Pharmaceutical Organic Chemistry 2 Short Question And Answers 2021
Question 1. Explain the reactivity and orientation of aniline towards electrophilic aromatic substitution reaction.
Answer:
Table of Contents
An electrophile refers to an electron-seeking species. Thus, an electrophilic substitution reaction refers to the reaction in which an electrophile substitutes another electrophile in an organic compound.
Anilines undergo the usual electrophilic reactions such as halogenation, nitration, and sulphonation.
- The functional group (-NH2. associated with aniline is an electron donating group and hence is very activating towards the electrophilic substitution reaction.
- Due to its various resonating structures, there’s an excess of electron or negative charge over the ortho- and para positions ofthe benzene ring than the meta-position. Thus, anilines are o- and p- directive towards electrophilic substitution reaction.
Halogenation:
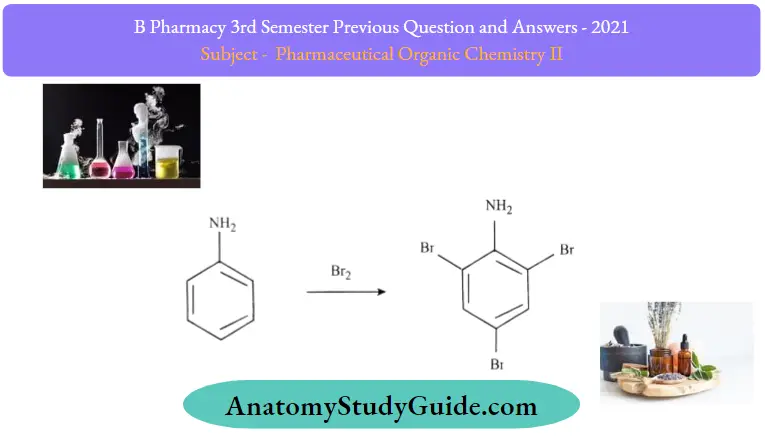
In the above reaction when aniline comes in the vicinity of bromine water, then the bromine molecule develops a polarity within itself bromine with a slightly positive charge that acts as an electrophile (electron seeking. and attacks the electron-rich ortho and para positions of aniline. A white-colored precipitate of 2,4,6-tribromoaniline is obtained at room temperature.
Nitration:
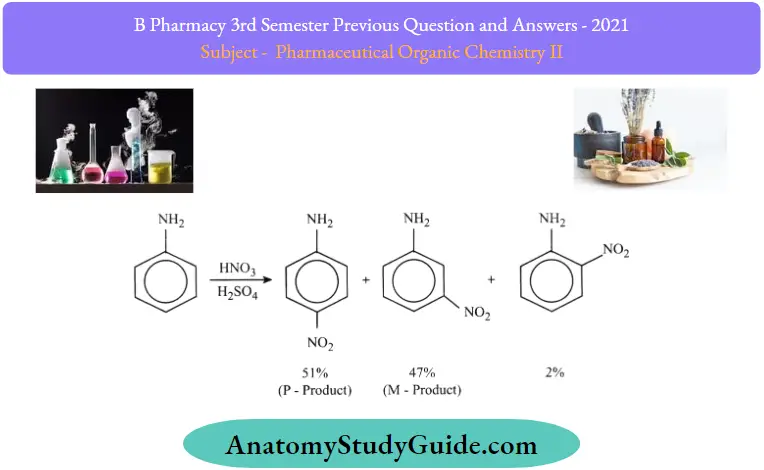
In this reaction, alongside the para isomer, the meta isomer is also observed. It is because the aniline molecule gets protonated in an acidic medium to become an anilinium ion which is meta-directing.
Sulphonation:
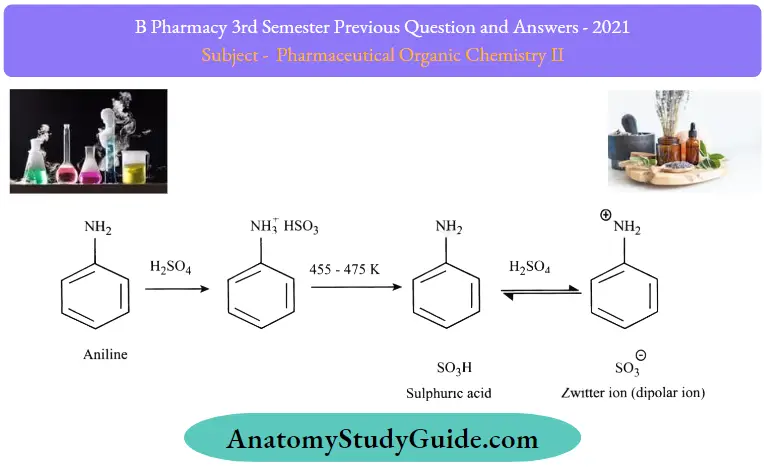
Sulphuric acid reacts vigorously with aniline to form anilinium hydrogen sulfate which on heating produces sulphanilic acid which in turn also has a resonating structure with zwitter ion as shown in the above figure.
- Zwitter ion refers to a dipolar ion in which both positive and negative charges exist and the molecule as a whole is neutral. It is also called inner salt sometimes.
- It differs from amphoteric ions in the sense that it has negative and positive charge simultaneously while amphoteric ions are either cationic or anionic at a time.
- Anilines don’t undergo Freidel crafts reaction because they react with ferric chloride of the reaction mixture which acts as catalyst for the reaction.
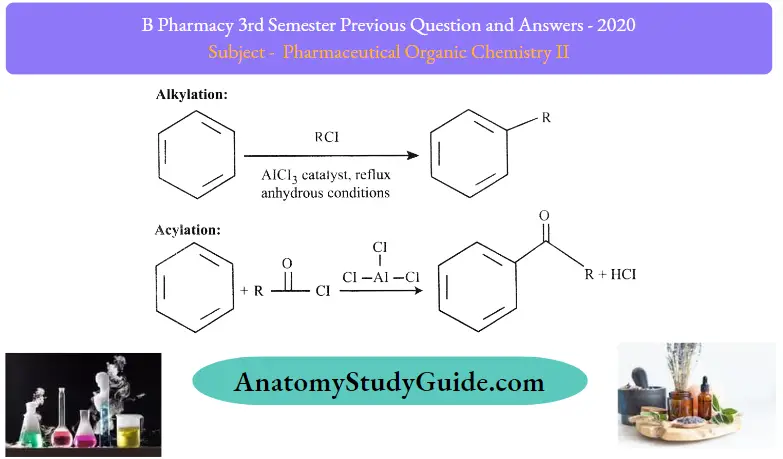
Question 2. Explain the Friedelcraft alkylation of benzene with limitations.
Or
Discuss The Mechanism of Friedelcraft alkylation
Answer:
A Friedel-Crafts reaction is an organic coupling reaction involving an electrophilic aromatic substitution that is used for the attachment of substituents to aromatic rings
Friedel-Crafts Alkylation
Friedel-Crafts Alkylation refers to the replacement of an aromatic proton with an alkyl group.
- This is done through an electrophilic attack on the aromatic ring with the help of a carbocation.
- The Friedel-Crafts alkylation reaction is a method of generating alkylbenzenes by using alkyl halides as reactants.
- The Friedel-Crafts alkylation reaction of benzene is illustrated below.
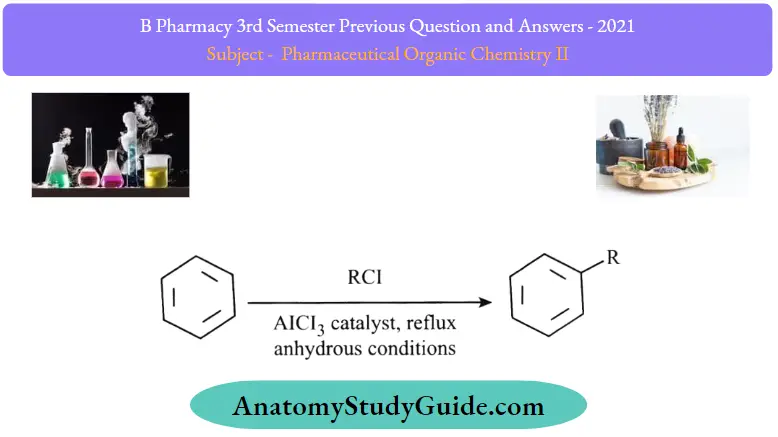
A Lewis acid catalyst such as FeCl3 or AlCl3 is employed in this reaction to form a carbocation by facilitating the removal of the halide. The resulting carbocation undergoes a rearrangement before proceeding with the alkylation reaction.
Friedel-Crafts alkylation Mechanism:
The Friedel-Crafts alkylation reaction proceeds via a three-step mechanism.
- Step 1: The Lewis acid catalyst (AlCl3). undergoes a reaction with the alkyl halide, resulting in the formation of an electrophilic carbocation.
- Step 2: The carbocation proceeds to attack the aromatic ring, forming a cyclohexadienyl cation as an intermediate. The aromaticity of the arena is temporarily lost due to the breakage of the carbon-carbon double bond.
- Step 3: The deprotonation of the intermediate leads to the reformation ofthe carbon-carbon double bond, restoring aromaticity to the compound. This proton goes on to form hydrochloric acid, regenerating the AlCl3 catalyst.
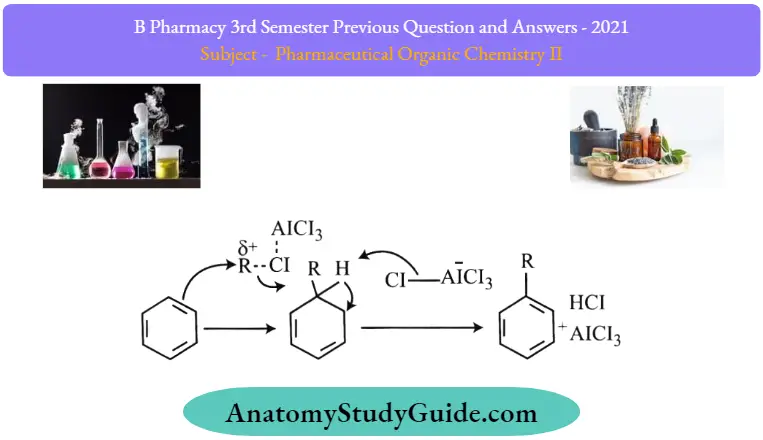
An illustration describing the mechanism ofthe FriedelCrafts alkylation reaction is provided above.
Friedel-Crafts alkylation Limitations:
Some important limitations of Friedel-Crafts alkylation are listed below.
- Since the carbocations formed by aryl and vinyl halides are extremely unstable, they cannot be used in this reaction.
- The presence of a deactivating group on the aromatic ring (such as an NH2 group. can lead to the deactivation of the catalyst due to the formation of complexes.
- An excess of the aromatic compound must be used in these reactions to avoid polyalkylation (the addition of more than one alkyl group to the aromatic compound.
- Aromatic compounds that are less reactive than monohalobenzenes do not participate in the Friedel-Crafts alkylation reaction.
It is important to note that this reaction is prone to carbocation rearrangements, as is the case with any reaction involving carbocations.
Question 3. Explain the effect of substituents on the basicity of aromatic amines.
Answer:
Because the nitrogen atom has a single electron, aromatic amines are basic. In comparison to aliphatic amines, they tend to be more acidic.
- Aromatic amines gain their basic character from electron-releasing groups.
- Forelectron releasing groups (para-substituted anilines. are more basic than orthosubstituted anilines. The para effect is responsible for this.
- Electron-withdrawing groups transform aromatic amines, causing them to lose their basic characteristics
- N,N-diemthyl aniline > N-methyl aniline > aniline
- In general, the more electrons present in a molecule, the more basic it is.
- Alkyl groups that donate electrons increase the basicity of amine molecules while nitrogen-containing groups such as -NO2 decrease its basicity.
- Because of the electron-donating property of alkyl groups attached to nitrogen atoms, the electron density around nitrogen atoms increases along with its ability to release electrons.
- Alkyl amines are more basic than ammonia because of the greater and easier release of electrons.
Question 4. Explain any one method to determine saponification value with its significance.
Answer:
Saponification value
Saponification Principle:
The saponification value is defined as the number of milligrams of KOH required to completely hydrolyze one gram of fat/ oil. In practice a known amount of the oil or fat is refluxed with an excess amount of standard alcoholic potash alum solution and the unused alkali is titrated against a standard acid.
Saponification Reaction:
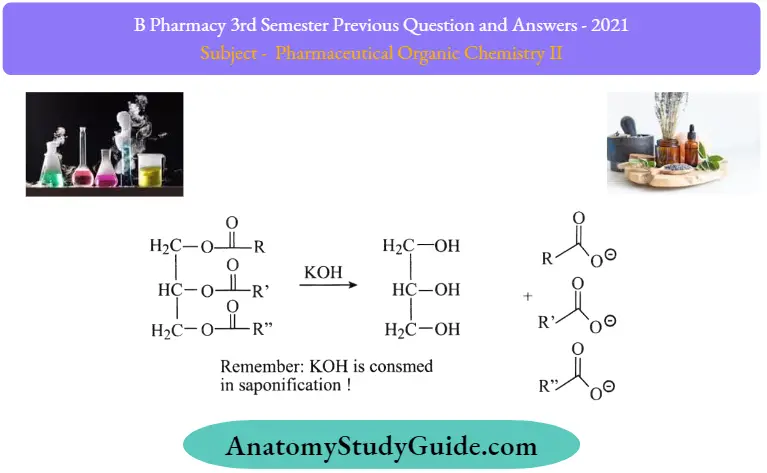
Saponification Procedure:
- About 2gms ofthe given oil/ fat is taken in a conical flask and weighed accurately.
- The oil/fat is dissolved in 25ml of N alcoholic potassium-hydroxide solution.
- Then the reaction mixture is refluxed using a water condenser in a water bath for half an hour.
- The resulting solution is cooled and titrated against NHCl solution adding 1ml of phenolphthalein.
- The number of ml of acid required is noted.
- An equal identical blank experiment is performed.
- The number of ml of hydrochloric acid is noted.
Saponification Calculation:
Saponification value = Volume of acid required to neutralize remaining KOH × Equivalent factor × 1000 / w = (b – a) × 0.02805 × 1000/w
Saponification Significance:
- It gives information concerning the character of the fatty acids of the fat- the longer the carbon chain.
- The less acid is liberated per gram of fat hydrolyzed.
Question 5. Outline any two syntheses and reactions of phenanthrene.
Answer:
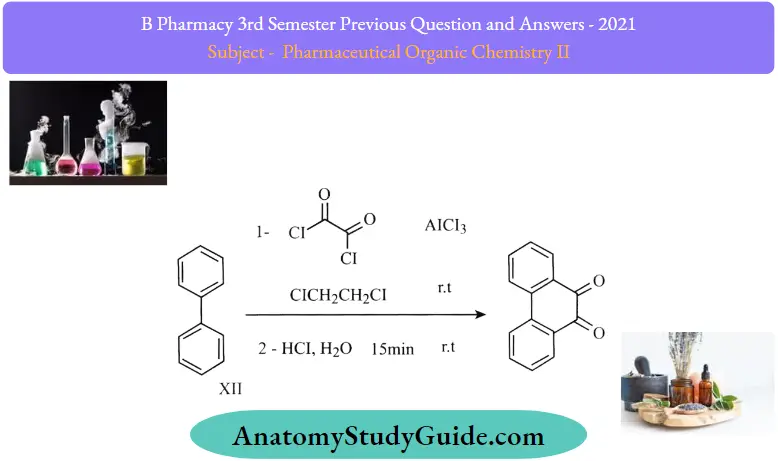
1. Howrth Method:
Question 6. Write any two syntheses and reactions of anthracene
Answer:
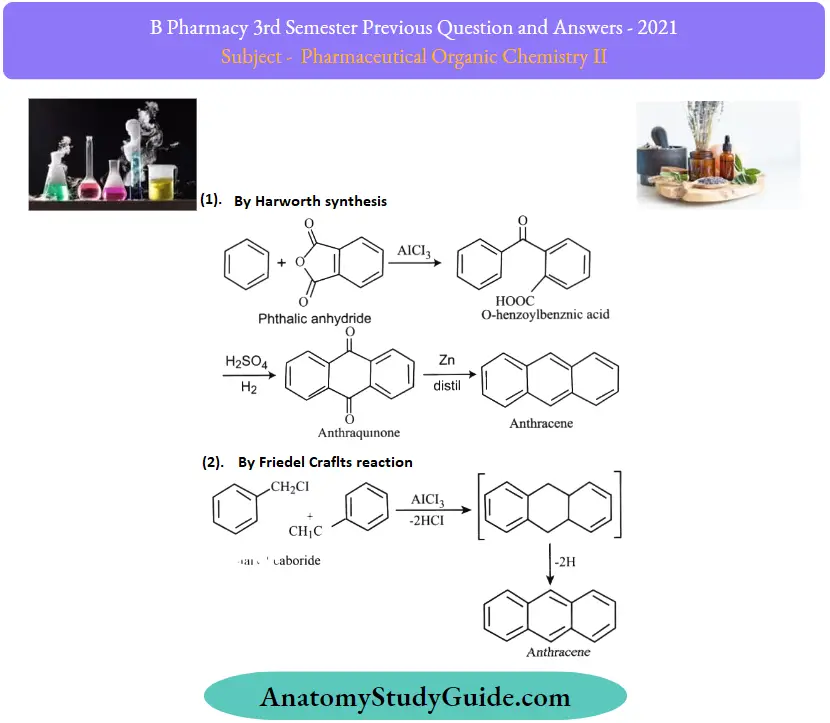
Question 7. Define angle strain. Explain why higher cycloalkanes are more stable than lower members.
Answer:
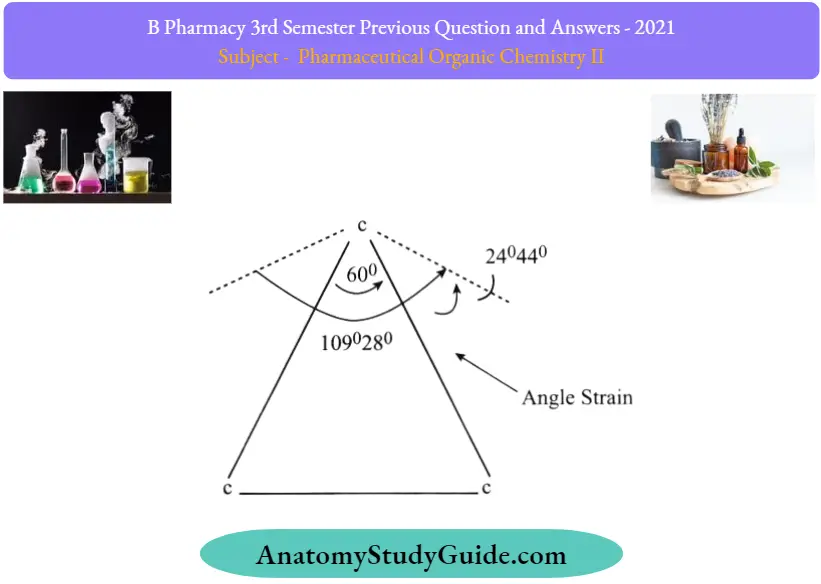
Angle strain:
Angle strain is the increase in the potential energy of a molecule due to bond angles deviating from the ideal values.
For example: Cyclopropane
Cycloalkanes are more stable than lower members:
The short-line structural formulas of cycloalkanes simply look like shapes such as a triangles, squares, etc. The internal angles ofthe shapes can be calculated with geometry, as shown below.
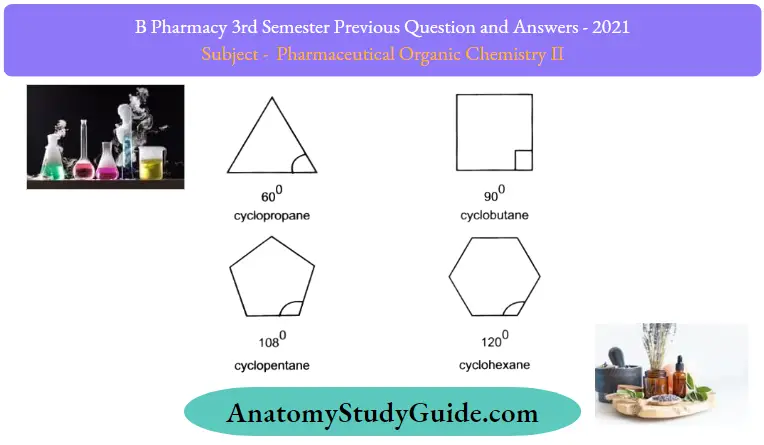
An interesting fact about the cycloalkanes is that they have different relative stabilities, and the stability depends on the size of the ring. It has been observed that cyclic compounds found in nature usually are in 5- or 6- 6-membered rings, and the 3- or 4-membered rings are rather rare.
To explain this stability difference, German chemist Adolf von Baeyer proposed the “Bayer Strain Theory”. Assuming all the rings are in a flat (or planar. shape, Bayer Theory suggests that the difference between the ideal bond angle (which is 109.5° for sp3 carbon. and the angle in the planer cycloalkane causes the strain, which is called angle strain.
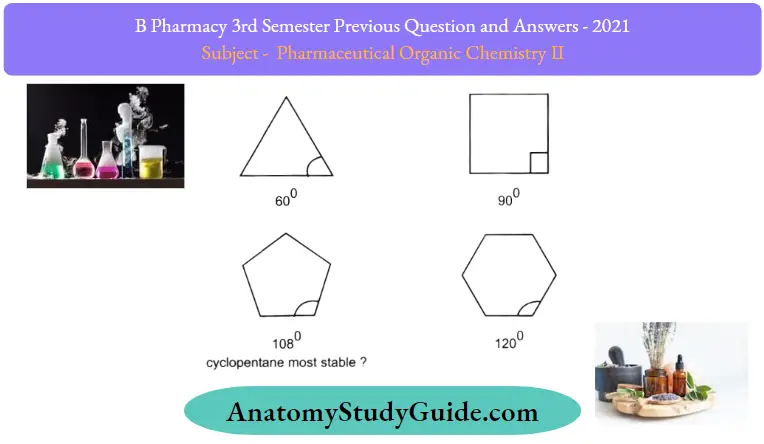
Question 8. Which cycloalkane is stable?
Answer:
However, experimental results show a different trend. It turns out that cyclohexane is the most stable ring that is strain-free and is as stable as a chain alkane.
Furthermore, cyclic compounds do not become less and less stable as the number of rings increases.
- To measure the relative stability of cycloalkanes, the heat of combustion (AHcomb). for each cycloalkane was measured.
- The heat of combustion is the amount of heat released when the compounds bums completely with oxygen.
- The cycloalkanes will be in higher energy levels than corresponding chain alkanes because of strain energy.
- Therefore, when cycloalkane bums, more heat will be released, so the difference of (AHcomb)between cycloalkane and the “strainless” chain alkane is just the amount of strain energy, as shown below.
- The larger the difference, the higher the strain energy of the cycloalkane
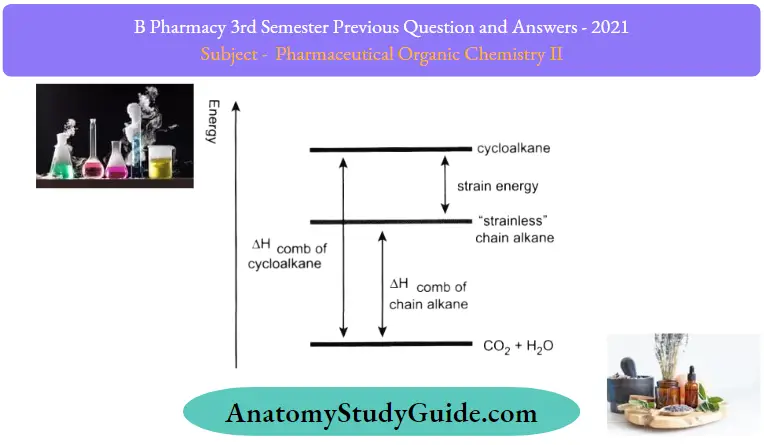
The relationship between heat of combustion and strain energy: Strain Energy(KJ/mol)
-
- Cyclopropane – 114
- Cyclobutane – 110
- Cyclopentane – 25
- Cyclohexane – 0
The major drawback of the Baeyer Theory was that we must assume that all the rings are flat. The highest stability of cyclohexane from experimental results indicates that the rings may not be in a planar shape. We will have a closer look at the actual shape and conformation of 3-, 4-, 5- and 6-membered cycloalkanes.
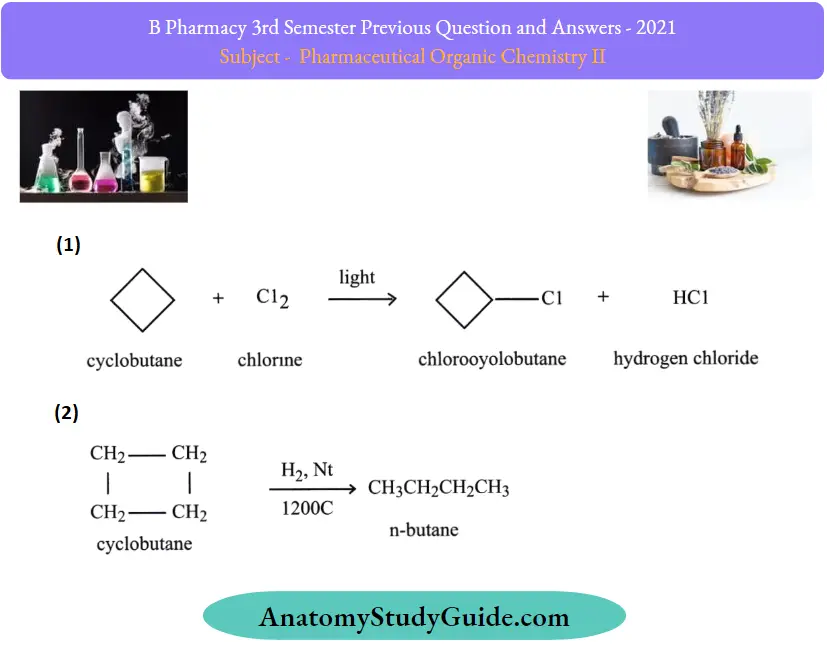
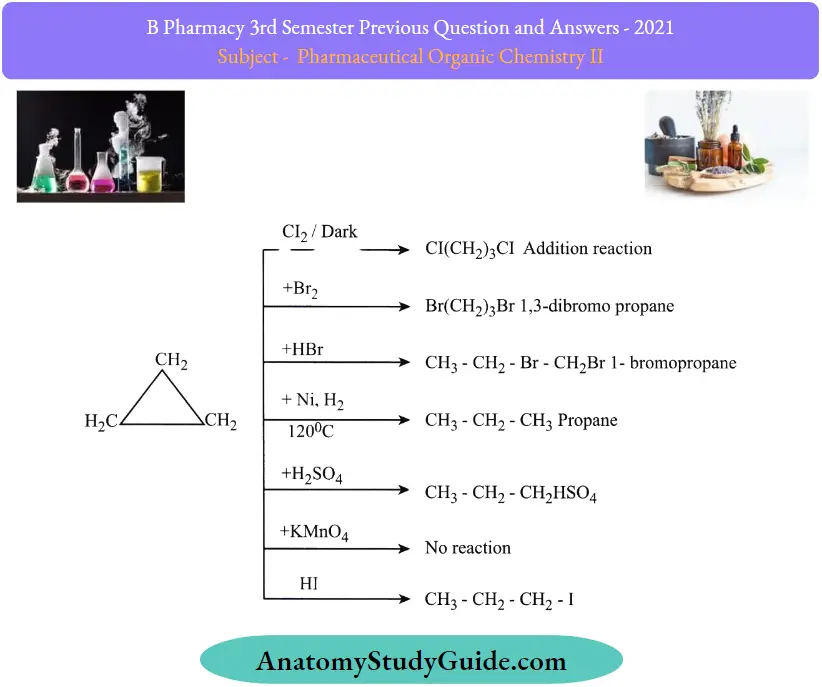
Question 9. Outline two reactions of cyclopropane and cyclobutane.
Answer:
Reactions of cyclobutane:
Reactions of cyclopropane:
Question 10. Write the structure and uses of saccharin and BHC.
Answer:
1. BHC Structure:

BHC Uses:
- Benzene hexachloride is used as an insecticide on crops, in forestry, for seed treatment.
- It is used in the treatment of head and body lice.
- It is used in pharmaceuticals.
- It is used to treat scabies.
- It is used in shampoo
2. Sacchari:
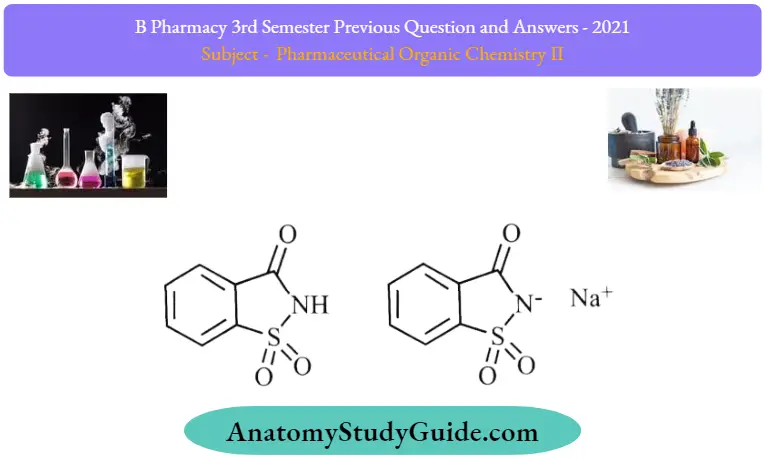
Sacchari Uses:
Added to sweeten low-calorie candies, jams, jellies, and cookies.
Phenol:
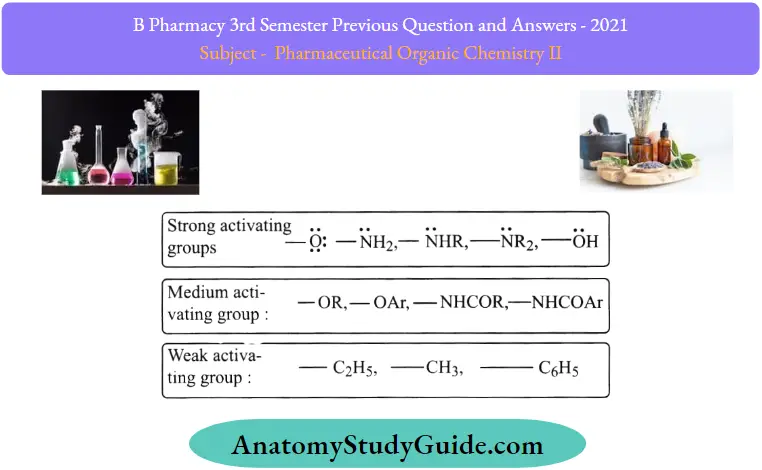
Question 11. Define activating and deactivating groups with examples.
Answer:
Activating and deactivating groups If a group increases the stability thereby increasing the overall reactivity ofthe electrophilic substitution reaction, then it can be termed an activating group. Similarly, if a group reduces the stability of the intermediate thereby reducing the reactivity, then it can be termed as a deactivating group.
Examples:
Activating groups
List of activating groups in organic chemistry:
Deactivating groups:
Nitro groups, carbon tetrachloride.
Question 12. Write the structure and uses of phenol and resorcinol.
Answer:
1. Resorcinol
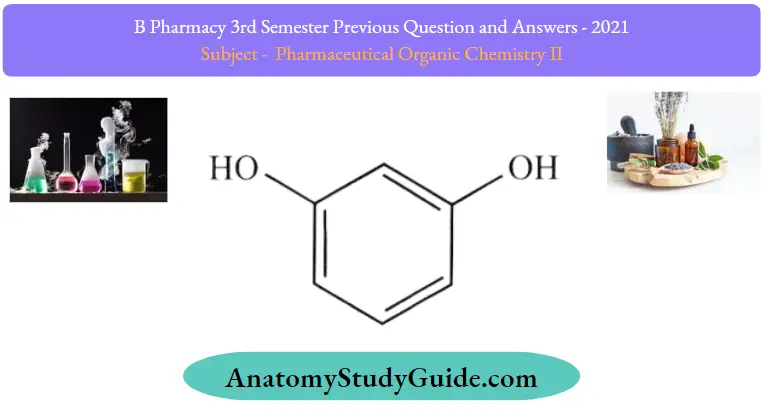
Resorcinol Uses:
Treat acne eczema, psoriasis, corns, roughed skin
2. Phenol

Phenol Uses:
- Disinfectant
- Antibacterial
- Antifungal
- Antiviral
- Oral analgesic
Question 13. Explain the synthetic uses of aryl diazonium salts
.Answer:
Ary
- Diazonium salts serve as an alternative to aryl halides, which have been widely used in transition metal-mediated cross-coupling reactions for carbon-carbon and carbon-hetero bond formation
- The electrophilic nature of diazonium salts arises from N2 being a good leaving group, which does not interfere with the reaction mixture and allows the use of ambient reaction conditions
Question 14. Define rancidity. Give its applications.
Answer:
Rancidity refers to the complete or incomplete hydrolysis or oxidation of fats and oils when exposed to air, light, moisture, and bacterial activity; this generally occurs in food items making them undesirable for consumption.
Rancidity Significance:
- Explains the oxygen level in the oils.
- Explains the oxygen damage in food.
- Explains how the natural structure of the food gets disturbed when the oxygen molecules interact with the oil and food resulting in changes in odor, and taste, and is not advisable for consumption
Question 15. Define ester value.
Answer:
The ester value is the number of mg of potassium hydroxide required to saponify the esters in 1.0 g of the substance. Wax esters are oxo esters of long-chain fatty acids esterified with long-chain alcohols
Question 16. What is Huckle’s rule? Give its significance.
Answer:
Huckel’s Rule is used to estimate the aromatic qualities of any planar ring-shaped molecule in the field of organic chemistry.
The Huckel 4n + 2 Pi Electron Rule
A ring-shaped cyclic molecule is said to follow the Huckel rule when the total number of pi electrons belonging to the molecule can be equated to the formula ‘4n + 2’ where n can be any integer with a positive value (including zero.
Examples of molecules following Huckel’s rule have only been established for values of ‘n’ ranging from zero to six. The total number of pi electrons in the benzene molecule depicted below can be found to be 6, obeying the 4n + 2 5π electron rule where n = 1
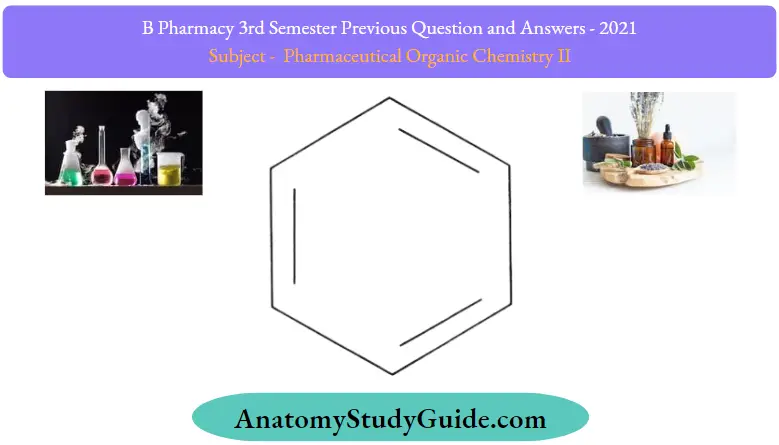
Thus, the aromaticity of the benzene molecule is established since it obeys the Huckel rule.
Huckle’s rule Importance:
- To know the aromaticity.
- To know the stability of geometric or connective arrangements with the same set of atoms.
Question 17. What is Friedalcraft’s acylation?
Answer:
The Friedel-Crafts acylation reaction involves the addition of an acyl group to an aromatic ring. Typically, this is done by employing an acid chloride (R-(C = O.-Cl). And a Lewis acid catalyst such as AlCl3 In a Friedel-Crafts acylation reaction, the aromatic ring is transformed into a ketone. The reaction between benzene and acyl chloride under these conditions is illustrated below.
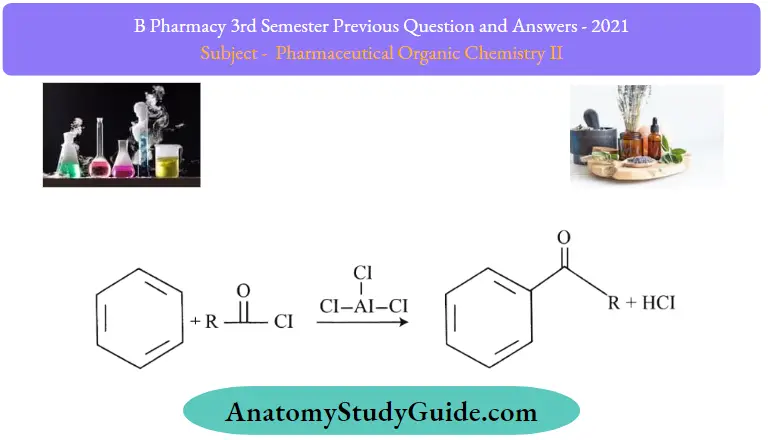
An acid anhydride can be used as an alternative to the acyl halide in Friedel-Crafts acylations. The halogen belonging to the acyl halide forms a complex with the Lewis acid, generating a highly electrophilic acylium ion, which has a general formula of RCO+ and is stabilized by resonance.
Friedel-Crafts acylations Mechanism:
Friedel-Crafts acylations proceed through a four-step mechanism.
Step 1: A reaction occurs between the Lewis acid catalyst (Al3Cl3. and the acyl halide. A complex is formed and the acyl halide loses a halide ion, forming an acylium ion which is stabilized by resonance.
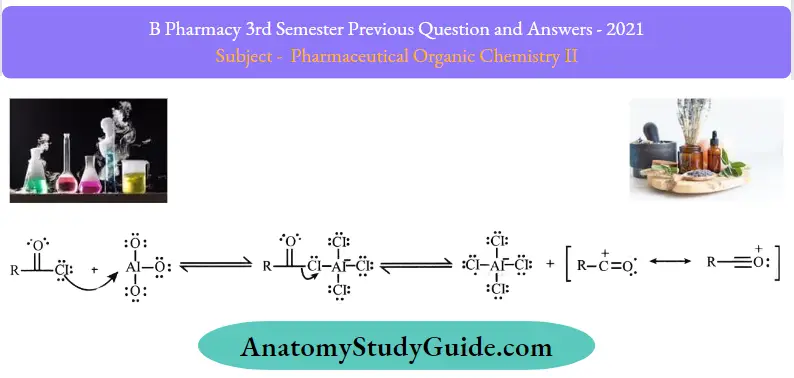
Step 2: The acylium ion (RCO+. goes on to execute an electrophilic attack on the aromatic ring. The aromaticity of the ring is temporarily lost as a complex is formed.
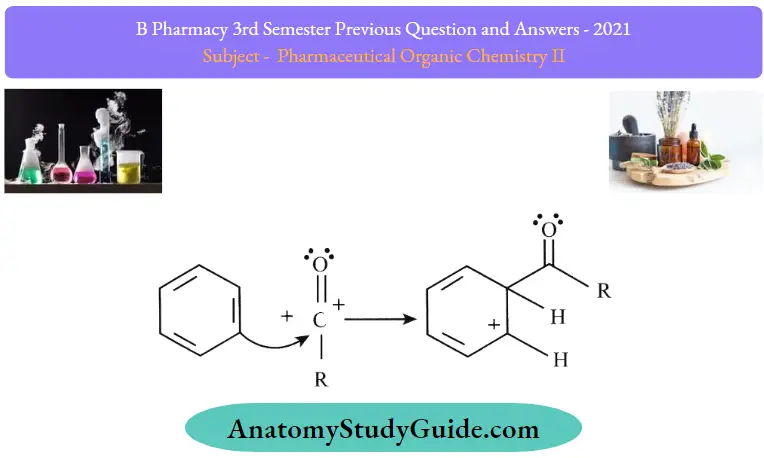
Step 3: The intermediate complex is now deprotonated, restoring the aromaticity to the ring. This proton attaches itself to a chloride ion (from the complexed Lewis acid., forming HCI. The Al3Cl3 catalyst is now regenerated.
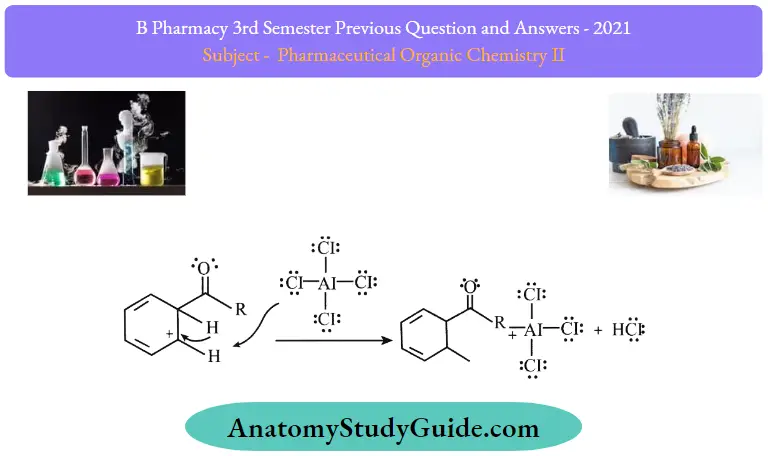
Step 4: The regenerated catalyst can now attack the carbonyl oxygen. Therefore, the ketone product must be liberated by adding water to the products formed in step 3. This step can be illustrated as follows.
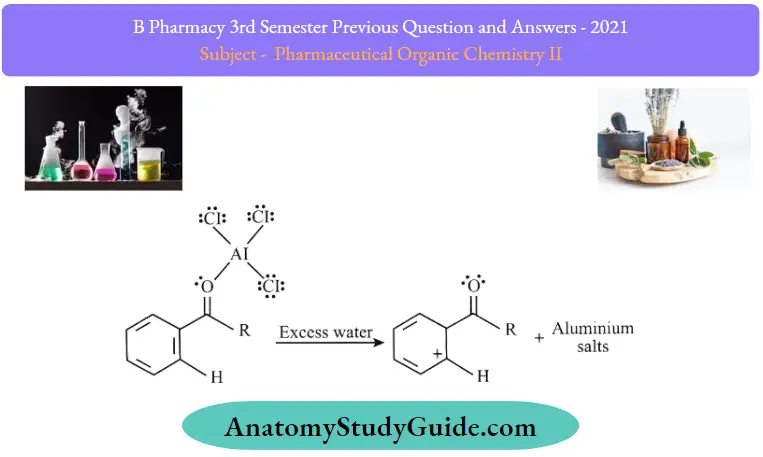
Thus, the required acyl benzene product is obtained via the Friedel-Crafts acylation reaction.
Friedel-Crafts acylations Limitations:
Despite overcoming some limitations of the related alkylation reaction (such as carbocation rearrangement and polyalkylation
- The Friedel-Crafts acylation reaction has a few shortcomings.
- The acylation reaction only yields ketones. This is because formyl chloride (H( C = O.Cl). decomposes into CO and HCl when exposed to these conditions.
- The aromatic compound cannot participate in this reaction if it is less reactive than a mono-halobenzene.
- Aryl amines cannot be used in this reaction because they form highly unreactive complexes with the Lewis acid catalyst.
The acylations can take place on the nitrogen or oxygen atoms when amine or alcohols are used
Question 18. Write the structure and uses of diphenylmethane.
Answer:
Diphenylmethane:
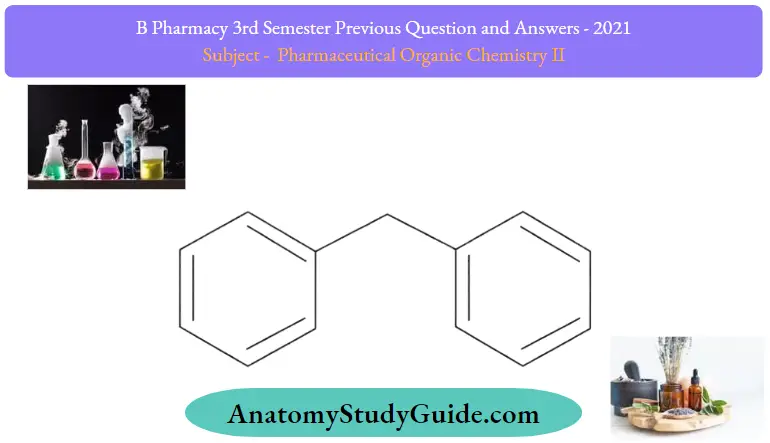
Diphenylmethane Uses:
- Plasticizer to improve dyeing properties.
- Lubricants
Question 19. Define Sache-Mohr theory.
Or
Explain Sache – Mohar theory
Answer:
According to Sachse- Mohr theory, the ring with six or more carbon atoms becomes free from the strain as all the carbon atoms are not forced into one plane. Hence, the carbon atoms occupy different planes where the normal tetrahedral angle is retained.
The rings formed are called Strainless rings.
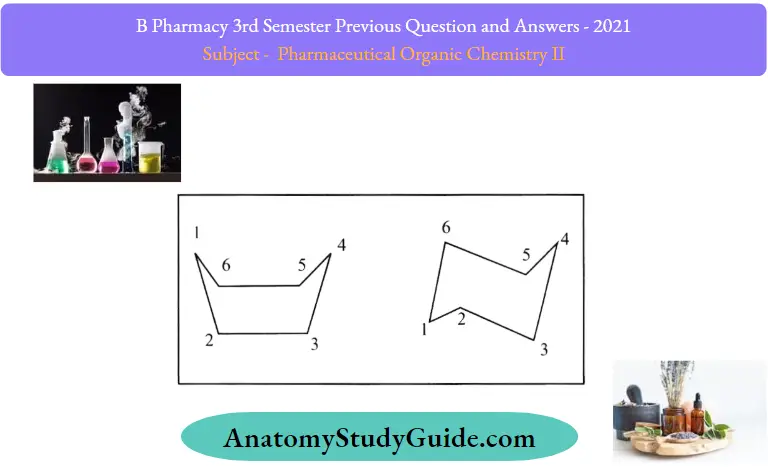
- For example: Cyclohexane exists in two puckered conformations.
- They are ‘Chair form’ and ‘Boat form’.
- Both these forms are without any angle strain. Hence, they are stainless rings.
The chair form of cyclohexane is more stable than the boat form due to the following reasons:
- In the chair conformation, the adjacent C- H bonds on all the neighboring carbon atoms are staggered. In the boat form, the adjacent C-H bonds on C2-C3 and C5-C6 are eclipsed. Hence energy of the boat form becomes more than the chair form.
- Out of the 12 hydrogens, six of them point up or down perpendicular to the plane of the molecule. These are called axial hydrogens. The other 6 hydrogens are found either above or below the plane of the molecule. They are equatorial hydrogens. In the boat form, the two axial hydrogens on C1, and C4 are closer than in chain form. Hence the energy of the boat form is more than chair form.
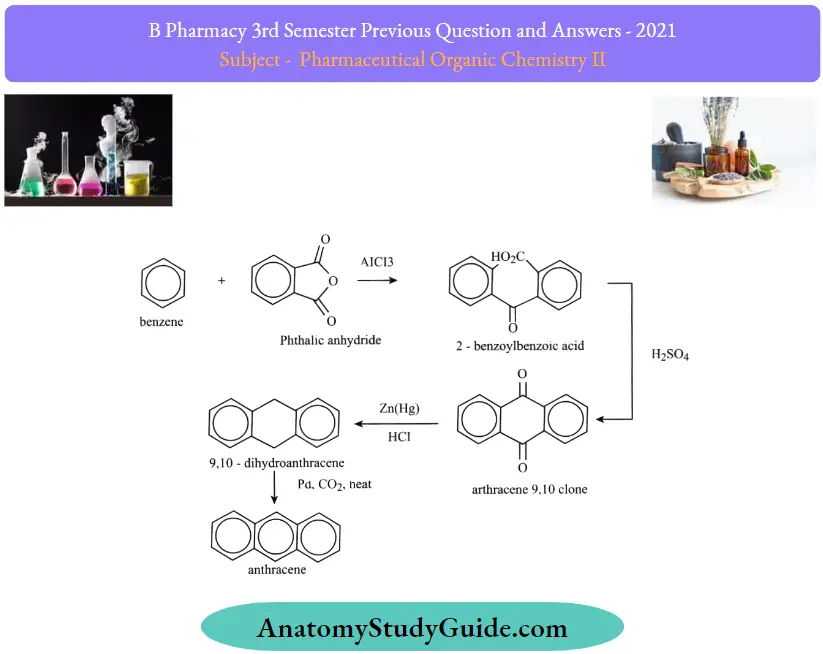
Question 20. Outline the synthesis of anthracene by the Haworth method.
Answer:
Question 21. Explain the Molecular orbital concept of cycloalkanes
Answer:
Molecular orbital concept of cycloalkanes:
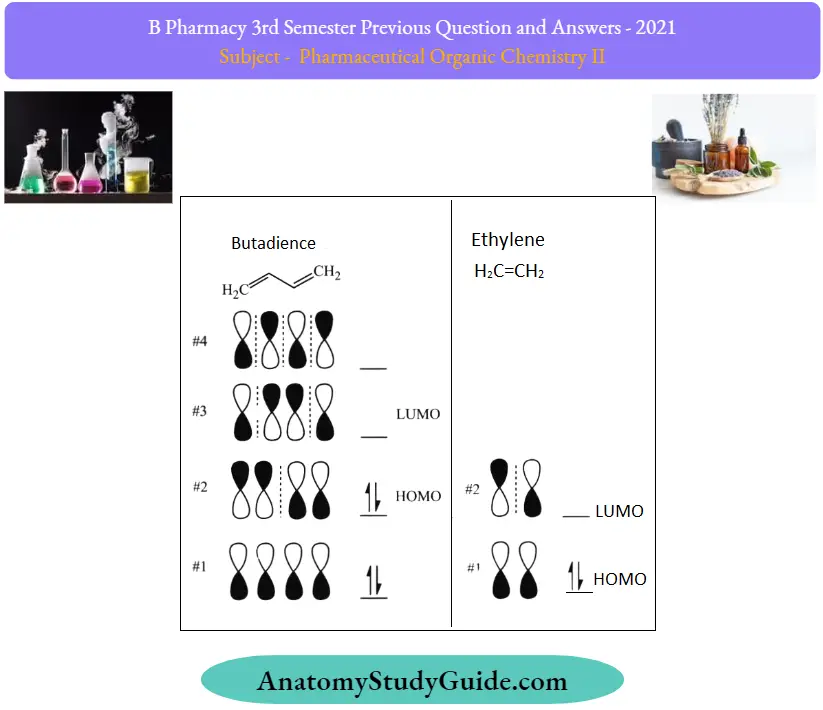
Principles of MO Theory The basic concept of MO theory is to describe the alternating patterns of orbitals that exist in pi-bond systems.
- We describe the orientation of each half of the dumbbell orbital centered around an atom as being bold or open.
- Remember, there are bonding and antibonding orbitals.
- Essentially, orbitals with lower numbers of nodes (switches in the orientation of the dumbbell orbital which we show with the dashed line) behave more like bonding orbitals while orbitals with higher numbers of nodes behave like anti-bonding orbitals.
- To determine what orbitals are occupied on your molecule, simply count
- How many pi bonds do you have in your conjugated system? Below is an example of butadiene. It has 4 pi electrons, so we fill the lowest molecular orbitals first, two in each orbital.
- Therefore, the highest occupied molecular orbital, or HOMO, in butadiene is #2.
- As a result, the lowest unoccupied molecular orbital, or LUMO, in butadiene is #3.
- We can extend this to ethylene and see that with 2 pi electrons, molecular orbital #1 is the HOMO, and molecular orbital #2 is the LUMO.
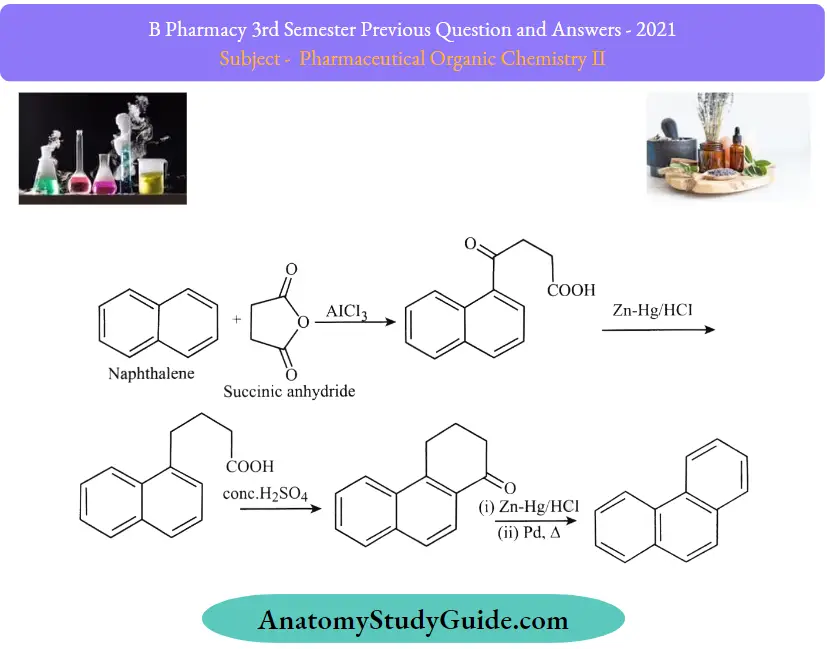
Question 22. Define and classify polynuclear hydrocarbons. Give any two syntheses of naphthalene
Answer:
A polynuclear aromatic hydrocarbon is a hydrocarbon made up of fused aromatic ring molecules. These rings share one or more sides and contain delocalized electrons. Another way to consider PAHs is molecules made by fusing two or more benzene rings
Synthesis of Naphthalene:
1. Synthesis of Naphthalene:
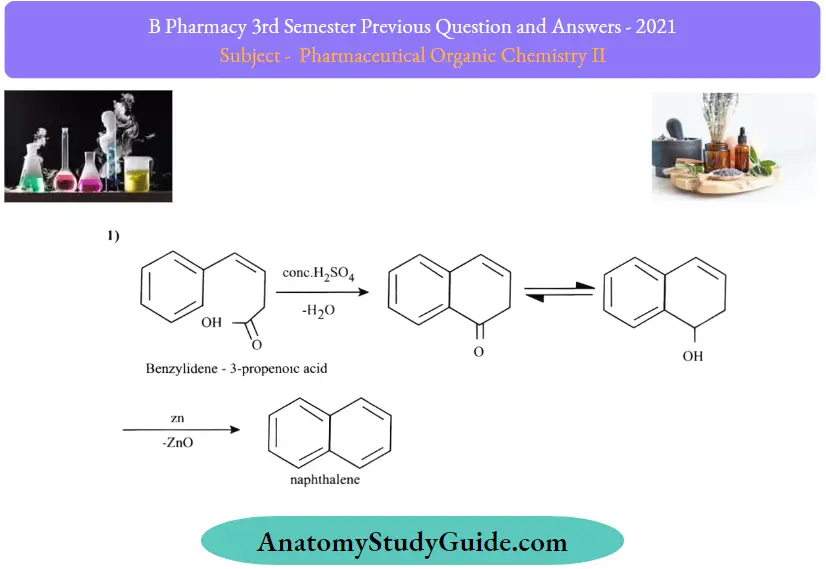
2. Haworth Synthesis:
In this process, benzene to naphthalene is produced. The benzene ring is first subjected to the acylation process. This reaction starts with the Friedel craft acylation of the benzene ring with the succinic anhydride, and then a series of reduction reactions undergo. Finally, a naphthalene molecule is produced by dehydrogenation. The structure of naphthalene is confirmed by the method of its synthesis
Howarth method:
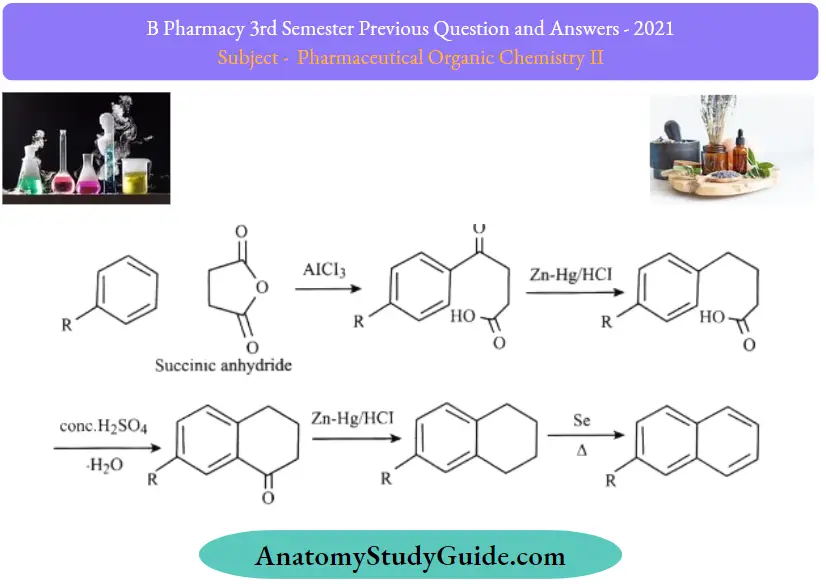
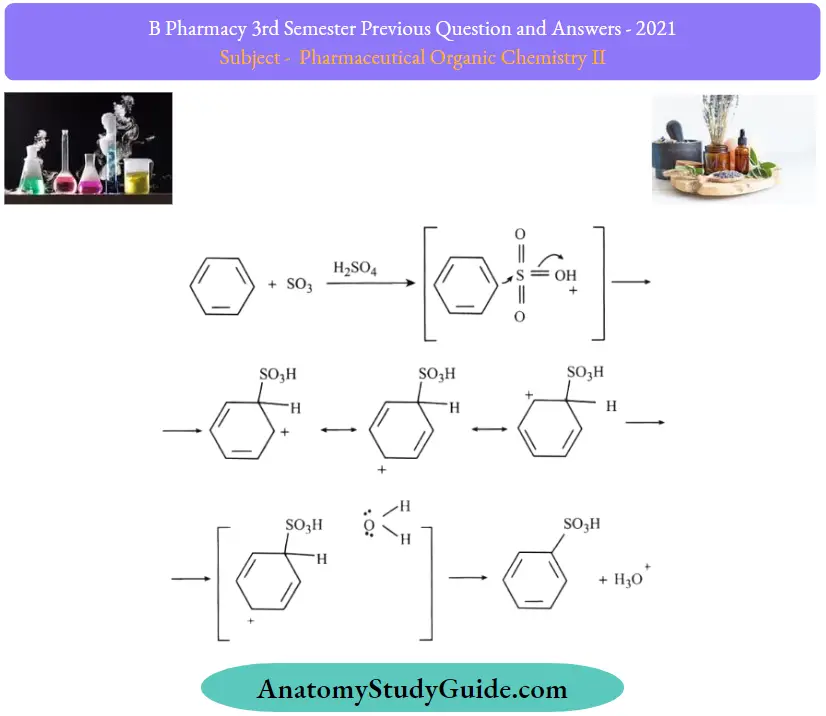
Question 23. Explain the reaction and mechanism of sulphonation of benzene.
Or
Answer:
The mechanism for Sulfonation of benzene:
Due to higher electronegativity, oxygen present in sulphuric acid pulls an electron towards itself, generating an electrophile. This attacks the benzene ring, leading to the formation of benzene sulfonic acid.
Question 24. Describe any one method to determine the Reichert-Meissi value with its significance
Answer:
The Reichert-Meissl value is the number of milliliters of 0.1N aqueous sodium hydroxide solution required to neutralize steam volatile water-soluble fatty acids distilled from 5g of oil/fat under the prescribed conditions. It is a measure of water-soluble steam volatile fatty acids chiefly butyric and caproic acids present in oil or fat.
Reichert-Meissi Principle:
- The material is saponified by heating with glycerol sodium hydroxide solution and then split by treatment with dilute sulfuric acid.
- The volatile acids are immediately steam distilled.
- The soluble volatile distillate is filtered out and estimated by titration with standard sodium hydroxide solution.
Reichert-Meissi Procedure:
Weigh accurately 5 ±0.1g of filtered oil or fat sample into a clean, dry, 300ml distilling flask. Add 20 ml of glycerine and 2 ml of concentrated sodium hydroxide solution, and heat by swirling over a flame until completely saponified, as shown by the mixture becoming clear.
- Cool the contents slightly and add 90 ml of boiling distilled water, which has been vigorously boiled for about 15 min.
- After thorough mixing the solution should remain clear.
- If the solution is not clear (indicating incomplete saponification) or is darker than light yellow (indicating over-heating), repeat the saponification with a fresh sample of the oil or fat.
- If the sample is old, the solution may sometimes be dark and not clear. Add about 0.6 – 0.7 gm of pumice stone grains and 50 ml of dilute sulfuric acid solution.
- Immediately connect the flask to the distillation apparatus.
- Place the flask on the asbestos board so that it fits snugly into the aperture. This will prevent the flame from impinging on the surface of the flask above the level of the liquid and avoid superheating.
- Heat very gently until the liberated fatty acids melt and separate.
- Then set the flame so that 110 ml of distillate shall be collected within 19 to 21 min.
- The beginning of the distillation is to be taken as the moment when the first drop of the distillate falls from the condenser in the receiving flask
- Keep the water in the condenser flowing at a sufficient speed to maintain the temperature of the outgoing water from the condenser between 15 and 20°C.
- Collect the distillate in a graduated flask. When the distillate exactly reaches the 110 ml mark on the flask, remove the flame and quickly replace the flask with a 25 ml measuring cylinder.
- Stopper the graduated flask and without mixing place it in a water bath maintained at 15°C for 10 min so that the 110 ml graduation mark is 1 cm below the water level in the bath.
- Swirl around the contents of the flask from time to time. Remove the graduated flask from the cold water bath, dry the outside, and mix the content gently by inverting the flask 4 to 5 times without shaking. Avoid wetting the stopper with the insoluble.
Filter the liquid through a dry, 9 cm Whatman No. 4 filter paper. Reject the first 2-3 ml of the filtrate and collect the rest in a dry flask. The filtrate should be clear. Pipette 100 ml ofthe filtrate and add 5 drops ofthe phenolphthalein solution, and titrate against standard 0. IN sodium hydroxide solution. Run a Blank Test without the fat, but using the same quantities of the reagents.
Reichert-Meissl Calculation:
Reichert-Meissl Value = (A – B) × N × 11
Where,
A = Volume in ml of standard sodium hydroxide solution required for the test;
B = Volume in ml in standard sodium hydroxide solution required for the blank; and
N = Normality of standard sodium hydroxide solution.
Reichert-Meissl Significance:
These determinations have been used principally for the analysis of butter and margarine. Butterfat contains mainly butyric acid glycerides.
- Butyric acid is volatile and soluble in water.
- No other fat contains butyric acid glycerides, and therefore, the ReichertMeissl value of the butter fat is higher than that for any other fat.
- Coconut oil and palm kernel oil contain appreciable quantities of caprylic capric and lauric acid glycerides
Question 25. Give any two chemical reactions of cyclobutane
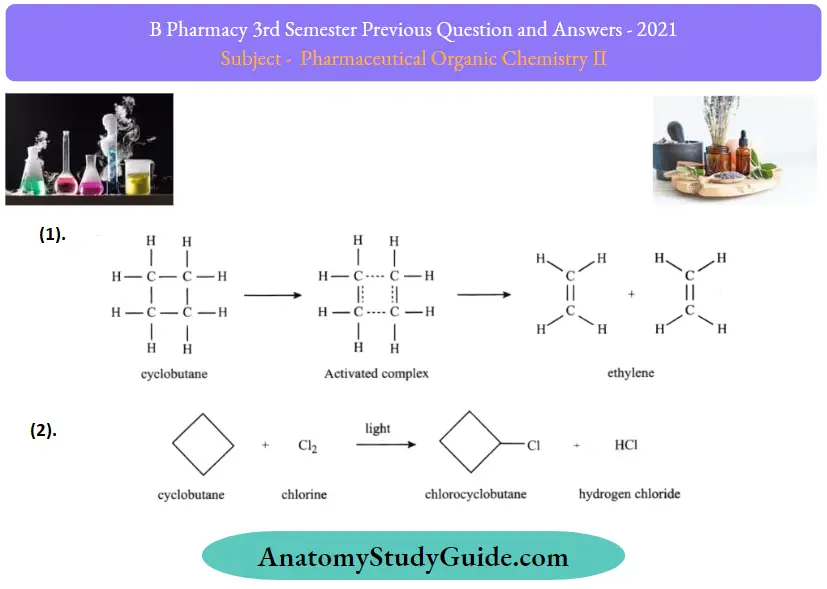
Question 26. Write a note on the drying of oils.
Answer:
A drying oil is an oil that hardens to a tough, solid film after a period of exposure to air, at room temperature. The oil hardens through a chemical reaction in which the components crosslink (and hence, polymerize) by the action of oxygen (not through the evaporation of water or other solvents).
- Drying oils are a key component of oil paint and some varnishes. Some commonly used drying oils include linseed oil, tung oil, poppy seed oil, perilla oil, and walnut oil.
- Drying oils consist of glycerol trivers of fatty acids. These esters are characterized by high levels of polyunsaturated fatty acids, especially alpha-linolenic acid.
- One common measure ofthe “siccative” (drying) property of oils is iodine number, which is an indicator of the number of double bonds in the oil.
- Oils with an iodine number greater than 130 are considered drying, those with an iodine number of 115-130 are semi-drying, and those with an iodine number of less than 115 are non-drying
Question 27. Give the composition of fats and oils
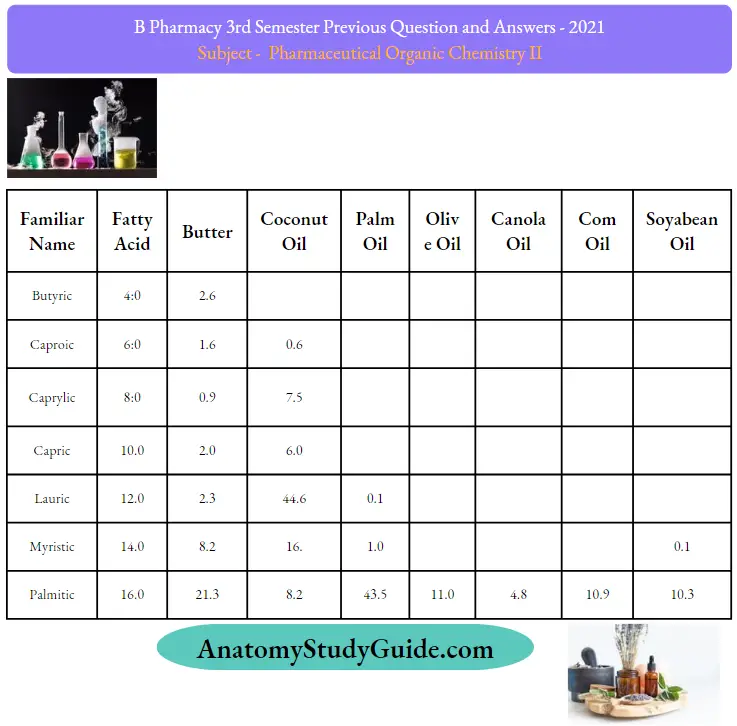
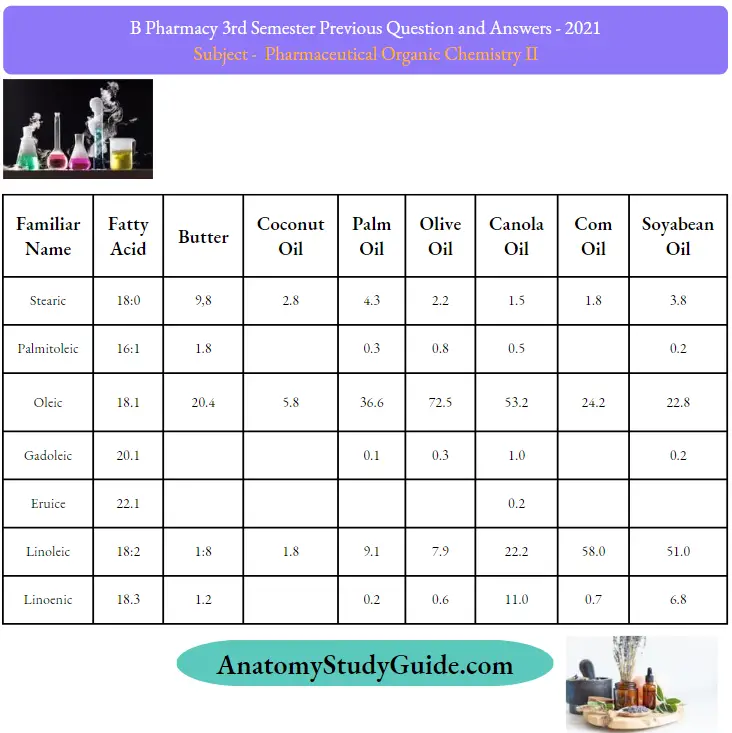
Question 28. Angle in cyclopropane?
Answer:
Question 29. What are fatty acids? Give an example of unsaturated fatty acid
Answer:
Fatty acids are a range of molecules with the general formula Cn H (2n + 1)COOH that occur naturally in many organisms, often combined with glycerol to form FATS.
There are two types of fatty acids:
- Saturated fatty acids
- Unsaturated fatty acids
Examples of saturated fatty acids: Are caprylic acid, lauric acid, palmitic acid, and stearic acid.
Examples of unsaturated fatty acids: Are palmitoleic acid, linoleic acid, arachidonic acid
Question 30. Give the structure and uses of o-cresol
Answer:
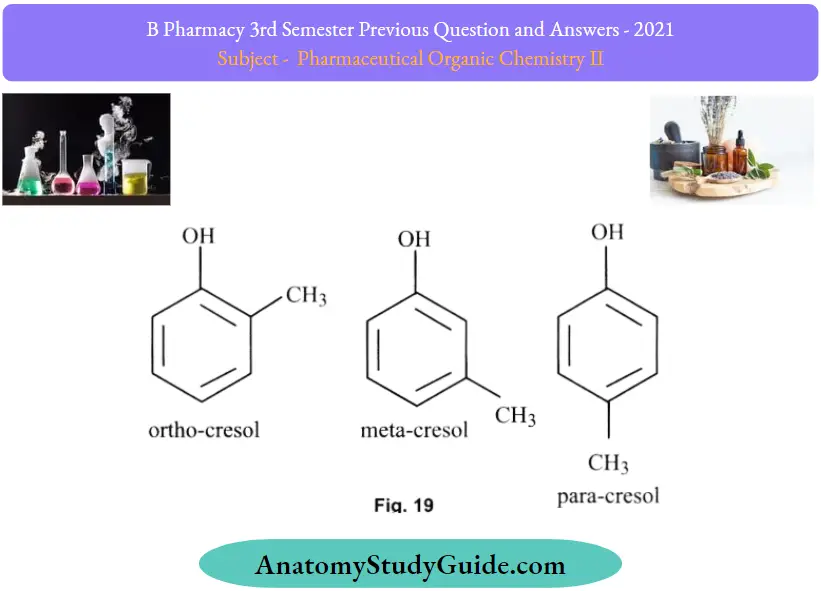
Question 31. Give the qualitative test of phenol.
Answer:
Litmus Test:
Scientists use litmus paper to test whether the given solution is acidic or basic. Red litmus paper turns blue while blue litmus paper remains unchanged in the presence of a base. Phenol turns blue litmus paper red. This shows that phenol is acidic. Carboxylic acid also gives this test. Compared to carboxylic acid phenol is weakly acidic and it does not give an effervescence with aqueous sodium carbonate.
Bromine water test:
Phenol undergoes electrophilic substitution reaction with bromine. When bromine water is added to an aqueous solution of phenol the brown color of bromine disappears and a white precipitate of tribromophenol is formed.
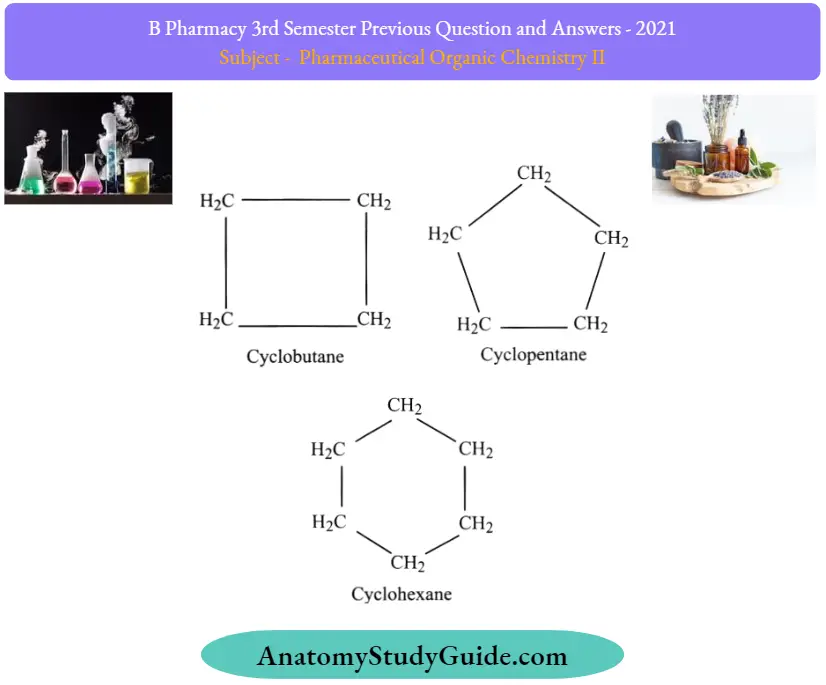
Question 32. Define cycloalkanes and give examples.
Answer:
Cycloalkanes are a class of hydrocarbons having a ring-like structure. This ring is formed due to their saturated nature, and they have three compounds of methane present in the structure which helps them in forming a ring. They have the general formula Cn H2n, Where n is said to be the number of carbon atoms present in the organic compound.
Some common examples of cycloalkanes are cyclopentane, Cyclobutane, cyclohexane, cycloheptane, cyclooctane
Question 33. Define deactivating groups. Give examples.
Answer:
If a group reduces the stability of the intermediate thereby reducing the reactivity, then it can be termed a deactivating group.
Examples:
NO2, CF3. CN, halogens, COOH
Pharmaceutical Organic Chemistry 2 Short Question And Answers 2020
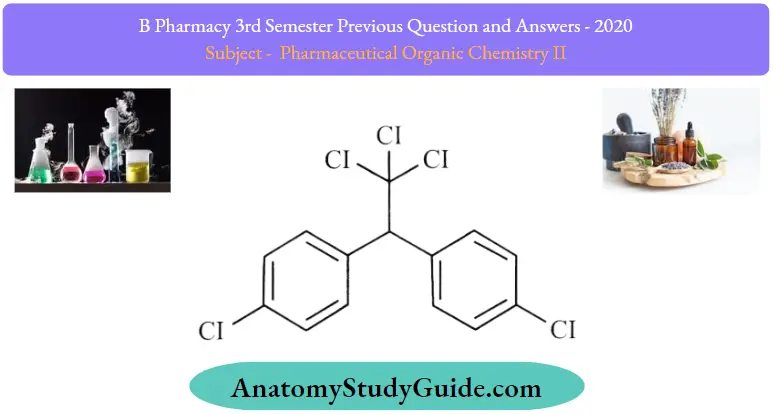
Question 1. Write the structure and uses of DDT
Answer:
DDT Structure:
Question 2. Give the structure and uses of naphthols
Answer:
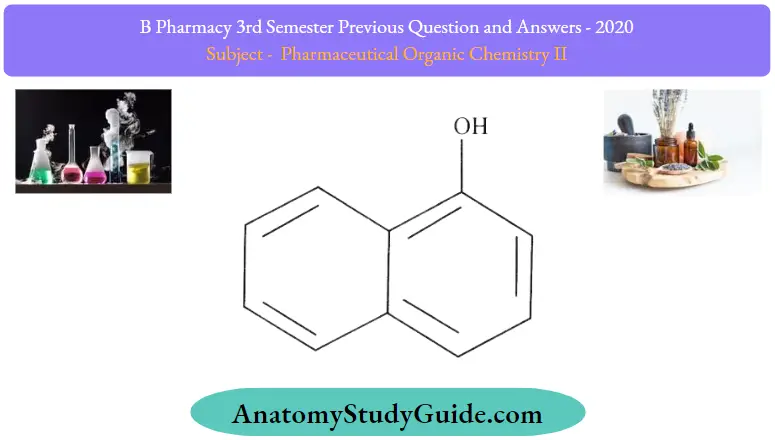
Naphthols Uses:
- Counterirritant in alopecia
- Anthelmintic
- Antiseptic
Question 3. What is Friedelcraft’s reaction?
Answer:
A Friedel-Crafts reaction is an organic coupling reaction involving an electrophilic aromatic substitution that is used for the attachment of substituents to aromatic rings. The two primary types of Friedel-Crafts reactions are the alkylation and acylation reactions.
Question 4. What is a cousin and Moffitt modification?
Answer:
In this theory, both are considered cyclopropane rings from sp³ hybridized CH2 groups. All sp² hybridized are posted 22 degrees outward from the imaginary line connecting the nuclei, resulting in about 20% less effective overlap than the C-C bond of ethane. For this reason, the bonds are often referred to as bent. This bending is considered for angle strain.
Pharmaceutical Organic Chemistry 2 Short Question And Answers 2019
Question 1. Give the effect of substituents on the acidity of phenols.
Answer:
Substituents have a great effect on the acidity and basicity of compounds. Substituents can increase or decrease the acidity of phenols. Let’s see how substituents affect the acidity of phenols. Let’s get started. Substituents are of
Two types i.e. Electron withdrawing and Electron donating.
- Electron withdrawing groups: These groups pull electrons from the ring and decrease electron density over the ring
- For example: NO2,-COCH3,-CHO, etc.
- Electron donating groups: These groups donate electrons to the ring and increase electron density over the ring.
- For example: – CH3,-NH2, -OH etc.
Now, according to Lewis’s acid-base theory, the acid is a species that can accept electrons.
- Now a species will be acidic if it accepts the electron. And to be able to accept electrons the species must be electron deficient.
- Less the electron density present in the electron-accepting group more easily it will accept electrons and the more acidic it will be.
- High electron density destabilizes the structure and makes it unable to accept electrons and it will be less acidic.
- So the effect of the substituent will depend upon the ability of the substituent to increase or decrease the electron density over the structure.
Effect Of Electron Withdrawing Groups:
As we know the electron-withdrawing groups (such as -NO2 -CHO,-CN, etc.) pull electrons towards them, and they ultimately decrease electron density over the hydroxyl group
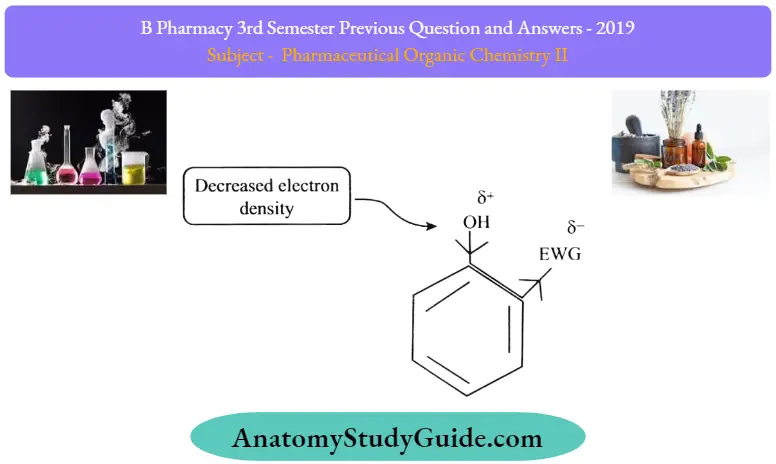
Effect Of Electron Donating Groups:
As the name suggests the electron donating groups (such as -NH2 -OH etc.) give electrons to the ring increase electron density over the oxygen in phenol, and destabilize the peroxide ion after releasing proton from phenol That makes the phenol to accept electrons and loosing proton. That decreases the acidity of phenol. That means electron-donating groups decrease the acidity of phenols.
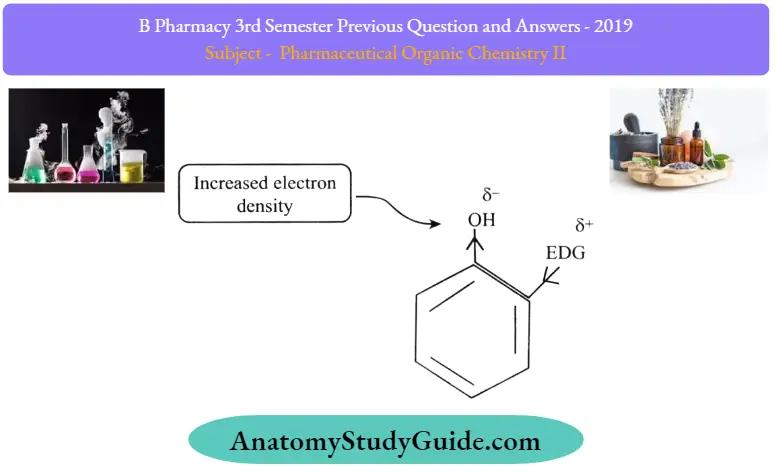
We can summarize it as electron-withdrawing groups increasing the acidity of phenols and electron-donating groups decreasing acidity of phenols. That’s how electron withdrawing and electron donating substituents affect the acidity of phenols.
Question 2. Describe any one method to determine iodine value with its Apparatus and Reagents.
Answer:
Iodine Apparatus:
- Glass stoppered Erlenmeyer flask 500 ml
- Measuring Cylinder (50 ml)
- Bulb pipette ( 20 ml capacity)
Iodine Reagents:
- Glacial Acetic Acid, free from ethanol: AR grade
- Carbon Tetrachloride or Chloroform: AR grade
- Potassium dichromate: AR grade
- Concentrated hydrochloric acid: AR grade
- Iodine Monochloride
- Preparation of Wij’s solution when using ampoules of iodine monochloride:
Question 3. Give a note on the hydrogenation of fats and oils.
Answer:
Hydrogenation:
Hydrogenation is a process that reduces the unsaturated fatty acid content of triglycerides by attaching hydrogen atoms at the point of unsaturation in the presence of a catalyst, usually Nickel.
- Hydrogenation is a process in which a liquid unsaturated fat is turned into a solid fat by adding hydrogen.
- Hydrogenation accomplishes two things-:
- It increases the melting point of the oil or fat.
- Resistance to oxidation and flavor deterioration.
- The hydrogenation process depends on several parameters:
- Pressure& Temperature,
- Type of catalyst,
- Speed of agitation etc.
Hydrogenation Mechanism:
The mechanism involved in fat hydrogenation is believed to be the reaction between unsaturated liquid oil and atomic hydrogen adsorbed on a metal catalyst. It is similar to the hydrogenation of alkenes.
Generally, hydrogenation reactions will not occur between hydrogen and organic compounds below 480 degrees Celsius without metal catalysts.
- Catalysts are responsible for binding the H2 molecule and facilitating the reaction between the hydrogen and the substrate.
- Platinum, palladium, rhodium, and ruthenium are known to be active catalysts that can operate at lower temperatures and pressures.
- Research is ongoing to procure non-precious metal catalysts that can produce similar activity at lower temperatures and pressures.
- Nickel-based catalysts, such as Raney nickel, have been developed, but still require high temperatures and pressures.
Monitoring End of Reaction Technique Used Principle Refractive:
- Index: Refractive Index decreases on hydrogenation
- Iodine Value: Iodine value decreases on hydrogenation
- Melting Point: Melting point increases on hydrogenation
- Peroxide Value: Peroxide value decreases on hydrogenation
- Color: Colour becomes darker on hydrogenation
Hydrogenation Significance:
- It increases the melting point ofthe oil or fat.
- Resistance to oxidation and flavor deterioration.
- The hydrogenation process depends on several parameters as pressure, temperature, type of catalyst, and speed of agitation
Question 4. Write the structure and uses of chloramine
Answer:
Chloramine:
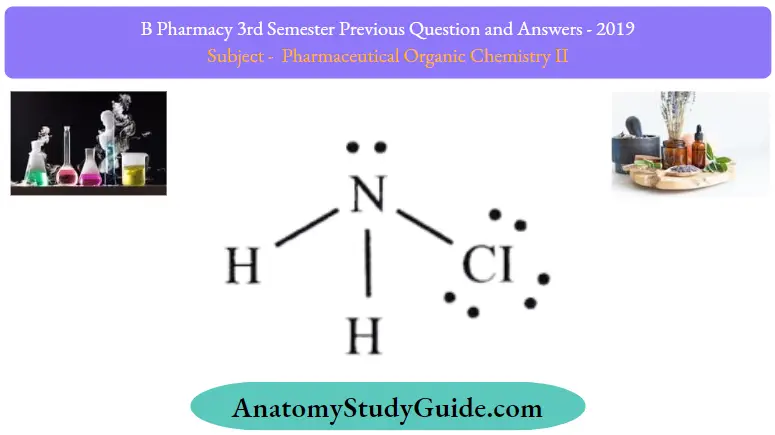
Chloramine Uses:
- Disinfectant
- Treat bums, wounds
- Used as oral mouthwash
Question 5. Define acid value. Give its significance.
Answer:
Acid value:
The milligrams of KOH required to completely neutralize free acid present in 1 g of oils is known as the acid value or neutralization number.
Acid value Significance:
- The acidity of oil is due to hydrolysis or oxidation of oil by atmospheric moisture leading to the formation of fatty acids.
- Lubricant oil with acid values greater than 0.1 corrodes metals, and forms gum, and sludge during operation.
Question 6. Define acetyl value. Give its significance.
Answer:
Acetyl value:
It is the number that expresses in milligrams the amount of potassium hydroxide required to neutralize the acetic acid liberated by the hydrolysis of one gram ofthe acetylated substance.
Acetyl value Significance:
- Wet chemical fire extinguishers: To extinguish cooking oils and fats, we use a saponification reaction. This is because cooking oils and fats have a flashpoint which is above 37 degrees which renders regular fire extinguishers useless.
- Creating hard and soft soaps: By using different types of alkali in the process the type of reaction product can be altered between hard and soft.
Using KOH: We can obtain soft soaps
Using NaOH: We can obtain hard soaps
Question 7. Give structure and uses of one medicinally important phenanthrene derivative.
Answer:
Morphine Structure:
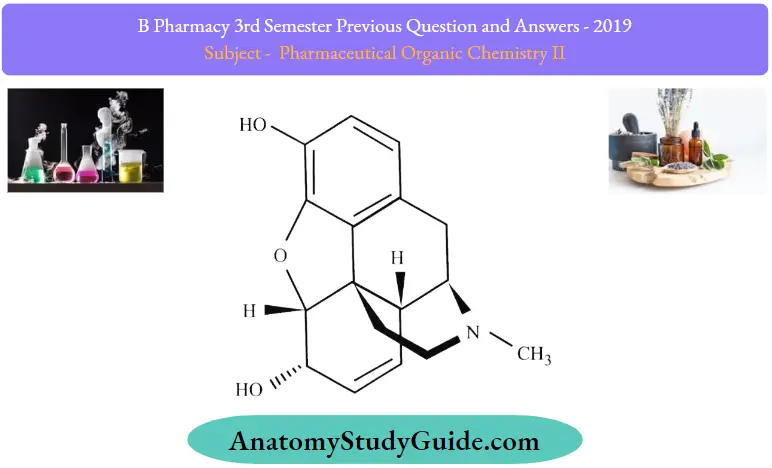
Morphine Uses:
- Acts as photoreceptor molecules
- Dyes
Question 8. Give the structure and medicinal uses of triphenylmethane.
Answer:
Triphenylmethane Structure:
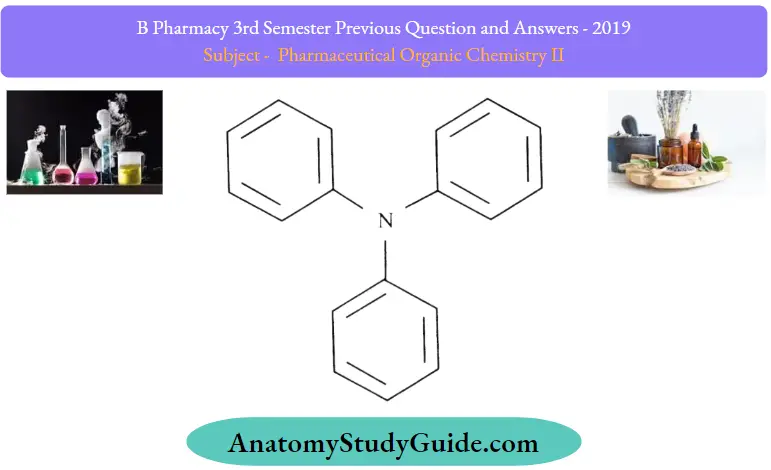
Triphenylmethane Uses:
- Acts as photoreceptor molecules.
- Dyes.
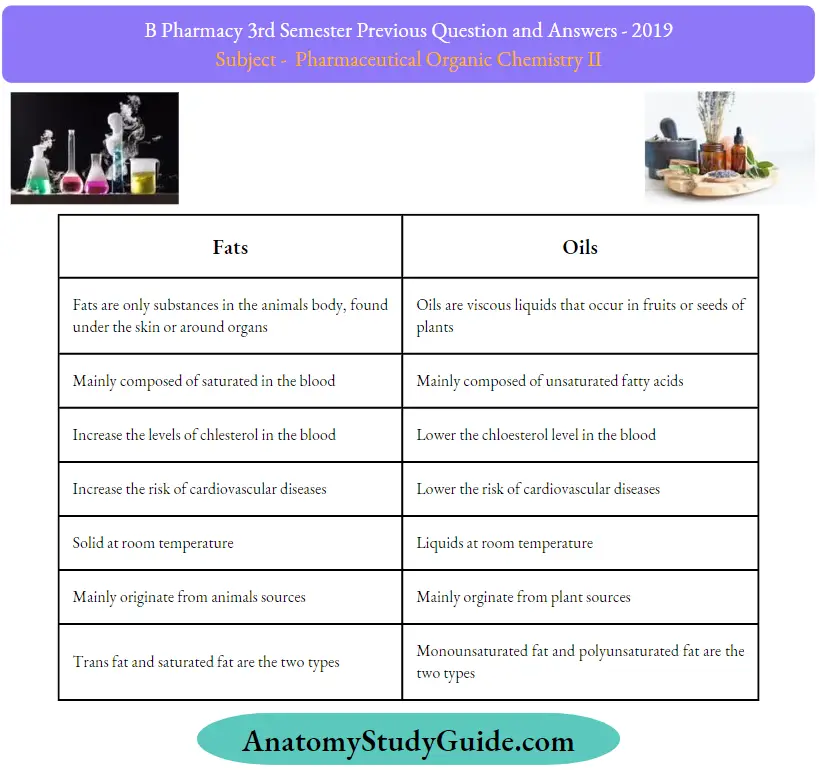
Question 9. Write the difference between oils and fats.
Answer:

Question 10. Define activating and deactivating groups with examples. Discuss the mechanism of sulphonation of benzene.
Answer:
Activating and deactivating groups If a group increases the stability thereby increasing the overall reactivity ofthe electrophilic substitution reaction, then it can be termed as activating group. Similarly, if a group reduces the stability of the intermediate thereby reducing the reactivity, then it can be termed a deactivating group.
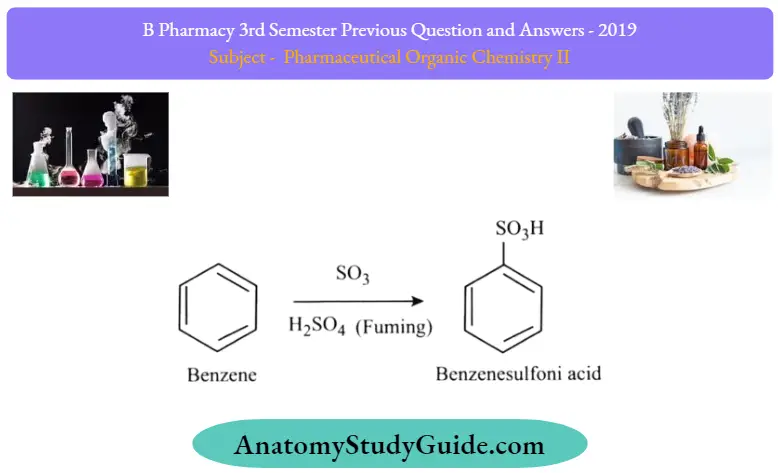
Sulfonation of Benzene:
Sulfonation is a reversible reaction that produces benzenesulfonic acid by adding sulfur trioxide and fuming sulfuric acid. It is important to note that the chemical formula of the sulfonic group is -S03H. The reaction is reversed by adding hot aqueous acid to benzenesulfonic acid to produce benzene.
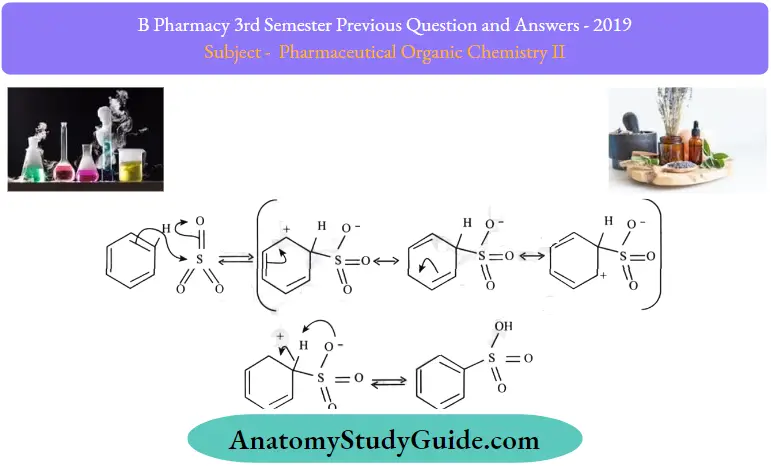
Sulfonation of Benzene Mechanism:
To produce benzenesulfonic acid from benzene, fuming sulfuric acid and sulfur trioxide are added. Fuming sulfuric acid also referred to as oleum, is a concentrated solution of dissolved sulfur trioxide in sulfuric acid. The sulfur in sulfur trioxide is electrophilic because the oxygens pull electrons away from it. After all, oxygen is very electronegative. The benzene reacts with the sulfur of sulfur trioxide to form the sigma complex. A subsequent proton transfer occurs to produce benzenesulfonic acid. All three steps are shown together in the mechanism below.
Reverse Sulfonation:
Sulfonation of benzene is a reversible reaction. Sulfur trioxide readily reacts with water to produce sulfuric acid and heat. Therefore, by adding heat to benzenesulfonic acid in diluted aqueous sulfuric acid the reaction is reversed
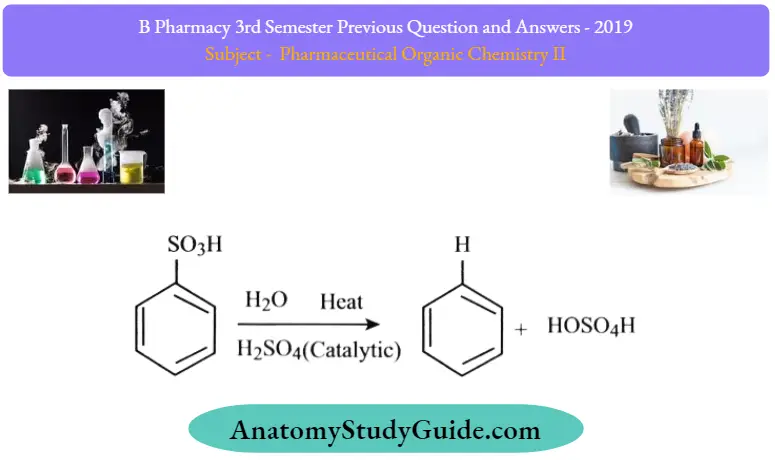
The reversibility of the sulfonation reaction creates an opportunity to prepare deuterated benzene. Isotopically labeled reagents can be useful in determining reaction mechanisms since the C-D bond is stronger than the CH bond.
Question 11. Explain the aromaticity, orbital picture, and resonance structure of benzene.
Answer:
Aromaticity of benzene:
Aromaticity is defined as a property of the conjugated cycloalkenes that enhances the stability of molecules due to the delocalization of electrons present in the n-n orbitals.
Aromatic molecules are said to be very stable, and they do not break so easily and also react with other types of substances. The organic compounds which are not said to be aromatic are known as aliphatic compounds. These might be in cyclic form, but only the aromatic rings have a special kind of stability
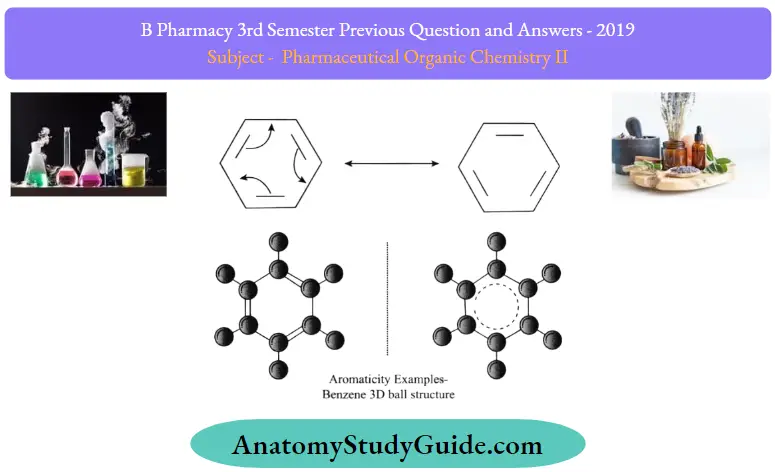
Aromatic compounds are less stable compounds, and they have many kinds of chemicals as well as synthetic uses. The nucleic acids and the amino acids that make up our cell structure make use of these aromatic compounds.
- But, the main thing is what makes aromatic compounds. The aromatics compounds are said to exhibit some special characteristics called rules which are given below-
- The aromatic compounds are always cyclic structures.
- Each element ofthe ring within the structure must and should have a p-orbital ring which is in a perpendicular form to the ring, and this makes it a planar molecule
- All the compounds obey Huckel’s Rule, i.e all the aromatic compounds should have the (4n+2) Pi number of electrons.
- The last one is that the organic compound has to be flat.
Orbital picture of benzene:
The structure of the benzene molecule is best described in terms of molecular orbital treatment theory. According to this theory, all the C-atoms in benzene are sp²- hybridized. The remaining six sp²- orbitals of six C-atoms overlap with the orbital of six H-atoms individually to form six sigma bonds.
Resonance structure of benzene:
Benzene is a very important aromatic hydrocarbon in organic chemistry. It has the chemical formula C6H6. The molecules of benzene have a cyclic structure consisting of alternating single and double bonds between adjacent carbon atoms. Each carbon atom is also bonded to one hydrogen atom. The two possible resonance structures of benzene are illustrated below.
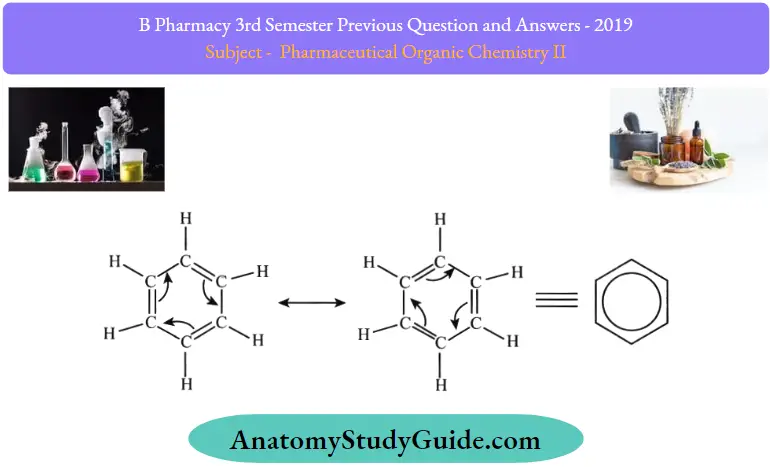
The benzene molecule is stabilized by resonance, the pi electrons are delocalized around the ring structure.
- This delocalization causes each carbon-carbon bond to have a bond order of 1.5, implying that they are stronger than regular C-C sigma bonds.
- In the resonance hybrid of benzene, the delocalization of pi electrons is described with the help of a circle inside the hexagonal ring.
- In benzene, Kekule first suggested two cyclohexatriene Kekule structures that have been taken together, they constitute the general structure as contributing structures.
- The hexagon replaces three double bonds in the hybrid structure on the right and represents six electrons in a collection of three molecular orbitals with a nodal plane in the molecule plane.
- Thus, the resonance structures of some molecules and polyatomic ions are discussed briefly in this article.
- Learn more about this concept and other related concepts such as hyperconjugation, resonance effect, and electron dot formula.
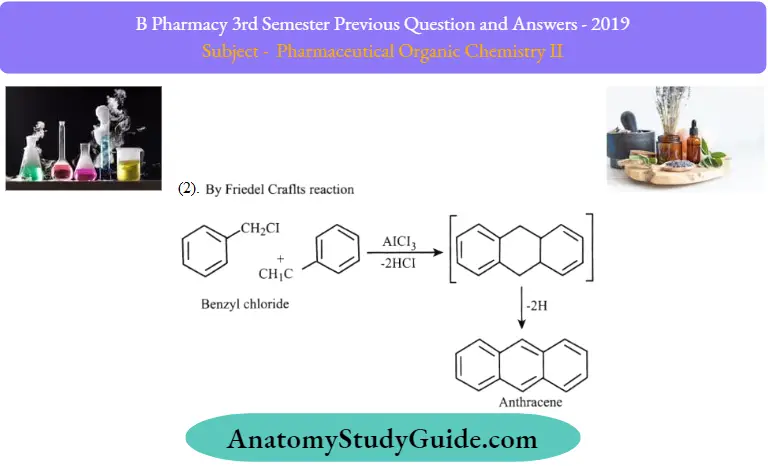
Question 12. Write any two syntheses and reactions of anthracene
1. By Haworth Synthesis:
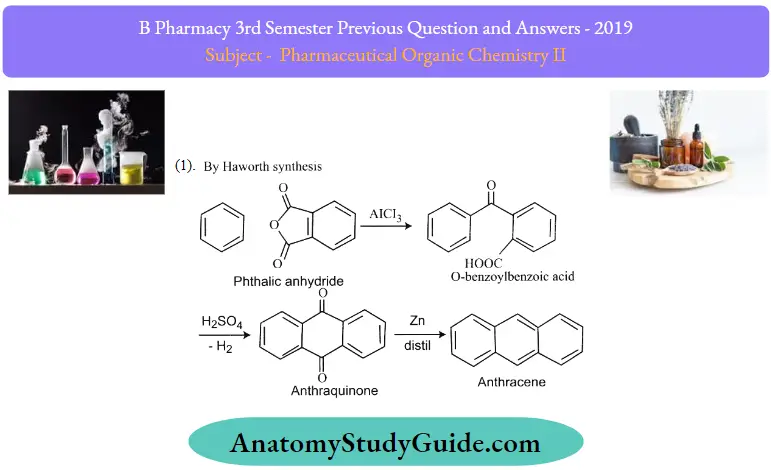
2. By Friedel Crafts Reaction:
Question 13. Describe any one method to determine iodine value with its significance.
Answer:
The iodine value is the number that expresses in grams the quantity of halogen, calculated as iodine which is absorbed by 100 gm of the substance.
Method for determination of iodine value:
- Wijs method (iodine monochloride method):
- Place an accurately weighed quantity ofthe substance under examination in a dry 500ml iodine flask, add 10ml of carbon tetrachloride, and dissolve.
- Add 20ml of iodine monochloride solution, insert the stopper, and allow to stand in the dark at 25°C for 30 minutes.
- Place 1 5ml of potassium iodide solution in the cup top, carefully remove the stopper, rinse the stopper and the sides ofthe flask with 100ml of water, shake, and titrate with 0.1M solution thiosulphate using starch as indicator. Note the number of ml required.
- Repeat the operation without the substance under examination note the number of required and calculate the iodine value
Calculation:
Iodine value = 1.269 x (b-a) / w
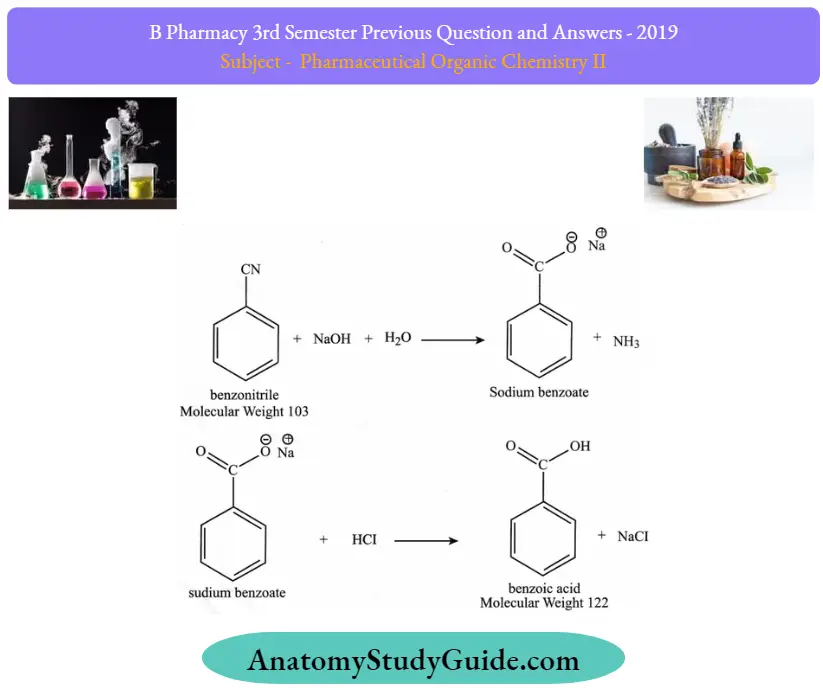
Question 14. Give the synthesis of benzoic acid
Answer:
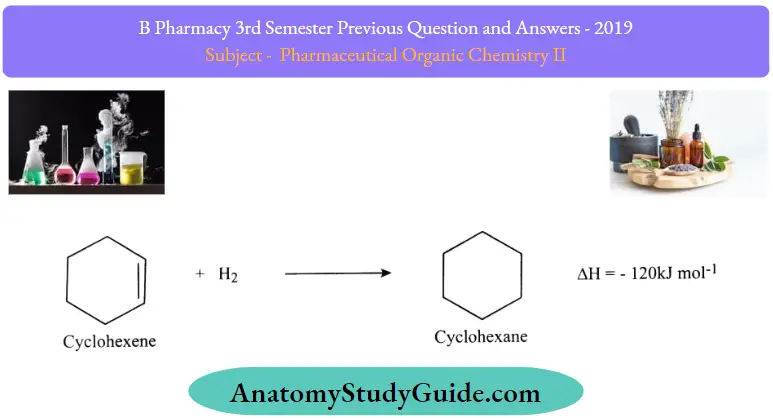
Question 15. Give additional reactions of cyclopropane.
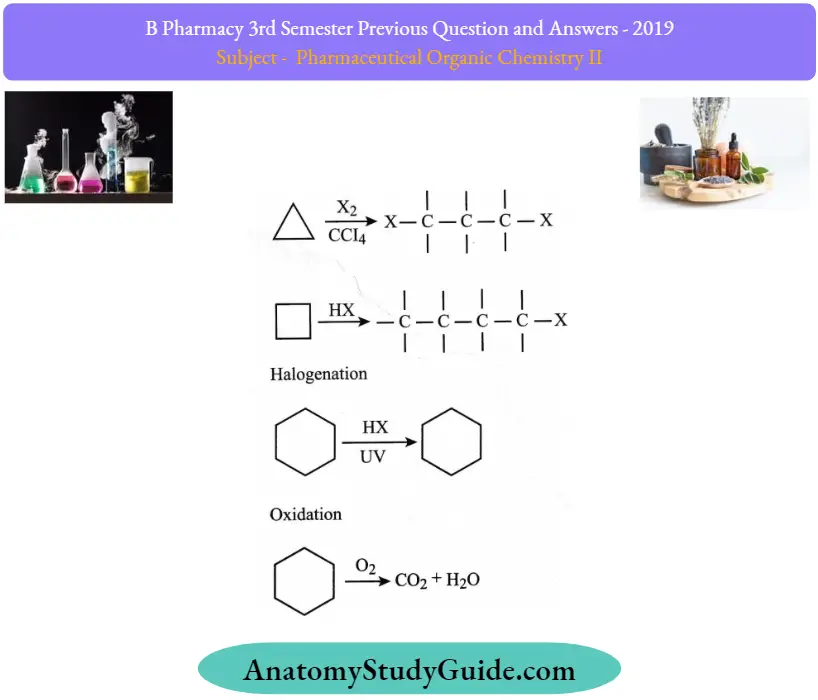
Answer:
1. Hydrogenation (ring-opening) :
Cycloalkanes undergo hydrogenation in the presence of catalysts like Ni or Pt to form the corresponding saturated hydrocarbons. The ease of hydrogenation decreases as the size of the ring increases. Higher Cycloalkanes having six or more carbon atoms are usually stable to hydrogenation.
Hydrogenation ofcyclohexene – one double bond
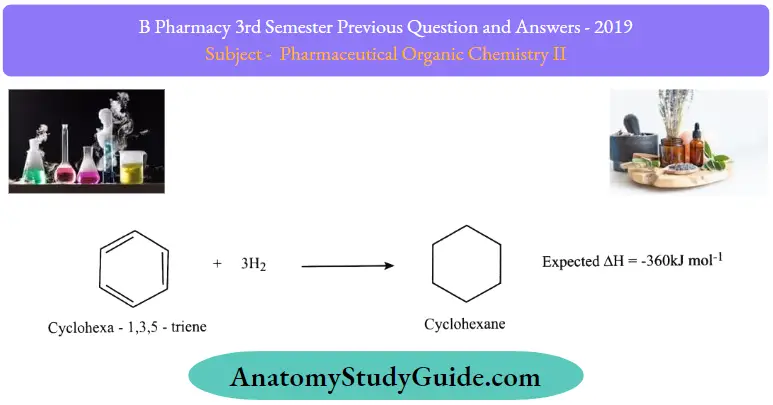
2. Hydrogenation:
Hydrogenation of Kekules benzene : Cyclohexa – 1,3,4, – triene – three double bonds
Halogenation:
Addition reaction of halogen (Leading to ring opening): Cyclopropane reacts with chlorine and bromine in the dark to form additional products.
Question 16. What is Sache-Mohr’s theory? Explain.
Answer:
Sache and Mohr proposed that seven rings can become free from strain if all the ring carbons are not forced into one plane, as meant by Baeyer. If a ring is assumed to have a folded condition then the normal tetrahedral angles of 109° 281 are retained and as a result, a strain within the ring is reduced.
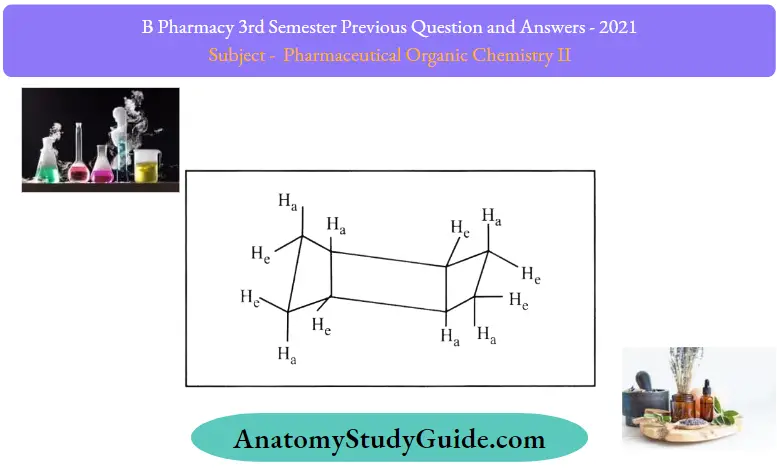
Cyclohexane exists in chair and boat form because of its shape:
Examination of the chair form of cyclohexane proves that the hydrogen atoms are divided into two categories. Six bonds of the hydrogen atom are found either straight up or down or almost perpendicular to the plane of the molecule. These are called axial hydrogen, and the other hydrogen lies slightly above or below the plane of the cyclohexane ring, and these are known as equatorial hydrogen.
Some of the different shapes of cyclohexane are:
- Chair form (more stable)
- Half chair form
- Twist boat
- Boat form (less stable)
Leave a Reply