Resin Restorations On Primary Teeth Introduction
Unfiled resins have been used in restorative dentistry since the early 1950s.
Table of Contents
Unfiled resins have very high water sorption and coefficient of thermal expansion, due to which they have very poor diametrical stability, inferior mechanical properties and below-par aesthetics.
Because of these disadvantages, composite resins have slowly replaced unfilled resins. Composite means ‘two brought into one’.
Composite resins are called filed resins. Two distinct materials coupled by a compatible medium lead to a significant improvement in desirable properties.
Read And Learn More: Paediatric Dentistry Notes
Resin Restorations On Primary Teeth Properties
Composite resins are used in paediatric dentistry due to their optimal physical properties and superior aesthetics.
The properties of composite resins are as follows:
- Resins are stronger materials than glass ionomers due to the hardness of the set resin, wear resistance, rigidity, fracture toughness and strength.
- The thermal properties of composites are not quite identical to those of the tooth structure.
- The coefficient of thermal expansion and thermal diffusivity is three to four times higher than that of the tooth structure. However, there is no demonstrable adverse clinical consequence of these thermal properties.
- Composite resins are not acceptably biocompatible. Monomer resin molecules can traverse through the dentinal tubules to initiate an adverse pulpal response.
- But polymerised resin does not cause any pulpal response. Glass ionomer lining on the dentin surface is mandatory under resin restoration. HEMA (hydroxyethyl methacrylate) is also a demonstrable allergen.
- Composite resins bond micromechanically and not chemically to the tooth structure. So, microleakage makes the tooth prone to secondary decay.
- Composite resins undergo degradation in the oral environment. The increased rate of wear and degradation is due to the following reasons:
- Unreacted methacrylate groups degrade more rapidly and are leached out of the resin.
- Hydrolytic degradation of barium and strontium glass filers may lead to the build-up of pressure resulting in crack formation.
- Composite resins allow water from the oral environment to be imbibed into the restoration. This property is termed water sorption.
- Water sorption takes place in the first 7–10 days before the setting reaction is complete. When water sorption is clinically high, it leads to an intrinsic colour change.
- Composites may also undergo extrinsic staining due to surface roughness. Colour stability is poor when the resins are not cured completely.
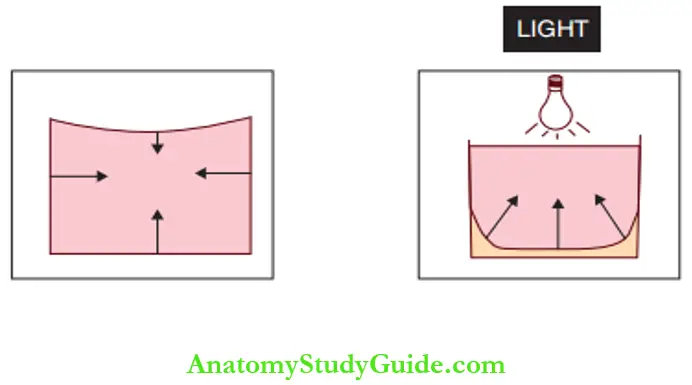
- Compositeresinsundergopolymerisationcontraction or shrinkage on the setting, leading to an approximate volumetric contraction of 1.0–2.5% and a strain at the tooth–resin interface.
- This contraction is towards the centre for self-cured resins and towards the light source for light-cured resins. The contraction can lead to poor marginal integrity.
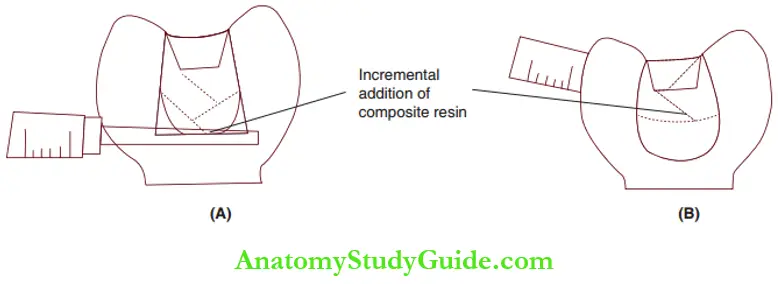
- It weakens the cusps, leading to potential cusp deformation and fracture. It can be prevented by an incremental resin build-up of the restoration. Each increment can be of 2 mm thickness.
- Unlike glass ionomers, composite resins are non-fluoride-releasing (except for compomers). Hence, there is no resistance to secondary decay.
Resin Restorations On Primary Teeth Composition
The composite resins are mainly composed of the resin matrix and the inorganic filler particles coupled by organosilane. Each component is described in detail as follows:
1. Resin matrix:
High-viscosity aromatic monomers such as bis-GMA (bisphenol-A dimethacry-acrylate) in combination with urethane dimethacrylate are the predominant constituents.
Low-viscosity aromatic monomers like TEGDMA (tri ethylene glycol dimethacrylate), EGDMA (ethylene glycol dimethacrylate) and HEMA are added to improve the handling properties of the composite resin.
2. Inorganic filer particles:
Silica, quartz, barium and strontium glasses and other similar inorganic materials are added to improve the physical properties of the resin.
The amount of filer concentration is expressed as ‘filer loading’. It is the percentage of filer present in a cured resin.
It ranges from 52% to 88% by weight. The filer loading can also be expressed by volume percentage which is 11–16% lesser for commercial composite resins.
3. Coupling agent:
The coupling agent is organosilane. The organic end couples with the resin matrix and the silane end couples with the filler particles.
It is coated on the filer particles and bonds the filer with the resin particles. The organosilane coupling agent usually used is methacryloxypropyl trimethoxysilane.
The quality of the coupling agent determines the longevity of the restoration. The loss of the coupling agent releases resin and filer particles and leads to surface breakdown.
4. Photoinitiator:
Composite resins are polymerised by either chemical activation or light-activated polymerisation. Chemically activated resins are called auto-cured resins and are used in bonding orthodontic brackets.
The light-activated resins are used in restorative dentistry. They contain photoinitiators like camphor quinone.
These initiators are activated by visible light of low wavelength (480–520 nm) and high frequency.
The photoinitiators release free radicals, which collide with the carbon-carbon double bond, releasing more free radicals while pairing up with one of the carbon atoms.
5. Inhibitors (stabilisers):
Inhibitors such as butylated hydroxytoluene prevent the auto-polymerisation of the resin by absorbing any available free radical in the system.
Unreacted methacrylate groups can amount to 25–56%. On light activation, 60% of the resins polymerise almost immediately; 10% will polymerise in the next 2 days.
Residual resins are unpolymerised and remain as monomers. Inhibitors are also called stabilisers since they maintain the monomer form of the resin.
6. Pigments:
Certain pigments are added so that the aforementioned components bond homogeneously with each other.
Composite Resin Restorations In Primary Teeth
Early childhood caries manifests itself as proximal caries prior to the involvement of labial or lingual surfaces.
These lesions on the primary anterior teeth are best restored with composite resins.
The following points have to be observed during the cavity preparation of composite resin restoration:
- All the carious enamel and infected dentin have to be removed.
- Affected dentin may be left
- Enamel margins need to be bevelled to increase the surface area for micromechanical bonding.
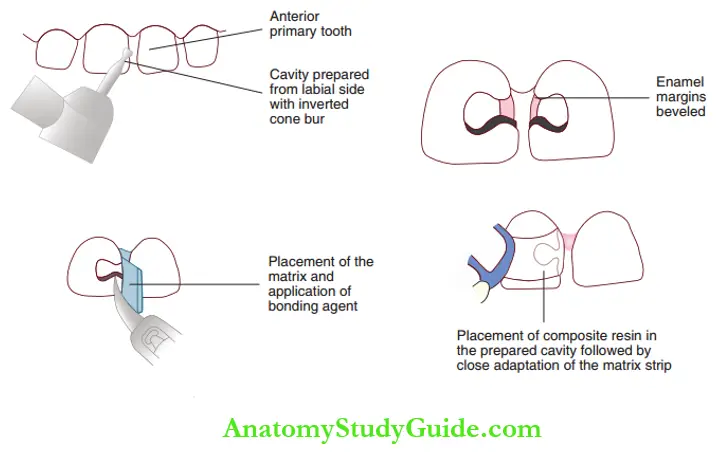
The cavity preparation for a proximal carious lesion on a primary anterior tooth begins from the labial side with an inverted cone bur.
The cavity preparation can be limited to the site of the carious lesion. The enamel margins are bevelled.
Incisal angle is included only if it is involved. Dentin is lined by Class 3 glass ionomer cement.
The cavity surface and glass ionomer surfaces are etched with 37% phosphoric acid gel for 90–120 seconds.
Etching time is longer owing to the higher content of the organic matrix in the enamel or dentin.
Etched surfaces are bonded with dentin bonding agents. The resin is placed in the prepared cavity with the Mylar matrix strip held in place. The resin is cured for 20–40 seconds.
Composite resin restorations for proximal caries in primary molars require a Mylar matrix and a light tan sitting plastic wedge. The resin is incrementally added and cured as depicted.
Compomers
Composite resins with added polyacid molecules are called compomers. They contain methacrylate resin molecules, inorganic filers and carboxylic acid molecules with other minor constituents.
Along with the conventional polymerisation of methacrylate groups, the carboxylic acid reacts with the inorganic filer, glass powders, in the presence of water by an acid-base reaction.
Hydrogel formation (discussed in Chapter 34) occurs within the resin structure. It results in a slow, low-level release of fluorides into the oral cavity.
Hence, compomers are fluoride-releasing composite resins. Fluoride release has been demonstrated throughout the lifetime of the restoration, but it is markedly lower than that in glass ionomers.
However, adhesion is micromechanical and not chemical. It requires acid etching and dentin bonding agent application as in the case of composite resins.
Properties such as compressive strength, hardness, fracture toughness and abrasion resistance are quite comparable to the composites.
They are relatively more biocompatible than composites. The optical properties and aesthetics of compomers are superior to those of glass ionomers.
Summary
1. Composition of composite resins:

2. Mechanical properties of composite resins that are of clinical significance:
- They are stronger materials than glass ionomer cement.
- The thermal properties of composites are not quite identical to the tooth structure.
- They are not acceptably biocompatible. Monomeric resin molecules can traverse through the dentinal tubules to initiate an adverse pulpal response.
- They bond micromechanically and not chemically.
- They undergo degradation in the oral environment.
- They may undergo extrinsic surface staining and intrinsic colour change in the oral environment.
- They undergo polymerisation contraction on set, leading to a volumetric contraction, which is towards the centre for self-cured resins and towards the light source for light-cured resins.
3. Clinical advantages/disadvantages

4. Composite resin restoration in primary teeth

5. Polyacid-modified composite resins (compomers) are composite resins with added polyacid molecules.
- Along with the conventional polymerisation of methacrylate groups, carboxylic acid reacts with glass powders.
- During setting, hydrogel formation gives a slow, low-level release of fluorides into the oral cavity. Hence, compomers are fluoride-releasing composite resins.
- Adhesion is micromechanical, not chemical. It requires acid etching and dentin bonding agent application as with composite resins.
- Their optical properties and aesthetics are superior to glass ionomers.
- Mechanical properties are quite comparable to composites.
- They are relatively more biocompatible than composites.
Leave a Reply