Thyroid And Antithyroid Drugs
Thyroid Drugs Introduction
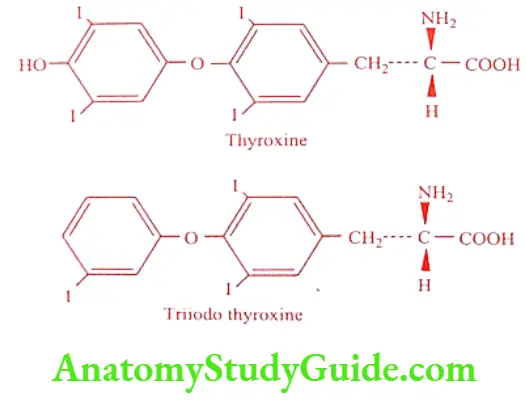
The thyroid gland is a large endocrine gland located deep in the neck, close to the upper portion of trachea. The gland is a source of two fundamental different types of hormones namely thyroxine (T4) and triiodothyroxine (T3), which are iodine containing derivatives of L-thyroxine. Both are vital for normal growth development and control of essential functions such as energy metabolism and protein synthesis. Thyroid gland is the only organ in mammals that has a capability to incorporate iodine into organic substances.
Table of Contents
Thyroxine was first isolated in crystalline form from a hydrolysis of thyroid gland by Kendall in 1915. Later its structure was established by Harington in 1927 and a year later Harington and Barger synthesized the hormone.
Bio-synthesis of Thyroid Hormone
Thyroid hormone 3, 3′- a-Diiodo – L – thyronine (T2) and 3, 3′, 5′-Triiodo-L- thyronine (T3) are formed in a giant prohormone molecule thyroglobulin, an iodinated glycoprotein. The formation of thyroid hormone depends on an exogenous supply of iodine. The iodine atom plays a unique role in the conformational preferences for T3 and T4 because of their large steric bulkiness. The formation of thyroid hormones involves the following complex sequences:
- Active uptake of iodide by follicular cells.
- Oxidation of iodide and formation of iodotyrosine.
- Coupling of iodotyrosine residue.
- Proteolysis of thyroglobulin and release of iodothyronines. (T4 and T3)
Active uptake of Iodide by follicular cells
The first step in the synthesis of thyroid hormone is the uptake of iodide from the blood by the thyroid gland. This is called iodine trapping. Iodide uptake is blocked by certain inorganic ions such as thiocyanate and perchlorate.
Oxidation of iodide and formation of Iodotyrosine
The iodide undergoes oxidation by a membrane bound enzyme thyroid peroxidase. The reaction results in iodination of tyrosyl residues of the thyroglobulin, present in the lumen of the thyroid follicle. The iodination furnishes L-3, 5- Diiodotyrosine (DIT) and about half the percentage of L-3-Iodotyrosine residues (MIT).
Coupling of Iodotyrosine residues
Coupling this two inactive iodotyrosine yield a hormonally active L-thyroxine and L-triiodothyronine. This reaction is also catalyzed by the same peroxidase enzyme. The hormone remains bound to thyroglobulin through peptide link and remain stored in the follicular colloid.
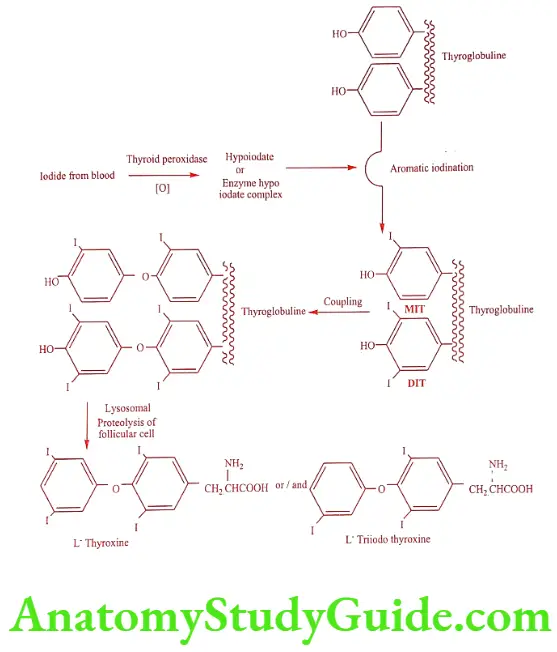
Proteolysis of Thyroglobulin and release of Iodothyronines
The secretion of thyroid hormone is mediated by the release of proteolytic enzyme from lysosome, which completely breaks down the thyroglobulin into its constituent amino acids releasing L-thyroxine and L-triiodo thyroxine into the circulation. The proteolysis is stimulated by TSH.
The iodinated tyrosine as metabolized and the iodine liberated in the form of iodide is recycled the thyroid hormones are transported in the blood in strong but non-covalent association with three major binding proteins; thyroid binding globulin, trans thyretin (formerly called T4 binding pre-albumin) and albumin. Protein binding of thyroid hormones protects them from metabolism and excretion.
Although L-triiodothyronine is secreted by the thyroid gland, the majority of it is produced by the mono-deiodination of L-thyroxine in the peripheral tissue by the enzyme iodothyronine deiodinase.
The hormonal production by the thyroid gland is regulated by the thyroid stimulating hormone (TSH). It is recognized that the rate of secretion of TSH is controlled by thyrotropin- releasing hormone secreted from the hypothalamus and by the quantity of thyroid hormone in the circulation.
Thyroid hormone regulates growth and development and also has calorigenic effect, affect metabolic processes, have cardio vascular effects causing more forceful and more rapid heartbeats and increasing cardiac output. Deficiency of thyroid hormone results in hypothyroidism. This causes goiter, cretinism and myxedema. An enlarged palpable thyroid gland is referred as goiter.
Cretinism, a syndrome of dwarfism, mental retardation and coarseness of the skin and facial features due to lack of thyroid hormone from birth (congenital hypothyroidism). Myxedema, a dry firm waxy swelling of the skin and subcutaneous tissues found in patient with under active thyroid glands (hypothyroidism).
Excessive secretion of thyroid hormone results in hyperthyroidism. This causes Grave’s disease characterized by thyrotoxicosis and ophthalmopathy.
Hashimoto’s disease is an auto immune condition in which antibodies are produced against normal thyroid tissues. It features include a firm swelling of the thyroid gland and partial or total failure of secretion of thyroid hormone.
Conformational Properties of Thyroid Hormones
The stereo chemistry of the thyroid hormones plays an important role in defining the activity of the hormone.
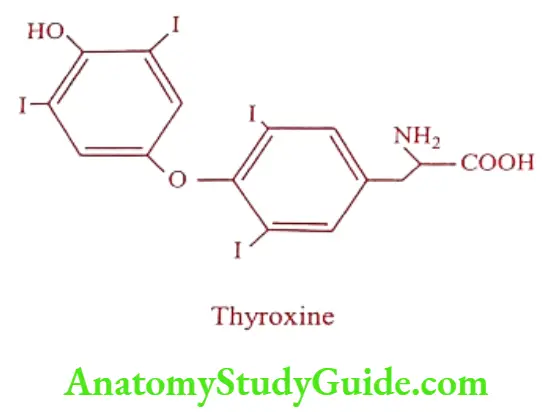
The diphenyl ether conformation is important for the activity. Because of the steric effect of the large iodine atoms in position 3 and 5 the planes of the two aromatic ring are perpendicular to each other which holds the ring at an angle of about 120°C.
The outermost phenolic ring can adopt conformation relative to the alanine side chain, which would be cisoid or transoid conformation. Both the conformation appears to be similar in energy. But the most favoured conformation has the carboxyl group at the maximum distance from the aromatic ring bearing the side chain.
The phenolic hydroxyl group is coplanar with its aromatic ring and the 3′-iodine atom is at distal position. The configuration at the a-carbon of the side chain is L or S in terms of Cahn- Inglod-Prelog convention. The D-isomer lowers the concentration of cholesterol in plasma, but due to its cardiac side effect it is not used much.
Thyroid hormone and dried thyroid glands are used in the treatment of in the are employed in treatment of hypothyroidism. The antithyroid drugs hyperthyroidism.
Synthetic – Thyroid Hormones
Synthetic, crystalline thyroid hormones are more uniformly absorbed than biological preparations and contain more precisely measured amount of active ingredients in their dosage forms.
Levothyroxine sodium (Levothroid, Unithroid, Thyronorm)
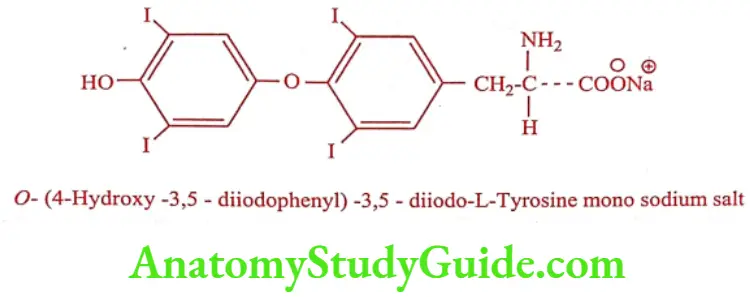
Synthesis
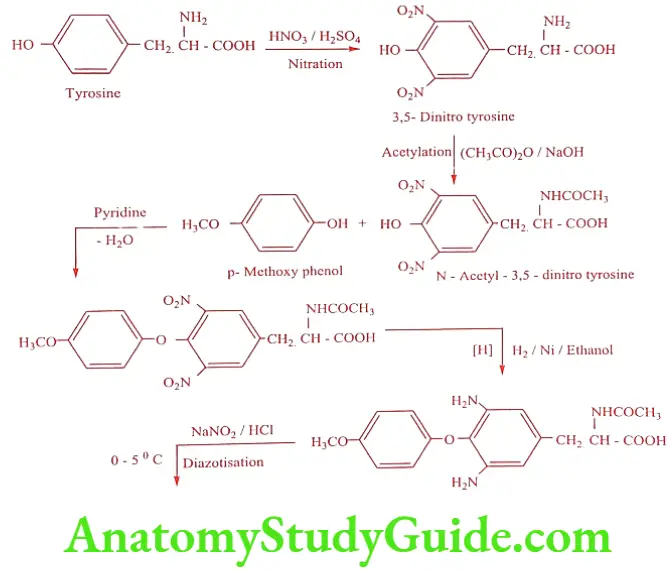
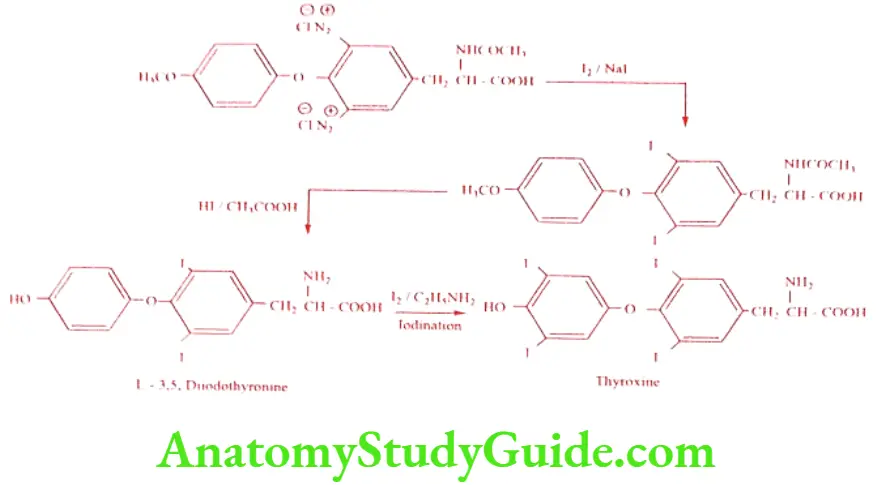
ADR: Nervousness, excitability, tremor and muscle weakness.
Dose: Initially 50 to 100μg daily may increase by 25 to 50μg at 4 weekly intervals until the thyroid deficiency is corrected.
Use: The dextro rotatory isomer of Thyroxine is used as hypocholesterolemic agent. It is recommended for the patients who do not have coronary artery diseases.
Liothyronine (T3) (Cytomel, Tertroxin, Triostat)
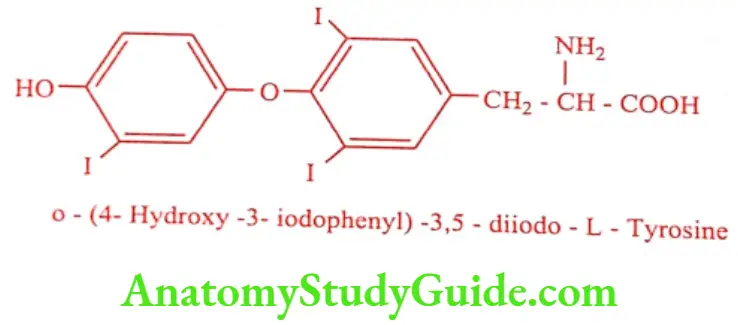
Liothyronine has a rapid onset of action. It is used in condition where rapid and short duration of action is required.
ADR: Insomnia, changes in menstrual cycle and increased appetite.
Dose: 60 to 75μg in 2 to 3 divided doses.
Use: It is used to treat conditions of thyroid deficiency and to treat goiter.
Liotrix (Thyrolar)
A mixture of sodium salt of Thyroxine and Liothyronine in the ratio of 4:1
ADR: Alopecia, asthenia and pruritus.
Dose: 100 to 200mg daily, should be taken in an empty stomach.
Use: It is used in replacement therapy in patient with hyperthyroidism.
Antithyroid Drugs Introduction
Antithyroid drugs can either directly or indirectly interfere with the synthesis, release or action of thyroid hormones and used to control hyperthyroidism. A number of linear and heterocyclic derivatives of thiourea inhibit the production of thyroid hormone by the thyroid gland. The mechanism of action is that of preventing iodination of tyrosine or coupling between iodotyrosine. They also inhibit the conversion of thyroxine to liothyronine in the periphery.
Antithyroid Drugs Classification
- Thioureylenes
- Thiouracil derivatives : eg. Methylthiouracil, Propylthiouracil.
- Imidazoles : eg. Methimazole, Carbimazole.
- Ionic inhibitors : eg. Thiocyanates, Perchlorates, Nitrates.
- Radioactive iodine : eg. 1311
- Iodides : eg. Lugol’s solution, Colloid iodine, Saturated solution of potassium iodide.
Thioureylenes
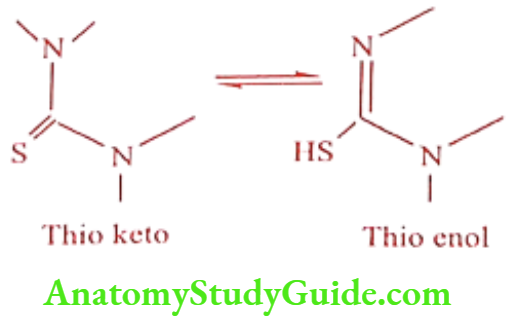
- The Ca thioketo / thioenol and unsubstituted N is essential for activity
- They act by inhibiting thyroid peroxidase enzyme
Thiouracil Derivatives
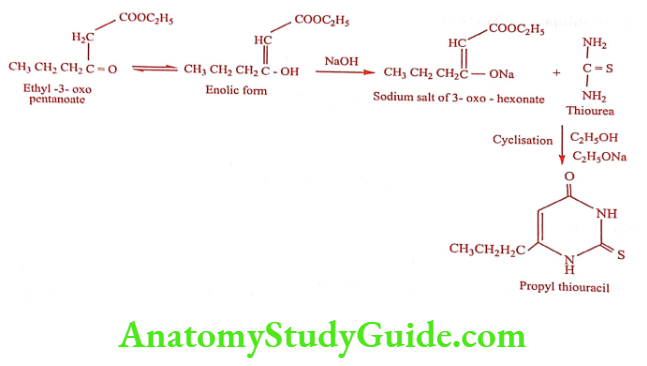
Propylthiouracil (Prothiugil)
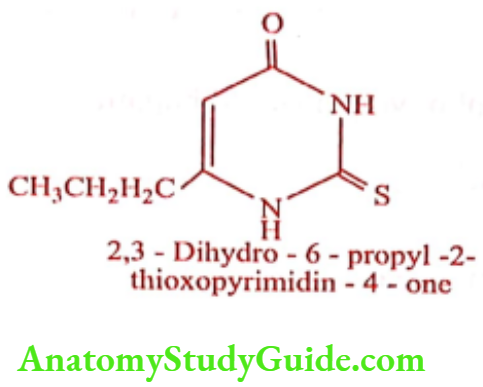
Synthesis
ADR: Agranulocytosis, thrombocytopenia and hepatic necrosis. Since is causes severe liver damage, it is recommended for people who cannot receive other treatment surgery, radioactive iodine or different medicaments.
Dose: 50 to 150mg thrice a day followed by 25 to 50mg for maintenance. It is not recommended for children.
Use: It is used to control hyperthyroidism in patients with Grave’s disease, in preparation of thyroidectomy and as an adjunct to radioiodine therapy.
Methylthiouracil (Thioryl, Tiorala)
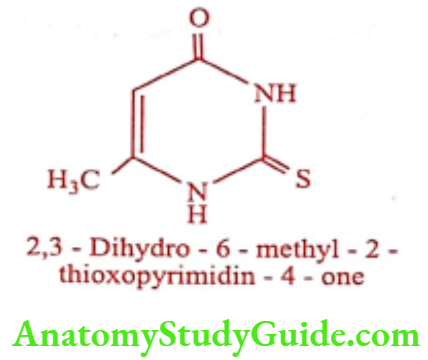
ADR: Agranulocytosis, thrombocytopenia and hepatitis.
Dose: 100mg three times daily.
Use: To treat hyperthyroidism diseases.
Imidazole Derivatives
Methimazole (Nortnyx)
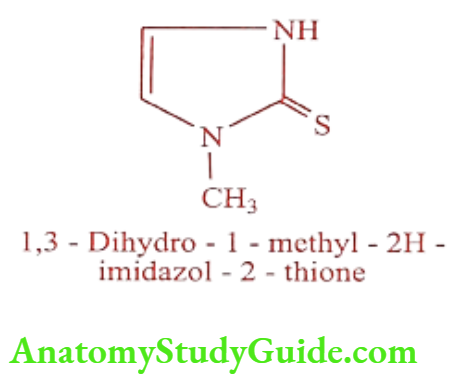
Synthesis
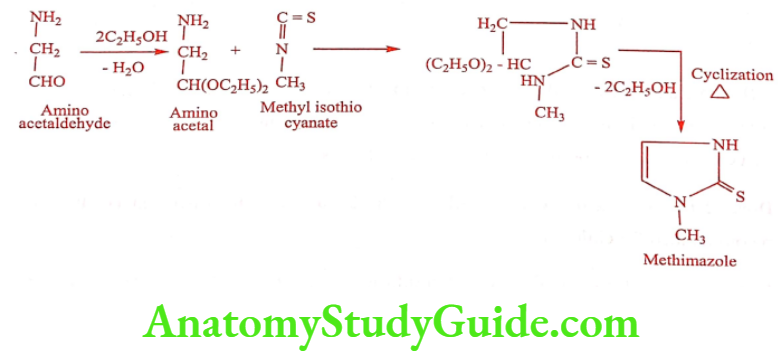
ADR: Long term use may lead to remission of hyperthyroidism. It may cause birth defect if it is used during pregnancy.
Dose: Three doses for every 8hrs. 15mg/day-for mild hyperthyroidism; 30mg/day-for moderate hyperthyroidism; 60mg/day-for severe hyperthyroidism.
Use: It is more potent than Propyl thiouracil and indicated in the treatment of hyperthyroidism.
Carbimazole (Thyrocab, Thyrozole)
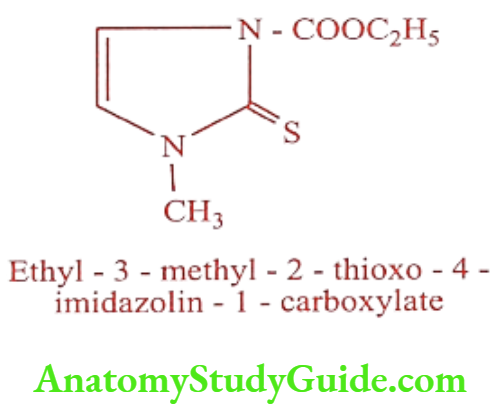
Synthesis
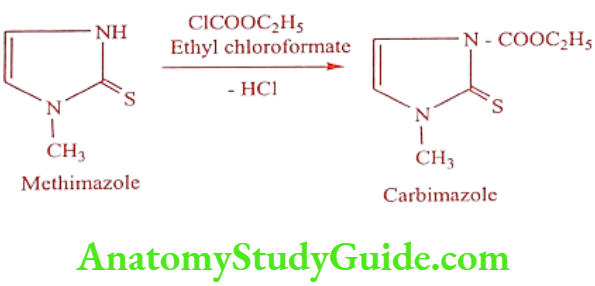
ADR: Mild leucopenia, sore throat, mouth ulcers and fever.
Dose: 20 to 60mg daily according to severity of disorders.
Use: To treat hyperthyroidism and thyrotoxicosis.
Ionic Inhibitors
Ionic inhibitors are the substances which block the iodine transport mechanism. The effective species are themselves anions resembling in some ways to iodide ions. Thiocyanate ion (SCN), a product of enzymatic hydrolysis of certain plant glycosides and perchlorate ion (CIO) are the prominent examples of ionic inhibitors. Perchlorate is ten times as active as thiocyanate. Although potassium or sodium perchlorate can be used to control hyperthyroidism, they may cause fatal aplastic anemia. Their use in therapeutics is abandoned except in very unusual circumstances.
Radioactive Iodine
Among the several isotopes of iodine, the isotope 131I is of greatest use. It has a half-life of 8 days and emits both X-ray and ẞ-particles. 131I is rapidly and efficiently taken up by the thyroid, incorporated into the amino acids and gets deposited in the colloid of the follicle. The ẞ-rays originating within the follicle destroy selectively cells of the thyroid with little or no damage to the surrounding tissue. Thus radioactive iodine in the form of sodium iodide (131) finds its use in the treatment of hyperthyroidism and in diagnosis of disorders of thyroid function.
Iodides
Iodides are used when it is imperative to control hyperthyroidism very rapidly as in a patient with thyrotoxic crisis or with congestive heart failure. For this reason, they should be given along with a thiourea drug. They are used in the preoperative preparation of a thyroid patient to reduce the vascularity of the gland. This is the most important use of iodides at present. It is given for 10 to 14 days pre-operatively in addition to a thiourea drug, but must be carefully avoided in patients on potassium perchlorate. Thyroxine given concurrently with antithyroid drugs can obviate the need for the pre-operative addition of iodide to therapy.
Leave a Reply