucosa consists of 2 layers superficial and deep. Between the two layers is the lamina propria composed of a network of fibro collagenic tissue with a few lymphocytes, plasma cells, macrophages and eosinophils.
The mucosa is externally bounded by muscularis mucosae:
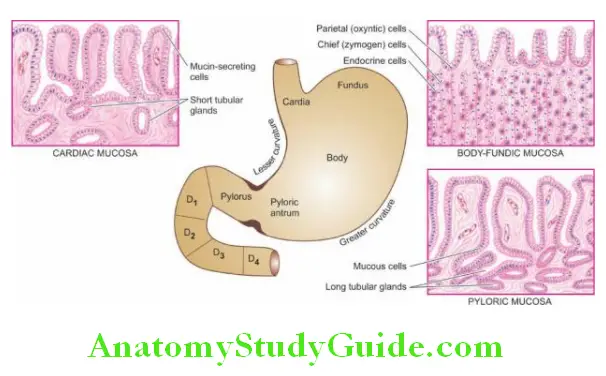
Superficial layer: It consists of a single layer of surface epithelium composed of regular, mucin-secreting, tall columnar cells with basal nuclei.
There is a rapid turnover of these cells. These dip down at places to form crypts (or pits or foveolae).
Cardiac mucosa is the transition zone between the oesophageal squamous mucosa and the oxyntic mucosa of the fundus and body with which it gradually merges.
Oxyntic mucosa lines both gastric fundus and body. Antral mucosa lines the pyloric antrum.
Deep layer: It consists of glands that open into the bottom of the crypts.
Depending upon the structure, these glands are of 3 types:
Glands of the cardia are simple tubular or compound tubular-racemose, lined by mucin-secreting cells. A few endocrine cells and occasional parietal and chief cells are also present.
Glands of the body-fundus are long, tubular and tightly packed which may be coiled or dilated.
There are 4 types of cells present in the glands of body-fundic mucosa: Parietal (Oxynticells are the most numerous and line the superficial (upper) part of the glands.
Parietal cells are triangular in shape and have dark-staining nuclei and eosinophilic cytoplasm. These cells are responsible for the production of hydrochloric acid in the gastric juice and the blood group substances.
Chief (Pepticells are the dominant cells in the deeper (lower) parts of the glands. Their basal nuclei are large with prominent nucleoli and the cytoplasm is coarsely granular and basophilic. These cells secrete pepsin from the gastric juice.
Mucin-secreting neck cells are small and fewer. These cells are present in the region of the narrow neck of the gastric glands i.e. at the junction of the glands with the pits.
Endocrine (Kulchitsky or Enterochromaffin) cells are widely distributed in the mucosa of all parts of the alimentary tract and are described later.
Glands of the pylorus are much longer than the body-fundic glands. These are simple tubular glands which are often coiled. They are lined mainly by small, granular, mucin-secreting cells resembling neck cells and occasional parietal cells but no chief cells.
Gastrin-producing G-cells are present predominantly in the region of anthropologic mucosa, with a small number of these cells in the crypts and Brunner’s glands of the proximal duodenum.
The secretory products of the gastric mucosa are the gastric juice and the intrinsic factor, required for absorption of vitamin B12.
Gastric juice consists of hydrochloric acid, pepsin, mucin and electrolytes like Na+, K+, HCO–3 and Cl-. Hydrochloric acid is produced by the parietal (oxyntic cells by the interaction of Cl- ions of the arterial blood with water and carbon dioxide in the presence of the enzyme, carbonic anhydrase.
The degree of gastric activity is correlated with the ‘total parietal cell mass’. Injection of histamine can stimulate the production of the acid component of the gastric juice, while the pepsin-secreting chief cells do not respond to histamine.
Physiologically, the gastric secretions are stimulated by the food itself.
Diseases of the stomach discussed in this chapter are congenital anomalies, inflammatory conditions (gastritis and peptic ulcer), and gastric tumours and tumour-like conditions. In the end, conditions producing haematemesis are summarised.
Congenital Anomalies
Pancreatic Heterotopia:
Heterotopic pancreatic tissue may present clinically as a gastric mass or may be an incidental finding. Symptomatic cases may present in newborns or later in life.
Grossly, it is seen as a mass projecting into the gastric lumen, generally in the region of the submucosa and less often in the muscular layer. In most cases, the mass is located in the region of the antrum or pylorus.
Microscopically, both normal mature pancreatic acinar and ductal tissue are seen. Islets are seen in about a third of cases.
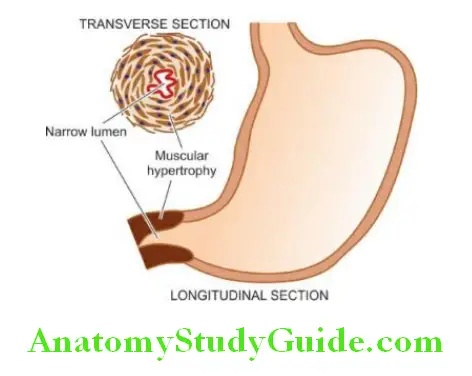
Pyloric Stenosis:
Hypertrophy and narrowing of the pyloric lumen occur predominantly in male children as a congenital defect (infantile pyloric stenosis).
The adult form is rarely seen, either as a result of the late manifestation of a mild congenital anomaly or may be an acquired type due to inflammatory fibrosis or invasion by tumours.
Etiology: Exact cause of congenital (infantile) pyloric stenosis is not known but it appears to have familial clustering and recessive genetic origin. The acquired (adult) pyloric stenosis is related to antral gastritis, and tumours in the region (gastric carcinoma, lymphoma, pancreatic carcinoma).
Morphologic Features: Grossly and microscopically, there is hypertrophy as well as hyperplasia of the circular layer of muscular in the pyloric sphincter accompanied by a mild degree of fibrosis.
Clinical Features: The patient, usually a first-born male infant 3 to 6 weeks old, presents with the following clinical features
- Vomiting, which may be projectile and occasionally contains bile or blood.
- Visible peristalsis usually noticed from the left to right side of the upper abdomen.
- Palpable lump, better felt after an episode of vomiting.
- Constipation.
- Loss of weight
Stomach: Normal Structure and Congenital Anomalies:
The stomach is a gland with a cavity. It has five anatomic zones: cardia, fundus, body, pyloric antrum and pylorus. Its wall consists of mucosa that varies in different zones, thick muscular and serosa.
Important congenital anomalies causing obstructive features are pancreatic heterotopia and pyloric stenosis.
Inflammatory Conditions:
Two important inflammatory conditions of the stomach are gastritis and peptic ulcer. Rarely, the stomach may be involved in tuberculosis, sarcoidosis and Crohn’s disease.
Gastritis:
The term ‘gastritis’ is commonly employed for any clinical condition with upper abdominal discomfort like indigestion or dyspepsia in which specific clinical signs and radiological abnormalities are absent.
The condition is of great importance due to its relationship with peptic ulcer and gastric cancer. Broadly speaking, gastritis may be of 2 types acute and chronic.
Chronic gastritis has further several subtypes.
Out of various classification schemes, the Sydney system of clinicopathologic classification of gastritis is the most widely accepted that takes into consideration the important etiologic role of Helicobacter pylori in various types of gastritis.
Acute Gastritis:
Acute gastritis is a transient acute inflammatory involvement of the stomach, mainly mucosa.
Etiopathogenesis: A variety of etiologic agents have been implicated in the causation of acute gastritis.
These are as follows:
1. Diet and personal habits
- Highly spiced food
- Excessive alcohol consumption
- Malnutrition
- Heavy smoking.
2. Infections
- Bacterial infections e.g. Helicobacter pylori, diphtheria, salmonellosis, pneumonia, staphylococcal food poisoning.
- Viral infections e.g. viral hepatitis, influenza, infectious mononucleosis.
3. Drugs
Intake of drugs like non-steroidal anti-inflammatory drugs (NSAIDs), aspirin, cortisone, phenylbutazone, indomethacin, preparations of iron, and chemotherapeutic agents.
4. Chemical and physical agents
- Intake of corrosive chemicals such as caustic soda, phenol, Lysol
- Gastric irradiation
- Freezing.

5. Severe stress
- Emotional factors like shock, anger, resentment etc.
- Extensive burns
- Trauma
- Surgery.
In acute gastritis, mucosal injury by any of the above agents causes acute inflammation by one of the following mechanisms
- Reduced blood flow, resulting in mucosal hypoperfusion due to ischaemia.
- Increased acid secretion and its accumulation due to H. pylori infection resulting in damage to the epithelial barrier.
- Decreased production of bicarbonate buffer.
Morphologic Features: Grossly, the gastric mucosa is oedematous with abundant mucus and haemorrhagic spots.
Microscopically, depending upon the stage, there is a variable amount of oedema and infiltration by neutrophils in the lamina propria. In acute haemorrhagic and erosive gastritis, the mucosa is sloughed off and there are haemorrhages on the surface.
Chronic Gastritis:
Chronic gastritis is the commonest histological change observed in biopsies from the stomach.
The microscopic change is usually poorly correlated to the symptomatology, as the histologic changes are observed in about 35% of endoscopically normal mucosal biopsies.
The condition occurs more frequently with advancing age; the average age for symptomatic chronic gastritis is 45 years which corresponds well with the age incidence of gastric ulcers.
Etiopathogenesis: In the absence of a clear aetiology of chronic gastritis, a number of etiologic factors have been implicated.
All the causative factors of acute gastritis described above may result in chronic gastritis too. Recurrent attacks of acute gastritis may result in chronic gastritis.
Some additional causes are as under:
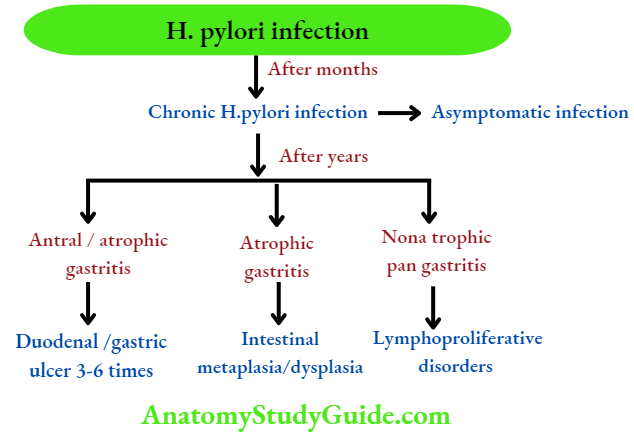
- Helicobacter pylori infection is strongly implicated in the etiology of chronic gastritis and is the most common cause of chronic gastritis.
- Autoimmune causes such as autoantibodies to gastric parietal cells in atrophic gastritis and autoantibodies against intrinsic factors are the next most common causes.
3. Other causes These include:
- Reflux of duodenal contents into the stomach, especially in cases which have undergone surgical intervention in the region of the pylorus.
- The associated diseases of the stomach and duodenum, such as gastric or duodenal ulcer, and gastric carcinoma.
- Chronic hypochromic anaemia, especially associated with atrophic gastritis.
The mechanism of chronic gastric injury by any of the etiologic agents is the cytotoxic effect of the injurious agent on the gastric mucosal epithelium, thus breaking the barrier and then inciting the inflammatory response.
Classification: Based on the type of mucosa affected (i.e. cardiac, body, pyloric, antral or transitional), guidelines for the clinicopathologic diagnosis as per classification given in Table have been proposed:
1. Type A Gastritis (Autoimmune Gastritis): Type gastritis involves mainly the body-fundic mucosa.
It is also called autoimmune gastritis due to the presence of circulating antibodies and is sometimes associated with other autoimmune diseases such as
Hashimoto’s thyroiditis and Addison’s disease. As a result of the antibodies against parietal cells and intrinsic factors, there is a depletion of parietal cells and impaired secretion of intrinsic factors.
These changes may lead to significant gastric atrophy where intestinal metaplasia may occur, and a small proportion of these patients may develop pernicious anaemia.
Due to the depletion of gastric acid-producing mucosal area, there is hypo- or achlorhydria, and hyperplasia of gastrin-producing G cells in the antrum resulting in hypergastrinaemia.
2. Type B Gastritis (H. pylori-related): Type B Gastritis mainly involves the region of antral mucosa and is more common.
It is also called hypersecretory gastritis due to excessive secretion of acid, commonly due to infection with H. pylori. These patients may have associated peptic ulcers. Unlike type gastritis, this form of gastritis has no autoimmune basis nor has an association with other autoimmune diseases.
3. Type AB gastritis (mixed gastritis, environmental gastritis, chronic atrophic gastritis): Type AB gastritis affects the mucosal region of A as well as B types (body-fundic and antral mucosa).
This is the most common type of gastritis in all age groups. It is also called environmental gastritis because a number of unidentified environmental factors have been implicated in its etiopathogenesis.
Chronic atrophic gastritis is also used synonymously with type AB gastritis because, in an advanced stage, there is a progression from chronic superficial gastritis to chronic atrophic gastritis, characterised by mucosal atrophy and metaplasia of intestinal or pseudo pyloric type.
Morphologic Features: Grossly, the features of all forms of gastritis are inconclusive. The gastric mucosa may be normal, atrophied, or oedematous.
Histologically, the criteria for categorisation are based on the following:
- The extent of inflammatory changes in the mucosa (i.e. superficial or deep).
- The activity of inflammation (i.e. quiescent or active; acute or chronic).
- Presence of any type of metaplasia (i.e. intestinal or pseudo pyloric).
The Sydney system of recording histologic changes in gastritis takes into account the following multiple parameters:
Etiology (H. pylori, autoimmune, NSAIDs, infections).
Location (pangastritis, predominant antral, predominant body-fundic).
Morphology (depth of inflammation superficial or deep, severity of inflammation, type of inflammation, atrophy, metaplasia).
Some special features (e.g. granulomas, eosinophilic gastritis, erosions, necrosis, haemorrhages).
Based on these criteria, morphologic features of various types of gastritis are as under:
1. Chronic Superficial Gastritis: As the name suggests there is an inflammatory infiltrate consisting of plasma cells and lymphocytes in the superficial layer of the gastric mucosa, but there are no histological changes in the deep layer of mucosa containing gastric
glands. Chronic superficial gastritis may resolve completely or may progress to chronic gastric atrophy.
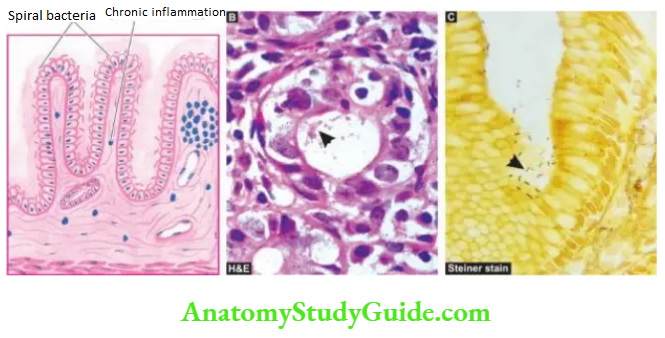
Its most common etiologic agent H. pylori, a spiral-shaped bacteria, was first reported by Warren and Marshall in Australia in 1982 as an inhabitant of the acid environment of the stomach causing gastritis. After initial scepticism, numerous workers subsequently verified that infection with H.
pylori is the most common etiologic agent associated with gastritis and its subsequent sequelae (Warren and Marshall shared Nobel Prize in medicine in 2005 for their discovery).
It is now known that H. pylori is causative for almost all active cases of chronic superficial gastritis and about 65% of quiescent cases. The organism is identified on the epithelial layer on the luminal surface and does not invade the mucosa.
It is not seen in areas with intestinal metaplasia. H. pylori gastritis can be diagnosed by the following techniques:
Invasive tests (Endoscopic biopsy):
histologic examination combined with special stains for identification of microorganisms Giemsa, Steiner silver or Warthin-Starry stains; biopsy urease test which is quick and simple but not fully sensitive; and culture of the microorganism that helps in determining specific antibiotic sensitivity.
Non-invasive tests:
serologic tests (Immunoblot, ELISwhich are cheap and convenient but may not be helpful in early follow-up cases; and
14C urea breath test. Although most patients with chronic superficial gastritis due to H. pylori remain asymptomatic, they may develop chronic atrophic gastritis, gastric atrophy, or peptic ulcer disease.
H. pylori infection is now considered an independent risk factor for gastric cancer: 3-6 fold increased risk for gastric adenocarcinoma and 6-50 times risk of MALT lymphoma.
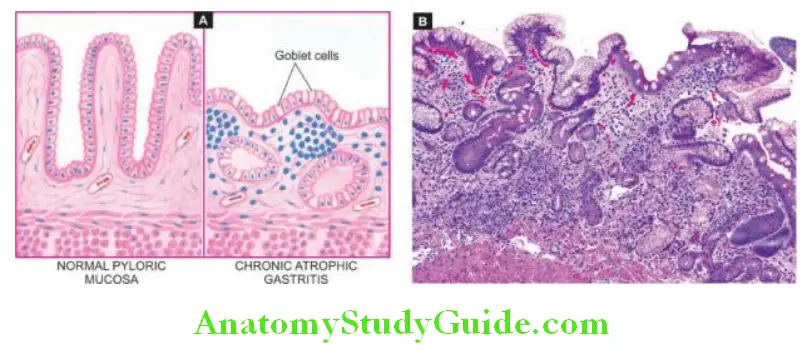
2. Chronic Atrophic Gastritis: In this stage, there is inflammatory cell infiltration in the deeper layer of the mucosa and atrophy of the epithelial elements including the destruction of the glands.
Two types of metaplasia are commonly associated with chronic atrophic gastritis:
Intestinal metaplasia is more common and involves antral mucosa more frequently. A characteristic histologic feature is the presence of intestinal-type mucous goblet cells; Paneth cells and endocrine cells may also be present.
Parietal cells are very few or absent. Intestinal metaplasia, focal or extensive, in chronic atrophic gastritis, is caused by H. pylori infection and is a precursor to gastric carcinoma. However, areas of intestinal metaplasia are not colonised by H. pylori.
Pseudopyloric metaplasia It involves the body glands which are replaced by proliferated mucus neck cells, conforming in appearance to normal pyloric glands. Its significance is not known.
3. Gastric Atrophy: In this, there is thinning of the gastric mucosa with loss of glands but no inflammation though lymphoid aggregates may be present.
4. Chronic Hypertrophic Gastritis (Menetrier’s Disease): This is an uncommon condition characterised pathologically by enormous thickening of gastric rugal folds resembling cerebral convolutions, affecting mainly the region of fundic-body mucosa and characteristically sparing antral mucosa.
The patients present with dyspepsia, haematemesis, melaena or protein-losing enteropathy.
Histologically, the gastric pits are elongated and tortuous. The mucosa is markedly thickened and parts of muscularis mucosae may extend into the thickened folds.
Epitheliumlined cysts are commonly seen in the glandular layer. Inflammatory infiltrate is usually mild but lymphoid follicles may be present. The condition is considered significant in view of the risk of developing cancer.
5. Miscellaneous Forms Of Chronic Gastritis: A few other types of gastritis which do not fit into the description of the types of gastritis described above are as under
Eosinophilic gastritis This condition is characterised by diffuse thickening of the pyloric antrum due to oedema and extensive infiltration by eosinophils in all the layers of the wall of the antrum. Eosinophilic gastritis probably has an allergic basis.
Chronic follicular gastritis: This is a variant of chronic atrophic gastritis in which numerous lymphoid follicles are present in the mucosa and submucosa of the stomach.
Haemorrhagic (or erosive) gastritis: In this condition, there are superficial erosions and mucosal haemorrhages, usually following severe haematemesis.
The causes for such erosions and haemorrhages are duodenal-gastric reflux, administration of non-steroidal anti-inflammatory drugs (NSAIDs), and portal hypertension.
Granulomatous gastritis: Rarely, granulomas may be present in the gastric mucosa such as in tuberculosis, sarcoidosis, Crohn’s disease, syphilis, various mycoses, and as a reaction to an endogenous substance or foreign material
Gastritis:
- Gastritis may be acute or chronic.
- Acute gastritis is caused by a variety of injurious agents in diet, infection, chemicals, certain drugs, chemicals and physical agents and severe burns.
- Chronic gastritis is most commonly due to H. pylori infection, or autoimmune causes; others are reflux of duodenal contents, other diseases of the stomach and duodenum, and chronic hypochromic anaemia.
- Based on microscopy, chronic gastritis is classified into superficial, atrophic, hypertrophic and specific types.
Peptic Ulcers:
Peptic ulcers are the areas of degeneration and necrosis of gastrointestinal mucosa exposed to acid-peptic secretions.
Though they can occur at any level of the alimentary tract that is exposed to hydrochloric acid and pepsin, they occur most commonly (98-99%) in either the duodenum or the stomach in the ratio of 4:1. Each of the two main types may be acute or chronic.
Acute Peptic (Stress) Ulcers:
Acute peptic ulcers or stress ulcers are multiple, small mucosal erosions, seen most commonly in the stomach but occasionally involving the duodenum.
Etiology: These ulcers occur following severe stress. The causes are as follows:
Psychological stress
Physiological stress as in the following:
- Shock
- Severe trauma
- Septicaemia
- Extensive burns (Curling’s ulcers in the posterior aspect of the first part of the duodenum).
- Intracranial lesions (Cushing’s ulcers developing from hyperacidity following excessive vagal stimulation).
- Drug intake (e.g. aspirin, steroids, furazolidone, indomethacin).
- Local irritants (e.g. alcohol, smoking, coffee etc).
Pathogenesis: It is not clear how the mucosal erosions occur in stress ulcers because actual hypersecretion of gastric acid is demonstrable in only Cushing’s ulcers occurring from intracranial conditions such as due to brain trauma, intracranial surgery and brain tumours.
In all other etiologic factors, gastric acid secretion is normal or below normal. In these conditions, the possible hypotheses for the genesis of stress ulcers are as under
- Ischaemic hypoxic injury to the mucosal cells.
- Depletion of the gastric mucus ‘barrier’ renders the mucosa susceptible to attack by acid-peptic secretions.
Morphologic Features:
Grossly, acute stress ulcers are multiple (more than three ulcers in 75% of cases). They are more common anywhere in the stomach, followed by decreasing frequency by occurrence in the first part of the duodenum.
They may be oval or circular in shape, usually less than 1 cm in diameter. Microscopically, the stress ulcers are shallow and do not invade the muscular layer.
The margins and base may show some inflammatory reaction depending upon the duration of the ulcers. These ulcers commonly heal by complete re-epithelialisation without leaving any scars.
Complications such as haemorrhage and perforation may occur.
Chronic Peptic Ulcers (Gastric and Duodenal Ulcers) If not specified, chronic peptic ulcers would mean gastric and duodenal ulcers, the two major forms of ‘peptic ulcer disease’ of the upper GI tract in which the acid-pepsin secretions are implicated in their pathogenesis.
Peptic ulcers are common in the present-day life of the industrialised and civilised world.
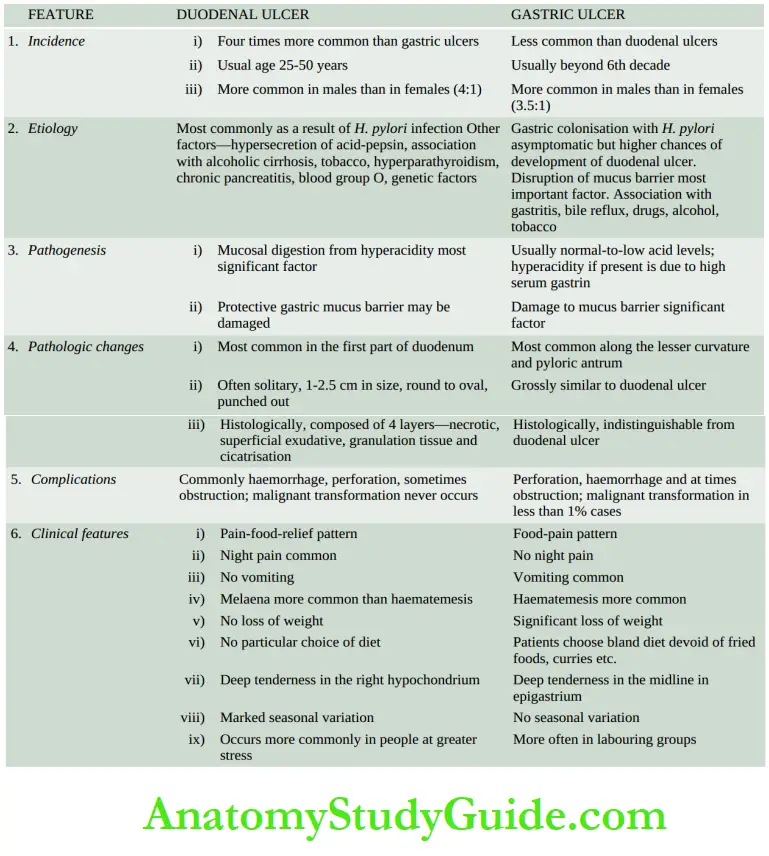
Gastric and duodenal ulcers represent two distinct diseases as far as their etiology, pathogenesis and clinical features are concerned.
However, morphological findings in both are similar and quite diagnostic. The features of gastric and duodenal peptic ulcers are described together below while their contrasting features.
Incidence: Peptic ulcers are more frequent in middle-aged adults. The peak incidence for duodenal ulcers is in the 5th decade, while for gastric ulcers it is a decade later (6th decade).
Duodenal as well as gastric ulcers are more common in males than in females. Duodenal ulcer is almost four times more common than gastric ulcer; the overall incidence of gastroduodenal ulcers is approximately 10% of the male population.
Etiology: The immediate cause of peptic ulcer disease is a disturbance in the normal protective mucosal ‘barrier’ by acid-pepsin, resulting in the digestion of the mucosa.
However, in contrast to duodenal ulcers, the patients of gastric ulcers have low-to-normal gastric acid secretions, though true achlorhydria in response to stimulants never occurs in benign gastric ulcers.
Besides, 10- 20% patients of with gastric ulcers may have coexistent duodenal ulcers as well. Thus, the aetiology of peptic ulcers possibly may not be explained on the basis of a single factor but is multifactorial.
These factors are discussed below but the first two H. pylori gastritis and NSAID-induced injury are considered the most important.
1. Helicobacter pylori gastritis About 15-20% of cases infected with H. pylori in the antrum develop a duodenal ulcer in their lifetime while gastric colonisation by H. pylori never develops ulceration and remains asymptomatic.
H. pylori can be identified in mucosal samples by histologic examination, culture and serology.
2. NSAID-induced mucosal injury Non-steroidal anti-inflammatory drugs are the most commonly used medications in developed countries and are responsible for direct toxicity, endothelial damage and epithelial injury to both gastric as well as duodenal mucosa.
3. Acid-pepsin secretions: There is conclusive evidence that some level of acid-pepsin secretion is essential for the development of duodenal as well as gastric ulcers.
Peptic ulcers never occur in association with pernicious anaemia in which there are no acid and pepsin-secreting parietal and chief cells respectively.
4. Gastritis: Some degree of gastritis is always present in the region of gastric ulcer, though it is not clear whether it is the cause or the effect of the ulcer. Besides, the population distribution pattern of gastric ulcers is similar to that of chronic gastritis.
5. Other local irritants: Pyloric antrum and lesser curvature of the stomach are the sites most exposed for longer periods to local irritants and thus are the common sites for the occurrence of gastric ulcers.
Some of the local irritating substances implicated in the aetiology of peptic ulcers are heavily spiced foods, alcohol, cigarette smoking, and unbuffered aspirin.
6. Dietary factors: Nutritional deficiencies have been regarded as etiologic factors in peptic ulcers e.g. occurrence of gastric ulcers in poor socioeconomic strata and higher incidence of duodenal ulcers in parts of South India.
However, malnutrition does not appear to have any causative role in peptic ulceration in European countries and the US.
7. Psychological factors: Psychological stress, anxiety, fatigue and ulcer-type personality may exacerbate as well as predispose to peptic ulcer disease.
8. Genetic factors: People with blood group O appear to be more prone to develop peptic ulcers than those with other blood groups. Genetic influences appear to have a greater role in duodenal ulcers as evidenced by their occurrence in families, monozygotic twins and association with HLA-B5 antigen.
9. Hormonal factors: Secretion of certain hormones by tumours is associated with peptic ulceration e.g. elaboration of gastrin by islet-cell tumour in Zollinger-Ellison syndrome, endocrine secretions in hyperplasia and adenomas of parathyroid glands, adrenal cortex and anterior pituitary.
10. Miscellaneous: Duodenal ulcers have been observed to occur in association with various other conditions such as alcoholic cirrhosis, chronic renal failure, hyperparathyroidism, chronic obstructive pulmonary disease, and chronic pancreatitis.
Pathogenesis: There are distinct differences in the pathogenetic mechanisms involved in duodenal and gastric ulcers under Duodenal ulcer There is conclusive evidence to support the role of high acid-pepsin secretions in the causation of duodenal ulcers.
Besides this, a few other noteworthy features in the pathogenesis of duodenal ulcers are as follows:
1. There is generally hypersecretion of gastric acid into the fasting stomach at night which takes place under the influence of vagal stimulation.
There is high basal as well as maximal acid output (BAO and MAO) in response to various stimuli.
2. Patients with duodenal ulcers have rapid emptying of the stomach so that the food which normally buffers and neutralises the gastric acid, passes down into the small intestine, leaving the duodenal mucosa exposed to the aggressive action of gastric acid.
3. Helicobacter gastritis caused by H. pylori is seen in 95-100% of cases of duodenal ulcers.
The underlying mechanisms are as under:
Gastric mucosal defence is broken by bacterial elaboration of urease, protease, catalase and phospholipase.
Host factors H. pylori-infected mucosal epithelium releases pro-inflammatory cytokines such as IL-1, IL-6, IL-8 and tumour necrosis factor-a, all of which incite an intense inflammatory reaction.
Bacterial factors Epithelial injury is also induced by cytotoxin-associated gene protein (CagA), while vacuolating cytotoxin (Vacinduces the elaboration of cytokines.
Gastric ulcer The pathogenesis of gastric ulcer is mainly explained on the basis of impaired gastric mucosal defences against acid-pepsin secretions.
Some other features in the pathogenesis of gastric ulcers are as follows:
- Hyperacidity may occur in gastric ulcers due to increased serum gastrin levels in response to ingested food in an atonic stomach.
- However, many patients with gastric ulcers have low-to-normal gastric acid levels. Ulcerogenesis in such patients is explained on the basis of the damaging influence of other factors such as gastritis, bile reflux, cigarette smoke etc.
- The normally protective gastric mucus ‘barrier’ against acid-pepsin is deranged in gastric ulcers. There is a depletion in the quantity as well as quality of gastric mucus. One of the mechanisms for its depletion is the colonisation of the gastric mucosa by H. pylori seen in 75-80% patients of with gastric ulcers.
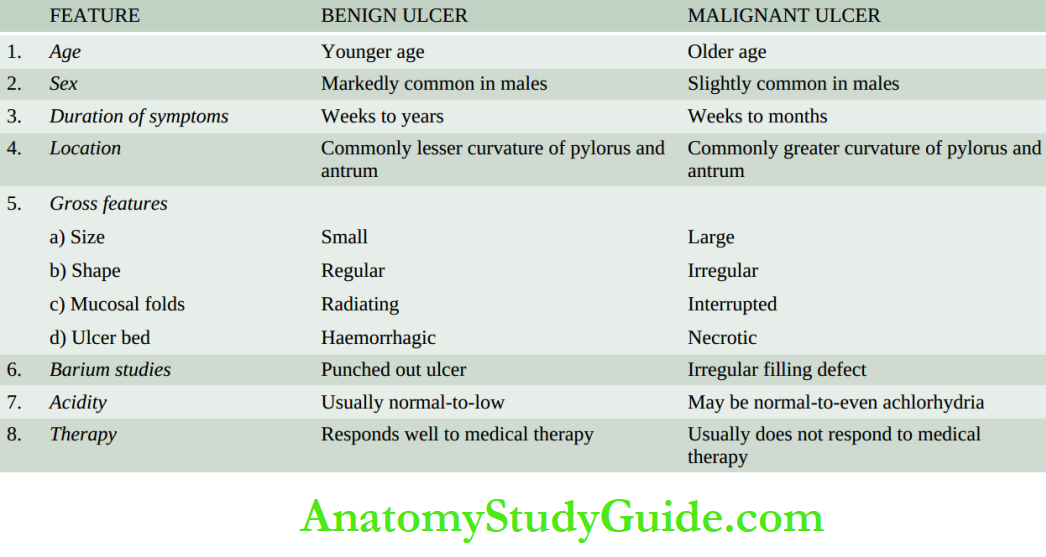
Morphologic Features: Gross and microscopic changes in gastric and duodenal ulcers are similar and quite characteristic. Gastric ulcers are found predominantly along the lesser curvature in the region of the pyloric antrum, more commonly on the posterior than the anterior wall.
Most duodenal ulcers are found in the first part of the duodenum, usually immediate post-pyloric, more commonly on the anterior than the posterior wall. Uncommon locations include ulcers in the cardia, marginal ulcers and in Meckel’s diverticulum.
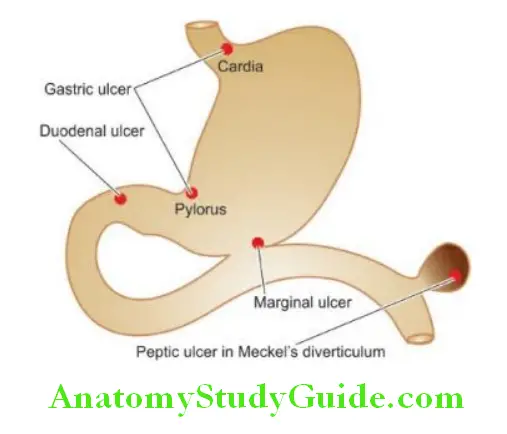
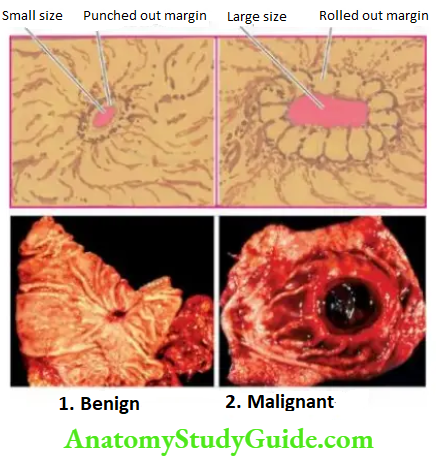
Grossly, typical peptic ulcers are commonly solitary (80%), small (1-2.5 cm in diameter), round to oval and characteristically ‘punched out’. Benign ulcers usually have flat margins at the level with the surrounding mucosa.
The mucosal folds converge towards the ulcer.
The ulcers may vary in depth from being superficial (confined to mucosa-deep ulcers penetrating into the muscular layer). In about 10-20% of cases, gastric and duodenal ulcers are coexistent. The vast majority of the peptic ulcers are benign.
Chronic duodenal ulcer never turns malignant, while chronic gastric ulcer may develop carcinoma in less than 1% of cases. Malignant gastric ulcers are larger, bowl-shaped with elevated and indurated mucosa at the margin.
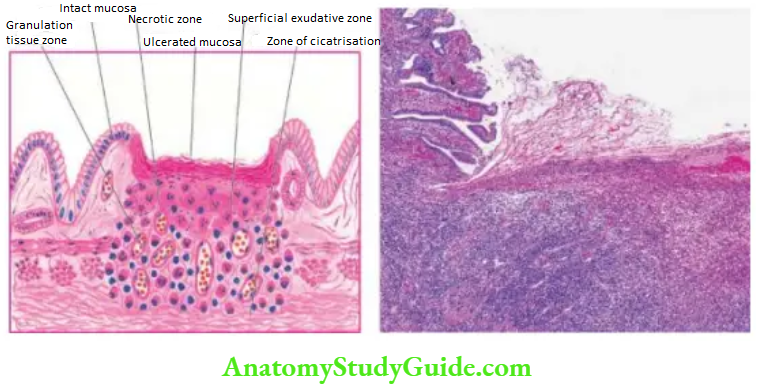
Microscopically, chronic peptic ulcers have 4 histological zones.
From within and outside, these are as under:
1. Necrotic zone lies on the floor of the ulcer and is composed of a fibrinous exudate containing necrotic debris and a few leucocytes.
2. Superficial exudative zone lies underneath the necrotic zone. The tissue elements here show coagulative necrosis giving an eosinophilic, smudgy appearance with nuclear debris.
3. Granulation tissue zone is seen merging into the necrotic zone. It is composed of nonspecific inflammatory infiltrate and proliferating capillaries.
4. Zone of cicatrisation is seen merging into a thick layer of granulation tissue. It is composed of dense fibro collagenic scar tissue over which granulation tissue rests. Thrombosed or sclerotic arteries may cross the ulcer which on erosion may result in haemorrhage.
Complications: Acute and subacute peptic ulcers usually heal without leaving any visible scar. However, healing of chronic, larger and deeper ulcers may result in complications.
These are as follows:
1. Obstruction: The development of fibrous scar at or near the pylorus results in pyloric stenosis. Healed duodenal ulcers cause duodenal stenosis. Healed gastric ulcers along the lesser curvatures may produce ‘hourglass’ deformity due to fibrosis and contraction.
2. Haemorrhage: Minor bleeding by erosion of small blood vessels in the base of an ulcer occurs in all the ulcers and can be detected by testing the stool for occult blood. Chronic blood loss may result in iron deficiency anaemia.
Severe bleeding may cause ‘coffee ground’ vomitus or melaena. A penetrating chronic ulcer may erode a major artery (e.g. left gastric, gastroduodenal or splenic artery) and cause a massive and severe haematemesis and sometimes death.
3. Perforation A perforated peptic ulcer is an acute abdominal emergency. Perforation occurs more commonly in chronic duodenal ulcers than in chronic gastric ulcers.
The following sequelae may result:
On perforation the contents escape into the lesser sac or into the peritoneal cavity, causing acute peritonitis.
Air escapes from the stomach and lies between the liver and the diaphragm giving the characteristic radiological appearance of air under the diaphragm.
Subphrenic abscesses between the liver and the diaphragm may develop due to infection. Perforation may extend to involve the adjacent organs e.g. the liver and pancreas.
4. Malignant transformation: The dictum ‘cancers ulcerate but ulcers rarely cancerate’ holds true for most peptic ulcers. chronic duodenal ulcer never turns malignant, while less than 1% of chronic gastric ulcers may transform into carcinoma.
Clinical Features: Peptic ulcers are remitting and relapsing lesions.
Their chronic and recurrent behaviour is summed up by saying:
‘Once a peptic ulcer patient, always a peptic ulcer patient.’ The two major forms of chronic peptic ulcers show variations in clinical features which are as follows:
1. Age: The peak incidence of duodenal ulcer is in the 5th decade while that for gastric ulcer is a decade later.
2. People at risk: Duodenal ulcer occurs more commonly in people faced with more stress and strain of life (e.g. executives, leaders), while gastric ulcer is seen more often in labouring groups.
3. Periodicity: The attacks in gastric ulcers last from 2-6 weeks, with intervals of freedom from 1-6 months. The attacks of duodenal ulcer, are classically worsened by ‘work, worry and weather.’
4. Pain: In gastric ulcers, epigastric pain occurs immediately or within 2 hours after food and never occurs at night. In duodenal ulcers, pain is severe, occurs late at night (‘hunger pain’) and is usually relieved by food.
5. Vomiting: Vomiting which relieves pain is a conspicuous feature in patients with gastric ulcers. Duodenal ulcer patients rarely have vomiting but instead, get heartburn (retrosternal pain) and ‘water brash’ (burning fluid into the mouth).
6. Haematemesis and melaena: Haematemesis and melaena occur in gastric ulcers in the ratio of 60:40, while in duodenal ulcers in the ratio of 40:60. Both may occur together more commonly in duodenal ulcers than in gastric ulcer patients.
7. Appetite: The gastric ulcer patients, though have good appetite but are afraid to eat, while duodenal ulcer patients have very good appetite.
8. Diet: Patients with gastric ulcer commonly get used to a bland diet consisting of milk, eggs etc and avoid taking fried foods, curries and heavily spiced foods. In contrast, duodenal ulcer patients usually take all kinds of diets.
9. Weight: Loss of weight is a common finding in gastric ulcer patients while patients with duodenal ulcers tend to gain weight due to frequent ingestion of milk to avoid pain.
10. Deep tenderness: Deep tenderness is demonstrable in both types of peptic ulcers. In the case of gastric ulcer, it is in the midline of the epigastrium, while in the duodenal ulcer, it is in the right hypochondrium.
Peptic Ulcers:
Peptic ulcers are the areas of degeneration and necrosis in the duodenum or the stomach. Each of the two main types may be acute or chronic.
The immediate cause of peptic ulcer disease is a disturbance in the normal protective mucosal ‘barrier’ by acid-pepsin.
The etiology of peptic ulcers is multifactorial but H. pylori gastritis and NSAID-induced injury are the two most important factors.
In the pathogenesis of duodenal ulcers, there is a role of high acid-pepsin secretions, while in gastric ulcers gastric mucosal defenses against acid-pepsin secretions are impaired.
Typical peptic ulcers are commonly solitary, small, round to oval and punched out.
Microscopically, chronic peptic ulcers have 4 histological zones: necrotic zone, superficial exudative, granulation tissue, and zone of cicatrisation. Important complications of peptic ulcers are obstruction, haemorrhage, perforation and, rarely malignant change.
Gastric Tumours And Tumour-Like Lesions:
While the WHO classification of benign tumours, premalignant lesions and malignant tumours of the stomach, a few tumour-like lesions or polyps also occur in the stomach which are described first.
Tumour-Like Lesions (Gastric Polyps):
Polyps are mucosal projections and appear like tumours due to their endoscopic and gross appearance. Polyps may occur in the entire GI tract but are most common in the colorectal region. Gastric polyps include three types: hyperplastic, fundic gland, and inflammatory fibroid.
1. Epithelial benign/premalignant lesions:
- Adenoma
- Intraepithelial neoplasia (low-grade, high-grade)
2. Epithelial malignant tumours
Carcinomas (90%)
- Adenocarcinoma
- intestinal type
- diffuse type
- Papillary adenocarcinoma
- Tubular adenocarcinoma
- Mucinous adenocarcinoma
- Signet-ring adenocarcinoma
- Adenosquamous carcinoma
- Squamous cell carcinoma
- Small cell carcinoma
- Undifferentiated carcinoma
2. Carcinoid tumour (3%)
3. Non-epithelial tumours (6%)
- Leiomyoma
- Schwannoma
- Granular cell tumour
- Glomus tumour
- Leiomyosarcoma
- A gastrointestinal stromal tumour (gist)
- Kaposi’s sarcoma
- Malignant lymphomas
4. Secondary tumours (1%)
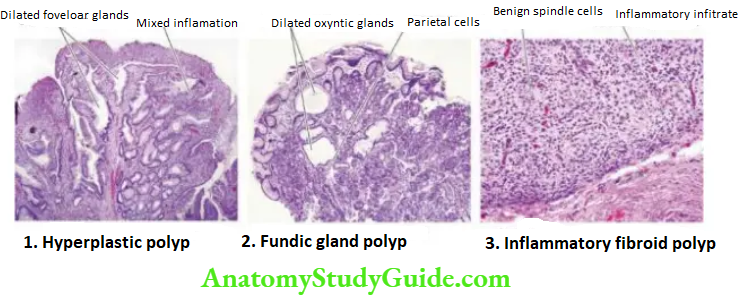
Hyperplastic Polyps:
Hyperplastic or inflammatory polyps are regenerative, non-neoplastic lesions which are the most common type (90%). They may be single or multiple and are more often located in the pyloric antrum.
Grossly, the lesions may be sessile or pedunculated, 1 cm or larger in size, smooth and soft. The surface may be ulcerated or haemorrhagic.
Microscopically, they are composed of irregular hyperplastic foveolar glands, which may show dilatation while the lamina propria shows oedema and mixed inflammation,
1. The lining epithelium is mostly superficial gastric type but antral glands, chief cells and parietal cells may be present. These lesions do not have cellular atypia and are benign.
Fundic Gland Polyps:
These are quite common and may occur sporadically or are seen in the background of familial adenomatous polyposis or Zollinger-Ellison syndrome.
Microscopically, the polyps contain dilated oxyntic glands lined by parietal and chief cells These polyps are also benign.
Inflammatory Fibroid Polyps:
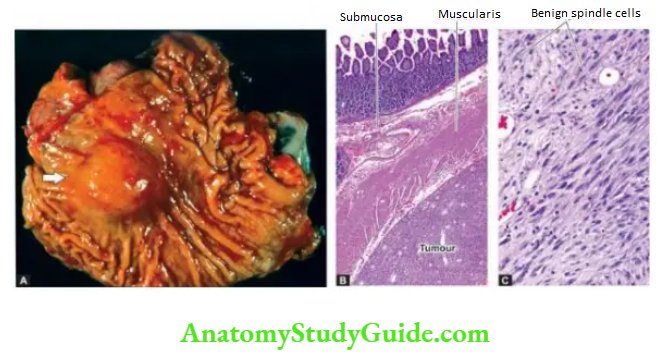
Inflammatory fibroid polyps, also called eosinophilic granulomatous polyps, are seen in the mucosa of the stomach or small intestine.
Microscopically, these polyps show the proliferation of benign spindle cells, inflammatory infiltration with a prominence of eosinophils, and proliferating blood vessels. Inflammatory fibroid polyps are reactive proliferations and not true neoplasms.
Epithelial Benign And Premalignant Lesions:
Adenomas (Adenomatous or Neoplastic Polyps):
Adenomas, also, referred to as adenomatous or neoplastic polyps, are true benign epithelial neoplasms and are much less common in the stomach than in the large intestine. They are found more often in the region of the pyloric antrum.
Gastric adenomas are commonly associated with atrophic gastritis and pernicious anaemia. Morphologically, adenomatous polyps of the stomach resemble their counterparts in the large bowel and are described.
The overlying epithelium frequently shows low- to high-grade dysplasia (or intraepithelial neoplasia hence these lesions are premalignant.
Mesenchymal Tumours:
The stomach may be the site for the occurrence of various uncommon benign mesenchymal tumours such as leiomyomas (most common), schwannomas, granular cell tumours and glomus tumours. They are usually firm, circumscribed nodules, less than 4 cm in size and appear as submucosal nodules.
They resemble in gross and microscopic appearance with their counterparts in other parts of the body. However, one mesenchymal tumour seen in the entire length of the gut called gastrointestinal stromal tumour (GIST), has attracted more attention in recent times.
Gastrointestinal Stromal Tumour (GIST):
The term GIST is used for a group of mesenchymal tumours composed of spindle cells or stromal cells that are related to interstitial cells of Cajal.
These cells are normally present in the entire length of the bowel, act as pacemakers of the gut, and thus coordinate peristalsis. GIST may occur anywhere along the gut but is most common in the stomach (60%), followed in descending frequency by the small intestine (30%).
The common age for the occurrence of GIST is 60-70 years.
Pathogenesis: Molecular basis of GIST has been extensively studied and is the first solid tumour in which targeted therapy was started based on molecular profiling of the tumour.
1. About 85% of cases of GIST have an activating mutation in the KIT gene in the tumour cells that encodes for tyrosine kinase (c-KIT). c-KIT mutation can be detected in the tumour cells by CD117 immunohistochemical stain.
2. Around 10-15% of cases of GIST contain a mutation in platelet-derived growth factor receptor alpha (PDGFR-a). These tumours more often have epithelioid morphology.
3. Rarely, GIST may have BRAF mutation.
Grossly, GIST is typically a solitary, firm and encapsulated tumour of varying size, commonly located in the submucosa or deeper layers.
Microscopically, the following features are present:
Classically, GIST is composed of interlacing bundles of uniform spindle cells with eosinophilic cytoplasm. The nuclei have indentation due to the perinuclear halo.
Differential diagnosis from other benign mesenchymal tumours such as leiomyoma and schwannoma is made by immunohistochemical staining for CD117.
Less often, a morphologic variant of GIST with epithelioid morphology may be seen.
Approximately 25% GISTs which are larger in size may have features of malignancy high N: C ratio, nuclear hyperchromatism, and high mitotic rate.
Malignant Tumours:
Gastric Carcinoma:
Incidence: Carcinoma of the stomach comprises more than 90% of all gastric malignancies and is the second most frequent cause of cancer-related mortality worldwide.
However, its incidence has been declining in Western countries and in traditionally high-incidence countries (e.g. Japan and Koredue to changing nutritional habits and early detection.
The highest incidence is between the 4th to 6th decades of life and is twice more common in men than in women.
Etiology: A number of etiologic factors have been implicated in the causation of gastric cancer.
These are as under:
1. H. pylori infection of the stomach causing chronic atrophic gastritis and intestinal metaplasia is an important risk factor for the development of gastric cancer.
Epidemiologic studies throughout the world have shown that seropositivity with H. pylori is associated with a 3 to 6 times higher risk of development of gastric cancer.
It may be mentioned here that a similar association of H.pylori infection exists with gastric lymphomas (MALT-type) as well.
2. Dietary factors: Epidemiological studies suggest that dietary factors are most significant in the aetiology of gastric cancer.
The evidence in support of this is multifold:
Occurrence of gastric cancer in the region of the gastric canal (i.e. along the lesser curvature and the pyloric antrum) where harmful foods exert their maximum effect.
Dietary nitrates are converted to carcinogenic nitrites by bacteria. Nitrates are found in dried, smoked and salted foods.
When these foods are either contaminated due to infection or due to H. pylori infection in the stomach, nitrates in them are converted into nitrites which are carcinogenic. Long-term consumption of such foods is certainly associated with a high risk of gastric cancer.
Intake of fresh green leafy vegetables and fruits lowers the risk of developing gastric cancer.
Tobacco smoke, tobacco juice and consumption of alcohol have all been shown to have a carcinogenic effect on the gastric mucosa.
3. Geographical factors There are geographic variations in the incidence of gastric cancer.
Japan, Korea, Chile and Italy have the highest recorded death rate from gastric cancer, while the incidence is considerably low in the US, UK and Canada.
The higher incidence in certain geographic regions is the result of environmental influences as observed from the finding of the incidence of gastric cancer in the next generation of Japanese immigrants to the US which is comparable to that of Native Americans.
4. Racial factors Within the country, different ethnic groups may have variations in the incidence of gastric cancer e.g. incidence is higher in Blacks, American Indians, and Chinese in Indonesia, and North Wales than in other parts of Wales.
5. Genetic factors: Genetic influences have some role in the etiology of gastric cancer. Not more than 4% of patients with gastric cancer have a family history of this disease.
Individuals with blood group A have a higher tendency to develop gastric cancer (Recall that the peptic ulcer is more common in individuals with blood group O).
A germline mutation in the E-cadherin gene inherited as an autosomal dominant pattern has been linked to the higher incidence of occult gastric cancer in younger individuals.
6. Pre-malignant changes in the gastric mucosa: There are certain conditions of gastric mucosa which have increased the risk of development of gastric cancer
- Hypo- or achlorhydria in atrophic gastritis of gastric mucosa with intestinal metaplasia.
- Adenomatous (neoplastic polyps of the stomach.
- Chronic gastric ulcer (ulcer-cancer), and its association with achlorhydria.
- Stump carcinoma in patients who have undergone partial gastrectomy.
Morphologic Features: Gastric carcinoma is most commonly located in the region of the gastric canal (pre-pyloric region) formed by lesser curvature, pylorus and antrum.
Other less common locations are the body, cardia and fundus. Pathogenetically, a sequential evolution of all gastric carcinomas from an initial stage of in situ carcinoma confined to mucosal layers called early gastric carcinoma (has been found. EGC eventually penetrates the muscular or beyond, resulting in advanced gastric carcinoma.
Accordingly, gastric carcinomas are broadly classified into 2 main categories:
- Early gastric carcinoma (EGC).
- Advanced gastric carcinoma, which has further subtypes.
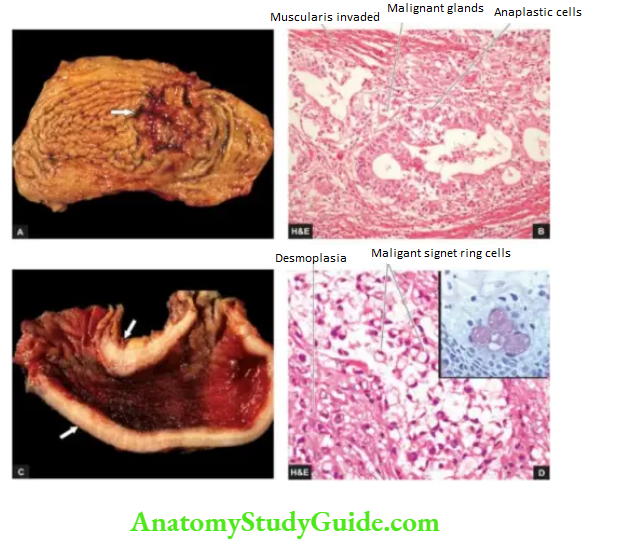
1. Early Gastric Carcinoma (EGC): EGC is the term used to describe cancer limited to the mucosa and submucosa.
The diagnosis of this condition has been made possible by extensive work on the histogenesis of gastric cancer by Japanese researchers by the use of a fibreoptic endoscope and gastrocamera. In Japan, EGC comprises 35% of newly diagnosed cases of gastric cancer.
Grossly, the lesion of EGC may have 3 patterns polypoid (protruded), superficial and ulcerated.
Histologically, EGC is a typical glandular adenocarcinoma, usually a well-differentiated type. The prognosis of EGC after surgical resection is quite good; the 5-year survival rate is 93- 99%.
Early gastric carcinoma must be distinguished from epithelial dysplasia seen in intestinal metaplasia such as in atrophic gastritis and pernicious anaemia.
2. Advanced Gastric Carcinoma: When the carcinoma crosses the submucosa into the muscularispropria or beyond, it is referred to as advanced gastric carcinoma.
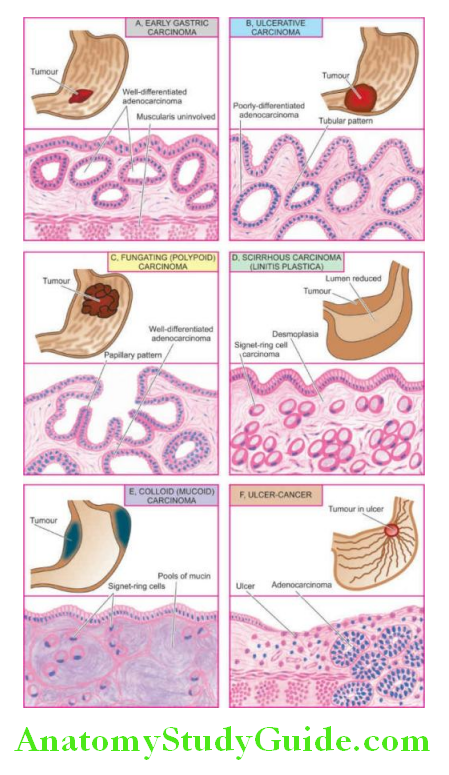
Grossly, advanced gastric carcinoma has the following 5 patterns:
1. Ulcerative carcinoma: This is the most common pattern. The tumour appears as a flat, infiltrating and ulcerative growth with an irregular necrotic base and raised margin. It is seen more commonly in the region of the gastric canal.
2. Fungating (polypoid) carcinoma: The second common pattern is a cauliflower growth projecting into the lumen, similar to what is commonly seen in the large intestine. It is seen more often in the fundus. The tumour undergoes necrosis and infection commonly.
3. Scirrhous carcinoma (Linitis plastica): In this pattern, the stomach wall is thickened due to extensive desmoplasia giving the appearance of a ‘leather-bottle stomach’ or ‘linitis plastic. The involvement may be localised to the pyloric antrum, or diffuse affecting the whole of the stomach from the cardia to the pylorus. The lumen of the stomach is reduced. There are no ulcers but rugae are prominent.
4. Colloid (Mucoid) carcinoma: This pattern is usually seen in the fundus. The tumour grows like a mass having a gelatinous appearance due to the secretion of large quantities of mucus.
5. Ulcer-cancer: The development of cancer in chronic gastric ulcers is a rare occurrence (less than 1%). The majority of ulcer cancers are malignant lesions from the beginning.
For confirmation of cancer in a pre-existing gastric ulcer, the characteristic microscopic appearance of a peptic ulcer should be demonstrable with one portion of the base or the margin of the ulcer showing carcinomatous changes. Differences between a benign and malignant gastric ulcer.
Histologically, gastric carcinoma is an adenocarcinoma having different architectural patterns and varying grades at the cytologic level. Conventionally, it has been divided by Lauren’s criteria into the following types
1. Intestinal type is most common comprising ~55% of cases. It is seen more often in association with preceding H. pylori gastritis and intestinal metaplasia. Intestinal-type adenocarcinoma is composed of glandular architecture lined by anaplastic epithelial cells.
2. Diffuse type comprises ~30% of cases and is seen more in younger patients and in females.
Diffuse type does not have a glandular pattern but is instead composed of diffuse anaplastic malignant cells containing mucin that may form signet ring cells.
3. Intermediate type comprises the remaining ~15% of cases. It combines features of both intestinal and diffuse patterns.
The WHO classification identifies 4 major histologic types:
1. Tubular carcinoma is the most common histologic type. It has irregularly placed diffuse glands lined by malignant cells.
2. Papillary adenocarcinoma is characterised by malignant cells covering fibrovascular cores.
3. Signet ring carcinoma is a poorly-differentiated carcinoma in which most of the tumour cells contain mucin vacuole in the cytoplasm pushing the nucleus to the periphery and forming a signet ring appearance.
The tumour cells may be arranged as trabeculae or incompletely formed glands and are often accompanied by marked stromal desmoplasia.
4. Mucinous adenocarcinoma is characterised by pools of mucin in >50% of tumour tissue. The tumour may have a glandular or diffuse pattern.
Spread:
Carcinoma of the stomach may spread by the following routes:
1. Direct spread by local extension is the most common feature of gastric carcinoma. The spread occurs mainly from the loose submucosal layer but eventually, muscularis and serosa are also invaded.
After the peritoneal covering of the stomach has been invaded, trans-coelomic dissemination may occur in any other part of the peritoneal cavity but ovarian masses (one-sided or both-sided) occur more commonly, referred to as Krukenberg tumours.
Submucosal spread occurs more often upwards into the oesophagus due to the continuity of the layers of the stomach with those of the oesophagus, while the spread downwards into the duodenum occurs less often due to the presence of pyloric sphincter and submucosal Brunner’s glands.
The tumour may directly involve other neighbouring structures and organs like lesser and greater omentum, pancreas, liver, common bile duct, diaphragm, spleen and transverse colon.
2. Lymphatic spread: Metastases to regional lymph nodes occur early, especially in scirrhous carcinoma.
The groups of lymph nodes involved are along the lesser and greater curvature around the cardia and supra pancreatic lymph nodes. Involvement of the left supraclavicular lymph node, Virchow or Troisier’s sign, is sometimes the presenting feature of gastric carcinoma.
3. Haematogenous spread: Blood spread of gastric carcinoma may occur to the liver, lungs, brain, bones, kidneys and adrenals. It occurs more commonly with the poorly-differentiated carcinoma.
The American Joint Committee on Cancer has developed a TNM staging system for gastric carcinoma based on tumour invasion (T), lymph node involvement (N) and distant metastasis (M) into earliest stage Tis N0 M0 (intraepithelial tumour) to most advanced stage Tany Nany M1.
Clinical Features: Gastric carcinoma may have diverse presentations.
The usual clinical features are as under:
- Persistent abdominal pain
- Gastric distension and vomiting
- Loss of weight (cachexia)
- Loss of appetite (anorexia)
- Anaemia, weakness, malaise.
The most common complication of gastric cancer is haemorrhage (in the form of haematemesis and/or melaena); others are obstruction, perforation and jaundice.
Gastric carcinoma remains undiagnosed until late when the symptoms appear.
Therefore, the prognosis is generally poor; 5-year survival rate is 5-15% from the time of diagnosis of advanced gastric carcinoma. However, the 5-year survival rate for early gastric carcinoma is far higher (93-99%) and hence the need for early diagnosis of the condition.
Carcinoid Tumour:
Carcinoid tumours are less common in the stomach and are usually non-argentaffin type but argent adenomas also occur. Their behaviour is usually malignant; they are described along with carcinoids in the small intestine.
Malignant Lymphoma:
Primary gastrointestinal lymphomas are defined as lymphomas arising in the gut without any evidence of systemic involvement at the time of presentation.
Secondary gastrointestinal lymphomas, on the other hand, appear in the gut after dissemination from another primary site.
Gastric lymphomas constitute over 50% of all bowel lymphomas; other sites being small and large bowel in decreasing order of frequency. The prognosis of primary gastric lymphoma is better than for intestinal lymphomas. Primary lymphoma of the stomach is the most common malignant gastric tumour (4%) next to carcinoma.
Clinical manifestations of gastric lymphomas may be similar to gastric carcinoma. Age lower than that for carcinoma (30-40 years as compared to 40-60 years in gastric carcinoma may occur even in childhood. Relationship with long-standing chronic H. pylori gastritis with lymphoid hyperplasia has been strongly suggested.
Grossly, gastric lymphomas have 2 types of appearances:
1. Diffusely infiltrating type, producing thickening of the affected gut wall, obliteration of mucosal folds and ulcerations.
The cut section shows lesions in the mucosa and submucosa but in late-stage whole thickness of the gut wall may be affected.
2. Polypoid type, which produces a large protruding mass into the lumen with an ulcerated surface. When present in the small intestine, it may cause luminal obstruction.
Lymph node involvement may occur in either of the two patterns.
Microscopically, gastric lymphomas are most often non-Hodgkin’s lymphomas of the following types
Low-grade mature small lymphocytic B-cell lymphomas referred to as MALToma are most common, arising from Mucosa Associated Lymphoid Tissue.
High-grade diffuse large B-cell lymphoma (DLBCL) may occur by the transformation of low-grade lymphoma.
Haematemesis:
Haematemesis is vomiting of blood which may be mild or massive. It may occur due to lesions in the oesophagus or stomach.
Haematemesis Of Oesophageal Origin:
1. Oesophageal Varices: Oesophageal varices are tortuous, dilated and engorged oesophageal veins, seen along the longitudinal axis of the oesophagus. They occur as a result of elevated pressure in the portal venous system, most commonly in cirrhosis of the liver.
Less common causes are portal vein thrombosis, hepatic vein thrombosis (Budd-Chiari syndrome) and pylephlebitis. The lesions occur as a result of bypassing of portal venous blood from the liver to the oesophageal venous plexus.
The increased venous pressure in the superficial veins of the oesophagus may result in ulceration and massive bleeding.
2. Mallory-Weiss Syndrome: In this condition, there are lacerations of mucosa at the gastro-oesophageal junction following minor trauma such as vomiting, retching or vigorous coughing. Patients present with upper gastro-oesophageal bleeding.
Figure 20.20 Malignant lymphoma stomach. The tumour is seen diffusely infiltrating lymphoid cells in the wall of the stomach.
(Reproduced
3. Rupture Of The Oesophagus: may occur following trauma, during oesophagoscopy, indirect injury (e.g. due to sudden acceleration and deceleration of the body) and spontaneous rupture (e.g. after overeating, extensive aerophagy etc).
4. Other Causes:
Oesophageal haematemesis may also occur in the following conditions:
- Bursting of an aortic aneurysm into the lumen of the oesophagus
- Vascular erosion by malignant growth in the vicinity
- Hiatus hernia
- Oesophageal cancer
- Purpuras
- haemophilia.
Haematemesis Of Gastric Origin:
These causes are as under:
- Chronic peptic ulcers (gastric as well as duodenal)
- Acute peptic ulcers (stress ulcers)
- Multiple gastric and duodenal erosions
- Carcinoma of the stomach
- A peptic ulcer in Meckel’s diverticulum
- Mallory-Weiss syndrome
- Anaemias
- Purpuras
- Haemophilia.
Gastric Tumours and Tumour-like Lesions:
- Tumour-like gastric lesions are polyps e.g. hyperplastic, fundic gland, and inflammatory fibroid polyps.
- Benign tumours are adenomas which may develop varying grades of dysplasia or intraepithelial neoplasia.
- The most common mesenchymal gastric tumour is the gastrointestinal stromal tumour (GIST). It is generally benign but 25% of cases, especially larger GISTs, may have features of malignancy.
- The most important etiologic factor in the pathogenesis of gastric carcinoma is the conversion of dietary nitrates into nitrites by bacteria which may be exogenous (by contamination) or
endogenous (by H. pylori). H. pylori-associated chronic gastritis and intestinal metaplasia are common preceding conditions. - The most common location of gastric carcinoma is the region of the pyloric canal.
- Early gastric carcinoma is an initial in situ stage.
- Advanced gastric carcinoma has several gross types: ulcerative, fungating, scirrhous, colloid, and ulcer cancer.
- A common histologic type is adenocarcinoma of varying grades. The most common patterns are tubular, papillary, mucinous and signet ring types.
- Gastric cancer may spread directly to adjacent structures and to distant sites by haematogenous and lymphatic routes.
- Other cancers in the stomach are lymphoma and carcinoid tumours.
Small Intestine
Normal Structure:
Anatomically, the small bowel has a length of 550-650 cm, includes the duodenum, jejunum and ileum and tends to become narrower throughout its course.
Histologically, the small bowel is identified by recognition of villi.
The wall of the small intestine consists of 4 layers:
1. The serosa is the outer covering of the small bowel which is complete except over a part of the duodenum.
2. The muscularis propria is composed of 2 layers of smooth muscle tissue an outer thinner longitudinal and an inner thicker circular layer. These muscles are functionally important for peristalsis.
Between the two layers of muscle lies the ganglionated plexus, the myenteric plexus of Auerbach.
3. The submucosa is composed of loose fibrous tissue with blood vessels and lacteals in it. It contains a ganglionated plexus, Meissner’s plexus, having fewer and smaller cells than Auerbach’s plexus.
4. The mucosa consists of glandular epithelium overlying the lamina propria composed of loose connective tissue and contains phagocytic cells and an abundance of lymphoid cells (Peyer’s patches in the ileum) and plasma cells.
It is supported externally by a thin layer of smooth muscle fibres, muscularis mucosae.
The mucous membrane is thrown into folds or plicae which are more in the jejunum and less in the ileum, thus increasing the absorptive surface enormously.
The absorptive surface is further increased by the intestinal villi.
Villi are finger-like or leaf-like projections which contain 3 types of cells:
Simple columnar cells perform absorptive function due to the presence of a brush border consisting of a large number of microvilli.
Goblet cells These are mucus-secreting cells and are interspersed between the columnar cells.
Endocrine cells These are scattered in the villi as well as are widely distributed throughout the gastrointestinal tract.
These cells have various synonyms as:
Kulchitsky cells, after the name of its discoverer.
Enterochromaffin cells, due to their resemblance to chromaffin cells of the adrenal medulla.
Argentaffin cells, as the intracytoplasmic granules stain positively with silver salts by a reduction reaction (argyrophilic cells, on the other hand, require the addition of an exogenous reducing substance for staining).
Endocrine cells, as these specialised cells are considered to be part of APUD cell system (having common properties such as Amine content, Amine Precursor Uptake and Decarboxylation).
APUD cells are considered to be endodermal in origin, while previously they were thought to be neural crest derivatives.
Other endocrine cells belonging to the APUD cell system are Ccells of the thyroid, chromaffin cells of the adrenal medulla, certain cells of the carotid body, bronchi, hypothalamus, pituitary and sympathetic ganglia.
Endocrine cells are heavily populated in the proximal small bowel as this is the most active site for absorption and secretory activities. They are sparse in the colon which is a less active site for such functions.
The duodenum contains distinctively branched Brunner’s glands present in the submucosa and going up to muscularis mucosae. The deeper layer of the mucosa of the small intestine elsewhere contains intestinal glands or crypts of Lieberkuhn. They are lined by columnar cells, goblet cells, endocrine cells and Paneth cells.
Paneth cells are normally exclusively found in the small intestine and occasionally in the caecum. These cells are characterised by the presence of supranuclear granules rich in lysozyme.
The blood supply of the whole of the small intestine, except the first part of the duodenum, is by the superior mesenteric artery which supplies blood by mesenteric arterial arcades and the straight arteries.
The main functions of the small intestine are digestion and absorption so that ultimately nutrients passing into the bloodstream are utilised by the cells in metabolism.
The mucosal layer of the small intestine has a remarkable capacity for regeneration and the new lining is laid every 3-4 days.
Conditions involving the intestine included in the discussion here are congenital anomalies, intestinal obstruction, ischaemic bowel disease, inflammatory bowel disease, enterocolitis, malabsorption syndrome, and small intestinal tumours.
Congenital Anomalies:
Meckel’s Diverticulum:
Meckel’s diverticulum is the most common congenital anomaly of the gastrointestinal tract, occurring in 2% of the population. It is more common in males.
The anomaly is commonly situated on the antimesenteric border of the ileum, about 1 meter above the ileocaecal valve. Like other true diverticula, Meckel’s diverticulum is an outpouching containing all the layers of the intestinal wall in their normal orientation.
It is almost always lined by a small intestinal type of epithelium; rarely it may contain islands of gastric mucosa and ectopic pancreatic tissue. The embryologic origin of Meckel’s diverticulum is from incomplete obliteration of the vitellointestinal duct (Other anomalies resulting from the remnants of the vitellointestinal duct are the vitelline sinus and vitelline cyst).
The common complications of Meckel’s diverticulum are perforation, haemorrhage and diverticulitis.
In addition to congenital Meckel’s diverticulum, acquired diverticula also occur in the small intestine. These are commonly multiple (diverticulosis), frequently located on the mesenteric border and are sometimes associated with malabsorption.
Other Anomalies:
There are a few uncommon anomalies of the small intestine:
Intestinal atresia is the congenital absence of the lumen, most commonly affecting the ileum or duodenum.
The proximal segment has a blind end which is separated from the distal segment freely, or the two segments are joined by a fibrous cord. The condition must be recognised early and treated surgically, as otherwise, it is incompatible with life.
Intestinal stenosis is a congenital narrowing of the lumen affecting a segment of the small intestine. The intestinal segment above the level of obstruction is dilated and below it is collapsed.
Intestinal malrotation is a developmental abnormality of the midgut (i.e. the portion of the intestine between the duodenojejunal flexure and the middle of the transverse colon).
Due to the failure of normal rotation of the midgut, the following consequences can occur:
- Exomphalos i.e. intestinal eventration at the umbilicus.
- Misplacement of the caecum, appendix and ascending colon.
- Mobile caecum.
Intestinal Obstruction:
Conditions which interfere with the propulsion of contents in the intestine are considered under the heading of intestinal obstruction.
The causes of intestinal obstruction can be classified under the following 3 broad groups:
1. Mechanical obstruction
It can occur as a result of the following causes:
- Internal obstruction (intramural and intraluminal)
-
- Inflammatory strictures (e.g. Crohn’s disease)
- Congenital stenosis, atresia, imperforate anus
- Tumours
- Meconium in mucoviscidosis
- Roundworms
- Gallstones, faecoliths, foreign bodies
- Ulceration induced by potassium chloride tablets prescribed to counter hypokalaemia.
2. External compression:
- Peritoneal adhesions and bands
- Strangulated hernias
- Intussusception
- Volvulus
- Intra-abdominal tumour.
2. Neurogenic obstruction: It occurs due to paralytic ileus i.e. paralysis of the muscles of the intestine as a result of shock after an abdominal operation or by acute peritonitis.
3. Vascular obstruction: Obstruction of the superior mesenteric artery or its branches may result in infarction causing paralysis.
The causes are as under:
- Thrombosis
- Embolism
- Accidental ligation.
Out of the various causes listed above, conditions producing external compression on the bowel wall are the most common causes of intestinal obstruction (80%). Some of these are described below.
Peritoneal Adhesions And Bands: Adhesions and bands in the peritoneum composed of fibrous tissue result following healing in peritonitis.
Rarely, such fibrous adhesions and bands may be without any preceding peritoneal inflammation and are of congenital origin. In either case, peritoneal bands and adhesions result in partial or complete intestinal obstruction by outside pressure on the bowel wall.
Hernia: Hernia is a protrusion of a portion of a viscus through an abnormal opening in the wall of its natural cavity. Inguinal hernias are more common, followed in decreasing frequency, by femoral and umbilical hernias.
When the contents of a hernia such as a loop of the intestine can be returned to the abdominal cavity, it is called reducible. When it is not possible to reduce a hernia due to large contents or due to adhesions in the hernial sac, it is referred to as irreducible.
When the blood flow in the hernial sac is obstructed, it results in strangulated hernia.
Obstruction to the venous drainage and arterial supply may result in infarction or gangrene of the affected loop of the intestine. The gross and microscopic appearance of the strangulated intestine is the same as that of infarction of the intestine.
Intussusception: Intussusception is the telescoping of a segment of the intestine into the segment below due to peristalsis. The telescoped segment is called the intussusceptum and the lower receiving segment is called the intussuscipiens.
The condition occurs more commonly in infants and young children, more often in the ileocaecal region when the portion of the ileum invaginates into the ascending colon without affecting the position of the ileocaecal valve. Less common forms are also-ileal and colo-colic intussusception.
In children, the cause is usually not known though enlargement of the lymphoid tissue in the terminal ileum has been suggested by some. In the case of adults, the usual causes are foreign bodies and tumours.
The main complications of intussusception are intestinal obstruction, infarction, gangrene, perforation and peritonitis.
Volvulus: Volvulus is the twisting of the loop of the intestine upon itself through 180° or more. This leads to obstruction of the intestine as well as cutting off the blood supply to the affected loop.
The usual causes are bands and adhesions (congenital or acquired) and long mesenteric attachment. The condition is more common in the sigmoid colon than in the small bowel.
Congenital Anomalies and Intestinal Obstruction:
- Meckel’s diverticulum is a common anomaly and is located in the ileum on the anti-mesenteric border about 1 meter above the ileocaecal valve.
- Intestinal atresia, stenosis and malrotation are uncommon anomalies.
- Intestinal obstruction may result from mechanical, neurogenic or vascular causes.
- Some of the common etiologies of intestinal obstruction are obstructed intestinal hernia, peritoneal bands, adhesions, intussusception and volvulus.
Ischaemic Bowel Disease:
Ischaemic lesions of the gastrointestinal tract may occur in the small intestine and/or colon; the latter is called ischaemic colitis or ischaemic enterocolitis and is commonly referred to as ischaemic bowel disease.
In either case, the cause of ischaemia is compromised mesenteric circulation, while the ischaemic effect is less likely to occur in the stomach, duodenum and rectum due to abundant collateral blood supply.
Depending upon the extent and severity of ischaemia, 3 patterns of pathologic lesions can occur:
1. Transmural infarction, characterised by full thickness involvement i.e. transmural ischaemic necrosis and gangrene of the bowel.
2. Mural infarction, characterised by haemorrhagic gastro enteropathy (haemorrhage and necrosis). The ischaemic effect in mural infarction is limited to the mucosa, submucosa and superficial muscular, while mucosal infarction is confined to mucosal layers superficial to muscularis mucosae.
3. Ischaemic colitis, due to chronic colonic ischaemia causing fibrotic narrowing of the affected bowel. These pathologic patterns are described below.
Transmural Infarction:
Ischaemic necrosis of the full thickness of the bowel wall is more common in the small intestine than in the large intestine.
Etiopathogenesis:
The common causes of transmural infarction of the small bowel are as under:
1. Mesenteric arterial thrombosis such as due to the following:
- Atherosclerosis (most common)
- Aortic aneurysm
- Vasospasm
- Fibromuscular hyperplasia
- Invasion by the tumour
- Use of oral contraceptives
- Arteritis of various types
2. Mesenteric arterial embolism arising from the following causes:
- Mural thrombi in the heart
- Endocarditis (infective and nonbacterial thrombotic)
- Atherosclerotic plaques
- Atrial myxoma
3. Mesenteric venous occlusion is a less common cause of full-thickness infarction of the bowel.
The causes are as under:
- Intestinal sepsis e.g. appendicitis
- Portal venous thrombosis in cirrhosis of the liver
- Tumour invasion
- Use of oral contraceptives
4. Miscellaneous causes:
- Strangulated hernia
- Torsion
- Fibrous bands and adhesions.
Morphologic Features: Grossly, irrespective of the underlying aetiology, infarction of the bowel is a haemorrhagic (red) type. A varying length of the small bowel may be affected.
In the case of colonic infarction, the distribution area of superior and inferior mesenteric arteries (i.e. splenic flexure) is more commonly involved.
The affected areas become dark purple and markedly congested and the peritoneal surface is coated with fibrinous exudate. The wall is thickened, oedematous and haemorrhagic.
The lumen is dilated and contains blood and mucus. In arterial occlusion, there is a sharp line of demarcation between the infarcted bowel and the normal intestine, whereas in venous occlusion the infarcted area merges imperceptibly into the normal bowel.
Microscopically, there is coagulative necrosis and ulceration of the mucosa and there are extensive submucosal haemorrhages. The muscular is less severely affected by ischaemia. Subsequently, inflammatory cell infiltration and secondary infection occur, leading to gangrene of the bowel.
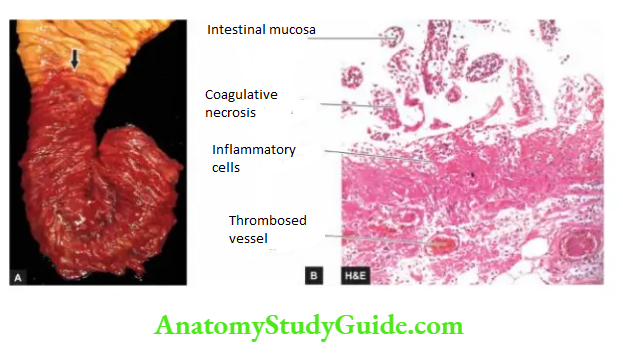
The condition is clinically characterised by ‘abdominal angina’ in which the patient has acute abdominal pain, nausea, vomiting, and sometimes diarrhoea. The disease is rapidly fatal, with a 50-70% mortality rate.
Mural And Mucosal Infarction (Haemorrhagic Gastroenteropathy):
Mural and mucosal infarctions are limited to superficial layers of the bowel wall, sparing the deeper layer of the muscular and the serosa. The condition is also referred to as haemorrhagic gastro enteropathy, and in the case of the colon as membranous colitis.
Etiopathogenesis: Haemorrhagic gastro enteropathy results from conditions causing nonocclusive hypoperfusion (compared to transmural infarction which occurs from occlusive causes).
These are as under:
- Shock
- Cardiac failure
- Infections
- Intake of drugs causing vasoconstriction e.g. digitalis, norepinephrine.
Morphologic Features:
Grossly, the lesions affect the variable length of the bowel. The affected segment of the bowel is red or purple but without haemorrhage and exudation on the serosal surface.
The mucosa is oedematous at places and sloughed and ulcerated at other places. The lumen contains haemorrhagic fluid.
Microscopically, there is patchy ischaemic necrosis of mucosa, vascular congestion, haemorrhages and inflammatory cell infiltrate.
The changes may extend into superficial muscularis but deeper layers of muscularis and serosa are spared.
Secondary bacterial infection may supervene resulting in pseudomembranous enterocolitis.
Clinically, as in transmural infarction, the features of abdominal pain, nausea, vomiting and diarrhoea are present, but the changes are reversible and curable.
With adequate therapy, normal morphology is completely restored in superficial lesions, while deeper lesions may heal by fibrosis leading to stricture formation.
Ischaemic Colitis:
Although this condition affects primarily the colon in the region of the splenic flexure, it is described here due to its apparent pathogenetic relationship with ischaemic injury.
Ischaemic colitis is characterised by chronic segmental colonic ischaemia followed by chronic inflammation and healing by fibrosis and scarring causing an obstruction (ischaemic stricture).
Grossly, the most frequently affected site is the splenic flexure; the other site is the rectum.
Ischaemic colitis passes through 3 stages:
Infarct, transient ischaemia and ischaemic stricture. However, the surgical submitted specimens generally are of the ischaemic stricture. The external surface of the affected area is fusiform or saccular.
On the cut section, there are patchy, segmental and longitudinal mucosal ulcers. Thus, the gross appearance can be confused with either of the two types of inflammatory bowel disease.
Microscopically, the ulcerated areas of the mucosa show granulation tissue. The submucosa is characteristically thickened due to inflammation and fibrosis.
The muscular may also show inflammatory changes and patchy replacement by fibrosis. The blood vessels may show atheromatous emboli, organising thrombi and endarteritis obliterans.
Necrotising Enterocolitis:
Necrotising enterocolitis is an acute inflammation of the terminal ileum and ascending colon, occurring primarily in premature and low-birth-weight infants within the first week of life and less commonly in full-term infants.
Etiology: The condition has been considered a variant of the spectrum of ischaemic bowel disease. Important factors in the aetiology of this disorder.
Thus, are as follows:
- Ischaemia
- Hypoxia/anoxia of the bowel due to bypassing of blood from the affected area
- Bacterial infection and endotoxins
- Establishment of feeding
- Infants fed on commercial formulae than breastfed, implying the role of immunoprotective factors.
Morphologic Features: Grossly, the affected segment of the bowel is dilated, necrotic, haemorrhagic and friable. The bowel wall may contain bubbles of air (pneumatosis intestinalis).
Microscopically, the changes are variable depending upon the stage. Initial changes are confined to the mucosa and show oedema, haemorrhage and coagulative necrosis.
A pseudomembrane composed of necrotic epithelium, fibrin and inflammatory cells may develop. As the ischaemic process extends to the subjacent layers, the muscle layer is also involved and may lead to perforation and peritonitis.
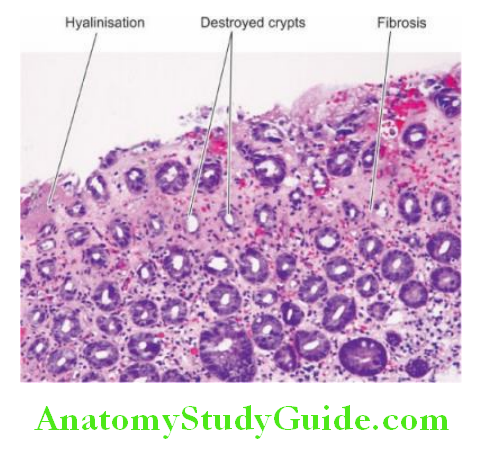
In healed cases, stricture formation, malabsorption and short bowel syndrome are the usual complications.
Ischaemic Bowel Disease:
- Ischaemic lesions of the small intestine and/or colon are called ischaemic enterocolitis.
- Depending upon the extent and severity of ischaemia, there are 3 patterns of pathologic lesions: transmural infarction, haemorrhagic gastro enteropathy and chronic ischaemic colitis.
- The causes for ischaemia may be due to mesenteric arterial or venous occlusion.
- Necrotising enterocolitis is an acute inflammation of the terminal ileum and ascending colon, occurring primarily in premature and low-birth-weight infants.
Inflammatory Bowel Disease (Crohn’S Disease And Ulcerative Colitis):
Definition:
The term ‘inflammatory bowel disease (IBD)’ is commonly used to include 2 idiopathic bowel diseases having many similarities but the conditions usually have a distinctive morphological appearance.
These 2 conditions are Crohn’s disease (regional enteritis) and ulcerative colitis:
Crohn’s disease or Regional enteritis: is an idiopathic chronic ulcerative IBD, characterised by transmural, non-caseating granulomatous inflammation, affecting most commonly the segment of the terminal ileum and/or colon, though any part of the gastrointestinal tract may be involved.
Ulcerative colitis: is an idiopathic form of acute and chronic ulcers-inflammatory colitis affecting chiefly the mucosa and submucosa of the rectum and descending colon, though sometimes it may involve the entire length of the large bowel.
Both these disorders primarily affect the bowel but may have systemic involvement in the form of polyarthritis, uveitis, ankylosing spondylitis, skin lesions and hepatic involvement.
Both diseases can occur at any age but are more frequent in 2nd and 3rd decades of life. Females are affected slightly more often.
Etiopathogenesis:
The exact aetiology of IBD remains unknown.
However, multiple factors are implicated which can be considered under the following 4 groups:
1. Genetic factors implicated in the etiopathogenesis of IBD are supported by the following pieces of evidence
- There is about a 3 to 20 times higher incidence of occurrence of IBD in first-degree relatives. This is due to genetic defects causing diminished epithelial barrier function.
- Overall, there is approximately a 50% chance of development of IBD (Crohn’s disease with about 60% concordance, ulcerative colitis with about 6% concordance) in monozygotic twins.
Although no specific and consistent gene association with IBD has been seen, a genome-wide search has revealed that disease-predisposing loci are present in chromosomes 16q, 12p, 6p, 14q and 5q.
In particular, CARD15 (caspase-associated recruitment domain-containing protein 15) on chromosome 16q is expressed by several cells in the intestinal mucosa which in mutated form results in loss of its function and predisposes an individual to about 50-times higher risk to develop Crohn’s disease.
HLA studies show that ulcerative colitis is more common in HLA-DRB1-alleles while Crohn’s disease is more common in HLA-DR7 and DQ4 alleles.
2. Immunologic factors: Defective immunologic regulation in IBD has been shown to play a significant role in the pathogenesis of IBD
1. Defective regulation of immune suppression In a normal individual, there is a lack of immune responsiveness to dietary antigens and commensal flora in the intestinal lumen.
The mechanism responsible for this is by activation of CD4+ T cells secreting cytokines inhibitory to inflammation (IL-10, TGF-ß) which suppress inflammation in the gut wall.
In IBD, this immune mechanism of suppression of inflammation is defective and thus results in uncontrolled inflammation.
2. Transgenic mouse experimental model studies Gene ‘knock out’ studies on colitis in mice have revealed that multiple immune abnormalities may be responsible for IBD as under
- Deletion of inflammation inhibitory cytokines (e.g. IL-2, IL-10, TGF-ß) or their receptors.
- Deletion of molecules responsible for T cell recognition (e.g. T cell antigen receptors, MHC).
- Interference with normal epithelial barrier function in the intestine (e.g. blocking N-cadherin, deletion of multidrug resistance MDR gene).
3. Type of inflammatory cells In both types of IBD, activated CD4+ T cells are present in the lamina propria and in the peripheral blood.
These cells either activate other inflammatory cells (e.g. macrophages and B cells) or recruit more inflammatory cells by stimulation of homing receptors on leucocytes and vascular endothelium.
There are two main types of CD4+ T cells in IBD:
TH1 cells secrete proinflammatory cytokines IFN-? and TNF which induce transmural granulomatous inflammation seen in Crohn’s disease. IL-12 initiates the TH1 cytokine pathway.
TH2 cells secrete IL-4, IL-5 and IL-13 which induce superficial mucosal inflammation, characteristically seen in ulcerative colitis.
3. Microbial factors: At different times, the role of a variety of microbes in the initiation of an inflammatory response by the body has been suspected.
Accordingly, several microorganism species (bacteria, viruses, protozoa and fungi have been suspected but without definite evidence Mycobacterium paratuberculosis, Salmonella, Shigella, Helicobacter, Clostridia, Bacteroides, Escherichia, Measles virus etc.
It has been seen that infants who have been administered antibiotics have a three times higher frequency of childhood IBD due to disturbed commensal microbial flora.
4. Environmental factors: In addition to the role of genetic factors, deranged T-cell mediated immunity and deranged microbiota, a role for several exogenous and environmental factors have been assigned
NSAIDs Long-term administration of nonsteroidal anti-inflammatory drugs is associated with a higher incidence of IBD.
Psychosocial factors It has been observed that individuals who are unduly sensitive, dependent on others and unable to express themselves, or some major life events such as illness or death in the family, divorce, interpersonal conflicts etc, suffer from the irritable colon or have an exacerbation of symptoms.
Patients of IBD in the West have been found to suffer from greater functional impairment as assessed by the sickness impact profile which is a measure of overall psychological and physical functioning.
Smoking Role of smoking in the causation of Crohn’s disease has been reported.
Oral contraceptives An increased risk to develop Crohn’s disease with long-term use of oral contraceptives has been found in some studies but there is no such increased risk for ulcerative colitis.
Thus, the consensus hypothesis in the pathogenesis of IBD involves the interlinked role of major etiologic factors i.e. in a genetically predisposed individual, the effects of environmental factors and deranged microbiota, resulting in dysregulation of mucosal immune function, which leads to chronic inflammation in IBD.
Morphologic Features:
The morphologic features of Crohn’s disease and ulcerative colitis are sufficiently distinctive so as to be classified separately.
Crohn’s Disease: Crohn’s disease may involve any portion of the gastrointestinal tract but affects most commonly 15-25 cm of the terminal ileum which may extend into the caecum and sometimes into the ascending colon.
Grossly, the characteristic feature is the multiple, well-demarcated segmental bowel involvement with intervening uninvolved ‘skip areas’.
The wall of the affected bowel segment is thick and hard, resembling a ‘hose pipe’. Serosa may be studded with minute granulomas.
The lumen of the affected segment is markedly narrowed. The mucosa shows ‘serpiginous ulcers’ while intervening surviving mucosa is swollen giving a ‘cobblestone appearance’. There may be deep fissuring into the bowel wall.
Histologically,
the characteristic features are as follows:
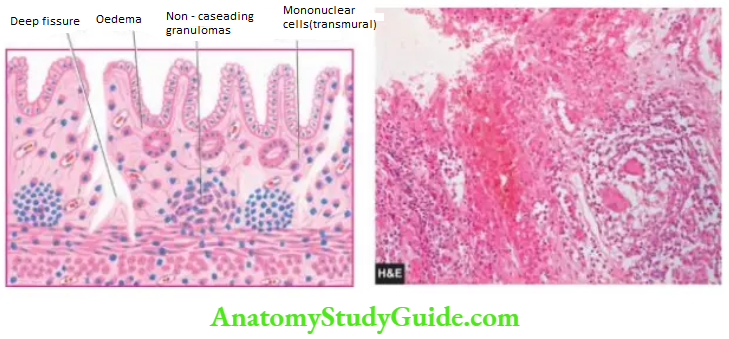
1. Transmural inflammatory cell infiltrate consisting of chronic inflammatory cells (lymphocytes, plasma cells and macrophages) is the classical microscopic feature.
2. Non-caseating, sarcoid-like granulomas are present in all the layers of the affected bowel wall in 60% of cases and may even be seen in the regional lymph nodes.
3. There is patchy ulceration of the mucosa which may take the form of deep fissures, accompanied by inflammatory infiltration of lymphocytes and plasma cells.
4. There is a widening of the submucosa due to oedema and foci of lymphoid aggregates.
5. In more chronic cases, fibrosis becomes increasingly prominent in all the layers disrupting the muscular layer.
Ulcerative Colitis: Classically, ulcerative colitis begins in the rectum, and in continuity extends upwards into the sigmoid colon, descending colon, transverse colon, and sometimes may involve the entire colon.
The colonic contents may rarely backflow into the terminal ileum in continuity, causing ‘back-wash ileitis’ in about 10% of cases.
Grossly, the characteristic feature is the continuous involvement of the rectum and colon without any uninvolved skip areas compared to Crohn’s disease.
The appearance of the colon may vary depending upon the stage and intensity of the disease because of remissions and exacerbations.
Mucosa shows linear and superficial ulcers, usually not penetrating the muscular layer. The intervening intact mucosa may form inflammatory ‘pseudopolyps.’
The muscle layer is thickened due to contraction, producing shortening and narrowing of the affected colon with loss of normal haustral folds giving a ‘garden-hose appearance’.
Histologically, ulcerative colitis because of remission and exacerbations, is characterised by alternating ‘active disease process’ and ‘resolving colitis.’
The changes in the ‘active disease process’ are as under:
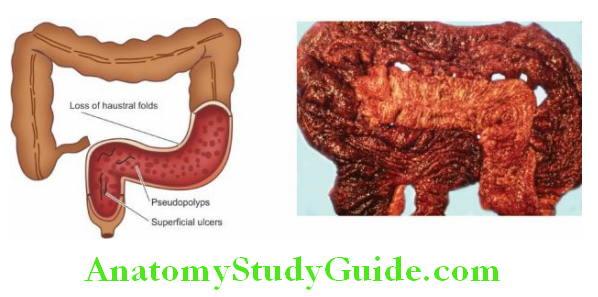
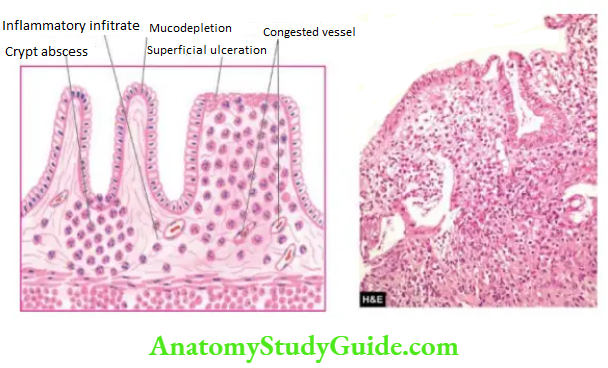
- Crypt distortion, cryptitis and focal accumulations of neutrophils forming crypt abscesses.
- Marked congestion, dilatation and haemorrhages from mucosal capillaries.
- Superficial mucosal ulcerations, usually not penetrating into the muscle coat, except in severe cases, and are accompanied by nonspecific inflammatory cell infiltrate of lymphocytes, plasma cells, neutrophils, some eosinophils and mast cells in the lamina propria.
- Goblet cells are markedly diminished in cases of active disease.
- Areas of mucosal regeneration and immunodepletion of lining cells.
- In long-standing cases, epithelial cytologic atypia ranges from mild to marked dysplasia and sometimes develops into carcinoma in situ and frank adenocarcinoma
Complications:
Complications of Crohn’s disease and ulcerative colitis are as under:
Crohn’s Disease:
- Malabsorption due to impaired absorption of fat, vitamin B12, proteins and electrolytes from the diseased small bowel.
- Fistula formation may occur in long-standing cases. These may be internal fistulae between the loops of the intestine, or external fistulae such as enterocutaneous, rectal and anal fistulae.
- Stricture formation may occur in chronic cases due to extensive fibrosis in the affected bowel wall.
- Development of malignancy in the small intestine as a late complication of Crohn’s disease is rarer than that in ulcerative colitis, but lymphoma may develop more often in Crohn’s disease than adenocarcinoma (seen in some long-standing cases of ulcerative colitis).
Ulcerative Colitis:
1. Toxic megacolon (Fulminant colitis) acute fulminating colitis in which the affected colon is thin-walled and dilated and is prone to perforation and faecal peritonitis. There is deep penetration of the inflammatory cell infiltrate into the muscle layer which is disrupted.
2. Perianal fistula formation may occur rarely.
3. Carcinoma may develop in long-standing cases of ulcerative colitis of more than 10 years duration.
4. Stricture formation almost never occurs in ulcerative colitis. The distinguishing features of the two conditions are summarised.
Inflammatory Bowel Disease:
- Inflammatory bowel disease (IBD) includes 2 idiopathic bowel diseases: Crohn’s disease (regional enteritis) and ulcerative colitis.
- Crohn’s disease is an idiopathic chronic ulcerative IBD, characterised by transmural, noncaseating granulomatous inflammation, affecting most commonly the segment of the terminal ileum and/or colon.
- Ulcerative colitis is an idiopathic acute and chronic ulcer-inflammatory colitis affecting chiefly the mucosa and submucosa of the rectum and descending colon.
- The exact aetiology of IBD is not known but genetic, microbial, immunologic and a few environmental factors have been implicated.
- Pathogenesis of IBD combines the interlinked role of three factors: genetic predisposition microbial flora and environmental factors, which lead to immune dysregulation that causes chronic inflammation.
- Complications of IBD are malabsorption, formation of fistula and stricture, toxic megacolon and development of malignancy in late cases.
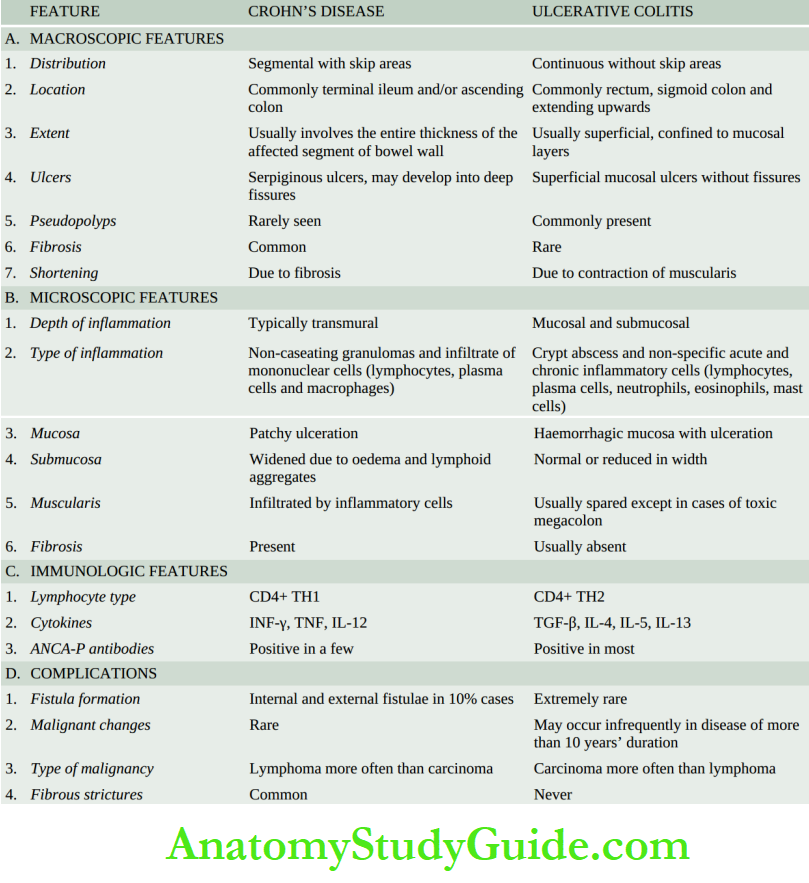
Infective And Other Enterocolitis:
Besides IBD, a variety of other acute and chronic inflammatory conditions affect small bowel (enteritis), large bowel (colitis), or both (enterocolitis); the last name is more common.
Hence, all these conditions involving small bowel and/or large bowel are described together here for better correlation of features.
After excluding IBD, various forms of inflammations of the bowel can be categorised broadly into infective enterocolitis and pseudomembranous enterocolitis.
Infective Enterocolitis:
These are a group of acute and chronic inflammatory lesions of the small intestine and/or colon caused by microorganisms (bacteria, viruses, fungi, protozoa and helminths).
All these are characterised by diarrhoeal syndromes. Pathogenetically speaking, these microorganisms can cause enterocolitis by 2 mechanisms by enteroinvasive bacteria producing ulcerative lesions, and by enterotoxin-producing bacteria resulting in non-ulcerative lesions.
A list of common microorganisms producing enterocolitis is presented in Table. A few important forms are described below.
Intestinal Tuberculosis:
Intestinal tuberculosis can occur in 3 forms primary, secondary and hyperplastic caecal tuberculosis.
1. Primary Intestinal Tuberculosis: Though an uncommon disease in the developed countries of the world, primary tuberculosis of the ileocaecal region is quite common in developing countries including India.
In the pre-pasteurisation era, it used to occur by ingestion of unpasteurised cow’s milk infected with Mycobacterium bovis.
But nowadays due to the control of tuberculosis in cattle and the pasteurisation of milk, virtually all cases of intestinal tuberculosis are caused by M. tuberculosis.
The predominant changes are in the mesenteric lymph nodes without any significant intestinal lesions.
Grossly, the affected lymph nodes are enlarged, matted and caseous (tabes mesenteric). Eventually, there is healing by fibrosis and calcification.
Microscopically, in the initial stage, there is a primary complex or Ghon’s focus in the intestinal mucosa as occurs elsewhere in primary tuberculous infection.
Subsequently, the mesenteric lymph nodes are affected which show a typical tuberculous granulomatous inflammatory reaction with caseation necrosis. Tuberculous peritonitis may occur due to the spread of the infection.
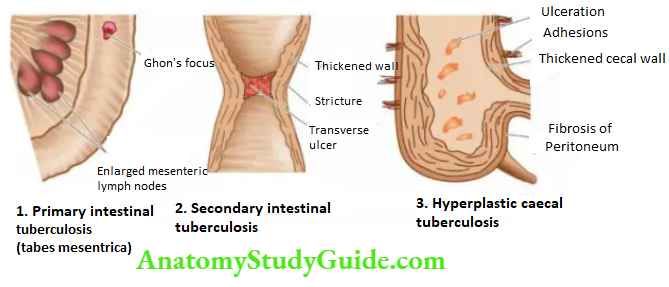
2. Secondary Intestinal Tuberculosis: Self-swallowing of sputum in patients with active pulmonary tuberculosis may cause secondary intestinal tuberculosis, most commonly in the terminal ileum and rarely in the colon.
Grossly, the intestinal lesions are more prominent than the lesions in regional lymph nodes as in secondary pulmonary tuberculosis.
The lesions begin in the Peyer’s patches or the lymphoid follicles with the formation of small ulcers that spread through the lymphatics to form large ulcers which are transverse to the long axis of the bowel (compared to typhoid ulcers of the small intestine).
These ulcers may be coated with caseous material.
Serosa may be studded with visible tubercles. In advanced cases, transverse fibrous strictures and intestinal obstruction are seen.
Histologically, the tuberculous lesions in the intestine are similar to those observed elsewhere i.e. presence of tubercles.
Mucosa and submucosa show ulceration and the muscularis may be replaced by a variable degree of fibrosis. Tuberculous peritonitis may be observed.
1. Bacterial enterocolitis
1. Entero-invasive bacteria
- M. Tuberculosis
- Salmonella
- Campylobacter jejuni
- Shigella
- Escherichia coli
- Yersinia enterocolitica
2. Enterotoxin-producing bacteria
- Vibrio cholera
3. Viral enterocolitis
4. Fungal enterocolitis
- Candida
- Mucor
4. Protozoal and metazoal infestations
- Giardia lamblia
- Entamoeba histolytica
- Balantidium coli
- Taenia solium
- Ascaris lumbricoides
- Ancylostoma duodenale
- Strongyloides stercoralis
3. Hyperplastic Ileocaecal Tuberculosis: This is a variant occurring secondary to pulmonary tuberculosis.
Grossly, the terminal ileum, caecum and/or ascending colon are thick-walled with mucosal ulceration. Clinically, the lesion is palpable and may be mistaken for carcinoma.
Microscopically, the presence of caseating tubercles distinguishes the condition from Crohn’s disease in which granulomas are non-caseating.
Besides, bacteriological evidence by culture or animal inoculation and the Mantoux test is helpful in the differential diagnosis of the two conditions.
Enteric Fever:
The term enteric fever is used to describe acute infection caused by Salmonella typhi (typhoid fever) or Salmonella paratyphi (paratyphoid fever). Besides these 2 salmonellae, Salmonella typhimurium causes food poisoning.
Pathogenesis: Typhoid bacilli are ingested through contaminated food or water. During the initial asymptomatic incubation period of about 2 weeks, the bacilli invade the lymphoid follicles and Peyer’s patches of the small intestine and proliferate.
Following this, the bacilli invade the bloodstream causing bacteraemia, and the characteristic clinical features of the disease like continuous rise in temperature and ‘rose spots’ on the skin are observed.
Immunological reactions (Widal’s test) begin after about 10 days and peak titres are seen by the end of the third week.
Eventually, the bacilli are localised in the intestinal lymphoid tissue (producing typhoid intestinal lesions), in the mesenteric lymph nodes (leading to haemorrhagic lymphadenitis), in the liver (causing foci of parenchymal necrosis), in the gallbladder (producing typhoid cholecystitis), and in the spleen (resulting in splenic reactive hyperplasia).
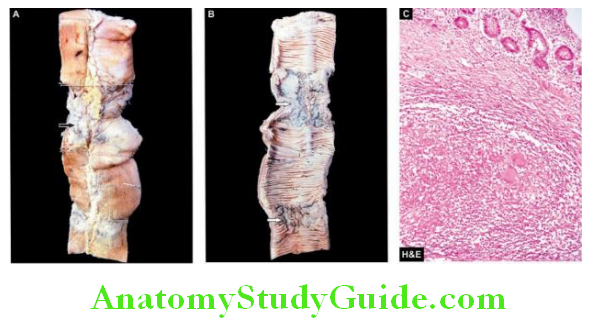
Morphologic Features: The lesions are observed in the intestines as well as in other organs.
1. Intestinal Lesions:
Grossly, the terminal ileum is affected most often, but lesions may be seen in the jejunum and colon.
Peyer’s patches show oval typhoid ulcers with their long axis along the length of the bowel (compared to tuberculous ulcers of the small intestine, described already).
Salient contrasting features of tuberculosis and typhoid ulcers. The base of the ulcers is black due to sloughed mucosa.
The margins of the ulcers are slightly raised due to inflammatory oedema and cellular proliferation.
There is never significant fibrosis and hence fibrous stenosis seldom occurs in healed typhoid lesions. The regional lymph nodes are invariably enlarged.
Microscopically, there is hyperaemia, oedema and cellular proliferation consisting of phagocytic histiocytes (showing characteristic erythrophagocytosis), lymphocytes and plasma cells.
Though enteric fever is an example of acute inflammation, neutrophils are invariably absent from the cellular infiltrate and this is reflected in the leucopenia with neutropenia and relative lymphocytosis in the peripheral blood.
The main complications of the intestinal lesions of typhoid are perforation of the ulcers and haemorrhage.
2. Other Lesions: Besides the intestinal involvement, various other organs and tissues showing pathological changes in enteric fever are as under:
- Mesenteric lymph nodes haemorrhagic lymphadenitis
- Liverfoci of parenchymal necrosis
- Gallbladdertyphoid cholecystitis
- Spleensplenomegaly with reactive hyperplasia
- Kidneysnephritis
- abdominal musclesZenker’s degeneration
- joints arthritis
- bone osteomyelitis
- Meninges meningitis
- Testis orchitis
- The persistence of organisms in the gallbladder or urinary tract may result in the passage of organisms in the faeces or urine creating a ‘carrier state’ which is a source of infection to others.
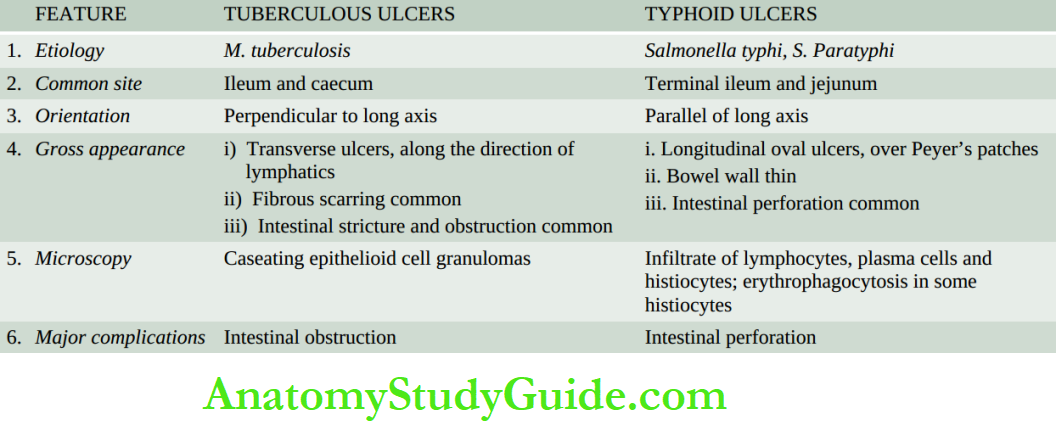
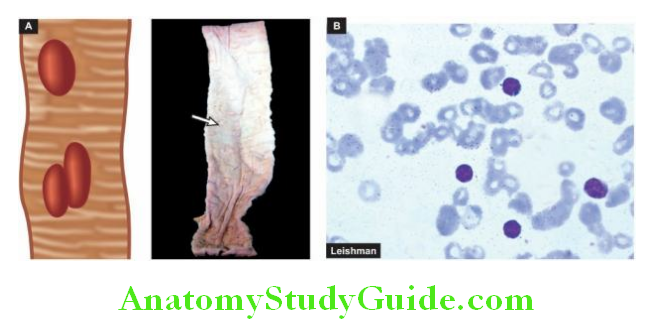
Bacterial Food Poisoning:
This is a form of acute bacterial illness that occurs following ingestion of food or water contaminated with bacteria other than those that cause specific acute intestinal infections like typhoid, paratyphoid, cholera or dysentery bacilli.
The illness results from either bacterial invasion or bacterial toxigenic effects on the bowel.
The commonest causes of bacterial food poisoning resulting in enteritis or enterocolitis are as under:
1. Staphylococcal food poisoning: Staphylococcus aureus infection acquired from contaminated food produces either mild food poisoning by enterotoxins or may cause a more severe form of the illness called pseudomembranous enterocolitis.
Staphylococcal food poisoning occurs due to the liberation of enterotoxins by the bacteria.
2. Clostridial food poisoning: Infection with anaerobic organisms Clostridium welch, following consumption of contaminated meat results in acute food poisoning. The illness occurs both by bacterial invasion as well as by toxins.
3. Botulism: This is a severe form of paralysing illness caused by the ingestion of the organism, Clostridium botulinum, which produces a neurotoxin.
4. Salmonella food poisoning (Salmonellosis): This is an infection (and not caused by toxins) occurring due to food contaminated by S. Typhimurium or S. enteritidis. The condition manifests with fever, vomiting, and diarrhoea. Death may result from the depletion of water and electrolytes.
Dysenteries:
The term ‘dysentery’ is used to mean diarrhoea with abdominal cramps, tenesmus and passage of mucus in the stools. Based on aetiology, there are 2 main forms of dysenteries bacillary and amoebic.
1. Bacillary Dysentery: Bacillary dysentery is the term used for infection by Shigella species: S. dysenteriae, S. flexneri, S. boydii and S. sonnei.
Infection occurs by the faecal-oral route and is seen with poor personal hygiene, in densely populated areas, and with contaminated food and water. The common housefly plays a role in the spread of infection.
Grossly, the lesions are mainly found in the colon and occasionally in the ileum.
Superficial transverse ulcerations of the mucosa of the bowel wall occur in the region of lymphoid follicles but perforation is seldom seen.
The intervening intact mucosa is hyperaemic and oedematous. Following recovery from the acute attack, complete healing usually takes place.
Microscopically, the mucosa overlying the lymphoid follicles is necrosed.
The surrounding mucosa shows congestion, oedema and infiltration by neutrophils and lymphocytes.
The mucosa may be covered by a greyish-yellow ‘pseudomembrane’ composed of fibrinosuppurative exudate.
The complications of bacillary dysentery are haemorrhage, perforation, stenosis, polyarthritis and iridocyclitis.
2. Amoebic Dysentery: This is due to infection by Entamoeba histolytica. It is more prevalent in tropical countries and primarily affects the large intestine. Infection occurs from ingestion of the cyst form of the parasite.
The cyst wall is dissolved in the small intestine from where the liberated amoebae pass into the large intestine.
Here, they invade the epithelium of the mucosa, reach the submucosa and produce the characteristic flask-shaped ulcers.
Grossly, early intestinal lesions appear as small areas of elevation on the mucosal surface. In advanced cases, typical flask-shaped ulcers having narrow necks and broad bases are seen. They are more conspicuous in the caecum, rectum and in flexures.
Microscopically, the ulcerated area shows a chronic inflammatory reaction consisting of lymphocytes, plasma cells, macrophages and eosinophils.
The trophozoites of Entamoeba are seen in the inflammatory exudate and are concentrated at the advancing margin of the lesion.
Intestinal amoebae characteristically have ingested red cells in their cytoplasm.
Oedema and vascular congestion are present in the area surrounding the ulcers.
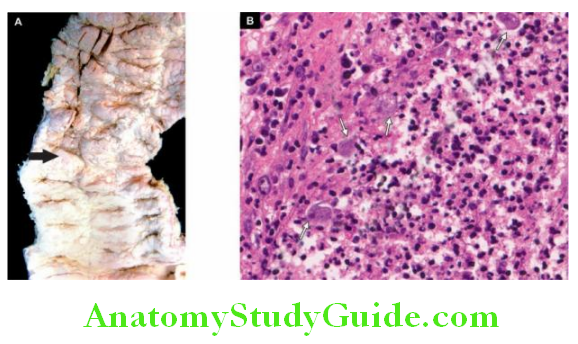
Complications of intestinal amoebic ulcers are amoebic liver abscess or amoebic hepatitis, perforation, haemorrhage and formation of amoeboma which is a tumour-like mass.
Pseudomembranous Enterocolitis (Antibiotic-Associated Diarrhoea):
Pseudomembranous enterocolitis is a form of acute inflammation of the colon and/or small intestine characterised by the formation of a ‘pseudomembrane’ over the site of mucosal injury.
Etiology: Numerous studies have established the overgrowth of Clostridium difficile with the production of its toxin in the aetiology of antibiotic-associated diarrhoea culminating in pseudomembranous colitis.
Oral antibiotics such as clindamycin, ampicillin and cephalosporins are more often (20%) associated with antibiotic-associated diarrhoea, while the development of pseudomembranous colitis may occur in 1-10% of cases.
Pseudomembrane formation may also occur in various other conditions as:
- Staphylococcal enterocolitis
- Bacillary (Shigelldysentery
- Candida enterocolitis
Morphologic Features: Grossly, the lesions may be confined, to the large intestine or small intestine, or both may be involved.
The mucosa of the bowel is covered by patchy, raised yellow-white plaques. Elsewhere, the mucosa is congested and may show small mucosal ulcerations.
Microscopically, the ‘pseudomembrane’ is composed of a network of fibrin and mucus,
which are entangled inflammatory cells and mucosal epithelial cells.
There is focal necrosis of surface epithelial cells. The lamina propria contains inflammatory cell infiltrate, mainly neutrophils.
The submucosa has congested capillaries and may show microthrombi. The inflammation spreads laterally rather than deeply.
Infective and Other Enterocolitis:
- Infective enterocolitis is caused by bacteria, viruses, fungi, protozoa and metazoa. Intestinal tuberculosis can occur as primary, secondary or hyperplastic ileocaecal type.
- In a classic case, there are multiple transverse ulcers and strictures causing intestinal obstruction.
- Enteric fever is an acute infection caused by Salmonella (typhoid fever). There are oval ulcers along the long axis of the small intestine which may be complicated by perforation.
- Bacterial food poisoning may be caused by staphylococci, Clostridia, and Salmonella.
- Dysentery is diarrhoea with abdominal cramps, tenesmus and passage of mucus in the stools, and may be bacillary and amoebic.
- Pseudomembranous enterocolitis is the acute inflammation of the colon and/or small intestine with the formation of a ‘pseudomembrane’. There is often an overgrowth of Clostridium difficile.
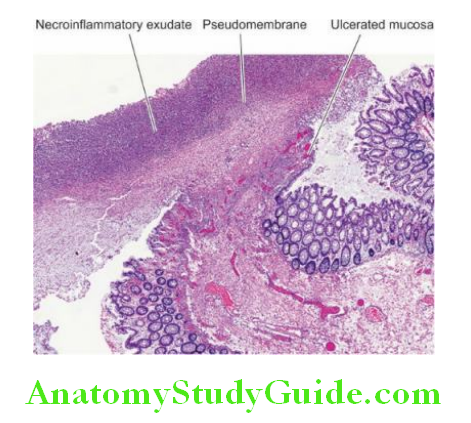
Malabsorption Syndrome:
Definition And Classification:
The malabsorption syndrome (MAS) is characterised by impaired intestinal absorption of nutrients especially of fat; some other substances are proteins, carbohydrates, vitamins and minerals.
MAS is subdivided into 2 broad groups:
Primary MAS is due to a primary deficiency of the absorptive mucosal surface and of the associated enzymes.
Secondary MAS, in which mucosal changes result secondary to other factors such as diseases, surgery, trauma and drugs. Each of the two main groups has a number of causes listed in Table.
Clinical Features:
The clinical manifestations of MAS vary according to the underlying cause.
However, some common symptoms are as follows:
- Steatorrhoea (pale, bulky, foul-smelling stools)
- Chronic diarrhoea
- Abdominal distension
- Barborygmi and flatulence
- Anorexia
- Weight loss
- Muscle wasting
- Dehydration
- Hypotension
- Specific malnutrition and vitamin deficiencies depend upon the cause.
Rimary Malabsorption
- Coeliac Sprue
- Collagenous Sprue
- Tropical Sprue
- Whipple’S Disease
- Disaccharidase Deficiency
- Allergic And Eosinophilic Gastroenteritis
2. Secondary Malabsorption
- Impaired Digestion
- Mucosal Damage E.G. In Tuberculosis, Crohn’S Disease, Lymphoma, Amyloidosis, Radiation Injury, Systemic Sclerosis
- Hepatic And Pancreatic Insufficiency
- Resection Of Bowel
- Drugs E.G. Methotrexate, Neomycin, Phenindione Etc.
2. Impaired Absorption
- Short Or Stagnant Bowel (Blind Loop Syndrome) From Surgery Or Disease Resulting In Abnormal Proliferation Of Microbial Flora
- Acute Infectious Enteritis
- Parasitoses E.G. Giardia, Strongyloides, Hookworms
3. Impaired Transport
- Lymphatic Obstruction E.G. In Lymphoma Tuberculosis, Lymphangiectasia
- Abetalipoproteinaemia
Investigations:
When MAS is suspected on clinical grounds, the following investigations (laboratory tests and endoscopic biopsy) may be carried out to confirm it:
Laboratory Tests:
1. Tests for fat malabsorption:
- Faecal analysis for fat content
- Microscopic analysis for faecal fat
- Blood lipid levels after a fatty meal
- Tests based on the absorption of radioactive-labelled fat.
2. Tests for protein malabsorption:
- Bile acid malabsorption
- Radioactive-labelled glycine breath test.
- Prothrombin time (vitamin K deficiency)
- Secretin and other pancreatic tests.
3. Tests for carbohydrate malabsorption:
- D-xylose tolerance test
- Lactose tolerance test
- Hydrogen breath test
- Bile acid breath test
4. Vitamin B12, malabsorption:
- Schilling test.
- Intestinal Mucosal Biopsy
A mucosal biopsy of the small intestine is essential for making the diagnosis of MAS and also evaluation of a patient on follow-up.
The availability of endoscopes has enabled easy viewing of affected mucosa directly and taking mucosal biopsy under vision; this has largely replaced the earlier per-oral Crosby-Kugler capsule biopsy of the small intestine.
The biopsy should first be examined under a dissecting microscope before histologic sectioning. Normal villous.
Under the dissecting microscope, the normal jejunal mucosa has tall, slender, finger-shaped or leaf-shaped villi.
It is lined by tall columnar absorptive epithelium and has scattered lymphocytes in the lamina propria.
Villous atrophy Variable degree of flattening of intestinal mucosa in MAS is the commonest pathological change in mucosal pattern and is referred to as villous atrophy.
It may be of 2 types partial and subtotal/total type. Partial villous atrophy is the mild form of the lesion in which villi fuse with each other and thus become short and broad, commonly called convolutions and irregular ridges.
The epithelial cells show compensatory hyperplasia suggesting a turnover of these cells. Lamina propria shows increased cellular infiltrate, predominantly of plasma cells.
Partial villous atrophy is commonly found in children and adults with diarrhoea, parasitic infestations, Crohn’s disease, ulcerative colitis and malabsorption due to drugs and radiation injury.
Subtotal/Total villous atrophy is the severe form of the lesion in which there is flattening of mucosa due to more advanced villous fusion. The surface epithelium is cuboidal and there is increased plasma cell infiltrate in the lamina propria.
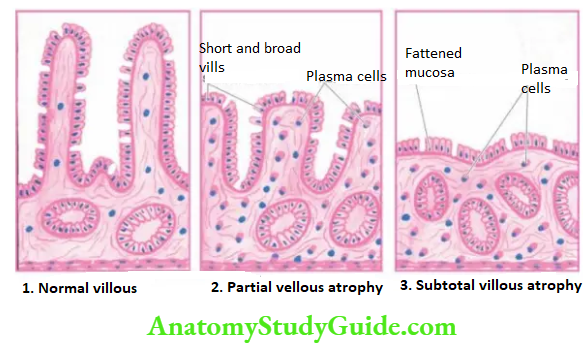
Subtotal and total villous atrophy is exhibited by a number of conditions such as nontropical sprue, tropical sprue, intestinal lymphomas, carcinoma, protein-calorie malnutrition etc.
Important Types Of Mas:
Coeliac Sprue:
Coeliac sprue, also known as non-tropical sprue, gluten-sensitive enteropathy or idiopathic steatorrhoea, is the most important cause of primary malabsorption occurring in temperate climates.
The condition is characterised by significant loss of villi in the small intestine and therefore diminished absorptive surface area.
The condition occurs in 2 forms:
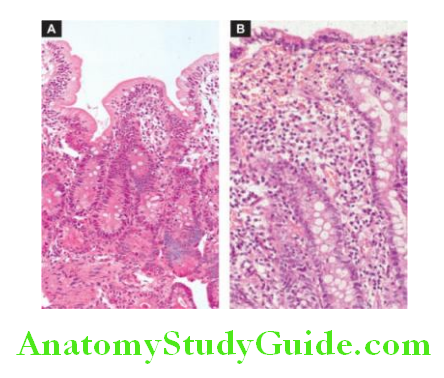
Childhood form is seen in infants and children and is commonly referred to as coeliac disease. Adult form, seen in adolescents and early adult life and used to be called idiopathic steatorrhoea.
In either case, there is genetic abnormality resulting in sensitivity to gluten (a protein) and its derivative, gliadin, present in diets such as grains of wheat, barley and rye.
Circulating antibodies to tissue transglutaminase IgA antigliadin and IgA anti-endomysial, are found in these patients and are helpful for making diagnoses.
The symptoms are usually relieved by the elimination of gluten from the diet.
The role of heredity is further supported by the observation of familial incidence and HLA association of the disease (HLA-DQ2 or HLA-DQ8). The exact pathogenesis of the condition is not clear.
However, the following hypotheses have been proposed for causing mucosal cell damage:
1. Hypersensitivity reaction as seen by gluten-stimulated antibodies.
2. Toxic effect of gluten due to inherited enzyme deficiency in the mucosal cells. Histologically, there are no differences in the pathological findings in children and adults.
There is a variable degree of flattening of the mucosa, particularly of the upper jejunum, and to some extent of the duodenum and ileum.
The surface epithelial cells are cuboidal or low columnar type.
There may be partial villous atrophy which is the replacement of normal villous pattern by convolutions, or subtotal villous atrophy characterised by a flat mucosal surface.
There is a variable degree of crypt hyperplasia.
Lamina propria shows an increased number of plasma cells and lymphocytes with increased intraepithelial lymphocytes.
The major sequela of long-term coeliac sprue is the increased incidence of intestinal carcinoma in these cases.
Collagenous Sprue:
This entity is regarded as the end result of coeliac sprue in which the villi are totally absent (total villous atrophy) and there are unique and diagnostic broad bands of collagen under the basal lamina of surface epithelium.
The condition is refractory to any treatment and the course is generally fatal. Some workers consider collagenous sprue as a variant of coeliac sprue without classifying it separately.
Tropical Sprue:
This disease, as the name suggests, occurs in individuals living in or visiting tropical areas such as Caribbean countries, South India, Sri Lanka and Hong Kong.
Pathogenesis of the condition is not clear but there is evidence to support enterotoxin production by some strains of E.
coli which causes intestinal injury. Severe cases are characterised by additional features such as macrocytic anaemia, glossitis and emaciation due to intestinal malabsorption of vitamin B12 and folate.
Histologically, there is usually partial villous atrophy and sometimes subtotal atrophy.
The lesions are relieved by removal of the patient from the tropical area and by oral administration of antibiotics but a gluten-free diet has no role in improvement.
Whipple’s Disease (Intestinal Lipodystrophy):
This is an uncommon bacterial disease involving not only the intestines but also various other systems such as the central nervous system, heart, blood vessels, skin, joints, lungs, liver, spleen and kidneys.
The disease is more common in males in the 4th to 5th decades of life.
Patients may present with features of malabsorption or may have an atypical presentation in the form of migratory polyarthritis, neurological disturbances and focal hyperpigmentation of the skin.
Histologically, the affected tissues show the presence of characteristic macrophages containing PAS-positive granules and rod-shaped micro-organisms (Whipple’s bacilli).
These macrophages are predominantly present in the lamina propria of the small intestine and mesenteric lymph nodes.
Patients respond very well to oral antibiotic therapy.
Protein-losing Enteropathies:
A number of disorders of the gastrointestinal tract are accompanied by excessive protein loss without a concomitant increase in protein synthesis, thus resulting in hypoproteinaemia.
These diseases are listed below:
- Whipple’s disease
- Crohn’s disease
- Ulcerative colitis
- Sprue
- Intestinal lymphangiectasia
- Menetrier’s disease (Hypertrophic gastritis)
Malabsorption Syndrome (MAS):
MAS is characterised by impaired intestinal absorption of fat; some other substances are proteins, carbohydrates, vitamins and minerals.
Clinically, common symptom of MAS is steatorrhoea i.e. pale, bulky, foul-smelling stools.
A mucosal biopsy of the small intestine is essential for making the diagnosis of MAS. It shows the presence of villous atrophy varying from partial to near total.
Sprue of various types causes MAS e.g. celiac (gluten-sensitive), tropical, and collagenous.
Small Intestinal Tumours:
Although the small intestine is about 6 meters long, for obscure reasons, benign as well as malignant tumours in it are surprisingly rare.
The most common benign tumours, in descending order of frequency, are Gastrointestinal stromal tumours (GIST), leiomyomas, adenomas and vascular tumours (haemangioma, lymphangioma).
Amongst the malignant tumours, the most frequently encountered, in descending frequency, are Carcinoid tumours, lymphomas and adenocarcinoma.
All these tumours are identical in morphology to those seen elsewhere in the alimentary tract. Carcinoid tumour, a peculiar neoplasm most common in the midgut, and malignant lymphoma
are described below.
Carcinoid Tumour (Argentaffinoma):
Carcinoid tumour or argentaffinoma is a generic term applied to tumours originating from neuroendocrine cells (synonyms argentaffin cells, Kulchitsky cells, enterochromaffin cells) and therefore also called neuroendocrine tumours.
Neuroendocrine cells are distributed throughout the mucosa of the GI tract.
These cells have secretory granules which stain positively with silver salts (argentaffin granules) or many stains after the addition of exogenous reducing agents (nonargentaffin or argyrophilic granules).
Accordingly, carcinoid tumours may be argentaffin or argyrophilic type. Depending upon the embryologic derivation of the tissues where the tumour is located, these are classified as foregut, midgut, and hindgut carcinoids.
Midgut carcinoids, seen in terminal ileum and appendix are the most common (60-80%) and are more often argentaffin positive.
Hindgut carcinoids, occurring in the rectum and colon are more commonly argyrophilic type and comprise about 10-20% of carcinoids.
Foregut carcinoids, located in the stomach, duodenum, and oesophagus are also argyrophilic type and are encountered as frequently as in the hindgut (10-20%).
Other uncommon locations are the bronchus, trachea, gallbladder, and Meckel’s diverticulum.
Appendix and terminal ileum, the two most common sites for carcinoids, depict variations in their age and sex incidence and biological behaviour
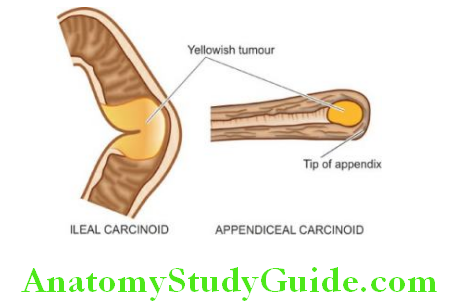
Appendiceal carcinoids, occur more frequently in 3rd and 4th decades of life without any sex predilection, are often solitary and behave as locally malignant tumours.
Ileal carcinoids, on the other hand, are seen more often in later age (7th decade) with female preponderance, are more commonly multiple and behave like metastasising carcinomas.
Morphologic Features: Grossly, all carcinoids are small, button-like submucosal elevations with intact or ulcerated overlying mucosa.
They are usually small; those larger than 2 cm are more often metastasising.
Ileal and gastric carcinoids are commonly multiple, whereas appendiceal carcinoids commonly involve the tip of the organ and are solitary. The cut section of all carcinoids is bright yellow.
Histologically, the features are as under:
Tumour cells may be arranged in a variety of islands solid nests, sheets, cords, trabeculae and clusters.
These cells have characteristic palisading at the periphery of islands. Acinar arrangements and rosettes are rarely seen.
The tumour cells are classically small, and monotonous, having uniform nuclei, salt and pepper chromatin and poorly-defined cell boundaries.
The argentaffin carcinoids show eosinophilic granules in the cytoplasm which stain positively by the argentaffin reaction.
Most carcinoids have infrequent mitotic figures. However, the cytologic features alone may be a poor guide for distinguishing clinically benign from malignant behaviour of the tumour, but all carcinoids infiltrate the bowel wall.
Common immunohistochemical stains for neuroendocrine differentiation of tumour cells are chromogranin A and synaptophysin while Ki-67/MIB1 is used as a proliferation marker.
Grading: Neuroendocrine tumours of the GI tract have been divided into three grades (grade 1 or well-differentiated, grade 2 or moderately differentiated, and grade 3 or poorly differentiated) based on the following characteristics
- Cellular morphology
- Presence or absence of necrosis
- Mitotic rate
- Ki-67 index
Grade 1 tumours have prolonged survival while grade 3 tumour has poor prognosis.
Carcinoid Syndrome: Carcinoid tumours that metastasise, especially to the liver, are sometimes associated with carcinoid syndrome.
The syndrome consists of the following features:
- Intermittent attacks of flushing of the skin of the face
- Episodes of watery diarrhoea
- Abdominal pain
- Attacks of dyspnoea due to bronchospasm
- Right-sided heart failure due to involvement of tricuspid and pulmonary valves and endocardium. A number of secretory products in a functioning carcinoid tumour have been demonstrated:
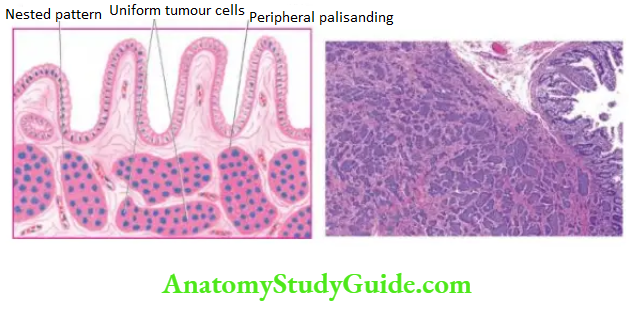
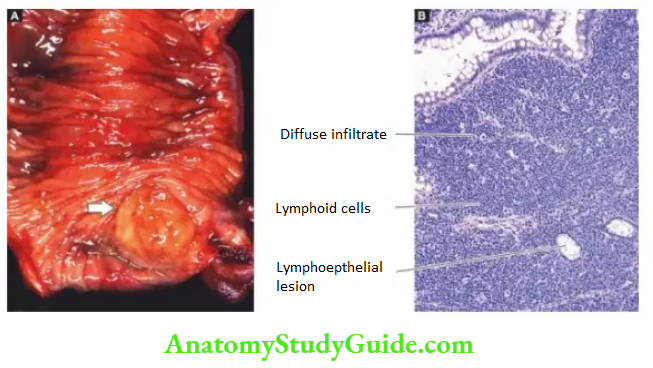
- 5-Hydroxytryptamine (5-HT, serotonin)
- 5-Hydroxytryptophan
- 5-Hydroxy-indole acetic acid (5-HIAA)
- Histamine
- Kallikrein
- Bradykinin
However, 5-HT and its degradation product, 5-HIAA, are particularly significant in the production of carcinoid syndrome.
5-HT, a potent vasodilator and smooth muscle stimulant, is normally synthesised in the endocrine cells of the gut from dietary tryptophan.
Tryptophan is first hydroxylated to 5-hydroxy tryptophan, then decarboxylated to 5-HT and further oxidised to 5-HIAA by the monoamine oxidase in the liver cells. It is then excreted in the urine.
This capacity to synthesise 5-HT and 5-HIAA is markedly elevated in primary and hepatic metastatic carcinoids.
Midgut carcinoids have rich decarboxylating enzymes and are thus able to produce large quantities of 5-HT and 5-HIAA, accounting for a high frequency of carcinoid syndrome in them.
Foregut and hindgut carcinoids, on the other hand, lack decarboxylating enzymes and, therefore, are less often associated with carcinoid syndrome.
Malignant Lymphoma:
Malignant lymphoma of the small intestine has a few peculiarities:
1. Presence and type of an underlying disorder e.g. long-term malabsorption syndrome, AIDS, Crohn’s disease.
2. Primary versus secondary lymphoma Extranodal lymphoma is more common in the gastrointestinal tract than elsewhere, it is important to rule out the spread of primary nodal lymphoma to the intestine.
3. Type of lymphoma and its cell lineage NHL of B-cell type are the main intestinal lymphomas because Hodgkin’s disease in the intestine is extremely rare.
B-cell NHL in the intestine includes immunoproliferative small intestinal disease (IPSID), low-grade B-cell NHL of MALT type, follicular lymphoma, diffuse large B-cell lymphoma (DLBCL), Burkitt’s lymphoma, mantle zone lymphoma.
T-cell NHL is rare and represents a complication of long-standing sprue. The morphology of NHL is identical to its counterpart described in the stomach.
Small Intestinal Tumours:
- Leiomyoma and GIST are more common benign tumours of the small intestine.
- Carcinoid tumour originates from neuroendocrine cells. It may occur in the midgut, hindgut and foregut.
- Grossly, a carcinoid is a small button-like nodule over mucosa.
- Microscopically, the tumour cells in carcinoids are monomorphic and have a typical nested pattern.
- Non-Hodgkin’s lymphoma of B-cell MALT type is another common malignant tumour in the small intestine.
Appendix
Normal Structure:
The appendix is a vestigial organ which serves no useful purpose in human beings but instead becomes the site of trouble at times.
It is like a diverticulum of the caecum, usually lying behind the caecum and varies in length from 4 to 20 cm (average 7 cm).
Histologically, the appendix has four layers in its wall mucosa, submucosa, muscular and serosa.
The mucosa has patchy distribution of crypts and the submucosa has abundant lymphoid tissue. Argentaffin and non-argentaffin endocrine cells are present in the base of mucosal glands just as in the small intestine.
The muscular of the appendix has two layers (inner circular and outer longitudinal) as elsewhere in the alimentary tract.
Two important diseases involving the appendix are appendicitis and appendiceal tumours.
Appendicitis:
Acute inflammation of the appendix, acute appendicitis, is the most common acute abdominal condition confronting the surgeon.
The condition is seen more commonly in older children and young adults and is uncommon at the extremes of age.
The disease is seen more frequently in the West and in affluent societies which may be due to variation in diet a diet with low bulk or cellulose and high protein intake more often causes appendicitis.
Etiopathogenesis: The most common mechanism is obstruction of the lumen from various etiologic factors that lead to increased intraluminal pressure.
This presses upon the blood vessels to produce ischaemic injury which in turn favours bacterial proliferation and hence acute appendicitis.
The common causes of appendicitis are as under:
1. Obstructive:
- Faecolith
- Calculi
- Foreign body
- Tumour
- Worms (especially Enterobius vermicularis)
- Diffuse lymphoid hyperplasia, especially in children.
2. Non-obstructive:
- Haematogenous spread of generalised infection
- Vascular occlusion
- Inappropriate diet lacking roughage.
Morphologic Features: Grossly, the appearance depends upon the stage at which the acutely-inflamed appendix is examined.
In early acute appendicitis, the organ is swollen and the serosa shows hyperaemia. In well-developed acute inflammation called acute suppurative appendicitis, the serosa is coated with fibrinopurulent exudate and engorged vessels on the surface.
In further advanced cases called acute gangrenous appendicitis, there is necrosis and ulcerations of mucosa which extend through the wall so that the appendix becomes soft and friable and the surface is coated with greenish-black gangrenous necrosis.
Microscopically, the most important diagnostic histological criterion is the neutrophilic infiltration of the muscular.
In the early stage, other changes besides acute inflammatory changes, are congestion and oedema of the appendiceal wall.
In later stages, the mucosa is sloughed off, the wall becomes necrotic, the blood vessels may get thrombosed and there may be neutrophilic abscesses in the wall. In either case, an impacted foreign body, faecolith, or concretion may be seen in the lumen.
Thus, there is a good correlation between macroscopic and microscopic findings in acute appendicitis.
Clinical Course:
The patient presents with features of the acute abdomen as under:
- Colicky pain, initially around the umbilicus but later localised to the right iliac fossa
- Nausea and vomiting
- Pyrexia of mild grade
- Abdominal tenderness
- Increased pulse rate
- Neutrophilic leucocytosis with toxic granules in neutrophils is the most significant laboratory finding.
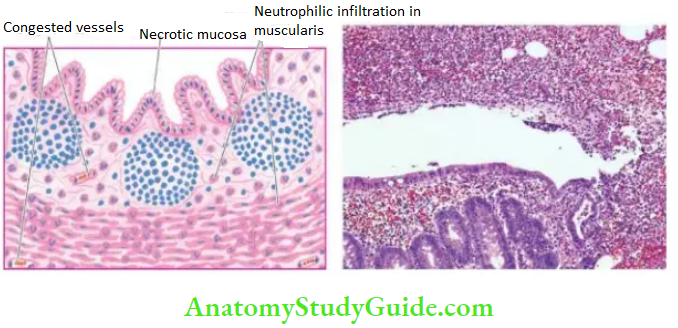
An attack of acute appendicitis predisposes the appendix to repeated attacks (recurrent acute appendicitis) and thus surgery has to be carried out.
If appendicectomy is done at a later stage following the acute attack (interval appendicectomy), pathological changes of healing by fibrosis of the wall and chronic inflammation are observed.
Complications:
If the condition is not adequately managed, the following complications may occur:
1. Peritoniti: A perforated appendix as occurs in gangrenous appendicitis may cause localised or generalised peritonitis.
2. Appendix abscess: This is due to the rupture of an appendix giving rise to a localised abscess in the right iliac fossa.
This abscess may spread to other sites such as between the liver and diaphragm (subphrenic abscess), into the pelvis between the urinary bladder and rectum, and in the females may involve the uterus and fallopian tubes.
3. Adhesions: Late complications of acute appendicitis are fibrous adhesions to the greater omentum, small intestine and other abdominal structures.
4. Portal pylephlebitis: Spread of infection into mesenteric veins may produce septic phlebitis and liver abscess.
5. Mucocele Distension: of the distal appendix by mucus following recovery from an attack of acute appendicitis is referred to as mucocele.
It occurs generally due to proximal obstruction but sometimes may be due to a benign or malignant neoplasm in the appendix.
An infected mucocele may result in the formation of empyema of the appendix.
Tumours Of Appendix:
Tumours of the appendix are quite rare. These include carcinoid tumour (the most common), adenocarcinoma and pseudomyxoma peritonei.
Carcinoid Tumour: It is already Both argentaffin and argyrophilic types are encountered, the former being more common.
Grossly, the carcinoid tumour of the appendix is mostly situated near the tip of the organ and appears as a circumscribed nodule, usually less than 1 cm in diameter, involving the wall but metastases are rare.
Histologically, the carcinoid tumour of the appendix resembles other carcinoids of the midgut.
Mucinous Adenocarcinoma: A well-differentiated mucinous adenocarcinoma called low-grade appendiceal neoplasm is specific to the appendiceal location.
In this tumour, the appendiceal wall is lined by atypical mucinous epithelium, and the lumen contains mucin that may extend into the wall of appendix or beyond.
When this mucinous material spreads to the peritoneal cavity, it produces a clinical condition termed pseudomyxoma peritonei which is accumulation of gelatinous mucinous ascites.
In assessing an ovarian mucinous tumour associated with pseudomyxoma peritonei, the condition of the appendix is always looked for the mucinous tumour of the ovary associated with mucinous ascites is presumed to be appendiceal origin unless proved otherwise.
Diseases of Appendix:
- Acute appendicitis is an acute abdomen associated with colicky pain and neutrophilic leucocytosis.
- Characteristically, there is neutrophilic infiltration in the muscularis propria.
- Complications of acute appendicitis are peritonitis, abscess formation, adhesions, portal pylephlebitis and mucocele.
- Tumours of the appendix include carcinoid and well-differentiated mucinous adenocarcinoma. The latter may produce pseudomyxoma peritonei.
Large Bowel
Normal Structure:
The large bowel consists of 6 parts the caecum, ascending colon, transverse colon, descending colon, sigmoid colon and rectum, and in all measures about 1.5 meters in length.
The serosal surface of the large intestine except the rectum is studded with appendices epiploic ae which are small, rounded collections of fatty tissue covered by the peritoneum.
Histologically, the wall of the large bowel consists of 4 layers as elsewhere in the alimentary tract serosa, muscular, submucosa and mucosa.
The mucosa lacks villi and there is preponderance of goblet cells over columnar epithelial cells. The lymphoid tissue is less abundant than in the small bowel but lymphoid follicles are seen in the caecum and rectum.
The muscular propria of the large intestine is quite peculiar the inner circular muscle layer ensheaths the whole length of the intestine, while the outer longitudinal muscle layer is concentrated into 3 muscle bands called Taenia coli.
The length of outer muscle layer is shorter than the length of the intestine and therefore, it forms the sacculations or haustra of the large intestine.
At the rectosigmoid junction, the three muscle bands fuse to form a complete covering.
The blood supply to the right colon is from the superior mesenteric artery which also supplies blood to the small bowel.
The remaining portion of the large bowel except for the lower part of the rectum receives blood supply from the inferior mesenteric artery. The lower rectum is supplied by haemorrhoidal branches.
The innervation of the large bowel consists of 3 plexuses of ganglion cells Auerbach’s or myenteric plexus lying between the two layers of muscular,
Henle’s plexus lies in the deep submucosa inner to the circular muscle layer, and the Meissner plexus lies in the superficial mucosa just beneath the mucosae.
These are interconnected by non-myelinated nerve fibres.
The anal canal, a 3-4 cm long tubular structure, begins at the lower end of the rectum, though is not a part of the large bowel, but is included here to cover simultaneously lesions pertaining to this region.
It is lined by keratinised or nonkeratinised stratified squamous epithelium. The anal verge is the junction between the anal canal and perineal skin, while the pectinate line is the squamocolumnar junction between the anal canal and the rectum.
Major groups of diseases of the large bowel discussed below are congenital and non-neoplastic miscellaneous conditions, colorectal polyps and tumours.
Congenital And Miscellaneous Non-Neoplastic Conditions:
Hirschsprung’S Disease (Congenital Megacolon):
The term ‘megacolon’ is used for any form of marked dilatation of the entire colon or its segment and may occur as a congenital or acquired disorder.
The congenital form characterised by the congenital absence of ganglion cells in the bowel wall (enteric neurons) is called Hirschsprung’s disease. As a result, the aganglionic segment remains contracted.
Genetically, Hirschsprung’s disease is a heterogenous disorder as under:
- Autosomal dominant inheritance with a mutation in RET proto-oncogene in some cases.
- Autosomal recessive form with a mutation in endothelin-B receptor gene in many other cases.
Clinically, the condition manifests shortly after birth with constipation, gaseous distension and sometimes with acute intestinal obstruction.
Its frequency is 1 in 5,000 live births, has a familial tendency in about 4% of cases and has a predilection for development in Down’s syndrome.
Pathogenesis lies in the failure of neuroblasts to migrate to the rectum which normally occurs at about 12 weeks of gestation.
Morphologic Features: Two types of biopsies may be done on infants suspected of having Hirschsprung’s disease full-thickness rectal biopsy, and suction biopsy that includes mucosa and submucosa.
Grossly, a typical case of Hirschsprung’s disease shows 2 segments a distal narrow segment that is aganglionic and a dilated proximal segment that contains a normal number of ganglion cells.
Microscopically, the distal narrow segment shows a total absence of ganglion cells of all three plexuses (Auerbach’s or myenteric plexus present between the two layers of muscular, deep submucosal or Henle’s plexus, and superficial mucosal or Meissner’s plexus) and prominence of non-myelinated nerve fibres.
Histochemical staining for acetylcholine esterase activity provides confirmation for identifying ganglion cells and nerve trunks.
Depending upon the length of the segment affected by aganglionosis in Hirschsprung’s disease, the following patterns are recognised
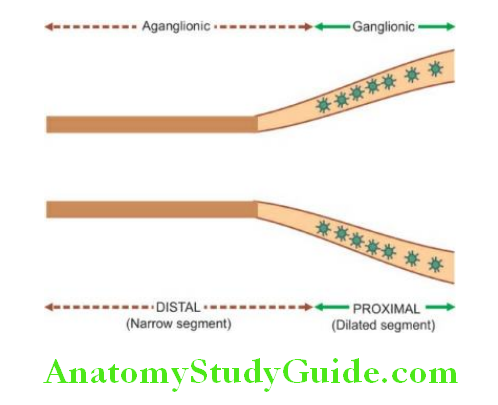
1. Classic form Aganglionosis from distal colorectal region to the proximal dilated colon.
2. Short segments (rectal and rectosigmoid) form Aganglionosis involving a few centimetres of the rectum and rectosigmoid only.
3. Ultra-short form Aganglionosis is in a very small segment which can be missed in a biopsy.
4. Long segment (subtotal coliform Aganglionosis involves most of the colon from the rectosigmoid to the ileocaecal valve, and sometimes may even extend into the small bowel.
5. Zonal colonic aganglionosis A short segment is involved in aganglionosis in which the ganglia cells are absent both above and below the aganglionic segment.
In addition to congenital megacolon discussed above, megacolon may occur from certain acquired causes such as
- Obstructive e.g. due to tumour, post-inflammatory strictures.
- Endocrine e.g. in myxoedema, cretinism.
- CNS disorders e.g. spina bifida, paraplegia, parkinsonism.
- Psychogenic e.g. emotional disturbances, psychiatric disorders.
- Chagas’ disease due to infection with Trypanosoma cruzi is the only example resulting in the acquired loss of ganglion cells. In all other acquired causes listed above, the bowel innervation is normal.
Colitis:
Colitis may occur in isolation but more commonly involvement of the small intestine is also present (enterocolitis).
In view of the considerable overlapping of enteritis and colitis, these lesions have already been described under the small intestine. The table presents a classification of the various types of colitis/enterocolitis.
Diverticulosis Coli:
Diverticula are the outpouchings or herniations of the mucosa and submucosa of the colon through the muscle wall.
Diverticular disease, as it is commonly known, is rare under 30 years of age and is seen more commonly as age advances.
Multiple diverticula of the colon are very common in Western societies, probably due to ingestion of a low-fibre diet but are seen much less frequently in tropical countries and in Japan.
Diverticulosis is often asymptomatic and may be detected as an incidental finding at autopsy.
However, a proportion of patients develop clinical symptoms such as low abdominal pain, distension, constipation and sometimes intermittent bleeding.
Based on the etiologic role of low fibre diet, the pathogenesis of diverticular disease of the colon can be explained as under
1. Ischaemic bowel disease
- Ischaemic colitis (‘membranous’ colitis)
2. Inflammatory bowel disease
- Ulcerative colitis
- Crohn’s disease
3. Other inflammatory lesions
- Infective enterocolitis (dysenteries bacillary, amoebic, other parasitic)
- ‘Pseudomembranous’ enterocolitis (antibiotic-associated diarrhoea)
- Necrotising enterocolitics
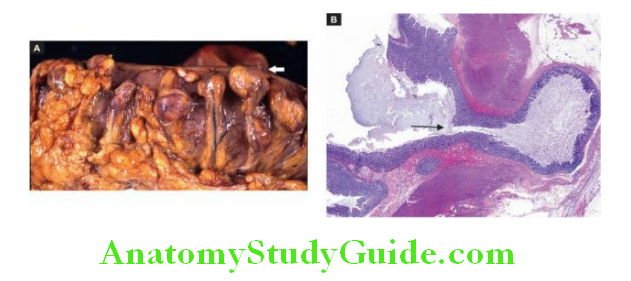
- Increased intraluminal pressure such as due to the low fibre content of the diet causes hyperactive peristalsis and thereby sequestration, of mucosa and submucosa.
- Muscular weakness of the colonic wall at the junction of the muscular with the submucosa.
Morphologic Features:
Grossly, diverticulosis is seen most commonly in the sigmoid colon (95%) but any other part of the entire colon may be involved.
They may vary in number from a few to several hundred.
They appear as small, spherical or flask-shaped outpouchings, usually less than 1 cm in diameter, commonly extend into appendices epiploic ae and may contain inspissated faeces. They are connected to the intestinal lumen by a narrow neck.
Histologically, the flask-shaped structures extend from the intestinal lumen through the muscle layer.
The colonic wall in the affected area is thin and is composed of atrophic mucosa, compressed submucosa and thin or deficient muscular.
However, muscularis propria in between the diverticular protrusions is hypertrophied.
While the diverticular disease may remain asymptomatic, inflammatory changes in the diverticula (diverticulitis) produce clinical symptoms.
The complications of diverticulosis and diverticulitis are perforation, haemorrhage, intestinal obstruction and fistula formation.
Haemorrhoids (Piles):
Haemorrhoids or piles are varicosities of the haemorrhoidal veins.
They are called internal piles if dilatation is of superior haemorrhoidal plexus covered over by mucous membrane, and external piles if they involve inferior haemorrhoidal plexus covered over by the skin.
They are common lesions in the elderly and in pregnant women. They commonly result from increased venous pressure.
Their possible causes include the following:
- Portal hypertension
- Chronic constipation and straining in stool
- Cardiac failure
- Venous stasis of pregnancy
- Hereditary predisposition
- Tumours of the rectum.
Microscopically, thin-walled and dilated tortuous veins are seen under the rectal mucosa (internal piles) or anal skin (external piles).
Secondary changes and complications that may occur include thrombosis, haemorrhage, inflammation, scarring and strangulation (prolapsed piles).
Melanosis Coli:
Melanosis coli is a peculiar condition in which mucosa of the large intestine acquires brown-black colouration.
The condition is said to occur in individuals who are habitual users of cathartics of anthracene type.
Grossly, the mucosal surface is intact and is pigmented brown-black.
Microscopically, a large number of pigment-laden macrophages are seen in the lamina propria. The nature of this pigment is found to be both melanin and lipofuscin.
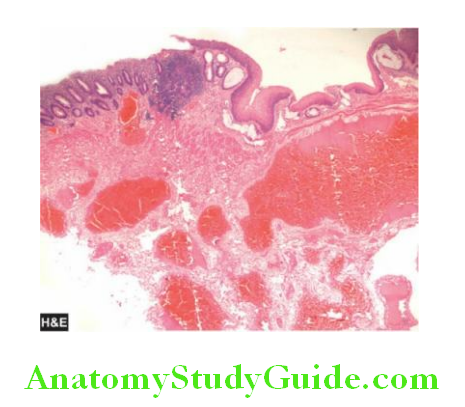
Angiodysplasia:
Angiodysplasia is a submucosal telangiectasia affecting the caecum and right colon that causes recurrent acute and chronic haemorrhage.
The condition is more common in the elderly past 6th decade. The pathogenesis is obscure but is possibly due to mechanical obstruction of the veins.
Miscellaneous Inflammatory Conditions:
Fistula-in-ano It is a well-known and common condition in which one or more fistulous tracts pass from the internal opening at the pectinate line through the internal sphincter onto the
skin surface.
The condition probably results from an infection of the anal glands.
Histologically, nonspecific inflammatory granulation tissue is seen lining the fistulous tract. Foreign body giant cells may be seen.
Anal fissure: It is an ulcer in the anal canal below the level of the pectinate line, mostly in the midline and posteriorly.
The common cause is trauma due to the passage of hard stools, followed by chronic infection.
Solitary rectal ulcer syndrome: It is a condition characterised usually by solitary, at times multiple, rectal ulcers with prolapse of rectal mucosa and development of proctitis.
The histological appearance is quite characteristic. Besides ulceration and inflammation of the rectal mucosa, lamina propria is occupied by spindle-shaped fibroblasts and smooth muscle cells.
The condition is also called a ‘localised form of colitis cystica profound and must be differentiated from a ‘diffuse form of colitis cystica profundal seen in cases of ulcerative colitis.
Submucosal cysts lined by foreign body giant cells and containing gas are also seen in ‘pneumatosis cystoids intestinal.
Congenital and Miscellaneous Nonneoplastic Conditions of Large Bowel:
- Megacolon characterised by the congenital absence of ganglion cells in the bowel wall is called Hirschsprung’s disease. The condition has a distal narrow segment that is aganglionic and a dilated proximal segment that contains a normal number of ganglion cells.
- Diverticulosis coli is the presence of outpouchings of the mucosa and submucosa of the colon through the muscle wall.
- Haemorrhoids or piles are the varicosities of the haemorrhoidal veins.
- Fistula-in-ano and anal fissures are common inflammatory conditions.
Colorectal Polyps And Tumours Of Large Bowel:
The large bowel is the most common site for a variety of benign and malignant tumours, the majority of which are of epithelial origin. Most of the benign tumours present clinically as polyps.
Classification of polyps, benign and malignant tumours of epithelial and nonepithelial origin, is presented.
Since lymphomas, neuroendocrine tumours and mesenchymal tumours are quite uncommon in the large bowel and resemble in morphology their counterparts elsewhere in the GI tract, the following discussion is centred on colorectal polyps and colorectal cancer.
At the end, a brief comment is given on tumours of the anal canal and tabulation of causes of GI bleeding has been done.
Colorectal Polyps:
Polyps are defined as protrusions from the mucous membrane into the lumen.
They are called pedunculated when they are attached to the surface with a stalk or a pedicle, and sessile when they are directly sitting on the mucosal surface.
Although polyps occur in the entire length of the bowel, they are much more common in the large intestine, especially in the rectosigmoid region than in the proximal colon.
Based on their origin, polyps are broadly classified into 2 groups nonneoplastic and neoplastic.
Non-neoplastic polyps have further subtypes depending upon their pathogenesis i.e. hyperplastic, inflammatory, hamartomatous etc.
Neoplastic polyps, on the other hand, are true tumours, which may be benign as well as premalignant. The WHO classification of colorectal polyps and tumours.
1. Colorectal polyps
- Non-neoplastic polyps
-
- Hyperplastic (metaplastic) polyps
- Juvenile (Retention) polyps and polyposis
- Peutz-Jeghers polyps and polyposis
- 2. Neoplastic polyps (Adenomas)
-
- Tubular adenoma
- Villous adenoma
- Tubulovillous adenoma
- Serrated polyps
2. Epithelial malignant tumours
- Carcinoma
- Adenocarcinoma
- Other carcinomas
- (Mucinous adenocarcinoma, signet-ring cell carcinoma, small cell carcinoma, adenosquamous carcinoma, undifferentiated carcinoma)
- Carcinoid tumour
3. Benign non-epithelial tumours
- (GIST, leiomyomas, leiomyoblastoma, neurilemmoma, lipoma and vascular tumours)
4. Malignant non-epithelial tumours
- Leiomyosarcoma, malignant lymphoma, malignant melanoma, angiosarcoma, Kaposi’s sarcoma)
5. Secondary tumours
1. Non-Neoplastic Polyps:
Non-neoplastic polyps are more common and include the following 3 subtypes:
1. Hyperplastic (Metaplastic) Polyps
Hyperplastic or metaplastic polyps are the most common among all epithelial polyps, particularly in the rectosigmoid.
They are called ‘hyperplastic’ because there is epithelial hyperplasia at the base of the crypts, and ‘metaplastic’ because there are areas of cystic metaplasia.
They may be seen at any age but are more common in the elderly (6th-7th decade).
Grossly, hyperplastic polyps are generally multiple, sessile, smooth-surfaced and small (less than 0.5 cm).
Microscopically, they are composed of long and cystically dilated glands and crypts lined by normal epithelial cells.
Their lining is partly flat and partly papillary.
The luminal border of the lining epithelium is often serrated or saw-toothed (see details under serrated polyps and adenomas below).
Hyperplastic polyps are usually symptomless and have no malignant potential.
2. Juvenile (Retention) Polyps and Polyposis:
juvenile or retention polyps are a form of hamartomatous polyps which occur more commonly in children below 5 years of age.
Solitary juvenile polyps occur more often in the rectum, while juvenile polyposis may be present anywhere in the large bowel.
Grossly, juvenile polyps are spherical, smooth-surfaced, about 2 cm in diameter and are often pedunculated.
Microscopically, the classical appearance is of cystically dilated glands containing mucus and lined by normal mucus-secreting epithelium. The stroma may show inflammatory cell infiltration if there is chronic ulceration of the surface.
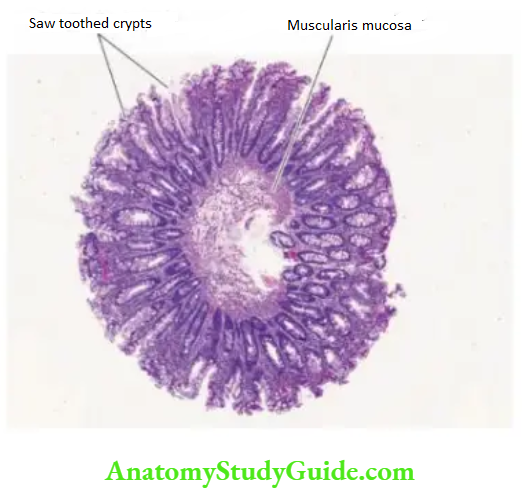
Most cases, on becoming symptomatic in the form of rectal bleeding, are removed. In common with other non-neoplastic polyps, they are also not precancerous.
3. Peutz-Jeghers Polyps and Polyposis:
Peutz-Jeghers syndrome is an autosomal dominant defect, characterised by hamartomatous intestinal polyposis and melanotic pigmentation of lips, mouth and genitalia.
The polyps may be located in the stomach, small intestine or colon but are most common in the jejunum and ileum. The most common age is adolescence and early childhood.
Grossly, these polyps are of variable size but are often large, multiple and pedunculated and more commonly situated in the small intestine.
Microscopically, the most characteristic feature is the tree-like branching of muscularis mucosae.
The lining epithelium is by normal-appearing epithelial cells. The glands may show hyperplasia and cystic change.
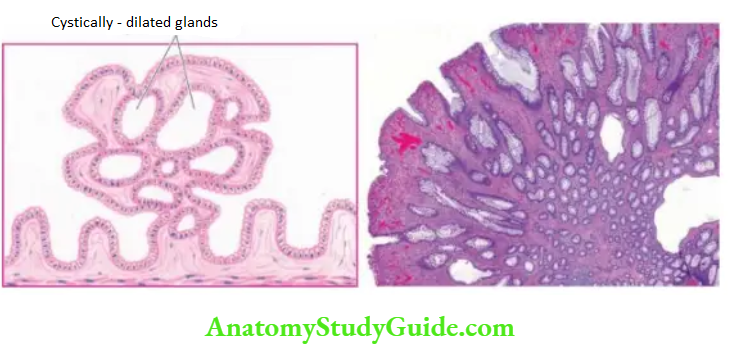
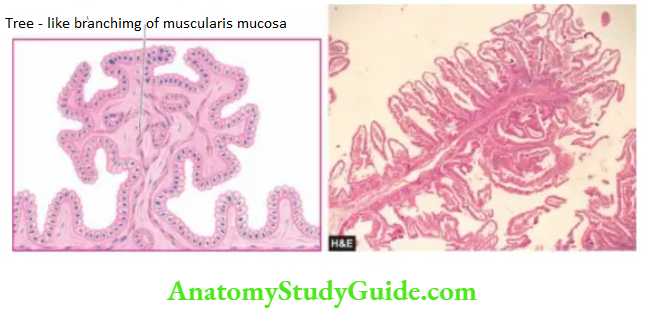
Peutz-Jeghers polyps do not undergo malignant transformation unless a coexistent adenoma is present.
However, patients with Peutz-Jeghers syndrome are more prone to certain other cancers such as of the pancreas, lungs, breast, ovary and uterus.
Neoplastic Polyps (Adenomas):
Neoplastic polyps are colorectal adenomas which have the potential for malignant change.
As per the WHO classification, there are 4 types of adenomas:
Tubular, villous tubulovillous, and serrated.
All these types of colorectal adenomas represent a difference in the growth pattern of the same neoplastic process and have variable biological behaviour but most of them are premalignant.
Adenomas may occur sporadically or may be present as part of familial adenomatous polyposis (FAP) syndrome with autosomal dominant inheritance pattern and is due to APC gene mutation.
This subject is discussed in detail under colorectal cancer.
1. Tubular Adenoma:
Tubular adenomas or adenomatous polyps are the most common neoplastic polyps (75%). They are common beyond 3rd decade of life and have slight male preponderance.
They occur most often in the distal colon and rectum. Tubular adenomas may remain asymptomatic or may manifest by rectal bleeding.
Grossly, adenomatous polyps may be single or multiple, sessile or pedunculated, varying in size from less than 1 cm too large, spherical masses with an irregular surface.
Usually, the larger lesions have recognisable stalks.
Microscopically, the usual appearance is of benign tumour overlying muscular mucosa and is composed of branching tubules which are embedded in the lamina propria.
The lining epithelial cells are of large intestinal type with diminished mucus-secreting capacity, large nuclei and increased mitotic activity.
However, tubular adenomas may show variable degrees of cytologic atypia ranging from atypical epithelium restricted within the glandular basement membrane called ‘carcinoma in situ’ to invasion into the fibrovascular stromal core termed frank adenocarcinoma.
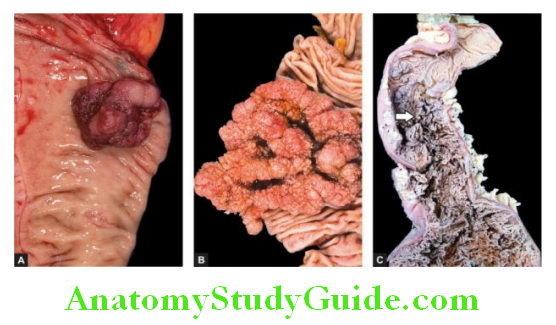
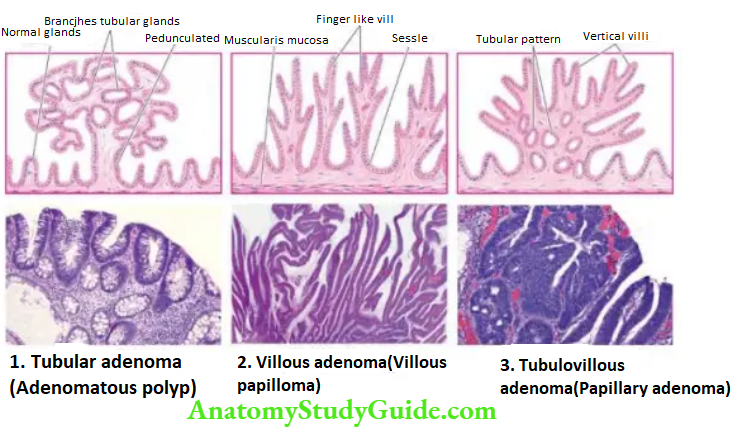
Malignant transformation is present in about 5% of tubular adenomas; the incidence being higher in larger adenomas.
2. Villous Adenoma:
Villous adenomas or villous papillomas of the colon are much less common than tubular adenomas.
The mean age at which they appear is 6th decade of life with approximately equal sex incidence.
They are seen most often in the distal colon and rectum, followed in decreasing frequency, by the rest of the colon.
Grossly, villous adenomas are round to oval exophytic masses, usually sessile, varying in size from 1 to 10 cm or more in diameter. Their surface may be haemorrhagic or ulcerated.
Microscopically, the characteristic histologic feature is the presence of many slender, fingerlike villi, which appear to arise directly from the area of muscularis mucosae.
Each of the papillae has a fibrovascular stromal core that is covered by epithelial cells varying from apparently benign to anaplastic cells. Excess mucus secretion is sometimes seen.
Villous adenomas are invariably symptomatic; with rectal bleeding, diarrhoea and mucus being the common features.
The presence of severe atypia, carcinoma in situ and invasive carcinoma are seen more frequently. Invasive carcinoma has been reported in 30% of villous adenomas.
3. Tubulovillous Adenoma:
Tubulovillous adenoma, also called papillary adenoma or villonodular adenoma, is an intermediate form of pattern between tubular adenoma and villous adenoma.
It is also known by other names like papillary adenoma and villa-glandular adenoma. The distribution of these adenomas is the same as for tubular adenomas.
Grossly, tubulovillous adenomas may be sessile or pedunculated and range in size from 0.5-5 cm.
Microscopically, they show intermediate or mixed patterns, characteristic vertical villi and a deeper part showing tubular patterns. The behaviour of tubulovillous adenomas is intermediate between tubular and villous adenomas.
4. Serrated Polyp and Adenoma:
Serrated lesions of the colorectal region are a new addition to colorectal polyps and adenomas.
A serrated lesion is a histologic diagnosis in which parts of crypts show a serrated or saw-toothed appearance.
Pathogenesis of serrated morphology is explained by decreased apoptosis of proliferated epithelial cells, causing diminished exfoliation that results in the accumulation and folding of epithelial cells in the serrated pattern.
Serrated lesions are quite common and comprise ~40% of all polyps.
Three types of serrated lesions have been described: hyperplastic polyp (65%), sessile serrated adenoma/polyp (35%), and traditional serrated adenoma (5%).
While hyperplastic polyps have already been explained under non-neoplastic polyps above, the morphology and significance of the other two entities have.
Sessile Serrated Adenoma/Polyp: It is a flat sessile lesion seen more commonly in the right colon. A Sessile serrated lesion is often larger than a hyperplastic polyp.
Microscopically, it shows the following features:
- Serration is present along the entire length of crypts.
- The crypt bases are dilated and branched giving an appearance resembling a boot or anchor.
Unlike hyperplastic polyps, sessile serrated adenomas/polyps have an increased propensity to transform into dysplasia and adenocarcinoma.
Traditional Serrated Adenoma: It is a larger, pedunculated and exophytic lesion which is more common in the distal (left) than right (proximal) colon.
Microscopically, the following features are seen:
- The surface of the adenoma is villiform.
- There is prominent crypt serration.
- The cells lining the crypts have pseudostratified nuclei and abundant eosinophilic cytoplasm.
Traditional serrated adenomas are premalignant and have a higher risk for the development of colonic adenocarcinoma.
The contrasting features of non-neoplastic and neoplastic colorectal polyps are given in Table.
Colorectal Cancer:
Colorectal cancer comprises 98% of all malignant tumours of the large intestine. The incidence of carcinoma of the large intestine rises with age; the average age of patients is about 60 years.
Cancer in the rectum is more common in males than females in the ratio of 2:1, while at other locations in the large bowel, the overall incidence is equal for both sexes.
The incidence of large bowel carcinoma shows wide variation throughout the world. It is the commonest form of visceral cancer accounting for deaths from cancer in the United States, next only to lung cancer.
Colorectal cancer is generally thought to be a disease of affluent societies because its incidence is directly correlated with the socioeconomic status of the countries.
Its prevalence is higher in Northern Europe than in South America,
Africa and Asia but in Japan, colon cancer is much less common than in the US although the incidence of rectal cancer is similar.
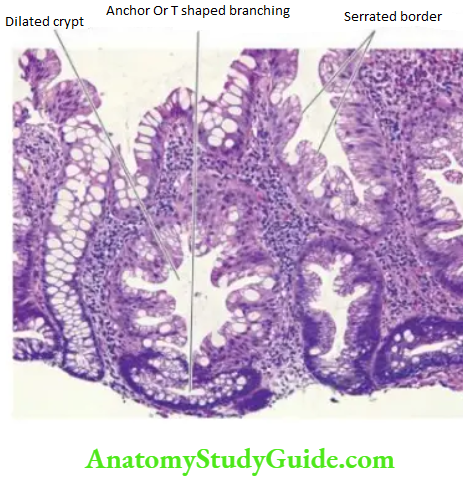
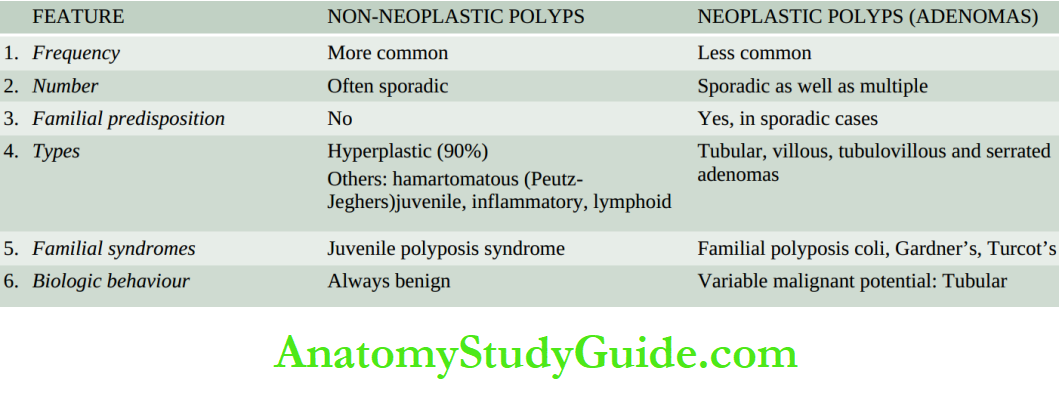
Etiology:
Two major factors diet and genetic susceptibility, play a major role in the etiology of colorectal cancer. Besides, there are a few other high-risk conditions.
1. Diet: Diet plays a significant part in the causation of colorectal cancer:
- A low intake of vegetable fibre-diet leading to low stool bulk is associated with a higher risk of colorectal cancer.
- Consumption of a diet rich in large amounts of animal fat by populations results in excessive cholesterol and its metabolites which may be carcinogenic.
- Excessive consumption of refined carbohydrates that remain in contact with the colonic mucosa for a prolonged duration changes the bacterial flora of the bowel, thus resulting in the production of carcinogenic substances.
2. Genetic Susceptibility:
Genetic factors may be involved in the aetiology of colorectal cancer in two possible ways:
Inherited syndrome of familial adenomatous polyposis (FAP), and hereditary non-polyposis colonic cancer (HNPCC).
Familial adenomatous polyposis (FAP) FAP is an autosomal dominant hereditary disease characterised by the presence of numerous adenomatous colorectal polyps (or adenomas) which have a tendency for progression to adenocarcinoma; the average number is about 1000.
FAP can be distinguished from multiple adenomas in which the number of adenomas is fewer, not exceeding 100.
FAP is due to germline mutations in APC (adenomatous polyposis college located on the long arm of chromosome 5 (5q21-22).
The average age at diagnosis of FAP is the 2nd and 3rd decade of life with equal incidence in both sexes.
Gardner syndrome is a variant of FAP in which there is a combination of FAP and certain extra-colonic lesions such as multiple osteomas (particularly of the mandible and maxilla), epidermoid cysts and desmoid tumours.
The number of polyps in Gardner’s syndrome is generally fewer than in the familial polyposis coli but their clinical behaviour is identical.
Turcot syndrome is another variant of FAP in which there is a combination of FAP with medulloblastoma, a malignant neoplasm of the brain. The malignant potential of familial polyposis coli is very high.
Colorectal cancer develops virtually in 100% of cases by the age of 50 years if not treated with colectomy.
There is strong evidence to suggest that colonic adenocarcinoma evolves from pre-existing adenomas, referred to as adenoma-carcinoma sequence.
The following evidence are cited to support this hypothesis:
In the case of early invasive cancer, the surrounding tissue often shows preceding changes of evolution from adenoma. hyperplasia? dysplasia? carcinoma in situ? invasive carcinoma.
The incidence of adenomas in a population is directly proportionate to the prevalence of colorectal cancer.
The risk of adenocarcinoma colon declines with endoscopic removal of all identified adenomas.
The peak incidence of adenomas generally precedes by some years to a few decades the peak incidence of colorectal cancer.
The risk of malignancy increases with the following adenoma-related factors:
A number of adenomas familial polyposis coli syndrome almost certainly evolves into malignancy.
Size of adenomas large size increases the risk.
Type of adenomas greater villous component associated with higher prevalence.
The molecular mechanism involved in it is described later.
Hereditary non-polyposis colonic cancer (HNPCC or Lynch syndrome) HNPCC is an autosomal dominant condition associated with multiple primary cancers at different sites (endometrium, ovary) including colorectal cancer without evidence of familial polyposis coli.
In HNPCC, colorectal cancer is seen in at least two generations of first-degree relatives, occurs at a relatively early age (= 50 years), is located more often in the proximal colon, and has a better prognosis than sporadic colon cancer cases.
It is due to germline mutations in mismatch repair genes, human mutL homolog abbreviated as hMLH2 located on chromosome 2 and hMLH1 on chromosome 3 resulting in DNA instability.
3. High-Risk Conditions: The presence of certain pre-existing diseases and some other factors increase the risk of developing colorectal cancer subsequently, e.g.:
Inflammatory bowel disease (especially ulcerative colitis).
Diverticular disease for a long duration. A low-fibre diet is implicated in the pathogenesis of diverticular disease as well.
Role of tobacco smoking in the development of colorectal cancer in younger patients.
Infection with Streptococcus bovis (a faecal organism) in the development of occult colorectal cancer.
Molecular Mechanism: Adenoma-Carcinoma Sequence Studies by molecular genetics have revealed that there are sequential multistep mutations in the evolution of colorectal cancer from adenomas by one of the following two mechanisms
1. Mutational pathway: This pathway of multiple mutations is generally associated with morphologically identifiable changes of transformation from adenoma to adenocarcinoma.
These changes are as under:
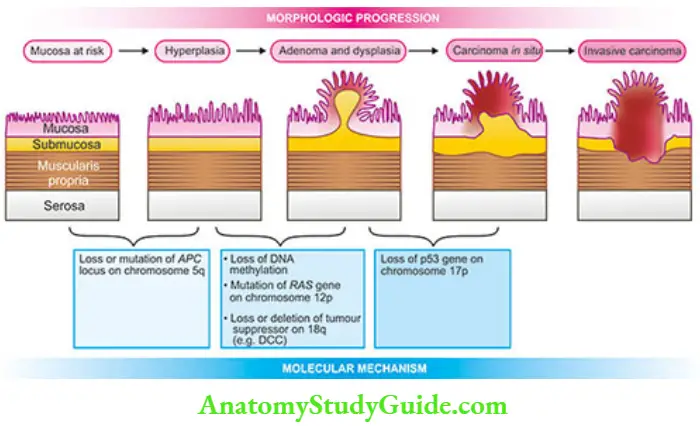
Loss of tumour suppressor APC (adenomatous polyposis college located on the long arm of chromosome 5 (5q) is observed in 80% of cases of sporadic colon cancer.
Since the function of the APC gene is linked to ß-catenin, the loss of the APC gene results in the translocation of ß-catenin to the nucleus where it activates transcription of other genes, mainly MYC and cyclin D1, both of which stimulate cell proliferation.
This is termed as APC mutation/ß-catenin mechanism.
A point mutation in the K-RAS gene follows the loss of the APC gene and is seen in 10 to 50% of cases of adenoma-carcinoma.
Deletion of DCC gene located on the long arm of chromosome 18 i.e. 18q (DCC for deleted in colorectal cancer) in 60-70% of cases of colon cancer.
Loss of the p53 tumour suppressor gene is seen in 70-80% of cases of colon cancer.
2. Microsatellite instability (MSI) mechanism: In this pathway also, there are multiple mutations but of different genes, and unlike APC mutation/ß-catenin mechanism, there are no morphologically identifiable changes.
This pathway accounts for 10-15% of cases of colon cancer. The basic mutation is the loss of the DNA repair gene.
This results in a situation in which repetitive DNA sequences (i.e. microsatellites) become unstable during the replication cycle, termed microsatellite instability, which is the hallmark of this pathway.
The significant DNA repair genes which are mutated in colon cancer are as under:
- TGF-ß receptor gene which normally inhibits cell proliferation but in mutated form allows the uncontrolled proliferation of colonic epithelium in adenoma.
- BAX gene which normally causes apoptosis but a defect in it results in loss of apoptosis and dysregulated growth.
While in 80-90% of cases of conventional adenomas of all types, the classic molecular mutational pathway involving APC, KRAS, and p53 discussed above is followed, 10-20% of cases of serrated adenomas follow the serrated mutational pathway involving BRAF/KRAS/MSI mechanisms.
Morphologic Features:
Distribution of primary colorectal cancer reveals that about 60% of the cases occur in the rectum, followed in descending order, by sigmoid and descending colon (25%), caecum and ileocaecal valve (10%); ascending colon, hepatic and splenic flexures (5%); and quite uncommonly in the transverse colon.
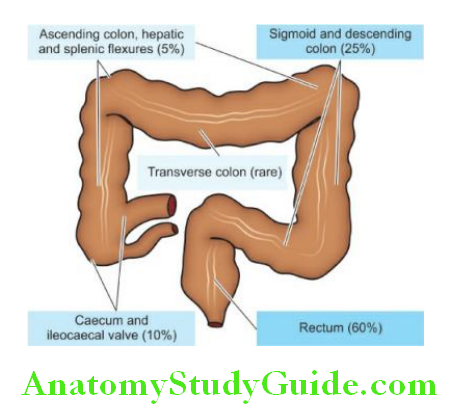
Grossly, there are distinct differences between the growth on the right and left half of the colon.
Right-sided colonic growths tend to be large, cauliflower-like, soft and friable masses projecting into the lumen (fungating polypoid carcinoma).
Left-sided colonic growths, on the other hand, have napkin-ring-like annular constriction i.e. they encircle the bowel wall circumferentially with increased fibrous tissue forming an annular ring and have central ulceration on the surface with slightly elevated margins (carcinomatous ulcers).
These differences in right and left colonic growths are probably due to the liquid nature of the contents in the ascending colon leaving space for luminal growth on the right side, while the contents in the left colon are more solid permitting the spread of growth into the bowel wall.
Contrasting features of right-sided and left-sided colon cancer.
However, early lesions in the left, as well as right colon, are small, button-like areas of elevation.
Microscopically, the appearance of right and left-sided growths is similar.
About 95% of colorectal carcinomas are adenocarcinomas of varying grades of differentiation, out of which approximately 10% are mucin-secreting colloid carcinomas.
The remaining 5% of tumours include uncommon microscopic patterns like small cell carcinoma, signet-ring cell carcinoma, undifferentiated carcinoma, and adenosquamous carcinomas; the latter is seen in the more distal colon near the anus.
Histologic grades indicating the degree of differentiation may vary:
Well-differentiated (15-20% cases), moderately-differentiated (60-70% cases) and poorly-differentiated (15-20% cases).
Spread:
Carcinoma of the large intestine may spread by the following routes:
1. Direct spread: The tumour spreads most commonly by direct extension in both ways circumferentially into the bowel wall as well as directly into the depth of the bowel wall to the serosa, pericolic fat, and sometimes into the peritoneal cavity.
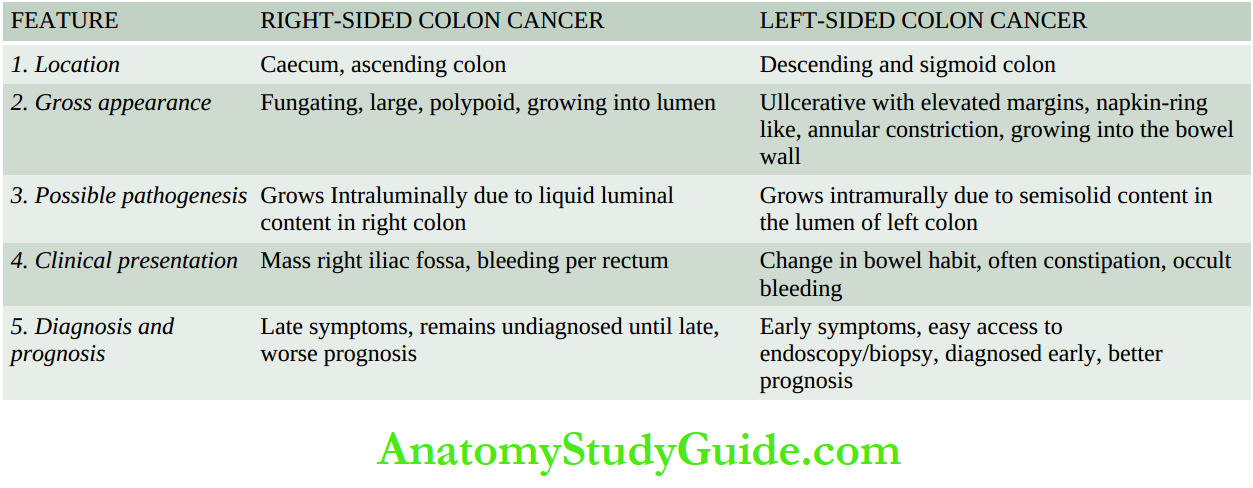
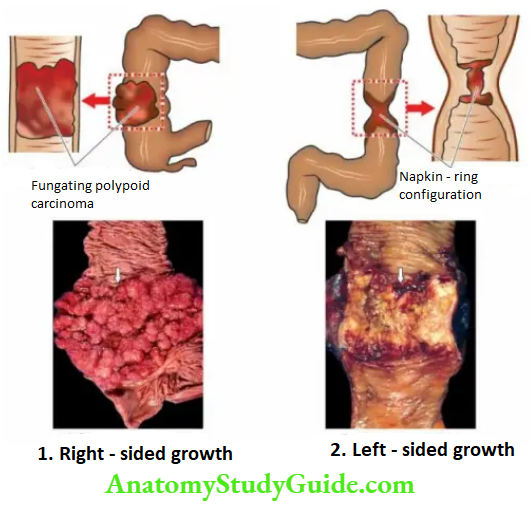
2. Lymphatic spread: Spread via lymphatics occurs rather commonly and involves, firstly the regional lymph nodes in the vicinity of the tumour, and then into other groups of lymph nodes like preaortic, internal iliac and sacral lymph nodes.
3. Haematogenous spread: Blood spread of large bowel cancer occurs relatively late and involves the liver, lungs, brain, bones and ovary.
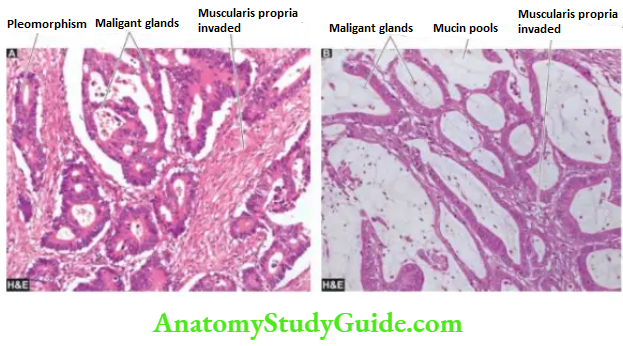
Clinical Features:
Clinical symptoms in colorectal cancer appear after a considerable time.
These are as follows:
- Occult bleeding (melaena)
- Change in bowel habits, more often in left-sided growth
- Loss of weight (cachexia)
- Loss of appetite (anorexia)
- Anaemia, weakness, malaise.
The most common complications are obstruction and haemorrhage; less often perforation and secondary infection may occur.
Aside from diagnostic methods like stool tests for occult blood, PR examination, proctoscopy, radiographic contrast studies and CT scans, recently the role of tumour markers has been emphasised.
Of particular importance is the estimation of carcinoembryonic antigen (a level which is elevated in 100% of cases of metastatic colorectal cancers, while it is positive in 20-40% of early lesions and 60-70% of advanced primary lesions.
However, the test may have prognostic significance only and is not diagnostic of colorectal cancer because it is positive in other cancers too such as of the lungs, breast, ovary, urinary bladder and prostate.
CEA levels are elevated in some non-neoplastic conditions also like ulcerative colitis, pancreatitis and alcoholic cirrhosis. Staging and Prognosis
The prognosis of colorectal cancer depends upon a few variables:
- Extent of the bowel involvement
- Presence or absence of metastases
- Histologic grade of the tumour
- Location of the tumour
The most important prognostic factor in colorectal cancer is, however, the stage of the disease at the time of diagnosis.
Three staging systems are in use:
- Dukes’ ABC staging (modified Duke’s includes stage D as well).
- Astler-Coller staging which is a further modification of Duke’s staging and is most widely used.
- TNM staging described by American Joint Committee is also used.
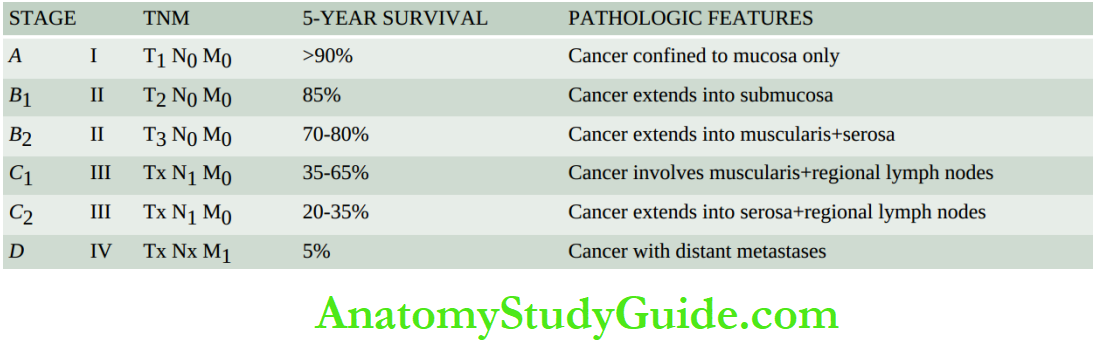
Tumours Of The Anal Canal:
Epithelial tumours of the anal canal are uncommon and may be a combination of several histological types.
Amongst the benign tumours of the anal canal, multiple viral warts called condyloma acuminata are the only tumours of note.
Malignant tumours of the anal canal include the following:
- Squamous cell carcinoma
- Basaloid carcinoma
- Mucoepidermoid carcinoma
- Adenocarcinoma (rectal, of anal glands, within anorectal fistulas)
- Undifferentiated carcinoma
- Malignant melanoma.
These tumours resemble in morphology with similar lesions elsewhere in the body.
Before closing the discussion on the GI tract, since GI bleeding from the upper (haematemesis), middle (small intestinal) and lower (melaenpart is a major presenting clinical feature of a variety of gastrointestinal diseases, a summary of its major causes.
Polyps and Tumours of Large Bowel:
Colorectal polyps may be non-neoplastic or neoplastic.
Non-neoplastic polyps are more common and include hyperplastic, juvenile (retention) and Peutz-Jeghers.
Neoplastic polyps or adenomas are of 4 types: tubular, villous, tubulovillous and serrated adenomas/polyps.
These adenomas have varying morphologic appearances and variable outcomes. The majority of adenomas are premalignant.
Colorectal polyps may occur as a part of syndromes e.g. familial polyposis, Gardener’s, Turcot’s etc.
Colorectal carcinoma is common and has an etiologic relationship to genetic predisposition and dietary factors.
Genetic susceptibility is linked to familial adenomatous polyposis (FAP) and hereditary nonpolyposis colonic cancer (HNPCor Lynch syndrome.
At the molecular level, colorectal carcinoma is explained by mutational pathway (APC mutation/ß-catenin mechanism, KRAS Mutation, loss of p53 tumour-suppressor gene) and microsatellite instability (MSmechanism.
Left-sided and right-sided colorectal cancers have distinct gross appearances but microscopy shows similar morphology of adenocarcinoma; ~10% are mucin-secreting colloid carcinoma. Histologic grade of moderately differentiated cancer is more common.
Colorectal cancer may spread by lymphovascular route as well as extend directly into adjacent tissues.
Three staging systems have been described for colorectal cancer: Dukes’ ABC, AstlerColler, and AJC-TNM.
The most common cancer of the anal canal is squamous cell carcinoma.
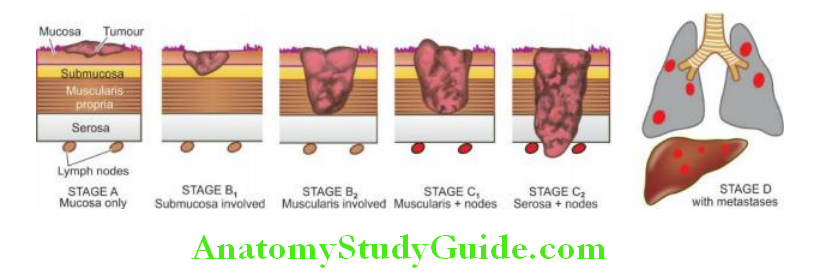
Peritoneum
Normal Structure:
The peritoneal cavity is lined by a layer of surface mesothelium derived from mesoderm. The lining rests on vascularised subserosal fibrous tissue.
Other structures topographically related to the peritoneum are retroperitoneum, omentum, mesentery and umbilicus.
These structures are secondarily involved in a variety of pathologic states which have been discussed along with the discussion of primary diseases.
Here, two conditions, peritonitis and primary tumours and tumour-like lesions, are briefly given.

Peritonitis:
Inflammatory involvement of the peritoneum may result from chemical agents or bacteria.
1. Chemical peritonitis can be caused by the following:
- Bile extravasated due to trauma or diseases of the gallbladder.
- Pancreatic secretions are released from the pancreas in acute haemorrhagic pancreatitis.
- Gastric juice leaked from the perforation of the stomach.
- Barium sulfate from perforation of the bowel during radiographic studies.
- Chemical peritonitis is localised or generalised sterile inflammation of the peritoneum.
2. Bacterial peritonitis: may be primary or secondary; the latter being more common. The primary form is caused by streptococcal infection, especially in children.
Secondary bacterial peritonitis may occur from the following disorders:
- Appendicitis
- Cholecystitis
- Salpingitis
- Rupture of peptic ulcer
- Gangrene of bowel
- Tuberculosis (specific inflammation).
Morphologic Features: Depending upon duration, the features in bacterial peritonitisvary. It may be generalised or may get localised by omentum such as in appendiceal abscess following acute appendicitis.
Depending upon the duration, the fluid accumulation varies from serious, turbid, and creamy to frankly suppurative.
The fluid may eventually resolve or may heal by organisation with the formation of fibrous adhesions.
Tumour-Like Lesions And Tumours:
Tumour-like conditions include idiopathic retroperitoneal fibrosis and mesenteric cysts while the peritoneum is the site for rare primary and many metastatic tumours.
Idiopathic Retroperitoneal Fibrosis:
Also known as Ormond’s disease or sclerosing retro peritonitis, this rare entity of unknown aetiology is characterised by diffuse fibrous overgrowth and chronic inflammation.
The condition is, therefore, more like inflammatory rather than neoplastic in origin.
It may be associated with a similar process in the mediastinum, sclerosing cholangitis and Riedel’s thyroiditis and is termed multifocal fibrosclerosis.
Though idiopathic, the etiologic role of ergot derivative drugs and autoimmune reactions has been suggested.
Mesenteric Cysts:
Mesenteric cysts of unknown aetiology and varying sizes may be found in the peritoneal cavity.
On the basis of their possible origin, they are of various types:
- A chylous cyst is a thin-walled cyst arising from lymph vessels and lined by endothelium.
- Pseudocysts are those which are formed following walled-off infection or pancreatitis.
- Neoplastic cysts occur due to cystic changes in tumours.
Tumours:
The peritoneum may be involved in malignant tumours primary and metastatic.
Mesothelioma is an example of a primary peritoneal tumour and is similar in morphology as in the pleural cavity.
Intra-abdominal desmoplastic small cell tumour is a recently described highly malignant tumour belonging to the group of other round cell or blue cell tumours such as small cell carcinoma lung, Ewing’s sarcoma, rhabdomyosarcoma, neuroblastoma and others.
Metastatic peritoneal tumours are quite common and may occur from dissemination from any intra-abdominal malignancy
Diseases of the Peritoneum:
- Peritonitis may be due to bacterial infection or chemical substances (e.g. bile, pancreatic secretion, gastric juice etc).
- Idiopathic retroperitoneal fibrosis has an unknown origin and is often multifocal.
- Mesenteric cysts of unknown aetiology may be seen in the peritoneal cavity.
- Metastatic tumours in the peritoneum are more common; rarely mesothelioma may occur in the peritoneum as a primary tumour
Leave a Reply