Question 1. Differentiate between prokaryotes and eukaryotes.
Answer:
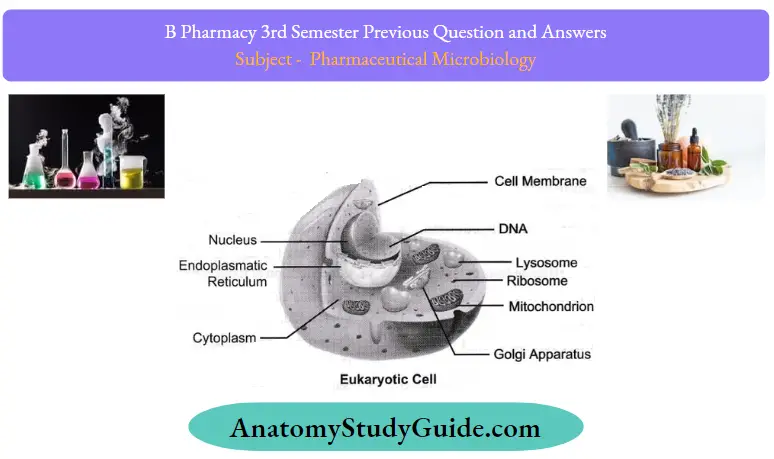
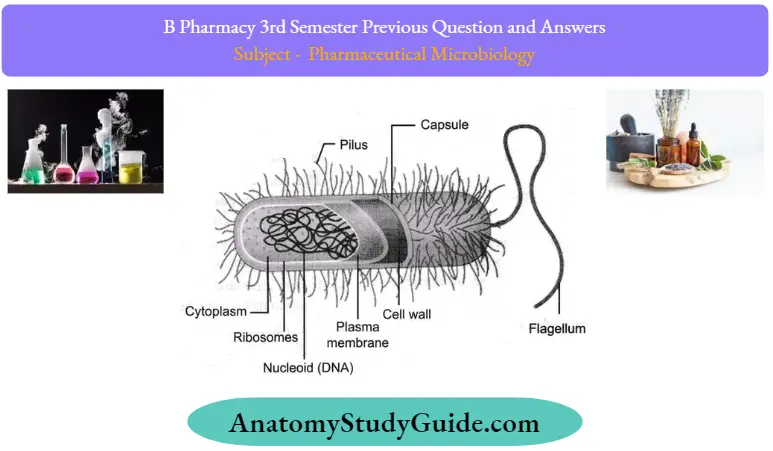
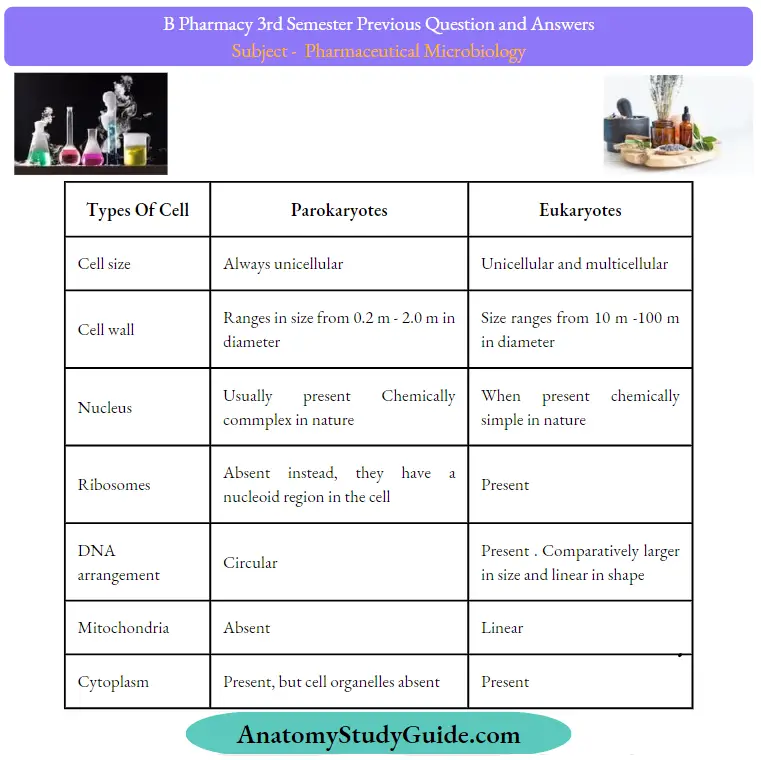
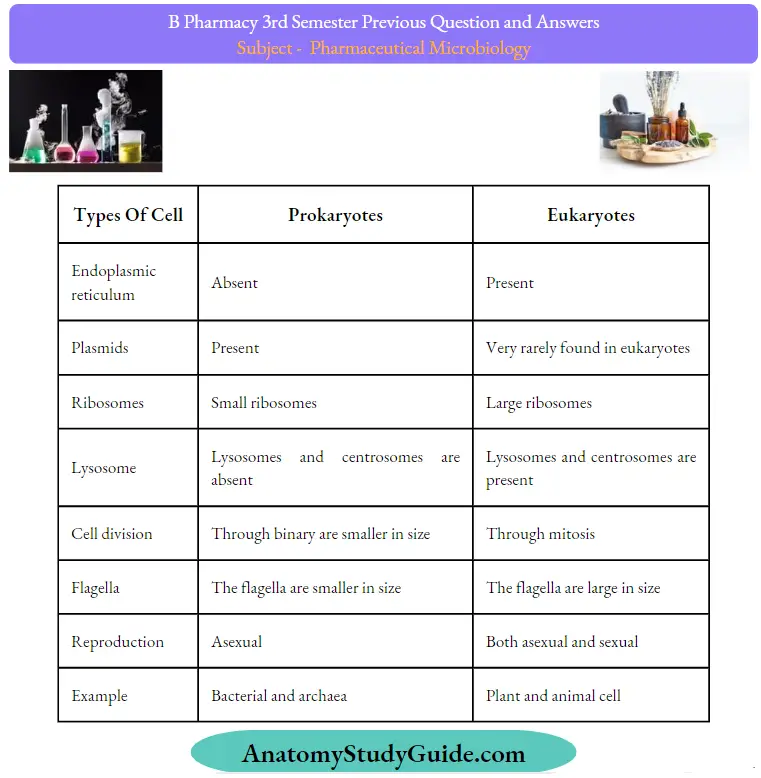
Types Of Cell:
Question 2. Explain the factors affecting sterilization.
Answer:
- Cleaning: Failure to adequately clean instruments results in higher bioburden, protein load, and salt concentration. These will decrease sterilization efficacy.
- Bioburden: The natural bioburden of used surgical devices is 100 to 103 organisms(primarily vegetative bacteria), which is substantially below the 105-106 spores used with biological indicators.
- Pathogen type: Spore-forming organisms are most resistant to sterilization and are the test organisms required for FDA clearance. However, the contaminating microflora used in surgical instruments consists mainly of vegetative bacteria.
- Protein: Residual protein decreases the efficacy of sterilization. However, cleaning appears to rapidly remove protein load.
- Salt: Residual salt decreases the efficacy of sterilization more than does protein load. However, cleaning appears to rapidly remove the salt load.
- Biofilm accumulation: Biofilm accumulation reduces the efficacy of sterilization by impairing exposure ofthe sterilant to the microbial cell.
- Lumen length: Increasing lumen length impairs sterilant penetration. May require forced flow through the lumen to achieve sterilization.
- Lumen diameter: Decreasing lumen diameter impairs sterilant penetration. May require forced flow through the lumen to achieve sterilization.
- Restricted flow: Sterilant mustcome into contact with microorganisms. Device designs that prevent or inhibit this contact, for example, sharp bends, blind lumens) will decrease sterilization efficacy.
- Device design and construction: Materials used in construction may affect compatibility with different sterilization processes and affect sterilization efficacy. Design issues (for example, screws, and hinges) will also affect sterilization efficacy.
Question 3. Write the significance of various reagents used in gram staining and acid-fast staining.
Answer:
Grams staining
- Application of the primary stain (crystal violet): Crystal violet stains all cells blue/purple
- Application of mordant: The iodine solution (mordant) is added to form a crystal violet-iodine(CVI) complex; all cells continue to appear blue.
- Decolorization step: The decolorization step distinguishes gram-positive from gram-negative cells.
- The organic solvent: Such as acetone or ethanol extracts the blue dye complex from the lipid-rich, thin-walled gram-negative bacteria to a greater degree than from the lipid-poor, thick-walled, gram-positive bacteria. The gram-negative bacteria appear colorless and gram-positive bacteria remain blue.
- Application of counterstain (safranin): The red dye safranin stains the decolorized gram-negative cells red/pink; the gram-positive bacteria remain blue.
Acid-fast staining
Carbol fuchsin, carbol-fuchsin, or carbolfuchsin, is a mixture of phenol and basic fuchsin, used in bacterial staining procedures. Carbol fuchsin is used as the primary stain dye to detect acid-fast bacteria because it is more soluble in the cell’s wall lipids than in the acid alcohol.
- Methylene blue: It functions as a counterstain and contains 3% of methylene blue. Methylene blue stains the decolorized cells of non-acid fast bacteria and make them appear blue. Unlike non-acid fast, an acid-fast bacteria will not take up the color of methylene blue and appear red.
- Acid alcohol: Acid alcohol is used as a decolorizing agent in the acid-fast staining procedure because acid binds to it and the stain sticks but it will decolorize nonacid-fast organisms. Ethyl alcohol will decolorize everything acid-fast and non-acid-fast
Question 4. Write about the classification of viruses.
Answer:
1. Based on Genetic Material Present:
Viruses are small, nonliving parasites, which cannot replicate outside of a host cell. A virus consists of genetic information – either DNA or RNA – coated by a protein. Accordingly, they are classified as DNA viruses and RNA viruses. The nucleic acid may be single or double-stranded, circular or linear, segmented or unsegmented.
- DNA viruses:
- As their name implies, DNA viruses use DNA as their genetic material.
- Some common examples of DNA viruses are parvovirus, papillomavirus, and herpesvirus.
- DNA viruses can affect both humans and animals and can range from causing benign symptoms to posing very serious health.
- RNA viruses:
- The virus that possesses RNA as genetic material are called RNA viruses.
- Rotavirus, poliovirus, yellow fever virus, dengue virus, hepatitis C virus, measles virus, rabies virus, influenza virus, and Ebola virus are examples of RNA viruses.
- DNA-RNA viruses:
- The RNA tumor viruses called Leukoviruses and
- Rous’s viruses unusually contain both DNA and RNA as genetic material.
2. based on the presence of several strands:
- Double-stranded DNA: It is found in pox viruses, the bacteriophages T2, T4, T6, T3, T7, and Lamda, herpes viruses, adenoviruses, etc.
- Single-stranded DNA: It is found in bacteriophages, X, and 74 bacteriophages.
- Double-stranded RNA: It has been found within viral capsids in the reoviruses of animals and the wound tumor virus and rice dwarf viruses of plants.
- Single-stranded RNA: It is found in most RNA viruses
- For example Tobacco mosaic viruses, influenza viruses, poliomyelitis, bacteriophage MS-2, and Avian leukemia virus.
3. Based on the Presence of Envelope:
The envelope is a lipid-containing membrane that surrounds some virus particles. It is acquired during viral maturation by a budding process through a cellular membrane Virus-encoded glycoproteins are exposed on the surface ofthe envelope. These projections are called peplomers.
- Enveloped Virus:
- DNA viruses: Herpesviruses, Poxviruses, Hepadnaviruses
- RNA viruses: Flavivirus, Toga virus, Coronavirus, Hepatitis D, Orthomyxovirus, Paramyxovirus, Rhabdovirus, Bunyavirus, Filovirus
- Retroviruses
- Non-Enveloped Virus:
- DNA viruses: Parvovirus, adenovirus and papovavirus.
- RNA viruses: Picomavirus, Hepatitis A virus, and Hepatitis E virus.
4. Virus Classification by Capsid Structure:
- Naked icosahedral: Hepatitis A virus, polioviruses
- Enveloped icosahedral: Epstein-Barr virus, herpes simplex virus, rubella virus, yellow fever virus, HIV-1
- Enveloped helical: Influenza viruses, mumps virus, measles virus, rabies virus
- Naked helical: Tobacco mosaic virus
Complex with many proteins: Some have combinations of icosahedral and helical capsid structures. Herpesviruses, smallpox virus, hepatitis B virus, T4 bacteriophage.
5. Based on Shapes of the Viruses:
Most ofthe animal vines are roughly spherical with some exceptions.
- Rabies virus: Bullet-shaped
- Ebola virus: Filamentous shaped
- Poxvirus: Brick shaped
- Adenovirus: Space vehicle shaped
6. Classification of Viruses based on Structure:
- Cubical viruses: They are also known as icosahedral symmetry vims
- For example: Reo vims, Picoma vims.
- Spiral viruses: They are also known as helical symmetry vims
- For example: Paramyxovirus, and orthomyxovirus.
- Radial symmetry virus:
- For example: Bacteriophage.
- Complex virus:
- For example Pox vims.
7. Based on the Type of Host:
The vims can be classified based on the type of host. They are:
- Animal Viruses: The vines that infect and live inside animal cells including man are called animal viruses.
- For example Influenza vims, rabies vims, mumps vims, poliovirus, etc. Their genetic material is RNA or DNA.
- Plant Viruses: The viruses that infect plants are called plant viruses. Their genetic material is RNA which remains enclosed in the protein coat. Some plant viruses are tobacco mosaic virus, potato virus, beet yellow virus turnip yellow virus, etc.
- Bacteriophages: Viruses that infect bacterial cells are known as bacteriophages or bacteria eaters. They contain DNA as genetic material. There are many varieties of bacteriophages. Usually, each kind of bacteriophage will attack only one species or only one strain of bacteria.
8. Classification of Viruses based on the Mode of Transmission:
- Virus transmitted through the respiratory route
- For example: Swine flu, Rhinovirus
- Virus transmitted through fecal-oral route:
- For example: HepatitisA virus, Poliovirus, Rotavirus
- Virus transmitted through sexual contacts:
- For example: Retrovirus
- Virus transmitted through blood transfusion:
- For example: the Hepatitis B virus, HIV
- Zoonotic virus: Vims transmitted through biting of infected animals
- For example Rabies vims, Alpha vims, Flavi vims
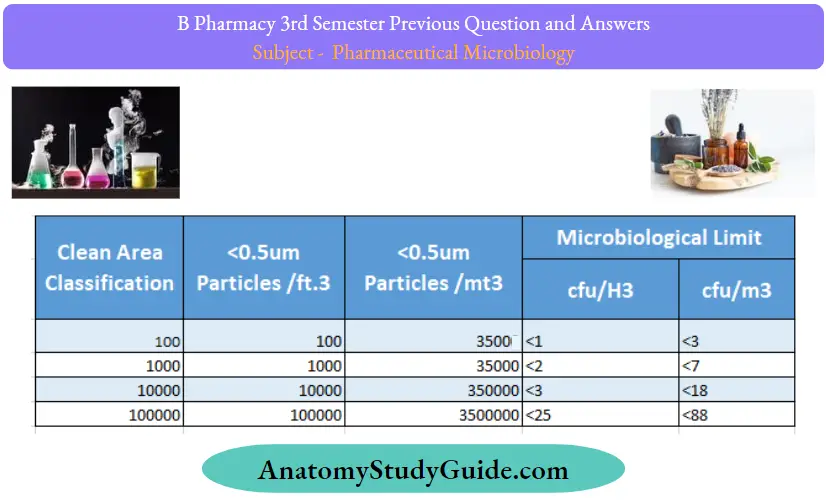
Question 5. Classify clean areas according to British standard and US standards.
Answer:
According to US FED STD 209E, the cleanroom classification is:
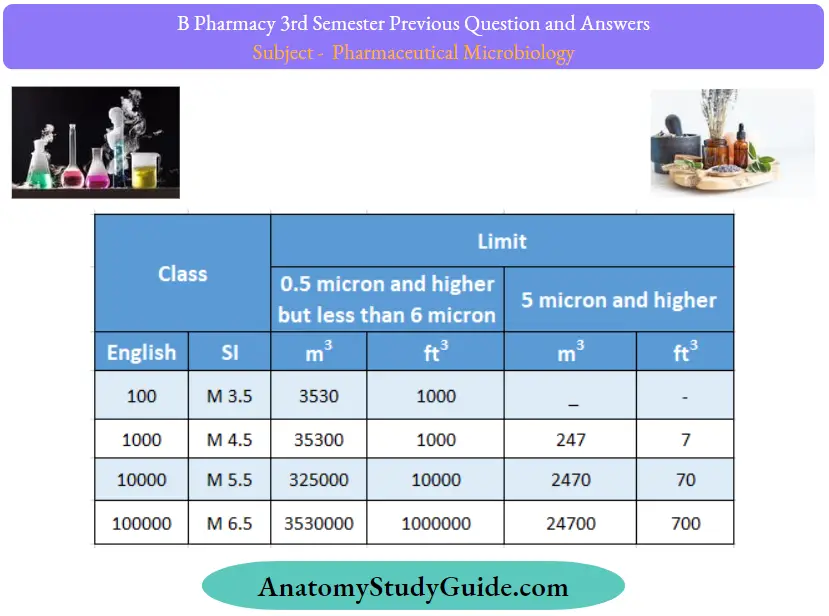
BS 5295 (British Standard):
The BSI offers the BS 5295 standard for specifying clean rooms and their classification. To classify clean rooms, they divided them into 10 categories.
Nevertheless, BS 5295 is superseded by ISO 14644 -1, as is FED STD 209E:
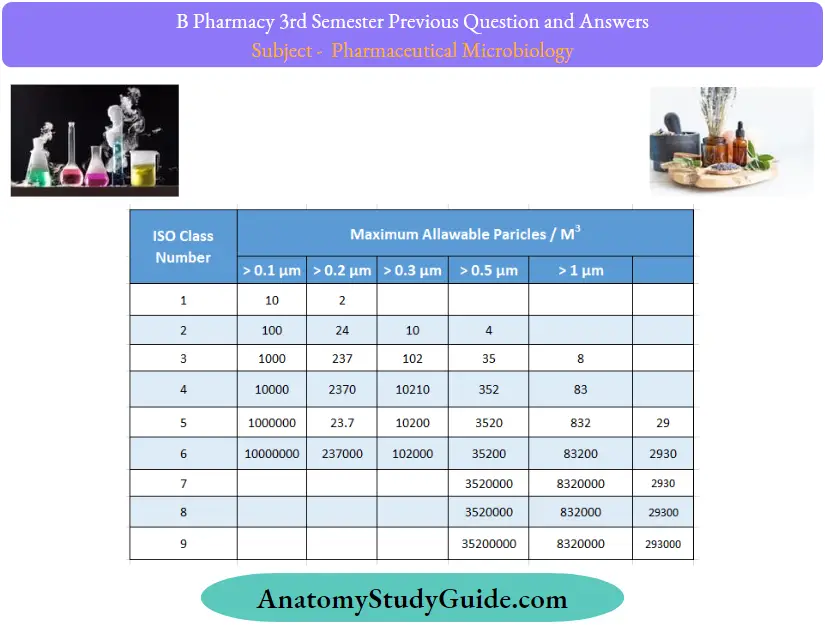
Clean Room Classification as per ISO 14644-1:2015:
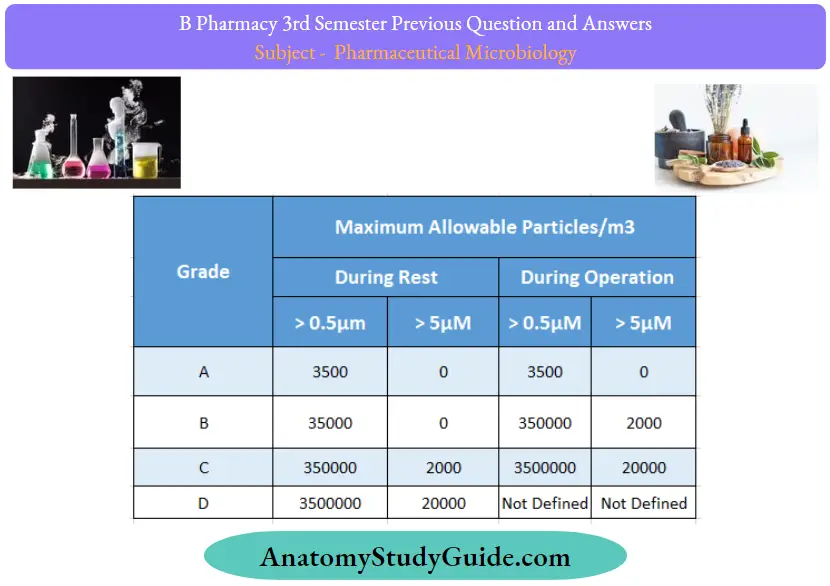
Cleanroom Classification as per the European Economic Commission
Question 6. Write briefly on different methods used for microbiological assay of antibiotics.
Answer:
The microbiological assay of an antibiotic is based upon a comparison ofthe inhibition of the growth of microorganisms by measured concentrations of the antibiotics under examination with that produced by the known concentration of a standard preparation ofthe antibiotic having a known activity.
Method 1: Cylinder Plate Method
Cylinder Plate Method Principle:
This method depends on the diffusion of an antibiotic from a vertical cavity through the solidified agar layer in the Petri plate. The growth of test microorganisms is inhibited entirely in a zone around the cylinder containing antibiotic solution.
Cylinder Plate Method Procedure:
- The nutrient agar is melted, cooled, and poured into a petri dish.
- 0.2 ml of known concentration of inoculum is spread on the surface of solidified agar by spread plate technique.
- Holes or cavities are made by a sterile borer.
- 0.2, 0.4, 0.6, 0.8, and 1 ml of antibiotic is poured into the cup of the agar plate and then incubated at 37°C for 24 hours.
- A zone of inhibition is observed for an antibiotic that has antimicrobial activity.
- The zone of inhibition is measured by scale from the backside ofthe plate from the center ofthe cavity or hole.
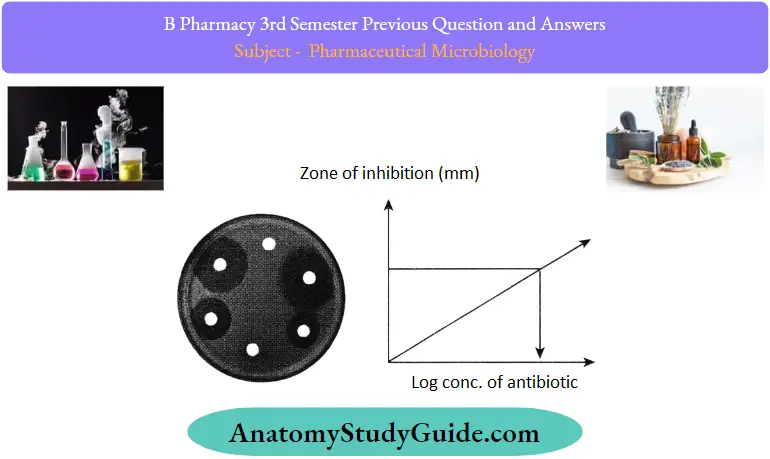
L = 3a+2b+c-e/5
H = 3e+2d+c-a/5
Where,
L = The calculated zone diameter for the lowest concentration ofthe standard curve response line.
H = The calculated zone diameter for the highest
concentration ofthe standard curve response line.
c = Average zone diameter of 36 readings of the reference point standard solution.
a, b, d, and e = Corrected average values for the other standard solutions, lowest to highest concentrations, respectively.
Two-Level Factorial Assay:
Parallel dilutions containing two levels of both the standard (SI, and S2) and unknown(U1 and U2) are prepared. Cavities of each plate are poured with diluted test samples along with standard. Finally, they are kept in the incubator, and the zone of inhibition is measured.
The potency is calculated by the following formula % potency = Antilog (2.0 + a log I)
Where,
a = (U1 + U2) – (S1 + S2) / (U1 + U2) + (S1 – S2)
U1 and U2 = The sums of the zone diameters with solutions ofthe unknown of high and low levels. SI and S2 =The sums of the zone diameters with solutions ofthe standard of high and low levels.
I = Ratio of dilutions.
If the potency of the sample is less than 60 percent or more than 150 percent ofthe standard, the assay is invalid and should be repeated using higher or lower dilutions of the same solution. The potency of the sample may be calculated from the expression.
Potency of sample = % Potency x Assemed potency of the sample/100
Method – 2 Tube Assay Method or Turbidity:
Tube Assay Method:
The method is based on the correlation between turbidity and changes in the microbial cell number. The turbidity is measured in a suitable liquid medium inoculated with microorganisms and finally, standard optical density or turbidimetric curves are used to estimate the number of microbial cells.
Tube Assay Principle:
This method depends on the inhibition of the growth of microbial culture in a uniform solution ofthe antibiotic in the fluid medium that is favorable to its rapid growth in the absence of antibiotics
Tube Assay Procedure:
- Five different concentrations ofthe standard solutions are prepared from the stock solution of standard as per step-wise increased ratio 4:5.
- The concentration of media is selected and the unknown substance solution is diluted to that concentration.
- 1 ml of each concentration ofthe standard solution as well as the test sample solution in each of the tubes is placed in duplicate.
- 9 ml of nutrient medium is added to each ofthe tubes.
- Side by side three control samples are prepared. One contained the inoculated culture medium, another is blank(0.5 ml of dilute formaldehyde solution) and the third one contained an uninoculated culture medium.
- All the tubes are placed in the incubator for 3-4 hours at 37°C and after incubation 0.5 ml of dilute formaldehyde is added in each tube.
- The growth of the test microorganism is measured by determining the absorbance at about 530 nm of each ofthe solutions in the tubes against the blank.

Calculation:
L = 3a+2b+c-e/5
H = 3e+2d+c-a/5
Where,
L = The calculated absorbance for the lowest concentration ofthe standard response line.
H = The calculated absorbance for the highest concentration of the standard response line.
a, b, c, d, and e = Average absorbance values for each concentration of the standard response line, lowest to highest concentrations, respectively.
Tube Assay Advantage:
The shorter incubation period(3-4 hours) for the growth ofthe test organism.
Tube Assay Disadvantage:
The presence of solvent residues and inhibitory substances affects more in this assay. Further, this method is not applicable for turbid or cloudy preparation.
Question 7. Discuss about advantages and disadvantages of animal cell culture
Answer:
Animal cell culture Advantages:
- Cell culture is superior to similar methods of biotechnology because it allows for the alteration of various physiological and physiobiological conditions such as pH, temperature, and Osmotic pressure.
- Animal cell culture is a great way to study issues that study cell metabolism and helps to understand cell biochemistry.
- It also permits observation ofthe effects of different chemicals like drugs and proteins on various cell types.
- The results of animal cell culture are consistent regardless ofthe type of cell that is utilized.
- This technique can also facilitate the identification of various cells on the basis in the appearance of markers, such as molecules, or through karyotyping.
- The usage of animal cells for testing and other purposes is a barrier to animal experiments.
- Animal cell cultures can be utilized to produce massive amounts of antibodies and proteins that otherwise would require an investment of a substantial amount
Animal cell culture Disadvantages:
- It is a highly specialized method that requires experienced personnel as well as the right conditions for aseptic treatment. It is a costly procedure because it requires expensive equipment.
- Subcultures of the cell line could result in different properties when different from that ofthe initial strain.
- The process produces a tiny amount of recombinant proteins which increases the cost of the procedure.
- Mycoplasma-related contamination and viral infections are common and are hard to identify and manage.
- The cells produced by this technique lead to instability due to the occurrence of aneuploidy chromosomal constitution.
Question 8. Write the contributions of Alexander Fleming.
Answer:
Alexander Fleming was a Scottish physician and microbiologist.
- He was well known for his discoveries of the antibiotic substance benzylpenicillin (Penicillin G) from the mold Penicillium Rubens in 1928.
- He discovered an antibacterial substance called “penicillin” from the Penicillium notatum
- In September 1928, during his research on
- Staphylococcus aureus, he left open a petri plate of Staphylococcus at his window. When he came back to his laboratory from a vacation with his family, he noticed that the plate in which he had left open had become contaminated with mold, which was later identified as Penicillium notatum.
- He noticed that all the bacterial cells near the molds had died. He isolated this mold from this plate and identified it as Penicillium genus.
- He also noticed that this was effective against all Gram-positive pathogens. In the beginning, he named it “mold juice
Question 9. List out the different phases of growth of bacteria.
Answer:
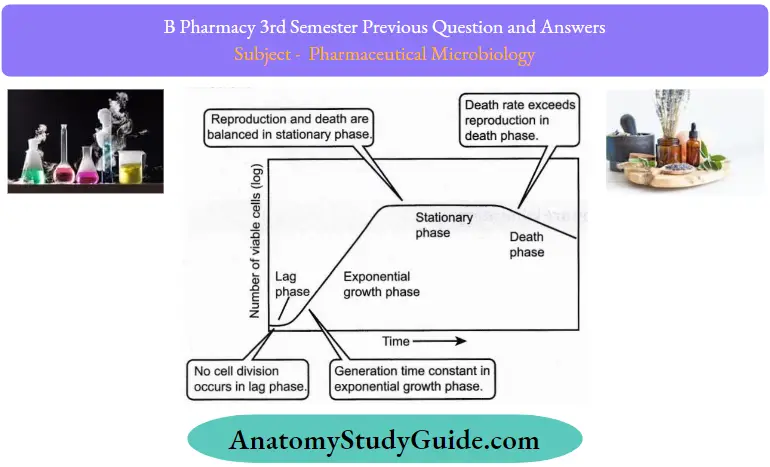
Question 10. Define sterilization.
Answer:
Sterilization is a process of destroying all germs including persistent microorganisms and bacteria. The whole process is difficult to achieve because chemicals are not always able to kill unwanted or hazardous leftovers.
Sterilization Methods:
- Sterile filtration.
- Disinfection.
- Heat radiation.
- Chemical method.
- Antiseptics.
- Cleaning
Question 11. Write the structure typical virion
Answer:
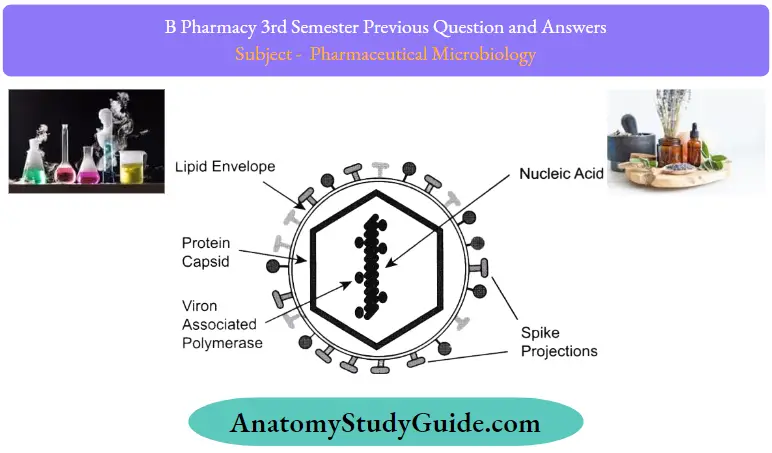
Question 12. Give two examples of alcoholic disinfectants. Mention its mechanism of action.
Answer:
Examples: Ethylalcohol, isopropylalcohol.
Mechanism of action:
They Damage cell membranes, denaturing essential microbial proteins interfering with metabolism, and resulting in cell lysis. Mixtures of alcohol and water are
Question 13. Write the principle of sterility testing.
Answer:
Sterility testing Principle:
The basic principle of the sterility test is that if bacteria or fungi are placed in a medium that provides the nutritive material and water and kept at a favorable temperature the organism will grow and their presence can be indicated by the turbidity in the clear medium.
Question 14. Outline interpretation sterility testing results.
Answer:
Interpretation of results:
Challenge organisms should clearly show visible growth of bacteria within 3 days, and fungi within 5 days in the test media containing products. Visually comparable to that in the control vessel without product.
Question 15. Draw a flow diagram of an aseptic area.
Answer:
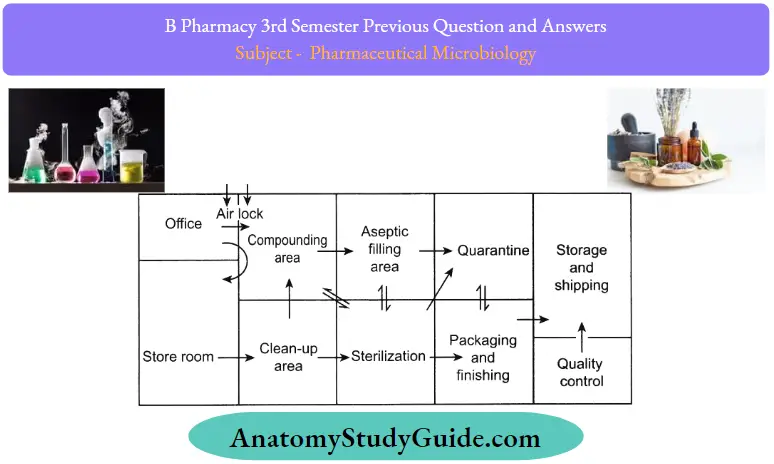
Question 16. What are the visible changes of microbial contamination in pharmaceuticals?
Answer:
- Viable growth
- Coloration
- Physical spoilage
Question 17. Mention any four major requirements for a cell culture laboratory.
Answer:
- Requirements are:
- Clean and sterile area.
- Preparation facilities.
- Animal house.
- Storage facilities
Question 18. Write the difference between Prokaryotes and eukaryotes
Answer:
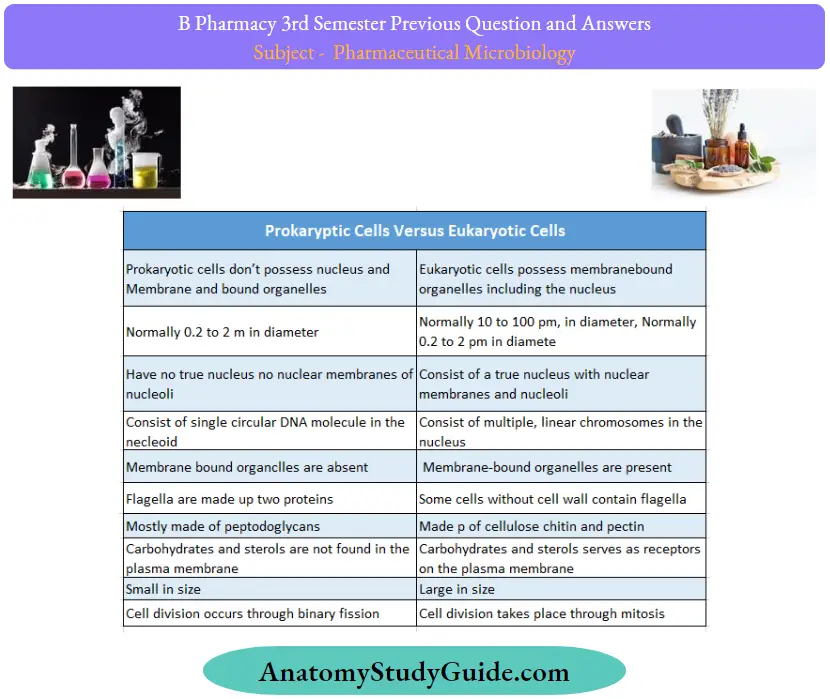
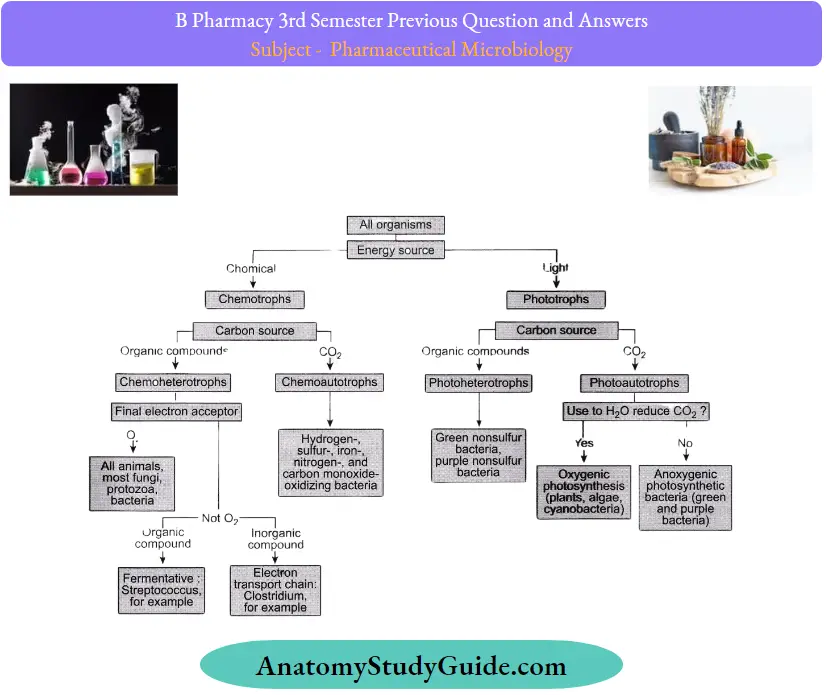
Question 19. Classify bacteria based on the nutritional requirements
Answer:
Question 20. What are the factors affecting the microbial spoilage of pharmacy?
Answer:
There are many factors affecting microbial spoilage of pharmaceutical products.
These include:
- Nutritional Factors
- Water
- Storage Temperature
- pH
- Package Design
These are:
- Nutritional Factors: Microorganisms utilize many formulation components as substrates for biosynthesis& growth.
- Water: It is the most important cause of the survival and growth of microorganisms.
- Storage Temperature: Spoilage of pharmaceuticals could occur potentially over the range of about 20°C to 60°C, although it is much less likely at the extremes.
- pH: Micro-organisms grow at neutral pH, therefore acidic or alkaline formulations are less susceptible to spoilage.
- Packaging Design: It can have a major influence on the microbial stability of some formulations in controlling the access of contaminants during both storage and use.
Other factors affecting microbial spoilage of pharmaceutical products include:
- Relative Humidity
- Oxygen Availability
- Osmotic Pressure
- Oxidation-Reduction Balance
- Surface Tension
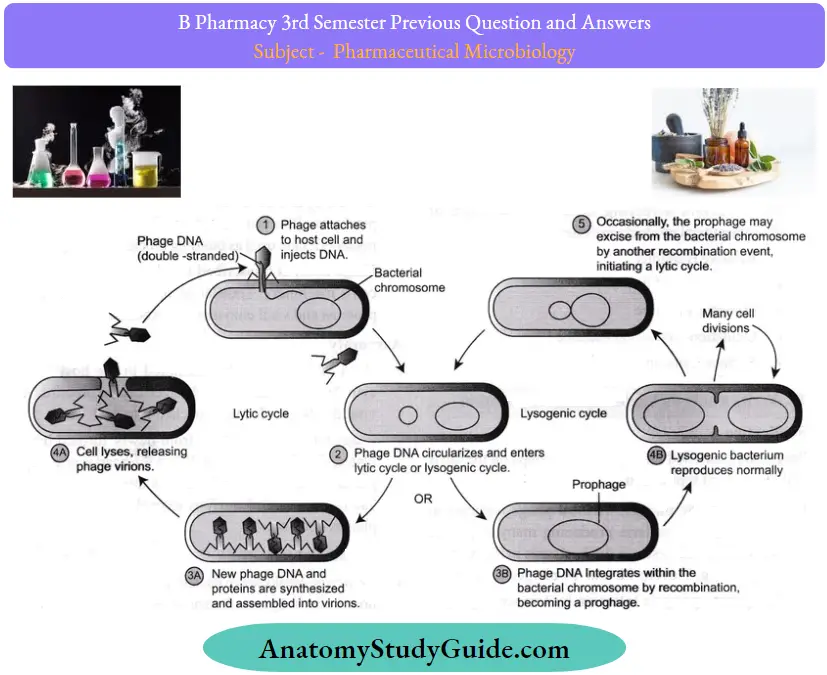
Question 21. Write a note on bacteriophage replication.
Answer:
Replication
Phages are classified into two major groups based on their mode of replication.
- Virulent (lytic phage): Virulent phages replicate in the susceptible bacteria producing many copies of themselves and destroying the host cells in the process by lysis,
- For example T-even; T2 and T4 phages of E. coli.
- Temperate phages: Infection by temperate phages can have either of two outcomes.
- Lytic growth or
- Lysogeny(non-lytic prophage state)
Replication of Lytic Phages: Adsorption/Attachment
Bacteriophage adsorption is the first step to start the infection process. The binding proteins of the bacteriophage mostly located the tail fibers interact and recognize specific receptors present on the bacterial cell wall.
The attachment of phage to its host cell results in morphological changes to both phage and bacterium facilitating the penetration of the phage into a host bacterium.
Penetration:
- Lysozyme-like enzyme found in the phage tails weakens the bacterial cell wall. The tail sheath contracts, hollow tube(core) penetrates the weakened cell wall and comes in contact with the cell membrane.
- Viral DNA moves from the head via the tube to the bacterial cytoplasm while the phage capsid remains outside.
Replication:
- Phage genes take control ofthe host cell’s metabolic machinery and direct host cells to produce only viral products. Bacterial DNA is disrupted and their nucleotides are used as building blocks for new phage.
- Phage DNA is transcribed to mRNA using the host cell’s machinery. Translation of mRNA and capsid proteins and viral enzymes are produced.
Assembly:
- The head of T4 Phages is assembled in the host cells’ cytoplasm from newly synthesized capsid proteins. A viral dsDNA molecule is packed into each head.
- Phage tails are assembled from newly formed base plates, sheaths, and collars. When the head is properly packed with DNA, each head is attached to a tail.
- Tail fibers are added and fully formed mature, infective phages develop.
Lysis and Release:
Lysozyme breaks down bacterial cell walls and bacterial host cells is lysed. Released phage infects other susceptible bacteria and the new infection process is started.
- Burst time: Time from adsorption to cell lysis, generally 20-40 minutes.
- Burst size (viral yield): Number of new virions released from each bacterial host cell. In T4 phage burst size is 50-200
Replication of Temperate Phages
- Temperate phages mostly exhibit lysogeny but can replicate by lytic pathways after induction. The best characterized temperate phage is the E. coli phage e.
- Lysogeny is a special type of latent viral infection. It’s a stable long-term relationship between the phages and its host in which the phage nucleic acid becomes incorporated into the host chromosome. The phage genome replicates as a prophage in the bacterial cell.
- In most lysogenic bacteria the genes required for lytic phage development are repressed and the production of infectious phage does not occur.
Process of Lysogeny:
- Lambda phages attach to bacterial cells and insert their linear DNA into the bacterial cytoplasm.
- PhageDNA circularizes and then integrates into the circular bacterial chromosome at a specific location. Insertion of lambda phage into a bacterium alters the genetic characteristics ofthe bacterium.
- Once established as a prophage, the virus can remain dormant for a long time. Each time a bacterium divides; the prophage is copied as a part of the bacterial chromosome and retained in the progeny bacteria. The period of bacterial growth with a prophage represents a lysogenic cycle.
- However, either spontaneously or in response to some outside stimulation, the prophage can become active and initiate a typical lytic cycle. This process is called induction.
Question 22. Mention the ideal characteristics of disinfectants.
Answer:
- Ideally, the disinfectant should have a wide spectrum of antimicrobial activity. It must be effective against a wide variety of infectious agents(Gram-positive and Gram-negative bacteria, acid-fast bacteria, bacterial endospores, fungi, and viruses) at high dilutions.
- It should act in the presence of organic matter
- It should not be toxic to humans or corrosive. In practice, this balance between effectiveness and low toxicity for animals is hard to achieve. Some chemicals are used despite their low effectiveness because they are relatively nontoxic.
- It should be stable upon storage and should not undergo any chemical change.
- It should be odorless or with a pleasant odor.
- It should be soluble in water and lipids for penetration into microorganisms.
- It should be effective in acidic as well as in alkaline media.
- It should have speedy action.
- If possible, it should be relatively inexpensive
Question 23. Enlist the types of staining. What is negative staining?
Answer:
Types of staining are:
- Simple staining
- Differential staining
- Gram staining
- Acid-fats staining
- Endospore staining
Negative staining
Negative staining is also referred to as “Indirect staining”. This technique helps to visualize various microorganisms by using both light and electron microscopy.
In bright field microscopy, the method of indirect staining involves the use ofthe liquid medium(black colored dyes) like Nigrosin and India ink that stains the background, leaving the bacteria unstained.
Negative staining Principle:
- Negative staining requires the use of an acidic stain such as India ink or nigrosin.
- The acidic stain, with its negatively charged chromogen, will not penetrate the cells because of the negative charge on the surface of bacteria.
- Therefore, the unstained cells are easily discernible against the colored background.
- The practical application of negative staining is twofold.
- First, since heat fixation is not required and the cells are not subjected to the distorting effects of chemicals and heat, their natural size and shape can be seen.
- Second, it is possible to observe bacteria that are difficult to stain, such as some spirilla.
- Because heat fixation is not done during the staining process, keep in mind that the organisms are not killed and slides should be handled with care
Negative staining Procedure:
- Place a small drop of nigrosin close to one end ofa clean slide.
- Using an aseptic technique, place a loopful of inoculum from the bacterial culture in the drop nigrosine and mix.
- Place a slide against the drop of suspended organisms at a 45° angle and allow the drop to spread along the edge ofthe applied slide.
- Push the slide away from the drop of suspended organisms to form a thin smear. Air-dry.
- Note: Do not heat-fix the slide.
- Examine the slides under oil immersion.
Question 24. Explain the cultivation of anaerobic bacteria.
Answer:
Bacterial Cultivation of anaerobic Bacteria
Main Principle:
Reduce the O2 content of the culture medium and remove any oxygen already present inside the system or in the medium.
- Oxygen is ubiquitous in the air so special methods are needed to culture anaerobic microorganisms.
- Several procedures are available for reducing the O2 content of cultures; some are simple but suitable mainly for less sensitive organisms, and others are more complex but necessary for the growth of strict anaerobes.
- Bottles or tubes filled to the top with culture medium and provided with tightly fitting stopper. Suitable for organisms not too sensitive to small amounts of oxygen.
- Addition of a reducing agent that reacts with oxygen and reduces it to water
For example: Thioglycolate in thioglycolate broth. After thioglycolate reacts with oxygen throughout the tube, oxygen can penetrate only near the top of the tube where the medium contacts air.
- Obligate aerobes grow only at the top of such tubes.
- Facultative organisms grow throughout the tube but are best near the top
- Microaerophiles grow near the top but not right at the
- Anaerobes grow only near the bottom of the tube, where oxygen cannot penetrate.
- A redox indicator dye called resazurin is added to the medium because the dye changes color in the presence of oxygen and thereby indicates the degree of penetration of oxygen into the medium.
- Strict anaerobes, such as methanogenic bacteria can be killed by even a brief exposure to O2. In these cases, a culture medium is first boiled to render it oxygen-free, and then a reducing agent such as H2S is added and the mixture is sealed under an oxygen-free gas
- All manipulations are carried out under a tiny jet of oxygen-free hydrogen or nitrogen gas that is directed into the culture vessel when it is open, thus driving out any O2 that might enter.
- For extensive research on anaerobes, special boxes fitted with gloves called anaerobic glove boxes, permit work with open cultures in completely anoxic atmospheres.
- Stringent anaerobes can be grown only by taking special precautions to exclude all atmospheric oxygen from the medium.
Such an environment can be established by using one ofthe following methods:
1. Pre-reduced media:
- During preparation, the culture medium is boiled for several minutes to drive off most of the dissolved oxygen. A reducing agent
- For Example, Cysteine is added to further lower the oxygen content. Oxygen-free N2 is bubbled through the medium to keep it anaerobic.
- The medium is then dispensed into tubes which are flushed with oxygen-free nitrogen, stoppered tightly, and sterilized by autoclaving.
- Such tubes are continuously flushed with oxygen-free CO2 using a cannula, restoppered, and incubated.
2. Anaerobic Chamber:
- This refers to a plastic anaerobic glove box that contains an atmosphere of H2, CO2, and N2. Culture
- All manipulations are carried out under a tiny jet of oxygen-free hydrogen or nitrogen gas that is directed into the culture vessel when it is open, thus driving out any 02 that might enter.
- For extensive research on anaerobes, special boxes fitted with gloves called anaerobic glove boxes, permit work with open cultures in completely anoxic atmospheres.
Question 25. What are the nutritional requirements for animal cell culture?
Answer:
The culture of animal cells and tissue is rather more difficult than that of microorganisms and plants because the latter synthesize certain chemical constituents(unlike microbes).
There are two types of media used for the culture of animal cells and tissue:
- The natural media and
- Artificial media.
1. Natural Media:
Natural media are the natural sources of nutrients sufficient for the growth and proliferation of animal cells and tissue.
There are three types:
- Plasma Clots: The most commonly used clots are plasma clots which have been in use for a long time. Plasma is now commercially available either in liquid or lyophilized state. It may also be prepared in the laboratory usually from the blood of male fowl.
- Biological Fluids: The various biological fluids used as a culture medium
- For example: Amniotic fluid, pleural fluid, aqueous humor from the eye, insect hemolymph, etc). Serum is the most widely used. Serum may be obtained from adult human blood, placental cord blood, horse blood, or calf blood of these foetal calf serum is commonly used.
- Tissue Extracts: Chick embryo extract is most commonly used but bovine embryo extract is also used. Others are the spleen, liver, bone marrow, leucocytes, etc. Tissue extracts can often be substituted by a mixture of amino acids and certain other organic compounds. For cell cultures, artificial media with or without serum are used.
2. Artificial Media:
Synthetic media are prepared artificially by adding several nutrients (organic and inorganic), vitamins, salts, O2, and CO2 gas phases, serum proteins carbohydrates, cofactors, etc. However, different types of synthetic media may be prepared for a variety of cells and tissues to be cultured. It can be prepared for different functions.
Synthetic media are of two types:
- Serum containing media and
- Serum-free media.
Serum itself has several disadvantages:
- It deteriorates within a year and differs with batches.
- Several batches are required if more than one
- cell types are used, which makes it difficult to maintain and co-culturing cells.
- Undesirable growth stimulation and inhibition may occur.
- It is not chemically defined and therefore, it is of variable composition.
- It may be a source of contamination by viruses, mycoplasma, prions, etc.
- It is the most expensive ingredient ofthe culture media.
Question 26. However, it has some advantages such as:
Answer:
- Serum contains a complete set of essential growth factors, hormones, attachment and spreading factors, binding and transport proteins.
- It binds and neutralizes toxins.
- It contains protease inhibitors.
- It increases buffering capacity.
- It provides trace elements and other nutrients
Question 27. What is enriched media?
Answer:
The addition of extra nutrients in the form of blood, serum, egg yolk, etc, to the basal medium makes an enriched medium. Enriched media are used to grow nutritionally exacting (fastidious) bacteria. Blood agar, chocolate agar, Loeffler’s serum slope, etc are a few examples of enriched media. Blood agar is prepared by adding 5-10% (by volume) blood to a blood agar base. Chocolate agar is also known as heated blood agar or lysed blood agar.
Question 28. What is normal flora?
Answer:
The normal flora of the human body refers to the microbial community that colonizes the skin and mucus membrane. Normal flora is sometimes called indigenous microbiota, as they reside within or on the human’s skin surface. Diverse microflora such as bacteria, fungi, and some protists are associated with or within the skin surface.
Question 29. Give the applications of animal cell culture in pharmacy.
Answer:
The main applications are:
- Model systems
- Toxicity testing
- Cancer research
- Virology
- Cell-based manufacturing
- Genetic counseling
- Genetic engineering
- Gene therapy
- Drug screening and development
Question 30. Classify clean area
Answer:
Question 31. What do you mean by primary culture?
Answer:
This is the cell culture obtained straight from the cells of a host tissue. The cells dissociated from the parental tissue are grown on a suitable container and the culture thus obtained is called primary cell culture. Such culture comprises mostly heterogeneous cells and most ofthe cells divide only for a limited time. However, these cells are very similar to their parents.
Question 32. What are sterilization indicators?
Answer:
Sterilization indicators are used to allow differentiation between processed and unprocessed items, and/or indicate gross failure ofa sterilization process. They are also biological indicators.
Question 33. What is thermal death time?
Answer:
Thermal death time is how long it takes to kill a specific bacterium at a specific temperature. It was originally developed for food canning and has found applications in cosmetics, producing salmonella-free feeds for animals (poultry), and pharmaceuticals.
Question 34. Differentiate between bacteria and fungi.
Answer:

Question 35. What is synchronous growth?
Answer:
Synchronous growth of a bacterial population is that during which all bacterial cells ofthe population are physiologically identical and in the same stage of cell division cycle at a given time. Synchronous growth of a bacterial population is that during which all bacterial cells ofthe population are physiologically identical and in the same stage of cell division cycle at a given time. Synchronous growth helps in studying particular stages or the cell division cycle interrelations μ
Question 36. Explain the principle and procedure involved in the MR-VP test.
Answer:
Methyl Red(MR) and Voges-Prauskauer(VP) tests are a part ofIMViC reactions, which are used in the identification of certain fermentative bacteria
For example: Enterobacteriaceae
- These tests are performed together because organisms are generally positive for one of them. Rarely, some organisms are positive for both tests.
- These tests are based on the fact that bacteria can ferment glucose into mixed acids or butylene glycol.
Methyl Red test (MR)
Methyl Red test (MR) Principle:
When grown in a glucose-containing medium, some bacteria can produce large amounts of mixed acids such as acetic acid, formic acids, or succinic acid from glucose fermentation.
- The amount of acids produced overcomes the buffering activity ofthe phosphate buffer included in the glucose phosphate broth thereby rendering the pH ofthe medium acidic. This acidity is tested by using
- Methyl Red is a pH indicator.
- MRreagent is an alcoholic solution of Methyl Red dye (also known as Cl Acid Red 2), which remains red at a pH of 4.4 or below.
MR test Procedure:
A tube containing(1%) glucose phosphate broth is inoculated with fresh culture ofthe test organism and incubated at 37oC for 24-48 hours. A volume of 2.5 ml of this suspension is transferred into a sterile tube and a few drops of MR reagent are added.
The culture suspension turns red instantly and remains stable if the test is positive or turns yellow if it is negative. Controls: Escherichia coli ATCC 25922 is positive control and Enterobacter cloacaeATCC 23355 is negative control
Voges Proskauer(VR) test
Voges Proskauer test Principle:
- Some fermenting bacteria undertake the butylene glycol pathway in the fermentation of glucose. These organisms do produce some organic acids but the chief end product of glucose fermentation is 2-3 butylene glycol(2-3 butanediol), a neutral product.
- However, the VP test detects an intermediate product, the acetyl methyl carbinol(acetoin).
- Upon the addition of KOH, acetoin is oxidized to diacetyl, which then reacts with the guanidine group of arginine (contained in the peptone) to give rise to a red-coloured product.
Voges Proskauer test Procedure:
1. Original VP test:
- The test organism is inoculated into glucose phosphate (MR-VP) broth and incubated at 30oC for five days (at least 48 hours).
- 2 ml ofthe suspension is transferred to another tube and a few drops of 10% KOH are added to it, shaken, and held at room temperature overnight.
- Appearance of red colour indicates a positive reaction. Several modifications to the VP test were made to hasten the process of oxidation of acetoin; these included the use of hydrogen peroxide, barium peroxide, potassium dichromate, sodium peroxide, ferric chloride, creatin etc.
2. Barritt’s method:
- The test organism is inoculated into glucose phosphate (MR-VP) broth and incubated at 37oC for 48 hours. 2.5 ml of this suspension is transferred to another tube, 0.6 ml of alcoholic solution of a-naphthol is added and shaken.
- To this, 0.2 ml of 40% KOH is added, the tube is shaken and left to stand at room temperature for 15 minutes.
- The appearance of red color indicates a positive test. If negative, the tube must be held for another 45 minutes. In this test, a-naphthol serves as a catalyst and a colour intensifies
- Since the use of a-naphthol was recommended by Maxwell Barritt, this method is also known as Barritt’s method.
3. Meara’s method:
- The test organism is inoculated into glucose phosphate(MR-VP) broth and incubated at 37°C for 48 hours. 2.5 ml of this suspension is transferred to another tube and 0.6 ml of O’Meara’s reagent (40% KOH + 0.3% creatine) is added.
- The tube is placed in a water bath at 37°C for 4 hours with intermittent shaking for aeration. Appearance of red color is a positive reaction. O’Meara’s reagent has been reported to be unstable.
4. Quicker VP tests:
The results of VP test can be obtained earlier by heavily inoculating a 0.5-1 ml volume of glucose phosphate broth and incubating at 37°C for 18-24 hours.
5. Barry’s rapid direct VP test:
- This test is performed on lactose-fermenting colonies (on MacConkey’s agar or TSI agar slant).
- These colonies contain detectable amounts of acetoin from the fermentation of sugars.
- Two drops of 0.5% creatine solution is taken in a tube and colonies are emulsified densely into it. To this, three drops of 5% a-naphthol(in 95% ethyl alcohol) is added and shaken.
- Next, two drops of 40% KOH are added and shaken. The appearance of pink-red color in 15 minutes is a positive reaction
6. Barry’s rapid indirect VP test:
- 0.2 ml of MR-VP broth is inoculated with a single colony ofthe test organism and incubated at 37°C for 4-6 hours.
- To this, two drops of 0.5% creatine solution is added and shaken.
- This is followed by the addition of three drops of a-naphthol and two drops of 40% KOH and shaken. The appearance of pink-red colour in 15 minutes is a positive reaction.
- Controls: Enterobacter cloacae ATCC 23355 is positive control and Escherichia coli ATCC 25922 is negative control
Question 37. Discuss sterilization by radiation.
Answer:
- Radiation Sterilization is a cold process that uses gamma radiation for sterilization of Healthcare Products.
- Controlled gamma energy which is released by radioisotopes such as
- Cobalt-60 is used for sterilization. Cobalt-60 is the most preferred radioisotope as it is readily available from a single nuclear reaction in a reactor and also cost-effective.
- Gamma radiation is characterized by deep penetration and kills microorganisms by destroying DNA structures.
- The process is suitable for Industrial scale sterilization. The radiation dose of 25 kGy (2.5 Mrad) is officially accepted for sterilization of healthcare products.
- Delivery of dose to the products is measured by dosimeter. Radiation sterilized products are acceptable by the Food& Drug Administration(FDA).
Advantages and Benefits of Radiation Sterilization
- Products of any shape can be sterilized: A product of any form and shape can be sterilized as the gamma rays are powerful enough to penetrate a high-density package and the product itself.
- Heat-sensitive materials can be sterilized: As it is a cold process with no known sources of heat, more sensitive products such as medical devices and pharmaceutical products can be safely sterilized.
- Flexibility in packaging: Radiation sterilization causes no long-term physical changes to the products as they can be packed individually in sealed bags and sterilized in the fully packaged form.
- Retention of product sterility: Since sterilization is affected after final packaging, product sterility is retained indefinitely provided the package is undamaged.
- Pre-packaged products: Radiation Sterilization enlarges the market for ready-to-use pre-packaged products. The process does not result in residual toxicity ofany form in the product.
- Safe to use: Products sterilized by this process do not become radioactive and are safe for use
High energy Electron Beam(EB) and X-ray machines are also used nowadays for this purpose.
The major difference In
- Gamma radiation and EB lie in their penetration powers, where gamma radiations can penetrate deep inside the products the electron beams do not have as good penetration power.
- Though X-rays have comparable penetration power to gamma radiation at a suitable energy range they are not yet very popular.
- The Board of Radiation and Isotope Technology (BRIT)/Department of Atomic Energy(DAE) had set up a radiation sterilization plant ‘ISOMED’ which has been in operation since 1974 for sterilization of medical products. Technology is effective, reliable and acceptable among users.
Question 38. Discuss the different methods of microbial assay of antibiotics.
Answer:
Microorganisms have found widespread uses in the performance of bioassays for
- Determining the concentration of certain compounds
- For example: Amino acids, vitamins, and some antibiotics) in complex chemical mixtures or in body fluids. Diagnosing certain diseases.
- Testing chemicals for potential mutagenicity or carcinogenicity.
- Monitoring purposes involving the use of immobilized enzymes.
- Sterility testing of antibiotics.
- Microbiological assays are used during production to determine potency and quality control. These are used to determine the pharmacokinetics of drugs in animals and humans.
- In antimicrobial chemotherapy to monitor, in managing, controlling the chemotherapeutic agents.
Methods of microbiological assay of antibiotics:
The following methods are used for microbiological assays
1. Disc diffusion method (cylindrical cup plate method):
- Use petri dishes or rectangular trays filled to a depth of 3-4 mm with a culture medium that has previously been inoculated with a suitable inoculum ofa susceptible test organism prepared as described below.
- The nutrient agar may be composed of two separate layers of which only the upper one may be inoculated.
- The concentration of the inoculum should be so selected that the sharpest zones of inhibition and suitable dose-response at different concentrations of the standard are obtained.
- When using the inoculum, an inoculated medium containing 1ml of inoculum per 100ml of the culture medium is usually suitable.
- When the inoculum consists of a suspension of vegetative organisms, the temperature of the molten agar medium must not exceed 48-50°C at the time of inoculation.
- The dishes or trays should be specially selected with flat bottoms.
- During the filling, they should be placed on a flat, horizontal surface so as to ensure that the layer ofthe medium will be of uniform thickness.
- With some test organisms, the procedure may be improved if the inoculated plates are allowed to dry for 30 minutes at room temperature before use, or refrigerated at 4°C for several hours.
- For the application ofthe test solution, small sterile cylinders of uniform size, approximately 10mm high and having an internal diameter of approximately, made of suitable material such as glass, porcelain, or stainless steel, are placed on the surface ofthe inoculated medium.
Instead of cylinders, holes 8-10mm in diameter may be bored in medium with a previously sterilized borer.
- Other methods of application ofthe test solution may also be used.
- The arrangement on the plate should be such that overlapping of zones is avoided.
- Solutions of the reference material of known concentration and corresponding dilutions ofthe test substance, presumed to be of approximately the same concentration, are prepared in a sterile buffer ofa suitable pH value.
- To assess the validity of the assay at least 3 different doses of the reference material should be used together with an equal number of doses of the test substance which has the same presumed activity as the solutions ofthe reference material.
The dose levels used should be in geometric progression, for example, by preparing a series of dilutions in the ratio 2:1.
- Once the relationship between the logarithm of the concentration of the antibiotic and the diameter of the zone of inhibition is approximately rectilinear for the system used, routine assays may be carried out using only 2 concentrations of the reference material and 2 dilutions ofthe test substance.
- Where a monograph gives directions for the initial preparation of a solution of the substance, this solution is then diluted as necessary with the appropriate sterile buffer.
- The solutions of the reference material and the test substance are preferably arranged in the form of a Latin square when rectangular trays are employed.
When Petri dishes are used, the solutions are arranged on each dish so that the solutions ofthe reference material and those ofthe test substance alternate around the dish and are placed in such a manner that the highest concentrations of the reference material and of the test substance are not adjacent.
- The solutions are placed in the cylinders or holes using a pipette that delivers a uniform volume of liquid.
- When the holes are used, the delivered volume should be sufficient to fill them almost completely.
- The plates are incubated at a suitable temperature, the selected temperature is controlled at +/- 0.5°C, for approximately 16 hours, and the diameters or areas of the zones of inhibition produced by the varied concentrations of the standard and of the test substance are measured accurately, preferably to the nearest 0.1 mm of the actualzone size, by using a suitable measuring device.
- From the results, the potency of the tested substance is calculated.
2. Turbidometric method:
Inoculate a suitable medium with a suspension of the chosen micro-organism having a sensitivity to the antibiotic to be examined such that a sufficiently large inhibition of microbial growth occurs in the conditions ofthe test.
- Use a known quantity of the suspension chosen to obtain a readily measurable opacity after an incubation period of about 4h.
- Use the inoculated medium immediately after its preparation.
- Using the solvent and the buffer solution indicated prepare solutions ofthe reference substance and of the antibiotic to be examined having known concentrations presumed to be an equal activity.
- In order for the validity of the assay may be assessed, use not fewer than 3 doses of the reference substance and 3 doses ofthe antibiotic to be examined which has the same presumed activity as the doses of the reference substance.
It is preferable to use a series of doses in geometric progression. To obtain the required linearity, it may be necessary to select from a large number 3 consecutive doses for the reference substance and the antibiotic to be examined.
Distribute an equal volume of each of the solutions into identical test tubes and add to each tube an equal volume of inoculated medium(for example, 1 ml ofthe solution and9ml ofthe medium).
- For the assay of tyrothricin, add 0.1ml of the solution to 9.9ml of inoculated medium.
- Prepare at the same time 2 control tubes without antibiotic, both containing the inoculated medium and to one of which is added immediately 0.5ml of formaldehyde R.
- These tubes are used to set the optical apparatus used to measure the growth.
- Place all the tubes, randomly distributed or in a Latin square or randomized block arrangement, in a water bath or suitable apparatus fitted with a means of bringing all the tubes rapidly to the appropriate incubation temperature.
Maintain them at that temperature for 3h to 4h, taking precautions to ensure uniformity of temperature and identical incubation time.
- After incubation, stop the growth of the microorganisms by adding 0.5ml of formaldehyde R to each tube or by heat treatment and measure the opacity of 3 significant figures using a suitable optical apparatus.
- Alternatively, use a method that allows the opacity of each tube to be measured after the same period of incubation.
- Calculate the potency using appropriate statistical methods.
- Linearity of the dose-response relationship, transformed or untransformed, is often obtained only over a very limited range.
- It is this range that must be used in calculating the activity and it must include at least 3 consecutive doses to permit linearity to be verified.
- In routine assays when the linearity ofthe system has been demonstrated over an adequate number of experiments using a three-point assay, a two-point assay may be sufficient, subject to agreement by the competent authority.
- However, in all cases of dispute, a three-point assay must be applied.
- Use in each assay the number of replications per dose sufficient to ensure the required precision.
- The assay may be repeated and the results combined statistically to obtain the required precision and to ascertain whether the potency of the antibiotic to be examined is not less than the minimum required.
3. Urease Assay:
- This assay is used for the assay of those antibiotics which inhibits protein synthesis.
- Aminoglycosides such as streptomycin, amikacin, kanamycin, gentamicin, netilmicin, and macrolides such as erythromycin, azithromycin, and clarithromycin.
- Certain micro-organisms like Proteus mirabilis
produce enzyme urease. - Production of urease indicated by an increase in pH of the media
- Antibiotics that inhibit protein synthesis, inhibit urea production so there is no further increase in pH. In this way, urease assay is done. This has very limited application.
4. Luciferase Assay:
- Luciferase is the enzyme that acts on luciferin protein produced by different micro-organisms’ presence of ATP and gives bioluminescence.
- This assay can be used to detect the very low amount of ATP present in bacterial culture. Drugs like Aminoglycosides inhibit ATP or reduce the level of ATP and luminescence doesn’t occur.
- If antibiotics inhibit the growth of micro-organisms, there is no ATP and no luminescence occurs.
5. Radioenzymatic Assay:
To detect the resistance of bacteria against different antibiotics, especially aminoglycosides and chloramphenicol
- If bacteria are resistant to these agents, they produce specific enzymes like aminoglycoside acetyltransferase(AAC), aminoglycoside adenyl transferase (AAD), and aminoglycoside phosphotransferase(APH) and thus become resistant to antibiotics
- In this case for aminoglycoside acetyltransferase (AAC) is radiolabelling agent 14C carbon isotope (with the help of cofactor [1- 14C] acetyl co-enzyme A) and for AAD and APH 3H (by [2- 3H] ATP) are used to detect the production of these aminoglycoside inactivating enzymes by micro-organisms.
- If microorganisms produce AAC, AAD, or APH, these enzymes become radiolabelled with their specific isotopes 14C/ 3H and give specific color changes.
- If color change is observed aminoglycoside resistance is inferred.
Question 39. Explain the different methods of maintenance of laboratory culture
Answer:
Laboratory culture Methods:
- Refrigeration
- Paraffin method
- Cryopreservation
- Lyophilization
1. Refrigeration:
- Pure cultures can be successfully stored at 0-4°C either in refrigerators or in cold rooms. This method is applied for a short duration(2-3 weeks for bacteria and 3-4 months for fungi) because the metabolic activities of the microorganisms are greatly slowed down but not stopped.
- Thus their growth continues slowly, nutrients are utilized and waste products are released in the medium. This results in, finally, the death of the microbes after some time.
2. Paraffin Method:
- This is a simple and economical method of maintaining pure cultures of bacteria and fungi. In this method, sterile liquid paraffin in poured over the slant (slope) of the culture and stored upright at room temperature.
- The layer of paraffin ensures anaerobic conditions and prevents dehydration of the medium. This condition helps microorganisms or pure culture to remain in a dormant state and, therefore, the culture is preserved for several years.
3. Cryopreservation:
- Cryopreservation(i.e., freezing in liquid nitrogen at- 196°C) helps the survival of pure cultures for long storage times.
- In this method, the microorganisms of culture are rapidly frozen in liquid nitrogen at -196°C in the presence of stabilizing agents such as glycerol, that prevent the formation ofice crystals and promote cell survival.
Lyophilisation (Freeze-Drying):
- In this method, the culture is rapidly frozen at a very low temperature (-70°C) and then dehydrated by vacuum. Under these conditions, the microbial cells are dehydrated and their metabolic activities are
stopped; as a result, the microbes go into dormant state and retain viability for years. - Lyophilized or freeze-dried pure cultures and then sealed and stored in the dark at 4°C in refrigerators. Freeze- drying method is the most frequently used technique by culture collection centers.
Question 40. Write the principle and procedure of acid-fast staining.
Answer:
Acid-fast staining Principle:
Acid-fast mycobacteria contain mycolic acid in their outer membrane, making the cells waxy and resistant to staining with aqueous based stains such as the Gram stain.
The primary stain, carbolfuchsin is applied to the cells, and heat and phenol are used to allow the stain to penetrate into the waxy surface of acid-fast microorganisms. The excess stain is removed with treatment by acid alcohol(ethanol and hydrochloric acid). A secondary stain, methylene blue, is then applied to the cells.
Ziehl-Neelsen stain reagents: Primary Stain:
0.3% Carbol-fuchsin. Dissolve 50 g phenol in 100 mL ethanol(95%) or methanol (95%). Dissolve 3 g Basic fuchsin in the mixture and add distilled water to bring the volume to 1 L.
- Decolorization Solution: Add 30 mL hydrochloric acid to 1 L of 95% denatured alcohol. Cool and mix well before use.
- Alternate decolorizing reagent (without alcohol): Slowly add 250 mL sulfuric acid(at least 95%) to 750 mL distilled water. Cool and mix well before using.
- Counterstain: 0.3% methylene blue. Dissolve 3 g nethylene blue in 1 L distilled water.
Acid-fast staining Procedure:
- Prepare and fix the specimen smear prior to staining.
- Place a small strip of blotting or filter paper over the top ofthe specimen, and place the slide over a boiling hot water bath on a mesh surface.
- Cover the filter paper with the primary stain, arbolfuchsin. Leave the slide on the water bath for 3 to 5 minutes. Continue to apply stain if the filter paper begins to dry.
- Remove the filter paper and rinse the slide with water until the solutionruns clear.
- Run an acid-alcohol decolorizer over the slide for approximately 10 to 15 seconds.
- Rinse the slide with water.
- Cover the smear with the secondary or counter stain, methylene blue, for 1 minute.
- Gently rinse the slide with water.
- Blot the slide dry with bibulous paper.
Acid-fast staining Result:
- Acid-fast: Bright red to intensive purple, Red, straight, or slightly curved rods, occurring singly or in small groups, may appear beaded
- Non-acid fast: Blue color; In addition, background material should stain blue.
Question 41. Explain the design and working of laminar airflow equipment.
Answer:
Airflow which is linear and positive up to working surfaces and thus prevents contamination of surrounding viable/nonviable particulate matter in aseptic handling.
A laminar flow cabinet consists ofthe following parts:
- Cabinet: The cabinet is made up of stainless steel with less or no gaps or joints preventing the collection of spores. The cabinet provides insulation to the inner environment created inside the laminar flow and protects it from the outside environment. The front ofthe cabinet is provided with a glass shield which in some laminar cabinets opens entirely or has two openings for the user’s hands to enter the cabinet.
- Working station: A flat working station is present inside the cabinet for all the processes to be taken place. Culture plates, burners, and loops are all placed on the working station where the operation takes place. The worktop is also made up of stainless steel to prevent rusting
- Filter pad/ Pre-filter: A filter pad is present on the top of the cabinet through which the air passes into the cabinet. The filter pad traps dust particles and some microbes from entering the working environment within the cabinet.
- Fan/ Blower: A fan is present below the filter pad that sucks in the air and moves it around in the cabinet. The fan also allows the movement of air towards the HEPA filter sp that the remaining microbes become trapped while passing through the filter.
- UV lamp: Some laminar flow hoods might have a UV germicidal lamp that sterilizes the interior ofthe cabinet and contents before the operation. The UV lamp is to be turnedon 15 minutes before the operation to prevent exposure of UV to the body surface of the user.
- Fluorescent lamp: Florescent light is placed inside the cabinet to provide proper light during the operation.
- HEPA filter: The High-efficiency particulate air filter is present within the cabinet makes the environment more sterile for the operation. The pre-filtered air passes through the filter which traps fungi, bacteria, and other dust particles. The filter ensures a sterile condition inside the cabinet, thus reducing the chances of contamination.
Laminar air flow equipment Working:
- The principle of laminar flow cabinet is based on the laminar flow of air through the cabinet.
- The device works by the use of an inwards flow of air through one or more HEPA filters to create a particulate-free environment.
- The air is taken through a filtration system and then exhausted across the work surface as a part of the laminar flow ofthe air.
- The air first passes through the filter pad or pre-filter which allows a streamlined flow of air into the cabinet.
- Next, the blower or fan directs the air towards the
- HEPA filters.
- The HEPA filters then trap the bacteria, fungi, and other particulate materials so that the air moving out of it is particulate-free air.
- Some of the effluent air then passes through the perforation present at the bottom rear end of the cabinet, but most of it passes over the working bench while coming out of the cabinet towards the face of the operator.
- The laminar flow hood is enclosed on the sides, and constant positive air pressure is maintained to prevent the intrusion of contaminated external air into the cabinet.
The procedure to be followed while operating a laminar flow cabinet is given below:
- Before running the laminar flow cabinet, the cabinet should be checked to ensure that nothing susceptible to UV rays is present inside the cabinet.
- The glass shield of the hood is then closed, and the UV light is switched on. The UV light should be kept on for about 15 minutes to ensure the surface sterilization ofthe working
- The UV light is then switched off, and a time period of around 10 minutes is spared before the airflow is switched on.
- About 5 minutes before the operation begins, the airflow is switched on.
- The glass shield is then opened, and the fluorescent light is also switched on during the operation.
- To ensure more protection, the working bench ofthe cabinet can be sterilized with other disinfectants like 70% alcohol.
- Once the work is completed, the airflow and florescent lamp both are closed and the glass shield is also closed.
Question 42. Discuss the types of microbial spoilage.
Answer:
Types of Microbial Spoilage:
1. Infection induced due to contaminated pharmaceutical products:
- Pharmaceutical products may get infected from many sources like raw materials, manufacturing, storage etc.
- Such infected products when consumed cause diseases.
- For example: Cholera, Pseudomonas infections, Botulism etc.
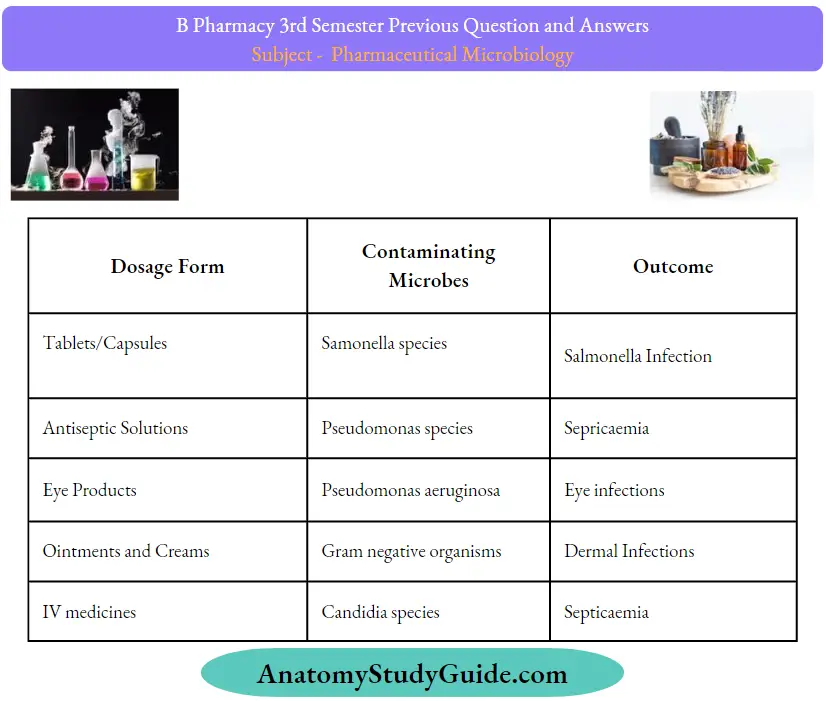
2. Physico-chemical spoilage:
In this kind of spoilage, there are some changes caused by microbial species and due to these changes the physical properties are also gets altered or deteriorated, thus it is called physico-chemical spoilage.
- Viable growth: Microbial cells form a viable layer over the surface of pharmaceutical formulation. This layer or the presence of microbial cells can be seen by the naked eyes. a layer of molds over syrups or sugar-containing products.
- Change in Color of the formulation: Occurs due to the change in the component’s chemical nature. Surface decoloration of tablets containing biological products by some mould
- Gas production: Some microbe contaminants in pharmaceutical formulations produce gases by their metabolic activities and form gas bubbles and foam over the formulation.
- For example: In carbohydrates containing formulations like syrups by yeasts.
3. Physical Spoilage:
These changes in physical appearance are caused by microbial activities in the formulation.
- Cracking of emulsion: Microbes eat off the emulsifying agent causing instability of emulsions resulting in separation of phases.
- Odor changes: Microbes due to their activity in the pharmaceutical product impart smell. Toluene smells due to spoilage of Balsam of Tolu by Penicillium species.
- Smell of geosmin to water phases by
- Actinomycetes contamination.
4. Biological spoilage:
Microbial contamination of the products results in the formation of chemicals in the products due to microbial metabolic activity, this is termed as Biological spoilage.
Mainly two types of chemicals are produced by microbes:
- Microbial Toxins.
- Microbial Metabolites.
Microbial Toxins: Endotoxins produced by some gram-positive bacteria like E.coli. Microbial Metabolites: Different types of amines and organic acids.
5. Chemical spoilage:
This occurs due to various types of chemical reactions mediated by contaminating microorganisms.
Question 43. Write the principle of sterility testing.
Answer:
Sterility testing Principle:
- If micro-organisms are placed in a media that provides nutrients and water and kept at a favorable temperature the organism will grow and their growth can be indicated turbidity in the originally clear medium.
- The sterility tests provide optimum conditions for the growth and multiplication of organisms, spores, etc, that might be a contaminant.
- It is not possible to claim that a batch of products is sterile unless the entire content of each batch has been tested.
- But these conditions are not possible because the preparation under test is either made unstable or is destroyed.
- Thus only a part of the batch can be sampled for testing.
Question 44. Discuss on basic requirements to establish the culture laboratory.
Answer:
- Cultivation should be done under aseptic conditions.
- The isolated plant part should get an appropriate environment that will help to divide the cell and to get an expression of internal potential.
Basic facilities are:
- Washing and storage facilities;
- Media preparation, sterilization, and storage room;
- Transfer area for aseptic manipulations;
- Culture rooms or incubators for maintenance of cultures under controlled conditions of temperature, light, and humidity;
- Observation or data collection area;
- Transplantation area.
1. Washing and Storage Facilities:
- An area with a large sink(lead-lined to resist acids and alkalis) and a draining area is necessary with provision for running water, draining boards or racks, and ready access to a deionized, distilled, and double-distilled apparatus.
- Space should also be available to set up drying ovens, washing machines, plastic or steel buckets for soaking labware, acid or detergent baths, pipette washers, driers, and cleaning brushes. For storage of washed and dried labware, the laboratory should be provided with dustproof cupboards or storage cabinets.
2. Media Preparation Room or Space:
This part is the central section of the laboratory where most of the activities are performed i.e., media preparation and sterilization of media and glassware needed for culture. There should be sufficient working bench as well as storage space.
Others:
- Different types of glassware
- Different kinds of balances
- Required chemicals
- Hot plates and Stirrer
- Water bath
- pH meter
- Autoclave and Hot air oven
- Microwave oven
- Vortex, Shaker
- Centrifuge
- Refrigerator and Freezer
- Storage cabinet (Dust-free)
3. Transfer Area:
- Tissue culture techniques can only be successfully carried out in a very clean laboratory with having dry atmosphere with protection against air-borne microorganisms.
- For this purpose, a sterile dust-free room/cabinet is needed for routine transfer and manipulation work.
- The ‘laminar airflow cabinet’ is the most common accessory used for aseptic manipulations nowadays.
- The cabinet may be designed with horizontal airflow or vertical airflow where the air is forced into the cabinet through a bacterial HEPA (High-Efficiency Particulate Air) filter.
- The air flows over the working bench at a constant rate which prevents the particles (microorganisms) from settling on the bench.
- Before operation in the laminar airflow cabinet, the interior of the cabinet is sterilized with ultraviolet(UV) germicidal light, and the floor of the cabinet with 70% alcohol.
- An inoculation chamber, a specially designed air-tight glass chamber fitted with UV light, may also be used as a transfer area.
4. Culture Room:
- Plant tissue cultures should be incubated under conditions of well-controlled temperature, illumination, photoperiod, humidity, and air circulation.
- Incubation culture rooms, commercially available incubator cabinets, large plant growth chambers, and walk-in environmental rooms satisfy these requirements.
- Culture rooms are constructed with proper airconditioning; perforated shelves to support the culture vessels, fitted with fluorescent tubes having a timing device to maintain the photoperiod, and black curtains may be used to maintain total darkness.
- For the suspension cultures, gyratory shakers are used.
- Air conditioners and heaters are used to maintain the temperature around 25 ± 2°C and humidity is maintained by uniform forced air ventilation.
- The lighting is also done in a measured amount i.e., 40-200 fc(foot-candle).
Data Collection Area:
The growth and development of tissues cultured in vitro are generally monitored by observing cultures at regular intervals in the culture room or incubators where they have been maintained under controlled environmental conditions.
Arrangements should be there where the observations can be done under aseptic conditions using a microscope. Special facilities are required for germplasm conservation i.e., cryopreservation accessories should be there.
Transplantation Area:
Plants regenerated from in vitro tissue culture are transplanted to soil in pots. The potted plants are ultimately transferred to the greenhouse but before transfer the tissue culture-grown plants are allowed for acclimatization under well-humid conditions and controlled temperature and controlled entry of sunlight
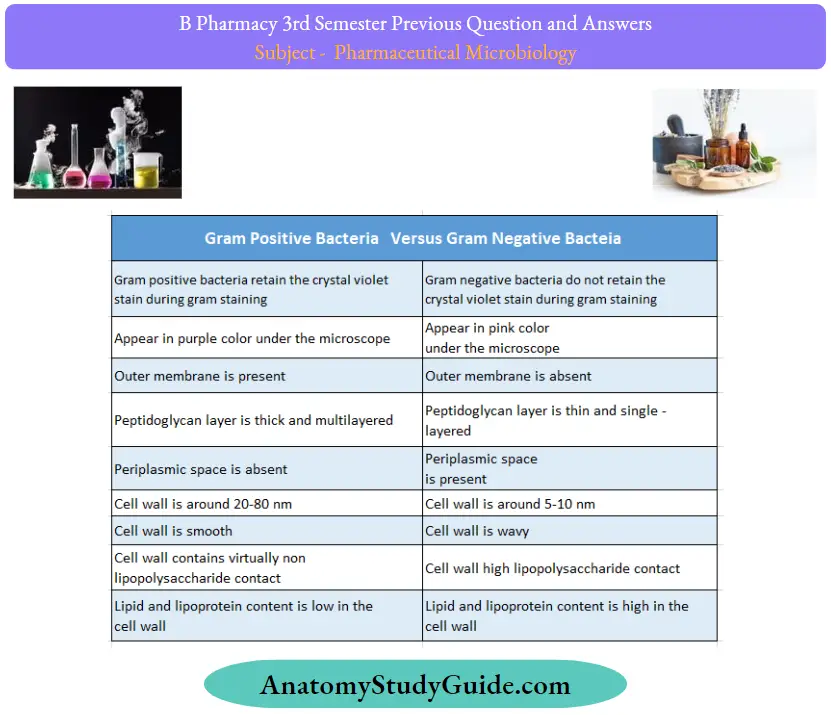
Question 45. Difference between gram-positive and gram-negative bacteria.
Answer:
Question 46. Holder method of pasteurization.
Answer:
Holder pasteurization is a gentle method, heating milk to 62.5 degrees Celsius for 30 minutes as opposed to the higher temperatures and shorter intervals of more aggressive alternatives. Holder pasteurization eliminates harmful bacteria but allows the milk to retain much of its immunological benefits, including significant amounts of secretory IgA and lysozyme.
Question 47. Biological indicator.
Answer:
A biological indicator is a microbiological test system consisting of a standardized viable population of specific microorganisms(usually bacterial spores) inoculated on a carrier contained within its primary pack ready for use and providing a defined resistance to a specified sterilization process.
They provide means to directly assess the microbial lethality of a sterilization process. When used in conjugation with physical or chemical process monitors, biological indicators indicate the effectiveness of a given sterilization process
Question 48. Sterility test for powders.
Answer:
For dry powder or lyophilized container, add the sterile water/0.1% peptone dissolve and then collect it in the sterile flask. 5.3.12 Filter the collected solution aseptically through 0.45|i membrane filter with the help of a vacuum
Question 49. Fungi.
Answer:
The fungi are traditionally considered heterotrophs, organisms that rely solely on carbon fixed by other organisms for metabolism. Fungi have evolved a high degree of metabolic versatility that allows them to use a diverse range of organic substrates for growth, including simple compounds such as nitrate, ammonia, acetate, or ethanol.
Fung Medicinal uses:
- Reduce cholesterol
- Treat anemia
- Help prevent cancer
- Prevent diabetes
- Improves bone health
- Helps in nutrient absorption
- he immune system
- Contributes to lower blood pressure
- Increases iron absorption
- Rich in selenium
- Weight loss
- Regenerates the skin
- Help in patients with Hepatitis B
- Everyday spice
- Reduce inflammation
- HIV defense
Question 50. Viable count.
Answer:
Viable count is a method used in cell culture to determine the number of living cells in a culture. This is different from other cell counting techniques because it makes a distinction between live and dead cells. The count represents the number of colony-forming units(CFU) per g(or per ml) ofthe sample.
Question 51. D value.
Answer:
The D-value or decimal reduction time(or decimal reduction dose) is the time(or dose) required, at a given condition
- For example – Temperature or set of conditions,
- To achieve a log reduction, that is, to kill 90% (or 1 log) of relevant microorganisms.
- A D-value is denoted with the capital letter “D”. D-values are sometimes used to express a disinfectant’s efficiency in reducing the number of microbes present in a given environment
Question 52. Applications of animal cell culture.
Answer:
Applications of animal cell culture are:
- Model systems
- Toxicity testing
- Cancer research
- Virology
- Cell-based manufacturing
- Genetic counseling
- Genetic engineering
- Gene therapy
- Drug screening and development
Question 53. Sterilization of surgical dressings.
Answer:
Sterilization of Surgical Dressings:
Materials like cotton wool balls, gauze swabs, ribbon gauze, bandages, etc,are used in surgery and inwards. These are commonly known as materials of surgical dressings.
The following stages are involved in the sterilization of surgical dressings:
- Packing or wrapping the unsterilized surgical dressings into a suitable container or packaging material. For this purpose, metal drums are used. These drums are perforated to facilitate the free flow of steam during sterilization. The surgical dressing can also be wrapped with fabrics like nylon film bags muslin cloth or calico cloth.
- The suitably packed dressings are correctly loaded into the sterilizer. The packing should be done in such a way that it ensures satisfactory steam penetration and air removal. So, the pack should be kept as small as possible. The heavy materials should not be sterilized with soft materials. The contents should be packed loosely and the spaces between the items and folds should be parallel.
- The sterilizer is closed and inside the sterilizer is replaced by steam. The fabric pack (surgical dressings) is exposed for 30-45 minutes at 121 degrees to sterilize the surgical dressings in it. When surgical dressings are packed in a metallic drum, a still longer time is required.
- Switch off the sterilizer and condense the steam inside it. The contents of the sterilizer are then removed. The warm packs are not placed on a cold surface. It is done to avoid condensation of steam into water which may cause contamination through porous wrappers.
- The containers are labeled indicating the date of sterilization to prevent overlong storage
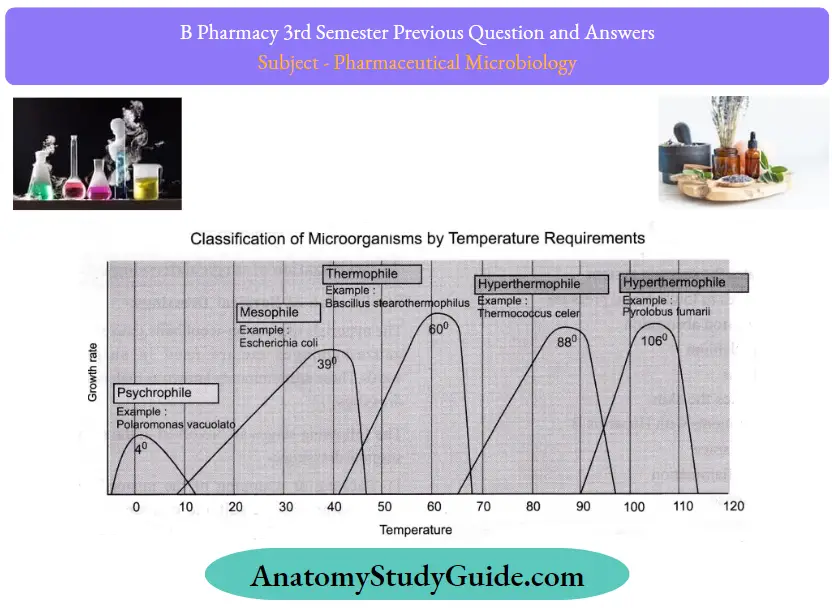
- Classify bacteria based on their temperature requirements with examples.
Question 54. Give the principle and working of phase contrast microscopy.
Answer:
Unstained living cells absorb practically no light. Poor light absorption results in extremely small differences in the intensity distribution in the image. This makes the cells barely, or not at all, visible in a brightfield microscope. Phase contrast microscopy is an optical microscopy technique that converts phase shifts in the light passing through a transparent specimen to brightness changes in the image
Phase contrast microscopy Principle:
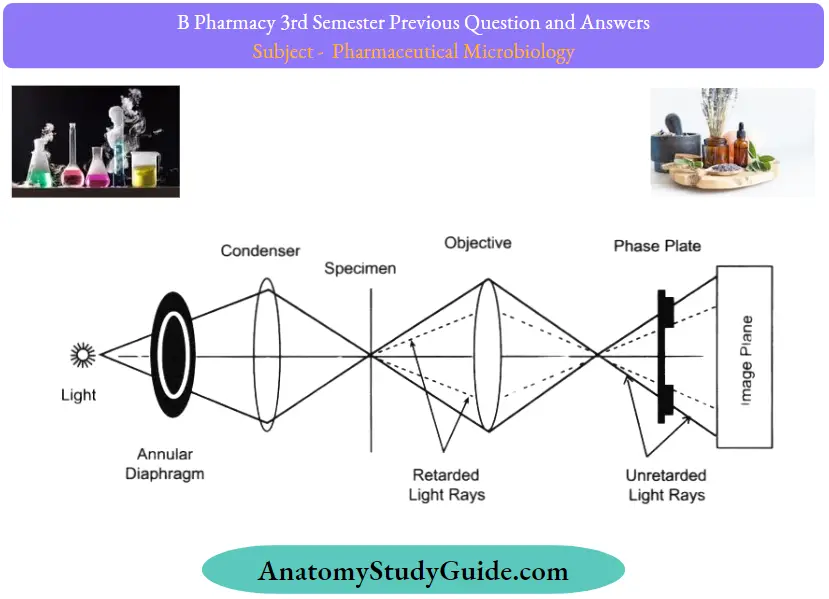
When light passes through cells, small phase shifts occur, which are invisible to the human eye. In a phase-contrast microscope, these phase shifts are converted into changes in amplitude, which can be observed as differences in image contrast.
Phase Contrast Microscopy Working:
- Partially coherent illumination produced by the tungsten-halogen lamp is directed through a collector lens and focused on a specialized annulus (labeled condenser annulus) positioned in the substage condenser front focal plane.
- Wavefronts passing through the annulus illuminate the specimen and either pass through undeviated or are diffracted and retarded in phase by structures and phase gradients present in the specimen.
- Undeviated and diffracted light collected by the objective is segregated at the rear focal plane by a phase plate and focused at the intermediate image plane to form the final phase-contrast image observed in the eyepieces.
Contrast microscopy Parts:
Phase-contrast microscopy is a specially designed light microscope with all the basic parts in addition to which an annular phase plate and annular diaphragm are fitted.
The annular diaphragm:
- It is situated below the condenser.
- It is made up of a circular disc having a circular annular groove.
- The light rays are allowed to pass through the annular groove.
- Through the annular groove ofthe annular diaphragm, the light rays fall on the specimen or object to be studied.
- At the back focal plane ofthe objective develops an image.
- The annular phase plate is placed at this back focal plane.
The phase plate:
- It is either a negative phase plate having a thick circular area or a positive phase plate having a thin circular groove.
- This thick or thin area in the phase plate is called the conjugate area.
- The phase plate is a transparent disc.
- With the help ofthe annular diaphragm and the phase plate, the phase contrast is obtained in this microscope.
- This is obtained by separating the direct rays from the diffracted rays.
- The direct light rays pass through the annular groove whereas the diffracted light rays pass through the region outside the groove.
- Depending upon the different refractive indices of different cell components, the object to be studied shows a different degree of contrast in this microscope.
Contrast microscopy Applications:
- Living cells (usually in culture),
- Microorganisms,
- Thin tissue slices,
- Lithographic patterns, fibers,
- Latex dispersions,
- Glass fragments, and
- Subcellular particles (including nuclei and other organelles).
Question 55. Describe the bacterial growth curve.
Answer:
The bacterial growth curve shows the following four distinct phases:
1. Lag phase:
- After a liquid culture broth is inoculated, the multiplication of bacteria does not start immediately. It takes some time to multiply.
- The time between inoculation and the beginning of multiplication is known as the lag phase.
- In this phase, the inoculated bacteria become acclimatized to the environment, switch on various enzymes, and adjust to the environmental temperature and atmospheric conditions.
- During this phase, there is an increase in size of bacteria but no appreciable increase in number of bacterial cells. The cells are active metabolically.
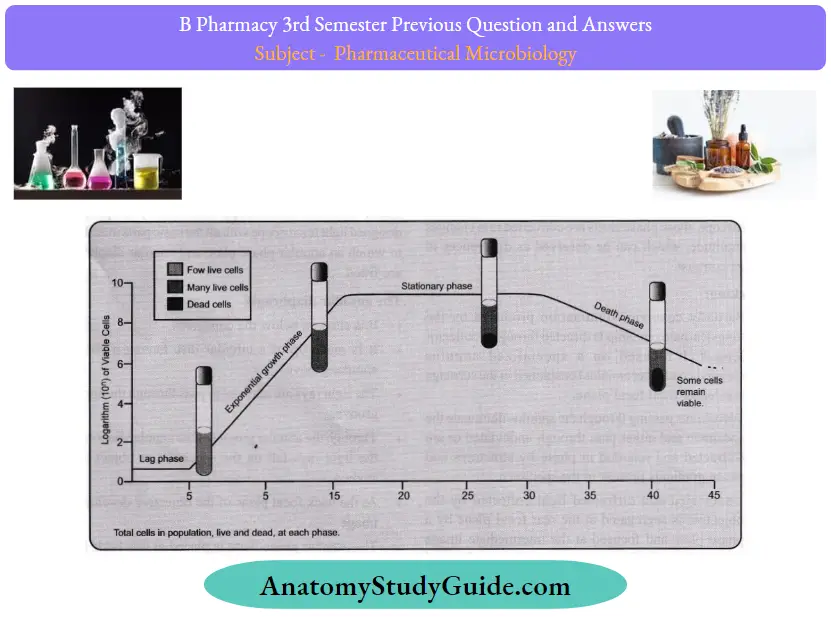
The duration of the lag phase varies with the bacterial species, nature of the culture medium, incubation temperature, etc. It may vary from 1 hour to several days.
2. Log phase:
- This phase is characterized by rapid exponential cell growth (i.e., 1 to 2 4 to 8, and so on).
- The bacterial population doubles during every generation. They multiply at their maximum rate.
- The bacterial cells are small and uniformly stained.
- The microbes are sensitive to adverse conditions, such as antibiotics and other antimicrobial agents.
- The growth rate is the greatest during the log phase.
- The log phase is always brief unless the rapidly dividing culture is maintained by constant addition of nutrients and frequent removal of waste products.
- When plotted on logarithmic graph paper, the log phase appears as a steeply sloped straight line.
3. Stationary phase:
- After the log phase, bacterial growth almost stops completely due to lack of essential nutrients, lack of water oxygen, changes in pH of the medium, etc., and accumulation of toxic metabolic wastes.
- It is during this phase that the culture is at its greatest population density.
- However, the Death rate of bacteria exceeds the rate of replication of bacteria.
- Endospores start forming during this stage.
- Bacteria become Gram variable and show irregular staining.
- Many bacteria start producing exotoxins.
4. Decline phase:
- During this phase, the bacterial population declines due to the death of cells.
- The decline phase starts due to
- Accumulation of toxic products and autolytic enzymes and
- Exhaustion of nutrients.
- Involution forms are common in this stage. Some cells assume various shapes, becoming long filamentous rods or branching or globular forms
that are difficult to identify. - Some develop without a cell wall and are referred to as protoplasts, spheroplasts, or L-phase variants (L-forms).
- When these involuted forms are inoculated into a fresh nutrient medium, they usually revert to the original shape ofthe healthy bacteria
Bacterial growth curve Significance:
- The study of bacterial growth curves is important when aiming to utilize or inoculate known numbers of the bacterial isolate, for example, to enhance plant growth, increase biodegradation of toxic organics, or produce antibiotics or other natural products at an industrial scale
- Knowledge of bacterial growth kinetics and bacterial numbers in a culture medium is important from both a research and commercial point of view.
- Growth kinetics is also useful for assessing whether particular strains of bacteria are adapted to metabolize certain substrates, such as industrial waste or oil pollution.
- Bacteria that are genetically engineered to clean up oil spills, for example, can be grown in the presence of complex hydrocarbons to ensure that their growth would not be repressed by the toxic effects of oil.
- Similarly, the slope and shape of growth curves produced from bacteria grown with mixtures of industrial waste products can inform scientists whether the bacteria can metabolize the particular substance, and how many potential energy sources for the bacteria can be found in the waste mixture.
Question 56. Write the procedure, merits, and demerits of membrane Alteration.
Answer:
A membrane filtration method involves the following steps:
- Sample collection: Collect water samples like groundwater (high in Cu and Zn) and wastewater (high in heavy metals) in the sterilized sample bottles. Then, dilute the water sample by making a volume of up to 100 ml.
- Selection of appropriate nutrient medium: For the isolation and enumeration of different pathogenic
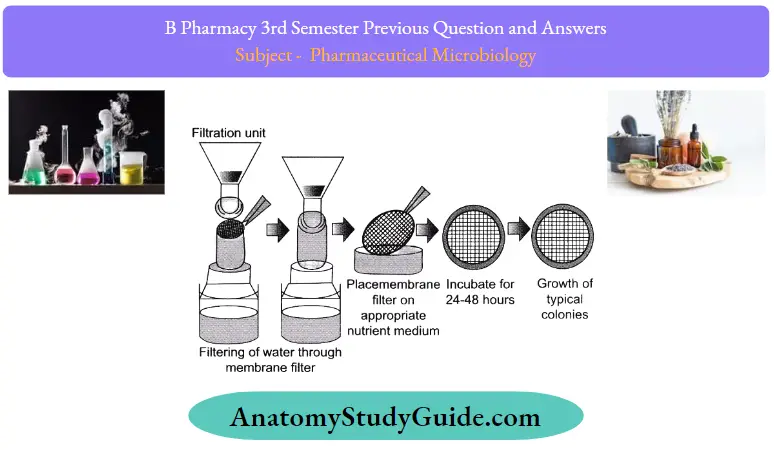
Microorganisms in water, we can use a variety of nutrient mediums:
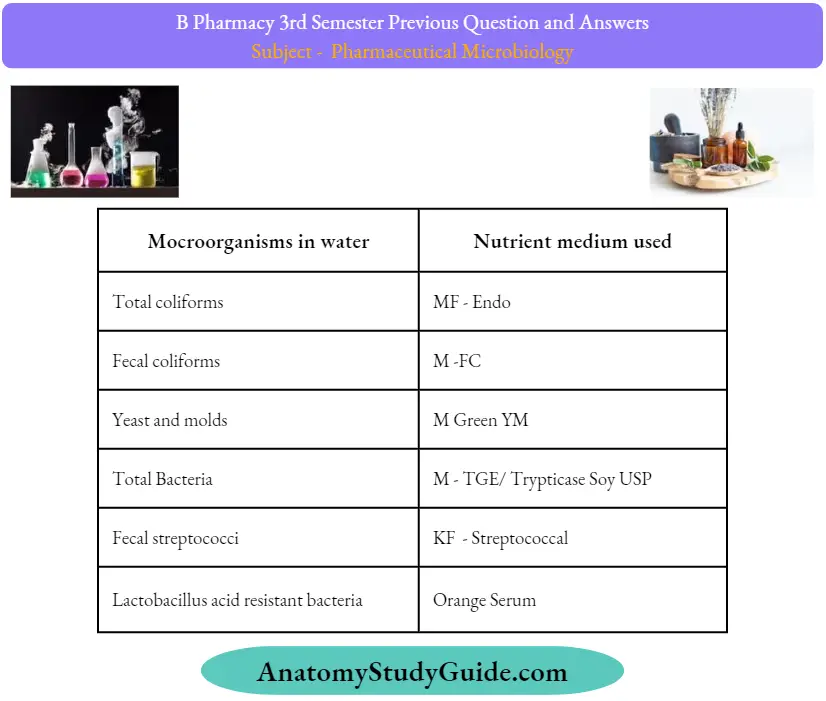
- After selecting an appropriate nutrient medium, prepare and transfer the media to the sterilized Petri plate containing an absorbent pad at the bottom.
- Soak the absorbent pad with the liquid broth. The saturation of the absorbent pad with the nutrient medium will promote microbial growth.
- Membrane filtration: For the membrane filtration of water, place a membrane filter over the carbon disk by using force sterilized under the flame.
- Then, flame the opening of the sample bottles and pour the test water sample, through the funnel.
- A vacuum system allows the complete separation of filtrate through the membrane filter. After complete separation of filtrate, rinse the funnel with the buffered water.
- Transfer of membrane filter: Flame the forces and take out the membrane filter from the filtration assembly.
- Then, place a membrane filter over the prepared petri dish containing an absorbent pad saturated with a liquid nutrient medium. Finally, incubate the Petri plate for 24-48 hours at 35-37 degrees Celsius temperature.
Nutrient medium Observation:
For the quantitative analysis, we need to count the number of colonies directly on the colony counter. Conversely, to test the water quality, we need to isolate and identify the type of microorganisms by subculturing the isolated colonies. Further, we could examine the specific type of microbial group through staining methods, microscopic observations, and biochemical tests
Nutrient medium Advantages:
- MF technique is an alternative to the most probable number method.
- One can test a large volume of water sample through the millipore filtration method.
- Membrane filtration consumes less time than the MPN method, as it only involves four to five steps to get the results.
- MF technique gives more relevant and more reliable results than the MPN method.
- One can enumerate the number of bacteria present in the water sample directly by using the colony counter.
Nutrient medium Disadvantages:
The membrane filtration method is not applicable to test the turbid water. There may be a chance of bacterial overgrowth, as the water contains many microorganisms
Question 57. What are the main sources of contamination of an aseptic room? How will you prevent it? An aseptic area is a premise in a clean area, designed, constructed, serviced, and used to prevent microbial contamination ofthe product.
Answer:
It is important to identify the sources from which the contaminants can enter the aseptic area to avoid the contamination of products under operation.
The important sources of contamination in an aseptic area are as follows:
- Personnel
- Buildings and Facilities
- Equipment and Utensils
- Raw Materials
- Manufacturing Process
- An HVAC (Heating, Ventilation, and Air Conditioning) System.
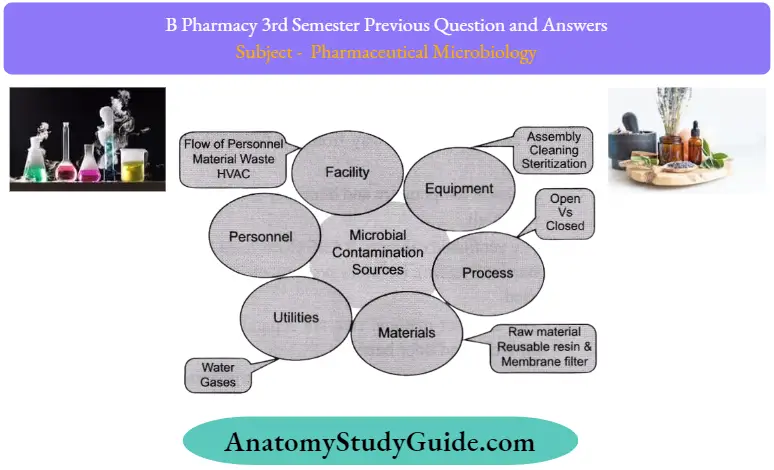
1. Personnel:
- Unauthorized personnel entering the facility, (This is why educational visits are denied in sterile areas).
- Insufficient gowning and protective equipment,
- Eating, drinking, or smoking within the storage and processing areas.
2. Buildings and Facilities:
These can also be important contributors to microbial contamination due to the following reasons:
- Inadequate size and organization of the space, which leads to selection errors (such as mixups or cross-contamination between consumables, raw materials, in-process materials, and finished products),
- Poor filth and pest controls,
- Rough floors, walls, and ceilings,
- Absence of air filtration systems,
- Inadequate lighting and ventilation systems,
- Poorly located vents, ledges, and drains, and
- Improper washing, cleaning, toilet, locker facilities, sanitary operation.
3. Equipment and Utensils:
They are the common sources of microbial contamination due to the following reasons
- Unsuitable design, size, corrosion-causing materials, and/or adulteration with lubricants, coolants, dirt, and sanitizing agents,
- Inadequate cleaning and sanitization,
- Inefficient cleaning and maintenance due to their designing
- Inappropriate calibration and irregular service, and
- Using defective equipment
4. Raw Materials:
- These are used during production and are considered a potential source of contamination due to the following reasons:
- Improper storage and handling, which leads to mix-ups or selection errors,
- Microbial or chemical contamination,
- Degradation due to extreme environmental conditions (like heat, cold, sunlight, moisture, etc.)
- Wrong labeling.
- Incorrect sampling and testing, and
- Using materials not meeting the acceptance criteria.
5. Manufacturing Process:
- During the manufacturing process, microbial contamination of raw materials, intermediates, or packaging materials can widely occur due to the following reasons
- Absence of facilities required for manufacturing of single product,
- Improper cleaning between batches to minimize the amount of product changeovers,
- Use of an open manufacturing system for exposing the product to the room environment, Improper zoning, Lacking an area line clearance (as per the approved procedures) after each cleaning process and between each batch, and lack of cleaning status labeling on all equipment and materials used within the manufacturing facility.
6. A HVAC (Heating, Ventilation, and Air Conditioning) System:
An inappropriate HVAC system is a possible source of microbial growth and also disperses contaminants throughout the manufacturing unit
This occurs due to the following reasons:
Organic materials accumulate in or near HVAC air intakes, Inadequate air filtration system, Inadequate magnitude of pressure differentials, which causes the flow of reversal, Incorrect ratio of fresh air to re-circulated air, Incapability of accessing ventilation dampers and filtering from outside the manufacturing areas, and Non-directional airflow within a production or primary packing areas. W
Methods of Prevention of Contamination:
1. Personnel:
- Access to production areas by unauthorized personnel should be restricted, and only trained ones should be allowed to enter. Adequate personnel hygiene should be maintained.
- The personnel should be given proper and regular training concerning hygiene as well to ensure that their activities do not hamper the product quality.
- Personnel entering the manufacturing area should wear protective clothing (over-garments, hair cover, beard or mustache cover, and overshoes).
- Personnel should avoid touching with naked hands the exposed products or any part of equipment in contact with the product.
2. Buildings and Facilities:
- In aseptic rooms, the differential air pressures should be higher than the adjacent controlled areas.
- Air filtration and air change rates should be set to attain the defined cleanroom class.
- Unidirectional (laminar flow) airflow should be maintained over critical areas at sufficient velocity to sweep particles away from the filling/closing area.
- Ambient temperature and humidity should not be very high.
- The ventilated cabinets, RABS (Restricted Access Barrier System), isolator systems, etc. should be used.
- The areas of unequal risk are separated by UV air locks and door barriers. All open-container processing should be performed within the isolator/RABS.
- The air pressure ofthe changing room should be negative concerning the manufacturing area corridor, but positive concerning the external adjacent areas.
- The ventilation dampers, specially designed filters, and other services should be positioned such that they can be accessed from outside the manufacturing areas (service voids or service corridors) for maintenance purposes.
An impermeable barrier should be used in appropriate areas to prevent cross-contamination between the two zones.
HVAC air distribution components should be used to prevent the spreading of contaminants generated within the room. All rooms and surfaces should be maintained and monitored for viable and non-viable particulates and this facility should be re-certified on a semi¬ annual basis.
3. Access to Areas Access:
to production, packaging, and QC areas by unauthorized personnel should be restricted. Personnel should gain access to these areas only via changing rooms. Materials should be accessed via specific routes (generally airlocks).
4. Building Requirements:
- Smooth, crack-free, and easily cleanable floors, walls, and ceilings should be used.
- Windows or viewing panels should be closed (opening), fixed with wall panels, and sealed to prevent the accumulation of dust and microbial contaminants.
- The designing of pipework, ventilation, and light points should be such that the Creation of spaces that are not easily cleanable is avoided.
- Sinks of stainless steel should be present within the production areas.
5. Cleaning and Disinfection:
- Time-to-time cleanliness and disinfection of the areas should be done. Cleaning agents of proper grades should be used to minimize health risks.
- Contact time, application, temperature, mechanical action, and the chemistry of cleaning agents should be considered during the cleaning process.
- The cleaning agents should not be directly applied on the product.
- The cleaning practices should be validated to prove that the process effectively controls microbial contamination.
- UtilitiesWater of pharmaceutical grade, microbiologically controlled and monitored should be used in the manufacturing of products.
- Clean and additive-free steam should be used for cleaning and sanitization of production tools and equipment, and to supply for autoclaves and humidification.
Question 58. Explain the principle involved in the microbiological assay of streptomycin.
Answer:
Urea medium, whether a broth or agar, contains urea and the phenol red as a pH indicator.
- Many organisms, especially those that cause urinary tract infections, produce the urease enzyme, which catalyzes the splitting of urea in the presence of water to release two molecules of ammonia and carbon dioxide.
- The ammonia combines with the carbon dioxide and water to form ammonium carbonate, which turns the medium alkaline, turning the indicator from its original orange-yellow color to bright pink.
- His test is performed as part of the identification of several genera and species of the Enterobacteriaceae family, including Klebsiella, Proteus, and some Citrobacter and Yersinia species, as well as some Corynebacterium species.
- The test is also useful to identify Cryptococcus, Brucella, Helicobacter pylori, and many other bacteria that produce the urease enzyme. Disks are available that combine urea and phenylalanine deaminase (PDA), allowing a one-disk test to identify Proteus, Providencia, and Morganella and to separate them from Klebsiella and Yersinia enterocolitica.
- The disk reactions are rapid and sensitive and allow for the rapid detection of agents of serious infections, Brucella, and Cryptococcus.
Microbiological assay Procedure:
1. Preparation of media:
About 24.52 grams ofthe dehydrated medium is dissolved in 950 ml of distilled water in a beaker. The solution is heated to bring it to a boil to dissolve the medium completely
- The prepared suspension is sterilized by autoclaving at 15 lbs pressure, 121°C for 15 minutes.
- The beaker is taken out following the autoclaving and cooled to 50°C. To the beaker, 50 ml of sterile 40% urea solution is added and mixed well.
- The medium is dispensed into tubes and set in a position to obtain agar slants.
2. Urease Activity:
A loopful of well-isolated colonies is taken with an inoculating loop and inoculated on the agar slants.
- The inoculation should be done on just the slant, and the butt shouldn’t be stabbed.
- The tubes are then incubated with loosened caps at 35 to 37°C. For non-fermenters, the tubes are incubated at 30°C.
- The tubes are observed for the development of pink color for as long as 7 days.
- If no growth is seen on the slant, further inoculation with heavy inoculum should be done.
3. Rapid Urease Test:
The Rapid urease test is also called the CLO test (Campylobacter-\ike organism) and is used for the rapid identification of Helicobacter pylori.
- For the rapid urease test, a biopsy is taken ofthe mucosal layer of the antrum of the stomach and placed on the urea broth with a phenol red indicator.
- The tube is then observed for the change in color from yellow to pink.
- A positive test is demonstrated by an intense magenta to bright pink color in 15 min to 24 hours. A negative test shows no color change.
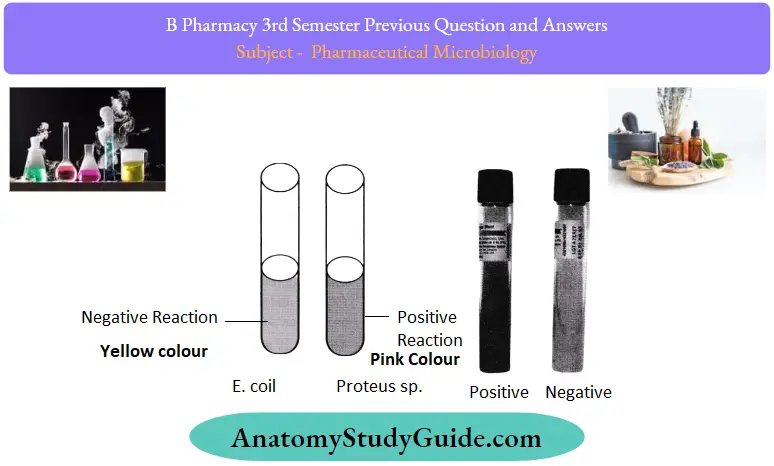
Question 59. Explain the sources of microbial contamination in pharmaceuticals.
Answer:
Contaminants can gain entry into a production process stream from several sources such as,
- Personnel,
- Poor facility design,
- Incoming ventilation air,
- Machinery and other equipment for production,
- Raw material and semi-finished material,
- Packaging material,
- Utilities,
1. Working Environment:
- Air is not a natural environment for microbes to grow as it does not contain water and nutrients needed for growth.
- However, the microbes are carried into the atmosphere and remain suspended on particles of dust, skin, clothing, moisture or nasal droplets, and sputum following sneezing or coughing.
- The microbial load ofthe air may increase during the handling of contaminated materials during dispensing, blending, and formulation processes.
- The use of starch and sugars in the dry state may increase fungal counts.
- Settle plates and different air sampling methods are commonly used for the isolation and identification of microbes present in the working environment.
- Microbes that are commonly isolated from the atmosphere are Staphylococcus species, Streptococcus spp., Bacillus spp., Microsporum spp., Aspergillus spp., Penicillium spp. etc.
2. Personnel:
Personnel who are supervising or performing drug manufacturing or control can be a potential source of microbiological contamination and a vector for other contaminants like, Natural skin flora of microorganisms can be a main source of contamination.
For eaxmple: Staphylococcus aureus, Sarcina spp., Alcaligenes spp. Etc, various dermatophytic fungi viz. Epidermophyton spp.,
- Microsporum spp. etc. If persons have wounds they may support
- organisms like, Staphylococcus aureus,
- Staphylococcus pyogenes, Proteus spp.,
- Pseudomonas aeruginosa etc.The respiratory discharge of the personnel may include organisms like Staphylococcus aureus,
- Staphylococcus salivarius, Neisseria pharyngitis,
- Klebsiella pneumoniae etc.
The main reasons for contamination from the personnel include:
- Lack of training in Direct contact between the operator’s hands and starting materials, primary packaging materials and intermediate or bulk productInadequate personnel cleanliness
- Access of unauthorized personnel into production, storage, and product control areas gowning and personnel protective equipment, and Malpractices like eating food, drinking beverages, or using tobacco in the storage and processing areas.
3. Buildings and Facility:
- The buildings and manufacturing facilities may also contribute to the contamination.
- Walls and ceilings of buildings are sources commonly for Aspergillus spp., Cladosporium spp., Penicillium spp., Bacillus spp. etc. They are mainly found in poorly ventilated buildings.
- These microorganisms utilize the contents ofthe wall paint as nutrients.
- The other reasons for contamination due to facility issues include Inadequate filth and pest controls.
- Rough floors, walls, and ceilings.Lack of air filtration systems.
- Improper lighting and ventilation. Poorly located vents, and drains.
- Inadequate washing, cleaning, toilet, and locker facilities to allow for sanitary operation, cleaning of facilities, equipment, and utensils; and personal cleanliness.
4. Equipment:
- The equipment and utensils used in processing, holding, transferring, and packaging are the common sources of pharmaceutical contamination.
- The main reasons for contamination from the equipment include
- Inappropriate design, size, and material leading to corrosion and accumulation of static material and/or adulteration with lubricants, coolants, dirt, and sanitizing agents.
- Improper cleaning and sanitization.
- The design prevents proper cleaning and maintenance.
- Improper calibration and irregular service, and Deliberate use of defective equipment.
5. Materials:
- The raw materials used for production can be a potential source of contamination. Animal-origin raw materials like gelatin, thyroid, and pancreas and Plant origin products like starch, acacia tragacanth, etc. are known sources of microbial contamination.
- Raw materials of mineral origin are usually free from microbes while some like zinc oxide, calamine, bentonite may contain spores of Clostridium spp.
- The main reasons for contamination from the raw materials include Storage and handling mistakes causing mix-ups or selection errors. Contamination with microorganisms or other chemicals.
- Degradation from exposure to excessive environmental conditions such as heat, cold, sunlight, moisture, etc.Improper labeling.
- Improper sampling and testing, andUse of materials that fail to meet acceptance specifications.
6. Manufacturing Process:
There are various opportunities for contamination of raw materials, intermediates, or packaging materials throughout the manufacturing process.
The main reasons for contamination during the manufacturing process include:
- Lack of dedicated facilities to manufacture a single product inappropriate cleaning in-between batches to minimize the amount of product changeoversUse of an open manufacturing system exposing the product to the immediate room environment. Inappropriate zoning.
- Absence of an area line clearance according to approved procedures following each cleaning process and between each batch
- Lack of cleaning status labeling on all equipment and materials used within the manufacturing facility.
7. Water:
- Water is one of the main constituents of many preparations and is also needed for washing and many processes.
- Fresh water contains many microbes, while water contaminated with sewage waste contains microbes of Proteus spp., Escherichia spp., Clostridium spp., Streptococcus species, Flavobacterium spp. etc.
- Softened water is commonly used for washing purposes in industry, microbes from Bacillus and Staphylococcus spp.
- May come in during the process. Water for industrial use may be treated with filtration or UV or stored at elevated temp.
- To discourage microbial growth in it.
8. Packaging:
- The packaging of pharmaceuticals plays a dual role; it holds the product and also prevents it from spoilage.
- The materials used for pharmaceutical labeling and packaging contain many species of microbes like Bacillus, Clostridium, Penicillium, etc.
- The containers and closures need to be sterilized before use
Question 60. Write briefly on the general requirements of the cell culture laboratory.
Answer:
Minimal Requirements for Cell Culture:
- Clean and quite sterile area
- Preparation facilities
- Animal House
- Microbiology laboratory
Storage facilities (for glassware, chemicals, liquids, and small equipment):
- Laminar-flow, sterilizer, incubator, refrigerator and freezer (-20°C), balance, CO2 cylinder, centrifuge, inverted microscope, water purifier, hemocytometer, liquid nitrogen freezer, slow cooling device (for freezing cells), pipette washer, a deep washing sink.
- Besides the basic and minimal requirements listed above, many more facilities may be beneficial for tissue cultures.
- These include air-conditioned rooms, containment room for biohazard work, phase-contrast microscope, fluorescence microscope, confocal microscope, osmometer, and high capacity centrifuge and time-lapse video equipment.
Culture Vessels:
- In the tissue culture technology, the cells attach to the surface of a vessel which serves as the substrate, and grow.
- Hence there is a lot of importance attached to the nature ofthe materials used and the quality of the culture vessels.
- The term anchorage-dependent cells is used when the cells require an attachment for their growth.
- On the other hand, some cells transform, and become anchorage-independent.
Question 61. Materials used for culture vessels:/’
Answer:
Glass:
Although glass was the original substrate used for culturing, its use is almost discontinued now. This is mainly because ofthe availability of more suitable and alternate substrates.
Disposable plastics:
Synthetic plastic materials with good consistency and optical properties are now in use to provide uniform and reproducible cultures. The most commonly used plastics are polystyrene, polyvinyl chloride (PVC), poly-carbonate, mines, and thermionic (TPX).
Types of culture vessels:
The following are the common types of culture vessels.
- Multiwall plates
- Petri dishes
- Flasks
- Stirrer bottles.
Question 62. Write the contributions of Robert Koch.
Answer:
He investigated the anthrax disease cycle in 1876 and studied the bacteria that caused tuberculosis in 1882 and cholera in 1883.
- He discovered bacteria such as anthrax bacilli, tubercle bacilli, and cholera bacilli. Koch observed the phenomenon of acquired immunity.
- He introduced solid media for the culture of bacteria. Koch pioneered the use of agar as a base for culture media. He developed the pour plate method and was the first to use solid culture media for the culture of bacteria.
- Koch also developed media suitable for growing bacteria isolated from the body.
Because of their similarity to body fluids, meat extracts, and protein digests were used as nutrient sources.
- The result was the development of nutrient broth and nutrient agar media that are still in wide use today.
- He also introduced methods for the isolation of bacteria in pure culture.
- He described the hanging drop method for testing motility. He introduced staining techniques by using aniline dye. He invented the hot air oven and steam sterilizer and also introduced methods to find out the efficacy of antiseptics.
Koch’s Phenomenon:
- Robert Koch observed that guinea pigs already infected with tubercle bacilli developed a hypersensitivity reaction when injected with tubercle bacilli or its protein.
- Since then, this observation has been called as Koch’s phenomenon.
- The medical applications of biotechnology still heavily depend on Koch’s principles of affirming the causes of infectious diseases.
- Write the applications of UV radiations. Detection of impurities.Structural elucidation of organic compounds.Quantitative determination of compounds.
- Detect the presence or absence of functional groups in the compound.
- Study the kinetic reactionsMolecular weight ofthe compounds can be measured.
Question 63. What is mycelium?
Answer:
Mycelium is the vegetative component of a fungus, which consists of white or cream-colored fungal threads or filaments recognized as hyphae. Mycelia is of great importance to the ecosystem as it helps to break down organic material.
They can eradicate pollutants such as petroleum products and pesticides, acting as biological filters. Because mycelia are made up of stiff cell walls, they can pass comparatively readily through the soil or other substrates. They can create fruiting bodies such as mushrooms when two consistent mycelia unite.
Question 64. How will you validate HEPA filters?
Answer:
During the test, a check is also made to ensure that all of the air within the clean room is passing successfully through the filter, ensuring there are no dead spots where contamination can build up.
Question 65. List out the different types of contamination
Answer:
Types:
- Physical contaminants
- Pyrogenic substances
- Chemical contaminants
- Biological contaminants
Question 66. What is the direct inoculation method of sterility testing?
Answer:
Direct Inoculation Method:
When the preparation to be tested has an antimicrobial effect, these effects must be reduced or neutralized by adding an appropriate substance to the specified test media, to diluents or solvents, or the preparation before testing.
Question 67. What is a secondary cell culture?
Answer:
- When a primary culture is sub-cultured, it is known as a secondary culture cell line or sub-clone.
- The process involves removing the growth media and disassociating the adhered cells (usually enzymatically).
- Sub-culturing of primary cells to different divisions leads to the generation of cell lines.
- During the passage, cells with the highest growth capacity predominate, resulting in a degree of genotypic and phenotypic uniformity in the population
- However, as they are sub-cultured serially, they become different from the original cell.
Leave a Reply